Drug Criteria & Outcomes: Agents used in the treatment of Alzheimer’s disease — Cholinesterase inhibitors: A therapeutic class review
Drug Criteria & Outcomes
Agents used in the treatment of Alzheimer’s disease — Cholinesterase inhibitors: A therapeutic class review
By Tamara Herring, PharmD, and Heather Nugent, PharmD
Scott and White Hospital and Health Plan,
Temple, TX
Introduction
Alzheimer’s disease (AD) is a progressive, neurodegenerative disease that affects both cognition and behavior.1,2 Approximately four million Americans are affected by this devastating illness, and an average of 100,000 AD patients die each year.1,2 AD costs the United States at least $100 billion per year.3
The disease first was described in 1907; however, the pathogenesis is still not completely understood.1,2 AD is thought to involve several neurotransmitter systems, the most common being the cholinergic system.1 Degeneration of cholinergic neurons is one of the anatomical hallmarks of the Alzheimer’s brain.2 Others include neurofibrillary tangles and neuritic plaques, which are abnormal collections of protein in the brain that lead to neuronal cell death.
Currently, no cure for AD exists, so it is important that new treatments be evaluated for cost-effectiveness. At this time, cholinesterase inhibitors (ChEIs) are the only class of drugs approved for the symptomatic treatment of Alzheimer’s disease.1 Tacrine (Cognex) was the first agent approved by the Food and Drug Administration (FDA) for AD in 1993.4 This review will cover the newer approved agents: donepezil (Aricept, by Eisai), rivastigmine (Exelon, by Novartis), and galantamine (Reminyl, by Janssen).
Clinical pharmacology
The human brain possesses two forms of cholinesterase: butyrlcholinesterase (BuChE) and acetylcholinesterase (AChE). Both forms hydrolyze acetylcholine (ACh). In the normal brain, AChE represents 99% of cholinesterase. However, in the Alzheimer’s brain, AChE represents only 65%, with BuChE representing the other 35%. BuChE concentrations may be higher in areas of neuritic plaques and may contribute to their formation.5
Donepezil, rivastigmine, and galantamine are centrally acting, reversible cholinesterase inhibitors. All of the agents basically work by the same mechanism, with a few slight differences. Donepezil and galantamine inhibit only AChE, whereas rivastigmine inhibits both AChE and BuChE. This difference gives rivastigmine the advantage of protecting more ACh from hydrolysis. In addition, rivastigmine is specific for a certain subtype of AChE (G1). This specificity does not affect the drug’s efficacy but may improve its adverse effect profile.6
By inhibiting the enzyme(s) that breaks down acetylcholine, these drugs allow ACh to remain in synapses of cholinergic neurons longer. Preserving cholinergic neuronal function is thought to improve the symptoms of AD. However, there is no evidence that these drugs affect the underlying disease process. As the disease progresses and more cholinergic neurons degenerate, these drugs likely will lose their effectiveness. The improvement seen with these agents can be significant, but usually is modest at best. Caregivers should be encouraged to recognize that "no change" is a positive sign.2,4,6
Indications
Donepezil, rivastigmine, and galantamine are indicated for treatment of mild to moderate dementia of the Alzheimer’s type.7-10
Pharmacokinetics
Table 1, below, shows the various pharmacokinetic parameters of the ChEIs.7-10
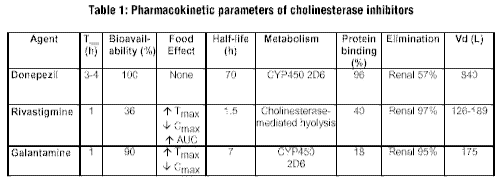 |
Hepatic disease. A study conducted in 10 patients with stable alcoholic cirrhosis demonstrated that the clearance of donepezil was decreased by 20% compared to healthy controls. Rivastigmine’s clearance was decreased by 60-65% in 10 patients with mild-to-moderate liver dysfunction compared to healthy controls. The clearance of galantamine was decreased by approximately 25% in eight patients with moderate hepatic impairment compared to normal volunteers, but was similar to normal volunteers in eight patients with only mild impairment.
