An Overview of Herbal Medicine for the Primary Care Provider: An Evidence-based Approach
An Overview of Herbal Medicine for the Primary Care Provider: An Evidence-based Approach
Author: Felise Milan, MD, Associate Professor of Clinical Medicine, Albert Einstein College of Medicine, Bronx, NY; Clinical Practice, Montefiore Medical Center, Bronx, NY.
Editor’s Note—Herbs can be defined in several different ways. In the botanical nomenclature, herbs are nonwoody seed-producing plants that die back at the end of the growing season. In the culinary arts, herbs are vegetable products used to add flavor or aroma to food. Herbal medicine (also called botanical medicine, phytomedicine) is the use of plant materials (medicinal herbs, medicinal botanicals, etc) to treat disease states or to improve health. There is physical evidence that the use of herbal medicine dates back to Neanderthal man.1 All cultures have long folk medicine histories that include the use of plants. One-quarter of our modern pharmaceuticals are derived from plant materials, and the use of many of those originated from the herbal lore of native peoples.1 Herbal medicines are considered by many to be dilute drugs with multiple active chemicals in any given plant. The active component(s) are actually not known for most herbal remedies, and therapeutic effects are often known to differ within different parts of the same plant (ie, stem, root, leaf, and berry).
From the ancient practices of Native Americans through the 1940s, most families and physicians in the United States used herbal remedies. Before the advent of modern medicines, textbooks of pharmacognosy (the study of the use of plants for medicinal purposes) listed hundreds of commonly used plant materials, and the studies were taught in medical schools. After the advances brought by 20th century technology, herbal medicine traditions were all but lost in this country. In the 1970s, a slow but steady increase in the use of more "natural" or "organic" therapeutic agents began. With the explosion of interest in many areas of complementary and alternative medicine (CAM) in the early 1990s, herbal medicine saw enormous growth in popularity and use.2 According to several surveys on use of CAM in the United States, herbal medicines are used by 12-20% of the general population, and the majority of that use is without the input of either a conventional or alternative health care provider.3-6 The herbal medicine market has seen unprecedented growth since the early 1990s with billions of dollars spent on herbal supplements each year by US consumers. That enormous growth coincided with a significant change in the regulations covering the sale and marketing of herbal remedies. In 1994, Congress passed the Dietary and Supplements Health and Education Act of 1994 (DSHEA), which severely restricted the FDA’s authority over these products. Under this legislation, no standardization of manufacturing or premarket safety evaluation of herbs is required. Several studies on the inconsistent quality of herbal products and reports of herb-drug interactions have heightened concerns about the safety of use of these products as medicines.7 However, the amount of quality research on herbal medicines has paralled its use by the public. There is a rapidly growing amount of literature suggesting efficacy for a number of herbal products in treating a variety of health problems.
With the acknowledgement that many of our patients are using these herbal remedies comes the responsibility of finding out what they are taking and the need for awareness of potential toxic effects or herb-drug interactions. This paper will describe the current regulations governing the manufacturing and marketing of herbal medicines and comment on how these issues may affect the safety of their use. It will also describe the different types of herbal preparations (extracts, tinctures, teas, etc), discuss the evidence for safety and efficacy of some of the more commonly used herbs, and highlight several clinical situations that carry significant risks of toxicity and/or adverse reactions. Finally, I will make some recommendations on discussing the subject with patients and list some resources for reliable information on herbal medicine.
Current Use of Herbal Medicine in the United States
Herbal medicine has consistently been found to be one of the most popular, if not the most popular, of the alternative therapies used in this country.3-6 The national surveys done by Eisenberg found that the use of herbal remedies increased 380% during the earlier part of the last decade (2.5% in 1990 to 12% in 1997). In 1994, the estimated market for herbal medicines was $1.6 billion annual retail sales, and by 1998 that figure was up to $4 billion.2 For the years 1999-2000, growth of total retail sales for herbal supplements slowed considerably, as did the rate of growth for dietary supplements overall (see Figure 1).8 This fall has been attributed to the negative media coverage about herbs in the past 2 years.9 Rates of use are highest among those with high levels of education and poor-perceived health status.6 Fewer surveys have been done among low income and non-English speaking populations. The surveys that have been done have found enormous variability between ethnic groups in their use of herbal medicine with an overall greater use of folk or traditional therapies.6
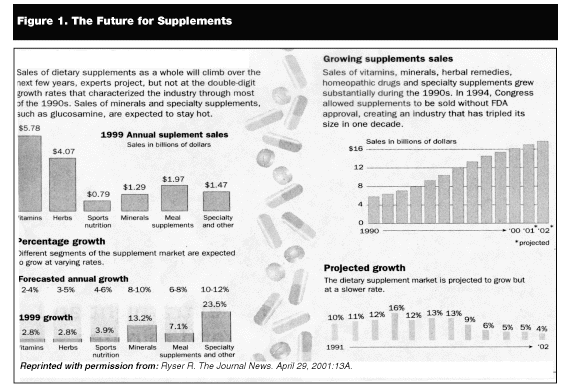
|
Common reasons for using herbs include: colds, burns, headaches, allergies, insomnia, PMS, depression, and menopause.10 The most commonly used herbs for 1998 and 1999 were Ginkgo biloba followed closely by St. John’s wort, ginseng, garlic, Echinacea/golden seal, saw palmetto, kava kava, cranberry, evening primrose oil, valerian root, and milk thistle. Since 1998, the greatest gains have been seen for milk thistle (used for chronic hepatitis) and saw palmetto (used for benign prostatic hypertrophy [BPH] and the greatest losses for ginkgo and St. John’s wort.11
Regulation of the Herbal Industry
In 1993, the FDA proposed tougher regulations governing the safety of vitamins, minerals, herbs, and other nutritional supplements. The supplement industry responded by trying to convince the public that these regulations would essentially eliminate the availability of these products from the market. In response to the flood of letters from consumers against these stricter regulations,12 Congress passed the DSHEA, which severely restricted the FDA’s authority over these products. This federal regulation created a new category of therapeutic, nonprescription substances called "dietary supplements" (herbs, vitamins, minerals, amino acids, or other nutritional supplements), which would not be subject to premarket safety evaluations but could also not make claims to prevent or treat disease. Manufacturers can make claims about the supplement’s effects on the "structure and function" of the body or on the consumer’s "well-being."13 All these products must carry the disclaimer: "These statements have not been evaluated by the FDA. This product is not intended to diagnose, treat, cure, or prevent any disease." These claims may not list characteristic signs and symptoms of a disease or mention the name of a disease or drug anywhere on the label of the product. These products may be touted as "improves immune function," "promotes joint health," and "maintains prostate health," but may not claim to "help fight infection," "relieve the pain of swollen joints," or "help maintain normal urine flow in older men." They may claim to affect common conditions associated with "natural states" such as postmenopausal hot flashes, premenstrual moodiness, and occasional constipation.
