Hypoglycemia
Hypoglycemia
Authors:
Cara O'Shaughnessey, DO, Diabetes Fellow, Ohio University Heritage College of Osteopathic Medicine, O'Bleness Health System, Athens, OH
Jay H. Shubrook Jr, DO, FACOFP, FAAFP, Associate Professor of Family Medicine and Director of Diabetes Fellowship, Ohio University Heritage College of Osteopathic Medicine, Athens, OH
Peer Reviewer:
Ademola Adewale MD, FAAEM, Director of Research and Medical Simulation, Assistant Program Director, Florida Hospital Emergency Medicine Residency, Orlando, FL
Authors' Note
In November 2011, the Centers for Disease Control and Prevention presented data on emergency hospitalizations because of adverse drug reactions. The report showed that insulin and oral hypoglycemic agents caused nearly 25% of the medication-induced hospitalizations in older adults in the United States. A total of 24,520 hospitalizations resulted from these medications. (www.nejm.org/doi/full/10.1056/NEJMsa1103053).
Hypoglycemia is common in diabetes. It can result from insulin treatment but also can occur in those taking oral medications. Although intensive glucose control has been shown to prevent microvascular complications and potentially decrease macrovascular complications in some populations, this benefit must be balanced with the detrimental effects of hypoglycemia. In fact, hypoglycemia may be the largest barrier to normalization of glucose control in diabetes.
The actual incidence of hypoglycemia is difficult to determine due to different assigned thresholds in studies. The American Diabetes Association (ADA) Workgroup on Hypoglycemia provided recommendations for defining hypoglycemia. Knowledge and use of the recommended levels in clinical practice and research studies may provide a better understanding of the commonality of hypoglycemia.
In patients with diabetes mellitus, hypoglycemia almost always is the result of a medical treatment. Although this is more likely to happen with insulin and sulfonylureas, it can happen with any medication. An understanding of how medications and insulin contribute to hypoglycemia will allow the primary care physician to optimize treatment protocols.
Hypoglycemia also can occur in people without diabetes. These spells can range from a nuisance to a serious condition that may require surgery. For example, those who have hypoglycemia during a period of fasting or hypoglycemia that is associated with weight gain should be evaluated for an insulinoma. Reactive hypoglycemia and alimentary hypoglycemia are more common and can be treated with dietary modification.
This article will review the spectrum of hypoglycemic disorders in clinical practice. It will include an overview of the classification of hypoglycemia, clinical presentations, evaluation, treatment, and prevention.
Introduction
Iatrogenic hypoglycemia as a result of the treatment of diabetes has been a concern since the first use of insulin in 1922.1,2 Treatment-induced hypoglycemia continues to be an important problem in patients with type 1 diabetes and in many patients with advanced type 2 diabetes.3-6 Hypoglycemia causes recurrent morbidity, and it is even sometimes fatal.3 The Diabetes Control and Complications Trial (DCCT) showed that intensive glycemic therapy decreases the frequency of long-term complications at the expense of increased risk of hypoglycemia.1,7,8 The ultimate goal for the glycemic management of diabetes is a lifetime of euglycemia without hypoglycemia.1 To date, this has not been attainable. Although there have been improvements in the glycemic management of diabetes, hypoglycemia continues to be a limiting factor leading to partially successful therapeutic regimens.9
Definition
In 1938, American surgeon Allen Whipple defined hypoglycemia by the following triad: typical signs and symptoms of hypoglycemia; documented low glucose level; and correction of signs and symptoms with normalization of glucose levels.10
Many glucose levels have been used as a cutoff for hypoglycemia. This variance has made it difficult to determine the true incidence of hypoglycemia as study populations cannot be compared. Some have used 70 mg/dL (3.9 mmol/L) as the lower limit of normal. Others favor a value of < 63 mg/dL (3.5 mmol/L) to avoid overclassification of hypoglycemia in asymptomatic patients.11,12 Other studies have defined a glucose level of < 56 mg/dL as mild and moderate as low as 36 mg/dL. In the clinical setting, many physicians have defined mild hypoglycemia as a glucose level < 70 mg/dL and severe hypoglycemia < 50 mg/dL (2.8 mmol/L). This makes it extremely difficult to make direct comparisons when the limits used in studies are not seen or reported in practice. Further, people in studies often are monitored closely and have access to early interventions, while those in the community typically do not seek care for these episodes (unless severe) or until their next appointment.
In 2005, the ADA Workgroup on Hypoglycemia defined hypoglycemia in patients with diabetes as "all episodes of an abnormally low plasma glucose concentration (with or without symptoms) that expose the individual to harm."14 The workgroup recommended that patients with drug-treated diabetes should become concerned about developing hypoglycemia when their self-monitored blood glucose level (SMBG) is < 70 mg/dL14 or is falling rapidly,3 although the cutoff value has been debated. The plasma glucose concentration of 70 mg/dL was used as it approximates the lower limit of the fasting non-diabetic glucose concentration range and the normal glycemic threshold for glucose counter-regulatory hormone activation.4 This value also is low enough to reduce glycemic defenses against further hypoglycemia in non-diabetic individuals3,13,15 and is higher than the glycemic level required to produce symptoms of hypoglycemia (~ 55 mg/dL [3.0 mmol/L]) or impair brain function in those without diabetes.16,17 Despite significant advances, glucose meters are not always precise, especially at low plasma glucose levels3,18 and can have an acceptable error rate of up to 16%. Therefore, a cutoff value of 70 mg/dL provides the patient with time to prevent a symptomatic hypoglycemic episode as well as some margin for potential inaccuracy of glucose meter readings.3 (See Table 1.)
Table 1: Types of Hypoglycemia

Clinical Classification
The ADA Workgroup proposed a classification of hypoglycemia in diabetes that includes the following: severe, documented symptomatic, asymptomatic, probable symptomatic, and relative hypoglycemia.14
Severe hypoglycemia requires the assistance of another individual to provide hypoglycemic treatment but does not require the measurement of a plasma glucose level during the event. Recovery of neurologic symptoms with a normal plasma glucose level provides sufficient evidence that a severe hypoglycemic event took place. The authors recommend that a glucose < 50 mg/dL also should be considered severe regardless of the signs or symptoms present.
An event that consists of the typical symptoms of hypoglycemia with a measured plasma glucose concentration < 70 mg/dL is classified as documented symptomatic hypoglycemia. This makes up the majority of hypoglycemic episodes and can occur in people with diabetes regardless of treatment and even in people who do not have diabetes.
Asymptomatic hypoglycemia is classified as an event with a measured plasma glucose concentration of < 70 mg/dL but without the typical symptoms of hypoglycemia. This is especially concerning if glucose levels are lower than 50 mg/dL or if it occurs frequently.
An event that consists of the typical symptoms of hypoglycemia but with a measured plasma glucose concentration > 70 mg/dL is classified as relative hypoglycemia. This is most often seen in people who are chronically hyperglycemic and who have become accustomed to being hyperglycemic. (See Table 2.)
