Special Supplement-The Evolution of Antiretroviral Therapy: Applying Clinical Trial Data to Optimize HAART in the Management of HIV
The Evolution of Antiretroviral Therapy: Applying Clinical Trial Data to Optimize HAART in the Management of HIV
Introduction
The primary therapeutic goals when initiating therapy in patients infected with HIV are maximal and long-lasting suppression of viral replication, maintenance or restoration of immunologic function, improvement of quality of life, and reduction of HIV-related morbidity and mortality.1 Highly active antiretroviral therapy (HAART) using combinations of antiretroviral agents has been shown to achieve these goals to a significant degree and reduce morbidity and mortality in the population of HIV-infected patients.2
Despite the proven efficacy of HAART, virologic failure may occur due to a number of identifiable factors.35 These factors include the potency of the specific antiretroviral regimen,4 patient adherence to the prescribed treatment regimen, and the ability of a chosen regimen to select for antiviral-resistant mutations.3
Although once-daily dosing of certain HAART agents may increase adherence, the most convenient regimens do not necessarily provide the most optimal efficacy. Recent data concerning the efficacy of certain triple nucleoside reverse transcriptase inhibitor (NRTI)-containing regimens in patients with HIV are important, since such data ultimately affect evidence-based guidelines for the selection of initial therapy in HIV-infected patients. This continuing education supplement will focus on studies that assessed triple nucleoside-containing regimens and the most recent guidelines from the Department of Health and Human Services (DHHS) for the timing of HAART initiation and recommended antiretroviral drug classes for managing treatment-naïve patients with HIV.
History and Goals of Antiretroviral Therapy
Since the recognition of HIV disease, the primary medical goal has been to decrease morbidity and mortality related to HIV infection and AIDS. Initially, there were no treatments for HIV itself, and health care providers largely treated opportunistic infections to forestall death. In 1987 zidovudine (ZDV, or AZT), an NRTI, was the first antiretroviral approved. Used as monotherapy, ZDV delayed HIV disease progression, but its efficacy was relatively short lived. As other NRTIs were approved, sequential monotherapy—discontinuing one NRTI and replacing it with another—and dual NRTI therapy were employed. While both approaches provided some benefit, they were not sufficient enough to make a substantial impact on disease progression. When non-nucleoside reverse transcriptase inhibitors (NNRTIs), a second class of antiretrovirals, became available, they were commonly added to patients’ current, failing regimens. As with the earlier approaches, this use of NNRTIs had little, if any, efficacy.
Today we understand why the earlier approaches did not work; monotherapy and dual therapy are not potent enough to suppress viral replication, allowing HIV to develop drug resistant mutations. Similarly, adding an NNRTI to a failing regimen did not provide sufficient antiviral activity, and resistance to the NNRTI would quickly develop. With the approval of protease inhibitors (PIs), researchers also discovered how to use antiretrovirals effectively. Using 3 drugs—initially a PI and 2 NRTIs—simultaneously suppressed viral replication in many patients. Because replication was minimized, development of mutations was uncommon. Two surrogate markers used in HIV clinical research—HIV RNA (viral load [VL]) and CD4 cell counts—responded well to what became know as combination therapy or HAART, and morbidity and mortality associated with HIV disease dropped dramatically.
According to the 2004 DHHS HIV treatment guidelines, the goals of antiretroviral therapy are:1
- Maximal and long-lasting suppression of viral replication;
- Maintenance or restoration of immunologic function;
- Improved quality of life; and
- Reduction of HIV-related morbidity and mortality.
Although these goals have been consistent over time, how to achieve them undergoes change as greater understanding of HIV and its treatment is attained through clinical research. For instance, while PI-based regimens are potent and can suppress viral replication, these regimens were and sometimes still are inconvenient and difficult to tolerate. Because long-term toxicity was also a concern, new approaches were sought. Regimens consisting of 3 NRTIs had early evidence of efficacy, and these regimens were often better tolerated, had a better long-term safety profile, and were convenient. Use of 2 NRTIs plus an NNRTI, rather than a PI, also demonstrated efficacy with good tolerability, safety, and convenience.
Clinical Trials: Recent Assessments of Various Triple Nucleoside Regimens as HAART for Treatment-Naïve HIV-Infected Patients
From a historical perspective, it is useful to review efficacy results from 2 earlier trials that assessed triple NRTI regimens when considering some of the emerging data from the past year. Early studies of triple NRTIs found comparable efficacy to PI-based regimens, at least for patients starting with a baseline VL <100,000 copies/mL. The convenience of triple NRTI regimens, particularly fixed dose ABC/3TC/ZDV, and their improved safety profile, made all NRTI regimens an attractive option for treatment-naïve patients. However, recent clinical studies have found comparatively poor results with several different triple NRTI combinations as first-line therapy.
