Thoracic Trauma: Principles and Practice of Emergency Stabilization, Evaluation, and Management - Part III
Thoracic Trauma: Principles and Practice of Emergency Stabilization, Evaluation, and Management
Part III: Injuries to the Heart and Aorta
Authors: Elizabeth B. Jones, MD, Assistant Professor, Department of Emergency Medicine, University of Texas-Houston Medical Center; Kimberly Chambers, MD, Assistant Professor, Department of Emergency Medicine, University of Texas-Houston Medical Center; Luis Haro, MD, Assistant Professor, Department of Emergency Medicine, Mayo Clinic, Rochester, MN.
Peer Reviewers: Larry B. Mellick, MD, MS, FAAP, FACEP, Chair and Professor, Department of Emergency Medicine, Section Chief, Pediatric Emergency Medicine, Medical College of Georgia, Augusta; Steven Winograd, MD, FACEP, Attending Physician, Department of Emergency Medicine, Allegan General Hospital, Allegan, MI; Southwestern Michigan Emergency Services, PC.
Injuries to the heart and great vessels are truly critical injuries. Both blunt and penetrating injuries to the great vessels often are instantly fatal. Of these injuries, only blunt cardiac injury has a reliably benign course. For years, there has not been much change in the diagnosis or treatment of these injuries. Patients with penetrating injury to the mediastinum traditionally underwent thoracotomy and patients with suspected blunt TAI underwent aortography. These standard treatments have changed in the last few years. The helical computed tomography (CT) is now routinely used in the diagnosis of blunt traumatic aortic injury (TAI), and in some centers a more selective approach to transmediastinal penetrating trauma has been advocated.
This article is the third in a three-part series on thoracic trauma. The first article reviewed initial treatment of all chest trauma and the use of radiologic tests in the diagnosis of chest trauma. The second article discussed injury to the thorax, lungs, esophagus, and diaphragm. This article contains a summary of radiologic diagnostic criteria for TAI.— The Editor
Injury to the Thoracic Great Vessels
Mortality from major vessel injury has risen from less than 1% in 1947 to as high as 15% in recent years.1 The increasing number of high-speed automobile accidents primarily has accounted for this rise. Major vessel injury is more frequent with penetrating trauma. Ninety percent of major vessel injuries are caused by penetrating trauma, and 10% by blunt trauma. The aorta is the most commonly injured vessel in blunt trauma and, because of its high mortality, aortic injury is the second most common cause of blunt traumatic death.2,3 The true incidence of aortic injuries is difficult to determine and varies with the population studied.
In an autopsy study of 530 motor vehicle fatalities, aortic injury was found in 90 victims (17%).4 One study found an incidence of 8% of aortic injuries in patients arriving to the emergency department (ED),5 while another found an incidence of 21% in a combined clinical and autopsy study.6 The ED clinician has a key role in the outcome of patients who sustain these injuries and survive to reach the ED. With prompt diagnosis and surgery, 71-84% will survive, whereas 90% will die without intervention.3,7
Other vessels, such as the aortic brachiocephalic branches, pulmonary arteries and veins, superior and inferior vena cava, and the innominate artery and vein, are less commonly injured but may cause significant injury and death.8-11 Unfortunately, not all injuries are a result of out-of-hospital events. Iatrogenic laceration of various vessels has been reported as a complication of central venous catheter insertion, chest tube insertion, intrathoracic counter-pulsation balloon insertion, over-inflation of Swan-Ganz catheter, and other diagnostic and therapeutic procedures.10-12
History. Thoracic major vessel injury has been recognized for more than 400 years. In 1557, Vesalius was the first to report the death of a patient from a ruptured aorta after being thrown from a horse. In 1922, Dfhanelidze reported the first successful repair of a puncture wound of the ascending aorta. De Bakey and Cooley successfully resected a posttraumatic thoracic aneurysm and replaced the aorta with a graft in 1954. Four years later, in 1958, Klassen completed the first successful repair of an acute traumatic thoracic aortic injury. The modern era of successful repair has been far more recent. As of 1978, there were fewer than 20 successful repairs reported in the English literature. Only in the past 20 years has diagnosis and repair of these injuries been successful.
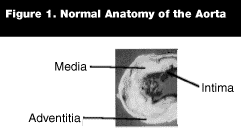
Definition. Throughout the medical literature, some of the terms used for blunt traumatic injury of the thoracic aorta are pathologically incorrect and can be clinically misleading. Descriptions of traumatic transections, dissections, and rupture are vague, ill-defined, and often confusing to the clinician. (See Figure 1 for normal anatomy of the aorta.)
The pathologic terminology is as follows:
Aortic Traumatic Rupture. A through and through complete, full thickness disruption of the wall, including intima, muscularis, and adventitia.
Aortic Traumatic Tear. Partial disruption of the aortic wall limited to the intima and muscularis. The adventitia is intact. The integrity and flow of the aortic lumen is maintained by the intact adventitia, which provides approximately 60% of the tensile strength of the aorta.
Aortic Traumatic Intimal Tear. Disruption is limited to the intima. The muscularis and adventitia are intact.
As the clinician cannot determine which injury is present on clinical grounds, the generic term TAI should be used until the injury is fully defined.
Most of the patients that we see in the ED have partial thickness injuries. The aorta is not "ruptured," but its wall is incompletely torn, with the adventitia intact.6 The natural history of acute TAI is dramatic. Of patients suffering a complete tear of the aorta, 80-85% exsanguinate at the scene of the injury or shortly after, while 15-20% arrive to the ED alive. Of these, 49% exsanguinate within the first 24 hours, 80% by the end of the first week, and 95% by four months after the injury. The remaining 2-5% are long-term survivors who may live a normal live span with a false aneurysm and ultimately die from an unrelated cause.2
Pathophysiology. Mechanism. In penetrating injury to the aorta, the mechanism is obviously direct trauma to the aorta. The partial thickness injuries often seen in blunt trauma are much less common with penetrating injuries. In blunt TAI almost any vertical deceleration impact has been described as directly responsible for aortic and other major vessel injuries: vehicular accidents, direct kicks from animals, crush injuries, falls from significant height, and air bag deployment.5,6,13,14 There are several theories of the mechanics of this injury:
• Sudden deceleration forces create a whiplash effect and result in a partial or complete tear of the vessel. The aorta lies mostly free in the chest, but the descending aorta is fixed at the ligamentum arteriosum and at the diaphragm. The pulmonary vessels are attached to the atria. Shear forces are created by the relative mobility of a portion of the vessel adjacent to a fixed portion. This causes flexion of the vessel and stress at the site of attachment, which may lead to tearing of the vessel.