Table 2: ADA Workgroup Definitions of Hypoglycemia in Diabetes
Type of hypoglycemia: Glucose level
Asymptomatic: < 70 mg/dL
Symptomatic: < 70 mg/dL
Severe hypoglycemia: Any low glucose that requires assistance from others
Relative hypoglycemia: > 70 mg/dL but typical symptoms
Frequency
Hypoglycemia occurs frequently in patients with type 1 diabetes.4-6,11,19,20 The average patient with type 1 diabetes experiences two episodes of symptomatic hypoglycemia per week,3,21 and typically one episode of severe, and at least temporarily disabling, hypoglycemia (with seizure or coma) per year.3,21 This does not include the numerous episodes of asymptomatic hypoglycemia with plasma glucose concentrations as low as 50-60 mg/dL (2.8-3.3 mmol/L). People can be low as much as 10% of the time.4,21 Severe hypoglycemic events have been reported to range from 62 to 170 episodes per 100 patient years in type 1 diabetes.3,4,21 In the Diabetes Control and Complications Trial (DCCT), patients with type 1 diabetes in the intensive therapy group had a higher overall rate of severe hypoglycemia compared to the conventional therapy group (61.2 vs 18.7 per 100 patient-years, respectively).8
Hypoglycemia also may affect patients with type 2 diabetes. This is most likely to occur in those treated with either insulin or insulin secretagogues.3-5,19,21 Hypoglycemia rates approach those of type 1 diabetes as the disease becomes more advanced and insulin deficiency ensues.3-5 The UK Hypoglycemia Study Group reported that the prevalence of severe hypoglycemia in patients with type 2 diabetes treated with insulin for < 2 years was only 7%, while the prevalence of severe hypoglycemia with type 2 diabetes > 5 years increased to 25%.22 The overall event rate for hypoglycemia in insulin-treated type 2 diabetes is approximately 30% of that in type 1 diabetes, according to a population-based, prospective study by Donnelly et al (1600 vs. 2400 per 100 patient-years, respectively).21 However, since there are 20 times more people with type 2 diabetes, most episodes of iatrogenic hypoglycemia will occur in patients with type 2 diabetes.
Since most patients with type 2 diabetes ultimately will require treatment with insulin, the rate of hypoglycemia increases with duration of diabetes. Tight glycemic control also is associated with an increased incidence of symptomatic hypoglycemia in patients with type 2 diabetes, as shown in three randomized controlled trials of intensive glycemic therapy.23-25 Another retrospective study found that the elderly (age 65 years or older) had nearly twice as many hypoglycemic episodes as those younger than 65 years.26
Pathophysiology
Normally, hypoglycemia is prevented or promptly ameliorated by redundant glucose counter-regulatory mechanisms.3,17 The critical physiological mechanisms involved include a marked reduction in insulin secretion as glucose levels drop within the physiological range; an increase in glucagon secretion or epinephrine secretion (if the former is absent) as glucose levels drop just below the physiological range; and an increase in cortisol and growth hormone secretion, both occurring with prolonged hypoglycemia.3 Plasma glucose levels will continue to drop if these defenses fail to halt the episode. When the plasma glucose concentration reaches 55 mg/dL (3.0 mmol/L) or less, hypoglycemic symptoms trigger the behavioral defense of food consumption.3,16,17
With the above physiologic defenses in mind, hypoglycemia in patients with type 1 or advanced type 2 diabetes is the consequence of insulin excess,3,9 defective glucose counter-regulation (decreased ability to suppress endogenous insulin production and increase glucagon secretion),9-11 or compromised behavioral defenses against falling plasma glucose concentrations.3,20
Table 3: Physiologic Response to Hypoglycemia

Table 4: Normal Response vs Diabetes Response to Hypoglycemia

Early in the course of type 2 diabetes, the incidence of hypoglycemia is relatively low, even with insulin treatment, because the above glycemic defenses are intact. However, with long-standing type 2 diabetes, b-cell failure leads to absolute endogenous insulin deficiency,22 as is seen in type 1 diabetes. Patients with type 1 and advanced type 2 diabetes become critically dependent on the third defense, epinephrine secretion, because they have lost the insulin and glucagon defenses.10,27,28 The epinephrine response to hypoglycemia often becomes attenuated in these patients,10,27-29 leading to the clinical syndrome of compromised glucose counter-regulation and increasing the risk of severe hypoglycemia by 25-fold.29 The glycemic thresholds for epinephrine activation can be shifted to lower plasma glucose levels due to recent and recurrent hypoglycemia27-28 as well as other potentially reversible factors including exercise and sleep.4,21,31-32 (See Tables 3 and 4, Figure 1.)
Figure 1: The Physiologic and Behavioral Responses to Dropping Glucose
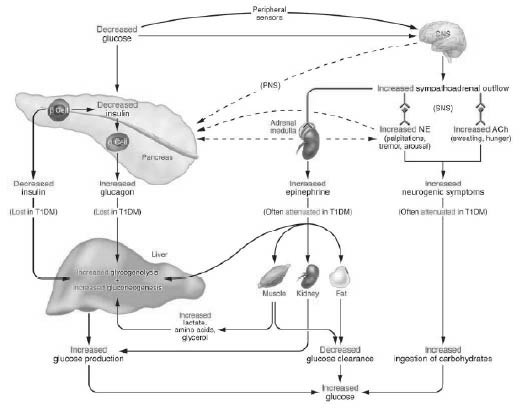
Reprinted with permission: Cryer PE. J Clin Invest 2006;116:1471-1743.
The most serious form of an attenuated response to the normal defenses to hypoglycemia is the syndrome of hypoglycemia-associated autonomic failure (HAAF). It is the result of the reduced sympathoadrenal, predominantly sympathetic neural response to hypoglycemia failure.10,31-32 The key concept of HAAF in patients with type 1 and advanced type 2 diabetes is that antecedent hypoglycemia leads to a vicious cycle of recurrent hypoglycemia by causing further impairment of glucose counter-regulation to subsequent hypoglycemia, thus leading to hypoglycemia unawareness.3,10 HAAF is a functional disorder that can be induced by prior hypoglycemia and reversed by avoidance of hypoglycemia for at least 2-3 weeks.10 Hypoglycemia unawareness is defined as the onset of neuroglycopenia before the appearance of autonomic warning symptoms.30 Patients with hypoglycemia unawareness lose the adrenergic symptoms that previously triggered the behavioral defense to consume carbohydrates and correct developing hypoglycemia.3,4 (See Figure 2.)
Table 5: Signs and Symptoms of Hypoglycemia

Figure 2: Hypoglycemia-Associated Autonomic Failure
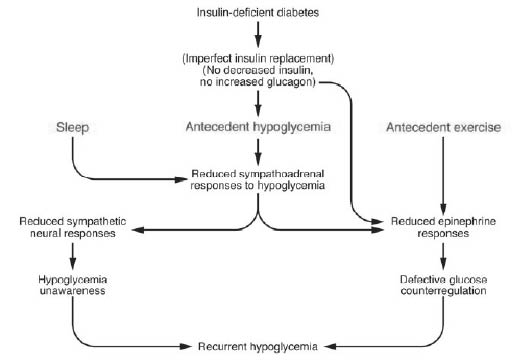
Reprinted with permission: Cryer PE. J Clin Invest 2006;116:1471-1743.
