Sexually Transmitted Diseases
Sexually Transmitted Diseases
Author:
Robert A. McKie, MD, Medical Director, Emergency Department, Veterans Affairs Medical Center, Boise, ID.
Peer Reviewer:
Ralph J. Riviello, MD, MS, FACEP, Associate Professor of Emergency Medicine, Drexel University College of Medicine, Philadelphia, PA.
Sexually transmitted diseases (STDs) are frequently encountered in clinical medicine. According to the Centers for Disease Control, approximately 2.8 million infections with Chlamydia trachomatis1 and 700,000 infections with Neisseria gonorrhea2 occur annually in the United States. Viral sexually transmitted diseases are frequently incurable, so prevalence data are a better measure of the overall burden of disease. It is estimated that 1 in 6 adults in the United States between the ages of 14 and 49 years have genital herpes.3 Genital herpes is caused by the herpes simplex virus 2 (HSV-2) and, increasingly, herpes simplex virus 1 (HSV-1).4 Roughly 1.2 million Americans are living with the human immunodeficiency virus (HIV).5 Human papilloma virus (HPV) can cause genital warts and several cancers. HPV is extraordinarily common about 20 million Americans are infected with HPV, and another six million infections occur annually.6 Syphilis, lymphogranuloma venereum, and chancroid are much less common, but can cause significant morbidity and mortality.7,8
Figure 1: Chlamydia Rates by Sex, United States, 1990-2010
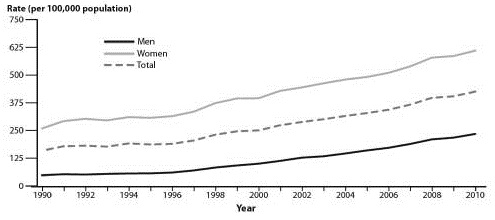
NOTE: As of January 2000, all 50 states and the District of Columbia have regulations that require the reporting of chlamydia cases.
Source: Centers for Disease Control and Prevention
The vast majority of patients with a new STD do not present to a sexually transmitted disease clinic.9 These patients typically present to outpatient clinics, emergency departments, and on inpatient wards. Underdiagnosis of sexually transmitted disease is a significant cause of morbidity and a major public health problem. For these reasons, it is essential that all health care providers are familiar with the diagnosis and treatment of sexually transmitted diseases.
General Principles
Sexually transmitted diseases have a broad range of manifestations. Presentations of sexually transmitted diseases can range from the completely asymptomatic patient to the moribund patient in septic shock. Some general principles should guide the evaluation of patients with sexually transmitted diseases.
History is essential to the proper diagnosis of sexually transmitted diseases. Patients should be asked about the number of male and female partners they have had. Patients should also be asked if they engage in insertive or receptive vaginal, oral, or anal sex. Several sexually transmitted diseases can present with oral, anal, or genital manifestations. Questions should be open-ended and non-judgmental. Sexual history can be an uncomfortable topic for patients and health care providers, and rapport is essential to having a frank discussion of sexual behavior. Patients should be asked about travel history and recent sexual contacts outside of the United States. Chancroid (Haemophilus ducreyi) is very uncommon in the United States, but is relatively common in parts of sub-Saharan Africa, Southeast Asia, and Latin America.10 Sporadic outbreaks have occurred in the United States and Europe. Lymphogranuloma venereum (LGV) is caused by serovars of Chlamydia trachomatis that are more common in tropical regions. HIV status can impact the presentation of some STDs, so physicians should ask patients about their HIV status.
Figure 2: Gonorrhea Rates, United States, 1941-2010
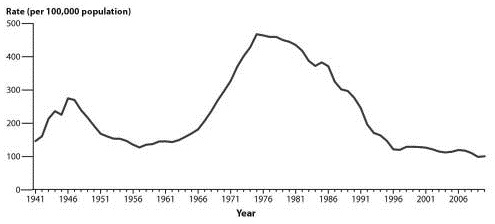
Source: Centers for Disease Control and Prevention
Genital ulcers present a diagnostic conundrum. While there are certain characteristics that suggest one diagnosis or another, it is essential to confirm the diagnosis whenever possible with laboratory tests. While certain characteristics of genital ulcers can assist with the diagnosis, history and physical examination may not be adequate for the diagnosis of STDs. Several types of genital ulcers can present in a similar fashion, and patients may be infected with more than one STD. Genital ulcers facilitate HIV transmission, and HIV testing should be offered to all patients with a genital ulcer.10
Some ulcer characteristics support the diagnosis of a specific sexually transmitted disease. The ulcers associated with genital herpes, chancroid, and lymphogranuloma venereum are generally painful. The chancre associated with primary syphilis and the nodules associated with Klebsiella granulomatis (granuloma inguinale or donovanosis) infection are usually painless. Firm, round erythematous papules suggest syphilis, while grouped vesicles prone to recurrence suggest genital herpes.10 Beefy, expanding granulomas that bleed easily suggest granuloma inguinale.11 Fluctuant lymph nodes suggest LGV or chancroid. Erosions may be seen in chancroid or after herpetic vesicles have ruptured. Painless ulcers that initially appear in the rectum, oropharynx, or vagina may go unnoticed by patients and health care providers.
Figure 3: Syphilis Reported Cases by Stage of Infection, United States, 1941-2010
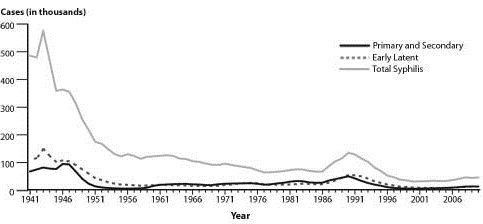
Source: Centers for Disease Control and Prevention
If laboratory testing does not reveal a diagnosis and the ulcer persists in the face of empiric antibiotic therapy, a non-sexually transmitted etiology should be considered. Behçet's disease and malignancy can present as ulcers of the oropharynx, genital tract, and anus.