CNAAB3005/CNA3014: Efficacy of Triple NRTI Therapy With ABC/3TC/ZDV
A phase 3, multicenter, randomized, double-blind trial, CNAAB3005, was conducted by Staszewski and colleagues to evaluate the efficacy and safety of ABC/3TC/ZDV (4 pills/day) compared with indinavir (IDV)/3TC/ZDV (8 pills/day) in antiretroviral-naïve HIV-infected patients.6 At week 48, in this treatment population in which approximately one-third of patients possessed baseline HIV RNA values >100,000 copies/mL, approximately half of patients in each arm had a VL <400 copies/mL. However, virologic suppression to <50 copies/mL was better in the IDV arm, and this difference was statistically significant when stratified by baseline VL >100,000 copies/mL. Analyses at week 48 showed advantages for the ABC arm for HIV RNA <400 copies/mL and <50 copies/mL. In contrast with CNAAB3005, ABC resulted in better virologic outcomes than IDV regardless of whether baseline VL was < or >100,000 copies/mL.7
ACTG 5095 and Observational Cohort Study of ABC/3TC/ZDV
The efficacy of the triple NRTI regimen ABC/3TC/ZDV has been assessed in ACTG 5095 (A5095) and in a large observational cohort study. In A5095, Gulick and colleagues conducted a phase 3, randomized, double-blind, placebo-controlled comparison of 3 PI-sparing regimens for the initial treatment of HIV infection in patients with a VL of >400 copies/mL and any CD4 cell count.8 Patients were monitored for virologic response and safety following randomization to a triple NRTI regimen (ABC/3TC/ZDV), a 4-drug regimen (ABC/3TC/ZDV + efavirenz [EFV]), or ZDV/3TC+EFV (control arm). The primary objectives of A5095 were to determine the safety and tolerability of the regimens. A5095 was also designed to compare virologic failure rates (defined as 2 consecutive HIV RNA ³ 200 copies/mL at week 16 or later) for ABC/3TC/ZDV + EFV and ZDV/3TC + EFV. Patients were stratified on the basis of whether they had a pretreatment VL> or <100,000 copies/mL. Baseline characteristics were comparable between study groups and patient demographics are listed (see Table 1, below).

Virologic failure occurred in 167 patients, 82/382 (21%) in the ABC/3TC/ZDV compared with 85/765 (11%) in the pooled EFV arms of the study; P<0.001, log rank test. Moreover, virologic failure occurred significantly earlier in the ABC/3TC/ZDV group compared with the EFV-containing treatment groups (P<0.001, log rank treatment comparisons). This finding was consistent, regardless of baseline VL < or >100,000 copies/mL (P<0.001 for each). At 48 weeks, 74% and 89% of patients had their HIV RNA suppressed to <200 copies/mL and 61% and 83% of patients achieved HIV RNA <50 copies/mL, in the ABC/3TC/ZDV and the EFV-containing treatment groups, respectively (intent-to-treat analysis). Grade 3 and 4 adverse events were similar between groups. In post-hoc analysis it was shown that even among patients who had their HIV RNA suppressed to at least 200 copies/mL, virologic failure occurred earlier in the ABC/3TC/ZDV group compared with EFV-containing treatment groups (P<0.001). The risk of virologic failure was approximately 7% over 3 months for patients receiving ABC/3TC/ZDV compared with 3.5% for subjects on the combined EFV-containing treatment groups.9 Based upon these interim results, the ABC/3TC/ZDV arm of A5095 was terminated.
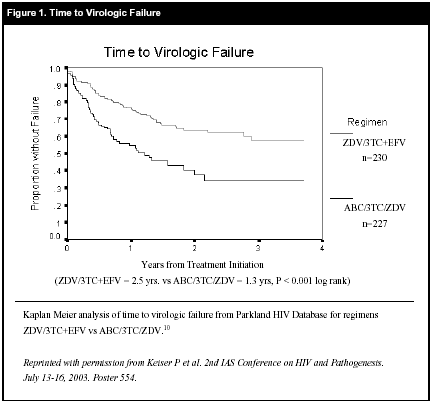
Similar results were found in an observational cohort comparison of ZDV/3TC+EFV to ABC/3TC/ZDV conducted in antiretroviral-naïve patients.10 In this cohort, the time to virologic failure was shorter in the ABC-containing arm (1.3 years) than in the EFV-containing arm (2.5 years) (P<0.001) (see Figure 1, below). This difference in time to treatment failure between the 2 groups also was demonstrated when results were stratified by baseline VL (< or >100,000 copies/mL) or by patients who responded to antiretroviral therapy by suppressing HIV RNA to <400 copies/mL.
