Ophthalmologic Emergencies in the Geriatric Patient
Ophthalmologic Emergencies in the Geriatric Patient
Author: MaryAnn Smith, MD, Emergency Medicine Physician, W.W. Backus Hospital, Norwich, CT.
Peer Reviewer: Eric R. Eggenberger, DO, Associate Professor, Michigan State University, Department of Neurology and Ophthalmology, East Lansing, MI.
In 1993, there were approximately 2.32 million visits to emergency departments (EDs) for ocular problems, and 51% of those cases were non-traumatic in nature.1 Ophthalmologic complaints, especially changes in vision or eye pain, are problems commonly evaluated in the ED.2 The elderly patient is at risk for particular ocular disorders, some of which rarely are encountered in the younger patient. Awareness of those ophthalmologic conditions that likely will be seen in the aged adult will help guide the history and physical examination and focus the differential diagnosis. Sudden visual impairment, painful or not, is alarming to any patient and almost invariably will cause the patient to present to the ED. Certain conditions, such as acute narrow-angle glaucoma, require rapid recognition and immediate, sight-preserving management or vision may be irreversibly lost. While loss of vision is a catastrophic event for any patient, elderly persons with limited hearing, balance, or motor skills are especially vulnerable for a poor long-term outcome.3-5
Physiologic changes of aging, particularly in the cardiovascular and immune systems, put the older patient at risk for a number of ophthalmologic conditions. The acute manifestations of glaucoma, retinal vessel disease, vitreoretinal hemorrhage, giant cell arteritis, herpes zoster, and optic nerve ischemia are likely to receive ED evaluation. Other diseases, such as diabetic retinopathy and macular degeneration, are important contributors to visual loss in the geriatric population. Although these are slow and insidious processes, they may present after an acute change. Diagnosis and treatment of ocular emergencies may be hampered by the presence of comorbid medical conditions. For example, dementia or central nervous system (CNS) disease may prevent the patient from communicating his or her symptoms clearly, or a patient with preexisting cardiac problems may experience significant side effects from topical ophthalmic preparations. Furthermore, factors such as social isolation and financial limitations also may pose difficulties. The emergency physician must meticulously assess a geriatric patient with a visual complaint to quickly recognize sight-threatening diseases and minimize the threat of blindness with both medical management and specialist involvement.
The emergency physician must be aware that geriatric patients are at high risk for ophthalmologic emergencies. A careful history and physical examination, with specialist involvement as necessary, may prevent the patient becoming blind—a devastating outcome.— The Editor
Acute, Narrow-Angle Glaucoma
Epidemiology. In the year 2000, it was estimated that 66.8 million people worldwide will have some form of glaucoma, with more than 6 million individuals becoming permanently blind from it.6 In the United States alone, approximately 1 million Americans older than age 65 have vision loss secondary to glaucomatous changes.7 While no cure exists, early treatment can control the progress of the disease and vastly reduce subsequent vision loss. Narrow-angle closure glaucoma makes up fewer than 10% of all cases of glaucoma,8 but it is characterized by sudden and painful onset, and is likely to bring the patient to the ED.
Pathophysiology. Glaucoma refers to many different disease processes that ultimately produce irreversible damage to the optic nerve. There are about 60 different types of glaucoma. The most common form, open-angle glaucoma, is a slowly progressive and often asymptomatic process, especially early in its course. Acute angle closure glaucoma, however, is a sudden condition of visual impairment and pain. Both types of glaucoma share the pathologic process of increased intraocular pressure (as a result of aqueous flow obstruction) and subsequent retinal and optic nerve ischemia.
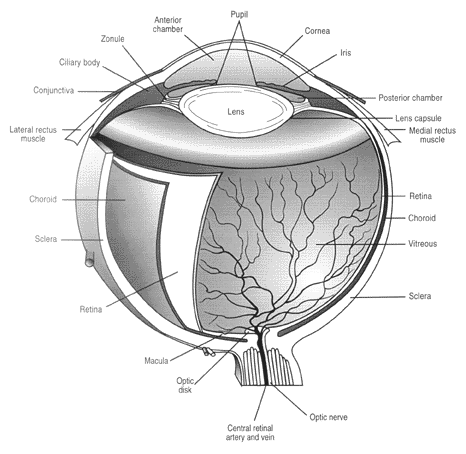
The pathologic mechanisms that produce increased intraocular pressure take place in the anterior and posterior chambers of the eye, both of which are located in the space anterior to the lens and bounded by the cornea. (See Figure 1.)
| Figure 1. Composition of the Orbit |
|
|
| Reprinted from: Carcia SE. Pediatric ocular trauma. Ped Emerg Med Reports 1998;3-87-98. |
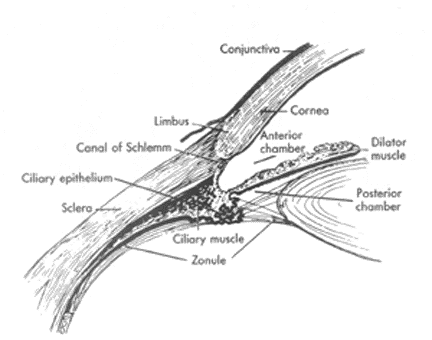
The iris divides these two spaces; the pupillary opening connects them. Aqueous humor is a low viscosity fluid created by the epithelium of the ciliary body, a posterior chamber structure that filters fluid and electrolytes from systemic blood utilizing carbonic anhydrase. Entering the posterior chamber from its peripheral location, aqueous humor flows behind the iris toward the pupil, through the pupillary opening, over the iris, and then outward toward the periphery. At the most lateral aspect of the iris, where the iris meets the cornea, aqueous humor leaves the anterior chamber through the trabecular network. Final disposition of the aqueous is accomplished in the trabecular network, with reabsorption into the systemic circulation via the episcleral vessels. (See Figure 2.)
| Figure 2. Anterior Eye Structures |
|
|
| Details are shown of the area of the limbus (junction of the cornea and sclera), ciliary body, and lens. Reprinted with permission from: Davson H. The Eye. 2nd ed. Vol. 1. New York: Academic Press; 1969. |
Turnover of aqueous humor is brisk, with complete volume replacement every 60 minutes. Any obstruction of the flow of aqueous humor, at any point along the pathway, will result in a precipitous rise in anterior and/or posterior chamber pressure. This increase in pressure is rapidly transmitted to the rest of the eye, including the retina and optic nerve. Ischemia can quickly set in, and if not reversed promptly, may cause irreversible cell damage.