Pelvic inflammatory disease (PID) is a potentially life-threatening complication of some sexually transmitted diseases. PID occurs when Chlamydia trachomatis, Neisseria gonorrhea, and/or vaginal flora infect the upper female genital tract. The upper female genital tract encompasses the uterus, the oviducts, and the ovaries. Right upper quadrant pain from perihepatic inflammation, also known as Fitzhugh-Curtis syndrome, is present in up to 15% of women with PID. The pain from perihepatitis may be pleuritic in nature and radiate to the right shoulder. The manifestations of PID range from mild, nonspecific abdominal pain to serious infection and sepsis.12,13
Figure 4: Chancroid Reported Cases, United States, 1981-2010
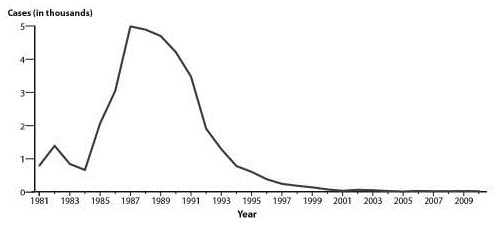
Source: Centers for Disease Control and Prevention
Pelvic inflammatory disease is a clinical diagnosis. PID should be suspected when a woman has otherwise unexplained abdominal pain plus adnexal tenderness, cervical motion tenderness, or uterine tenderness. The presence of one or more of the following findings increases the specificity of the diagnosis of PID: fever greater than 101°F, mucopurulent vaginal or cervical discharge, positive tests for Neisseria gonorrhea or Chlamydia trachomatis, elevated erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP), or abundant WBCs in vaginal fluid. Adnexal masses should alert the clinician to a potential tubo-ovarian abscess. Tubo-ovarian abscess is an inflammatory mass of the upper female genital tract and/or surrounding pelvic organs.14 Tubo-ovarian abscesses are typically diagnosed in patients with PID. Ruptured tubo-ovarian abscesses are life-threatening and usually require emergent surgery and broad-spectrum antibiotic therapy. In the absence of advanced therapy, the mortality for ruptured abscesses approaches 50%.14
The most specific diagnosis of PID can be made by imaging studies, laparoscopy, or uterine biopsy.12 Several imaging modalities are useful in the diagnosis of PID. Magnetic resonance imaging or ultrasonography may demonstrate thickened, fluid-filled tubes, with or without free pelvic fluid, or other tubo-ovarian changes.12 Doppler studies may reveal tubal hyperemia. Laparoscopy may demonstrate edema or erythema of the oviducts, purulent exudate, or other abnormalities in the upper female genital tract. In patients with perihepatic involvement, laparoscopy reveals purulent exudates on the surface of the liver, with so-called violin-string adhesions attached to the liver capsule. Uterine biopsy in patients with PID demonstrates findings consistent with endometritis.13
Figure 5: Genital Warts Initial Visits to Physicians' Offices, United States, 1966-2010
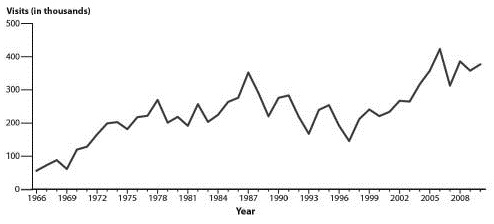
NOTE: The relative standard errors for genital warts estimates of more than 100,000 range from 18% to 30%.
Source: Centers for Disease Control and Prevention, IMS Health, Integrated Promotional Services. IMS Health Report, 1966–2010.
Chlamydia
In 2010, more than 1.3 million cases of Chlamydia trachomatis were diagnosed in the United States. The CDC estimates that an even larger number of cases goes undiagnosed.1 Chlamydia can infect the urethra, cervix, upper female genital tract, eyes, rectum, and the oropharynx.12
Chlamydia trachomatis is a gram-negative, obligate, intracellular organism. C. trachomatis is unable to grow and reproduce outside of eukaryotic cells. The bacterium most frequently infects the cells of the cervix in women and the urethra in men. The infective C. trachomatis is called an elementary body. The elementary body enters the cell and forms a reticulate body. The reticulate body utilizes the resources of the eukaryotic cell to grow and reproduce. As an obligate intracellular organism, C. trachomatis is able to grow and reproduce using the adenosine triphosphate and other biochemical products of the host eukaryotic cell. After several cycles of replication, the host cell lyses and releases infective elementary bodies.11
Asymptomatic chlamydia is frequently diagnosed through routine screening or when a patient learns that a sex partner was infected with chlamydia. Screening for Chlamydia trachomatis, Treponema pallidum, and hepatitis B is all a part of routine prenatal care. Testing for HIV and Neisseria gonorrhea is also frequently offered as a part of prenatal care. Regular screening for chlamydia, gonorrhea, syphilis, and HIV is recommended in a wide range of patient populations.12
The majority of women with isolated chlamydial cervicitis are asymptomatic.15 If symptoms are present, they typically include vaginal discharge, intermenstrual bleeding, and dyspareunia. If the urinary tract is involved, dysuria and pyuria are frequently present. When chlamydial infections involve the upper female genital tract, abdominal pain, vaginal discharge, and vaginal bleeding are common symptoms. In patients with anorectal chlamydia, pruritus, rectal discharge, and rectal bleeding may be present. Chlamydia can damage the oviducts and cause infertility and ectopic pregnancy. Chlamydia can also be passed from mother to infant. Chlamydial infection in infants can cause pneumonia and ocular infections. Ocular infections range from conjunctivitis to trachoma. Trachoma is a serious infection of the eyes that scars the conjunctiva and turns the eyelashes inward. Once directed inward, the eyelashes damage the cornea. Trachoma has caused millions of cases of blindness worldwide. Oropharyngeal chlamydia is relatively uncommon and generally occurs in patients who have a history of performing oral sex. As mentioned above, some serovars of C. trachomatis cause LGV, a far more malignant chlamydial infection.12,15
On exam, patients with cervicitis may have cervical discharge, edema, friability, or ulcers. If the patient also has abdominal pain along with cervical motion tenderness, uterine tenderness, or adnexal tenderness, consider the diagnosis of PID.12,15 Patients with urethral involvement may have white blood cells on urinalysis.
