Diabetic Foot and Leg Wounds: Principles, Management, and Prevention
Diabetic Foot and Leg Wounds: Principles, Management, and Prevention
Author: Michael B. Strauss, MD, FACS, AAOS, Medical Director, Baromedical Department, Long Beach Memorial Medical Center, Long Beach, Calif; Associate Clinical Professor of Orthopaedics, Harbor/UCLA Medical Center, Torrance, Calif; and Orthopaedic Consultant, PACT (Preservation-Amputation, Care and Treatment Program), Veterans Administration Medical Center, Long Beach, Calif.
Editor’s Note—Foot and leg wounds occur in about 15% of the diabetic mellitus population in the United States. With approximately 15% of the health care dollar spent on diabetes care, foot wound prevention and management have an important role in the management of this patient group. The Diabetes Control and Complications Trial (DCCT) demonstrated that complications of diabetes can be markedly reduced with comprehensive management. This extends to diabetic foot problems also.
The following article by Dr. Michael Strauss provides direction to the primary care physician (PCP) for managing the diabetic foot. Strauss’ experiences (as evidenced by his many citations in the references) provide logical and medically sound advice for the management and prevention of diabetic foot wounds (DFW).
This paper includes several unique features in addition to the currently accepted management recommendations for diabetic foot problems. These include: 1) 3 principles for understanding the management of diabetic foot and leg wounds; 2) a simplified wound scoring system that allows the clinician quickly to ascertain the severity and level of care needed for the patient’s wound; 3) 4 treatment strategies for wound management and what the roles of the PCP are for each strategy; 4) 4 logical approaches to prevent wound recurrences and new DFW problems; 5) a simplified grading and management table for diabetic skin and nail care; and 6) a goal score to assess patient motivation and compliance.
The PCP is the essential link between the diabetic patient and the wound specialist. The PCP will likely be the first physician contact for the patient with a new diabetic foot problem and the "gate-keeper" to ensure proper preventive care measures are followed after a wound is healed. With information presented in this paper, diabetic foot complications can be reduced in a magnitude similar to the reduction in general diabetic complications that resulted from the DCCT research group’s findings.
Introduction
Approximately 16 million patients in the United States have diabetes mellitus.1 As of 1994, it was estimated that almost 15% of the health care dollar (92-105 billion a year) was spent on diabetic care.2,3 Prevention of complications has taken an increasingly important role in medicine and nowhere are the benefits more apparent than in the diabetic.4 In one randomized sample, foot symptoms were observed in approximately two thirds of diabetics.5 Although care of the DFW would seem to be surgical, the prevention and early recognition of the problem will likely be done by the patient’s PCP. Especially important is the recognition of risk factors for DFWs such as deformities, peripheral vascular disease, neuropathy, history of previous DFW, and/or coexistence of an amputation. Likewise optimal medical management is essential to achieve the best possible outcomes for wound and infection problems. This paper discusses principles dealing with DFWs, treatment strategies for managing the wound, and measures to prevent healed wounds from recurring or new wounds from developing.
Principles in Managing Diabetic Foot and Leg Wounds
Three principles provide the basis for understanding the healing of DFWs. Successful management is fundamental to these principles. When followed, they make the management of DFWs logical. For the majority of patients with DFWs, the principles can be achieved through the cooperative efforts of the patients’ PCPs and their surgical, wound team consultants.
Principle I: Oxygen and Blood Flow Requirements for Healing
Markedly increased blood flow and oxygen availability are required to heal a wound and control infection.6,7 Blood supply and oxygen requirements for noncritical tissues such as the foot and leg are low in steady state, resting situations. This concept is apparent in the patient with advanced peripheral vascular disease who develops a relatively minor wound in one foot. Because of inadequate blood flow, the wound does not heal—resulting in a below knee amputation. The other limb survives, although studies demonstrated that perfusion and oxygenation were essentially the same to each lower extremity.
Oxygen and blood flow requirements for wound healing and infection control probably need to increase 20-fold or more. This is based on several observations. The first is the reserve capacity of the vascular tree. An average sized human has a blood volume of approximately 5 L. The estimated potential capacity of the vascular system is at a minimum 120 L.8 This allows for a potential increase in blood flow to noncritical tissues by a factor of 24. The rates of oxygen uptake for neutrophils to kill bacteria increases as much as 50-fold during the respiratory burst.9 Blood flow between rest and maximum exercise increases as much as 25-fold in muscles and oxygen uptake, and cardiac outputs also have large reserve capacities.8 Consequently, there is an enormous reserve capacity for blood flow and oxygen delivery to wounds and infection sites. If this reserve capacity becomes unavailable due to disease or other factors, wound healing will be compromised. The wound becomes labeled as nonhealing. This is one definition for a problem wound. A quantitative method to define the problem wound is presented later.
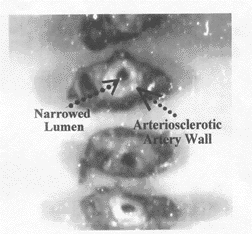
There are several reasons why blood flow and oxygen delivery are unable to increase to the wound site in the patient with DFWs (see Table 1) and thereby lead to failure for the wound to heal. The most important is arteriosclerosis with narrowing of the lumen of the blood vessel (see Figure). Although flow in such vessels may be adequate for steady state situations, it does not have the potential to increase enough to meet the demands for wound healing. Revascularization, angioplasty, and hyperbaric oxygen are interventions used to improve blood flow and oxygen availability. However, as will be discussed, medical interventions are not only effective in improving blood flow and oxygen delivery to the wound but can improve the effectiveness of the above interventions, also.