Renal disease. In eight patients with moderately impaired renal function, the clearance of rivastigmine was 64% lower compared to normal controls, but was 43% higher in eight patients with severe renal impairment. A reason for this could not be determined. In a study with galantamine, area under the curve (AUC) was increased by 37% and 67%, respectively, in patients with moderate and severe renal impairment, compared to normal volunteers.
Age/gender/race. No formal pharmacokinetic studies have examined the effects of age, gender, and race on donepezil’s clearance. However, data from clinical trials and retrospective analysis suggest that these factors have no influence. Mean oral clearance of rivastigmine was 30% lower in elderly patients, but gender and race did not affect the clearance. In trials with galantamine, concentrations were 30-40% higher in elderly patients; clearance was about 20% lower in females; and race did not affect the clearance.
Smokers. A population pharmacokinetic analysis showed that the oral clearance of rivastigmine is increased by 23% with concurrent nicotine use.
Efficacy 1,11-19
With a large proportion of the U.S. population aging, the prevalence of AD is expected to increase dramatically unless a cure is found. The ChEIs have been proved efficacious compared to placebo. Unfortunately, there is a paucity of head-to-head comparative trials among the various agents. The following will review some of the pivotal trials leading to the approval of each of the three agents.
Donepezil. Rogers et al conducted a 15-week (12 weeks active followed by three weeks placebo washout) randomized, double-blind, placebo-controlled (RDBPC), multicenter trial in 468 patients with mild to moderately severe AD. The study’s objective was to evaluate the efficacy and safety of two doses of donepezil compared to placebo.
Inclusion in this study required a diagnosis of probable AD, a Mini-Mental State Examination (MMSE) score of 10-26, and no complicating comorbidities such as delirium, delusions, depression, or a history of substance abuse. Patients with other uncontrolled medical disorders were excluded. (Diagnosis of probable AD in all of the trials discussed here was determined by criteria of the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association and/or Diagnostic and Statistical Manual [DSM] criteria.)
After randomization, patients received placebo, donepezil 5 mg, or donepezil 10 mg. Approximately 90% of patients were on concomitant medications; however, medications that could affect CNS functioning were not allowed. Primary outcome measures of this trial were the ADAS-cog and the CIBIC-plus. Safety assessments included vital signs, EKG, routine labs, and adverse event monitoring. The results from the intent-to-treat (ITT) analysis are presented in Table 2.
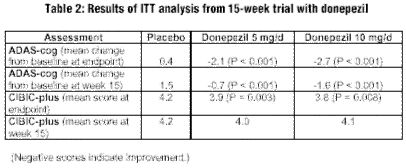 |
Another trial conducted by Rogers et al looked at 473 patients with mild to moderately severe AD to evaluate the efficacy and safety of two doses of donepezil vs. placebo. This RDBPC multicenter study consisted of 24 weeks of active treatment followed by a six-week placebo washout period. Methods of this study (inclusion/exclusion criteria, outcome measures, etc.) were similar to the 15-week study discussed previously. The results of the ITT analysis are presented in Table 3.
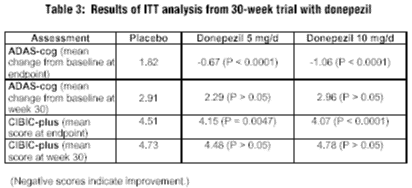 |
Rivastigmine. Rosler and colleagues conducted a 26-week, RDBPC multicenter study of 725 patients with mild to moderately severe AD. The goal of the study was to assess the efficacy and safety of two doses of rivastigmine vs. placebo. Participants had to have a diagnosis of probable AD and an MMSE score of 10-26.