Prior to DSHEA, these supplements were regulated as foods and could not be placed on the market without approval by the FDA. Now the manufacturers must notify the FDA within 30 days after the product is released to the public. The FDA may warn the public if it feels a product may be hazardous but can only remove the product from the market if it proves it unsafe in a court of law or if the director of the Department of Health and Human Services (DHHS) determines the supplement to be hazardous in an administrative hearing.
The herbal supplement Ma Huang or ephedra provides a useful illustration of the consequences of the current regulation. Supplements containing ephedra alkaloids are consumed widely both alone and in combination with caffeine and guarana (a plant-based stimulant) for weight loss, athletic performance, and increased energy. They have been sold legally as "herbal phen-fen" or illegally as "herbal ecstasy."14 From 1994 to 1997, the FDA received more than 800 reports of serious adverse events14 associated with the use of a product containing ephedra. Proposals made by the FDA to restrict the amount of ephedra alkaloids per recommended serving and to increase the warnings on the labels of these products were firmly resisted by the herbal industry, which questioned the reliability of these adverse event reports. After sponsoring a 2-day hearing on the safety of ephedra products, the Office of Women’s Health (OWH) at the NIH recently filed a petition with the DHHS requesting that this issue be readdressed.15 In addition, in an independent review of 140 serious adverse events reported to the FDA from June 1997 to March 1999 recently published in the New England Journal of Medicine, it was concluded "that dietary supplements that contain ephedra alkaloids pose a serious health risk to some users."16
Standardization and Quality Control
Ephedra not only illustrates the imbalance of power between the supplement industry and the FDA to determine regulation of herbal products, but it also illustrates the problems encountered from lack of strict standardization or quality controls. Prior to DSHEA, the production of herbs and other supplements was governed by the good manufacturing practices (GMPs) for foods. GMPs specifically for dietary supplements have not yet been agreed upon. Standardization and quality control are challenging issues as the potency and constituents of any herbal product can be affected by a multitude of factors including: genetic variation; growing and harvesting conditions; storage and handling; processing and preparation; and which part of the plant is used (root vs leaf vs whole plant, etc).17 Chemical constituents can also vary between similar species but may be marketed without the consumers’ awareness of those differences. For example, Echinacea purpurea herb, E pallida root extract, and E augustifolia whole plant juice are all often sold simply as "Echinacea."
Several studies commissioned by consumer groups have demonstrated that herbal products sold in the United States vary widely in the concentrations of their active ingredients and that those variations are not reflected on the product label.7 Consumer Reports has published studies done on ginseng12,18 revealing variations of relevant constituents from 5% to 140% of the labeled amounts and different brands of the same herb containing up to 10 times as much as another. Several studies published in the scientific literature have found the same to be true for ephedra products.7,19 In one study, following the label directions on a series of 9 ephedra products would have given patients anywhere between 5 mg and 89 mg of ephedrine alkaloids daily.7 The FDA recommends no more than 8 mg per dose and 24 mg per day.14
Along with variations in active constituents, there is well-documented evidence of adulteration in herbal products made in Asia.20 In a study conducted by the California Department of Health, 32% of the 260 products tested contained undeclared pharmaceuticals or heavy metals.20
All of these issues leave many clinicians with concerns about the reliability and safety of herbal products. Even if there are good efficacy data to support the use of some products, do these findings apply to other preparations of the same herb? So what are the solutions to the current lack of standardization and reliability of the herbal market?
Potential Solutions for Improved Herbal Quality
There is a technology that many manufacturers are already using to identify and standardize their herbal products. High-performance liquid chromatography (HPLC) can produce a "fingerprint" chromatogram that identifies the key components in an herbal product and their relative proportions (see Figure 2). The use of HPLC is not currently required but does appear on the packaging of some products and can probably be interpreted as a marker of quality. Some experts feel that it is also safe to assume that herbal products manufactured by one of the large well-established pharmaceutical companies may also be of reasonable quality.
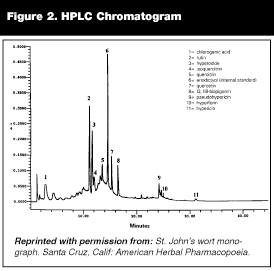
|
Regulatory changes will also hopefully improve the situation. While GMPs for dietary supplements have not yet been approved, there are reports that the FDA is working with the herbal industry to establish GMPs, which can then be enforced. The FDA has recently drafted proposals21 that would allow the submission of foreign marketing data for a botanical drug substance not previously marketed in the United States to demonstrate the products’ safety and effectiveness for a specific use and qualify for classification as an over-the-counter (OTC) drug. The most significant of the proposed changes would greatly increase the financial incentive to seek OTC status for new herbal products. Under the drafted proposal, a company that develops and gains approval for an herbal medicine can market that product exclusively for 5 years even if they do not have a patent.
These new regulations may bring the United States closer to the European herbal markets, which have more lenient approval for selling herbs as medicines but are more stringent and organized in regulating the standardization of these products. For example, in Germany herbal medicine is extremely popular and is closely regulated by Commission E in its Ministry of Health. Practitioners routinely prescribe herbal medicines that have been approved by Commission E and are paid for by their national insurance plan. Commission E produces a monograph on each herbal product after evaluating it for absolute proof of safety and "reasonable" proof of efficacy. Asian countries with long traditions of herbal medicine (ie, China, Japan, Korea) require little proof of safety and efficacy, relying instead on the principle of "traditional use." In other words, herbs that have been widely used for many centuries are presumed safe for consumption.
Herbal Preparations
The composition and, thereby, the clinical activity of any herbal medicine is determined not only by which part(s) of the herbs are used but how they are prepared (see Figure 3). The raw plant may be crushed and its juice extracted or, more commonly, it is dried. This dried herb may then be either ground into a powder to be made into capsules or tablets or immersed in a solvent (alcohol, hexane, etc) that will extract certain constituents. As the active chemical(s) are not known for most herbal remedies, many of these methods of preparation have been passed down through generations of herbalist tradition. There are clear scientific rationales for the extraction processes of several standardized extracts on the market (ie, Egb761 Ginkgo biloba extract, saw palmetto berry extract-SPBE) for which the active constituents have been identified.