In narrow-angle glaucoma, the obstruction is located at the iris-cornea angle. Normally, the iris is not in contact with the cornea; in fact, the angle between them is more than 40 degrees. Any force that pushes the iris forward closes the angle, blocking off the trabecular network. There are two predominant causes of narrow-angle glaucoma: pupillary block and plateau iris.
In a normal eye, the iris is positioned anterior to, but not touching, the lens. In the normal course of aging, the lens enlarges, and assumes a slightly anterior position. This creates the possibility of iris-lens apposition. If the iris comes in contact with the lens, the pupillary opening is sealed off, creating pupillary block. Aqueous humor still is produced, but is trapped within the posterior chamber. As pressure accumulates, the iris bows forward. The forward curve closes the iris-cornea angle. Residual aqueous humor in the anterior chamber, which is now compressed by the iris, cannot drain out and pressure continues to rise. Plateau iris is a condition in which the iris has an unusual curved shape at the periphery, placing it closer to the cornea and creating a congenitally narrow-angle. Even slight changes in the iris angle can block aqueous flow. In either condition, increased anterior/posterior chamber pressure is communicated throughout the eye, and retinal circulation is impeded.
Clinical Presentation. Patients at risk are 50-69 years of age,9 and of Caucasian, Inuit, Chinese, or Asian descent. Women are affected 3-4 times more often then men, and far-sighted persons also are at higher risk. Typical symptoms are sudden onset of unilateral eye pain, perception of colored halos around lights, and headache. The headache often is severe, may be accompanied by nausea or vomiting, and can mimic a migraine. The attack may be unprovoked, or may be precipitated by anything that causes pupil dilation such as entering a dark environment or mydriatics. Anticholinergics and beta-agonists (including inhaled agents) have produced sudden attacks of narrow-angle glaucoma.10 The patient may recall similar, transient attacks in the past.
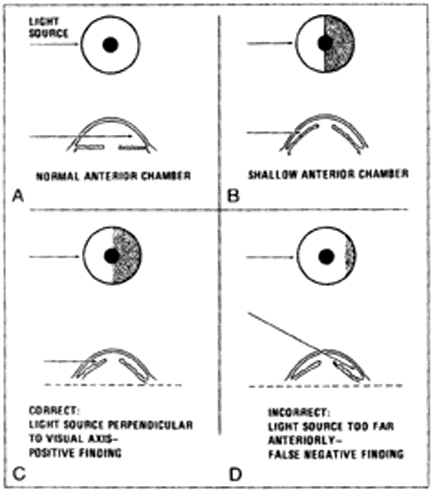
Physical examination reveals an eye with a diffuse conjunctivitis and ciliary flush. The cornea may be clouded or hazy due to edema, and small blisters may appear on the surface. Due to iris ischemia, the pupil is non-reactive. The pupil usually is mid-dilated, which is the position of maximal iris-lens contact. Small, gray-white anterior lens opacities (glaukomflecken) may be seen on the cornea; these are evidence of previous episodes of increased pressure. Palpation of the eye, which should be done gently, will find the globe to be rock hard. Exam of the opposite eye may suggest a congenitally shallow anterior chamber. A slit lamp exam or oblique flashlight test will demonstrate a shallow anterior chamber and iris bowing. (See Figure 3.)
| Figure 3. Oblique Flashlight Test |
|
|
| Reprinted with permission from: Bresler MJ, Hoffman RS. Prevention of iatrogenic acute narrow-angle glaucoma. Ann Emerg Med 1981;10:535-537. |
Applied tonometry will demonstrate increased intraocular pressure. While applanation tonometry remains the gold standard, most emergency physicians should have availability of and familiarity with either the Tonopen or Schiotz tonometer. Any measurement of more than 22 mmHg is abnormal, but in acute narrow-angle glaucoma, pressures may rise to more than 70 mmHg.
Treatment. Initial control of increased intraocular pressure is attempted using topical and oral/parenteral medications. These agents temporarily decrease intraocular pressure in different ways: decreasing aqueous humor production, volume, or facilitation of reabsorption. There are several classes of medication available. Topical beta-blockers are the first line of therapy. Beta-blockers constrict the blood vessels supplying the ciliary body, thereby decreasing blood supply and aqueous production. Topical alpha-agonists enhance trabecular network outflow by dilating the episcleral vessels. Carbonic anhydrase inhibitors decrease aqueous production by inhibiting synthesis within the ciliary epithelium. A relatively new option, topical prostaglandin agonist (latanoprost), augments aqueous elimination by increasing uveoscleral outflow.11 Miotics, or cholinergic compounds, constrict the pupil by stimulating the iris but may not be effective if the iris is ischemic, and should not be used until the intraocular pressure is lower than 40 mmHg. Miotics also are not indicated if the patient has had cataract surgery or a lens implant. Osmotic diuretics decrease the overall volume of the eye by pulling water out of the vitreous and aqueous humor. Another simple, non-invasive option is to have the patient lie completely supine, which may allow gravity to pull the lens away from the iris. Definitive treatment of narrow-angle glaucoma, however, is surgical. Laser iridectomy creates a small hole in the peripheral iris, and provides a permanent, alternative pathway for aqueous humor flow.