Several approaches exist for the diagnosis of chlamydia in women. Samples may be collected by endocervical swab on speculum exam, urine sample, ureteral swab, or self-collected vaginal swab. For reasons of comfort, urine samples are typically more acceptable to many patients. Chlamydia trachomatis is notoriously difficult to culture, and the diagnosis is typically made by nucleic acid amplification tests (NAAT) or direct fluorescent antibody testing. In some forensic cases, chlamydia will be cultured using eukaryotic cell cultures. Neisseria gonorrhea infection frequently co-exists with chlamydia, and testing for both organisms should be performed in all patients.12 Empiric therapy for gonorrhea may be considered in some patients with chlamydia.
Treatment of chlamydia in women with cervicitis can involve either a single dose of 1 gram of azithromycin or seven days of 100 mg of doxycycline taken twice daily. Doxycycline is the cheaper drug, but azithromycin compliance can approach 100% if the patient is observed taking the full dose of azithromycin in the emergency department or clinic. This dose of azithromycin can cause nausea and vomiting. Erythromycin, levofloxacin, and ofloxacin are alternative agents for the treatment of chlamydia. Levofloxacin, ofloxacin, and doxycycline should be avoided in pregnant women.12 The treatment of pelvic inflammatory disease is more complex and is described later in the article.
In men, C. trachomatis typically infects the urethra. Patients with chlamydial urethritis experience dysuria and thin, watery, or mucoid discharge from the penile meatus.12,16 The diagnosis of C. trachomatis urethritis should be considered in sexually active men with urine that is positive for leukocyte esterase or has greater than 10 white blood cells per high powered field. A Gram stain of urethral secretions that shows greater than 5 white blood cells per high powered field also establishes the diagnosis of urethritis. Occasionally, the infection can spread to the epididymis or the prostate. These patients typically experience unilateral testicular pain, pelvic pain, or swelling of the epididymis. Testicular torsion is a surgical emergency that may mimic the symptoms of epididymitis. Chlamydia epididymitis should be suspected in patients with severe unilateral testicular pain, especially if there is no evidence of infection on diagnostic studies. Patients with LGV present with lymphadenopathy and signs or symptoms of systemic illness. Rectal LGV can cause inflammatory rectal or retroperitoneal masses. Rectal chlamydia is usually asymptomatic, but it can present with rectal bleeding, tenesmus, or itching. Anoscopic examination may reveal friable mucosa and discharge.12,16
Once urethritis or other chlamydial infection is suspected, the diagnosis in men typically involves performing NAAT on samples obtained from urethral swabbing or urine. Proctitis, if suspected, may prompt anoscopy with rectal swabbing for NAAT testing.
Chlamydia urethritis in men can be treated with the same regimens that are used to treat cervicitis in women. Epididymitis can be treated with 100 mg of doxycycline twice daily for 10 days plus 250 mg of IM ceftriaxone. Pending culture results, proctitis should be treated with 100 mg of doxycycline twice daily for 7 days plus 250 mg of ceftriaxone IM. Lymphogranuloma venereum requires more intensive therapy. Recommended regimens include 100 mg of doxycycline twice daily for 21 days or erythromycin base 500 mg four times daily for 21 days. Azithromycin 1 gram by mouth weekly is probably also effective. Buboes (swollen infected lymph nodes) are frequently present in LGV, and these may require aspiration or incision and drainage. If buboes are not properly drained, rupture and sinus tract formation can result.12
In men who engage in insertive anal intercourse, enteric organisms should also be considered as a potential cause of epididymitis. In this situation, the treatments of choice include 500 mg of oral levofloxacin once daily for 10 days or 300 mg of oral ofloxacin twice daily for 10 days plus 250 mg of intramuscular ceftriaxone.12
Chlamydial conjunctivitis should be considered in infants with conjunctivitis, especially if the mother has untreated chlamydia. Topical therapy is not adequate as monotherapy for chlamydial conjunctivitis. Topical ophthalmologic agents given to newborns for prevention of gonococcal conjunctivitis are not effective in preventing chlamydial infections. Oral therapy with weight-based erythromycin is adequate monotherapy. Chlamydia trachomatis should also be considered as a possible cause of pneumonia in infants up to 3 months old, especially if the mother had untreated chlamydia at the time of delivery. Treatment is weight-based erythromycin for 14 days. The cure rate for this therapy is about 80%, so patients must be followed up to ensure that therapy is adequate. Prenatal screening and treatment has significantly reduced the burden of chlamydial infections in newborns.12