Patients were randomized to placebo, rivastigmine 1-4 mg/day, or rivastigmine 6-12 mg/day. Doses were increased weekly by 1.5 mg/day during weeks 1-12. During this time, no dose decreases were allowed. During weeks 13-26, doses could be increased or decreased within the allotted range in order to achieve the highest tolerable dose.
Primary outcome measures were the ADAS-cog, the CIBIC-plus, and the PDS. Safety measures included physical exams, EKGs, vital signs, routine labs, and adverse event monitoring. Table 4 depicts results from the last observation carried forward (LOCF) analysis at week 26.
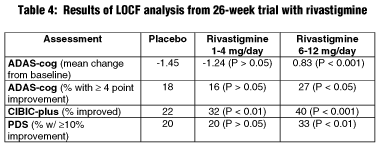 |
Corey-Bloom et al conducted a 26-week RDBPC multicenter study of 699 patients with mild to moderately severe AD to evaluate the efficacy and safety of lower-dose rivastigmine (1-4 mg/day) and higher-dose rivastigmine (6-12 mg/day) vs. placebo.
Patients had to have a diagnosis of probable AD, an MMSE score of 10-26, and a CT or MRI consistent with AD within the last 12 months before inclusion. Primary efficacy measures were the ADAS-cog, CIBIC-plus, and the PDS. Safety measures were the same as in the above study.
Study participants received rivastigmine 1-4 mg/day, rivastigmine 6-12 mg/day, or placebo. A fixed-dose weekly titration schedule was used through week 7, with flexible dosing allowed in weeks 8-26, provided the doses stayed in the allotted range. Table 5 presents results from the ITT analysis performed at 26 weeks. All data represent the mean change from baseline with 95% confidence intervals.
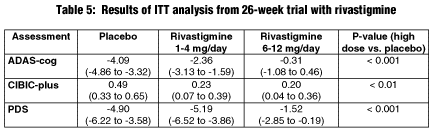 |
Galantamine. Raskind et al conducted a six-month RDBPC multicenter trial of 636 patients with mild-to-moderate AD. The objective of the study was to evaluate the efficacy and tolerability of galantamine 24 mg/day and galantamine 32 mg/day vs. placebo using a weekly titration schedule. Inclusion criteria included a six-month history of progressive dementia, a diagnosis of probable AD, an MMSE score of 11-24, and an ADAS-cog score of > 12.
After a four-week placebo run-in, patients were randomized to placebo, galantamine 24 mg/day, or galantamine 32 mg/day in a 1:1:1 ratio. Primary efficacy measures included the ADAS-cog and the CIBIC-plus. Safety measures included physical exams, EKGs, vital signs, routine labs, and adverse event monitoring. Ninety-one percent of patients had comorbid conditions, and 69% completed the study. Of the 350 patients who entered the six-month extension, 76% completed the study. An observed case analysis (OC) and an ITT analysis were performed. Results from the ITT analysis after six months are presented in Table 6.
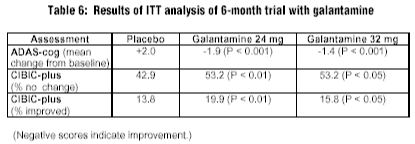 |
All patients in the open-label extension received galantamine 24 mg/day. Specific data were not given; however, the patients who received galantamine 24 mg/day for the entire 12 months maintained ADAS-cog scores above baseline. Patients who received placebo the first six months improved but not to the degree of patients who continually had received galantamine 24 mg/day. Patients originally in the galantamine 32 mg/day group had ADAS-cog scores that fell below baseline but were still above those patients originally in the placebo group after 12 months.
Tariot et al conducted a five-month RDBPC multicenter trial of 978 patients with mild-to-moderate AD to evaluate the efficacy and tolerability of galantamine 16 mg/day and galantamine 24 mg/day vs. placebo. Inclusion criteria included a six-month history of progressive dementia with a diagnosis of probable AD, MMSE score 10-22, and an ADAS-cog score of > 18. Primary efficacy and safety measures were the same as in the above trial.