• Compression of vessels caused by inferior and posterior rotation of anterior thoracic bony structures (manubrium, first rib, clavicles), cause pinching and shearing of the interposed aorta as they strike the vertebral column.
• The intraluminal pressure tolerance of a healthy adult aorta is estimated to be about 50 mmHg. This can be exceeded in a high velocity impact. A sudden increase in intra-aortic pressure, called "the water-hammer pulse" is thought to be one reason why aortic and other cardiac injuries occur.
A combination of these mechanisms likely accounts for most of the injuries. It is essential to recognize these mechanisms, as they frequently represent the only clue to the possibility of an "occult" major vessel injury.
One prospective study evaluated the mechanism of injury in 1561 patients with aortic injury over a 5.5-year period. The only statistically significant risk factor for blunt aortic injury was a high-speed motor vehicle accident (> 60 mph). No significant association was found between frontal and side impacts, ejection, associated fatalities, sudden deceleration, automobile destruction and aortic injury.15 Interestingly, of the 116 patients who had "fall" as their mechanism, only two had blunt TAI and not one had blunt TAI after a fall of fewer than 10 feet.
Location. Of all blunt TAI, 54-65% involve the proximal descending aorta, 10-14% the ascending aorta or arch of the aorta, 12% the distal aorta, and 13-18% involve multiple sites.6 The incidence of other major vessel injuries is unknown, but they are not unusual. They frequently are reported in the literature, especially in penetrating trauma, but also in motor vehicle collisions, falls from significant heights, and as a result of iatrogenic diagnostic or therapeutic maneuvers. One series of blunt injuries to the chest reported a ratio of one tear of the branch of the aortic arch for every four aortic injuries.9
Patients who sustain ascending aortic tears have a 70-80% incidence of associated lethal injuries, such as pericardial tamponade, aortic valve tear, and coronary artery injuries. Most of these patients die instantly or before arrival to the ED.
Prehospital Management and Transport. While prehospital trauma life support protocols do not change major vessel injury, there are particular management issues that need to be addressed. Military antishock trousers application in patients with these injuries significantly increases the chance of death during transport and hospitalization in both pediatric and adult populations.16 This device elevates blood pressure by increasing afterload and is the equivalent to placing a cross clamp distal to the potential injury—clearly a counter productive maneuver. Injuries to the great vessels should be treated as a "load and go" situation by emergency medical services (EMS). There is no effective "in field" therapy for these injuries.
Traditionally, all trauma victims have been treated with large volume fluid resuscitation with the goal of increasing blood pressure to normal or supranormal levels. A 1994 study published in the New England Journal of Medicine found that immediate crystalloid resuscitation increased the incidence of mortality and adult respiratory distress after penetrating torso trauma.17 This has lead some to advocate fluid restriction after penetrating trauma to permit moderate hypotension (systolic blood pressure of 60-90 mmHg). The goal is to avoid blood pressure increase and to prevent disruption of any protective soft perivascular clot that has formed. It is difficult to recommend "permissive hypotension" currently. The study that found it to be beneficial has not been replicated. While it is reasonable that excessive fluid administration could dislodge a forming clot, it is hard to imagine how hypotension could benefit a patient with cardiac tamponade. For now, the best interpretation of this interesting research is to emphasize the need to get the patient definitive care as soon as possible and to avoid excessive fluid administration.
Emergency Center Evaluation. All patients should be examined and managed following strict advanced trauma life support (ATLS) protocols.
History. Unfortunately, history is not always reliable. Patients frequently are intoxicated or have altered sensorium secondary to their injuries. Witnesses are not available, and family members usually are "in route." EMS personnel are essential and many times are the only source of information about the injured patient. EMS personnel can obtain history not only from bystanders, police, and first responders but also from the patient before intubation. Specific information that should be gathered includes severity of the impact, time of extrication, intrusion into the vehicle, vital signs, Glasgow Coma Scale (GCS) score, amount of blood loss, and stability during transport. In penetrating trauma, the length of the knife and firearm type, number of rounds fired, and the patient’s distance from the firearm all are important.
When he or she is able to give a history, the patient may complain of chest pain, interscapular pain, or difficulty breathing. Unfortunately, the most common symptom, interscapular or retrosternal chest pain, is only present in 25% of cases of blunt aortic injury.18 Uncommon indicators of major vessel injury include hoarseness caused by compression of the laryngeal nerve, dysphagia caused by esophageal compression, and extremity pain caused by low flow ischemia.
Physical Exam. A potential pitfall, even for the most seasoned clinician, is to disregard the potential of aortic injury based on the patient’s general appearance, vital signs, or lack of chest wall trauma. These clinical indicators are totally unreliable. If found, pulse deficits, abnormal vital signs, chest wall trauma, and interscapular pain increase the suspicion of aortic injury, but the absence of such clues does not exclude aortic injury. Up to 50% of the patients with proven blunt TAI may have no external physical sign of injury.18 There are some physical findings that increase the suspicion of blunt aortic injury. (See Table 1.) When present, a fast and effective inclusion-exclusion of great vessel injury is necessary. Every patient with a significant mechanism of injury should be thought of as having an aortic injury until proven otherwise.