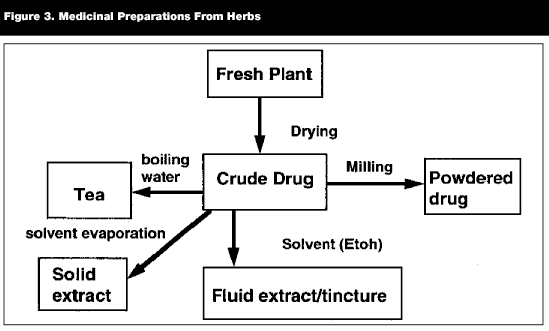
|
Commonly Used Herbs
Saw Palmetto
Plant: Serenoa repens—American Dwarf Palm Tree—indigenous to the southeastern United States.
History and Clinical Use: The Native Americans used saw palmetto for centuries for urogenital problems. It is currently the most popular therapy in Europe for BPH.
Preparations: A lipid-soluble extract made from the berry (SPBE) is usually standardized to 85-95% fatty acids and sterols. The most widely studied preparation is permixon, which is a hexane extract standardized to 90% free fatty acids with a small amount of sterols, flavonoids, and other compounds.
Active Ingredients: Beta-sitosterol, esterified sterols.22
Mode of Action: While the exact mechanism is largely unknown, there are several popular theories. The most widely accepted theory is that SPBE works like finasteride by inhibiting 5-alpha reductase. Other proposed mechanisms include: inhibition of dihydrotestosterone, antiestrogen effects, and antiandrogen effects.22
Efficacy: As herbal medicines go, saw palmetto is one of the best studied. There have been multiple studies and 2 meta-analyses both in Europe and in the United States that have consistently shown decreases in urinary symptom scores, increased peak urine flow rates with saw palmetto as compared to placebo, and effects comparable to finasteride with significantly less impotence.23-25 Several studies have shown, however, that even though SPBE may improve symptoms, it does not reduce prostate size.24 One recent randomized, placebo-controlled trial of a combination of saw palmetto and nettle root followed men for 6 months and evaluated them with ultrasound-guided biopsy. They found that while prostate volume remained unchanged, there was contraction of the epithelium and decreased swelling of the prostate tissue.24 The limited data comparing SPBE to alpha-blockers have shown the alpha-blockers to be superior to SPBE in improving symptomatology.22 The most widely studied saw palmetto preparation is permixon. The literature is limited by small sample sizes and varying SPBE preparations. The National Center for Complementary and Alternative Medicine (NCCAM) of the NIH is currently funding a large, year-long, randomized, controlled trial of saw palmetto.
Safety: Saw palmetto is well tolerated. There are no consistently reported adverse effects from its use at recommended doses. There are no long-term data on its effects on lipids, bone density, or other systems that may be affected if it does have hormonal activity. Several studies have shown that SPBE does not affect PSA levels.22,24 However, there have been many case reports to the contrary, and many urologists feel that the data contradict their clinical experience with saw palmetto and are concerned that use of the herb will falsely lower PSA.26
Drug Interactions: None reported.
Dose/Cost: 160 mg b.i.d.; cost ranges from $15-54 per month.
St. John’s Wort
Plant: Hypericum perforatum.
History and Clinical Uses: The ancient Greeks and 19th century European physicians used St. John’s wort for mood disorders. It is the most widely prescribed antidepressant in Germany surpassing all other prescriptions including selective serotonin reuptake inhibitors (SSRIs).
Preparation: An ethanolic extract of the flowering parts of the plant is usually standardized to 0.2-0.3% hypericin. The concentration of hypericin/flavonoids varies greatly by genetic variation and time of harvesting. A standardized German extract (Ze117) has been used in many studies.
Active Ingredients: Hypericin has been postulated for some time to be the active ingredient even though it does not cross the blood-brain barrier. Newer data have cast some doubt on this, as well as implicating another substance, hyperforin, as the active ingredient. Hypericin and hyperforin contents vary independently from plant to plant. This may account for the variability of the study’s findings.
Modes of Action: Hypericin has been inconsistently shown to exhibit MAO inhibition and peripheral serotonin activity. Hyperforin may have dopaminergic, noradrenergic, and serotonergic activity.
Efficacy: Until recently, the data on St. John’s wort’s efficacy as an antidepressant were fairly consistent. Several meta-analyses27-29 and a large multicenter trial in Germany30 concluded that St. John’s wort was better than placebo and equivalent to low-dose tricyclic antidepressants in treating mild-to-moderate depression and with fewer side effects than the tricyclics. There has been 1 study comparing a standardized St. John’s wort extract (Ze117) to fluoxetine 20 mg/d in 240 subjects with mild-to-moderate depression.31 It found that while efficacy was similar for the 2 therapies, St. John’s wort was better tolerated. The first multicenter, placebo-controlled trial in the United States has recently reported different results.32 It was concluded that treatment with a standardized extract of St. John’s wort for 8 weeks was found to be no better than placebo (see Figure 4) in treating moderate-to-more severe depression. While overall response rates did not differ, the number of patients experiencing more complete recovery ("remission rates") was greater in the St. John’s wort group over placebo (14.3% vs 4.9%). Some have questioned the validity of these results as the placebo response rate was 18.6%, well below the usual 30% seen in depression studies. The study was funded by Pfizer, the maker of Zoloftä.33
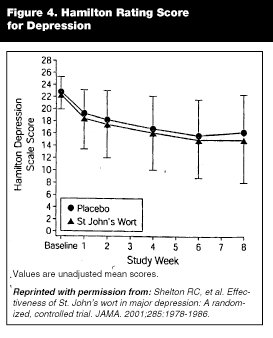
|
Safety: While many studies have shown St. John’s wort to be better tolerated than tricyclic antidepressants, recently there have been many concerns raised about St. John’s wort’s safety. Photosensitivity has been seen in animals grazing on St. John’s wort. There was a recent report from a trial finding that St. John’s wort produced a significant phototoxicity in patients with HIV.34 Other adverse effects reported with St. John’s wort include dry mouth, dizziness, fatigue, and constipation.
Drug Interactions: The most significant concerns about St. John’s wort have centered on its interactions with other medications that are metabolized by cytochrome P450 in the liver. There have been multiple case reports of St. John’s wort decreasing the levels of theophylline and cyclosporin, decreasing patients’ INR on coumadin, and causing breakthrough bleeding in patients on oral contraceptives.35 One study found that the digoxin levels of volunteers taking St. John’s wort was significantly lower than those taking placebo, and the effect increased with the number of days on St. John’s wort.35 Another important interaction that has been recognized reinforces the potential danger of using St. John’s wort in conjunction with other antidepressants. Five elderly patients on antidepressants (4 on SSRIs and 1 on nefazadone) and St. John’s wort exhibited symptoms characterisitic of serotonin syndrome (delerium, tremor, headache, myalgias, and restlessness).36
Dose/Cost: 300 mg t.i.d. of extract standardized to 0.3% hypericin; cost ranges from $14-25 per month.