Hypoglycemia unawareness occurs more frequently in patients who are older, in those who achieve tight glycemic control, and during times of increased insulin sensitivity such as exercise and sleep.33 Hypoglycemia also begets hypoglycemia, so those who suffer from frequent hypoglycemic episodes or those who have autonomic diabetic neuropathy are more likely to have future episodes of hypoglycemia. In addition, hypoglycemic unawareness increases the risk for severe hypoglycemia by 6-fold.34 (See Table 5.)
Nocturnal hypoglycemia is common in patients with type 1 diabetes and is usually asymptomatic.34-35 Almost 50% of all episodes of severe hypoglycemia in patients with type 1 diabetes occur at night during sleep.35 Nocturnal hypoglycemia, a form of hypoglycemia unawareness, can lead to disruption of sleep and delays in correction of the hypoglycemia.3,35 A study that looked at continuous glucose monitoring at night in patients with type 1 diabetes showed that nocturnal hypoglycemia occurred on more than half the nights studied, with an average duration over 2.5 hours.36 Unsuspected recurrent episodes of nocturnal hypoglycemia play a role in the pathophysiologic mechanisms of hypoglycemia unawareness and HAAF by reducing the sympathoadrenal responses to subsequent hypoglycemia.35
Symptoms and Signs
Hypoglycemia may cause adrenergic and/or neuroglycopenic symptoms, although these can sometimes be nonspecific and relatively insensitive.9 Adrenergic symptoms may include tremors, palpitations, anxiety, arousal (catecholamine-mediated, adrenergic), sweating, hunger, and paresthesias (acetylcholine-mediated, cholinergic).37-38 Neuroglycopenic symptoms may include cognitive impairment, behavioral/personality changes, difficulties speaking, dizziness, blurred vision, weakness, incoordination, hemiplegia, arrhythmias, seizures, coma, and even death.38-39 Common signs of hypoglycemia include diaphoresis and pallor,3 as well as slight elevations in heart rate and systolic blood pressure. Neuroglycopenic manifestations are usually recognized by others. These symptoms and signs can be mild, moderate, or severe, depending on how low the glucose falls and a variety of other factors. Manifestations of hypoglycemia may vary from person to person and can even vary within the same patient on different occasions.40 Because the manifestations of hypoglycemia are nonspecific, many episodes go unrecognized. The patient may not notice any symptoms/signs of hypoglycemia, even though these are apparent to others. Older adults tend to have more neuroglycopenic manifestations compared with neurogenic manifestations, and may present with hypoglycemia later in the hypoglycemia process.33
Complications
Hypoglycemia is often the most important limiting factor in the glycemic management of diabetes.3-6,9,29 Iatrogenic hypoglycemia can cause a vicious cycle of recurrent hypoglycemia, resulting in morbidity and preventing the maintenance of euglycemia over time.3-6 Hypoglycemia often causes recurrent physical morbidity, recurrent or persistent psychosocial morbidity, or both in most patients with type 1 diabetes and many with type 2 diabetes.41 In elderly patients with diabetes, even mild episodes of hypoglycemia may lead to adverse outcomes. The neuroglycopenic symptoms of hypoglycemia can increase the risk of falls, fracture, and nursing home placement.42
Because glucose is an obligate metabolic fuel for the brain, hypoglycemia can have potentially devastating effects on the brain.9,16 Hypoglycemia can cause functional brain failure that is corrected in most cases after the plasma glucose concentration is raised.3,16 Recurrent hypoglycemia has been associated with cognitive impairment in young children and elderly patients with diabetes, but the extent of these effects are unknown in other age groups. The DCCT showed that patients with repeated episodes of hypoglycemia did not perform differently in multiple psychosocial and neurobehavioral parameters from those who rarely had hypoglycemia.41 Prolonged and profound hypoglycemia may result in permanent neurological damage and even brain death,16 although these effects are rare.43
It is also rare, but possible, that diabetic hypoglycemia will cause death.43 In patients with type 1 diabetes, hypoglycemic mortality estimates range from 6% to 10%.41,43-44 Although fatal hypoglycemia has been documented in type 2 diabetes, mortality rates are currently unknown.4-5 In large, randomized, controlled trials testing aggressive glycemic control in patients with type 2 diabetes, hypoglycemia occurred more frequently in the intensive therapy groups (A1C of 6.4% to 6.9%).23-25 In the ACCORD trial,23 there were more deaths in the intensive therapy group; and in the VADT trial,25 a history of severe hypoglycemia was a significant predictor of cardiovascular death. Although the exact cause of mortality in these trials is not known, it is plausible that iatrogenic hypoglycemia may have resulted in cardiac arrhythmias, thus possibly contributing to an untimely death.3 The "dead in bed syndrome" is an unexpected death of a person with type 1 diabetes that occurs while sleeping at night3,46 and is thought to be caused by a fatal ventricular arrhythmia.3,46,47 It is believed that the sympathoadrenal response to hypoglycemia may cause a transiently prolonged QT interval and increased QT dispersion, resulting in a fatal arrhythmia.3,46-47 (See Table 6.)