| Table 1. Physical Findings that Increase Suspicion of Blunt Aortic Injury | ||
| • | Hypotension: | Up to 50% of patients with proven aortic rupture present with hypotension. |
| • | Hypertension: | Up to 72% of patients may be hypertensive before fluid or vasoactive drug administration. The hypertension may result from a stretching stimulus of the sympathetic afferent nerve fibers, located in the area of the aortic isthmus. |
| • | Pseudocoarctation syndrome: |
Acute onset of upper extremity hypertension, along with absent or diminished femoral pulses. Reported to occur in up to one third of the patients and thought to occur secondary to the compression of the aortic lumen by the periaortic hematoma. |
| • | Expanding hematoma: |
At the thoracic outlet. |
| • | Interscapular murmur: |
Reported in up to 31% of the patients. |
| • | Palpable fracture: | Of the sternum. |
| • | Palpable fracture: | Of the thoracic spine. |
| • | Hemothorax: | Chest tube with an initial "rush" of more than 1500 mL or more 200-300 mL/h is suggestive of a major vessel injury and an indication for urgent thoracotomy. |
|
|
||
Diagnostic Studies. In the last five years, the area of diagnosis of blunt TAI has been the purpose of many studies. Several have attempted to clarify what a normal chest radiograph (CXR) means in the setting of a possible TAI. There have been significant advances in the diagnosis of TAI by CT and transesophageal echocardiography (TEE). The vast majority of patients with penetrating great vessel injury will be unstable or have indications for surgical exploration. The following brief discussion applies to suspected blunt TAI except where specifically noted. A more thorough discussion appears in the first of this series of articles. (See Emergency Medicine Reports Aug. 27, 2001 [2001;22:201-210].)
Chest Radiograph. The most valuable initial screening tool for chest trauma is the CXR. Upon the patient’s arrival, a supine anterioposterior 36-inch CXR should be performed. For penetrating injuries, it is important to place radiopaque markers to identify the entrance and exit sites. Radiographic evidence of major vessel injury in penetrating trauma includes:
• large hemothorax;
• foreign bodies near or in the trajectory of great vessels; and
• "missing" missile in a patient with a gunshot wound to the chest, suggesting distal embolization in the aortic tree.
For blunt trauma, a standard CXR as described before and possibly a mediastinal view x-ray with thoracic spine technique are used to more closely evaluate the mediastinal structures. The CXR is a good screening tool when coupled with knowledge of mechanism of injury and physical exam.19 The absence of mediastinal abnormalities has been reported to rule out blunt TAI.15,19,20 More conservative reports warn that 2-7% of blunt TAI patients have a normal mediastinum on CXR.5,21-23 One author analyzed 52 articles identifying the radiological findings in 656 patients and found that 7.3% of patients had a normal mediastinum.
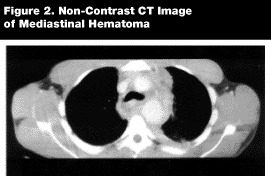
On the CXR, physicians should look for signs of mediastinal hematoma: wide mediastinum, depression of the left mainstem bronchus, extension of the left paraspinal stripe above the aortic arch, widening of the right paratracheal stripe, apical pleural cap, and obscured aortic knob. Mediastinal hematoma can be caused by aortic bleeding, but most commonly it is not of aortic origin. It is more often due to tears of other mediastinal vessels (e.g., the azygous and hemiazygos, spinal, intercostal, and mammary arteries and veins).9 The most common cause of mediastinal hematoma is spinal fracture (21.4%).5 Specificity of an abnormal mediastinum is only 10%. However, CXR showing an abnormal mediastinum is the clue to a major vessel injury and should be thought of as the source of bleeding until proven otherwise. The absence of a mediastinal hematoma has been reported to have a negative predictive value of 98%.33 (See Figure 2.)
Chest Computed Tomography Scan. CT evaluation of the thoracic aorta has been possible since the advent of this technology in the late 1970s. By the 1980s, improvements of CT technology and increased availability resulted in widespread application for the evaluation of aortic pathology. One of the most critical points when analyzing literature in CT diagnosis of blunt TAI is to pay close attention to the technology used. Spiral CT in the early 1990s gave birth to CT angiography and redefined the role of CT for imaging of the thoracic aorta. Unlike conventional CT, which obtains images sequentially in a stepwise fashion, spiral CT scanners have the ability to image the entire aorta in a single, volumetric breath-hold scan.24 When coupled with the administration of 120-150 mL of contrast at 3-4 mL/sec administered via power injectors, this technique is referred to as "CT angiography" because the images can be manipulated to mimic the appearance of a conventional angiogram.
A normal spiral CT virtually excludes TAI. A recent prospective study of 112 patients found that spiral CT had excellent results for detecting all aortic injuries. All CT scans were confirmed by aortography or surgery. The authors recommended that "all trauma patients with high risk-deceleration injuries undergo routine helical CT irrespective of the CXR findings."5 Mirvis reported a sensitivity of 97% and specificity of 99.8% for aortic injury, and sensitivity of 99.3% and specificity of 87.1% for mediastinal hemorrhage.25 Garant reported that helical CT was more sensitive than aortography (100% vs 94.4%).26 Fabian et al documented sensitivity and specificity of 100% and 83%, respectively, for spiral CT; and 92% and 99%, respectively, for conventional angiography.27
All these recent data have been very exciting and have seriously challenged conventional aortography as the gold standard in blunt TAI. It is likely that helical CT will replace aortography in the majority of the trauma centers, with angiography reserved only for indeterminate scans or for patients undergoing angiography for other reasons (e.g., treatment of hemorrhage from pelvic fracture.)
CT scan also has been used for the evaluation of penetrating trauma to the torso. In the stable patient with a gunshot wound to the torso, CT scan has been used to determine the trajectory of the bullet in an effort to reduce exploratory surgery. This approach is limited in scope, however. In the one study evaluating this approach, of 2123 torso gunshot wound patients evaluated, only 50 were stable enough to undergo CT scan.28 Centers seeing small numbers of torso gunshot wound patients may be better off using standard operating room exploration of these wounds.
Catheter Arteriography. In penetrating trauma, catheter angiography is indicated for suspected innominate, carotid, or subclavian arterial injuries. This study is essential for localizing the injury and planning the appropriate incision. Proximity of a missile trajectory to the brachiocephalic vessels, even without physical finding of vascular injury, is an indication for arteriography.16 Aortography also may be useful in hemodynamically stable patients with suspected penetrating aortic injuries, but has several limitations. A "negative" aortogram may represent a vessel that has "sealed off" or may indicate the column of aortic contrast overlies a small area of extravasation.