| Table 1. Impairments to Oxygen Delivery and Blood Flow in Problem Wounds | ||||
| Condition | Pathophysiology | Comments | ||
|
|
||||
| Distal vessel arteriosclerosis | Vessel lumen narrowed | Flow limited to the size of the arteriosclerotic vessel Lumen (see Figure 1). There is no capacity to increase flow by vasodilatation. When localized blockage exists or is in large, more proximal vessels, angioplasty and/or revascularization can increase blood flow. | ||
| Edema | Increased diffusion distance between capillary and cell | Diffusion distance is approximately equal to the square root of the juxta capillary oxygen concentration in the tissue fluid. Hyperbaric oxygen can increase diffusion distance of oxygen 3-fold through tissue fluids.22 | ||
| Congestive heart failure | Impaired circulation; fluid retention | Usually very responsive to medical interventions. | ||
| Capillary basement membrane thickening | Interferes with diffusion of oxygen and nutrients from the capillary to the target cells | A normal phenomenon with aging; basement membrane thickening is accelerated in the diabetic. | ||
| Impaired red blood cell deformability | With RBC aging, hypoxia and sepsis deformability is impaired | The 7.5 micron in diameter RBC must elongate (deform) to pass through the 5.0 micron in diameter capillary where oxygen exchange occurs. | ||
| Auto sympathectomy | Nonresponsive to neuro-impulses; no automatic vasomotor activity | A frequent finding in diabetics. | ||
| Capillary hypoxia; chemical | Injury to the microvasculature distal to sympathetic regulation and arteriosclerosis | Observations: increased thromboxane; increased free radicals; decreased free radical scavengers, increased white blood cell adherence; and decreased nitric oxide. | ||
| Osmotic and glycosylation effects of hyperglycemia | Possibly a cause of increased blood viscosity and impairment | The control of hyperlycemia significantly reduces complications of diabetes (Diabetes Control and Complications Trial).30 | ||
|
|
||||
|
Figure
Cross section (nonmagnified) of medium sized artery and vein removed from an amputated extremity of a diabetic. The cross section’s surface area has been reduced by more than 80% due to arteriosclerosis. Flow through the narrowed lumen was probably adequate for the steady state situation, but insufficient to meet the increased demands for wound healing. Nonhealing of a leg wound was the reason for the amputation. From Poiseuille’s Principle we know that an increase in the diameter of a vessel, increases the flow by the 4th power; that is, if the vessel diameter is doubled, flow would increase 16-fold. It is obvious that the potential to increase flow in this patient’s arteriosclerotic artery no longer existed. |
Principle II: Stages of Healing
Three stages are recognizable in the healing DFWs (see Table 2). Although each stage has characteristic findings, healing is a continuum of responses with one stage transitioning into another. The reason that most problem DFWs do not heal is because of the inability to generate an inflammatory response. The inflammatory response defines the initial phase of healing in the normal, adequately vascularized wound. Oxygen requirements to mobilize and activate fibroblasts and white cells are higher during the inflammatory phase than at any other phases of wound healing.7 When oxygenation availability to the wound is inadequate, the inflammatory stage is thwarted. The wound fails to show signs of healing, and the limb is at risk for amputation. With appropriate interventions, approximately 80% of problem wounds will heal.10 The wounds evolve through 3 stages.
|
Table 2. Stages in the Healing of Problem Wounds |
|||||
| Stage | Clinical Findings |
Interventions | Goals | Duration | Comments |
|
|
|||||
| Latency | Minimal changes in appearance of wound base | Medical management to control
sepsis, fluid balance, diabetes, and nutrition; Surgical management to provide adequate exposure to the wound, to remove necrotic material and exposed bone |
Clean vascular wound base, access for optimal wound care, soft tissue and bone preparation for coverage | 10 days to 2 weeks |
If the wound does not worsen during this stage, the prognosis for healing is good |
| Gran- ulation |
The angiogenic response (initiated during the latency phase culminates in the formation of a granulating wound base) | Maintenance of a moist physiological wound environment |
Completely vascularized wound base |
2 weeks | Additional studies (angiography and transcutaneous 02 monitoring)are done during this stage, if needed. Angioplasty and/or revascularization may be done, if indicated, from the studies |
| Coverage | A wound ready for coverage and/or closure | Skin graft, delayed closure and/or partial foot amputation | Healed wound | 3 weeks | Once the wound is healthy, the coverage/closure options are usually obvious |
|
|
|||||
Latency Stage. This is the first stage in healing of the problem wound. During this stage, the wound appears to be resting. However, changes take place in the wound environment and the host that make it possible for the wound to transition into the other stages of healing. During this stage, measures to control sepsis, improve fluid balance, augment oxygenation, eliminate edema, and provide adequate exposure to the wound are instituted. A clean wound environment must be established with removal of necrotic and/or avascular infected tissue by sharp debridement, chemical debridement, or both. Wound dressings are selected for their ability to control infection, eg, bacteriostatic/bacteriocidal solutions. Hyperbaric oxygen is a useful adjunct at this time to improve wound oxygenation and initiate the inflammatory response.11 Its use at this time is largely a clinical decision since edema and induration interfere with accurate transcutaneous oxygen measurements. The latency stage, once appropriate strategies are introduced, is usually completed in 10-14 days.
Granulation Stage. This stage begins when cellulitis and sepsis have been resolved and the wound begins to generate a granulating base. Wound dressings are switched to more physiological agents. Debridement of any remaining nonviable tissue and bony deformities, if present, are done. However, if the nonviable tissue is mummifying, small in size, and the demarcation between viable and nonviable tissue sharp, it is appropriate to allow auto amputation to occur. We have observed that improvements in nutrition as measured by prealbumin levels parallel the appearance of granulation tissue in the wound. If the wound is not improving as expected, juxta-wound transcutaneous oxygen measurements are made and vascular surgery consultation are obtained. From these evaluations, decisions will be made regarding the role for hyperbaric oxygen, angiography, angioplasty, and/or revascularization.12 The granulation phase requires about 2 weeks to complete.