Table 6: Risk Factors for Hypoglycemia
Excess of insulin
Exogenous insulin
Oral secretagogues
Decreased clearance of insulin
Renal insufficiency
Renal failure
Decrease in glucose production
Late effects from alcohol
Critical illness such as intake sepsis
Decreased glucose intake
Missed or late meal
Increased glucose uptake
Exercise independent of insulin
Increased insulin sensitivity
Exercise, sleep
Risk Factors
The pathophysiology of hypoglycemia in patients with type 1 and advanced type 2 diabetes can help provide insight into the potential risk factors and even prevention of hypoglycemia.4 Generally, these risk factors may include insulin excess, defective counterregulatory responses, intensive therapy, as well as others that are multifactorial.3,4
Insulin Excess: Absolute or relative insulin excess may occur in patients with diabetes in the following scenarios leading to hypoglycemia:3,4
- Exogenous insulin or insulin secretagogue (sulfonylureas, meglitinides) doses that are excessive, taken at an unsuitable time, or of the inappropriate type.36 Exogenous insulin and insulin secretagogues are the most common medications associated with hypoglycemia.4,48 The prevalence of mild hypoglycemic symptoms has been reported in 30-50% of patients treated with insulin and 16-20% in patients treated with sulfonylureas.48-51 Insulin secretagogues stimulate endogenous insulin secretion, suppress hepatic and renal glucose production, stimulate glucose utilization, and can cause hypoglycemia even in the setting of intact glycemic defenses if given in sufficient doses. Among the commonly used insulin secretagogues, those with a longer duration of action, such as glyburide, are associated with hypoglycemia more often.48,52-54
- Intensive therapy. Hypoglycemia and hypoglycemia unawareness occur more frequently in patients treated with intensive therapy.8 In fact, hypoglycemia is the most common adverse effect of intensive insulin therapy in both the hospital and outpatient settings.3
Decrease in glucose levels
- Exogenous glucose influx is decreased, which may occur during an overnight fast, or with small, delayed, or missed meals.4
- Endogenous glucose production is decreased, which may occur as a result of alcohol consumption or in patients with sepsis. Alcohol consumption is often the cause of, or a contributing factor to, hypoglycemia seen in patients who present to the emergency department.55 Alcohol inhibits gluconeogenesis and can result in hepatic glycogen depletion and hypoglycemia, especially if there is limited food consumption.3 Sepsis is a relatively common cause of hypoglycemia3,56-57 and is likely due to cytokine-induced inhibition of gluconeogenesis in the setting of glycogen depletion, in addition to cytokine-accelerated glucose utilization.57
- Insulin-independent glucose utilization is increased. Exercise increases glucose utilization by muscle and can cause hypoglycemia in patients treated with insulin.3,58 Even when plasma glucose levels are near normal or moderately elevated at the start of exercise, hypoglycemia can occur both during or shortly after exercise.3,58
- Conditions in which sensitivity to insulin is increased. This is often seen after weight loss, hours after exercise, following improved glycemic control, in the middle of the night, after withdrawal of glucocorticoid therapy,3 and occasionally in patients with type 1 diabetes mellitus who develop adrenal insufficiency.34,39 Among the latter patients, sensitivity to insulin is increased because of loss of the gluconeogenic effect of cortisol and the hyperglycemic effects of epinephrine.32,50
Decreased clearance of insulin
- Insulin clearance is decreased. This is commonly seen in patients with acute or chronic renal failure3 who are treated with insulin or insulin secretagogues. Hypoglycemia in patients with chronic kidney disease may also be due to impaired gluconeogenesis and reduced renal glucose production.3
Defective counter-regulatory responses. Insulin excess alone explains only a minority of episodes of hypoglycemia.3,59 Impaired counter-regulatory response is the primary risk factor for subsequent hypoglycemia.59 People with type 1 diabetes do not have the normal insulin and glucagon responses to hypoglycemia. As they experience hypoglycemia, the glycemic threshold becomes lower for the sympathoadrenal response to subsequent hypoglycemia which leads to defective glucose counter-regulation.10 Risk factors for HAAF include:
- The degree of absolute endogenous insulin deficiency3,19,60-62 that determines the extent of the insulin and glucagon defenses as glucose levels drop.
- A history of severe hypoglycemia, hypoglycemia unawareness, or both.3,19,60-61
- Recent antecedent hypoglycemia, exercise, or sleep.10,63-64
- Long duration of diabetes3,66 and a history of classical diabetic autonomic neuropathy,67-68 which are associated with more severe HAAF.
Other risks tend to be multifactorial, and may not be clearly defined, such as hypoglycemia in hospitalized patients. A recent meta-analysis suggested the greatest mortality occurred in hospitalized patients with spontaneous, not medication-induced, hypoglycemia.69
- These factors may include renal insufficiency, changes in caloric intake without adjustments in medications, excessive metabolic activity in the face of severe illness, inappropriate glucose monitoring, failure to reconcile admission orders, inappropriate continuation of outpatient medications, frequent transfers between hospital units, frequent travel for procedures and interventions, and inappropriate use of an insulin sliding scale.70-72
Prevention
The clinical approach to preventing hypoglycemia while improving glycemic control includes acknowledging the issue, applying the principles of intensive glycemic therapy, and considering the potential risk factors for iatrogenic hypoglycemia.9
Hypoglycemia should be addressed during every patient contact, especially with those who are treated with insulin or an insulin secretagogue. Critical review of the patient's blood glucose readings or continuous glucose sensing data will help detect both the frequency and severity of hypoglycemia, even if the patient does not raise this concern. Patient concerns about hypoglycemia should be addressed and discussed because fear of hypoglycemia can be a major barrier to glycemic control. Patients are more likely to have intentional hyperglycemia in the months after experiencing a frightening episode of severe hypoglycemia.73 Many patients will add unscheduled snacks between meals and bedtime to prevent future hypoglycemic episodes. When ingestion of food is not for nutrition purposes, but rather to prevent lows, it is termed "defensive eating." With the range of medications and insulins available today, the authors of this review feel that defensive eating is no longer necessary. It is far better to make patients' insulin match their lives, rather than make their lives match their insulin.
Unless the cause is easily amendable, frequent or severe hypoglycemia should lead to consideration of significantly changing the treatment regimen based on the review and application of the principles of aggressive glycemic therapy.1 These principles include: patient self-management achieved through education and empowerment, ongoing professional guidance and support, frequent SMBG testing or continuous glucose sensing, flexible and individualized drug regimens, and individualized glycemic goals.3
Diabetes self-management is critically important in the maintenance of glycemic control, especially as the treatment regimen becomes more complex. Patient self-management can be improved through professional guidance and support, which is best provided by a chronic care model conducted by a diabetes care team.74 Patients should be educated about the recognition and treatment of hypoglycemia. They also should be informed that the typical hypoglycemic manifestations may be impaired after an episode of hypoglycemia, especially if severe. Frequent SMBG data can help detect glycemic variability and thus provide appropriate rationale for modification of the treatment regimen. In fact, the best way for a patient to prevent hypoglycemia is to check SMBG levels frequently, be aware of conditions that increase the risk of hypoglycemia, and be prepared to treat hypoglycemia at all times. This also means carrying testing supplies and items to treat hypoglycemia with them all of the time.
Treatment regimens should be flexible and individualized to minimize the risk of hypoglycemia. There have been studies that have identified difference even among medications within the same class. Among the insulin secretagogues, hypoglycemia is less common with glimepiride compared with glyburide.52-54 Analog insulins have less hypoglycemia compared with NPH, regular, or premixed insulin.75-77 Rapid-acting insulin analogs (Lispro, Aspart, Glulisine) have been shown to reduce nocturnal hypoglycemia in patients with type 1 diabetes.75 Long-acting insulin analogs (Glargine, Determir) have been shown to reduce nocturnal hypoglycemia and symptomatic hypoglycemia in patients with type 1 and type 2 diabetes.75-77 Hypoglycemia reduction may involve switching to a different secretagogue or insulin type that has a lower risk of hypoglycemia, or reducing the dose of these medications.