Treatment of children with chlamydial and other STD infections differs slightly from the treatment of adults. Sexual abuse should always be considered, and child protective services should be alerted. Weight-based erythromycin oral therapy for 14 days is preferred in children who weigh less than 45 kilograms; children who weigh more than 45 kilograms and are younger than 8 years of age should be treated once with 1 gram of oral azithromycin. In children older than 8 years of age, 1 gram of oral azithromycin or 100 mg of doxycycline twice daily for 7 days are both appropriate therapies.12
Sexual partners of patients with C. trachomatis should also be evaluated and/or treated for chlamydia. This prevents both the morbidity of untreated chlamydia infection in the sex partner and re-infection of the patient. In some circumstances, patient-delivered partner therapy (PDPT) is appropriate. PDPT involves giving the patient a prescription for antibiotic therapy for sex partners, along with instructions about medication allergies and side-effects, symptoms of more serious conditions like PID, and recommendations that the partner seek medical attention. The advantage of PDPT is that it provides an opportunity to treat patients who might otherwise not seek treatment. The disadvantage to PDPT is that patients are treated without being evaluated for other sexually transmitted diseases or more complicated manifestations of STDs like PID. PDPT is not legal in all jurisdictions, so physicians should know the rules of their locality before prescribing PDPT.12
Gonorrhea
More than 300,000 cases of gonorrhea were diagnosed in the United States in 2010.2 As with chlamydia, the CDC estimates that an even larger number of gonorrhea infections goes undetected. Like chlamydia, Neisseria gonorrhea can cause infections of the urogenital tract, the oropharynx, and the rectum. In addition, gonorrhea can cause disseminated infections with joint pain, septic arthritis, and pustular skin lesions. Disseminated gonorrhea can be life-threatening.12
Neisseria gonorrhea is a gram-negative diplococci. N. gonorrhea is relatively fastidious and requires specialized media for growth. Pilli facilitate the attachment of N. gonorrhea to human cells.11
In women, gonorrhea is frequently asymptomatic. When symptoms are present, they can include vaginal discharge and vaginal itching. Gonococcal infection of the urethra presents as pyuria and dysuria.12,17
In men, the most common site of N. gonorrhea infection is the urethra. In sharp contrast to chlamydia, gonococcal urethritis is asymptomatic in only about 10% of men. Most men experience dysuria and/or a large volume of purulent penile discharge. Epididymitis can also occur with N. gonorrhea infection in men.12,17
Both men and women can experience N. gonorrhea infections of the pharynx and rectum. These infections are usually asymptomatic. Men and women can also experience gonococcal bacteremia and septic arthritis. Gonorrhea can also be a cause of conjunctivitis.12,17
Diagnosis of N. gonorrhea involves collection of samples in a similar fashion to C. trachomatis. A Gram stain of male urethral samples that reveals intracellular gram-negative diplococci is highly specific for N. gonorrhea. In women and in patients with non-urethral sites of gonorrhea, gram stain is not adequate. The use of NAAT and culture is appropriate in patients with gonorrhea. As with C. trachomatis, culture may be important in forensic examinations. Culture can also provide useful information about sensitivity in patients who fail first-line treatment.12,17
Treatment of N. gonorrhea infections varies by site. Uncomplicated urethral, rectal, and cervical infections can be treated with a single dose of 250 mg of IM ceftriaxone or 400 mg of oral cefixime plus one dose of 1 gram of azithromycin or 100 mg of doxycycline twice daily for 7 days. Adding azithromycin or doxycycline to a cephalosporin covers co-infection with C. trachomatis, if present, and generally also covers N. gonorrhea. N. gonorrhea is double-covered to reduce the risk of antibiotic resistance. For gonococcal pharyngitis, only the ceftriaxone-containing regimen is advised. Because fluoroquinolone-resistant strains of N. gonorrhea are increasingly common worldwide, fluoroquinolones should generally not be used for treatment of gonorrhea.12 Spectinomycin is highly effective for the treatment of gonorrhea, but is currently not available in the United States. Spectinomycin is not available in an oral preparation and is relatively ineffective for the treatment of pharyngeal gonococcus.12
Until April 2007, N. gonorrhea could be treated with oral medications. At that time, fluoroquinolones were no longer recommended for the treatment of gonorrhea and PID in the United States. Unfortunately, at that time, cefixime was not available in the United States.19 High-dose therapy with a one-time dose of 2 grams of azithromycin may be used to treat uncomplicated gonorrhea. This therapy can cause significant nausea, and monotherapy with lower doses of azithromycin increases the risk of inadequate therapy and antibiotic resistance. Fortunately, in April 2008, cefixime again became available in the United States. Treatment of N. gonorrhea remains dependent on the cephalosporin class of antibiotics.