After a four-week placebo run-in, patients were randomized to placebo, galantamine 8 mg/day, galantamine 16 mg/day, or galantamine 24 mg/day in a 2:1:2:2 ratio. The 8 mg/day group was used to test for a dose-response effect and was not powered to detect efficacy. A four-week titration schedule was used. Ninety-seven percent of patients had comorbid conditions, and 96-98% were taking concomitant medications. Eighty percent of the patients completed the study. Both OC and ITT analyses were performed. The results of the ITT analysis after five months are presented in Table 7.
 |
Conclusions. Each agent is significantly more effective than placebo for at least one dosage range. Higher doses seem to provide more benefit. The drugs’ beneficial effects are lost if the drugs are discontinued. Higher doses have a higher incidence of adverse effects.
Adverse reactions 6-10
The adverse reaction profile of each of these agents is similar. The major adverse reactions seen with ChEIs are cholinergic excess and gastrointestinal irritation (nausea, vomiting, anorexia, and diarrhea). The GI effects of these drugs can be minimized by administration with food and slow titration of doses. The effects are usually mild to moderate, transient, and most prevalent during dose escalation periods. Table 8 summarizes the adverse events observed in clinical trials.
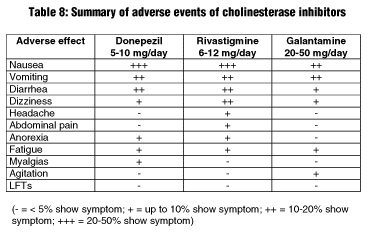 |
Contraindications 7-10
Table 9 details the contraindications for these three agents.
 |
Warnings (the following apply to all three agents) 7-10
Anesthesia. ChEIs are likely to exaggerate succinylcholine-type muscle relaxation during anesthesia.
Cardiovascular. Due to their cholinomimetic actions, ChEIs may have vagotonic effects on heart rate (i.e., bradycardia). This may be important in patients with conduction abnormalities, brady-arrhythmias, or sick-sinus syndrome and in those taking other bradycardic drugs. Syncopal episodes were reported with donepezil, rivastigmine, and galantamine in trials; however, the occurrence was rare.
Gastrointestinal. ChEIs, as predicted by their cholinomimetic actions, can be expected to increase gastric acid secretion, thus putting patients at increased risk for development of ulcers. Patients who have a history of peptic ulcer disease and/or use NSAIDs should be monitored closely for signs of active or occult GI disease. The ChEIs also have been shown to cause nausea, vomiting, anorexia, and diarrhea at therapeutic doses.
Genitourinary. Although not observed in any of the clinical trials involving the ChEIs, these agents may cause bladder outflow obstruction.
Neurologic. Cholinomimetics are believed to have some potential seizure activity. However, seizure activity also may be a manifestation of AD.
Pulmonary. Because of their cholinomimetic actions, these agents should be prescribed with caution in patients with pre-existing asthma or obstructive pulmonary disease.
Precautions 7-10
Pregnancy. Donepezil has a Pregnancy Category C classification. Rivastigmine and galantamine are Pregnancy Category B.
Pediatric use. No studies have been done with these agents in any pediatric diseases. Therefore, their use in pediatric patients is not recommended.
Drug interactions 7-10,20
Digoxin/warfarin. None of these agents were found to have significant effects on the pharmacokinetics of digoxin or warfarin.
Use with anticholinergics. All of these agents have the ability to interfere with the activity of anticholinergic medications.
Use with cholinomimetics and other cholinesterase inhibitors. These agents are expected to act synergistically when used concomitantly with succinylcholine, other ChEIs, similar neuromuscular blockers, or cholinergic agonists.
Donepezil. In vitro drug displacement studies were performed for donepezil and other drugs such as furosemide, digoxin, and warfarin. Donepezil had no effect on the binding of these drugs and vice versa.