In blunt trauma, aortography is still largely considered the "gold standard" for aortic injury. Aortography provides information about brachiocephalic injuries that might be missed by other techniques.9 There are obvious drawbacks to aortography: invasiveness, cost, iodinated-dye exposure, and the need to transfer patients out of the trauma suite, as well as time to do the test. In one study, time was measured from the time of arrival to the angiography suite until time to thoracotomy. The average time was a disappointing 90 minutes. This is especially critical in light of the 12.5-20% mortality in the first six hours after admission that has been reported with this injury.29
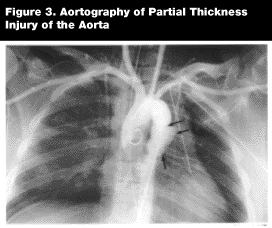
The advantage of aortography over TEE and spiral CT is that it localizes the injury precisely, even in the case of multiple aortic tears. (See Figure 3.) It also is more accurate in the diagnosis of other great vessel injuries. In a recent study of 72 patients with aortic rupture, 1% had an injury to the distal aorta and 19% had injuries to other aortic arch branches. The authors conclude that if TEE had been the only test, these injuries would have been missed.30 The sensitivity and specificity of aortography is 73-100% and 99%, respectively.31 Fabian reported a sensitivity of 92% and specificity of 99%.21 Another potential advantage of aortography is the ability to perform simultaneous coronary angiography, often indicated in patients with lesions in the ascending aorta or suspected atherosclerotic disease. This adjunctive diagnosis may give the patient the opportunity to have coronary artery bypass grafting together with a surgical repair of the aortic rupture.
Intravenous digital subtraction angiography (IVDA) is an exciting and relatively new modality that has been the diagnostic study of choice in many centers in recent years; it can be performed in 15-30 minutes, is less time consuming, is less expensive, utilizes less contrast, and its accuracy is comparable to conventional angiography.32
Transesophageal Echocardiography (TEE). TEE is an excellent screening tool for blunt TAI and can be the only test used to determine that a patient requires surgery. TEE has the advantage of being a bedside test, does not require contrast, is faster than aortography, gives information on other cardiovascular structures, and is obviously less invasive. In a prospective, nonrandomized study of 160 patients suspected of having blunt TAI, TEE had a sensitivity and specificity of 100%, and aortography had a sensitivity of 73% and a specificity of 99%. However, three (2.5%) of the TEE studies were read as equivocal and the patients required subsequent aortography.33 In another study, 93 patients underwent TEE followed by aortography. The sensitivity and specificity of TEE was 100% and 98%, respectively.34 Another prospective study of 134 patients demonstrated a sensitivity of 93% and specificity of 98%. Patients undergoing procedures solely based on TEE had a significantly shorter (30 min vs 71 min) time to surgery than those who had angiography as the sole diagnostic test. TEE also detected pericardial effusion, valvular disruption, and blunt myocardial injury.35,36 There is excitement about this test, but its utility is limited by the experience of the operator. It is contraindicated in patients with cervical and esophageal injuries and is limited in the evaluation of other aortic branch injuries. Nasogastric tubes can result in an artifact.37
The medical literature states that, at the present time, there is no clear algorithm that seems to be followed by the major, academic trauma centers.5,16,19,21,38 There are centers that exclude aortic injury based on their data that state: "the lack of mediastinal hematoma excludes aortic injury."19,20 They use a normal mediastinum on CXR or spiral CT to exclude mediastinal hematoma. Other authors advocate thoracic aortography solely on the basis of a high-velocity, abrupt-deceleration mechanism if the patient is unstable.15,16,23 Some advocate spiral CT in this situation. If the patient is stable with normal mediastinum and physical exam, some authors advocate admission with 4-6 hours interval CXR and again at 24 hours to detect the development of suggestive signs of great vessel injury.16 One study reported a case and found other cases in the literature in which this approach resulted in delayed rupture and death of several patients. In all cases, aortic injury was diagnosed at postmortem examination.23 At the present time, the best approach for the ED clinician is to work in conjunction with their radiology and surgical colleagues to develop protocols according to their experience, capabilities, and resources to diagnose blunt TAI quickly and safely.
Treatment. The most important issue in the ED, other than diagnosis, is to attempt to increase the chances of survival for the patients. Pharmacological treatment can be helpful while operative management is being planned. In a few cases management is nonoperative.
Pharmacological Adjuncts. Pharmacological management is identical to that of management of aortic dissection, as the same principal applies. Blood pressure should be maintained between 100 and 120 mmHg. Beta-blockers can decrease the pulse pressure and minimize shear forces acting on the aorta, therefore protecting the intact adventitia. Esmolol, because of its short-acting, titratable effect, is the preferred agent for this purpose. If needed, nitroprusside can be added to reach the target blood pressure.16,21,27,39,40 Unfortunately, many centers do not practice this routinely. In one multicenter study of blunt TAI, only 17% of the patients had this therapy. Twenty-four patients in the study who were hemodynamically stable died while awaiting thoracotomy.21 In the ED, while waiting for diagnostic studies or operative management, blood pressure control is the only effective therapy.
Surgical Management. There is controversy regarding the optimal operative management of acute thoracic aortic injuries. There are three accepted methods of repair. These include the "clamp and sew," active or passive shunting, and total cardiopulmonary bypass. No single technique is superior.41 The central point of discussion is the method used to protect against spinal cord ischemia. Whatever the surgical technique, it seems that one cannot avoid paraplegia. Despite the hopes that technical maneuvers or monitoring techniques would reduce the incidence of paraplegia, a 5-7% paraplegia rate persists. The reasons for this complication are many: inadequate radicular artery flow, prolonged preoperative and intraoperative hypotension, prolonged aortic cross clamping, intraoperative ligation of intercostal arteries that supply radicular arteries, and anatomical variability of the radicular artery. In a recent multicenter study in France that included 174 aortic isthmus disruptions, the three techniques were compared: Nine patients had paraplegia (5.2%), four of 69 (5.7%) in the "clamp and sew" group, two of 93 (4.3%) in the extracorporeal circulation group, and one of 12 (8.3%) in the shunt group. Overall mortality was 19%.41 The group at Maryland reviewed 192 patients with TAI who had surgical repair within the last 27 years (1971-1998); they divided the groups in four—Group I (1971-75), II (1976-85), III (1986-94), and IV (1994-98). Despite advances in technology and surgical techniques they had no statistically significant improvements in mortality rate over the four periods. There were 22 cases of paraplegia in 167 patients (13%). However, in their last group (IV) of 67 patients, there were no cases of paraplegia. This may be secondary to the use of heparin coated circuit and the centrifugal pump as an adjunct to the repair.38
There also is some controversy regarding primary repair as opposed to graft repair. Recently, most of the literature has been favorable to primary repair if it is feasible, arguing that it is faster; has less potential for infection and suture line dehiscence; and gives similar, if not lower, rates of mortality and postoperative paraplegia. Grafts usually are placed if there is extensive tension on the vessel walls or jaggedly torn ends of the vessel. Direct repair seems to be the preferred method in children, because it obviates the need for reoperation and graft replacement as the child grows.