Gonococcal conjunctivitis can be treated with a single dose of 1 gram of IM ceftriaxone. Irrigation of the eye with saline may be considered as an adjunctive therapy. Patients with disseminated N. gonorrhea infection should be hospitalized and treated with either 1 gram of ceftriaxone IV or IM every 24 hours or 1 gram cefotaxime or ceftizoxime IV every 8 hours. Gonococcal meningitis and endocarditis are treated with higher doses of intravenous ceftriaxone. Infants with gonococcal conjunctivitis and other N. gonorrhea infections are usually treated with ceftriaxone. Topical preparations of silver nitrate or antibiotics are administered to newborns to prevent gonococcal conjunctivitis. This diagnosis should be suspected in infants with conjunctivitis that did not receive prophylaxis or in infants who have mothers with a known diagnosis of, or risk factors for, gonorrhea. As with chlamydia, patients with gonorrhea may be offered PDPT in some circumstances to ensure that sex partners are also treated for gonorrhea. Testing for HIV and other sexually transmitted diseases should be offered to patients with N. gonorrhea.12,18
Syphilis
In 2006, 36,000 cases of syphilis were diagnosed in the United States. By 2010, that number had increased to more than 45,000. Although less common than chlamydia or gonorrhea, syphilis remains a major public health issue. Rates of syphilis infection have generally increased in recent years. If undiagnosed, syphilis can cause catastrophic, life-threatening complications. Syphilis infections are increasingly common among men who have sex with men.6
Syphilis is caused by Treponema pallidum, which is a gram-negative spirochete that resembles a corkscrew. T. pallidum is typically spread by mucocutaneous contact. Pregnant women can pass syphilis to their infants.11
Syphilis is a disease with an extraordinarily broad range of presentations. The diverse range of presentations has earned it the moniker "the great imitator." Typically, syphilis can be divided into three distinct phases. Primary syphilis involves a firm, painless ulcer called a chancre. The chancre appears at the initial site of T. pallidum inoculation. This generally resolves and is followed by secondary syphilis. Secondary syphilis most commonly involves a widespread rash that is most prominent on the hands and feet. Systemic symptoms such as fever, weight loss, and malaise may also be present. Tertiary syphilis appears after 10 or more years of untreated syphilis and can involve cardiovascular complications such as ruptured aneurysms, multiple neurologic complications, and rubbery skin lesions called gummas. Asymptomatic syphilis infection of less than one year is called early latent syphilis, and asymptomatic syphilis infection of greater than one year is called late latent syphilis. Latent syphilis infection of unknown duration is considered, unsurprisingly, latent syphilis of unknown duration.11,12
The diagnosis of syphilis starts with clinical suspicion. The diagnosis of syphilis should be considered in patients with typical syphilitic chancres or rash. If the painless chancre occurs in the rectum or the vagina, it may go unnoticed by the patient. Like other sexually transmitted diseases, the diagnosis of syphilis may be made by screening of asymptomatic patients. Less frequently, syphilis will present as a vascular catastrophe, such as a ruptured abdominal aortic aneurysm or a neurologic event such as stroke, dementia, visual disturbances, or loss of vibratory sensation.11,12
Serum tests and chancre material are used to make the diagnosis of syphilis. The Venereal Disease Research Laboratory (VDRL) and the Rapid Plasma Reagin (RPR) are both non-treponemal tests for syphilis. These tests are called non-treponemal because they look for antigens to mammalian cells that are usually present in T. pallidum infections. Other infectious and autoimmune conditions can cause false positives. For this reason, a treponemal test is used to confirm the diagnosis. Treponemal tests look for antibodies to actual components of T. pallidum. Treponemal tests remain positive in most patients after effective treatment of syphilis. For this reason, treponemal tests alone are also not adequate for the diagnosis of syphilis. Serum expressed from an abraded chancre can be examined under dark-field microscopy or fluorescent antibody microscopy for the presence of spirochetes.11,12
Penicillin G is the drug of choice for syphilis treatment. Primary, secondary, and early latent syphilis in adults should be treated with 2.4 million units of benzathine penicillin IM. Patients with late latent syphilis and syphilis of unknown duration are given three weekly doses of 2.4 million units of benzathine penicillin IM. Oral penicillin and other preparations of intramuscular penicillin are not adequate for the treatment of syphilis. Children are treated with weight-based doses of benzathine penicillin IM. Alternatives to penicillin are not well-studied, but doxycycline, ceftriaxone, and azithromycin are all likely to be effective in treating early syphilis.12
If neurosyphilis is suspected, the patient will require cerebrospinal fluid analysis and may require ophthalmologic consultation for ocular manifestations of syphilis. If the diagnosis of neurosyphilis is confirmed, high doses of intravenous penicillin G are required for 10 to 14 days. Patient with non-neurologic tertiary syphilis are usually treated with 2.4 million units of IM benzathine penicillin weekly for 3 weeks. Treatment of syphilis in pregnancy typically involves intravenous penicillin.12
Penicillin remains the cornerstone of syphilis treatment. Penicillin has been used to treat syphilis for more than 50 years and remains highly effective. Penicillin allergy can result in fatal reactions when penicillin is administered, but the majority of patients who report penicillin allergy can be treated with penicillin under proper conditions. Patients who report penicillin allergy may no longer be allergic, or may not ever have had a true allergy to penicillin. Skin testing should be performed, if available, on patients who report penicillin allergy. High-risk patients, including patients with a history of anaphylaxis and patients with asthma, should first be tested with 1/100th dilutions of penicillin skin tests.12
Patients with negative skin tests can generally be treated with penicillin safely. Patients with positive skin tests should be desensitized to penicillin prior to receiving full-dose penicillin therapy. Desensitization can also be performed for patients with a history of significant penicillin allergy if skin testing is not available. Patients require proper monitoring during testing, and resources to rescue the patient from anaphylaxis and other severe reactions must be available. From a practical standpoint, penicillin desensitization usually requires hospital admission.12
Patients being treated for syphilis should also be informed that they may experience an acute febrile reaction during the first 24 hours following initiation of antibiotic therapy. This syndrome, also known as the Jarisch-Herxheimer reaction, includes myalgias, fever, and headache. It can be treated with acetaminophen or other antipyretics. The reaction appears to be more prominent in patients with early syphilis.12
As with other sexually transmitted diseases, patients with syphilis should be offered testing for HIV and other STDs. Sex partners should be informed, evaluated, and treated, but PDPT is not appropriate for patients with syphilis.
Chancroid, Granuloma Inguinale, Trichomoniasis, and Genital Herpes
Chancroid. Only 24 cases of chancroid were diagnosed in the United States in 2010. This disease remains relatively common in several parts of the world. This diagnosis should be considered in patients with painful genital ulcers and sexual contacts from or a history of travel to endemic regions. Less frequently, local outbreaks of chancroid can occur in the United States and other developed nations.10
Testing for chancroid is not widely available in the United States. Several laboratories have developed their own tests for H. ducreyi, and if the diagnosis is suspected, physicians should check with a local lab for available testing. As a result, chancroid is often a clinical diagnosis.
