Update on Current Management of Atrial Fibrillation
Update on Current Management of Atrial Fibrillation
Authors:
Sula Mazimba, MD, MPH, Cardiology Fellow, Kettering Medical Center, Dayton, OH.
Mauricio Anaya-Cisneros, MD, Cardiology Fellow, Kettering Medical Center, Dayton, OH.
Analkumar Parikh, MD, Internal Medicine Resident, Kettering Medical Center, Dayton, OH.
Peer Reviewer:
Tushar N. Shah, MD, FACC, FASE, FAHA, FACP, The Heart Attack and Stroke Prevention Center at The Austin Center for Living, Austin, TX.
This is an adaptation of an article that originally appeared in the October 2011 issue of Primary Care Reports.
Introduction
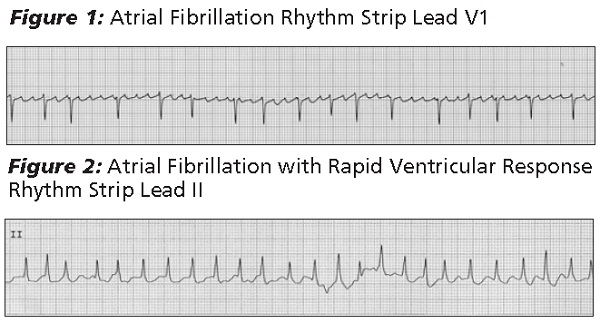
Atrial fibrillation (AF) is an irregular, disorganized, electrical activity of the atria.1 It is characterized by uncoordinated atrial activity that leads to inefficient atrial contraction and impaired ventricular filling. The ability of ventricles to maintain stroke volume and cardiac output can then become impaired, resulting in a back up of blood in the lungs and inadequate perfusion of vital organs. The electrocardiographic (ECG) characteristics of AF include the absence of regular P wave activity, which is replaced by rapid oscillations or fibrillatory waves that vary in size, shape, and timing. The fibrillatory waves occur at a rate greater than 300 per minute and are typically best visualized on the V1 electrocardiographic lead. (See Figure 1.) The ventricular response in AF is fortunately less than this fast atrial rate due to the prolonged refractory period of AV nodal conduction. In a typical case of atrial fibrillation occurring in an adult with a healthy heart, the ventricular response is between 160 and 180 beats per minute (bpm). Aging and disease may affect AV nodal conduction and slow the ventricular response. In addition, autonomic function as well as medications that affect propagation of the action potential through the AV node may also result in a slower ventricular rate. The ventricular response in atrial fibrillation is usually irregular, but at times, if the ventricular rate is fast, the ventricular rhythm may appear "pseudo-regular" and be mistaken for a junctional tachycardia. (See Figure 2.)
Classification
There are various classification systems for AF. The one that is used most commonly employs the temporal nature of AF and the therapeutic response to treatment strategies. (See Table 1.) A useful concept for the management of patients with AF in the ED is "recent-onset," which refers to onset within the past 48 hours. When AF is first detected, it is categorized as new-onset AF. When a patient experiences two or more episodes of AF, it is characterized as recurrent AF. Recurrent AF is further divided into three sub-categories: paroxysmal AF, persistent AF, and permanent AF. Paroxysmal AF is characterized by episodes of AF that self-terminate.2 Most patients with paroxysmal AF will have the AF terminate spontaneously within 7 days without treatment. Persistent AF is one that is sustained for more than 7 days. In these patients, pharmacologic or direct-current cardioversion is considered in symptomatic patients but does not change the designation. AF that is completely refractory to cardioversion or one where the option for cardioversion has been abandoned is termed permanent AF. In permanent AF, the goal is rate control and anticoagulation to prevent thromboembolic events.
Table 1: Classification of Atrial Fibrillation
|
Type of Atrial Fibrillation |
Comments |
|
Lone Atrial Fibrillation |
No underlying structural heart disease or inciting event. Patients often are younger than 60 years and do not need anticoagulation. |
|
Recent-onset Atrial Fibrillation |
Symptoms or other findings indicate onset within the past 48 hours. |
|
Paroxysmal Atrial Fibrillation |
AF that spontaneously reverses to sinus rhythm, usually within a week. |
|
Persistent Atrial Fibrillation |
AF lasting longer than 7 days |
|
Permanent Atrial Fibrillation |
AF that is refractory to cardioversion. The goal of treatment is rate control and consideration for anticoagulation. |
|
Secondary Atrial Fibrillation |
AF attributable to a specific cause |
|
Nonvalvular Atrial Fibrillation |
AF without associated valvular heart disease |
Figure 3: Atrial Fibrillation in a Patient with WPW Syndrome Mimicking Ventricular Tachycardia
These classifications are not mutually exclusive. For instance, the arrhythmia may change from paroxysmal pattern to persistent or permanent. Lone AF is characterized by an absence of recognizable structural heart disease or inciting events and usually occurs in individuals younger than 60 years of age.3 Secondary AF is characterized by situations in which AF is attributable to a primary problem such as acute myocardial infarction, surgery, or hyperthyroidism. Nonvalvular AF is restricted to cases in which there is an absence of associated valvular heart disease, usually mitral valve disease (especially mitral stenosis), a prosthetic heart valve (usually mitral), or a history of mitral valve repair.
Epidemiology
AF is the most common sustained cardiac arrhythmia. It is estimated that about 2.3 million people in the United States have the disease condition.4 The increase in the prevalence of AF is partly a reflection of the increase in the elderly population as well as improved survival of patients with myocardial infarction and heart failure.5 Hospital admissions with the principal diagnosis of AF have increased by about two-thirds during the last two decades6 and account for one-third of hospitalizations for cardiac rhythm disorders. Other factors contributing to the rise in prevalence of AF include the corresponding increase in the prevalence of chronic heart disease as well as the widespread use and availability of noninvasive monitoring devices to diagnose AF.