Lower Efficacy With Stavudine, Didanosine, and ABC Triple NRTI Regimen
The triple NRTI regimen stavudine (d4T), didanosine (ddI), and ABC was evaluated in a randomized, controlled, open-label trial by Gerstoft and colleagues who compared it to ZDV + 3TC in combination with either ritonavir (RTV)/saquinavir (SQV) or nelfinavir (NFV)/nevirapine (NVP). It had been assumed that the triple NRTI regimen possessed a low potential for the cross-resistance mutations K65R and L74V—previously relatively rare in vivo when ABC and ddI were combined.11 The primary endpoint was plasma HIV RNA <20 copies/mL after 48 weeks. At week 48 (intent-to-treat [ITT] population), a significantly lower proportion of patients demonstrated a VL <20 copies/mL in the d4T/ddI/ABC arm (43%) compared with the NFV/NVP-containing arm (69%) (P<0.01). This regimen was particularly poor for patients with a higher baseline VL or AIDS, who were significantly less likely to achieve a VL <20 copies/mL by week 48. Additionally, side effects were also more frequent in the triple NRTI arm (e.g., mitochondrial toxicity, neuropathy, and lactatemia). The investigators suggest that a reason for the frequent treatment failure with this NRTI combination might have been an acceleration of cross-class resistance. Also, the K65R mutation that was presumably avoidable with this triple NRTI regimen was found in 5 patients.
Tenofovir DF (TDF)/ABC/3TC Versus EFV/ABC/3TC in Antiretroviral-naïve HIV Patients
TDF/ABC/3TC in treatment-naïve HIV-infected patients was evaluated in ESS30009 and a pilot study by Farthing. In ESS30009, an open-label, randomized, multicenter trial, Gallant and colleagues compared the virologic suppression associated with TDF/ABC/3TC versus EFV/ABC/3TC in adult, antiretroviral-naïve HIV patients with a VL >5000 copies/mL.12 Subsequent to several cases of early virologic non-response in the TDF/ABC/3TC treatment arm, an unplanned interim analysis was conducted in patients with 8 or more weeks of HIV RNA data (see Table 2 and Figure 2). Virologic non-response was defined as <2.0 log10 decline in HIV RNA by treatment week 8 or a 1.0 log10 increase above nadir on any subsequent visit. By week 8, virologic non-response was reported for 50/102 (49%) in the TDF/ABC/3TC treatment group compared with 5/92 (5.4%) patients in the EFV/ABC/3TC group (P<0.001). For subjects with virologic non-response to TDF/ABC/3TC, with available genotypes (n=36), 100% had developed the M184V mutation. Over half of those patients with M184V also had developed K65R (n=23). Because of these results, the TDF/ABC/3TC arm was terminated with no change in the EFV/ABC/3TC arm. Long-term data from the EFV/ABC/3TC treatment group are forthcoming.
Similarly, Farthing and colleagues have reported early virologic failure in a pilot study in treatment-naïve HIV-infected patients who received TDF/ABC/3TC.13 In that study (n=20 enrolled; 9 patients with a VL >100,000 copies/mL; 3 withdrawals), the mean VL declined from 82,381 copies/mL at baseline to only 14,898 copies/mL by week 4; 8219 copies/mL by week 8. Moreover, from 17 patients, 9 (52%) demonstrated VL rebound: 1 at week 4, 6 at week 8, and 2 at week 16. The study was prematurely terminated due to these suboptimal efficacy findings, which also suggest a particularly high risk for early virologic failure with this regimen in patients with an initial VL >100,000 copies/mL. A small retrospective observational study prompted by results of the Farthing study found that 5 of 8 patients had virologic failure subsequent to a switch from an antiretroviral regimen in which they were fully suppressed to the TDF/ABC/3TC combination.14 As such, this combination also appears to be suboptimal as an alternate treatment for patients on successful regimens, as well as in treatment-naïve HIV-infected patients.
A number of explanations for these high rates of virologic failure with the TDF/ABC/3TC regimen have been proposed, including low genetic barrier to resistance and pharmacokinetic interactions. However, preliminary results from another pilot study in which early virologic failure occurred in patients who received this combination point to a low genetic barrier to resistance as the major cause of failure. In that study, this hypothesis is supported by the association of adequate expected plasma Cmin of the 3 antiretroviral agents, high proportion of early virologic failure, and high rate of rapid selection for the K65R and M184V mutations in patients with a plasma VL above 4 log10 copies/mL.15
Atlantic Study: An Assessment of d4T/ddI/3TC
The Atlantic Study, a randomized, open-label trial compared the relative efficacy of 2 convergent treatment regimens (in which all antiretrovirals target reverse transcriptase [i.e., NRTIs and NNRTIs]) and one divergent regimen (targeting reverse transcriptase and protease) in treatment-naïve HIV-infected patients with plasma HIV RNA >500 copies/mL and CD4 counts >200 x 106 cells/L.16 Patients were recruited from the regular patient population at 17 international centers and randomized to receive d4T and ddI plus either IDV, NVP, or 3TC. The primary endpoint was the proportion of patients in the ITT population with a plasma VL <500 copies/mL after 48 weeks of treatment. Treatment failure was defined as plasma VL <500 copies/mL at any point after 24 weeks follow-up.