Patients with suspected chancroid can be treated with a number of antibiotic regimens, including 250 mg ceftriaxone IM once, a single oral dose of 1 gram of azithromycin, 500 mg of oral ciprofloxacin twice daily for 3 days, or longer courses of erythromycin. On reexamination, ulcers should improve significantly in a week if proper therapy is initiated. If buboes are present, these should be aspirated or drained to prevent rupture or sinus tract formation.12
Granuloma Inguinale. Granuloma inguinale or donovanosis is caused by Klebsiella granulomatis. It causes painless, erythematous granulomas on the genitals. Patients may also have rubbery lymph node enlargement. As with chancroid, this is rarely encountered in the United States. Granuloma inguinale is endemic in India, Papua New Guinea, the Caribbean, southern Africa, and parts of Australia.12 Diagnosis typically requires tissue biopsy. Tissue samples reveal grouped organisms in mononuclear cells. This finding is known as a Donovan body, after the physician who first noted this finding in patients with granuloma inguinale.11
Granuloma inguinale is typically treated with 100 mg of doxycycline twice daily for 3 weeks. Alternative agents include azithromycin, ciprofloxacin, sulfa-trimethoprim, and erythromycin.12 Chloramphenicol is highly effective for the treatment of K. granulomatis,11 but should be avoided because of the risk of aplastic anemia in patients given this drug.
Trichomoniasis. Trichomoniasis is a very common condition that is caused by the protozoan Trichomonas vaginalis. The CDC estimates that 3.7 million Americans have T. vaginalis infections. In women, trichomoniasis is characterized by a large volume of foul-smelling vaginal discharge. Vulvar irritation can also be present. In some women, the symptoms are subtle or absent altogether. In men, T. vaginalis infection of the urethra is frequently asymptomatic, but some men experience symptoms of urethritis. T. vaginalis has not been found to infect the oral cavity, and infection of the rectum is rare.12
Testing for T. vaginalis traditionally involved wet preparation microscopy. Today, point-of-care tests are available that are more sensitive and specific than wet preparation microscopy. In men, urethral swabs, urine, and semen can all be tested for T. vaginalis.12
Treatment of T. vaginalis involves a single oral dose of 2 grams of either metronidazole or tinidazole. Alternatively, patients may take 500 mg of oral metronidazole twice daily for 7 days. Topical preparations are less effective than oral agents. As with other STDs, sex partners should be evaluated and treated for T. vaginalis.12
Genital Herpes. Genital herpes can be caused by HSV-1 or HSV-2. Traditionally, HSV-1 caused peri-oral infection, and HSV-2 caused genital infection. In recent years, this distinction has blurred, likely in part because of the growing popularity of oral sex. Among women and men who have sex with men, HSV-1 is an especially common cause of genital herpes. HSV-2 antibodies are present in about one in six adults of reproductive age, and HSV-1 antibodies occur in the majority of individuals. As a general rule, HSV-2 is much more prone to genital recurrence than HSV-1. Patients who are infected with one type of HSV can be infected with another type of HSV in what is known as a non-primary infection.12,20
Patients with HIV have unique issues with HSV infection. As with other genital ulcers, HSV increases the risk of acquiring and transmitting HIV. This may be true even when visible genital ulcers are not present. In general, clinical manifestations of HSV infection are more severe in patients with HIV. After initiation of antiretroviral therapy for HIV, HSV infections may become more symptomatic as the immune system is reconstituted.12
The diagnosis of HSV-1 and HSV-2 can be established in a number of ways. If ulcers are present, these can be unroofed and the contents sent for viral culture or polymerase chain reaction. Serum tests can be useful for differentiating between HSV infections once the body has mounted an antibody response.12,20
The ulcers of genital herpes can be treated with antiviral drugs. Initial outbreaks can be managed with acyclovir, valacyclovir, or famciclovir for 7-10 days. This reduces the severity and duration of the initial episode. Recurrent episodes are treated with shorter courses of the same drugs. Suppressive therapy reduces viral shedding and reduces the risk of transmitting genital herpes to uninfected sex partners. In general, viral shedding from genital sites is much more common with HSV-2 than HSV-1. Patients with genital herpes should be educated about the risk of transmitting HSV to uninfected partners, the importance of partner notification, and the role of suppressive therapy and barrier contraception in reducing the risk of transmission. Many individuals do not know that they have herpes, and individuals with herpes can transmit the infection while they are asymptomatic. The diagnosis of a lifelong viral infection may be traumatic, and patients should also be offered counseling and reassurance.12,20
Neurologic complications of genital herpes include encephalitis and urinary retention. Patients with encephalitis and other serious HSV infections should be treated with intravenous acyclovir.12,20
Women can pass genital herpes to infants during delivery. Neonatal herpes is a potentially life-threatening condition that can infect the skin, mucous membranes, central nervous system, and other organs. In general, the risk of transmission is low unless the woman acquires HSV infection near the time of delivery. To prevent this, women who do not have genital herpes should avoid sex with partners who may have HSV during the third trimester. Women without a history of orolabial herpes should refrain from oral sex during the third trimester to prevent acquisition of HSV-1 genital herpes. Women with symptomatic genital herpes should consider cesarean delivery to reduce the risk of neonatal herpes. The treatment of neonatal herpes is complex and should be done in consultation with a pediatric infectious disease specialist.12
Human Immunodeficiency Virus
The treatment of HIV has advanced dramatically in the past three decades and is beyond the scope of this article. While overall mortality has declined, the prevalence of HIV disease has increased. Patients with HIV are living very long lives with the help of antiviral therapy. Emergency room physicians should be aware that antivirals, especially protease inhibitors, alter metabolic profiles significantly. Antivirals can worsen glucose tolerance and lipid profiles. While there is some variation in the data, long-term treatment with antiretroviral drugs appears to significantly increase the risk of cardiovascular disease in proportion to the duration of protease inhibitor therapy.21