Although effective, all of the anti-glaucoma agents have systemic side effects that can be dangerous to the elderly patient.12-15 Table 1 lists some of the more common side effects associated with each class of medication.
| Table 1. Glaucoma Medications | ||||
| Drug | Route | Examples | Mechanism | Side Effects |
| Beta-blocker |
T |
Timolol |
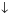 aqueous production aqueous production |
Myocardial depression Bronchospasm Bradyarrhythmia Depression |
| Alpha-adrenergic agonist |
T |
Apraclonidine |
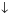 aqueous production aqueous production |
Hypertension Tachyarrhythmia Tremor Allergic conjunctivitis Headache |
 trabecular outflow trabecular outflow |
||||
| Carbonic anhydrase inhibitor |
T |
Dorzolamine Brinzolamide |
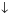 aqueous production aqueous production |
Corneal irritation Keratitis Headache Vertigo Blood dyscrasia Renal calculi |
|
O |
Acetozolamide | |||
| Miotics | T |
Pilocarpine |
Iris constriction | Headache Ocular pain Hypotension Dyspnea |
 aqueous outflow aqueous outflow |
||||
| Prostaglandin analog | T |
Latanoprost |
 uveoscleral outflow uveoscleral outflow |
Iris pigmentation Corneal irritation Myalgias Arthralgias |
| Osmotic diuretic |
O |
Glycerine |
 vitreous volume vitreous volume |
Hypotension CHF |
|
T = topical O = oral ¶ IV = intravenous |
||||
| Adapted from: Quillen DA. Common causes of vision loss in elderly patients. Am Fam Physician 1999;60:99-108; Singh K, Bautista RD. Advances in glaucoma therapy. Int Ophthalomol Clin 1999:39:1-15. | ||||
Retinal Vascular Occlusions
Epidemiology. Symptomatic retinal vascular occlusions are a relatively uncommon event, and are very uncommon in the younger patient population. The risk factors for both arterial and venous vascular occlusions are those which predispose individuals to thrombotic or embolic events. Arterial and venous occlusions are more common in men and usually affect persons in the fifth to sixth decades of life. Profound visual loss is not uncommon, especially in central retinal artery occlusion, and prognosis for visual recovery is poor.
Pathophysiology. Central retinal artery occlusion (CRAO) can be an embolic or thrombotic event. Emboli are more common, and typically originate from the common or internal carotid arteries, but may also originate from a fibrillating atria or rheumatic heart disease. Occlusion of the central retinal artery rapidly causes ischemia to the entire retina, and loss of vision results. The occlusion may be temporary and the patient will quickly recover his or her vision; such transient episodes are termed amaurosis fugax. In some cases, the emboli may lodge in a more distal or arteriole branch with less severe visual loss. As the retina progressively becomes more ischemic, edema sets in. Irreversible damage occurs in fewer than 60 minutes. Persons with arteriosclerotic vascular disease, hypertension, and hyperlipidemia are at risk for this condition. Giant cell arteritis is a common precipitator of arterial occlusions.
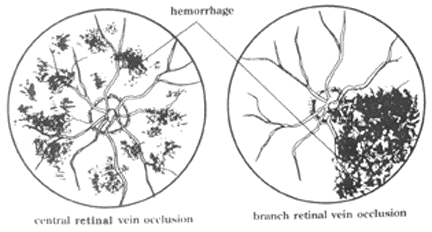
Central retinal vein occlusion (CRVO) has similar risk factors, but the mechanism is thrombotic in nature. In this situation, venous occlusion produces edema, hemorrhages, and nerve fiber infarcts (cotton wool spots). Retinal blood supply is impeded, with subsequent retinal ischemia. Branch retinal venous occlusions may occur at arteriovenous crossings, where the vessel is slightly bent or angled, and only a segment of the retina is ischemic. (See Figure 4.) Additional risk factors for central venous occlusion include diabetes, collagen-vascular disease, and hyperviscous coagulopathic disorders.
| Figure 4. Central and Branch Retinal Vein Occlusion |
| Reprinted with permission from: James F. Collins, MD. The Center for Eye Care. West Islip, New York. |
Clinical Presentation. Retinal vascular occlusion, either arterial or venous, causes sudden, painless monocular vision loss. The blindness may begin as a graying-out, or be described as a shade pulling down. Most often, there is no precipitating event. Physical examination reveals significant loss of visual acuity but an otherwise normal-appearing external eye. When there is an arterial occlusion, fundoscopy will show a pale gray or white retina. The fovea appears cherry-red, as a result of the underlying intact choroid vasculature showing through the underperfused retina. If any arteries can be visualized, there may be segmentalization or "boxcarring," which is indicative of blood flowing very slowly. In the case of venous occlusion, the retina is dark red and congested, with engorged, prominent vessels, cotton wool spots, and flame hemorrhages. The optic disc may appear edematous. In this instance, tonometry will discover normal or even slightly low intraocular pressures. Fluorescein angiography can determine localization of the obstruction, and also has a role in treatment decisions, especially for CRVO.
Treatment. No effective therapy exists to treat retinal vascular occlusions. In the past, ocular massage, carbon monoxide inhalation, anterior chamber paracentesis, and many other interventions have been recommended for arterial occlusions, but all were found to have little or no therapeutic value.19-20 At best, noninvasive maneuvers have produced sporadic benefits.21 Although treatment results often are poor for CRAO, the standard of care remains emergent treatment attempts and an ophthalmologic evaluation; failing to attempt treatment is a potential medico-legal pitfall. For CRVO treatment, there are no definite emergent options. Prompt ophthalmologic follow-up is indicated, however.
Intra-arterial fibrinolytics are a promising new therapy, which have shown some efficacy in treating both arterial22-25 and venous26,27 occlusions. No randomized trials have been completed, and it remains a controversial intervention with many potential complications. Recognizing that retinal vessel occlusions may represent a symptom of systemic vascular disease and providing appropriate follow-up for this problem may be the most valuable intervention for the patient. Ophthalmologic follow-up also is necessary, as the ischemic retina soon will stimulate the growth of new vessels, a process known as neovascular retinopathy. As mentioned in the discussion of vitreous hemorrhage, retinopathy can lead to other ocular problems and should be monitored closely.
Vitreous Hemorrhage
Epidemiology. Vitreous hemorrhage can produce a spectrum of disease from minor, annoying "floaters" (small, grayish opacities that shift with head movement and settle with gravity) to total vision loss. The incidence is 7 in 100,000 for the general population, with elderly persons at higher risk.28 The three most common etiologies for vitreous hemorrhage—posterior vitreous detachment, diabetic retinopathy, and retinal vessel occlusion—most commonly occur in the older adult.
Pathophysiology. Vitreous humor is a transparent, gel-like substance that is composed of water, collagen fibrils, and hyaluronic acid. Filling the vitreous cavity (posterior surface of the lens to the retinal surface), vitreous humor makes up 80% of total eye volume and maintains the shape of the eye. With scanty cellular elements, it is an almost completely transparent substance that provides a clear pathway for the visual axis. Unlike aqueous humor, vitreous turnover is extremely slow. Hemorrhage within the vitreous comes from one of two sources: ruptured retinal vessels or, rarely, an extraretinal source (i.e., subarachnoid bleed).