Of the 298 patients enrolled in the study, 198 (66%) were observed through week 48; 139 (47%) through week 96. At 48 weeks follow-up, suppression of VL to <50 copies/mL in the on-treatment (OT) population was significantly lower in the 3TC-containing arm (58.7%) compared with the IDV- (80.3%) and NVP-containing arms (80.7%) (P=0.004). Moreover, at 96 weeks follow-up, both the ITT and OT populations in the 3TC arm had a significantly lower proportion of patients with a VL <50 copies/mL. The incidences of grade 3 and 4 adverse events, as well as the rate of discontinuation due to an adverse event, were comparable between the treatment arms, indicating equivalent tolerability. The convergent regimen used in this trial, then, was inferior with regard to the potency of viral suppression compared with the divergent regimens.
CLASS: Triple NRTI d4T/3TC/ZDV Versus NNRTI- and PI-based HAART
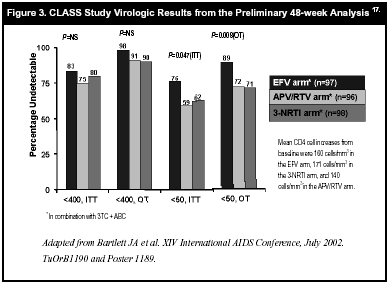
In the 96-week, open-label, randomized, multicenter Clinically Significant Long-term Antiretroviral Sequential Sequencing (CLASS) study, Bartlett and colleagues compared EFV, amprenavir (APV)/RTV, or d4T added to the nucleoside backbone 3TC/ABC in treatment-naïve, HIV-infected adults with plasma HIV RNA >5000 copies/mL.17 Forty-two percent of patients presented with a VL >100,000 copies/mL. At week 48, the proportion of patients who achieved a VL <50 copies/mL were 76%, 59%, and 62% for the EFV-, APV/RTV-, and d4T-containing arms, respectively (ITT: P=0.047; observed: P=0.008) (see Figure 3, below). Moreover, of patients with a baseline VL >100,000 copies/mL, 77% of patients who received EFV achieved a VL <50 copies/mL compared with 55% in the triple NRTI arm.
Efficacy From Trials With Quadruple PI-Sparing Therapy
In light of the recent evidence of suboptimal efficacy with certain triple NRTI regimens reviewed above, the efficacy of PI-sparing quadruple therapy (i.e., triple NRTI + NNRTI or triple NRTI + a nucleotide) is of substantial clinical interest.
ABC/3TC/ZDV in combination with TDF was studied in a pilot, open-label, multicenter study in 88 treatment-naïve subjects with HIV (baseline median VL and CD4 count 5.1 log10 copies/mL and 226 cells/mm3, respectively).18 By week 24, 78% (42/54) and 67% (36/54) had VL <400 copies/mL and <50 copies/mL, respectively (observed analysis). Those with virologic non-response were more likely to have had baseline VL >100,000 copies/mL.
ABC/3TC/ZDV + EFV was assessed in a multicenter, open-label, pilot study (CNAF3008) conducted in 31 treatment-naïve HIV-infected adults (baseline VL 4.69 log10 copies/mL, CD4 cell count 322 cell/mm3, 13/31 VL >100,000 copies/mL).19 Potent and durable antiretroviral activity resulted from this combination by week 48 with a median reduction of 2.7 log10 copies/mL by week 4 and 58% of patients having reduced their VL to <50 copies/mL at week 8.
ACTG 384, a randomized, double-blind comparison of EFV and NFV paired with either 3TC/ZDV or ddI/d4T, used a multifactorial approach to evaluate a number of treatment strategies. Results of this study showed improved outcomes when first-line treatment consisted of EFV/3TC/ZDV and no significant difference in durability between a single 4-drug regimen and 2 sequential 3-drug regimens.20,21
Summary
Results from earlier studies comparing the triple NRTI regimen ABC/3TC/ZDV with PI-based regimens found comparable efficacy results, at least for patients with lower baseline viral loads. The convenience, tolerability, and safety profile of ABC/3TC/ZDV made it, and other triple NRTI combinations, an attractive option for first-line treatment. More recent studies, however, have found high rates of virologic failure with ABC/3TC/ZDV and several other triple NRTI regimens. In addition, HIV treatment has changed considerably over the last few years. NNRTIs offer a low pill burden, with EFV dosed QD and NPV dosed BID. With PIs, ritonavir boosting allows greater convenience with high potency. Newer PIs also offer advantages. For instance, atazanavir is dosed once daily and has a favorable lipid profile, and fosamprenavir has a lower pill burden than the parent drug, amprenavir. As a result, while potent, tolerable, convenient, and safe regimens are still needed, today’s options are much better than a few years ago.