Formerly, the only treatment was open surgery of the thorax, in which poor prognosis is related to the lesion or other associated disorders. However, in 1994, Dake and colleagues first reported the treatment of a traumatic thoracic aortic aneurysm with a self-expanding endovascular prosthesis.42 The procedure has to be done under general or spinal anesthesia, with cardiovascular surgery and immediate bypass available for failed stenting. A percutaneous left brachial artery pigtail catheter is inserted over a guidewire, which is used for the intraoperative aortogram. Then the femoral or iliac artery is used for introduction of the stent graft system. Exact placement of the graft system is determined by angiography; when in the appropriate position the sheath is removed to deploy the stent graft. The spring stent expands and conforms the anatomy of the normal aorta, then a balloon is blown to keep enough pressure against the aortic wall and a repeat arteriogram is used to verify placement, free perfusion of the stent graft, and lack of leakage. Recently there have been reports of successful acute traumatic rupture repairs with this innovative technique.43 Safety and feasibility of repair of thoracic aortic aneurysms also is recently being studied with acceptable results.44 Endovascular treatment seems to be a viable alternative to surgery, though its long-term validity remains to be confirmed.
Nonoperative Management. Nonoperative management of blunt tears of the thoracic aorta has been reported with increasing frequency since 1981, especially in extremely sick patients who might not survive surgery.16,45 This approach is for patients who have a high likelihood of death if taken for immediate repair or with associated injuries that are uniformly fatal. This is never a decision made by the emergency physician. It is mainly based on a small number of case series. Galli and associates studied 42 patients with TAI; 21 patients had immediate repair with a 19% mortality rate, compared with no deaths in 21 patients who had an operation after intensive medical therapy and treatment of their associated injuries.46 Patients with conditions listed in Table 2 have been reported to benefit from non-surgical management.
| Table 2. Indications for Conservative Management of Traumatic Aortic Injury | |
| • | Severe injury to the central nervous system (survival limited or poor quality of life) |
| • | Major burns with a high risk of infection |
| • | Sepsis |
| • | Contaminated open wounds with high risk of infection |
| • | Severe respiratory insufficiency |
| • | Hemodynamic instability and metabolic failure |
| • | Presence of a " nonthreatening lesion" (an intimal defect alone) |
| • | False aneurysm |
The Injured Heart
Penetrating trauma of the heart is largely an occurrence of metropolitan violence, caused by long knives, ice picks, stilettos, and gunshot wounds. It is rarely caused by accidents or by iatrogenic misadventures from central venous catheter placements, percutaneous dilatation of coronary arteries, or Swan-Ganz placements. Injuries caused by penetrating trauma to the heart are a major cause of death in the young.
Penetrating Trauma. History and Background. In 1883, Theodore Billroth, obviously pessimistic of successful surgical repair of the heart, wrote, "The surgeon who should attempt to suture a wound to the heart would lose the respect of his colleagues." Thirteen years later, in 1896, Rehn carried out the first successful human cardiorrhaphy. Hill performed the first in the United States on a 13-year-old boy on an kitchen table in Alabama in 1902. By 1959, Isaccs analyzed 60 cases of cardiac injuries and found a remarkable survival of 89% for stab wounds and 43% for gunshot wounds. Today, repair of penetrating cardiac wounds is routine in most major trauma centers of this country and of the rest of the world.47
Location. The distribution of these injuries over a 20-year experience was published by Wall and colleagues and included 711 injuries. Forty percent were to the right ventricle, 40% to the left, 24% to the right atrium, and 3% to the left. In one-third of the injuries, multiple structures will be involved. In 5% of the cases, a laceration to the coronary arteries occurs, which is rarely proximal and can be ligated without coronary artery bypass. Rarely, injuries involve intracardiac structures such as papillary muscles, valves, or septum. Coronary artery injuries may produce a rapid cardiac tamponade or may present as coronary occlusion and myocardial insufficiency.
Clinical Features in Pathophysiology. Generally, patients will present with symptoms of pericardial tamponade or hemorrhagic shock. The presentation is determined by the size of the penetration, rate of bleeding, and the chamber involved, as well as the patient’s ability to compensate. Stab wound patients present with cardiac tamponade in 80% of cases with or without shock. The mechanism appears to be that after a sharp object penetrates the pericardium and the myocardial wall, the pericardium is sealed by a clot or adjacent fat. The egress of blood into the pericardium as low as 60-100 mL will impede ventricular filling, reduce the stoke volume, and increase catecholamine output. The resultant increase in right heart filling shifts the septum to the left, and further decreases left ventricular filling, creating a lethal cycle of worsening shock. One key component to keep in mind, especially when deciding on mode of transportation to another facility, is that the progression from a compensated to an uncompensated tamponade may be sudden and profound.
In contrast to stab wounds, gunshot wounds are often large injuries and are incapable of sealing themselves. Therefore they often present with significant hemodynamic compromise. Only 20% present with tamponade. Small perforations can be sealed by the left ventricular wall, and to a lesser extent, the right ventricle and atria. The ability to seal a perforation is proportionate to the wall’s muscle mass.
Diagnosis. The classic findings of tamponade with Beck’s triad of reduced blood pressure, muffled heart tones, and distended neck veins are present in fewer than 10% of the cases. The triad is not specific and also may result from tension pneumothorax. The findings of pulsus paradoxus or Kussmaul’s sign are unreliable for diagnosing tamponade. Any evidence of an entry wound in the epigastrium or the precordium within 3 cm of the sternum carries a risk for penetrating injury to the heart. High-velocity gunshot wounds not in this area can ricochet and injure the heart and other structures. Patients present to the ED stable, in shock, or in full arrest. Most gunshot wound patients present in profound shock or cardiac arrest secondary to severe hemorrhage.