Retinal vessels generated in response to chronic ischemia ("neovascularization"), as in the case in diabetic retinopathy and retinal vessel occlusion, are inherently fragile and prone to rupture. Bleeding may be spontaneous or provoked by relatively minor trauma. Diabetic retinopathy is probably the greatest risk factor for vitreous hemorrhage.29 Vessel rupture also may be associated with vitreous-retinal detachment. Aging causes a slow contraction of the vitreous, as the central gel liquifies and collagen fibers condense. As the vitreous collapses, it tends to separate or detach from the retinal epithelium in the posterior area. Posterior vitreous detachment actually is a common and often asymptomatic event in the older adult. If vitreous separation is uneven or sudden, it can exert a pull on the retina. Retinal tears or detachments can occur. Sudden detachment can be precipitated by blunt trauma, rapid eye movements during sleep, or a sudden head position change. Whatever the cause, bleeding into the vitreous leads to rapid clot formation. The extent of visual impairment is related to the size, density, and location of the clot in relation to the visual axis. Other causes of vitreous hemorrhage include macular degeneration, ocular trauma (much more common in younger patients), and sickle cell-related retinopathy. Anticoagulants and blood dyscrasias do not appear to contribute significantly to the risk for vitreous hemorrhage.30
Clinical Presentation. If hemorrhage is preceded by retinal detachment, the patient may experience flashes of light (photopsias). Otherwise, vitreous hemorrhage is experienced as unilateral, painless vision loss, often described as haze, shadows, cobwebs, or smoke. Once blood clots are formed, these will appear as floaters. Visual acuity may not be affected if the clot is away from the visual axis.
Physical examination will reveal an unremarkable external eye. If the hemorrhage is large and light cannot reach the macula, a small afferent pupillary defect occasionally may be noted. (See Figure 5.)
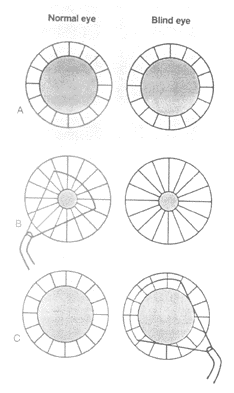
| Figure 5. Afferent Pupillary Defect |
|
|
| Because consensual light response is guaranteed by double hemidecussation of afferent pupillary fibers, pupillary size is the same in the totally blind eye as in the opposite, normal eye (A). Illumination of opposite, normal eye causes direct and consensual papillary miosis (B). Swinging a flashlight to illuminate the blind eye results in apparent dilation of the pupil of the blind eye in response to light (C). Consensual dilation of the blind eye occurs because of loss of illumination of the normal eye. The swinging flashlight test can detect very small monocular visual losses and is extremely useful for distinguishing between monocular hysterical blindness and real visual loss (e.g., retrobulbar neuritis). |
| Reprinted with permission from: Kosmorsky GS. Sudden Painless Visual Loss: Optic Nerve and Circulatory Disturbances. In: Clinics in Geriatric Medicine. W.B. Saunders Co.; 1999;15:9. |
Fundoscopic exam will reveal a normal appearing retina if the bleed is small, or the retina may be completely obscured. Sometimes a red haze is evident if the hemorrhage occurred recently. A large retinal detachment may be visible as a gray flap. Changes of retinopathy frequently are seen. Early ophthalmologist consultation is necessary, especially if retinal detachment is suspected. Evaluation of an obscured retina may be accomplished using ultrasound, which can identify unseen retinal detachments.
Treatment. There are no immediate treatment options for vitreous hemorrhage. Blood within the vitreous humor is metabolized very slowly, a consequence of minimal cellular elements within the avascular gel. Clot resorption proceeds at a very slow rate, approximately 1% per day. Vision may slowly improve over weeks to months. Patients who experienced hemorrhage as a complication of diabetic retinopathy or retinal vessel occlusion tend to have poor recovery, and many will require surgical intervention. ED care for vitreous hemorrhage, after excluding any other treatable conditions such as unrecognized diabetes or trauma, should include early ophthalmic follow-up. Pars vitrectomy, a surgical procedure to remove the vitreous and replace it with a clear substrate such as silicone gel or sodium hyaluronate, may be necessary in severe cases.
Giant Cell (Temporal) Arteritis
Epidemiology. Giant cell (temporal) arteritis is an important cause of visual loss in the elderly patient, and unlike many other causes of blindness, it is potentially preventable. Statistics concerning giant cell arteritis in the literature vary widely, probably due to the difficulty with establishing a single, uniform definition of the disease. Although most often associated with the classic lateralized, temporal headache, ocular manifestations of giant cell arteritis are present in one-half of all affected patients. Vision loss can be more concerning to the patient than headache pain, and may be the impetus for ED evaluation. Furthermore, ocular symptoms may present in the absence of any other systemic problem. This unusual presentation, termed occult giant cell arteritis, has as much potential for permanent visual impairment but is particularly difficult to diagnose.31 Giant cell arteritis usually is not seen before age 50, affects women more then men, and is more common in white individuals. A family history of the disease may be present, as there appears to be some genetic predisposition. For unknown reasons, the incidence of giant cell arteritis appears to be rising.32 If left untreated, up to 50% of patients will become unilaterally blind from this disease.
Pathophysiology. The etiology of this disease remains unknown, although age-related changes in vessel walls has been suggested. Affecting mostly large and medium-size vessels, giant cell arteritis is characterized by vessel wall granulomatous inflammation and accumulation of multinucleated giant cells and other cellular elements within the vessel wall. This frequently occurs in the temporal artery, producing the characteristic, palpable temple cord and associated scalp tenderness. Inflammation also may extend into the ocular circulatory system. If the ophthalmic artery is affected, circulation to the entire eye will be compromised (ocular ischemic syndrome, to be discussed later). Giant cell arteritis has a tendency to involve the posterior ciliary arteries, which provide the majority of blood to the choroid and optic nerve head. Inflammation within the posterior ciliary artery results in optic nerve head ischemia and eventual infarction. Central retinal artery involvement also may occur.