Although the prevalence of AF in the general population is estimated at 0.4% to 1%,5 it increases to about 8% in those older than 80 years.7 The median age of individuals with AF is 75 years of age.8 The age-adjusted prevalence of AF among men has increased by 50% over the last two decades.7 The incidence of AF across the various age groups is higher in men9; however, the absolute number of women with AF is higher in the age group above 75 years of age, because women comprise the majority of the group in this age demographic.8
Etiology
Several etiological factors have been implicated in the genesis of AF. The underlying pathological process that is noted in most patients with AF is atrial fibrosis and loss of atrial muscle; these are normal age-related changes that may explain the increased prevalence of AF in the elderly.10,11 Other inciting conditions, such as valvular heart disease, hypertrophic cardiomyopathy,12 ischemia, and hypertension, may accelerate these histologic changes beyond what is expected due to normal aging. Nonhomogeneity of conduction created by the juxtaposition of normal atrial fibers next to patchy areas of fibrosis serves as a nidus for the initiation and maintenance of AF.13,14 The extent of fibrosis predicts a poor prognosis for the therapeutic efficacy of various treatment interventions and for the maintenance of sinus rhythm in patients with AF.15 Atrial fibrillation, in turn, further remodels atrial tissue structurally by accelerating atrial fibrosis and electrophysiologically through the progressive shortening of effective refractory periods, further perpetuating the arrhythmia.16,17 Other factors potentially involved in the induction or maintenance of AF include inflammation, autonomic nervous system activity, atrial ischemia, atrial dilation, anisotropic conduction,18 and structural changes associated with aging.19-22
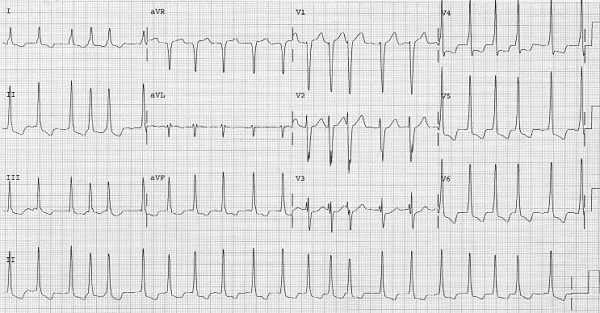
A particularly dangerous setting for AF occurs with conduction across an accessory pathway, which exists in Wolf-Parkinson-White syndrome (WPW).23 In a normal heart, the refractory period of the AV node protects the ventricles from over stimulation from the rapid and chaotic atrial impulses. In the WPW syndrome, a bypass tract provides an alternative path for the atrial impulse to depolarize the ventricles. The bypass tract in the WPW syndrome typically has a shorter refractory period than the AV node, allowing for more frequent conduction of atrial impulses into the ventricles and a faster ventricular response. When an atrial impulse travels down the bypass tract to the ventricles, depolarization of the ventricular myocardium is via slower cell-to-cell spread, not the rapid His-Purkinje system, and so the QRS complex is prolonged. If atrial fibrillation occurs in a patient with WPW syndrome, a rapid ventricular rate can mimic ventricular tachycardia with wide, rapid QRS complexes. (See Figure 3.) This ventricular response is potentially dangerous and can degenerate into ventricular fibrillation.24,25
AF commonly complicates cardiac surgery, where the incidence after coronary artery bypass approaches 15% in patients younger than 65 years and 30% in patients older than 65 years of age.26 The incidence of AF in patients after valve replacement surgery is about 60%.26 In non-cardiac surgery cases, the most powerful predictors of AF include dilated cardiomyopathy, valvular heart disease, hypertension, history of myocardial infarction, and advanced age.27 The presence of left atrial enlargement and ventricular hypertrophy is associated with an increased incidence of AF.
There are two prevailing mechanisms for the electrophysiologic basis of AF and both are supported by experimental data. These mechanisms may coexist in the same patient and are not mutually exclusive.
The first mechanism is termed "enhanced automaticity." AF originates from localized areas of atrial myocardium that have electrical properties which generate rapid spontaneous impulses and serve as a substrate for localized reentry and sustained AF. These tissues most commonly have been found in the distal pulmonary veins as they enter the left atrium, but they also have been identified in superior vena cava, ligament of Marshall, left posterior free wall, crista terminalis, and coronary sinus.28-30 Utilizing catheter-based electroanatomic mapping and ablation of these foci allows AF to be extinguished successfully in some patients.
The second mechanism is termed "conduction abnormality." AF is dependent on a larger mass of tissue distributed throughout the atria that possesses non-uniform refractoriness and conduction. Such non-uniformity favors the development of multiple simultaneous daughter wavelets of depolarization as electrical activity propagates across the atria and provides a milieu for diffuse reentry through the atrium and maintenance of AF.