Coverage Phase. Once the wound is granulated, coverage and/or closure of the wound is done. For the healthy, vascular-based wound, coverage decisions are usually easy to make. A number of choices are available including: 1) allowing the wound to heal by secondary intention; 2) coverage by artificial skin substitutes; 3) split thickness skin grafting; 4) delayed primary flap closures; 5) partial amputations; and 6) innovative amputations with complex flaps. Although this portion of the wound management is surgical, the patient’s medical status must be optimized to achieve the best results. Usually the closed wound is stable after 2-3 weeks, and resumption of activities can be done at this time. This signals the end of the coverage phase. During this stage, the patient is usually transferred to a lower level of care than that required in the acute hospital setting, and physical therapy and rehabilitation is started. Once the wound is healed, the patient and his/her family are instructed in measures to prevent wound recurrences and new wound occurrences.
Principle III: The Hierarchy of Metabolic Demands For Wound Closure
Different wound coverage techniques have different oxygen requirements. Although it is said that healing is independent of the length of the incision, wound size and depth make a difference in healing of the DFW. The smaller the size of the wound, the more likely healing will be possible with a marginal blood supply. For each succeeding layer of the 3 tissue planes (including skin and subcutaneous; muscle and tendon; and bone and joint), I suggest that the requirements for oxygen and blood flow double for successful healing. For problem wounds involving all layers of tissue to the bone and joint level, the full reserve capacity (ie, 20-fold increase) in blood flow and oxygen delivery is probably required for healing.
There is a hierarchy of oxygen and perfusion requirements for the different coverage and closure choices.13 The coverage choice that requires the least demands is the wound that is covered with a thin crust or eschar with sharp demarcation between the adjacent healthy skin and the eschar. Epithelialization occurs at the margins, and healing is observed even in very dysvascular wounds. The next level of demand would be seen with the split thickness skin graft and the artificial skin substitutes. Supplemental hyperbaric oxygen skin grafts will be preserved in relatively hypoxic wound environments many times.14 Simple incisions represent the third level of oxygen requirements for wound healing. Without adequate blood supply to the margins, wound slough, dehiscence, and/or infection will occur. Composite flaps, as seen in partial ray resections and forefoot, mid-foot amputations involve healing through all 3 layers of tissue. The full blood flow and oxygen delivery reserve capacity is required for healing at this level of the hierarchy. Finally, the most demanding type of wound healing is in the deep cavity involving all 3 layers of tissues. The wound must contract in size and form sufficient granulation tissue to fill in the defect. Infection control in the defect is dependent on a blood supply that provides enough oxygen for white blood cell bacterial killing and delivers enough antibiotics to be effective. The pairing of the wound closure technique with the estimated potential for healing makes it possible to select the closure technique that is most appropriate for the patient’s wound.
Treatment Strategies for Problem DFWs
The healing of DFWs is not by chance. Once a problem wound is recognized, appropriate interventions, precisely timed in the healing continuum, are essential to achieve the best possible outcomes.15 The first step, the assessment, is necessary to determine the seriousness of the wound. There are various grading systems for diabetic foot wounds. Previously, I defined the problem wound qualitatively. However, I recommend using a grading system that is based on 5 objective criteria, can classify the severity of the wound, will quantify improvement, and is easy to use.16 Each criterion is graded with objective parameters on a scale of 2 (best) to 0 (worst) (see Table 3a). From the wound score a wound is classified as healthy, problem, or futile.
|
Table 3a. Strauss Wound Grading System |
|||
| Criteria | 2 Points* | 1 Point* | 0 Points* |
|
|
|||
| Appearance (wound base) |
Red | White/Yellow | Black |
| Size | > Thumb print | Thumb print-to-fist | > fist |
| Depth | Skin, subcutaneous | Muscle, tendon | Bone, joint |
| Infection | Colonized | Cellulitis | Sepsis** |
| Perfusion*** | Palpable pulse (warm skin temperature and/or normal capillary refill) | Doppler pulse (cool skin temperature, and/or sluggish capillary refill) | None (cold skin temperature and/or no capillary refill) |
|
|
|||
| * Use half points for intermediate or mixed findings | |||
| ** Cellulitic wound with one or more of the following: leucocytosis, elevated blood sugars, fever, positive blood culture, necrotizing soft tissue infection. | |||
| ***The most difficult to assess. Transcutaneous oxygen measurements and angiography may be needed to supplement this criterion. In the diabetic with calcified arteries, we have found Doppler studies and use of the ankle-brachial index to be a poor predictor of healing potential. | |||
|
|
|||
The wound score provides a quantitative method to define a problem wound and a logical approach to management (see Table 3b). A healthy wound will heal with minimal attention. One hundred percent good outcomes are expected. A futile wound will probably not heal without angioplasty and/or revascularization, high patient motivation, and very concerted wound healing team efforts. The problem wound is the wound type in which healing is expected to occur in 80% of more of the cases with appropriate treatment strategies.10 Once a decision has been made to salvage a problem DFW, the following 4 strategies need to be followed.15
| Table 3b. Wound Classification and Predicted Outcomes Based on the Wound Score | ||
| Wound Classification |
Wound Score |
Outcome |
| Healthy | 8-10 | 100% healing |
| Problem | 4-7* | 80% healing** |
| Futile*** | 0-3* | Amputation |
| * When scores are between 3 and 4, other considerations such as the patient’s level of health (ie, host status), nutrition status, and motivation (see Tables 9a, b) need to be considered before making a decision to amputate. | ||
| ** With appropriate management strategies | ||
| *** Healing highly unlikely in futile wounds, unless revascularization and/or angioplasty is effective in improving perfusion. | ||
Strategy 1: Wound Appearance. The wound must be clean enough that other strategies will be effective. The wound should be prepared such that wound coverage and closure strategies can be done in the future. Management of the wound base is a critical component of this strategy and is guided by the appearance criterion used for scoring a wound (see Table 3a). If wound base is red (a score of "2" on the appearance criterion), physiological agents such as normal saline should be used to maintain a moist, healthy environment.