To date it appears insulin pump therapy does not reduce the hypoglycemia risk. According to a recent systematic review of 15 randomized trials, there is no difference of mild, nocturnal, or severe hypoglycemia between continuous subcutaneous insulin infusion and multiple daily injection therapy with insulin analogs.78 However, when the pump uses sensor-augmented continuous subcutaneous insulin infusion, it has been shown to achieve lower A1C levels without an increase in hypoglycemia.79
Ultimately, glycemic goals should be individualized to match the health and life expectancy of the individual patient. Although intensive glycemic therapy has been shown to prevent or delay microvascular complications, including retinopathy, nephropathy, and neuropathy, the risk of hypoglycemia is also increased.7,59,80,81 The ADA suggests a goal HbA1C below 7% for many adults, although a more stringent goal can be sought in select patients as long as it can be achieved without significant hypoglycemia.1 Severe or frequent hypoglycemia is an absolute indication for setting less-aggressive glycemic goals. In addition, patients who are very young or old; patients who have multiple comorbidities, advanced microvascular or macrovascular complications; or patients with a limited life expectancy should have relaxed glucose goals that reduce the risk of hypoglycemia.1
There are a number of special circumstances that can increase the risk of hypoglycemia. These can include exercise-related hypoglycemia and hypoglycemia from alcohol consumption. Hypoglycemia can occur during exercise via noninsulin-mediated glucose uptake. However, the hypoglycemia risk can remain elevated for a number of hours after aerobic exercise. Since hypoglycemia may occur several hours after exercise, other actions may need to be considered, such as eating a bedtime snack to prevent nocturnal hypoglycemia.54 Post-exercise hypoglycemia can be prevented by frequent SMBG testing and insulin dose reduction and/or carbohydrate ingestion prior to or during exercise.
Patients should be informed that they may experience hypoglycemia with alcohol consumption and thus may need to check SMBG more frequently or ingest carbohydrates to prevent this from occurring. For patients with nocturnal hypoglycemia, bedtime snacks have been the traditional approach for prevention, although this has not been shown to be more effective than no snack at all.82 Regular blood glucose monitoring at bedtime or continuous glucose monitoring overnight and the utilization of short- and long-acting insulin analogues may help to prevent or reduce the frequency of nocturnal hypoglycemia.
For patients with hypoglycemia unawareness or one or more episodes of severe hypoglycemia, it is recommended that glycemic goals be raised to avoid hypoglycemia for a 2- to 3-week period.1 This recommendation is based on the findings that avoidance of hypoglycemia for 2-3 weeks has been shown to reverse hypoglycemia unawareness, at least partially, and reduce the risk of future episodes in most affected patients by improving the reduced epinephrine component of defective glucose counter-regulation.83-85 (See Table 7.)
Table 7: Foods to Treat Hypoglycemia
4 oz or ½ cup pure juice or soda
4 teaspoons table sugar
8 oz of nonfat milk
4-5 saltine crackers
3-4 glucose tablets
1 tablespoon honey
5-6 hard candies like life savers
Treatment
Patients with drug-treated diabetes should undertake defensive actions when their plasma glucose concentration is < 70 mg/dL because of the concern of developing hypoglycemia.14 Such actions include repeating the SMBG level to confirm hypoglycemia, consuming carbohydrates, modifying the treatment regimen, and abstaining from tasks such as driving or operating heavy equipment while hypoglycemic.14 (See Table 8.)
Table 8: Treatment of Hypoglycemia

Most episodes of asymptomatic or mild-to-moderate symptomatic hypoglycemia are effectively self-treated by the consumption of carbohydrates. Plasma glucose concentration should be raised to the normal range as quickly as possible in all cases of hypoglycemia. Suh et al showed that post-hypoglycemic glucose reperfusion may contribute to neuronal death in a rodent model of extreme hypoglycemia.86 It is therefore important to avoid post-treatment hyperglycemia, especially after an episode of profound, prolonged hypoglycemia.
The "15-15 rule" can be followed in the majority of cases in adults. This involves the ingestion of 15 g of fast-acting carbohydrates and recheck of SMBG 15 minutes later to ensure that the glucose level is > 70 mg/dL. If the SMBG level is not within the recommended range, the steps should be repeated until it is achieved.1 Items that contain 15 g of fast-acting carbohydrates include: 4 ounces of carbohydrate-containing juice or regular, non-diet soda; 8 ounces of milk; 3-4 glucose tablets; 1 tube of glucose gel; 5-6 hard candies; 2 tablespoons of raisins; 4 or 5 saltine crackers; 4 teaspoons of sugar; and 1 tablespoon of honey or corn syrup. Food that contains fat (like candy bars) or protein (cheese) should be avoided initially, since they slow the absorption of glucose.
In adults, 15-20 g of carbohydrates are usually sufficient to raise blood glucose to a safe range without causing hyperglycemia.1,3 Clinical improvement should occur within 15-20 minutes of treatment. Although most episodes of hypoglycemia are reversed once the glucose level is raised to the normal range, the glycemic response to oral glucose may be transient, especially in basal or intermediate insulin-induced hypoglycemia (often < 2 hours). Therefore, a long-acting carbohydrate with protein, such as a meal or more substantial snack, if the next meal is > 1 hour away, should be consumed shortly after the plasma glucose is raised to prevent recurrent symptoms.1,3 It is also recommended that patients check their SMBG levels serially after treatment with carbohydrates to ensure that they continue to remain within the euglycemic range, as individual responses may vary.3 People usually feel bad when they are hypoglycemic, and rebound hypoglycemia is common due to ingestion of more carbohydrates than needed for the situation. Training patients and their families on a prompt but appropriate response to hypoglycemia may reduce this overshoot.
Patients treated with insulin or a secretagogue in combination with an alpha-glucosidase inhibitor should only consume pure glucose (dextrose) to treat symptomatic hypoglycemia. Alpha-glucosidase inhibitors slow the digestion of other carbohydrates, such as table sugar (sucrose), and thus consumption of non-dextrose carbohydrates will be less effective in raising blood sugar.
In cases of severe hypoglycemia, when the patient is unconscious or is unable/unwilling to ingest carbohydrates, glucagon treatment is necessary and requires the assistance of another individual.3 Relatives and close friends should be trained to recognize the signs and symptoms of hypoglycemia and provide treatment should this complication occur. If hypoglycemia is suspected, a blood glucose level should be checked initially, especially if the patient is unconscious, to ensure that hyperglycemia is not the culprit. If this is not possible, then empiric treatment for hypoglycemia should be performed.3
Glucagon, injected subcutaneously or intramuscularly in a dose of 1 mg in adults, can be lifesaving and should be administered if there is no established intravenous access. Glucagon should be prescribed for all patients at risk for developing severe hypoglycemia, and caregivers or family members should receive education and training for administration.1 To restate this, the authors feel that all patients who are on insulin replacement, regardless of type of diabetes mellitus, should have glucagon available to treat severe hypoglycemia.
Glucagon is a hormone that rapidly counters the metabolic effects of insulin in the liver through stimulation of glycogenolysis and gluconeogenesis. Injected glucagon can raise blood glucose levels by 30-100 mg/dL within minutes and can lead to transient, but significant hyperglycemia.3 Resolution of hypoglycemic symptoms with recovery of consciousness usually occurs within 10-15 minutes after glucagon administration. Nausea and vomiting may follow 60-90 minutes later. Therefore, it is important that a patient be placed onto his side if he is unconscious to avoid the risk of aspiration. Once the patient is awake and able to tolerate oral food, he initially should consume a fast-acting carbohydrate and then a long-acting carbohydrate with protein, since glucagon depletes glycogen stores and potentially can lead to rebound hypoglycemia. If the patient remains unconscious 10 minutes after the glucagon injection, another injection should be given and emergency personnel should be contacted immediately.