Reversible Causes of AF
AF can be caused by reversible conditions such as alcohol intake (holiday heart syndrome), surgery, electrocution, acute myocardial infarction, pericarditis, myocarditis, pulmonary embolism or other pulmonary disorders, hyperthyroidism, as well as other metabolic disorders. (See Table 2.) Treatment of the underlying conditions will result in correction of the disorder and maintenance of normal sinus rhythm. AF in the setting of acute myocardial infarction is associated with poor prognosis.26 AF also may be associated with other arrhythmias such as atrial flutter or AV nodal reentrant tachycardias. Treatment of the primary arrhythmias reduces or eliminates the incidence of recurrent AF.31
Table 2: Secondary Causes of Atrial Fibrillation
|
Cause |
Examples and Comments |
|
Atrial pressure elevation |
Mitral or tricuspid valve disease: Mitral valve disease is a more common cause than tricuspid Myocardial disease: Often dilated cardiomyopathy Hypertension: Especially if associated with left ventricular hypertrophy Intracardiac tumors or thrombi |
|
Atrial ischemia |
AF occurs in about 20% of AMI patients Associated with involvement of the atrial branches off the right coronary artery or the left circumflex artery |
|
Inflammatory or Infiltrative Atrial Disease |
Pericarditis Myocarditis Amyloidosis Age-related fibrosis |
|
Toxins |
Alcohol: Holiday heart syndrome Caffeine: Weak evidence |
|
Endocrine |
Hyperthyroidism Pheochromocytoma |
|
Surgery |
Thoracic Pulmonary Esophageal |
|
Neoplastic |
Primary or metastatic disease in or adjacent to the atrial wall |
|
Congenital heart disease |
Atrial septal defect Epstein's anomaly |
|
Central nervous system disorders |
Subarachnoid hemorrhage Ischemic or nonhemorrhagic large stroke |
|
Familial |
Associated with defects in potassium ion channels Inherited in an autosomal dominant pattern |
Symptoms and Manifestation of AF
Patients with AF have variable modes of presentation and this arrhythmia may occur in the presence or absence of heart disease. AF may be self-limited with minimal symptoms or may require urgent medical intervention due to symptomatic hemodynamic compromise manifesting as severe palpitations, chest pain, dyspnea on exertion or at rest, and rarely with syncope. Some patients may be completely asymptomatic until they present with a thromboembolic complication.
A patient's pattern of AF can be characterized by its number of episodes, duration, frequency, mode of onset, triggers, and response to therapy, but these features may be impossible to discern when AF is first encountered in an individual patient.
For most patients, the symptoms are nonspecific.32 It is understandable how patients with persistent or permanent AF can be affected, with reduced exercise tolerance and poorer functional reserve. What is not always appreciated is that even patients with paroxysmal AF have significant functional impairment, sometimes comparable to those with severe cardiac conditions such as coronary atherosclerosis.33 Fortunately, the quality of life in patients with permanent AF can be improved through rate control medications, rhythm control medications, or ablation of the AV node and implantation of the pacemaker.34-36
Approach to a Patient with AF
Diagnosis of AF requires ECG confirmation of an irregular ventricular activity with the presence of an atrial fibrillatory wave pattern. Once the AF diagnosis has been made, the initial priority in the ED is to assess the hemodynamic consequences of the arrhythmia. Unstable patients, such as those in circulatory shock, with acute pulmonary edema, or with acute ischemic chest pain, may necessitate urgent cardioversion.
In stable patients (those able to tolerate the arrhythmia), determining the cause of the arrhythmia and defining the associated cardiac and extra-cardiac factors related to the etiology is useful. A careful history and physical examination is important. It is important to look for new-onset angina or congestive heart failure, as this may necessitate the need for early cardioversion. For those with episodic or recurrent AF, determine the frequency, duration, and precipitating factors, and the mode of termination of the arrhythmia. Patients should be assessed for underlying heart disease and other reversible causes of the arrhythmia, such as the use of alcohol, presence of thyroid diseases, or other metabolic disorders. Unless thyroid function has been recently checked, consider a routine thyroid-stimulation hormone (TSH) determination in patients with recent-onset AF, as almost 10% of these patients in a recent study from Italy were found to be hyperthyroid.37
Investigations
Diagnosis of AF requires ECG documentation, preferably by a 12-lead recording, but by at least a single-lead recording during the arrhythmia. In patients with implanted pacemakers or defibrillators, the diagnosis of paroxysmal AF may be made from detection of the arrhythmia from retrievable memory functions of the device.38
In evaluating the ECG in patients with AF, look at the ventricular response. A ventricular response less than 120 in a patient not on medications suggests AV nodal disease. A prolonged QRS-complex suggests infranodal conduction disease or a bypass tract. Assess for left ventricular hypertrophy and evaluate the ST-segments and T-waves for evidence of ischemia. It is also important to measure electrocardiographic intervals such as the R-R, QRS, and QTc for the purposes of determining and monitoring the therapeutic intervention. A chest X-ray is important to determine the presence of pulmonary and vascular abnormalities.
For patients with recent-onset AF, a transthoracic echocardiogram is useful to identify valvular heart disease, determine chamber dimensions, and detect the presence of pericardial disease, left ventricular hypertrophy, and left atrial thrombus. The echocardiogram is not necessary for urgent rate or rhythm control in the ED, so such a study can wait until the patient is stable.
For patients with symptoms of tachyarrhythmias such as AF but who do not have an arrhythmia present on ED evaluation, Holter monitoring or event recording provided on discharge may be useful to detect paroxysmal episodes provided the device is available and can be analyzed on follow-up. Holter monitoring can also be a means of evaluating rate control for patients with chronic AF who report continued symptoms despite an acceptable heart rate during physician assessment.
Approach to Patients with Recent-Onset AF
In patients with minimal or no symptoms, it is often difficult to determine whether the initial episode of AF is really the first episode or whether it's part of the spectrum of recurrent AF. Patients who are hemodynamically stable with self-limited episodes of AF may not need an antiarrhythmic therapy. Conversely, AF patients who have hemodynamic instability, such as angina, pulmonary edema, or with concomitant pre-excitation, will benefit from urgent cardioversion. Patients with AF and concomitant pre-excitation have unpredictable response to antiarrhythmic therapy.39 It is recommended that patients with underlying pre-excitation syndromes be treated with cardioversion for acute AF.