Clinical Presentation. The ocular symptoms experienced by a patient with giant cell arteritis are diverse. Visual loss is common, but may range from subtle to severe. The patient may relate a recent history of amaurosis fugax or diplopia. Distortions such as colored vision, hallucinations, or photopsias have been reported. While monocular complaints are usual, simultaneous bilateral involvement has been documented. Eye pain is not typical; in one study, fewer than 10% of patients reported any ocular discomfort,33 although the majority of patients will complain of headache. Examination of the eye may demonstrate the findings of retinal artery occlusion or optic nerve edema, or may be unremarkable. If present, other somatic complaints commonly associated with giant cell arteritis, such as lateralized headache, jaw claudication, myalgias, or polymyalgia rheumatica, likely will support the clinical suspicion. Laboratory testing can be very helpful with the diagnosis. A markedly elevated erythrocyte sedimentation rate (ESR) and C-reactive protein level are consistent with the diagnosis; however, normal values do not rule out the presence of the disease. It is important to remember that the ESR increases with age: A simple rule of thumb for estimating the upper range of a "normal" ESR in this age group is age/2 for males and (age + 10)/2 for females. While an elevated sedimentation rate is found in about 90% of affected patients, laboratory testing is not 100% sensitive, and the diagnosis may be made on clinical grounds alone.34,35 New imaging modalities, such as color doppler flow studies and laboratory studies, soon may be available to aid with diagnosis.36 The gold standard for diagnosis remains temporal artery biopsy.
Treatment. The first step in successful treatment is recognition of the disease. Including giant cell arteritis in the differential diagnosis of any elderly patient with a visual complaint or unexplained headache, unless another disease process is identified, allows the best chance for correct diagnosis. Early treatment with corticosteroids, which suppress arterial inflammation, is of paramount importance. It has been well-documented that the most important factor in preservation of visual function is prompt initiation of steroid therapy. Prednisone, taken in daily doses of 60-100 mg, should be started immediately. Some studies have recommended considerably higher dosages, up to 2 mg/kg/day, if there is blindness or bilateral eye involvement.37-39 If blindness is present at the time of diagnosis, it may be prudent to administer the first dose of steroids as an IV bolus.38 Methylprednisolone, often in very high doses (1 g/day), can be used for this purpose. For patients who are unable to take steroids, and also for steroid-resistant cases, immunosuppressive agents such as methotrexate or azathioprine may confer some benefit.40-42 Referral to an ophthalmologist for temporal artery biopsy should take place as soon as possible, but can be delayed for up to two weeks if necessary. Treatment should not be withheld for fear of altering the biopsy results. Steroid administration also will help protect the unaffected eye, which is at significant risk of involvement by the disease. Other ED interventions may include non-steroidal anti-inflammatory drugs (NSAIDs) or narcotics to relieve the patient’s pain; some cases may require admission to the hospital. Although a rapid (24-48 hours) resolution of symptoms can be expected with treatment, once vision is lost most patients do not recover visual function. Thus the key remains early diagnosis, treatment, and prevention of visual loss.
Nonarteritic Anterior Ischemic Optic Neuropathy (NAION)
Epidemiology. Ischemic optic neuropathy (ION) is characterized by painless, monocular loss of vision that is due to sudden cessation of ocular blood flow involving the posterior ciliary artery and infarction of the optic nerve head. The infarction may occur close to the optic disk and produce detectable disk swelling (anterior or AION), or, more rarely, may affect the retrobulbar optic nerve with no discernable disk changes (posterior or PION). If associated with giant cell arteritis, the condition is termed arteritic ION; otherwise, the condition is termed nonarteritic. Nonarteritic anterior ischemic optic neuropathy (NAION) is a common cause of vision loss in the elderly population. In patients older than 50 years, NAION is the most common acute optic neuropathy, with an estimated incidence of 2.3 per 100,000 annually.43
Pathophysiology. Nonarteritic anterior optic neuropathy is characterized by infarction of the anterior optic nerve that is unassociated with inflammation, mass compression, or demyelinization disease. It often strikes individuals in their 60s and 70s, and is seen in association with hypertension, ischemic heart disease, hypercholesterolemia, tobacco use, and diabetes.44,45 Familial reports of NAION and a marked Caucasian predilection suggest there is a genetic component to the disorder. NAION also has been reported following cardiac bypass surgery and cataract extraction. The precise pathophysiology remains unknown, but postulated mechanisms include vasculopathy associated with the aforementioned risk factors, and a small cup-disc ratio that results in watershed zones which are more susceptible to ischemia. NAION does not appear to be an embolic process. A recent study examining prothrombotic risk factors (hypercoagulable states) also demonstrated that NAION is not primarily a thrombotic process.45 Although the main risk factors and triggering events remain the subject of debate, NAION is likely the result of a compromised microcirculation in the optic nerve head.
Clinical Presentation. NAION usually is characterized by abrupt, unilateral loss of visual acuity and visual field. Vision loss often is described as altitudinal; that is, the upper or lower half of the visual field is missing. Inferior loss is more common. A large "black dot" (central scotoma) also may be reported. Visual loss usually is maximal at onset and fairly stable, although there is a subset of patients in whom visual acuity deteriorates over several days or weeks.46 There usually is no precipitating event or warning sign. Pain or other visual symptoms are atypical.
Physical examination will reveal a swollen optic disc and an afferent pupillary defect. Peripapillary hemorrhages may be seen in the immediate vicinity of the disk, but the remainder of the retinal exam should be unremarkable.
Treatment. Treatment should begin by ruling out giant cell arteritis as a cause. If history, physical examination, or laboratory testing suggest giant cell arteritis, the patient should be started on steroid therapy immediately. For NAION, no treatment is available for the affected eye. Some patients will have spontaneous improvement; in one study, 31% of patients demonstrated an increase in vision 24 months after diagnosis.47 Optic nerve decompression surgery has not been shown to increase visual recovery.48 Approximately 10% of patients with this disease will develop NAION in the other eye as well. Aspirin may decrease the frequency of second eye involvement.49 Any patient with NAION who can tolerate aspirin (325 mg/day) should begin this therapy immediately. Risk factor reduction (lowering cholesterol, cessation of tobacco, and control of blood sugar) also should be emphasized.