CYP450. In vitro studies indicate that donepezil shows a low rate of binding to isoenzymes 2D6 and 3A4. Therefore, little likelihood exists that donepezil affects the clearance of drugs metabolized by the two isoenzymes. It is not known if donepezil has any enzyme-induction potential. Donepezil was found to have no significant effects on the pharmacokinetics of theophylline and cimetidine. Inducers and inhibitors of CYP2D6 and CYP3A4 may affect donepezil’s metabolism, but there is no evidence to prove a clinical significance.
Rivastigmine. Because rivastigmine is metabolized primarily through esterase hydrolysis, no significant interactions with any drugs metabolized through the CYP450 system are expected. No pharmacokinetic interactions were observed between rivastigmine and diazepam or fluoxetine. A population pharmacokinetic analysis of 625 patients showed that rivastigmine has no significant effect on the kinetics of several commonly prescribed classes of drugs (antacids, antihypertensives, antidiabetics, NSAIDs, estrogens, salicylate analgesics, antianginals, and antihistamines).
Cimetidine. An in vivo study found that cimetidine increased the bioavailability of galantamine by approximately 16%.
Ketoconazole. Ketoconazole, a strong CYP450 3A4 inhibitor, increased the AUC of galantamine by 30%.
Erythromycin. Erythromycin is a moderate inhibitor of CYP3A4 and had a minimal (10%) effect on the AUC of galantamine.
Paroxetine. Paroxetine is a strong inhibitor of CYP450 2D6. Coadministration increased the bioavailability of galantamine by about 40%.
Galantamine is thought to have minimal potential as an enzyme (CYP450) inhibitor.
Dosage and administration 7-10
Donepezil. The initial dose of donepezil is 5 mg once daily. After 4-6 weeks on this dose, patients may be escalated to 10 mg once daily. Donepezil should be taken just before bedtime and may be taken without regard to food.
Rivastigmine. The recommended starting dose of rivastigmine is 1.5 mg twice daily. If well-tolerated, the dose may be increased to 3 mg twice daily after a minimum of two weeks. The titration period may be extended to four weeks to lessen the incidence of adverse reactions. Following the 3 mg twice-daily dose, the dose may be increased to 4.5 mg and then 6 mg twice daily. The maximum recommended dose is 6 mg twice daily. If side effects become problematic, patients should discontinue treatment for several doses and then restart at the same or next lower dose. Dosing adjustment is not necessary in renal or hepatic impairment as the dose is titrated to individual tolerability. Rivastigmine should be taken with food.
Galantamine. Galantamine should be started at 4 mg twice daily. After a minimum of four weeks, the dose may be increased to 8 mg twice daily if it is well-tolerated. An additional increase to 12 mg twice daily may be attempted after another four weeks. The maximum recommended dose is 24 mg/day. If therapy is interrupted for several days or more, patients should be started on the lowest dose and retitrated up to the previous dose. Galantamine should be given with food. In patients with moderate renal or hepatic impairment, the daily dose of galantamine should not exceed 16 mg. Galantamine is not recommended for use in patients with severe renal or hepatic impairment.
How supplied 7-10
Donepezil is supplied as 5 and 10 mg film-coated round tablets. Rivastigmine is supplied as 1.5, 3, 4.5, and 6 mg capsules. Galantamine is supplied as 4, 8, and 12 mg tablets.
Conclusion 3-5,7-10
Donepezil, rivastigmine, and galantamine are approved for the symptomatic treatment of AD. Unfortunately, there is a lack of data to suggest that one of the agents is superior; thus, agent selection is difficult. Numerous pharmacoeconomic studies have proved that treating AD with one of these agents is indeed cost-effective. Nursing home care costs an average of $42,000-$70,000 per patient per year. Clinical trials prove that each of the approved ChEIs is significantly better than placebo in slowing disease progression. However, if patients do not respond to one agent, it is unlikely they will respond to another.