Spontaneous conversion to sinus rhythm with recent-onset AF is common, occurring in up to 30% while in the ED40 and in about 25% within a week after ED discharge.41 In patients with lone AF, spontaneous conversion within 24 hours is reported in up two-thirds of patients.3 Conversely, if the AF has been present for 7 days or more, the chance for spontaneous conversion is rare.42,43
Table 3: Intravenous Drugs Used for Rate Control in Atrial Fibrillation
|
Drug |
Initial Dose (Adult > 70 kg) |
Subsequent Dose |
Comments |
|
Diltiazem |
15-20 mg (0.25 mg/kg) IV over 2 min |
25 mg (0.35 mg/kg) IV over 2 min at 15 min after first dose if the initial response is not adequate When adequate response achieved, start 5 mg/h IV infusion, titrate infusion to ventricular rate (max rate 20 mg/h) |
Side effects include hypotension, bradycardia, and heart failure |
|
Verapamil |
5-10 mg (75-150 mcg/kg) IV over 2 min |
10 mg (0.15 mg/kg) IV over 2 min at 30 minutes after the first dose if the initial response is not adequate |
|
|
Esmolol |
500 mcg/kg IV over 60 sec |
50 mcg/kg per min IV infusion Repeat initial dose if inadequate response in 2-5 min and increase infusion rate in 50 mcg/min increments (max infusion rate 300 mcg/kg per min) |
Side effects include hypotension, bradycardia, bronchospasm, and heart failure Use beta-blocker with caution in patients with existing hypotension, history of heart failure with depressed ejection fracture, and reactive airways disease (asthma or COPD) |
|
Metoprolol |
5 mg IV |
May repeat 5 mg every 5 min for two additional doses (max total dose 15 mg) |
|
|
Propranolol |
30 mcg/kg IV over 60 sec |
May repeat initial dose twice if inadequate response in 2-5 min (max dose 0.1 mg/kg) |
|
|
Digoxin |
0.25 to 0.5 mg IV |
Not typically required |
Peak effect for rate control may take up to 6 hours |
In AF patients who are hemodynamically stable, rate control with intravenous diltiazem, beta-blocker, or digoxin is appropriate. (See Table 3.) The goal is a heart rate less than 100. Depending on the response, some patients may achieve rate control and symptom relief with rapid transition to oral maintenance of these drugs. (See Table 4.) On occasion, patients may undergo spontaneous conversion in the ED while receiving these rate control medications. In this case, the infusion can be stopped and the patient monitored for a period of time, usually a few hours, to assess for recurrence. If none, the patient can be discharged, usually with a prescription for an oral rate control medication with follow up to check for recurrence and assessment of the etiology.
Table 4: Oral Drugs Used for Ventricular Rate Control as an Outpatient
|
Drug |
Typical Daily Dose (Adult > 70 kg) |
Comments |
|
Diltiazem |
30-90 mg QID |
May use extended-release preparations with the same total daily amount given BID Caution in patients with heart failure and reduced ejection fraction |
|
Verapamil |
60-90 mg QID |
|
|
Metoprolol |
25-100 mg PO BID |
Caution in patients with reactive airways disease (asthma or COPD) |
|
Propranolol |
20-60 mg PO QID |
|
|
Digoxin |
0.125 – 0.25 mg |
Useful when added to calcium channel or beta-blocker to control resting heart rate but allow for some increase in heart rate with exercise |
If the AF onset or duration is deemed to be less than 48 hours in a patient free of significant left ventricular dysfunction, mitral valve disease, or previous embolism, then restoration to sinus rhythm may be attempted with either pharmacological means (see Table 5) or direct current shock.
Table 5: Intravenous Drugs Used for Chemical Conversion of Atrial Fibrillation
|
Drug |
Initial Dose (Adult > 70 kg) |
Subsequent Dose |
Comments |
|
*Not FDA approved, available in Europe |
|||
|
Procainamide |
1 g IV over 60 min |
1-4 mg/min IV infusion |
Converts 50-60% of recent-onset AF after initial dose Major side effect is myocardial depression and hypotension |
|
Ibutilide |
1 mg IV over 10 min |
May repeat in 10 min |
Converts about 75% of recent-onset AF Torsades occurs in about 4% and ventricular tachycardia in about 5% Monitor patients for 4-6 h after infusion |
|
Propafenone |
2 mg/kg IV over 10 min |
Not typically used |
Converts 60-80% of recent-onset AF Avoid in patients with coronary artery disease or cardiomyopathy |
|
Flecainide |
2 mg/kg IV over 10 min |
Not typically used |
Converts 75-90% of recent-onset AF Avoid in patients with coronary artery disease or cardiomyopathy |
|
Amiodarone |
6 mg/kg IV over 30-60 min |
50-75 mg/h IV infusion over 24 h |
Slower onset and lower conversion rates than other agents Major side effect is hypotension |
|
Vernakalant* |
3 mg/kg IV over 10 min (max dose 339 mg) |
If conversion does not occur within 15 min after end of initial infusion, second dose of 2 mg/kg IV over 10 min (max dose 226 mg) can be given |
Converts about 60% of recent-onset AF Low incidence of bradycardia or hypotension in patients without heart failure Caution in patients with heart failure |
The currently available antiarrhythmic agents used for chemical conversion of recent-onset AF are, to varying degrees, proarrhythmic and myocardial depressants. The patient should be on continuous cardiac rhythm monitoring during the IV infusion and the infusion should be stopped if there is evidence of impaired electrical conduction (prolongation of the QRS or QT intervals) or ventricular arrhythmias (torsades de pointes or ventricular tachycardia). A novel atrial-selective antiarrhythmic, vernakalant, is available for use in Europe and may obtain FDA approval for use in the United States after additional study. In ED patients with recent-onset AF, placebo-controlled and open-label trials in six countries found that 60% of patients converted to sinus rhythm within 90 minutes after the infusion.44 The adverse side effect of vernakalant is generally tolerable in patients without a history of heart failure, but the incidence of hypotension and ventricular arrhythmias in patients with heart failure is about 17% and 7%, respectively.