Several studies have demonstrated the link between long-duration protease inhibitor therapy and cardiovascular disease. Protease inhibitor therapy of less than 18 months has generally not been associated with increased cardiovascular mortality. A study of veterans in the United States showed no increased risk of heart disease in HIV patients treated with antiretrovirals for a median of 15 months. Protease inhibitor therapy of greater than 18 months has been shown in a number of studies to be associated with an increased risk of myocardial infarction. In the French Hospital Cohorts study of HIV-positive men, increasing duration of protease inhibitor therapy was shown to increase the risk of heart disease. Men with greater than 18 months of protease inhibitor use had more than twice the risk of myocardial infarction of men with less than 18 months of protease inhibitor therapy. The multi-national D:A:D Study group found that antiretroviral therapy correlated with an increased risk of cardiovascular disease in proportion to years of exposure. For the first seven years of therapy, the risk of cardiovascular events increased by 27% per year. The data in all studies are not entirely consistent or without flaws, but the general trend appears to be toward more cardiovascular disease in patients with HIV who are taking long-term antiretroviral therapy. The increase in cardiovascular disease among patients taking protease inhibitors could not be explained by changes in lipids alone.21
These data emphasize the importance of cardiovascular disease among patients with HIV taking antiretroviral therapy. HIV infection may also speed the progression of vascular disease by increasing inflammation and endothelial damage. Patients with HIV are also far more likely to use tobacco than the population at large. As death rates from traditional opportunistic infections and cancers have declined among patients with AIDS, treatment and prevention of cardiovascular disease among patients taking antiretroviral therapy is increasingly important. Emergency medicine physicians should be alert to the very high risk of cardiovascular disease in patients with HIV who have been treated with protease inhibitors for a long period of time.21
Diagnosing primary HIV infection is another area in which emergency medicine physicians can impact public health. The benefits of long-term antiretroviral therapy are well-known, and patients cannot benefit from antiretroviral therapies unless they know that they are HIV positive. In addition, viral loads are quite high in the first few weeks following acquisition of HIV. During this time, blood and genital secretions of HIV patients are highly infectious, and informing patients of their HIV status during primary infection has major public health implications. Studies of couples who have sero-discordant HIV status have shown that transmission is most likely to occur when viral loads are highest in the early and late phases of HIV infection.22
Primary HIV is a nonspecific febrile syndrome that resembles mononucleosis. Aseptic meningitis, lymphadenopathy, and skin rash may also be present. The diagnosis should be strongly suspected in patients who inject drugs and engage in high-risk sexual behaviors. This emphasizes the importance of asking patients with poorly defined viral syndromes about sexual history and substance use.22
HIV testing in acute infection is complicated by the fact that the body has frequently not yet mounted an antibody response. Viral load and antibody studies should be ordered. Positive testing for p-24 antigens also establishes the diagnosis of HIV, but is less sensitive than testing for viral load. If antibody studies are negative or indeterminate and viral load is high, a new diagnosis of HIV should be suspected. A viral load of < 10,000 may be a false positive in patients who are negative for HIV antibodies. In this situation, repeat viral load testing confirms the diagnosis of HIV if the viral load is rising.22
Pre-exposure prophylaxis is another promising and controversial development in HIV prevention. Pre-exposure prophylaxis involves giving high-risk patients antiviral drugs to prevent HIV acquisition. In one study, emtricitabine and tenofovir (FTC-TDF) or placebo was given to 2,499 HIV seronegative men or transgender women who have sex with men. After a median follow-up of 1.2 years, the group randomized to FTC-TDF had a statistically significant 44% reduction in acquisition of HIV. Among patients with study drugs detected in plasma and cell samples, the relative risk reduction in HIV acquisition was greater than 90%.23
The study was not without concerns. Patients given FTC-TDF had more nausea and unintentional weight loss than patients given placebo. Administration of some antiretroviral drugs in patients with newly acquired HIV infection may increase the risk of drug resistance. Tenofovir can impair renal function, but this usually resolves with discontinuation of the drug.23 Finally, patients in the study were given comprehensive information about HIV prevention. Without this training, it is not clear what impact FTC-TDF would have on risky sexual behaviors. While this drug combination is effective for preventing HIV acquisition, it provides little or no protection against many other STDs.
Patients may also present to emergency departments after potentially being exposed to HIV. In health care workers exposed to HIV occupationally, a case control study showed that zidovudine reduced the risk of HIV acquisition by more than 80%. These data have been extrapolated to nonoccupational HIV exposures. These exposures may occur in the setting of a sexual assault, barrier contraception failure during sex with an HIV-positive individual, anonymous unprotected sex, or injection drug use. In patients with chronic exposure to HIV, education on preventative measures and pre-exposure prophylaxis may be more appropriate. Antiretroviral drugs are also highly effective in preventing the transmission of HIV from mother to infant, but the regimens are beyond the scope of this article.
Postexposure prophylaxis (PEP) may be offered to patients up to 72 hours after a potential exposure to HIV. HIV testing is essential, both to avoid giving PEP to patients who are already HIV positive and to document the patient's HIV negativity at the time of the exposure. ELISA tests can rapidly determine the HIV status of the patient. The decision to give PEP must be guided by the risk of the encounter and the desire of the patient to pursue PEP. The CDC has published guidelines about who should be offered PEP. HIV specialists can also be helpful in making these decisions. The risk of acquiring HIV from a single episode of consensual unprotected vaginal intercourse with an HIV-positive individual is about 0.1 to 0.2%. With consensual receptive anal intercourse, the risk may be 3% or higher per episode. In traumatic sex, sex in the presence of genital ulcers, or sex with individuals with acute HIV infection, the risk of infection is probably much higher.12 Treatment should continue for 28 days, and the patient should receive close follow-up and repeat testing for seroconversion.24
Human Papilloma Virus
In the United States, there are about 6 million new cases of human papilloma virus annually.6 Most of these individuals clear the virus without incident. Some remain chronically infected. HPV causes genital warts and some malignancies.12 HPV has been linked to cancers of the cervix, anus, head and neck, vulva, penis, and vagina.6
HPV vaccination is highly effective in preventing cervical cancer. The two available vaccines (Cervarix and Gardasil) cover HPV 16 and 18, which cause the majority of cervical cancers. Gardasil also covers two strains of HPV that cause genital warts.12 Data are still evolving with other cancers, but there is good reason to suspect that other HPV-related cancers will also be prevented by HPV vaccination.