Herpes Zoster Ophthalmicus
Epidemiology. Viral infections of the eye are common among patients of all ages; ocular viral pathogens include herpes simplex virus, adenovirus, enterovirus, and others. Herpes zoster ophthalmicus represents a particularly painful and sight-threatening viral infection that tends to affect older persons. The incidence of zoster increases with age, with approximately 20 cases per 1000 in patients older than 70 years. In all individuals who have had chicken pox, 50% will, by age 80, have experienced at least one episode of herpes zoster. Herpes zoster can appear in almost any part of the body, with ophthalmic involvement in 15% of all cases. Elderly individuals generally experience more pain and complications. Recovery from herpes zoster may be prolonged, and up to 15% of patients will have sequelae.
Pathophysiology. After an episode of chicken pox, varicella zoster virus resides asymptomatically in the sensory nerves. Reactivation may be triggered by many factors, and in the geriatric patient, decreased cell mediated immunity is thought to contribute.50,51 Latent in the trigeminal ganglion, varicella virus is triggered to become active and travels down the trigeminal and ophthalmic nerve. Once it reaches the peripheral nerves, inflammation and vasculitis produce typical skin lesions on the forehead, face, and eyelid. Vesicular and hemorrhagic bullae are characteristic. Direct viral invasion of corneal epithelium results in conjunctivitis and keratitis. Reactive inflammation may produce uveitis or scleritis. Neurologic involvement can result in ocular motor palsies, diffuse orbital vasculitis, or optic neuritis. Zoster iritis, when the iris becomes inflamed and ischemic, may result in long-term visual consequences. Postherpetic neuralgia, a debilitating condition of chronic pain and altered sensation, is a dreaded complication.
Clinical Presentation. The patient who presents with herpes zoster ophthalmicus may recall a prodrome of flu-like symptoms, including headache, fever, malaise, and anorexia, about a week before the rash develops. Viremia is the likely cause of the prodrome, which is then followed by pain that is often severe along the affected dermatome. A vesicular rash is then noted, although there are reported cases of zoster without observable lesions. If the subsequent rash is noted to have spread to the tip of the nose (nasociliary branch), ocular involvement is usually present. The eye is red and diffuse conjunctivitis is seen. The patient may complain of photophobia and lacrimation. Slit lamp and fluorescein exam may reveal keratiti (either punctate or dendritic). Dendrites of herpes zoster are smaller and more superficial than the fern-like dendrites seen in herpes simplex infection. Cranial nerve palsies may occur. Overall, the patient may appear ill.
Treatment. Antivirals are the mainstay of treatment, and best results are achieved when therapy is started within 72 hours of the attack. Antivirals reduce the severity and duration of the current symptoms, and may reduce the incidence of post-infection complications such as post-herpetic neuralgia.52,53 Topical antivirals do not play a role. Oral acyclovir has been the mainstay of treatment, but other antivirals, such as valacyclovir and famcyclovir, are just as effective and easier to dose.54-57 Table 2 lists recommended dosing regimens.
| Table 2. Antiviral Medications | ||
| Medication | Dosage |
Duration |
| Acyclovir | 800 mg 5 x day | 7-21 days |
| Valacyclovir | 1 g 3 x day | 7-14 days |
| Famcyclovir | 500-750 mg 3 x day | 7 days |
Ocular Ischemic Syndrome
Epidemiology. The ocular ischemic syndrome is a little known, uncommon disease of older patients, typically affecting persons in their 60s. There are an estimated 1800 cases per year.60 It describes symptoms and signs that occur when the eye is globally inadequately perfused, usually due to severe stenosis of carotid vessels. Ischemia involves both the posterior ciliary and central retinal artery circulations. More commonly associated with gradual vision loss, it can be the etiology of sudden blindness and eye pain. Most importantly, the recognition and diagnosis of ocular hypoperfusion should alert the practitioner to the likely presence of advanced, even imminently life-threatening, carotid artery disease.
Pathophysiology. Ocular hypoperfusion, defined as inadequate blood flow to the orbit that usually is due to severe stenosis within the carotid vessels, and usually is the common carotid or the internal carotid artery. The ophthalmic artery is the first branch off of the internal carotid artery. Obstruction to blood flow also can be secondary to carotid dissection or artherosclerotic disease in the aorta. The entire eye is globally deprived of circulation, but the sensitive retina and optic nerve fiber cells are the most susceptible to infarction. Progression of the retinal damage may worsen suddenly and manifest as sudden visual loss. Retinal arteries are normally low-flow, high-resistance vessels, and further reduction in flow, such as that caused by carotid stenosis, may lead to stasis and thrombosis and precipitate retinal artery or vein occlusion. Risk factors include diabetes, hypertension, coronary artery disease, and peripheral vascular disease.
Clinical Presentation. The symptoms of ocular hypoperfusion are diverse and may mimic other ocular emergencies. Visual loss, whether gradual or sudden, is the most common symptom. Bilateral involvement, not seen in many other conditions, is not uncommon with this disease. One key symptom is eye pain, a dull ache that extends from the eye toward the temple, that is termed "ocular angina."61 The patient may complain of poor vision after exposure to bright light, a phenomenon known as light-induced amaurosis, in which pigment synthesis is impaired and retinal recovery is delayed.62 Examination of the eye may show signs of retinal ischemia such as pale color and the "cherry red" fovea. Retinal arteries unusually are narrow due to vasoconstriction. Neovascularization of the iris, called rubeosis iridis, is almost pathognomonic for this disease.60 Tonometry may be normal or reveal hypotony, with intraocular pressures of less than 10 mmHg. A complete exam of the patient’s vascular system often reveals diffuse and severe disease. Indeed, symptoms consistent with light-induced amaurosis should alert the clinician to the possibility of significant carotid occlusion.
Treatment. Visual prognosis for patients with ocular ischemic syndrome is poor, and their overall health is at significant risk. For patients with this disease, five-year morbidity has been measured at 40%, with most deaths attributed to cardiac problems or stroke.63 Surgical intervention, such as carotid endarterectomy, may be indicated. Endarterectomy has been shown to have some effect on improving vision,63 but results have been variable.64 In the ED, as a temporizing measure, a patient with suspected ocular ischemic syndrome might benefit from further lowering of the intraocular pressure.65 Prompt referral for vascular and retinal consults is mandatory.