Synchronized electrical cardioversion is an acceptable alternative for patients seen in the ED with recent-onset AF with a success rate of 70-90% for conversion to sinus rhythm.45,46 Electrical cardioversion can be performed using two different electrical waveforms: monophasic and biphasic. Biphasic shocks have a higher success rate than monophasic shocks, so biphasic shocks are preferred. About 60% can be converted with 100 J, and more than 80% can be converted with 200 J. Some patients may require a larger amount of energy. When possible, procedural sedation with the attendant monitoring and resuscitation equipment should be available. As noted before, some patients with recent-onset AF will undergo spontaneous conversion and a "wait and see" approach is also an acceptable alternative to patients who might be eligible for ED electrical cardioversion.47
A trial of chemical conversion with IV procainamide followed by electrical cardioversion if needed has been named the "Ottawa Aggressive Protocol" after the city of the home institution.48 This approach was able to achieve a 90% conversion rate for ED patients with recent-onset atrial fibrillation. Relapse was seen in about 8% of patients within a week after discharge. Other studies of ED cardioversion for recent-onset AF have found it is safe to discharge patients home if they are stable and do not have another condition that necessitates hospitalization.49 The observed relapse rate for AF in these studies was 3% to 17%, so patients should be informed of this possibility.
For patients with duration of symptoms longer than 48 hours, one option is to assess for the presence of left atrial thrombus with a transesophageal echo, and if there is no evidence of a thrombus on imaging, then proceeding with cardioversion. Another acceptable option would be to fully anticoagulate for 4 weeks before attempting cardioversion and to maintain oral anticoagulation for 6-12 weeks after successful cardioversion to prevent thromboembolic events. For those patients with failed cardioversion or early recurrence, rate control with long-term anticoagulation is one reasonable strategy, with the option for repeat cardioversion at a later date.
Post-ED Management
There are three long-term strategic objectives in the management of AF: rate control, rhythm control, and prevention of thromboembolism. These three objectives can be achieved by pharmacological as well as non-pharmacological means.
Heart Rate Control vs. Rhythm Control
Patients with symptomatic AF that has been present for some time need medication for rate control and anticoagulation to prevent embolic events with the long-term goal of conversion to sinus rhythm. If rate control does not offer symptomatic relief, then restoration of sinus rhythm becomes more important.
Randomized trials comparing outcomes of rhythm to rate-control treatment strategies in patients with AF have found that there is no difference in mortality or stroke rate between patients with rate-controlled strategies vs. rhythm control. The AFIRM trial (Atrial Fibrillation Follow up Investigation of Rhythm Management) found rate control to be non-inferior to rhythm control for the prevention of death and morbidity.50 There is conflicting data regarding the quality of life on the two treatment strategies.
Rate control may be a reasonable treatment strategy in elderly patients with persistent AF. In younger patients, particularly those with paroxysmal AF, rhythm control may be a better alternative. What constitutes adequate rate control is not clearly defined; however, heart rate between 60-80 bpm at rest and between 90-115 bpm during moderate exercise is considered an acceptable goal. A more recent study, RACE II (Rate Control Efficacy in Permanent Atrial Fibrillation), evaluated clinical outcomes between more lenient controls of heart rate in patients with AF vs. strict heart rate control.51 Strict heart rate control was defined as resting heart rate of < 80 bpm and a heart rate of < 110 bpm with moderate exercise. Lenient control was resting heart rate of < 110 bpm. In this study at 3 years, the primary endpoints were death from cardiovascular causes, hospitalization for heart failure, stroke, systemic embolization, bleeding, and life-threatening arrhythmias. At 3 years, the cumulative incidence of primary outcomes was statistically significant, with 12.9% in the lenient group vs. 14.9% in the strict heart rate control group. Based on these findings, the most recent updated management guidelines by the American College of Cardiology advocate a more lenient heart rate control with a caution that patients with normal left ventricular function in whom a lenient heart control strategy is adopted should have LV function monitored for deterioration.
Patients with tachycardia that is allowed to run unabated for sometime may have deterioration in left ventricular function. The cardiomyopathy that ensues following a sustained period of tachycardia is referred to as tachycardia-induced cardiomyopathy. This type of cardiomyopathy usually resolves within 6 months of achieving adequate control of heart rate.52
In patients with systolic dysfunction, defined as LV function less than 35%, there was no difference in clinical outcomes between patients treated for rhythm control vs. those treated for rate control.53
Anticoagulation in AF
Thromboembolism is the most important complication of AF.54 Non-valvular AF accounts for about one-third of all strokes per year in patients older than 65 years.55 Even in patients with AF who do not have clinical evidence of stroke, about 6-25% of patients will have evidence of intra-atrial thrombus on TEE.56 The risk of stroke in patients without heart disease and younger than 60 years is 0.5% per year.3 The risk of stroke increases with age and is about 4.2% for those aged between 70-79 years and 5.1 between 80-89 years.57 Known risk factors that increase the risk for stroke include advanced age, female gender, hypertension, left ventricular dysfunction, diabetes mellitus, and previous history of stroke. Heart failure can cause up to a 4.3-fold increase in stroke.58 In patients with non-valvular AF, prior stroke or TIA is the strongest independent risk factor for stroke. There is no difference in the stroke rates between patients with paroxysmal AF and those with permanent AF. In the stroke prevention in Atrial Fibrillation III trial, the annual rate of ischemic stroke in those with paroxysmal AF was 3.2%, while in those with permanent AF it was 3.3%.59
Embolic Risk Stratification
Echocardiography is a valuable tool for risk stratification of AF patients who are at an increased risk for developing thromboembolic events. Patients with impaired LV dysfunction, enlarged left atrial dimensions, and clot in the left atrial appendage are at increased risk. Other features on echocardiography indicative of high risk for embolic potential include spontaneous echo contrast indicating low velocity flow in the atrium and complex atheromatous plaque in the thoracic aorta.