Pericardiocentesis has a limited role in the diagnosis of pericardial tamponade because false positives and false negatives are common. Patients who present with a penetrating precordial or epigastric wounds without hemodynamic compromise are particularly challenging, as they may have an occult injury to the heart and can deteriorate quickly. Aaland and Sherman reviewed the literature and reported eight cases of delayed pericardial tamponade (3-76 days) after knife wounds.48
Subxiphoid Pericardial Window. One option for making the diagnosis of occult cardiac injury is subxiphoid pericardial window. This procedure, described in the surgical literature, is performed by making a vertical midline incision over the xiphoid process and upper epigastrium, elevating and excising the xiphoid, and incising the pericardial sac. If there is hemorrhage/injury present through the pericardial window, the incision is extended to a median sternotomy for cardiorrhaphy. The recent enthusiasm with ultrasonography in the initial evaluation of the trauma patient has diminished the role of the subxiphoid window.
Two-Dimensional Echocardiography. This is a rapid, noninvasive, low cost, and accurate method of detecting occult cardiac injury. In a prospective study, 2-D echocardiography was found to have 96% accuracy, 97% specificity, and 90% sensitivity for predicting cardiac injury. This study suggested that in the absence of technicians who can perform the study, four hours of training in ultrasonography may be sufficient to identify pericardial fluid. Eighty percent of the ED physicians performed echocardiography without missing the diagnosis.49 Another prospective study reported that the accuracy diminished in patients with hemothorax, with a sensitivity of 56%, specificity of 93%, and accuracy of 90%; in the absence of hemothorax, accuracy was the same as that reported with subxiphoid pericardial window.50 Rozycki and colleagues reported a sensitivity of 100% and a specificity of 97.3% for pericardial fluid using the focused assessment for the sonographic examination of the trauma victim (FAST).50 The increasing use of FAST supports the role of sonography as the diagnostic modality of choice for detecting occult hemopericardium, but has yet to be evaluated in large multicenter studies.
Prehospital Management. Prompt transport to the emergency center without attempts of stabilization in the field has been shown to improve outcome in penetrating cardiac trauma. The only procedure or prognostic factor that has proven to be beneficial in increasing survival rates is endotracheal intubation and CPR for fewer than 5 minutes.52
Emergency Department Management. Management of such patients follows ATLS guidelines. After the primary survey, fully examine entrance and exit wounds and other factors contributing to the patient’s condition. In the hemodynamically compromised patient, the operating room should be notified immediately upon arrival of the patient to the ED. Most patients receive a central venous line to monitor CVP and to have a large bore access site for fluid administration.
The indications for ED thoracotomy are profound shock without response to fluid administration, patients who are agonal and impending arrest upon arrival, and loss of vital signs in transit. If the hypotensive patient responds to fluids, thoracotomy is withheld and the patient should immediately be taken to the operating room.53
The definitive treatment of cardiac injuries is cardiorrhaphy through a thoracotomy or sternotomy. This should be accomplished as soon as possible and, ideally, in the operating room.
Outcomes. Several reports with large numbers of patients have survival rates in penetrating injuries that range from 31% to 90%. Collected series have stratified survival rates at 0%, 30%, 40%, and 50% for patients presenting to the ED dead, in arrest, in an agonal state, and in profound state of shock, respectively.54 Stab wounds have a better prognosis than gunshot wounds. Stab wounds to the heart have approximately a 70-80% survival rate, and gunshot wounds 30-40%.53
Blunt Cardiac Trauma
Blunt trauma to the heart ranges from minor injury to frank rupture. The minor injury is a nonspecific condition variously termed cardiac contusion or myocardial concussion. More specific abnormalities that can be demonstrated and diagnosed consistently are injuries to the pericardium, cardiac valves, papillary muscles, chordae tendinae, septal wall, and coronary vessels. The extreme end of the spectrum is the often-fatal cardiac rupture.
The frequency and prognostic value of minor cardiac injury in blunt chest trauma is controversial. This entity appears to be the least important of the diagnoses in the spectrum of blunt cardiac injury.47 The most important thing seems to be that when cardiac abnormalities do occur, they need to be treated with or without the diagnosis of myocardial contusion, and that in the absence of these abnormalities, a diagnosis of myocardial contusion is irrelevant.
Significant cardiac events are rare in young patients with anterior chest wall concussion, which represent the majority of patients evaluated in trauma. Cardiac markers, echocardiography, cardiac scintigraphy, and gated blood pool scanning have all been studied, and none of them were highly sensitive or specific for this purpose. When the diagnosis is made in these young patients, the outcome was unrelated to abnormal tests. Cardiac morbidity is rare in young patients with initial ECG abnormalities. Initial ECG abnormalities are better indicators of cardiac complications in the elderly, in patients with preexisting cardiac pathology, and in critically ill patients, irrespective of the diagnosis.
Based on this discussion, it is recommended that asymptomatic patients with anterior chest wall concussion should not be admitted to intensive care units, and that no further work up should be done. Elderly patients or patients with preexisting cardiac disease should be monitored, but no further testing should be done unless they become symptomatic. Symptomatic patients should be admitted to the ICU for an evidence-based work up that seeks specific injuries.53
Injury to the Pericardium. Isolated pericardial disruption occurs in 29% of blunt traumatic cardiac injuries. As an isolated injury, the importance lies in the fact that the heart can herniate partially through a disruption and cause cardiovascular compromise and death. The pericardium can tear from direct impact or from increase intra-abdominal pressure. Disruptions occur more commonly in the left side and may range from small to larger tears the entire length of the pericardium. Clinical exam may reveal pericardial rub; chest x-ray may suggest the diagnosis by demonstrating pneumopericardium or displacement of the heart. These findings require surgical consultation.