If the wound base is white or yellow (a score of "1" on the appearance criterion), interventions are done to remove the exudate and/or superficially necrotic material. Enzymatic debriding agents are useful for this purpose. In-office or on-the-ward limited debridements are effective in removing necrotic debris over a vascular wound base. The debridements are "limited" in as much the goal is to remove only obvious and easily accessible necrotic material in a serial fashion. This is in contrast to an in-operating room debridement in which the goal is to establish surgical margins. Limited debridements conserve the most possible tissue and usually are well tolerated pain-wise due to the removal of only necrotic tissue and the patient’s neuropathy. Likewise, blood loss is almost negligible. Hydrotherapy (whirlpool or pulsatile lavage) can soften and hydrate the tissues so the limited debridements can even be more effective. If the wound base is black (a score of "0" on the appearance criterion), then debridement to viable vascularized tissues is required.
Mechanical problems, such as deformities, contractures, and muscle imbalances, should be corrected and nonviable bone removed. Osteotomies, ostectomies, joint resections, joint manipulations, and tenotomies are surgical techniques used to achieve these goals. Frequently, these measures can be done in the nonoperating room setting with equipment as simple as scalpel, forceps, scissors, or bone ronguer.17 When anesthesia and an operating room are required, a surgeon, with an interest in problem wounds, should be requested. The final consideration regarding wound appearance is that of the nutritional status of the patient. Wounds in malnourished patients heal poorly, if at all. The status of the patient’s nutrition is easily established by measuring albumin and prealbumin levels.18 The clinical nutrition consultant can optimize the patient’s diet especially if factors such as obesity, dysphagia, renal insufficiency, or combinations of these are present and complicate nutrition care.
Strategy 2: Wound Protection. The problem wound must be kept as motionless as possible in order to provide an optimal environment for healing. Motion through a healing site can be destructive. Motion contributes to wound edema, bleeding, slough of threatened flaps, shearing of new, tenuous blood vessels, wound dehiscence, and bacterial multiplication. These may disrupt the healing that occurred while the wound was at rest.
Wound protection is achieved with appropriate dressings and immobilization techniques: Dressings should be bulky, padded, compression types that remain in place even when the patient moves. A good dressing is one that stays in place when the patient transfers or shift positions but is easy to remove for dressing changes. Elastic wraps (Ace®) over a bulky dressing keep the dressing in place, provide supplemental and gentle compression, and are easy to remove (without scissors). In addition, elastic bandages are reusable and obviate the need for using tape on the skin. The elimination of scissors to remove a dressing reduces a potential source of contamination if scissors are used without sterilization from one patient to another.
Casts and splints are other techniques to protect and immobilize a wound. In general, casts are much more desirable in as much as they control motion and provide uniform compression much better than splints. Splints do not have the strength of casts, are heavier for equal strengths, and are not appropriate for weight bearing. Fiberglass casts offer strength with much less weight and maintain their strength even if accidentally wetted in contrast to plaster casts. Casting for DFWs should be creative, ie, strong, yet allow access to the wound by either an extended opening near the cast end, if the wounds are in that location, or windows to provide access. The use of cast windows is controversial. Windows reduce the strength of the cast so reinforcement of the cast, if weight bearing is to be done, may be necessary. Meticulous attention to replacing the cast window after each dressing change is essential to prevent cast window edema. Casts with windows may need to be changed frequently since moist dressings may wet the padding and cause skin maceration. Bi-valving the cast is another way of meeting the requirements of protection, immobilization, and access to the wound for inspections and dressing changes. Although the use of total contact casts is considered a standard of practice by many, this technique for DFWs is not recommended. I feel the underlying deformities first need to be corrected and then frequent wound inspections and dressing changes done postoperatively. Total contact casts require weekly or semiweekly changes, and healing of problem wounds usually takes weeks to months to achieve using this technique. If the underlying deformity(ies) causing the wound is not corrected, there is a high likelihood that the wound will recur after the total contact cast is discontinued. Selected shoes and removable leg casts are useful because they are easy for the patient to don and remove while meeting the goals above. Examples include post-op shoes, wedged shoes, Chukka boots, removable walker casts, off-loading diabetic boots, and Charcot restraining orthotic walkers.
Surgical stabilization of DFWs is needed for special situations. When wounds are so large that other techniques will not provide adequate stabilization and access, temporary percutaneous pinning of joints can be very useful. This technique is used after contracture releases—especially of the Achilles tendon—to manage equinus contractures in association with DFWs. Temporary pin fixation axially through the ankle is recommended when a short mid-foot amputation is done. Because of the short foot, casting and splinting cannot counteract the loss of leverage from removal of the forefoot. Pinning of the ankle prevents the "unbalanced" foot from developing equinus contractures and pressure sores during the period of wound healing. If the patient walks on a shortened foot with an equinus contracture, breakdown over the flap end is likely to occur. Internal fixation for wound stabilization is rarely indicated due to the high risk of infection and the potential to create new wounds in order to gain access to the fusion site. As in the management of the wound appearance, wound protection techniques should be done in collaboration with the foot surgeon with a special interest in DFWs.