Table 6: Assessment of Stroke Risk in Nonvalvular Atrial Fibrillation, the CHADDS2 Risk Scores60
Criteria Value
Prior stroke or TIA 2
Age above 75 years 1
Hypertension 1
Diabetes mellitus 1
Heart failure 1
Table 7: Oral Drugs Used for Suppression of Atrial Fibrillation
|
Drug |
Typical Daily Dose (Adult > 70 kg) |
Comments |
|
Dronedarone |
100 mg PO BID |
Used in lone AF |
|
Propranolol |
20-60 mg PO QID |
Used in lone AF |
|
Metoprolol |
25-100 mg PO BID |
Used in lone AF |
|
Flecainide |
100 mg PO BID |
Avoid in patients with coronary artery disease or cardiomyopathy |
|
Propafenone |
150-300 mg PO TID |
Avoid in patients with coronary artery disease or cardiomyopathy |
|
Sotalol |
80 mg PO BID |
Proarrhythmic, especially if QTc prolonged, history of torsades de pointes, or hypokalemia |
|
Dofetilide |
500 mcg PO BID |
Proarrhythmic Contraindicated if QTc > 440 msec or CrCl < 20 mL/min |
|
Amiodarone |
600 mg PO daily |
There are various assessment tools that have been used to identify patients at risk for developing thromboembolic events. One of the most commonly used tools for categorizing the risk of patients for thromboembolic disease is CHADS2 (Cardiac Failure, Hypertension, Age, Diabetes, Stroke [doubled]).60 (See Table 6.) Patients with two or more points on the CHADS2 score have greater than a 4% yearly risk of stroke and are generally considered to be candidates for anticoagulation.
Oral Anticoagulant Therapy
Patients with permanent AF should be considered for anticoagulation therapy to prevent thromboembolism. Aspirin therapy has modest protection against stroke in patients with AF.61-63 Oral anticoagulation with warfarin is much more effective in the prevention of stroke in patients with AF than aspirin alone.64 The goal of warfarin treatment is the INR range of 2.0 and 3.0.65 At this INR, there is balance between thromboembolic protection and adverse risk of bleeding.
Two recent studies found that the combination of aspirin and clopidogrel in AF did not show improvement in clinical outcomes when compared with warfarin.66,67 However, a combination of aspirin and clopidogrel can be considered in patients who cannot tolerate other oral anticoagulants. This combination has a slight increase in the bleeding risk. A combination of aspirin, warfarin, and clopidogrel is acceptable combination therapy in patients with AF, prosthetic valves, and recently placed drug-eluting stents.68 A new antithrombotic, dabigatran, recently was approved by the FDA for thromboembolic protection in patients with AF. This drug has the advantage of not requiring serial monitoring of the degree of anticoagulation like warfarin. Dabigatran at 150 mg twice-daily dosage was associated with lower rates of strokes and systemic embolization when compared to warfarin and was also associated with a comparable risk of bleeding.69
Oral Antiarrhythmic Drugs for Suppression of Atrial Fibrillation
Oral antiarrhythmic therapy can be used to suppress episodes or recurrence of AF. (See Table 7.) These drugs can be used on a chronic basis, or taken immediately at an increased amount by the patient at home upon the first symptoms of a recurrence, a protocol termed "pill in a pocket." The two agents most commonly used in this "pill in a pocket" protocol are flecainide 300 mg or propafenone 600 mg. Antiarrhythmic medications enhance the chances of successful restoration to sinus rhythm by about 90% if therapy is initiated early and in adequate doses.70
Before the initiation of antiarrhythmic drugs, reversible causes of AF should be identified and corrected. Selection of an appropriate agent is based on patient safety. Patients should be assessed for the presence or absence of underlying heart disease.71
In patients with lone AF, dronedarone or a beta-blocker can be tried as initial therapy. Flecainide, propafenone, and sotalol are as effective. Amiodarone and dofetilide are reasonable alternatives. Patients taking antiarrhythmic medications must be aware of the drug–drug interactions and potential adverse effects. Patients should be monitored for QRS-complex and QT-interval prolongation.
Selection of Antiarrhythmic Drugs in Special Situations
Heart Failure. Patients with heart failure are prone to ventricular arrhythmias, so amiodarone or dofetilide are acceptable options in these patients.72,73
Coronary Artery Disease. In patients with stable CAD, beta-blockers are the drug of choice. Sotalol is also acceptable.
Hypertensive Heart Disease. Patients with left ventricular hypertrophy are prone to torsades. Dronedarone is an antiarrhythmic medication that has been recently approved by the FDA for maintenance of sinus rhythm. It is similar to amiodarone but lacks an iodine moiety and therefore lacks iodine-related toxicities associated with the use of amiodarone. Dronedarone was found to increase the time to recurrence of AF.74 It also slows the ventricular rate by 11-13 bpm.75 Dronedarone is less efficacious than amiodarone but has a better tolerability profile. It is contraindicated in patients with recent decompensated heart failure or left ventricular systolic dysfunction.76 More recently there have been reports of dronedarone causing hepatic failure.77 Serial monitoring of liver function tests are indicated in patients on this medication.
Summary
AF is the most common sustained arrhythmia seen in clinical practice. The prevalence of AF is increasing due to the aging of the population and the increased survival of patients with chronic heart disease. Hemodynamic impairment and thromboembolic events related to AF result in significant morbidity, mortality, and cost. The main goals of management of patients with AF are threefold: control of symptoms through the maintenance of sinus rhythm, prevention of tachycardia-mediated cardiomyopathy through adequate rate control, and protection from thromboembolic complications by appropriate anticoagulation in patients who are at increased risk. Early referral to a cardiologist for specialized therapies is an important element in the management of AF patients who are refractory to routine medical management.
References
1. Definition of terms related to cardiac rhythm. Am Heart J 1978;95:796-806.
2. Sopher SM, Camm AJ. Atrial fibrillation: Maintenance of sinus rhythm versus rate control. Am J Cardiol 1996;77:24A-37A.
3. Kopecky SL, et al. The natural history of lone atrial fibrillation. A population-based study over three decades. N Engl J Med 1987;317:669-674.
4. Kannel WB, et al. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: Population-based estimates. Am J Cardiol 1998;82:2N-9N.