Injury to Endocardium and Septum. Nonpenetrating injury to these structures is uncommon, but not rare. Rupture of the valves is the most common, especially in patients with preexisting disease of these structures. One study reported 546 cases of blunt cardiac trauma and reported a 9% incidence of injury to the valves. The aortic, mitral, and tricuspid valves, respectively, were involved.54 The injury should be suspected in patients that present with chest pain, respiratory difficulty, and a new murmur. Echocardiography or cardiac catheterization are the studies of choice. Treatment can be medical in minor defects, as some of them will close; major defects with significant shunts usually are corrected by surgery.
Injury to the Coronary Vessels. Coronary thrombosis may result from the intimal injury of blunt cardiac trauma. It usually is indistinguishable from atherosclerotic myocardial infarction and is treated as such. Prognosis usually is better, but formation of a ventricular aneurysm can be a late complication that leads to rupture, cardiac failure, embolism, or arrhythmias. The diagnosis is made by cardiac catheterization, and an operative approach usually is necessary to correct the lesion.
Cardiac Rupture. Blunt cardiac rupture accounts for 5% of motor vehicle accident (MVA) fatalities. It is documented in 15% of MVA deaths.54,56 Before the 1900s, the literature on this entity was limited to autopsy reports. In 1935, Bright and Beck made the observation that of 152 patients dying from cardiac rupture from blunt trauma, 30 patients lived 30 minutes or longer.56 Desforges reported the first successful repair of a ruptured right atrium in the United States in 1955. By 1986, Calhoon reported a 70% survival rate in patients with blunt rupture to the heart.
The mechanism for rupture of the heart is essentially the same as those for TAI, as described previously. Actually, 20% of these patients also have simultaneous aortic rupture.55
Fifty percent or more of patients with this injury present with no vital signs to the ED and have minimal survival. The presenting symptoms are those of cardiac tamponade or severe hemorrhage. In patients who present stable, delay in diagnosis is common. As in penetrating trauma to the heart, no clinical features are reliably diagnostic and there must be a very high index of suspicion. A precordial murmur that sounds like a splashing millwheel may be heard but is rare. ECG may reveal bundle branch block, or if herniation occurs, axis deviation. Ultrasound may confirm pericardial fluid. The treatment of these injuries is essentially the same as treatments previously described for penetrating trauma to the heart.
Penetrating and blunt injuries to the heart are seen more and more frequently. Diagnosis is difficult and requires a high index of suspicion—the reward for which is a remarkable survival rate even in patients who present in extremis.53
References
1. Trupka A, Waydhas C, Hallfeldt KJ, et al. Value of thoracic computed tomography in the first assessment of severely injured patients with blunt chest trauma: Results of a prospective study. J Trauma 1997;43:405-412.
2. Smith R, Chang F. Traumatic rupture of the aorta: Still a lethal injury. Am J Surg 1988;152:660-663.
3. LoCiero J, Mattox KL. Epidemiology of chest trauma. Surg Clin North Am 1989;69:15.
4. Williams JS, Graff JA, Uku JM, et al. Aortic injury in vehicular trauma. Ann Thorac Surg 1994;57:726-730.
5. Demetriades D, Gomez H, Velmahos GC, et al. Routine helical computed tomographic evaluation of the mediastinum in high risk blunt trauma patients. Arch Surg 1998;133:1084-1088.
6. Katyal D, McLellan BA, Brenneman FD, et al. Lateral impact motor vehicle collisions: Significant cause of blunt traumatic rupture of the thoracic aorta. J Trauma 1997;42:769-772.
7. Munshi IA, Engelman D. The image of trauma. Thoracic aortic transection secondary to blunt trauma. J Trauma 2000;48: 985.
8. Schmidt CA, Jacobson JG. Thoracic aortic injury: A ten-year experience. Arch Surg 1984;119:1244.
9. Bolton RJ. Blunt aortic transection with aberrant right subclavian artery. J Trauma 1999;47:597-598.
10. Pan D, Moore HG, Angood P, et al. Aortic injury resulting from attempted subclavian central venous catheter placement. J Trauma 1999;47:403-405.
11. Sadd AG, Zaidi A, Pugh ND, et al. Iatrogenic mammary arteriovenous fistula caused by a sternal wire. Ann Thorac Surg 1999;68: 625-632.
12. Siersema PD, Tan TG, Sutorius FF, et al. Massive hemorrhage caused by a perforating Gianturco-Z stent resulting in an aortoesophageal fistula. Endoscopy 1997;29:416-420.
13. Reid C, Livesey A, Egleston CV. Aortic rupture as a result of low velocity crush. J Accid Emerg Med 1999;16:299-230.
14. Pasic M, Ewert R, Engel M, et al. Aortic rupture and concomitant transection of the left bronchus after blunt chest trauma. Chest 2000;117:1508-1510.
15. Dyer DS, Moore EE, Ilke DN, et al. Thoracic aortic injury: How predictive is mechanism and is chest computed tomography a reliable screening tool? A prospective study of 1561 patients. J Trauma 2000;48:673-683.
16. Mattox KL, Wall MJ, LeMaire SA. Injury to the Thoracic Great Vessels. In: Trauma. 4th ed. Old Tappan, NJ: Appleton & Lange; 2000:559-582.
17. Bickell WH, Wall MJ Jr, Pepe PE, et al. Immediate versus delayed fluid resuscitation for hypotensive patients with penetrating torso injuries. N Engl J Med 1994;331:1105-1109.
18. Markovchick V, Wolfe R. Cardiovascular Trauma. In: Rosen P, et al. Emergency Medicine: Concepts and Clinical Practice. 4th ed. St. Louis, MO: Mosby; 1998.
19. Harris JH. Acute traumatic aortic injury. Chest. In: Harris JH, Harris WH. The Radiology of Emergency Medicine. 4th ed. Philadelphia: Lippincot Williams & Wilkins; 2000:496-527.
20. Lee J, Harris JH, Duke J, et al. Noncorrelation between thoracic skeletal injuries and acute traumatic aortic tear. J Trauma 1997;43: 400-404.
21. Fabian TC, Richardson JD, Croce MA, et al. Prospective study of blunt aortic injury: Multicenter trial of the American Association for the Surgery of Trauma. J Trauma 1997;42:374-383.