Echinacea
Plant(s): Purple coneflower in the daisy family—there are 9 species of Echinacea. The 3 most commonly marketed are E purpurea, E pallida, and E angustifolia.
History and Clinical Uses: Native Americans have used Echinacea preparations as antiseptics and analgesics for centuries.11,37 Other clinical conditions for which Echinacea has been used include: upper and lower respiratory infections and other infections as an immunostimulant, toothaches, abdominal pain, and topically for snake bites and other wound healing. Echinacea is extremely popular in both Europe and the United States, with sales exceeding $70 million in the United States in 1998.
Preparations: Many different preparations are available varying by species and part of plant used. Preparations include: using whole plant dried or extract, plant juice, root extract, above-ground parts only dried, or extract. There is no uniform extraction process. The 2 preparations that have been approved for use in Germany are E purpurea plant juice (made from above-ground parts) and E pallida root tincture.
Active Ingredients and Mode of Action: There are 7 known active constituents: polysaccharides, flavonoids, essential oils, polyacetylenes, alkamides, caffeic acid derivatives, and glycoproteins. Their proposed immunological mechanisms of actions include: stimulating phagocytosis, increasing lymphocyte activity, and tumor necrosis factor (TNF). In wound healing, the proposed mechanisms of action include: inhibition of tissue and bacterial hyaluronidase, promotion of the formation of mesenchymal mucopolysaccarides, and of the differentiation of fibrocytes from fibroblasts.
Efficacy: There have been several randomized, controlled trials38-40 and 2 systemic reviews41,42 looking at the use of Echinacea for the prevention and/or treatment of viral upper respiratory infections. The data for the use of Echinacea in the prevention of colds, even in those who experienced more than 3 in the previous year,39 have been largely negative. The data looking at the use of Echinacea in the treatment of colds have been more mixed. The multitude of preparations of Echinacea used and studied has produced a heterogeneous literature and has prohibited the usefulness of a meta-analysis to further evaluate its efficacy.
Safety: There have been few reports of adverse effects in studies on Echinacea. However, there have been reports of allergic reactions to the herb ranging in severity from mild urticaria to severe asthma and anaphylaxis.37 Given the immunological effects that Echinacea is purported to have, there is a theoretical risk of use in persons with immunodeficiency or autoimmune disorders. Due to the concern for depressed immunity from prolonged use, there is a recommendation to not use Echinacea for more than 8 weeks consecutively. There are no data to support the usefulness of "drug holidays" for those who want to use it long-term.
Drug Interactions: There are no known drug interactions. There is a theoretical caution against the use of Echinacea with immunosuppressants.
Dose/Cost: Dose is dependent on preparation. Some examples include: 6-9 mL of E purpurea plant juice per day, echinaforce (6.78 mg E purpurea extract per tablet) 3 tablets twice daily, E pallida root tincture 900 mg/d. There is a wide variation in cost depending on preparation, approximately $8-20 per month.
Golden Seal
Plant: Hydrastis canadensis is a member of the buttercup family. The name golden seal comes from its bright yellow rhizome. It grows wild in the woodland areas in the eastern United States. Over-harvesting in the 1980s and 1990s led to its classification as an endangered species. It is now being cultivated in Washington state as a cash crop.
History and Clinical Uses: Native Americans have used golden seal for its antimicrobial properties as a treatment for stomach ulcers, a diuretic, and as an eye wash for sore eyes. Topically it was used for eczema, itching, and wounds. Since the 1970s, it has been used by some to mask urine tests for illicit drugs including morphine, cocaine, and marijuana. It is frequently combined in herbal preparation with Echinacea as an immune stimulant or anticold remedy.
Preparations: It is available as dried root and rhizome in capsules, tinctures, extracts, salves, and ointments. It is not typically standardized to any 1 component and because over-collection has led to high prices, it is often adulterated with other herbs.
Active Ingredient and Mode of Action: The active ingredients are a group of alkaloids—the most significant of which are berberine and hydrastine. There is some in vitro data that indicate that berberine has some antimicrobial activity through inhibition of adherence of microbes to host cells.43
Efficacy: There are no clinical studies of golden seal in humans and limited animal data.
Safety: With short-term use in recommended doses (less than 500 mg of berberine or 8-10 g of dry root), no adverse effects have been reported. However, long-term use and larger doses have been reported to cause digestive disorder, hallucinations, and delirium. There have been reported cases of death from overdose. The LD50 of berberine in humans is 27.5 mg/kg.43
Drug Interactions: None reported.
Dose/Cost: For cold and flu, 1 or 2 500-mg capsules are recommended 2-3 times a day. One teaspoon of herb or tincture can be added to a cup of boiling water and used as a tea 3 times a day. Cost can vary between $16-38/month.
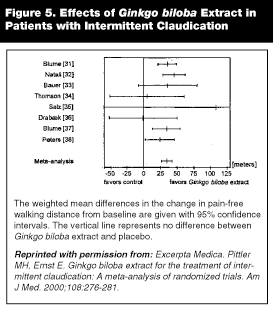
|
Valerian
Plant: Valeriana officinalis.
History and Clinical Uses: There is a historical record of valerian’s use dating back to Hippocrates and Galen. Valerian is currently used as an anxiolytic and a sleep aide.
Preparations: There are a great variety of valerian preparations made from the root including: extracts standardized to anywhere from .015% to 1% valerenic acid, full-strength root tinctures, and pure root. A tea can be made by steeping 2-3 g of dried root in 150 mL of boiling water. Many commercial products marketed for stress, sleep, and "for women" contain valerian in combination with ginseng, kava kava, evening primrose, and passion flower.
Active Ingredients and Mode of Action: There are many constituents thought to be responsible for the sedating and anxiolytic properties of valerian. Valerenic acid and other sesquiterpenes are the best studied. There is some evidence that these compounds inhibit the catabolism of GABA and may also bind loosely to benzodiazepene receptors.44
Efficacy: Most of the studies evaluating valerian’s effect on sleep in normal patients and those with insomnia have been small, performed in Germany, and have used differently prepared valerian root extracts. There have been, however, at least 2 randomized, blinded, crossover studies that used polysomnography to evaluate the effect of valerian on sleep architecture. They found that after several doses of valerian, patients with insomnia had improved sleep latency45 and quality with an increase in REM sleep compared to the placebo study period.46 There are insufficient data to comment on valerian’s effectiveness in treating anxiety.