5. Go AS, et al. Prevalence of diagnosed atrial fibrillation in adults: National implications for rhythm management and stroke prevention: The AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA 2001;285: 2370-2375.
6. Friberg J, et al. Rising rates of hospital admissions for atrial fibrillation. Epidemiology 2003;14:666-672.
7. Furberg CD, et al. Prevalence of atrial fibrillation in elderly subjects (the Cardiovascular Health Study). Am J Cardiol 1994;74:236-241.
8. Feinberg WM, et al. Prevalence, age distribution, and gender of patients with atrial fibrillation. Analysis and implications. Arch Intern Med 1995;155: 469-473.
9. Phillips SJ, et al. Prevalence of cardiovascular disease and diabetes mellitus in residents of Rochester, Minnesota. Mayo Clin Proc 1990;65:344-359.
10. Mary-Rabine L, et al. The relationship of human atrial cellular electrophysiology to clinical function and ultrastructure. Circ Res 1983;52:188-199.
11. Psaty BM, et al. Incidence of and risk factors for atrial fibrillation in older adults. Circulation 1997;96:2455-2461.
12. Robinson K, et al. Atrial fibrillation in hypertrophic cardiomyopathy: A longitudinal study. J Am Coll Cardiol 1990;15:1279-1285.
13. Allessie M, et al. Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovasc Res 2002;54: 230-246.
14. Frustaci A, et al. Histological substrate of atrial biopsies in patients with lone atrial fibrillation. Circulation 1997;96: 1180-1184.
15. Bailey GW, et al. Relation of left atrial pathology to atrial fibrillation in mitral valvular disease. Ann Intern Med 1968; 69:13-20.
16. Hobbs WJ, et al. Reversal of atrial electrical remodeling after cardioversion of persistent atrial fibrillation in humans. Circulation 2000;101:1145-1151.
17. Zipes DP. Electrophysiological remodeling of the heart owing to rate. Circulation 1997;95:1745-1748.
18. Spach MS. Non uniform anisotropic cellular coupling as a basis for reentrant arrhythmias. In: DiMarco JP, Prystowsky EN, eds. Atrial Arrhythmias: State of the Art. Armonk, NY: Futura; 1995: 123-147.
19. Prystowsky EN, Katz AM. Atrial fibrillation. In: Textbook of Cardiovascular Medicine. Philadelphia: Lippincott-Raven; 1998:1661.
20. White CW, et al. The effects of atrial fibrillation on atrial pressure-volume and flow relationships. Circ Res 1982;51: 205-215.
21. Kamkin A, et al. Mechanically induced potentials in atrial fibroblasts from rat hearts are sensitive to hypoxia/reoxygenation. Pflugers Arch 2003;446:169-174.
22. Spach MS. Non-uniform anisotropic cellular coupling as a basis for reentrant arrhythmias. 1995:123-147.
23. Chen PS, et al. New observations on atrial fibrillation before and after surgical treatment in patients with the Wolff-Parkinson-White syndrome. J Am Coll Cardiol 1992;19:974-981.
24. Klein GJ, et al. Ventricular fibrillation in the Wolff-Parkinson-White syndrome. N Engl J Med 1979;301:1080-1085.
25. Dreifus LS, et al. Recurrent Wolff-Parkinson-White tachycardia in an infant: Successful treatment by a radio-frequency pacemaker. Am J Cardiol 1971;28: 586-591.
26. Creswell LL, et al. Hazards of postoperative atrial arrhythmias. Ann Thorac Surg 1993;56:539-549.
27. Prystowsky EN. Tachycardia-induced-tachycardia: A mechanism of initiation of atrial fibrillation. In: DiMarco JP, Prystowsky EN, eds. Atrial Arrhythmias: State of the Art. Armonk, NY: Futura; 1995.
28. Benjamin EJ, et al. Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study. JAMA 1994;271:840-844.
29. Tsai CF, et al. Initiation of atrial fibrillation by ectopic beats originating from the superior vena cava: Electrophysiological characteristics and results of radiofrequency ablation. Circulation 2000;102:67-74.
30. Schmitt C, et al. Biatrial multisite mapping of atrial premature complexes triggering onset of atrial fibrillation. Am J Cardiol 2002;89:1381-1387.
31. Lin WS, et al. Catheter ablation of paroxysmal atrial fibrillation initiated by non-pulmonary vein ectopy. Circulation 2003;107:3176-3183.
32. Kerr C, et al. Follow-up of atrial fibrillation: The initial experience of the Canadian Registry of Atrial Fibrillation. Eur Heart J 1996;17 Suppl C:48-51.
33. Dorian P, et al. The impairment of health-related quality of life in patients with intermittent atrial fibrillation: Implications for the assessment of investigational therapy. J Am Coll Cardiol 2000;36:1303-1309.
34. Wood MA, et al. Clinical outcomes after ablation and pacing therapy for atrial fibrillation: A meta-analysis. Circulation 2000;101:1138-1144.
35. Jung W, Luderitz B. Quality of life in patients with atrial fibrillation. J Cardiovasc Electrophysiol 1998;9(8 Suppl):S177-186.
36. Hohnloser SH, et al. Rhythm or rate control in atrial fibrillation Pharmacological Intervention in Atrial Fibrillation (PIAF): A randomised trial. Lancet 2000;356:1789-1794.
37. Buccelletti F, Carroccia A, Marsiliani D, et al. Utility of routine thyroid-stimulation hormone determination in new-onset atrial fibrillation in the ED. Am J Emerg Med 2011;29:1158-1162.
38. Savelieva I, Camm AJ. Clinical relevance of silent atrial fibrillation: Prevalence, prognosis, quality of life, and management. J Interv Card Electrophysiol 2000;4:369-382.