The Use of HAART in Treatment-Naïve HIV-Infected Patients
HAART is the standard of care in the treatment of HIV/AIDS; and the DHHS Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents specify a number of multiple-drug combinations.1 Due to the vast armamentarium of antiretroviral agents available, many combinations are possible. The timing and proper selection of the initial therapy for HIV infection is a clinical decision with profound consequences. For instance, the use of PI-based HAART (NNRTI-sparing) regimens preserves NNRTIs for use in the event of treatment failure; NNRTI-based (PI-sparing) regimens preserve PIs for subsequent antiretroviral use. The use of a triple NRTI regimen preserves both PI- and NNRTI-based regimens. The timing for initiation of HAART is also important and requires consideration of the benefits and risks of early versus delayed therapy. A substantial body of evidence now exists to guide clinicians as to the timing and selection of initial therapy in the untreated patient with HIV.
PI-based and PI-sparing First-line HAART Regimens. The proper selection of the initial antiretroviral regimen is important because: 1) initial therapy generally is more successful than subsequent regimens in reducing VL; and 2) proper sequencing of initial therapy will not limit possibilities for second-line treatment in the event of treatment failure.6 Regimens of a PI plus 2 different NRTIs have the advantage of the longest experience and most available clinical data. Conversely, the disadvantages of traditional PI-based regimens may include complexity, the large number of pills patients must take (as many as 1416 pills daily), long-term toxicity, and the potential to compromise the effectiveness of future PI regimens. However, newer treatment strategies, such as using ritonavir as a boosting agent, and newer PIs, such as atazanavir, can mitigate these drawbacks.
PI-sparing HAART regimens have been shown to provide long-term control of HIV replication,20 and there are a number of advantages to NNRTI-based regimens, including their antiretroviral potency and possibility of reserving PIs for potential use in the event that they are required.16 An additional advantage of NNRTI-based therapy is the regimen’s simplicity and requirement for fewer pills. Triple-NRTI therapy, another PI-sparing approach, appears to have relatively inferior potency for initial use in HIV-infected patients based upon the recent evidence presented in this supplement.
DHHS Recommended Regimens for the Initial Treatment of HIV. Selection of any regimen should be individualized and based upon respective advantages and disadvantages, and when possible, selected in an evidence-based manner. A number of factors need to be considered when initiating antiretroviral therapy in previously untreated patients (see Table 3, below). The most recent DHHS guidelines define either preferred or alternative HAART regimens. Regimens designated as preferred have clinical data that suggest optimal efficacy and durability with acceptable tolerability and ease of use. Alternative regimens have clinical data to support their efficacy, but have inherent disadvantages compared with preferred regimens, such as inferior antiretroviral activity, durability, tolerability, or ease of use. The most extensively studied and well-characterized HAART regimens are NNRTI-based (e.g., 1 NNRTI + 2 NRTIs), PI-based (1 or 2 PIs + 2 NRTIs), or triple NRTIs.1 The DHHS (March 2004) preferred and alternative HAART regimens are listed in Figures 4a, 4b, and 4c.

The recommendation that EFV be utilized as the preferred NNRTI-based regimen is based upon evidence from 2 large studies that compared it to PI-based regimens. At 48 weeks in the open-label DMP006 (assessed EFV and IDV on a backbone of ZDV/3TC [n=450]), significantly more patients in the EFV arm (70%) achieved a VL <400 copies/mL, compared with the IDV arm (48%; P<0.001). Moreover, a subgroup analysis determined the EFV-containing regimen is significantly more effective than the IDV-containing regimen in its ability to achieve VL <50 copies/mL in patients with a baseline VL >100,000 copies/mL.22 Importantly, the durability of response (DOR) of the EFV regimen was substantiated by a 144-week follow-up in which 55% and 52% in the EFV/ZDV/3TC arm compared with 34% and 30% in the IDV/ZDV/3TC arm had a VL <400 copies/mL and <50 copies/mL, respectively (P<0.05).22 Further, in a 6-arm comparison of sequential 3-drug regimens (ACTG 384), patients received EFV or nelfinavir (NFV) as part of an initial regimen, with background NRTI agents randomized by factorial design (ddI + d4T versus ZDV + 3TC). Compared with NFV, patients who received EFV initially demonstrated fewer events of virologic failure; also, a more favorable benefit was observed for EFV among those assigned to ZDV + 3TC. As such, this study strongly suggested EFV/ZDV/3TC as a particularly useful initial antiretroviral regimen.24
The use of PIs in combination with NRTIs has been evaluated in a number of controlled trials. Due to its low tolerability, regimens with full-dose ritonavir (RTV) are not recommended. However, low-dose (RTV-boosted) regimens often are used due to their reduced pill burden, improved scheduling, and elimination of food restrictions (i.e., IDV). A large study evaluated the DHHS-preferred PI-based regimen of the boosted PI lopinavir (LPV)/RTV (400 mg/100 mg BID) compared with NFV (750 mg TID), each with 2 NRTIs. At week 48, the boosted PI was demonstrated superior to NFV in maintaining a VL <400 copies/mL (75% vs 63%; P<0.001), VL <50 copies/mL (67% vs 52%; P<0.001), and was well tolerated.1,24 Noting that there are few trials comparing LPV/RTV with other boosted-PI regimens or with EFV-based regimens, the authors base the preference for LPV/RTV on "virologic potency, patient tolerance, and pill burden."1
The DHHS guidelines now recommend triple NRTI regimen ABC + 3TC (or d4T) + ZDV only be used for the initial treatment of HIV when NNRTI- and PI-based regimens cannot or should not be used. Moreover, the triple RTI regimens ABC + TDF + 3TC or ddI + TDF + 3TC are not recommended for use as the sole combination antiretroviral regimen initially or at any time.1 These new recommendations are based upon the literature reviewed previously in this supplement.68,12,16,17,26
Guidelines: Initiating Antiretroviral Therapy for the HIV-Infected Patient. Although there is strong evidence in favor of treating HIV-infected patients with CD4 counts <200 cells/mm3, the optimal time to initiate therapy for asymptomatic patients with CD4 counts >200 cells/mm3 is not known.1 A carefully balanced assessment of the pros and cons of initiating therapy must be undertaken.