22. Woodring JH. The normal mediastinum in blunt traumatic rupture of the thoracic aorta and brachiocephalic arteries. J Emerg Med 1990;8:467-476.
23. Savitt D. Traumatic aortic rupture: Delayed presentation with a normal chest radiograph. Am J Emerg Med 1999;17:285-287.
24. Kalender WA, Seissler W, Klotz E, et al. Spiral volumetric CT with single breath hold technique, continuous transport, and continuous scanner rotation. Radiology 1990;176:181-183.
25. Mirvis SE, Shanmuganathan K, Miller BH, et al. Traumatic aortic injury: Diagnosis with contrast enhanced thoracic CT—five-year experience at a major trauma center. Radiology 1996;200: 413-422.
26. Garant ML, Menke PG, Fabian T, et al. Blunt traumatic aortic rupture: Detection with helical CT of the chest. Radiology 1995;197: 125-133.
27. Fabian TC, Davis KA, Gavant ML, et al. Prospective study of blunt aortic injury: Helical CT is diagnostic and antihypertensive therapy reduces rupture. Ann Surg 1998;227:666-676.
28. Grossman MD, May AK, Schwab CW, et al. Determining anatomic injury in selected torso gunshot wounds. J Trauma 1998; 446-456.
29. Feliciano DV. Trauma to the aorta and major vessels. Chest Surg Clin N Am 1997;7:305-322.
30. Ahrar K, Smith D, Bansal RC, et al. Angiography in blunt thoracic aortic injury. J Trauma 1997;42:665.
31. Soyer R, Burnett A, Piwnica A, et al. Traumatic rupture of the thoracic aorta with reference to 34 operated cases. J Cardiovasc Surg 1980;21:295-302.
32. Mirvis SE, Pais SO, Gens DR. Thoracic aortic rupture. Advantages of intraarterial digital subtraction angiography. Am J Roentgenol 1986;146:987.
33. Sturn JT, Hankins DG, Young G. Thoracic aortography following blunt chest trauma. Am J Emerg Med 1990;8:92.
34. Buckmaster MJ, Kearney PA, Johnson SB, et al. Further experience with transesophageal echocardiography in the evaluation of thoracic aortic injury. J Trauma 1994;37:989.
35. Smith MD, Cassidy JM, Souther S, et al. Transesophageal echocardiography in the diagnosis of traumatic rupture of the aorta. N Engl J Med 1995;332:356-362.
36. Leonard G, Wayne IO. Repair of traumatic aortic valve disruption and descending aortic transection. Ann Thorac Surg 2000;69: 1251-1253.
37. Urban BA, Bluemke DA, Johnson KM, et al. Imaging of thoracic aortic disease. Cardiol Clin 1999;17:659-682.
38. Attar S, Cardarelli MG, Downing SW, et al. Traumatic aortic rupture: Recent outcome with regard to neurologic deficit. Ann Thorac Surg 1999;67:959-965.
39. Pate JW, Fabian TC, Walker W. Traumatic rupture of the aortic isthmus: An emergency? World J Surg 1995;19:119.
40. Hilgenberg AD, Logan DL, Akins CW, et al. Blunt injuries of the thoracic aorta. Ann Thorac Surg 1992;53:233.
41. Tatou E, Steinmetz E, Jazayeri S, et al. Surgical outcome of traumatic rupture of the thoracic aorta. Ann Thorac Surg 2000;69:70-73.
42. Dake MD, Miller DC, Semba CP, et al. Transluminal placement of endovascular stent grafts for the treatment of descending thoracic aortic aneurysm. N Engl J Med 1994;331:1729-1734.
43. Brunix G, Wery D, Dubois E, et al. Emergency endovascular treatment of an acute traumatic rupture of the thoracic aorta complicated by a distal low-flow syndrome. Cardiovasc Intervent Radiol 1999; 22:515-518.
44. Grabenwoger M, Hutschala D, Ehrlich MP, et al. Thoracic aortic aneurysms; Treatment with endovascular self-expandable stent grafts. Ann Thorac Surg 2000;69:441-445.
45. Maggisano R, Nathems A, Alexandrova NA, et al. Traumatic rupture of the thoracic aorta: Should one always repair immediately? Ann Vasc Surg 1995;9:44-52.
46. Galli R, Pacini D, Di bartolomeo R, et al. Surgical indications and timing of repair of traumatic rupture of the thoracic aorta. Ann Thorac Surg 1998;65:461-464.
47. Ivatury RR. The injured heart. In: Mattox KL, Feliciano DV, Moore EE. Trauma. 4th ed. New York, NY: Appleton and Lange; 2000: 545-556.
48. Aaland MO, Sherman RT. Delayed pericardial tamponade in penetrating chest trauma: Case report. J Trauma 1991; 31:1563.
49. Jimenez E, Martin M, Krukenkamp I. Subxiphoid pericardiotomy versus echocardiography: A prospective evaluation of the diagnosis of occult cardiac injury. Surgery 1990;108:676.
50. Meyer DM, Jessen ME, Grayburn PA. Use of echocardiography to detect occult cardiac injury after penetrating thoracic trauma. A prospective study. J Trauma 1995;39:902.
51. Rozycki GS, Schmidt JA, Ochsner MG, et al. The role of the surgeon performed ultrasound in patients with possible penetrating cardiac wounds. A prospective multicenter study. J Trauma 1998; 45:190.
52. Durham LA, Richardson RJ, Wall MJ, et al. Emergency center thoracotomy: Impact of prehospital resuscitation. J Trauma 1992; 32:775.
53. Brown B, Grover FL. Trauma to the heart. Chest Surg Clin N Am 1997;7:325-341.
54. Milhalm FH, Grindlinger GA. Survival determinants in patients undergoing emergency room thoracotomy for penetrating chest injury. J Trauma 1986;26:702.
55. Parmley LF, Manion WC, Mattingly TW. Nonpenetrating traumatic injury to the heart. Circulation 1958;18:371.
56. Bright EB, Beck CS. Nonpenetrating wounds of the heart: A critical and experimental study. Am Heart J 1935;10:293-321.
57. Fulda G, Brathwhite CEM, Rodriguez A. Blunt traumatic rupture of the heart and pericardium: A 10 year experience (1979-1989). J Trauma 1991;31:167.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.