Strategy 3: Wound dressings. One thousand or more agents are available for wound dressings. Agents need to be selected according to the wound appearance and what the immediate goals of the wound management are. The cardinal rule when selecting dressing materials is to make the wound as "physiological" as possible. Initially, this rule may need to be deferred until the infected material and the necrotic tissue in the wound base have been eliminated. As the wound improves, the choices of wound dressings become larger and the selection easier. Many times, 2 or more agents are selected to achieve combined effects such as using an agent to debride necrotic material in combination with one that absorbs secretions. To simplify selection, the agents may be divided into 4 categories (see Table 4).
| Table 4. Wound Dressing Choices | ||||
| Category | Effect | Examples* | Benefits | Disadvantages |
|
|
||||
| Gauze moistened with solutions | Maintenance of moist environment; remove debris from wound |
Normal saline, acetic acid solution, Dakins® solution; Flagyl® solution | Economical; useful for large as well as small wounds; control of odor |
Time consuming and frequency of dressing change |
| Covering agents | Maintains moist environment | Op Site®; Tegaderm® Xeroform®; Polymem® Duoderm®; Mepitel®; Oasis® and Apligraft® | Less frequent dressing changes. Oasis®; Apligraft® are artificial skin substitutes | Fluid collection under coverings can cause maceration of periwound skin |
| Absorptive agents | Maintain moist environment while removing secretions | Kaltostat®; Aquasel® Allevyn®; Mepilex® Wound-Vac® | Highly effective in removing secretions and exudates from wound |
Costly when used in large wounds, may require frequent dressing changes |
| Salves, ointments, and creams with additives | a. Moisturizing | Curasol®; Safgel®; Carrasyn® | Limited to relatively small wounds (except Silvadene® for burns) | Some agents very costly |
| b. Enzymatic | Acuzyme®; Panafil® | |||
| c. Bacteriostasis | Acticoat®; Lodosorb gel®; Bactroban®; Bacitracin®; Silvadene®; Polysporin® | |||
| d. Naturopathic | Biafin® | |||
| e. Anti- inflammatory | Hydrocortisone®; Benedryl® | |||
| f. Growth factor | Regranex® | |||
| * These examples are not intended to be a complete list of available agents, nor does inclusion in the list imply endorsement of the agent. The examples are products we have used in our in-patient and out-patient wound programs. | ||||
|
|
||||
The first group of dressing materials are the gauze dressings that are moistened with solutions such as normal saline. Agents can be added to normal saline such as acidic acid, bleach, Flagyl®, etc to provided bacteriostatic effects to the solution. A normal saline dressing is the standard for measuring effectiveness and economies in wound dressings. A moist wound optimizes the environment for healing, prevents cell desiccation, and decreases bacterial load and debris with its frequent changes. The major disadvantage of moist dressings, however, is the time and frequency they require in order to be effective. In addition, expertise is needed to apply these dressings so the wound remains moist and the surrounding tissue remain dry and do not become macerated. The moist, frequently changed dressing may be the only dressing option for the large wound with a necrotic base and in wounds with osteomyelitic bone in the base.
A second group of dressing materials are used when wounds are nonexudative with healthy vascular bases. Many agents are available that can minimize the number of dressing changes while maintaining a moist environment. Examples include hydrogels, occlusive and semi-occlusive membranes, and sponge-like materials. Artificial skin substitutes (Oasis® and Apligraft®) are biological coverings that can stimulate epithelialization of the defect while keeping the wound base physiological. These dressings, although much more expensive than the normal saline dressings can reduce nursing time for managing wounds and, in some cases, promote wound healing without the need for skin grafting.
The third wound dressing choice is used for wounds that are highly exudative. These agents are designed to absorb secretions. Generic terms for these materials include hydrophilic beads, alginates, and highly absorptive sponge-like materials. The wound vacuum (Wound-Vac®) has been useful for difficult to manage exudative wounds.19 A trimmed sponge that conforms to the wound shape is placed in the wound and connected to a vacuum apparatus that has controls to adjust the pressure and frequency of suction. The Wound-Vac® effectively removes exudates while promoting wound contraction and blood flow. Nursing care for dressing changes is only needed semi-weekly.
The fourth category of dressing materials are those agents that are basically gels, ointments, or salves that have special additives (see Table 4). Moisturizing agents, a popular additive, help to maintain the physiological environment. A second category of additives are the enzymatic debriding agents. A third category are agents with bacteriocidal and/or fungicidal additives. A fourth category of additives are those with naturopathic effects. A fifth category are the ointments with anti-inflammatory additives. Finally, there are those agents with growth factors in them. The use of these agents require judgment and experience. Some of those agents are expensive; costing several hundred dollars for a 15 g tube. The appearance of the wound and what is thought to be the factor most needed for healing should determine the type of agent to use.
Strategy 4: Wound Oxygenation. Wounds require adequate perfusion to heal. Juxta-wound tissue oxygen tensions of 30-40 mm are required for wound healing and infection control.20 Wound oxygenation strategies often complement each other. An important consideration for achieving this strategy is edema reduction. Edema decreases oxygen availability by increasing the diffusion distance of O2 from the capillary to the cell. If it occurs in a confined space, circulation may be compromised due to collapse of the microcirculation (ie, compartment syndrome). The foot has more closed spaces, potential compartments and tendon sheaths than almost any other structure in the body. Edema reduction is achieved through elevation, elastic wraps, support hose, immobilization, pain control, pneumatic compression devices, and hydrotherapy. Many patients with advanced peripheral vascular disease dangle their legs for pain relief. This increases blood flow due to the gravity effect, but the persistent dependent position of the legs causes edema, venous stasis, stasis dermatitis, and stasis ulcers. The foot wound is especially susceptible to the edema formation due to its propensity to be in the dependent position for sitting, standing, and walking activities.