Early initiation of antiretroviral therapy for HIV-infected patients has significant advantages, which include:1
- Earlier suppression of viral replication;
- Preservation of immune function;
- Prolongation of disease-free survival;
- Reduced risk of developing treatment resistance with complete viral suppression; and
- Possible decreased risk of HIV transmission.
Conversely, a number of potential risks are associated with early initiation of antiretroviral therapy, such as:
- Drug-related reduction in quality of life;
- Inconvenience of some treatment regimens with high daily pill counts resulting in reduced adherence;
- Serious drug-related toxicities associated with certain antiretroviral drugs;
- Development of treatment-associated resistance if viral suppression is suboptimal;
- Limitation of future treatment options due to premature cycling of available drugs; and
- Unknown durability of current treatment regimens.
Although delayed therapy postpones treatment-related negative effects on quality of life and the development of drug-associated toxicities, preserves treatment options for later in the course of the disease, and delays the development of drug resistance, potential risks must be considered. These risks include possible preventable—but irreversible—damage to the immune system, more difficult suppression of viral replication associated with later-stage disease, and increased risk for HIV transmission to other individuals during the longer untreated period.1
All of these factors must be considered in the clinical decision concerning whether to initiate antiretroviral therapy. Accordingly, the DHHS and International AIDS Society (IAS) recommendations for the timing of initial therapy are shown in Figures 5a and 5b. Although it is clear that patients with CD4 cell counts <200 cells/mm3 should receive treatment, the decision to initiate treatment for asymptomatic patients with higher cell counts should be based upon several criteria, including patient comprehension of the regimen and interest in participation in a complex treatment protocol.27
Summary
The DHHS guidelines for the initial treatment of HIV-infected patients continue to evolve according to emerging evidence. The current guidelines present treatment recommendations in a user-friendly format for the health care professional. It is clear that the selection of a particular antiretroviral drug regimen for the initial treatment of HIV is an important clinical decision with potential long-term therapeutic implications. Recent evidence concerning suboptimal efficacy supports the DHHS recommendation that antiretroviral triple NRTI combinations should only be used when NNRTI- or PI-based regimens cannot or should not be used; certain triple NRTI regimens are not recommended for use initially or at any time as sole therapy. In the current DHHS guidelines, the preferred NNRTI is efavirenz and the preferred PI is the combination LPV/RTV.1 Another important clinical decision in the management of the untreated patient with HIV is the timing of initiation of antiretroviral therapy in asymptomatic patients whose CD4 count is >200 cells/mm3. This decision should be individualized with consideration given to the advantages and disadvantages of early versus delayed therapy for each patient.
References
1. Department of Health and Human Services (DHHS). Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Bethesda, Maryland, March 23, 2004.
2. Palella FJ Jr, Delaney KM, Moornay AC, et al. Declining morbidity and mortality among patients with advanced Human Immunodeficiency Virus infection. N Engl J Med. 1998; 338:853860.
3. Masquelier B, Peytavin G, Leport C, et al. Mechanisms of early virologic failure in antiretroviral-naive patients starting protease-inhibitor-containing regimens: The APROVIR study. Infect Dis. 2002; 186:15031507.
4. Descamps D, Flandre P, Calvez V, et al. Mechanisms of virologic failure in previously untreated HIV-infected patients from a trial of induction-maintenance therapy. Trilège (Agence Nationale de Recherches sur le SIDA 072) study team. JAMA. 2000;283:205211.
5. Peyrière H, Guillemin V, Lotthe A, et al. Reasons for early abacavir discontinuation in HIV-infection patients. Ann Pharmacother. 2003;37:13921397.