Summary
Eye complaints commonly are seen among older ED patients. Emergency physicians must be aware that older patients, in particular, are at risk for ophthalmologic emergencies and that many of these conditions are potentially sight-threatening. Optimal outcomes depend on quickly assessing these disorders and beginning appropriate treatment. (See Figure 6.)
| Figure 6. Visual Loss Algorithm | |
| GCA
= giant cell arteritis OIS = ocular ischemic syndrome VH = vitreous hemorrhage |
CRAO
= central retinal
artery occlusion CRVO = central retinal vein occlusion NAION = nonarteritic anterior ischemic optic neuropathy |
| Emergency physicians also must communicate effectively with their ophthalmology colleagues to arrange further assessment and follow-up. | |
References
1. McCaig LF, Stussman BJ. National Hospital Ambulatory Medical Care Survey: 1996 Emergency Department Summary. Hyattsville, MD: National Center for Health Statistics, Centers for Disease Control and Prevention; 1997. Advance Data From Vital and Health Statistics, No. 293.
2. Nash EA, Margo CE. Patterns of emergency department visits for disorders of the eye and ocular adnexa. Arch Ophthalmol 1998; 116:1222-1226.
3. Glynn RJ, Seddon JM, Krug JH, et al. Falls in elderly patients with glaucoma. Arch Ophthalmol 1991;109:205-210.
4. Owsley C, McGwin G, Ball K. Vision impairment, eye disease, and injurious motor vehicle crashes in the elderly. Ophthalmic Epidemiol 1998;5:101-103.
5. Parrish RK, Gedde JJ, Scott IU, et al. Visual function and quality of life among patients with glaucoma. Arch Ophthalmol 1997; 115:1447-1455.
6. Quigley HA. Number of people with glaucoma worldwide. Br J Ophthalmol 1996;80:389-393.
7. Pizzarello LD. The dimensions of the problem of eye disease among the elderly. Ophthalmol 1987;94:1191-1195.
8. Erie JC, Hodge DO, Gray DT. The incidence of primary angle-closure glaucoma in Olmsted County, Minnesota. Arch Ophthalmol 1997;115:177-181.
9. Congdon N, Wang F, Tielsch JM. Issues in the epidemiology and population-based screening of primary angle closure glaucoma. Surv Ophthalmol 1992;36:411-423.
10. Hall SK. Acute angle-closure glaucoma as a complication of combined beta-agonist and ipratropium bromide therapy in the emergency department. Ann Emerg Med 1994;4:844-847.
11. Palmberg P. A topical carbonic anhydrase inhibitor finally arrives. Arch Ophthalmol 1995;113:985-986.
12. Diamond JP. Systemic adverse effects of topical ophthalmic agents. Implications for older patients. Drugs Aging 1997;11: 352-360.
13. Nygaard HA, Hording G. Adverse effects of local use of beta-blocker in glaucoma: A literature review and a survey of reports and the adverse drug reaction authority 1985-1995. Tidsskr Nor Laegeforen 1997;117:2019-2021.
14. Johns MD, Ponte CD. Acute pulmonary edema associated with ocular metipranolol use. Ann Pharmacother 1995;29:370-373.
15. Diggory P, Heyworth P, Chau G, et al. Unsuspected bronchospasm in association with topical timolol—A common problem in elderly people: Can we easily identify those affected and do cardioselective agents lead to improvement? Age Ageing 1994;1:17-21.
16. Choong YF, Irfan S, Menage MJ. Acute angle closure glaucoma: An evaluation of a protocol for acute treatment. Eye 1999;13:613-616.
17. Zimmerman TJ, Kooner KS, Kandarakis AS, et al. Improving the therapeutic index of topically applied ocular drugs. Ann Ophthalmol 1984;102:551-553.
18. Singh K, Bautista RD. Advances in glaucoma therapy. Int Ophthalomol Clin 1999:39:1-15.
19. Atebara NH, Brown GC, Cater J. Efficacy of anterior chamber paracentesis and Carbogen in treating acute nonarteritic central retinal artery occlusion. Ophthalmology 1995;102:2029-2034.
20. Beatty S, Au Eong KG. Acute occlusion of the retinal arteries: Current concepts and recent advances in diagnosis and management. J Accid Emerg Med 2000;17:324-329.
21. Neubauer AS, Mueller AJ, Schriever S, et al. Minimally invasive therapy for clinically complete central retinal artery occlusion-results and meta-analysis of literature. Klin Monatsbl Augenheilkd 2000;217:30-36.
22. Beatty S, Au Eong KG. Local intra-arterial fibrinolysis for acute occlusion of the central retinal artery: A meta-analysis of the published data. Br J Ophthalmol 2000;84:914-916.
23. Padolecchia R, Puglioli M, Ragone MC, et al. Superselective intraarterial fibrinolysis in central retinal artery occlusion. Am J Neuroradiol 1999;20:565-567.
24. Weber J, Remonda L, Mattle HP, et al. Selective intra-arterial fibrinolysis of acute central retinal artery occlusion. Stroke 1998;29: 2076-2079.
25. Richard G, Lerche RC, Knospe V, et al. Treatment of retinal arterial occlusion with local fibrinloysis using recombinant tissue plasminogen activator. Ophthalmology 1999;106:768-773.
26. Glacet-Bernard A, Kuhn D, Vine AK, et al. Treatment of recent onset central retinal vein occlusion with intravitreal tissue plasminogen activator: A pilot study. Br J Ophthalmol 2000;84: 609-613.
27. Vallee JN, Masin P, Aymard A, et al. Superselective ophthalmic arterial fibrinolysis with urokinase for recent severe central retinal venous occlusion: Initial experience. Radiology 2000;216:47-53.
28. Lindgren G, Sjodell I, Lindblom B. A prospective study of dense spontaneous vitreous hemorrhage. Am J Ophthalmol 1995;119: 458-465.