39. Falk RH. Proarrhythmia in patients treated for atrial fibrillation or flutter. Ann Intern Med 1992;117:141-150.
40. Vinson DR, Hoehn T, Graber DJ, et al. Managing emergency department patient with recent-onset atrial fibrillation. J Emerg Med 2012;42:139-148.
41 Fundaro C, Galli A, Paglia S, et al. Atrial fibrillation in emergency department: Prevalence of sinus rhythm 1 week after discharge. Emerg Med J 2011 March 25 [Epub]
42. Danias PG, et al. Likelihood of spontaneous conversion of atrial fibrillation to sinus rhythm. J Am Coll Cardiol 1998;31:588-592.
43. Reisinger J, et al. Prospective comparison of flecainide versus sotalol for immediate cardioversion of atrial fibrillation. Am J Cardiol 1998;81:1450-1454.
44. Stiell IG, Dickinson G, Butterfield NN, et al. Vernakalant hydrochloride: A novel atrial-selective agent for the cardioversion of recent-onset atrial fibrillation in the emergency department. Acad Emerg Med 2010;17:1175-1182. Erratum in Acad Emerg Med 2011;18:224.
45. Cristoni L, Tampieri A, Mucci F, et al. Cardioversion of acute atrial fibrillation in the short observation unit: Comparison of a protocol focused on electrical cardioversion with simple antiarrhythmic treatment. Emerg Med J 2011;28:932-937.
46. Bellone A, Etteri M, Vettorello M, et al. Cardioversion of acute atrial fibrillation in the emergency department: A prospective randomized trial. Emerg Med J 2012;29:188-191.
47. Doyle B, Reeves. "Wait and see" approach to the emergency department cardioversion of acute atrial fibrillation. Emerg Med Internat 2011;2011:545023. Epub 2011 Nov 17.
48. Stiell IG, Clement CM, Perry JJ, et al. Association of the Ottawa Aggressive Protocol with rapid discharge of emergency department patients with recent-onset atrial fibrillation or flutter. CJEM 2010;12:181-191.
49. Von Bresser K, Mills AM. Is discharge to home after emergency department cardioversion safe for the treatment of recent-onset atrial fibrillation? Ann Emerg Med 2011;58:517-520.
50. Sherman DG, et al. Occurrence and characteristics of stroke events in the Atrial Fibrillation Follow-up Investigation of Sinus Rhythm Management (AFFIRM) study. Arch Intern Med 2005;165: 1185-1191.
51. Van Gelder IC, et al. Lenient versus strict rate control in patients with atrial fibrillation. N Engl J Med 2010;362:1363-1373.
52. Nerheim P, et al. Heart failure and sudden death in patients with tachycardia-induced cardiomyopathy and recurrent tachycardia. Circulation 2004;110: 247-252.
53. Roy D, et al. Rhythm control versus rate control for atrial fibrillation and heart failure. N Engl J Med 2008;358:2667-2677.
54. Warfarin to prevent thromboembolism in chronic atrial fibrillation. Lancet 1989;1:670.
55. Hinton RC, et al. Influence of etiology of atrial fibrillation on incidence of systemic embolism. Am J Cardiol 1977;40: 509-513.
56. Grimm RA, et al. Should all patients undergo transesophageal echocardiography before electrical cardioversion of atrial fibrillation? J Am Coll Cardiol 1994;23:533-541.
57. Kannel WB, et al. Epidemiologic features of chronic atrial fibrillation: The Framingham study. N Engl J Med 1982;306:1018-1022.
58. Kannel WB, et al. Coronary heart disease and atrial fibrillation: The Framingham Study. Am Heart J 1983;106:389-396.
59. Hart RG, et al. Stroke with intermittent atrial fibrillation: Incidence and predictors during aspirin therapy. Stroke Prevention in Atrial Fibrillation Investigators. J Am Coll Cardiol 2000;35:183-187.
60. van Walraven C, et al. A clinical prediction rule to identify patients with atrial fibrillation and a low risk for stroke while taking aspirin. Arch Intern Med 2003;163:936-943.
61. Secondary prevention in non-rheumatic atrial fibrillation after transient ischaemic attack or minor stroke. EAFT (European Atrial Fibrillation Trial) Study Group. Lancet 1993;342:1255-1262.
62. Petersen P, et al. Placebo-controlled, randomised trial of warfarin and aspirin for prevention of thromboembolic complications in chronic atrial fibrillation. The Copenhagen AFASAK study. Lancet 1989;1:175-179.
63. Ezekowitz MD, et al. Warfarin in the prevention of stroke associated with nonrheumatic atrial fibrillation. Veterans Affairs Stroke Prevention in Nonrheumatic Atrial Fibrillation Investigators. N Engl J Med 1992;327:1406-1412.
64. Fuster V, et al. ACC/AHA/ESC 2006 guidelines for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 guidelines for the management of patients with atrial fibrillation) developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Europace 2006;8:651-745.
65. Hylek EM, et al. Effect of intensity of oral anticoagulation on stroke severity and mortality in atrial fibrillation. N Engl J Med 2003;349:1019-1026.
66. Connolly SJ, et al. Effect of clopidogrel added to aspirin in patients with atrial fibrillation. N Engl J Med 2009;360:2066-2078.
67. Connolly S, et al. Clopidogrel plus aspirin versus oral anticoagulation for atrial fibrillation in the Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE W): A randomised controlled trial. Lancet 2006;367:1903-1912.
68. Holmes DR, Jr., et al. Combining antiplatelet and anticoagulant therapies. J Am Coll Cardiol 2009;54:95-109.
69. Connolly SJ, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009;361:1139-1151.
70. Boriani G, et al. Conversion of recent-onset atrial fibrillation to sinus rhythm: Effects of different drug protocols. Pacing Clin Electrophysiol 1998;21(11 Pt 2):2470-2474.
71. Prystowsky EN. Management of atrial fibrillation: Therapeutic options and clinical decisions. Am J Cardiol 2000;85: 3D-11D.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