6. Staszewski S, Keiser P, Montaner J, et al. Abacavir-lamivudine-zidovudine vs indinavir-lamivudine-zidovudine in antiretroviral-naive HIV-infected adults: A randomized equivalence trial. JAMA. 2001;285:11551163.
7. Vibhagool A, Cahn P, Schechter M, et al. Abacavir/combivir (ABC/COM) is comparable to indinavir/combivir in HIV-1 infected antiretroviral therapy naive adults: Preliminary results of a 48-week open label study (CNA 3014). 1st International AIDS Society Conference on HIV Pathogenesis and Treatment; July 811, 2001; Buenos Aires, Argentina. Abstract 63.
8. Gulick RM, Ribaudo HJ, Shikuma CM, et al. ACTG 5095: A comparative study of 3 protease inhibitor-sparing antiretroviral regimens for the initial treatment of HIV infection. 2nd IAS Conference on HIV Pathogenesis and Treatment; July 1316, 2003; Paris, France. Abstract 41.
9. NIH National Institute of Allergy and Infectious Diseases (NIAID). Notice to Physicians. Bethesda, Maryland; March 10, 2003.
10. Keiser P, Nassar N, Smillie J, et al. An observational cohort comparison of zidovudine-lamivudine-efavirenz to zidovudine-lamivudine-abacavir in anti-retroviral naïve individuals. 2nd IAS Conference on HIV and Pathogenesis; July1316, 2003; Paris, France. Poster 554.
11. Gerstoft J, Kirk O, Obel N, et al. Low efficacy and high frequency of adverse events in a randomized trial of the triple nucleoside regimen abacavir, stavudine, and didanosine. AIDS. 2003;17:20452052.
12. Gallant JE, Rodriguez A, Weinberg W, et al. Early non-response to tenofovir DF (TDF) + abacavir (ABC) and lamivudine (3TC) in a randomized trial compared to efavirenz (EFV) + ABC and 3TC: ESS30009 unplanned interim analysis (oral presentation). 43rd Interscience Conference on Antimicrobial Agents and Chemotherapy; September 1417, 2003; Chicago, Illinois. Oral Poster H-1722a.
13. Farthing C, Khanlou H, Yeh V, et al. Early virologic failure in a pilot study evaluating the efficacy of once-daily abacavir (ABC), lamivudine (3TC), and tenofovir DF (TDF) in treatment-naive HIV-infected patients. 2nd Annual AIDS Society Meeting; July 1316, 2003; Paris, France. Abstract 43.
14. Hoogewerf M, Regez RM, Schouten WEM, et al. Change to abacavir-lamivudine-tenofovir combination treatment in patients with HIV-1 who had complete virologic suppression. Lancet. 2003;362:19791980.
15. Landman R, Peytavin G, Descamps D et al. Low genetic barrier to resistance is a possible cause of early virologic failurs in once-daily regimen of abacavir, lamivudine, and tenofovir: The Tonus Study. 11th Conference on Retroviruses and Opportunistic Infections; February 811, 2004; San Francisco, CA. Session 13, Oral Abstract 52.
16. van Leeuwen R, Katlama C, Murphy RL et al. A randomized trial to study first-line combination therapy with or without a protease inhibitor in HIV-1-infected patients. AIDS. 2003;17:987999.
17. Bartlett JA, Johnson J, Herrera G, et al. Abacavir/lamivudine (ABC/3TC) in combination with efavirenz (NNRTI), amprenavir/ritonavir (PI), or stavudine (NRTI): ESS40001 (CLASS) preliminary 48-week results. XIV International AIDS Conference, July 712 2002, Barcelona, Spain. Abstract 1189.
18. Elion R, Cohen C, DeJesus E, et al. COL40263: Resistance and efficacy of once-daily Trizivir and Tenofovir DF in antiretroviral naïve subjects. 11th Conference on Retroviruses and Opportunistic Infections. Feb. 811, 2004; San Francisco, CA. Session 13, Oral Abstract 53.
19. de Truchis P, Force G, Welker Y, et al. Efficacy and safety of a quadruple combination Combivir + abacavir + efavirenz regimen in antiretroviral treatment-naive HIV-1-infected adults: La Francilienne. J AIDS. 2002;31:178182.
20. de la Rosa R, Ruiz-Mateos E, Rubio A, et al. Long-term virological outcome and resistance mutations at virological rebound in HIV-infected adults on protease inhibitor-sparing highly active antiretroviral therapy. J Antimicrob Chemother. 2004;53:95101.
21. Shafer R, Smeaton L, Robbins G, et al. Comparison of four-drug regimens and pairs of sequential three-drug regimens as initial therapy for HIV-1 infection. N Engl J Med. 2003;349(24):2304-2315.
22. Staszewski S, Morales-Ramirez J, Tashima KT, et al. Efavirenz plus zidovudine and lamivudine, efavirenz plus indinavir, and indinavir plus zidovudine and lamivudine in the treatment of HIV-1 infection in adults. N Engl J Med. 1999;341:18651873.