29. Dana MR, Werner MS, Viana MA. Spontaneous and traumatic vitreous hemorrhage. Ophthalmology 1993;100:1377-1383.
30. Spraul CW, Grossniklaus HE. Vitreous hemorrhage. Surv Ophthalmol 1997;42:3-39.
31. Hayreh SS, Podhajsky PA, Zimmerman B. Occult giant cell arteritis: Ocular manifestations. Am J Ophthalmol 1998;125:521-526.
32. Salvarani C, Gabriel SE, O’Fallon WM, et al. The incidence of giant cell arteritis in Olmstead County, Minnesota: Apparent fluctuations in a cyclic pattern. Ann Intern Med 1995;123:192-194.
33. Hayreh SS, Podhajsky PA, Zimmerman B. Ocular manifestations of giant cell arteritis. Am J Ophthalmol 1998;125:509-520.
34. Hayreh SS, Podhajsky PA, Raman R, et al. Giant cell arteritis: Validity and reliability of various diagnostic criteria. Am J Ophthalmol 1997;123:285-296.
35. Ellis JD, Munro P, McGettrick P. Blindness with a normal erythrocyte sedimentation rate in giant cell arteritis. Br J Hosp Med 1994;52:358-359.
36. Ghanchi FD, Dutton GN. Current concepts in giant cell (temporal) arteritis. Surv Ophthalmol 1997;42:99-123.
37. Hayreh SS. Ophthalmic features of giant cell arteritis. Baillieres Clin Rheumatol 1991;5:431-459.
38. Liu GT, Glaser JS, Schatz NJ, et al. Visual morbidity in giant cell arteritis. Clinical characteristics and prognosis for vision. Ophthalmology 1994;101:1779-1785.
39. Rosenfeld SI, Kosmorsky GS, Klingele TG, et al. Treatment of temporal arteritis with ocular involvement. Am J Med 1986;80:143-145.
40. Hernandez GC, Soriano C, Morado C. Methotrexate treatment in the management of giant cell arteritis. Scand J Rheumatol 1994;23: 295-298.
41. Krall PI, Mazance DJ, Wilke WS. Methotrexate for corticosteroid resistant polymyalgia rheumatica and giant cell arteritis. Clev Clin J Med 1989;56:253-257.
42. Wilke WS, Hoffman GS. Treatment of corticosteroid resistant giant cell arteritis. Rheum Dis Clin North 1995;21:59-71.
43. Johnson LN, Arnold AC. Incidence of nonarteritic and arteritic anterior ischemic optic neuropathy: Population based study in the state of Missouri and Los Angeles county. J Neuroophthalmol 1994;14:38-44.
44. Selvin GJ, Townsend JC. Clinical pearls in optometric management of the geriatric patient. J Am Optom Assoc 1994;65:49-57.
45. Salomen O, Huna-baron R, Kurtz S, et al. Analysis of prothrombotic and vascular risk factors in patients with nonarteritic anterior ischemic optic neuropathy. Ophthalmology 1999;106:739-742.
46. Potarazu S. Ischemic optic neuropathy: Models for mechanism of disease. Clin Neurosci 1997;4:264-269.
47. Ischemic Optic Neuropathy Decompression Trial: Twenty-four month update. Arch Ophthalmol 2000;118:793-798.
48. The Ischemic Optic Neuropathy Decompression Trial Research Group. Optic nerve decompression surgery for nonarteritic anterior optic neuropathy is not effective and may be harmful. JAMA 1995;273:625-632.
49. Salomon O, Huna-Baron R, Steinberg DM, et al. Role of aspirin in reducing the frequency of second eye involvement in patients with non-arteritic anterior ischemic optic neuropathy. Eye 1999;13: 357-359.
50. Weksler ME. Immune senescence. Ann Neuro 1992;35:35-37.
51. Burke BL, Steele RW, Beard OW, et al. Immune responses to varicella-zoster in the aged. Arch Intern Med 1982;142:291-293.
52. Kost RG, Straus SE. Post-herpetic neuralgia-pathogenesis, treatment, and prevention. N Engl J Med 1996;335:32-42.
53. Marsh RJ. Herpes zoster ophthalmicus. J R Soc Med 1997;90:7670-674.
54. Tyring S, Barbarash RA, Nahlik JE. Famcicovir for the treatment of acute herpes zoster: Effects on acute disease and postherpetic neuralgia; A randomized, double-blind placebo-controlled trial. Ann Intern Med 1995;123:89-96.
55. De Greef H. Famciclovir, a new oral antiherpes drug: Results of the first controlled clinical study demonstrating its efficacy and safety in the treatment of uncomplicated herpes zoster in immunocompetent patients. Int J Antimicrob Agents 1995;4:241-246.
56. Beutner KR, Friedman DdJ, Forszpaniak C, et al. Valacyclovir compared with acyclovir for improved therapy for herpes zoster in immunocompetent adults. Antimicrob Agents Chemother 1995;39: 1546-1553.
57. Colin J, Prisant O, Cochener B, et al. Comparison of the efficacy and safety of valacyclovir and acyclovir for the treatment of herpes zoster ophthalmicus. Ophthalmology 2000;107:1507-1511.
58. Kaufman HE. Treatment of viral diseases of the cornea and external eye. Prog Retin Eye Res 2000;19:69-85.
59. Morgan R, King D. Characteristics of patients with shingles admitted to a district general hospital. Postgrad Med J 1998;74:101-103.
60. Kosmorsky GS. Sudden painless visual loss: Optic nerve and circulatory disturbances. Clin Geriatr Med 1999;15:1-13.
61. Brown GC, Margargal LE. The ocular ischemic syndrome. Clinical, fluorescein angiographic and carotid angiographic features. Int Ophthalmol 1988;11:239-251.
62. Roberts DK, Sears JM. Light-induced amaurosis associated with carotid occlusive disease. Optom Vis Sci 1992;69:889-897.
63. Silvalingam A, Brown GC, Margargal LE, et al. The ocular ischemic syndrome. Mortality and systemic morbidity. Int Ophthalmol 1989;13:187-191.
64. Silvalingam A, Brown GC, Margargal LE . The ocular ischemic syndrome III. Visual prognosis and the effect of treatment. Int Ophthalmol 1991;15:15-20.
65. Mizener JB, Podhajsky P, Hayreh SS. Ocular ischemic syndrome. Ophthalmology 1997;104:859-864.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.