Safety: Clinical studies done on more than 12,000 patients for as long as 14 days have reported few significant adverse effects of valerian. There may be some sedation or morning drowsiness at higher doses.44 However, extended use has been associated with a withdrawal syndrome similar to that seen with benzodiazepenes.47
Drug Interactions: There is a theoretical danger of mixing valerian with other sedating substances with common mechanisms of action such as: alcohol, benzodiazepenes, and barbiturates.
Dose/Cost: The dose ranges greatly based on preparation. 400-900 mg of valerian root extract can be taken 1-2 hours before bedtime. The cost can range from $7-20 a month.
Kava (also known as kava kava)
Plant: Piper methysticum.
History and Clinical Uses: Kava has a long folk tradition for use as a sedative and anxiolytic. It has been used throughout the Polynesian Islands for ceremonial rituals for thousands of years. It is used similar to the way alcohol is used in Western societies. Its use for anxiety and stress has been increasing, and it is a common ingredient in the compound herbal preparations marketed for this purpose.
Preparations: Most kava root extracts are standardized to between 30-70% kava-lactone content. It can also be taken as a tea made from 2-4 g of dried root and 150 mL of boiling water. In the Polynesian Islands, a drink is prepared by masticating the roots and mixing the resulting liquid with water.
Active Ingredients and Mode of Action: The active ingredients are called kava lactones or pyrones. Their mode of action is unknown. They are not thought to affect benzodiazepene or GABA receptors.
Efficacy: There are several small European trials that were done to assess the efficacy of kava in the symptomatic treatment of anxiety. One systematic review and meta-analysis48 reported that all 7 double-blind randomized, controlled studies meeting their strict methodological criteria found kava to be superior to placebo in treating anxiety. Pooled data from 3 of the studies found significant improvement in Hamilton Anxiety Rating Scale for Anxiety (HAM-A) scores with kava.
Safety: While short-term use is felt to be safe, long-term use has been associated with "kawaism" or kava addiction and Kava dermopathy (a pellagra-like syndrome unresponsive to therapy with niacin) in the Polynesian Islands. Kava can cause drowsiness and impaired motor reflexes but is not felt to impair cognitive function like benzodiazepenes.49
Drug Interactions: There has been a report of coma resulting from the combined intake of kava and alprazolam.50 There is also a theoretical danger of mixing kava with other sedating substances such as alcohol and barbiturates.
Dose/Cost: For kava root extract products standardized to 70% kava lactones, the recommended dose is 100 mg 3 times a day. A cup of tea 3 times a day can be used with the tea made by adding boiling water to 2-4 g of root. Cost is about $10-15 per month.
Feverfew
Plant: Tanacetum parthenium (a daisy-like perennial).
History and Clinical Use: Feverfew is used for fever, menstrual irregularities, arthritis, and most commonly for migraine headache.
Preparations: Dried whole leaf is used standardized to 0.2-0.7% parthenolide. Many products, however, are not standardized and the amount of feverfew in each capsule can vary greatly.
Active Ingredients and Mode of Action: The only currently identified ingredient thought to possess clinical activity is parthenolide. It has been proposed that parthenolide inhibits prostaglandin synthesis and has been found to inhibit platelet aggregation.
Efficacy: There are little data on feverfew. One randomized, controlled, crossover trial published in the Lancet51 did find a significant decrease in number and severity of headaches with fewer associated symptoms like nausea and vomiting. A Cochrane Review on feverfew52 reviewed 4 methodologically sound trials. Three of the trials suggested beneficial effects of feverfew, but the fourth, which was negative, was the largest and had the highest methodological quality. While there are insufficient data to draw any firm conclusions on the efficacy of feverfew, its use in migraine prevention seems promising.
Safety: No reported consistent adverse effects.
Drug Interactions: Given the antiplatelet aggregation effects of feverfew, there is a theoretical risk of using feverfew with anticoagulants, antiplatelet drugs, or nonsteroidal anti-inflammatory drugs (NSAIDs).
Dose/Cost: 125 mg once or twice a day of dried whole leaf standardized to .2% parthenolide. Cost can vary as much as $4-25 per month.
Ginseng
Plant: Panax ginseng (Asian ginseng or Chinese ginseng); Panax quinquefolius (American ginseng); (Eleutherococcus senticosus (Siberian ginseng—not a true ginseng).
History and Clinical Uses: For thousands of years, ginseng has been used throughout Asia for medicinal purposes. It has been used throughout the world as an "adaptogen," or a substance that can help the body to adapt to the stress it encounters in its environment, or "it’s good for what ails you." It is also used to increase energy, decrease glycemia, and as an aphrodisiac. Wild American ginseng is so widely sought after that it is classified as an endangered species.
Preparations: Products are rarely standardized. Asian ginseng is typically used as a powder of root extract. American ginseng is used as the pure root. Because of the decreasing availability of ginseng, especially American ginseng, products are often adulterated with other herbs and sometimes do not contain any detectable ginseng at all.
Active Ingredients and Mode of Action: The active ingredients are known as either ginsenosides or panaxans of which there are many subtypes. Ginsenoside Rb-1 is thought to be responsible for the hypoglycemic effect of ginseng by decreasing islet cell insulin concentration.58 The mode of actions of the other ginsenosides is unknown.
Efficacy: A review of 16 studies on ginseng for multiple indications (ie, physical performance, psychomotor performance, cognitive function) found "no compelling evidence for any of these indications."54 There was a recent double-blind, randomized, placebo-controlled study done in Canada that found American ginseng significantly decreased the glycemic response when taken 40 minutes before a glucose challenge in normal subjects or when taken either 40 minutes before or at the time of a glucose challenge in diabetics.53
Safety: Little is known about the safety of long-term ginseng use. There are few consistently reported or validated adverse effects despite previous concerns about estrogenic effects.
Drug Interactions: Ginseng has been reported to lower the international normalized ratio (INR) in patients on coumadin.55 There is a theoretical risk of causing hypoglycemia in diabetics by augmenting their hypoglycemia therapies.
Dose/Cost: For American ginseng, the recommended dose is 0.25-0.5 g of root 2 times daily. For Asian ginseng, the recommended dose is 0.6-3.0 g of powdered root extract 1-3 times a day. The cost of ginseng can be high, especially for true American ginseng ($30-50/mo), while Asian ginseng or Korean ginseng is often less expensive ($10-40/mo).