23. Arribas J, Staszewski S, Nelson M, et al. 3-year durability of response with an efavirenz (EFV)-containing regimen: 144-week follow-up of Study 006. Presented at: 11th European Congress of Clinical Microbiology and Infectious Diseases; April 1, 2002; Istanbul, Turkey. Abstract 043.
24. Robbins G, De Gruttola V, Shafer R, et al. Comparison of sequential three-drug regimens as initial therapy for HIV-1 infection. N Engl J Med. 2003; 349:22932303.
25. Walmsley S, Bernstein B, King M, et al. Lopinavir-ritonovir versus nelfinavir for the initial treatment of HIV infection. N Engl J Med. 2003;346:20392046.
26. Jemsek J, Hutcherson P, Harper E. Poor virologic responses and early emergence of resistance in treatment naïve, HIV-infected patients receiving a once daily triple nucleoside regimen of didanosine, lamivudine, and tenofovir DF. 11th Conference on Retroviruses and Opportunistic Infections. Feb. 811, 2004; San Francisco, CA. Session 13, Oral Abstract 51.
27. Yeni PG, Hammer SM, Carpenter CC, et al. Antiretroviral treatment for adult HIV infection in 2002: Updated recommendations of the International AIDS Society-USA Panel. JAMA. 2002;288(2):222-235.
28. Manion DJ. Early virologic non-response in patients with HIV infection treated with lamivudine, abacavir, and tenofovir. GlaxoSmithKline Drug Warning, July 2003.
CME Questions
1. Which of the following statements is false about earlier approaches to HIV therapy?
A. Didanosine was the first
antiretroviral approved.
B. Monotherapy and dual therapy were not potent enough to suppress viral replication.
C. The addition of an NNRTI to a failing regimen did not provide sufficient
antiviral activity.
D. All of the above statements are true.
2. According to the 2004 DHHS guidelines for the treatment of HIV, the goals of antiretroviral therapy are:
A. maximal and long-lasting
suppression of viral replication.
B. maintenance of restoration of immunologic function.
C. improved quality of life.
D. reduction of HIV-related morbidity and mortality.
E. All of the above.
3. Concerning the ACTG 5095 trial, all of the following are true except:
A. All arms of the trial
are currently ongoing with results forthcoming.
B. Patients were randomized to receive ABC/3TC/ZDV, ABC/3TC/ZDV + EFV, or ZDV/3TC/EFV.
C. Virologic failure occurred significantly more often in patients in the ABC/3TC/ZDV
arm compared with the pooled EFV arms.
D. Virologic failure occurred earlier in the ABC/3TC/ZDV group compared with
the EFV-containing treatment groups.
4. In the open-label trial in which the triple NRTI regimen d4T/ddI/ABC was compared with RTV/SQV or NFV/NVP:
A. It was determined that
the triple NRTI regimen is particularly effective for patients with higher baseline
VL.
B. The primary endpoint was plasma RNA <50 copies mL after 48 weeks.
C. Side effects were more frequent in the triple NRTI arm.
D. The K65R mutation was not found in any patients.
5. Which of the following statements is false?
A. ESS30009 compared the
virologic suppression associated with ABC/3TC/ZDV versus EFV/ABC/3TC.
B. A pilot study by Farthing and colleagues reported early virologic failure
in treatment-naïve HIV-infected patients who received TDF/ABC/3TC.
C. A possible explanation for the high rate of virologic failure with TDF/3TC/ABC
is a low genetic barrier to resistance .
D. Both B and C are false.
6. The Atlantic Study was a randomized, open-label trial that compared the relative efficacy of 1 convergent and 2 divergent regimens.
A. True
B. False
7. Which of the following statements is true?
A. In the CLASS study,
a high baseline VL was predictive of a better virologic response in the triple
NRTI arm.
B. A pilot study with ABC/3TC/ZDV +TDF resulted in a VL <50 copies/mL in
67% of patients by week 24.
C. A pilot study with ABC/3TC/ZDV + EFV resulted in a VL <50 copies/mL in
58% of patients at week 8.
D. Both B and C are correct.
8. According to the DHHS guidelines, which of the following is not a preferred antiretroviral regimen for the initial treatment of HIV?
A. EFV + 3TC + ZDV
B. LPV/r + 3TC + ZDV
C. ABC + TDF + 3TC
D. LPV/r + 3TC + d4T
9. Regimens with a PI plus 2 different NRTIs for the initial treatment of HIV possess the longest clinical experience and most available clinical data.
A. True
B. False
10. The advantages of initiating antiretroviral therapy early in HIV-infected patients include:
A. earlier suppression
of viral replication.
B. preservation of immune function.
C. reduced risk of developing treatment resistance with partial viral suppression.
D. increased risk of viral transmission.
E. Both A and B are correct.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