Optimization of cardiac and renal function are essential medical interventions for management of DFWs. Improved perfusion augments oxygen, growth factor, and substrate delivery to the wound. Diuresis can help to mobilize extracellular fluid and reduce edema. The vascular surgeon or radiologist may improve oxygenation through angioplasty and revascularization. If outflow is poor and the arterial disease diffuse, benefits from these techniques alone may be inadequate to meet oxygenation and perfusion demands for wound healing.
Hyperbaric oxygen (HBO) is another method for improving oxygenation to the DFW. HBO is a particularly useful adjunct in the hypoxic DFW and therefore can be an adjunct to angioplasty and revascularization. It increases plasma and tissue oxygen tensions 10-fold.21 With HBO, oxygen delivery is no more flow dependent (vs red blood cell oxygen delivery) than the other substances physically dissolved in the plasma. With the increased oxygen tensions, diffusion is increased 3-fold through tissue fluids at standard treatment pressures.22 Juxta-wound transcutaneous oxygen measurements can indicate which wounds need and/or are benefited by HBO.23 These studies help to objectify the use of this modality.
Many pharmacological agents can improve flow and other rheological properties of blood (see Table 5). They include red blood cell deforming agents, blood vessel dilating agents (both local [eg, nitroglycerin patches and systemic]), and anticoagulants.24 Use of these agents require judgment. Many have significant side effects and require monitoring. The PCP will be largely responsible for using these strategies.
| Table 5. Pharmacological Agents to Improve Flow and/or Alter Blood Rheology | ||
| Agent | Example | Comments (side effects) |
| Red blood deformability | Trental® | Observations suggest a complementary effect with hyperbaric oxygen |
| Antiplatelet adhesive agents | Aspirin Ticlid® |
Gastrointestinal upset from aspirin |
| Systemic vasodilators | Calcium channel
blockers (Procardia®) |
Hypotension |
| Local vasoconstrictors | Nitroglycerin ointments Pletal® |
Flushing, headaches |
| Anticoagulants | Coumadin® Fragmin® Heparin® Lovenox® |
Bleeding; monitoring needed for Coumadin® and Heparin |
Prevention of New and Recurrences of Healed Diabetic Foot Wounds
Once the DFW is healed, the goal, of course, is to keep the wound from recurring. Preventive measures start with the PCP, who is often the first to be told of a new problem or wound recurrence by the patient or family. Four target areas need to be addressed to prevent DFW recurrences and/or the formation of new wounds.
First, skin and toenail care must be optimized. Diabetics tend to have dry skin. This should be managed by moisturizing the skin with soaks and then lubricating the skin with petroleum- or lanolin-based agents. This skin care treatment helps to maintain the turgor, moisture content, and pliability of the skin. Foot skin care needs to be done on a daily or, in some cases, in which the skin is exceedingly dry and scaly, twice-a-day basis. With dry skin, fissures develop and lead to moisture and debris accumulation. This causes the skin to macerate. Bacteria can multiply in the debris, penetrate the macerated skin leading to cellulitis and ulceration. Skin grading and management is easily done with a 4-point grading system (see Table 6).25 Nail care is equally important. The nails of diabetics need to be trimmed on a regular basis. Trimming must be done by someone experienced in working with dystrophic, fungus-infected nails. Nail evaluation and management like skin care can be done on a 4-point grading system (see Table 7).26
| Table 6. Evaluation and Management of Skin Care | ||
| Grade | Finding | Intervention |
| 1 | Moist, pliable, soft healthy skin | None; compliment patient |
| 2 | Mild-to-moderately dry and scaly skin | Do foot skin care (FSC)* on a daily basis |
| 3 | Very dry and scaly skin | In-office FSC plus daily care at home |
| 4 | Scaling and plaquing of skin | Hydrotherapy, debridement of plaques and FSC in-office followed by daily FSC at home |
| * | Foot Skin Care | |
| a. | Moisturize 10 minutes with shower, bath, or wet towel wrap | |
| b. | Dry | |
| c. | Lubricate and massage skin with petroleum jelly or similar agent. | |
| d. | Wipe off all residual lubricant especially between toes with a soft cloth; the skin should not feel greasy after this step. | |
| Table 7. Evaluation and Management of Nail Care | ||
| Grade | Finding | Intervention |
| 1 | Healthy, normal length | Compliment |
| 2 | Healthy, but long | Simple trimming |
| 3 | Thick, dystrophic, fundus infected | Trim, debride, file (rotary drill) until tissue paper thin |
| 4 | Long, rams horn shapes with debris imbedded in nail margins | Formal debridements with rongers and/or bone cutters |
The appearance of new calluses and erythematous areas on the skin suggest abnormal pressure build-ups or shear stresses and are the second prophylactic area to address. They require immediate attention. Calluses and impending pressure sores can rapidly progress to ulcers that can quickly penetrate to bone. Abscesses, cellulitis, and osteomyelitis can be the consequences. A step-wise approach to these prewound problems must be instituted. Initially, calluses are shaved and inserts to offload the pressure areas are placed in the patient’s shoes. Principles of offloading include relieving the pressure under the wound with cutouts in the foot pad and using the other healthy weight-bearing portions of the foot to load share the offloaded portion. Most of the time, these simple measures are adequate. Muscle imbalances causing clawing of the toes, which are very common in the diabetic foot, may contribute to the deformity. Contracture releases and manipulation of the contracted joints can be done percutaneously in an office setting. This improves the biomechanics of the forefoot by eliminating the winching effect the clawed toe has on driving the metatarsal head plantarward. If these techniques are not effective, then attention to the bone and/or bursal deformity must be given with surgery. Bone deformities in the foot such as bunions, spurs bunionettes, and subluxations/dislocations may require surgical management if shoe wear and orthotics are not effective.