Chasteberry
Plant: Vitex agnus-castus also known as "Monk’s pepper."
History and Clinical Use: Chasteberry gets its name from the belief that eating the berries would inspire chastidy. Chasteberry has been used in Europe for many years to treat infertility, mastodynia, and menstrual complaints, including PMS.
Preparations: Chasteberry can be used as the whole dried fruit or as an aqueous extract of the berry standardized to 0.5% agnuside (thought to be an inactive ingredient).
Active Ingredient and Mode of Action: The active ingredients include iridoid glycosides, flavonoids, volatile oils, and progestins. There is some evidence that chasteberry is anti-androgenic. It has been shown in animal and in vitro studies to inhibit prolactin and possess dopaminergic activity.56
Efficacy: There is some evidence from German studies that chasteberry is effective in treating the symptoms of PMS. One randomized, controlled study found that in women who had both high prolactin levels and PMS, the chasteberry was effective in reducing both their symptoms and prolactin levels.56 A recent double-blind, randomized, controlled trial treated women with a standardized chasteberry extract and found significant improvement in both emotional and physical symptoms of PMS.57
Safety: In studies, chasteberry has been extremely well tolerated. Other than minor gastrointestinal (GI) upset, headache, or allergic reactions, no adverse effects have been reported for chasteberry.
Drug Interactions: There are theoretical concerns about chastberry’s ability to interfere with dopamine antagonists and oral contraceptives. No interactions have been reported.
Dose/Cost: Recommended dose is either 0.25-1.0 g of dried fruit 3 times daily or 175-225 mg of extract 1-2 times a day. Cost ranges from $12-36 per month.
Milk Thistle
Plant: Silybum marianum.
History and Clinical Uses: Milk thistle has been used as a "liver protectant" for centuries. It has been used for cirrhosis, chronic hepatitis, and mushroom poisoning.
Preparations: An extract from the fruit and seed is standardized to 70% silymarin. Milk thistle is not very soluble in water and is not effective if taken as a tea.
Active Ingredient and Mode of Action: Silymarin is the active ingredient that consists of 4 flavanolignans—the most active of which is silybin. Silymarin is thought to protect hepatocytes by altering the cell membrane to prevent penetration by toxins, increase protein synthesis, and exhibit antioxidant properties.58
Efficacy: There are a small but promising amount of data showing milk thistle to be effective in decreasing mortality from alcoholic cirrhosis and chronic hepatitis.59 There are also 2 studies showing IV silymarin decreases mortality in Amanita mushroom poisoning.60 Milk thistle is approved in Germany for the treatment of cirrhosis, hepatitis, and similar liver conditions.
Safety: In the studies that have been done to date, milk thistle has appeared to be well tolerated.
Drug Interactions: No drug interactions have been reported.
Dose/Cost: The recommended dose of 70% silymarin extract is 200-400 mg/d. Cost ranges from $10-46 per month.
Cranberry
Plant: Vaccinium macrocarpan.
History and Clinical Uses: Cranberry has its greatest use as a food. It is also used for the treatment and prevention of urinary tract infections (UTIs).
Preparations: Cranberry for medicinal purposes can be used as undiluted cranberry juice, raw berries, or capsules containing dried berries.
Active Ingredient and Mode of Action: It was previously thought that cranberry acidified the urine. It is now known that proanthocyandin is the active ingredient and it works by interfering with bacterial adherence to the urinary tract epithelium. Fructose may play an anti-infective role.
Efficacy: A double-blind, randomized, controlled trial showed that drinking 300 mL of cranberry juice cocktail daily for 6 months significantly reduced the frequency of bacteriuria and pyuria in older women.61 There is little evidence that cranberry can cure an acute UTI.
Safety: Drinking more than 3 or 4 L of cranberry juice at a time can cause diarrhea. In recommended doses, there are no adverse effects.
Drug Interactions: There are none reported.
Dose: For UTI prevention, the recommended dose is 3 oz cranberry juice a day. For UTI treatment, it is 12-22 oz per day.
Potentially Toxic Herbs and Herb/Drug Interactions
Hepatotoxicity. There are multiple herbs that have been shown to be hepatotoxic (see Table 1). Most of the herbs listed have been associated with hepatotoxicity through multiple case reports where the same clinical scenario is seen with the same herb on multiple occasions. Some of them have also been associated with hepatic toxicity by case reports where withdrawal of the herb resulted in recovery and reintroduction-caused recurrence. While the herbs listed are more commonly associated with reversible hepatic damage, they have also been associated with at least 1 case of fatal liver failure or cirrhosis.62-64
| Table 1. Hepatotoxic Herbs | |||
| Herb | Clinical Use | Clinical Outcome | Miscellaneous |
| Germander | weight loss | acute nonviral hepatitis | banned in France |
| Chapparal | antioxidant | acute symptomatic hepatitis | FDA warning 1992 |
| Jin Bu Huan | sedative, analgesic | acute symptomatic hepatitis | |
| Pennyroyal oil | abortifacient, to flavor teas |
severe hepatic toxicity | |
| Comfrey(Pyrrolizidinealkaloids) | multiple uses | hepatic veno-occlusive disease | use restricted in United Kingdom,Canada, Australia, and Germany |
Nephrotoxicity. One of the most concerning stories of toxicity from an herbal therapy began in 1992 when physicians in Belgium began to see multiple cases of extensive renal interstitial fibrosis in women who had been treated at a diet clinic with a certain Chinese herbal therapy. It was later concluded that one of the botanicals (Stephania tetrandra) in the diet aide had been inadvertently substituted with Aristolochia fangchi, a botanical known to contain aristolochic acid, which has been shown to be highly nephrotoxic and carcinogenic in animals.65
To date, more than 100 cases of "Chinese herbal nephropathy" have been reported in Belgium with 70 requiring either dialysis or transplantation. Such a significant number of these patients had been diagnosed with renal or urothelial carcinoma that many of the patients underwent prophylactic nephrectomy. When the removed kidneys were examined, 46% of those patients were found to have carcinoma of the kidney or urinary tract.65 Chinese herbal nephropathy has now been reported in the United Kingdom, France, and the United States.66 The use of herbs containing aristolochic acid has been banned in Belgium, Canada, United Kingdom, France, Australia, and Germany. They are still available in the United States.67 The FDA has recently issued a letter on its web site warning physicians of the safety concerns about the use of herbs containing aristolochic acid.66