There are no efficacy or safety data to guide the management of hypoglycemia if glucagon is not available prior to the arrival of emergency personnel. In the absence of other options, some experts suggest treatment with either glucose gel or cake frosting placed in the buccal mucosa or table sugar placed sublingually. Other experts do not recommend these options since there is no supporting evidence87 and there also is a potential risk of aspiration. Hypothetically, an epinephrine injection will also raise the glucose but should only be used when other evidence-based treatments are not available.
In the hospital, the standard parenteral therapy for hypoglycemia is intravenous (IV) glucose. A standard initial glucose dose is 25 g of 50% glucose (dextrose) IV. Usually, the glycemic response is only transient and thus a subsequent dose or glucose infusion often is required. It is recommended that patients ingest food with carbohydrates as soon as they are able to tolerate food by mouth.3 Even more importantly, if an inpatient experiences hypoglycemia, the treating team should examine the clinical care for a cause. This can be caused by decreased oral intake, comorbid illness, or mistimed oral diabetes or insulin therapy. Some patients who present to the emergency room with hypoglycemia will require hospitalization for prolonged treatment and observation, as commonly seen with sulfonylurea overdoses. Octreotide has also been used to treat hypoglycemia induced by sulfonylureas.88
After an episode of severe hypoglycemia, it is recommended that a fundamental review of the treatment regimen and the glycemic goals be performed, unless the cause was easily explainable.3
Hypoglycemia in Patients without Diabetes
Although hypoglycemia is a common event in persons with diabetes, it is a rare event in those without diabetes because of the effectiveness of the key physiologic defenses in response to declining plasma glucose levels.89 A triad of symptoms, known as Whipple's triad, should be used to diagnose hypoglycemia in persons without diabetes.10 This triad includes symptoms and/or signs consistent with hypoglycemia, a documented low plasma glucose concentration, and relief of the symptoms or signs after the plasma glucose level is raised. Only those non-diabetic individuals with documented Whipple's triad should undergo evaluation and management of hypoglycemia2 to avoid unnecessary workup and costs.
Table 9: Hypoglycemia in Those without Diabetes

Hypoglycemia in patients without diabetes can be categorized clinically based on whether the patient is well- or ill-appearing and/or treated with certain medications (see Table 9).89 In those patients who are well-appearing, hypoglycemia usually is caused by either endogenous hyperinsulinism or accidental, surreptitious, or malicious administration of an insulin secretagogue or insulin.89 Although insulinomas are the prototypical cause of endogenous hyperinsulinemic hypoglycemia, other causes should be considered including nesidioblastosis, noninsulinoma pancreatogenous hypoglycemia syndrome (NIPHS), post-gastric bypass hypoglycemia, autoimmune hypoglycemia, accidental or surreptitious ingestion of an insulin secretagogue, a mutation of the insulin receptor, or exercise-induced hyperinsulinemia.89
It should be noted that there are now case reports of people who have developed hyperinsulinemic hypoglycemia after undergoing bariatric surgery. These cases can occur years after surgery and are difficult to treat. As there is an increasing number of people undergoing these procedures, this is an important potential complication of which physicians should be aware.90
Drugs, including insulin, insulin secretagogues, alcohol, and others, are the most common cause of hypoglycemia, especially in the setting of critical illness or when enteral or parenteral nutrition is disrupted.89 In patients who are ill-appearing and not receiving treatment with culprit medications, renal failure, hepatic failure, cardiac failure, sepsis, and inanition are the most common causes of hypoglycemia.14,89 Although uncommon, hypoglycemia can occur in patients with cortisol or growth hormone deficiencies after prolonged fasting.2 Fasting hypoglycemia also can occur rarely in patients with a non-islet cell tumor, which is typically a large mesenchymal or epithelial tumor (hepatomas, adrenocortical carcinomas, carcinoids), caused by the overproduction of an incompletely processed form of insulin-like growth factor II.2
Reactive hypoglycemia. Reactive hypoglycemia was first described in 1924.91 This condition occurs in people who have a mismatch between insulin secretion and glucose absorption. Reactive hypoglycemia is defined as hypoglycemia that occurs within 4 hours of ingestion of food.20 Patients who experience reactive hypoglycemia typically initially will experience mild early post-meal hyperglycemia, and this will be followed with a vigorous insulin response with a 75 mg/dL drop in glucose that overshoots the normal range and results in mild hypoglycemia. Although the recommended diagnostic workup is controversial,92 many clinicians will utilize a 3- to 4-hour glucose tolerance test or results of the patient's mixed nutrient meal documentation. This condition often is considered a prediabetic condition. Reactive hypoglycemia is treated by small frequent meals with low carbohydrate content.
Alimentary hypoglycemia. Another uncommon but clinically relevant form of hypoglycemia is alimentary hypoglycemia.93 This tends to occur in young thin women who experience rapid transit through the gastrointestinal tract. This condition is very worrisome to the patient but rarely results in severe hypoglycemia. Further, it is not related to diabetes mellitus and does not predict an increased risk of diabetes mellitus in the future. This is more common in people with irritable bowel syndrome. This is treated with antispasmotic agents such as dicyclomine and hyoscyamine. These medications, which have not been studied in controlled clinical trials for this condition, seem to make a large difference in our clinical experience.
Summary
Hypoglycemia is a common and significant problem in diabetes. It is the rate-limiting factor for intensive glucose treatment. Hypoglycemia can be divided into mild, severe, asymptomatic, and relative hypoglycemia. The treatment of hypoglycemia varies based on its severity, cause, and location of treatment. Physicians should routinely ask patients about hypoglycemia and should be on the lookout for defensive eating and hypoglycemic unawareness. In addition, physicians should be able to apply the proper treatment of hypoglycemia in relation to the severity of the symptoms and the environment in which it occurs. Finally, physicians should be prepared to change diabetes treatment when hypoglycemia is recurrent or severe.
References
1. American Diabetes Association. Standards of medical care in diabetes2011. Diabetes Care 2011;34(Suppl. 1):S11-S61.
2. Fletcher AA, Campbell WR. The blood sugar following insulin administration and the symptom complex: Hypoglycemia. J Metab Res 1922;2:637-649.
3. Cryer PE, et al. Evaluation and management of adult hypoglycemic disorders: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 2009;94:709-728.
4. Cryer PE. Hypoglycemia in Diabetes. Pathophysiology, Prevalence and Prevention. American Diabetes Association, Alexandria, VA, 2009.
5. Cryer PE. The barrier of hypoglycemia in diabetes. Diabetes 2008;57:3169-3176.
6. McCrimmon RJ, Sherwin RS. Hypoglycemia in type 1 diabetes. Diabetes 2010;59:2333-2339.
7. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977-986.
8. The Diabetes Control and Complications Trial Research Group. Hypoglycemia in the Diabetes Control and Complications Trial. Diabetes 1997;46:271-286.