Factors to consider when choosing any drug include ease of dosing, drug interactions, adverse reactions, comparative efficacy, and cost. Each of the agents examined here has a similar adverse effect profile and comparable cost. Costs are listed in Table 10. Little or no data are available regarding comparative efficacy. The distinguishing factors for these agents include dosing flexibility (i.e., donepezil is available in two strengths, rivastigmine is available in four strengths), interactions with other drugs, and pharmacology (AChE inhibition vs. BuChE inhibition).
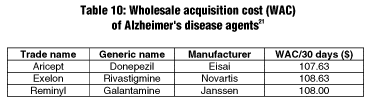 |
References
1. Exelon Scientific Product Monograph. East Hanover, NJ: Novartis Pharmaceuticals; April 2000.
2. Dipiro JT, ed. Pharmacotherapy: A Pathophysiologic Approach. 4th ed. Stamford, CT: Appleton & Lange; 1999.
3. Alzheimer’s Association. Available at: http://www.alz.org/hc/overview/stats.htm. Accessed: April 17, 2001.
4. Grutzendler J, Morris JC. Cholinesterase inhibitors for Alzheimer’s disease. Drugs 2001; 61:41-52.
5. Exelon Product Monograph: Overview of Efficacy. East Hanover, NJ: Novartis Pharmaceuticals; April 2000.
6. Weinstock M. Selectivity of cholinesterase inhibition: Clinical implications for the treatment of Alzheimer’s disease. CNS Drugs 1999; 12(4):307-323.
7. "Cognex Product Information." Physician’s Desk Reference, 55th edition. Montvale, NJ: Medical Economics Co.; 2001, pp. 1242-1246.
8. "Aricept Product Information." Physician’s Desk Reference, 55th edition. Montvale, NJ: Medical Economics Co.; 2001, pp. 1181-1183.
9. Exelon product information. East Hanover, NJ: Novartis Pharmaceuticals; April 2000.
10. Reminyl product information. Titusville, NJ: Janssen Pharmaceuticals; March 2001.
11. Sajatovic M, Ramirez L. Rating Scales in Mental Health. Hudson, OH: Lexi-Comp Inc., 2001.
12. Farlow M, et al. A controlled trial of tacrine in Alzheimer’s disease. JAMA 1992; 268:2523-2529.
13. Knapp MJ, et al. A 30-week randomized controlled trial of high-dose tacrine in patients with Alzheimer’s disease. JAMA 1994; 271:985-991.
14. Rogers SL, et al. Donepezil improves cognition and global function in Alzheimer disease: A 15-week, double-blind, placebo-controlled study. Arch Intern Med 1998; 158:1021-1031.
15. Rogers SL, et al. A 24-week, double-blind, placebo-controlled trial of donepezil in patients with Alzheimer’s disease. Neurology 1998; 50:136-145.
16. Rosler M, et al. Efficacy and safety of rivastigmine in patients with Alzheimer’s disease: International randomised controlled trial. BMJ 1999; 318:633-640.
17. Corey-Bloom J, et al. A randomized trial evaluating the efficacy and safety of ENA 713 (rivastigmine tartrate), a new acetylcholinesterase inhibitor, in patients with mild to moderately severe Alzheimer’s disease. Int J Geriatr Psychopharmacol 1998; 1:55-65.
18. Raskind MA, et al. Galantamine in AD: A 6-month randomized, placebo-controlled trial with a 6-month extension. Neurology 2000; 54:2261-2268.
19. Tariot PN. A 5-month, randomized, placebo- controlled trial of galantamine in AD. Neurology 2000; 54:2269-2276.
20. Hutchison TA, et al, eds. "Tacrine Drug Evaluation." DRUGDEX System. Greenwood Village, CO: MICROMEDEX Inc. Vol. 108 (expires June 2001).
21. Scott and White Health Plan data, April 2001.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