Risks of Herbs During Surgery. There are several herbal medicines that have been reported to cause or have the potential to cause problems in patients having surgery. Asian ginseng, feverfew, garlic, ginger, and ginkgo all inhibit platelet aggregation and may increase bleeding, especially in patients on anticoagulants or antiplatelet medications. Asian ginseng and St. John’s wort have both been reported to have the opposite effect on warfarin and have resulted in a significant lowering of patients’ INR.68 Herbs that have significant sedating effects can have an additive effect with anesthetic agents. This has been seen with kava, valerian, and St. John’s wort.69 The American Society of Anesthesiologists has cautioned their members to ask all patients about their use of herbal supplements and advise patients who are taking herbal medicines to stop them 2-3 weeks prior to surgery.70
Talking to Patients about Herbal Medicine
At the present time, there is a relative paucity of information about herbal medicines to guide the clinician. It is clear, however, that with or without our guidance our patients are using herbal preparations in significant numbers and not telling us about it most of the time.3,4 The Eisenberg survey found that among the adults taking prescription medications (44% of sample), only 1 in 5 reported the concurrent use of herbs or megavitamins to their physician.4 It is our responsibility to get a full history of all the herbals that our patients are taking. Ask the questions with interest in a nonjudgmental way to overcome the patient’s resistance to sharing this information. It is important to acknowledge the limits of your knowledge in this area and how much you are willing to do to increase that knowledge. How each clinician proceeds from that point may differ depending on their interpretation of the information available. Some may feel comfortable recommending certain herbal therapies in selected clinical scenarios, and others may not feel comfortable giving any degree of approval for patients’ use of herbals. It is important to provide patients with the information they need to choose herbal therapies in the safest way possible if that is their choice. Patients should avoid compound herbal products that contain many different herbs and herbal treatments with known adverse or toxic effects. They should avoid mixing certain herbs and prescription medications and be advised to watch for any interactions if they do take these concurrently. There is not enough information available to safely recommend herbal therapies for long-term use or for use during pregnancy, lactation, or in children. Patients need to know that the current regulations governing the manufacturing and marketing of herbal products do not mandate standardization by the FDA. The contents of some of these products have been shown not to match the ingredients listed on the label. Herbal remedies have been found to contain significantly more or less than the amount listed. Some contain adulterants with pharmaceuticals and some are contaminated with heavy metals. There may be greater safety in using products that are "standardized extracts," made by larger pharmaceutical companies or who have used HPLC to ensure the purity and identity of its ingredients. The most important message of all to patients is that "natural" does not always mean safe.
Summary
Since the beginning of time, man has been using the plants of the earth to treat the ails of its people. In the 20th century, these traditional therapies were all but lost with the advent of modern pharmaceuticals. Many factors have contributed to the resurgence seen with herbal medicine. What is clear is that these products are imminently available and being used by our patients and that we need this information. How safe and efficacious these products are is less clear. The number of methodologically strong studies being published on herbs is growing and will hopefully clarify some of these issues. At this point, some herbal therapies (the vast minority, unfortunately) appear to be safe and perhaps even efficacious while others are clearly neither. Many are calling for more stringent controls on the industry to increase our confidence in the quality and safety of these products. We can only hope that some of the changes currently being proposed will help to clarify what is now a difficult and murky clinical issue.
References
1. US Government Printing Office. Alternative Medicine; Expanding Medical Horizons. A Report to the NIH on Alternative Medical Systems and Practices in the United States. US Government Printing Office 017-040-00537-7.
2. Brevoort P. The Booming US Botanical Market. Herbalgram. 1998;44:33-47.
3. Eisenberg DM, et al. Unconventional medicine in the United States. Prevalence cost and patterns of use. N Engl J Med. 1993;328:246-252.
4. Eisenberg DM, et al. Trends in alternative medicine use in the U.S. 1990-1997. JAMA. 1998;280:1569-1575.
5. The Integrator for the Business of Alternative Medicine. Sacramento, Calif: Landmark Healthcare, Inc.; 1999.
6. Palinkas LA, Kabongo ML. The use of CAM by primary care patients. J Fam Pract. 2000;49:1121-1130.
7. O’Mathuna DP. Where to with herbals? Altern Ther Health Med. 2000;6:34-35.
8. Ryser R. The Journal News. April 29, 2001:13A.
9. Blumenthal M. Herbs sales down 15% in mainstream market. Herbalgram. 2001;51:69.
10. Brody J. American gamble on herbs as medicines. New York Times/Science Times. February 9, 1999:1, 6, 7.
11. Blumenthal M. Herb market levels off after 5 years of boom. Herbalgram. 1999;47:64-65.
12. Herbal Rx promises and pitfalls. Consumer Reports. March 1999:44-48.
13. Dietary Supplement Health and Education Act of 1994. December 1, 1995. Available at: http://www.cfsan.fda.gov/~dms/dietsupp.html. Accessed June 28, 2001.
14. FDA Talk Paper. US Department of Health and Human Services. FDA announces the availability of new ephedrine and "street drug alternative" documents. Available at: http://www.fda.gov/bbs/topics/ANSWERS/ANS01008.html. Accessed June 28, 2001.
15. Blumenthal M. Ekpheda update. Herbalgram. 2000;50:64-65.
16. Haller CA, Benowitz NL. Adverse cardiovascular and central nervous system events associated with dietary supplements containing ephedra alkaloids. N Engl J Med. 2000;343:1833-1838
17. Schulz V, Hansel R, Tyler VE. Rational Phytotherapy. New York, NY: Springer; 1998:6.
18. Herbal roulette. Consumer Reports. November 1995:698-705.
19. Gurley BJ, Gardner SF, Hubbard MA. Content versus label claims in ephedra-containing dietary supplements. Am J Health Syst Pharm. 2000;57:963-969.
20. Ko RJ. Adulterants in Asian patent medicines. N Engl J Med. 1998;339:847.
21. Bayne HM. FDA publishes new draft guidance for botanical drug products. Herbalgram. 2000;50:68-69.
22. Gerber GS. Saw palmetto for the treament of men with lower urinary tract symptoms. J Urol. 2000;163:1408-1412.
23. Wilt TJ, et al. Saw palmetto extracts for treatment of BPH. JAMA. 1998;280:1604-1609.
24. Marks LS, et al. Effects of a saw palmetto herbal blend in men with symptomatic BPH. J Urol. 2000;163:1451-1456.
25. Wilt TJ, MacDonald R, Ishani A. Beta-sitosterol for the treatment of benign prostatic hyperplasia: A systematic review. BJU Int. 1999;83(9):976-983.