9. Cryer PE, et al. Hypoglycemia in diabetes. Diabetes Care 2003;26:1902-1912.
10. Whipple AO. The surgical therapy of hyperinsulinism. J Int Chir 1938;3:237-276.
11. Cryer PE. Diverse causes of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med 2004;350:2272-2279.
12. Frier BM. Defining hypoglycaemia: What level has clinical relevance? Diabetologia 2009;52:31-34.
13. Amiel SA, et al. Effect of intensive insulin therapy on glycemic thresholds for counterregulatory hormone release. Diabetes 1988;37:901-907.
14. Workgroup on Hypoglycemia, American Diabetes Association. Defining and reporting hypoglycemia in diabetes: A report from the American Diabetes Association Workgroup on Hypoglycemia. Diabetes Care 2005;28:1245-1249.
15. Davis SN, et al. Effects of differing antecedent hypoglycemia on subsequent counterregulation in normal humans. Diabetes 1997;46:1328-1335.
16. Cryer PE. Hypoglycemia, functional brain failure, and brain death. J Clin Invest 2007;117:868-870.
17. Cryer PE. The prevention and correction of hypoglycemia. In: Jefferson L, et al, eds. Handbook of Physiology; Section 7. The Endocrine System. Volume II. The endocrine pancreas and regulation of metabolism. New York: Oxford University Press 2001;1057-1092.
18. A multicenter study of the accuracy of the One Touch Ultra home glucose meter in children with type 1 diabetes. The Diabetes Research in Children Network (DirecNet) Study Group. Diabetes Technol Ther 2003;5:933-941.
19. UK Hypoglycaemia Study Group. Risk of hypoglycaemia in types 1 and 2 diabetes: effects of treatment modalities and their duration. Diabetologia 2007;50:1140-1147.
20. Cryer P. 2008 Glucose homeostasis and hypoglycemia. In: Kronenberg H, eds. Williams Textbook of Endocrinology. 11th ed. Philadelphia: Saunders; 2007:1503-1533.
21. Donnelly LA, et al. Frequency and predictors of hypoglycaemia in type 1 and insulin-treated type 2 diabetes: A population-based study. Diabet Med 2005;22:749-755.
22. UK Prospective Diabetes Study 16. Overview of 6 years' therapy of type II diabetes: A progressive disease. UK Prospective Diabetes Study Group. Diabetes 1995;44:1249-1258.
23. Gerstein HC, et al. Effects of intensive glucose lowering in type 2 diabetes. The Action to Control Cardiovascular Risk in Diabetes Study Group. N Engl J Med 2008;358:2545-2559.
24. The ADVANCE Collaborative Group. Intensive blood control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358:2560-2572.
25. Duckworth W, et al. Glucose control and vascular complications in veteran with type 2 diabetes. N Engl J Med 2009;360:129-139.
26. Curkendall SM, et al. Incidence and cost of hypoglycemia among patients with type 2 diabetes in the United States: Analysis of a health insurance database. JCOM 2011;18:455-462.
27. Dagogo-Jack SE, et al. Hypoglycemia-associated autonomic failure in insulin-dependent diabetes mellitus. Recent antecedent hypoglycemia reduces autonomic responses to, symptoms of, and defense against subsequent hypoglycemia. J Clin Invest 1993;91:819-828.
28. Segel SA, et al. Hypoglycemia-associated autonomic failure in advanced type 2 diabetes. Diabetes 2002;51:724-733.
29. Amiel SA, et al. Hypoglycemia in Type 2 diabetes. Diabetic Med 2008;25:245-254.
30. White NH, et al. Identification of Type 1 diabetic patients at increased risk for hypoglycemia during intensive therapy. N Engl J Med 1983;308:485-491.
31. Cryer PE. Mechanisms of sympathoadrenal failure and autonomic failure and hypoglycemia in diabetes. J Clin Invest 2006;116:1470-1473.
32. Cryer PE. Mechanisms of hypoglycemia-associated autonomic failure and its component syndromes in diabetes. Diabetes 2005;54:3592-3601.
33. Spollett G. Type 2 diabetes across the lifespan. In: The Art and Science of Diabetes Self-Management Education Desk Reference. 2nd ed. Chicago, IL: American Association of Diabetes Educators; 2011.
34. Gold AE, et al. Frequency of severe hypoglycemia in patients with type I diabetes with impaired awareness of hypoglycemia. Diabetes Care 1994;17:697–703.
35. Allen KV, Frier BM. Nocturnal hypoglycemia: clinical manifestations and therapeutic strategies toward prevention. Endocr Pract 2003;9:530-543.
36. Gross TM, et al. Detection of unseen hypoglycemia using continuous glucose monitoring (Abstract). Diabetologia 2001;43:A5.
37. Towler DA, et al. Mechanism of awareness of hypoglycemia. Perception of neurogenic (predominantly cholinergic) rather than neuroglycopenic symptoms. Diabetes 1993;42:1791-1798.
38. Hepburn DA, et al. Symptoms of acute insulin-induced hypoglycemia in humans with and without IDDM. Factor-analysis approach. Diabetes Care 1991;14:949-957.
39. Cryer PE. Hypoglycemia. Chapter 339. In: Harrison's Principles of Internal Medicine. Fauci AS, et al. Vol 2, 17th ed. New York: McGraw Hill; 2008:1596-1607.
40. Cox DJ, et al. Perceived symptoms in the recognition of hypoglycemia. Diabetes Care 1993;16:519-527.
41. Cryer PE. Hypoglycaemia: The limiting factor in the glycaemic management of type I and type II diabetes. Diabetologia 2002;45:937-948.
42. Matyka K, et al. Altered hierarchy of protective responses against severe hypoglycemia in normal aging in healthy men. Diabetes Care 1997;20:135-141.
43. Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study Research Group; Jacobson AM, et al. Long-term effect of diabetes and its treatment on cognitive function. N Engl J Med 2007;356:1842-1852.
44. Skrivarhaug T, et al. Long-term mortality in a nationwide cohort of childhood onset type 1 diabetic patients in Norway. Diabetologia 2006;49:298-305.
45. Feltbower RG, et al. Acute complications and drug misuse are important causes of death for children and young adults with type 1 diabetes: Results from the Yorkshire Register of diabetes in children and young adults. Diabetes Care 2008;31:922-926.
46. Tattersall RB, Gill GV. Unexplained deaths of type 1 diabetic patients. Diabet Med 1991;8:49-58.
47. Lee SP, et al Influence of autonomic neuropathy on QTc interval lengthening during hypoglycemia in type 1 diabetes. Diabetes 2004;53:1535-1542.
48. White JR. The contribution of medications to hypoglycemia unawareness. Diabetes Spectrum 2007;20:77-80.
49. Cohen MR, et al. Survey of hospital systems and common serious medication errors. J Healthc Risk Manag 1998;18:16-27.
50. Fischer KF, et al. Hypoglycemia in hospitalized patients. Causes and outcomes. N Engl J Med 1986;315:1245-1250.
51. Zammitt NN, Frier BM. Hypoglycemia in type 2 diabetes: Pathophysiology, frequency, and effects of different treatment modalities. Diabetes Care 2005;28:2948-2961.
52. Gangji AS, et al. A systematic review and meta-analysis of hypoglycemia and cardiovascular events: a comparison of glyburide with other secretagogues and with insulin. Diabetes Care 2007;30:389-394.