Appropriate shoe wear is the third measure for preventing new and recurrent DFWs. MEDICARE (HCFA) has provisions for diabetic footwear (see Tables 8a, b).26 In 1993, at the direction of Congress, the Therapeutic Show Bill was passed to provide shoe wear and orthotics to diabetics with the goal of preventing DFWs in the high-risk patient. Medicaid programs have provisions for diabetic footwear but vary from state to state. Diabetics need to be fitted with shoes that provide support, yet have a spacious toe box. Extra-depth padded inserts should be placed in their shoes to minimize foot plantar contact pressures. Once new shoes are obtained for the diabetic, they should only be worn 15-20 minutes initially; then the feet should be inspected for pressure areas and abrasions. If initial new shoe wear is okay, the shoe wear time should be increased progressively. Many times, diabetic shoe wear must be "fine-tuned" by the supplier at the direction of the prescribing physician. The feet need to be inspected daily for sores. White socks should be worn and changed daily. White is desirable since any stain from a new sore will be easy to recognize. Attention must be given to proper fitting of the socks, also. If they are too tight, they can impede circulation, especially in the lower pressure venous side leading to edema. If too loose, slipping, sliding, and wrinkles in the socks can lead to foot sores.
| Table 8a. Requirements for Diabetic Therapeutic Shoe Benefits | ||
| • | Medicare Part B coverage | |
| • | Have satisfied Medicare’s $100 deductible for the current year | |
| • | Certifying statement from the treating physician that includes: | |
| a. | documentation of diabetes | |
| b. | the treatment plan requires therapeutic shoes. | |
| c. | one or more conditions (ie, risk factors) associated with the diabetic foot are present. | |
| • | A prescription for therapeutic shoes by a qualified physician (ie, knowledgeable about diabetic shoes and inserts). | |
| • | The therapeutic shoes must be supplied by a retail store that covers footware covered by the therapeutic Bill, pedorthist, or other qualified individual. | |
| Table 8b. Diabetic Therapeutic Shoe Benefits | |
| • | One pair of off-the-shelf depth shoes and 3 pairs of multidensity inserts over a 1-year period or any one of the following permutations over a 1-year period: |
| One pair of off-the-shelf depth shoes, including modifications and 2 additional pairs of multi-density inserts. | |
| One pair of custom-molded shoes and 2 additional pairs of multidensity inserts. | |
| • | Medicare reimbursement of 80% of reasonable and allowed Medicare fees to a maximum amount of $134 on a pair of shoes and $68 for a pair of inserts. The patient is responsible for paying the remaining 20% of the bill at the time the shoes and/or inserts are dispensed, if the supplier is accepting assignment. If not, the patient needs to pay the supplier and submit the paper work directly to Medicare for reimbursement. |
Two very difficult-to-manage gait-related problems are shear and rotary stresses that may occur in the foot with walking. These problems are aggravated by weakness, imbalances, and partial foot amputations. Orthotics, braces, and custom-designed shoes may control these problems. If not, consideration for surgical stabilization of the joint must be given. Some patients with these problems prefer to use a wheelchair for most ambulatory activities and weight bear on the "at-risk" feet only for transfers. This is a reasonable decision when the alternatives of new DFW occurrences or surgeries that do not always have good outcomes are considered.
The final preventive measure is the use of appropriate aids for walking. There is a hierarchy of levels of ambulation from bedridden to wheelchair to household to community. The level of ambulation should always be documented, and, if there is deterioration, reasons should be sought. Walking aids such as crutches, walkers, and canes may be used at any level. Balance problems coupled with weakness are frequent in the diabetic patient with neuropathy especially after prolonged convalescence to manage a DFW. Physical therapy evaluation and management is recommended to achieve the highest level of safe ambulation. In addition, improvements in gait patterns can reduce shear stresses in the feet and reduce the likelihood of new DFWs developing. In collaboration with the physical therapist, the decision may need to be made to limit the patient to wheelchair ambulation.
Conclusions
Healing of DFWs requires appropriate and timely interventions as well as patient and family cooperation. The wound score offers a quick assessment for healing potential and management. We have also developed a goal score to assess patient cooperation (see Tables 9a, 9b).27 If the goal score is low, we recommend a major amputation for managing the DFW. In such patients, new problems will likely develop once their care is no longer supervised, or the potential for ambulation so poor that it is not appropriate to expend resources to heal a wound. Conversely, a high goal score encourages us to optimally manage, the DFW using the team approach with the 4 strategies previously described. Healing is expected in 80% of the problem DFW group in motivated patients.
| Table 9a. The Patient Goal Score | |||
| Criteria | 2 Points* | 1 Point* | 0 Points |
| Motivation (to avoid amputation) | Strong | Some | None |
| Comprehension (of problem) | Full | Partial | None |
| Compliance (diet monitoring, skin care, etc) | Consistent | Inconsistent | None |
| Family support | Excellent | Fair | None |
| Self care(activities of daily living, eating, dressing, hygiene, etc) | Independent | Partial | None |
| Note: Use half points for intermediate findings or mixed findings | |||
| Table 9b. Goal Score Interpretations | |
| Score | Recommended Management |
| 8-10 | Foot wound salvage regardless of wound score unless the wound and adjacent tissue are totally avascular, pain is uncontrollable such as in reflex sympathetic dystrophy, the wound has failed to heal with optimal wound management strategies, and/or infection in the wound is uncontrolled. |
| 4-7 | Salvage if a healthy or problem wound |
| 0-3 | Major (transtibial or higher) amputation recommend- ed regardless of wound score |
Once a DFW is healed, much can be done to prevent recurrences. The PCP is the essential link between the patient and the wound specialist. With a knowledge of wound healing principles, treatment strategies, wound classification, and preventive measures, the PCP has much to contribute to the diabetic foot care team. Even more important is the role the PCP has in ensuring the patient with diabetes is compliant with skin care, nail care, foot/skin inspections, and appropriate shoe wear. When a diabetic foot problem arises, the PCP will likely be the first physician from whom the patient seeks advice.