26. Barrette EP. Saw palmetto for BPH: An update. Alternative Medicine Alert. 2001;4(3):25-28.
27. Linde K, et al. St. John’s wort for depression. BMJ. 1996; 313:253-258.
28. Linde K, Mulrow CD. St. John’s Wort for Depression (Cochrane Review). Oxford, England: Update Software; 1999.
29. Gaster B, Holroyd J. St. John’s wort for depression: A systematic review. Arch Intern Med. 2000;160:152-156.
30. Woelk H. Comparison of St. John’s wort and imipramine for treating depression: Randomized controlled trial. BMJ. 2000; 321:536-539.
31. Schrader E. Equivalence of St. John’s wort extract (Ze117) and fluoxetine: A randomized, controlled study in mild-moderate depression. Int Clin Psychopharmacol. 2000;15:61-68.
32. Shelton RC, et al. Effectiveness of St. John’s wort in major depression. JAMA. 2001;285:1978-1986.
33. Questions raised about latest St. John’s wort study in JAMA [press release]. Austin, Tex: American Botanical Council; April 13, 2001. Available at: http://www.herbalgram.org/press/041301press.html. Accessed June 28, 2001.
34. Gulick RM, et al. Phase I studies of hypericin in HIV-infected adults. Ann Intern Med. 1999;130:510-514.
35. Ernst E. Second thoughts about safety of St. John’s wort. Lancet. 1999;354:2014-2016.
36. Lantz MS, et al. St. John’s wort and antidepressant drug interactions in the elderly. J Geriatr Psychiatry Neurol. 1999;12:7-10.
37. Kattapong VJ. Echinacea for colds. Alternative Medicine Alert. 2000;3(11):128-130.
38. Melchart D, et al. Echinacea root extracts for the prevention of upper respiratory tract infections. Arch Fam Med. 1998;7: 541-545.
39. Grimm W, Muller H. A randomized, controlled trial of the fluid extract of E purpurea on the incidence and severity of colds and respiratory infections. Am J Med. 1999;106:138-143.
40. Gunning K. Echinacea in the treatment and prevention of upper respiratory tract infections. West J Med. 1999;171:198-200.
41. Barrett B, et al. Echinacea for upper respiratory infections. J Fam Pract. 1999;48:628-634.
42. Melchart D, et al. Echinacea for Preventing and Treating the Common Cold (Cochrane Review). Oxford, England: Update Software; 2001.
43. Natural Medicines Comprehensive Database. Stockton, Calif: Therapeutic Research; 2001:504-505.
44. Natural Medicines Comprehensive Database. Stockton, Calif: Therapeutic Research; 2001:1053.
45. Hardy ML. Valerian root for insomnia. Alternative Medicine Alert. 1999;2:85-96.
46. Donath F, et al. Critical evaluation of the effect of valerian extract on sleep structure and sleep quality. Pharmacopsychiatry. 2000;33:47-53.
47. Garges HP, et al. Cardiac complications and delirium associated with valerian root withdrawal. JAMA. 1998;280:1566-1567.
48. Pittler MH, Ernst E. Efficacy of kava extract for treating anxiety: Systematic review and meta-analysis. J Clin Psychopharmacol. 2000;20:84-89.
49. Natural Medicines Comprehensive Database. Stockton, Calif: Therapeutic Research; 2001:626.
50. Almeida JC, et al. Coma from the health food store: Interaction between kava and alprazolam. Ann Intern Med. 1996;125: 940-941.
51. Murphy JJ, et al. Randomized, double-blind, placebo-controlled trial of feverfew in migraine prevention. Lancet. 1988;23: 189-192.
52. Pittler MH, Vogler BK, Ernst E. Feverfew for Preventing Migraine (Cochrane Review). Oxford, England: Update Software; 2001.
53. Vuksan V, et al. American ginseng reduces postprandial glycemia in nondiabetic subjects and subjects with type 2 diabetes. Arch Intern Med. 2000;160:1009-1013.
54. Vogler BK, Pittler MH, Ernst E. The efficacy of ginseng. A systematic review of randomized, clinical trials. Eur J Clin Pharmacol. 1999;55:567-575.
55. Miller LG. Herbal medicinals. Arch Intern Med. 1998;158: 2200-2211.
56. Klepser T, Nisly N. Chaste tree berry for premenstrual syndrome. Alternative Medicine Alert. 1999;2(6):64-67.
57. Schellenberg R. Treatment for the premenstrual syndrome with agnus castus fruit extract: Prospective, randomized, placebo-controlled study. BMJ. 2001;322:134-137.
58. Natural Medicines Comprehensive Database. Stockton, Calif: Therapeutic Research; 2001:733.
59. NCCAM Clearinghouse. Hepatitis C: Treatment Alternatives. Available at: http://nccam.nih.gov/fcp/factsheets/hepatitisc/hepatitisc.htm. Accessed June 28, 2001.
60. Foster S. Milk Thistle. Austin, Tex: American Botanical Council; 1996.
61. Avorn J, et al. Reduction of bacteriuria and pyuria after ingestion of cranberry juice. JAMA. 1994;271:751-754.
62. Larson AM, Carithers RL. Hepatotoxicity due to herbal medications. UpToDate, Inc. 2001.
63. Ernst E. Harmless herbs? A review of the recent literature. Am J Med. 1998;104:170-178.
64. U.S. Food and Drug Administration Center for Food Safety and Applied Nutrition. Illnesses and injuries associated with the use of selected dietary supplements. Unsubstanciated Claims and Documented Health Hazards in the Dietary Supplement Marketplace. 1993. Available at: http://www.cfsan.fda.gov/~dms/ds-ill.html. Accessed June 28, 2001.
65. Nortier JL, et al. Urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi) N Engl J Med. 2000; 342:1686-1692.
66. U.S. Food and Drug Administration Center for Food Safety and Applied Nutrition. Botanical products determined by FDA to contain aristolochic acids. April 9, 2001. Available at: http://www.cfsan.fda.gov/~dms/ds-bot3.html. Accessed June 28, 2001.
67. Kessler DA. Cancer and herbs. N Engl J Med. 2000;342: 1742-1743.
68. Fugh-Berman A. Herb-drug interactions. Lancet. 2000;355: 134-138.
69. Hardy ML. Herb-drug interactions: An evidence-based table. Alternative Medicine Alert. 2000;3(6):64-69.
70. Anesthesiologists warn: If you’re taking herbal products, tell your doctor before surgery [press release]. Park Ridge, Ill: American Society of Anesthesiologists. Available at: http://www.asahq.org/PublicEducation/herbal.html. Accessed June 28, 2001.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