53. Bolen S, et al. Systematic review: Comparative effectiveness and safety of oral medications for type 2 diabetes mellitus. Ann Intern Med 2007;147:386-399.
54. Szoke E, et al. Effects of glimepiride and glyburide on glucose counterregulation and recovery from hypoglycemia. Metabolism 2006;55:78-83.
55. Amiel S. Hypoglycemia in Patients with type 1 diabetes. In: Lebovits HD, ed. Therapy for Diabetes and Related Disorders. 5th Ed. Alexandria VA: American Diabetes Association; 2009: 372-375.
56. Miller SI, et al. Hypoglycemia as a manifestation of sepsis. Am J Med 1980;68:649-654.
57. Maitra SR, et al. Alterations in tissue glucose uptake during the hyperglycemic and hypoglycemic phases of sepsis. Shock 2000;13:379-385.
58. Camacho RC, et al. Glucoregulation during and after exercise in health and insulin-dependent diabetes. Exerc Sport Sci Rev 2005;33:17–23.
59. Epidemiology of severe hypoglycemia in the diabetes control and complications trial. The DCCT Research Group. Am J Med 1991;90:450-459.
60. Allen C, et al. Risk factors for frequent and severe hypoglycemia in type 1 diabetes. Diabetes Care 2001;24:1878-1881.
61. Muhlhauser I, et al. Risk factors of severe hypoglycaemia in adult patients with type I diabetes a prospective population based study. Diabetologia 1998;41:1274-1282.
62. Steffes MW, et al. Beta-Cell function and the development of diabetes-related complications in the Diabetes Control and Complications Trial. Diabetes Care 2003;26:832-836.
63. Jones TW, et al. Decreased epinephrine responses to hypoglycemia during sleep. N Engl J Med 1998;338:1657-1662.
64. Banarer S, Cryer PE. Sleep-related hypoglycemia-associated autonomic failure in type 1 diabetes: Reduced awakening from sleep during hypoglycemia. Diabetes 2003;52:1195-1203.
65. Kilpatrick ES, et al. Relating mean blood glucose and glucose variability to the risk of multiple episodes of hypoglycaemia in type 1 diabetes. Diabetologia 2007;50:2553-2561.
66. Pedersen-Bjergaard U, et al. Severe hypoglycaemia in 1076 adult patients with type 1 diabetes: Influence of risk markers and selection. Diabetes Metab Res Rev 2004;20:479-486.
67. Bottini P, et al. Contribution of autonomic neuropathy to reduced plasma adrenaline responses to hypoglycemia in IDDM: Evidence for a nonselective defect. Diabetes 1997;46:814-823.
68. Meyer C, et al. Effects of autonomic neuropathy on counterregulation and awareness of hypoglycemia in type 1 diabetic patients. Diabetes Care 1998;21:1960-1966.
69. Boucai L, et al. Hypogylcemia-associated mortality is not drug-associated but linked to comorbidities. Am J Med 2011;124:1029-1035.
70. Smith WD, et al. Causes of hyperglycemia and hypoglycemia in adult inpatients. Am J Health Syst Pharm 2005;62:714-719.
71. Fischer KF, et al. Hypoglycemia in hospitalized patients: Causes and outcomes. N Engl J Med 1986;315:1245-1250.
72. Ben-Ami H, et al. Drug-induced hypoglycemic coma in 102 diabetic patients. Arch Intern Med 1999;159:281-284.
73. Irvine AA, et al. Fear of hypoglycemia: Relationship to physical and psychological symptoms in patients with insulin-dependent diabetes mellitus. Health Psychol 1992;11:135-138.
74. Warm EJ. Diabetes and the chronic care model: A review. Curr Diabetes Rev 2007;3:219-225.
75. Gough SC. A review of human and analogue insulin trials. Diabetes Res Clin Pract 2007;77:1-15.
76. Hirsch IB. Insulin analogues. N Engl J Med 2005;352:174-183.
77. Gough K, et al. Long-acting insulin analogues versus NPH insulin (human isophane insulin) for type 2 diabetes mellitus. Cochrane Database Syst Rev 2007:CD005613.
78. Fatourechi M, et al. Hypoglycemia with intensive insulin therapy: A systematic review and meta-analysis of randomized trials of continuous subcutaneous insulin infusion versus multiple daily injections. J Clin Endocrinol Metab 2008;94:729-740.
79. Bergenstal RM, et al. Effectiveness of a sensor-augmented insulin pump therapy in type 1 diabetes. N Engl J Med 2010;363:311-320.
80. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;52:837-53.
81. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;52:854-865.
82. Raju B, et al. Nocturnal hypoglycemia in type 1 diabetes: An assessment of preventive bedtime treatments. J Clin Endocrinol Metab 2006;91:2087-2092.
83. Fanelli CG, et al. Meticulous prevention of hypoglycemia normalizes the glycemic thresholds and magnitude of most of neuroendocrine responses to, symptoms of, and cognitive function during hypoglycemia in intensively treated patients with short-term IDDM. Diabetes 1993;42:1683-1689.
84. Fanelli C, et al. Long-term recovery from unawareness, deficient counterregulation and lack of cognitive dysfunction during hypoglycaemia, following institution of rational, intensive insulin therapy in IDDM. Diabetologia 1994;37:1265-1276.
85. Cranston I, et al. Restoration of hypoglycaemia awareness in patients with long-duration insulin-dependent diabetes. Lancet 1994;344:283-287.
86. Suh SW, et al. Hypoglycemic neuronal death is triggered by glucose reperfusion and activation of neuronal NADPH oxidase. J Clin Invest 2007;117:910-918.
87. Gunning RR, Garber AJ. Bioactivity of instant glucose. Failure of absorption through oral mucosa. JAMA 1978;240:1611-1612.
88. Boyle PJ, et al. Octreotide reverses hyperinsulinemia and prevents hypoglycemia induced by sulfonylurea overdoses. J Clin Endocrinol Metab 1993;76:752-756.
89. Cryer PE. Glucose homeostasis and hypoglycemia. In: Kronenberg HM, et al., eds. Williams Textbook of Endocrinology. 11th ed. Philadelphia, PA: Elsevier Health Sciences; 2008: 1503-1533.
90. Amiel S. Hypoglycemia in patients with type 1 diabetes. In: Therapy for Diabetes and Related Disorders. 5th ed. Alexandria VA: American Diabetes Association; 2009: 372-375.
91. Harris S. Hyperinsulinemia and Dysinsulinism. JAMA 1924;83:729-733.
92. Brun JF, et al. Postpranidal reactive hypoglycemia. Diabetes Metab 2000;26:337-351.
93. Leichter SB. Alimentary hypoglycemia: A new appraisal. Am J Clin Nutr 1979;32:2104-2113.
In November 2011, the Centers for Disease Control and Prevention presented data on emergency hospitalizations because of adverse drug reactions. The report showed that insulin and oral hypoglycemic agents caused nearly 25% of the medication-induced hospitalizations in older adults in the United States.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