With the number of diabetics in the United States and their requirement for health care resources, the PCP is expected to have a large diabetic patient population. It is the fourth or fifth most common reason a patient seeks help from the PCP.28 Fifteen percent of diabetics are expected to develop an extremity ulcer sometime during the course of their disease, and diabetes is the leading cause of lower extremity amputations in the United States.29 The DCCT study showed that careful management of blood sugar levels reduced diabetic complications significantly, perhaps by as much as two thirds.3,30 With proper patient selection (by using the wound and goal scores), appropriate treatment strategies and attention to the measures used to prevent new wounds and recurrences of healed wounds, complications in the diabetic foot can be reduced to an equal degree.
References
1. National Institutes of Health. Diabetes in America. 2nd ed. Washington, DC: National Institutes of Health; 1995.
2. American Diabetes Association. Direct and Indirect Costs of Diabetes in the United States in 1992. Alexandria, Va: American Diabetes Association.
3. Levy D. Diabetes. A key to lower health costs. USA Today. April 12, 1994;2.
4. Position statement: Third-party reimbursement for diabetes care, self-management education, and supplies. Diabetes Care. 2000;23(Suppl 1):S111.
5. Rosenquist U. An epidemiological survey of diabetic foot problems in the Stockholm country. Acta Med Scand Suppl. 1984; 687:55-60.
6. Hohn, DC. Oxygen and lecocyte microbial killing. In: Davis JC, and Hunt TK, eds. Hyperbaric Oxygen Therapy. Bethesda, Md: Undersea Medical Society, Inc; 1997:101-110.
7. Hunt TK, Zederfeldt B, Goldstick TK. Oxygen and healing. Am J Surg. 1969;118:521-525.
8. Guyton AC, Hall JE. Textbook of Medical Physiology. 9th ed. Philadelphia, Pa: WB Saunders Co; 1996:171-172, 1065-1067.
9. Babior BM. The respiratory burst of phagocytes. J Clin Invest. 1984;73:599-601.
10. Borer KM, Borer RC Jr., Strauss MB. Prospective evaluation of a clinical wound score to identify lower extremity wounds for comprehensive wound management. Undersea Hyperb Med. 2000;27(Suppl):34.
11. Hyperbaric oxygen therapy. In: Committee Report, Undersea and Hyperbaric Medical Society. Kensington, Md; 1999:27-34.
12. Strauss MB, Borer RC Jr., Borer KM. An algorithm approach to decision making in problem wounds. Undersea Hyperb Med. 2000;27(Suppl):35.
13. Strauss MB, Groner-Strauss WS. Hyperbaric oxygen in the treatment of chronic bone and joint infections. In: Stein H, ed. Sirot 1997 Haifa Inter-Meeting, Scientific Proceedings. London, England: Freund Publishing House, Ltd; 1997:47-51.
14. Bowersox JC, Strauss MB, Hart GB. Clinical experience with hyperbaric oxygen therapy in the salvage of ischemia skin flaps and grafts. Journal of Hyperbaric Medicine. 1986;1:141-149.
15. Strauss MB, Pinzur MS. Treatment strategies for problem diabetic foot wounds [abstract]. AAOS. 2001;2:SE 64:675-676.
16. Strauss MB. Problem wounds: How to promote healing. Consultant. 2000;40(13):2259-2273.
17. Strauss MB, Hart JD. KISS (Keep It Simple and Speedy) procedures for problem foot wounds. Undersea Hyperb Med. 2001;28(Suppl):35-36.
18. Strauss MB, et al. Nutrition assessment of problem wounds. Undersea Hyperb Med. 2000;27(Suppl):3-36.
19. Argenta LC, Morykwas MS. Vacuum-assisted closure: A new method for wound control and treatment: Clinical experience. Ann Plast Surg. 1997;38(6):563-576.
20. Hunt TK, Pai MP. The effect of varying ambient oxygen tensions on wound metabolism and collagen synthesis. Surg Gynecol Obstet. 1972;135:561-567.
21. Boerema I, et al. Life without blood. A study of the influence of high atmospheric pressure and hypothermia on dilution of the blood. J Cardiovasc Surg. 1960;1:133-146.
22. Extracorporeal Circulation for Open-Heart Surgery. Peirce EC, ed. Springfield, Ill: Charles C. Thomas; 1969:83-84.
23. Borer RC Jr., Borer KM, Strauss MB. Prospective serial transcutaneous oxygen challenge measures in problem lower extremity wounds. Undersea Hyperb Med. 2000;27(Suppl):40.
24. Appel M, et al. Augmenting transcutaneous oxygen tensions in dysvascular test [sic, patients] with the 1-2-3 protocol. Undersea Hyperb Med. 2001;28(Suppl):60-61.
25. Strauss MB, Hart JD, Winant DM. Preventive foot care. A user friendly system for patients and physicians. Postgrad Med J. 1998;103(5):223-245.
26. Balanced Budget Act of 1997. US Government Printing Office. 1997:115-116. (Publication #869-033-00034-1.)
27. Strauss MB, Smith LV. The goal score as an aid to decision in foot salvage vs. leg amputation. Undersea Hyperb Med. 2001; 28(Suppl):68.
28. LeRoith D, Taylor SI, Olefsky JM, eds. Diabetes Mellitus: A Fundamental and Clinical Text. Philadelphia, Pa: Lippincott Williams & Wilkins; 1996.
29. Reiber GE, Boyko EJ, Smith DG. Lower extremity foot ulcers and amputations in diabetes. In: Harris MI, Cowie C, Stern MP, eds. Diabetes in America. 2nd ed. Washington DC: US Government Printing Office; 1995. (NIH publication 95-1468.)
30. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977-986.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.