Emergency physicians are frequently asked to evaluate rectal bleeding. In addition to the usual diagnoses, anal cancer should be considered, which can be related to HPV infection. This is especially true among men who engage in receptive anal intercourse, women with a history of cervical cancer, patients with perianal HPV infections, and patients who have undergone organ transplantation.25 In addition to rectal examination and anoscopy, patients should be questioned about risk factors for anal HPV infection. Patients should also be referred to their primary care provider for anal PAP tests if they are at high risk.25 The rate of anal cancer among men who practice insertive anal intercourse is similar to the rate of cervical cancer among women prior to the advent of PAP testing.25
Treatment of Pelvic Inflammatory Disease
Women with PID generally require more intensive therapy than patients with uncomplicated cervicitis. Patients with mild to moderate PID can usually be treated as outpatients with 100 mg of oral doxycycline twice daily for 14 days and either 250 mg of intramuscular ceftriaxone or 2 grams of intramuscular cefoxitin administered with 1 gram of oral probenecid. Metronidazole 500 mg twice daily for 14 days may be added to any of these regimens.12
Women should be admitted to the hospital and treated with intravenous antibiotics when severe PID is present. Severe PID is defined by pregnancy, nonadherence or intolerance to oral medications, the presence of nausea, vomiting, high fever, tubo-ovarian abscess, or lack of clinical response to oral antibiotics.12 Clinical judgment should also guide the decision to admit patients to the hospital. Patients in whom surgical emergencies cannot be excluded should also be admitted to the hospital.12 Cholecystitis, ascending cholangitis, appendicitis, and diverticulitis can all clinically mimic PID.
Hospitalized patients with severe PID may be treated with a number of regimens. These include 100 mg of doxycycline oral or IV every 12 hours plus cefotetan 2 grams IV every 12 hours or cefoxitin 2 grams IV every six hours. Alternative regimens include clindamycin 900 mg IV every 8 hours plus IV or IM weight-based gentamicin dose either once or every 8 hours or 100 mg of oral or IV doxycycline every 12 hours plus 3 grams of IV ampicillin sulbactam every 6 hours.
Tubo-ovarian abscess should be suspected in any patient with an adnexal mass. Ruptured tubo-ovarian abscesses are surgical emergencies that require urgent surgical intervention and intravenous antibiotic therapy.26 Patients with ruptured tubo-ovarian abscesses frequently present with an acute abdomen and sepsis. Nonruptured tubo-ovarian abscesses have traditionally been drained surgically, but intravenous antibiotics without surgery may be adequate for patients with small tubo-ovarian abscesses. If patients worsen or do not improve on antibiotics alone, surgical intervention may be indicated. Some tubo-ovarian abscesses are treated with a combination of antibiotics and minimally invasive surgical drainage.27 Antibiotic regimens are complex and typically involve 2-3 drug therapy. Decisions about specific antibiotic therapies should be made in concert with the surgeon managing the patient.
Correction
In the April 9, 2012, issue of Emergency Medicine Reports, CME question 10 should read: "A stroke in the posterior circulation may present with all of the following except."
We apologize for the error.
References
1. Centers for Disease Control and Prevention. http://www.cdc.gov/std/chlamydia/STDFact-Chlamydia.htm. Accessed 4/30/2012.
2. Centers for Disease Control and Prevention. http://www.cdc.gov/std/gonorrhea/STDFact-gonorrhea.htm. Accessed 4/30/2012.
3. Centers for Disease Control and Prevention. http://www.cdc.gov/std/Herpes/STDFact-Herpes.htm. Accessed 4/30/2012.
4. http://bmj-sti.highwire.org/content/85/6/416.abstract. Accessed 5/11/12.
5. Centers for Disease Control and Prevention. http://www.cdc.gov/hiv/topics/surveillance/basic.htm#hivest, accessed 4/30/2018
6. Centers for Disease Control and Prevention. http://www.cdc.gov/std/HPV/STDFact-HPV.htm#common. Accessed 4/30/2012.
7. Centers for Disease Control and Prevention. http://www.cdc.gov/std/syphilis/STDFact-Syphilis.htm. Accessed 4/30/2012.
8. Centers for Disease Control and Prevention. http://www.cdc.gov/std/stats10/other.htm. Accessed 4/30/2012.
9. Centers for Disease Control and Prevention. http://www.cdc.gov/std/stats10/tables/a2.htm. Accessed 4/30/2012.
10. Centers for Disease Control and Prevention. http://www.cdc.gov/std/treatment/2010/genital-ulcers.htm#chancroid.
11. Forbes BA, Sahm DF, Weissfeld AS. Bailey and Scott's Diagnostic Microbiology, 12th edition. St. Louis: Mosby Elsevier: 2007.
12. Centers for Disease Control and Prevention. http://www.cdc.gov/std/treatment/2010/pid.htm.
13. UpToDate.com. Clinical features and diagnosis of pelvic inflammatory disease. Last edited 3/19/2012.
14. UpToDate.com. Epidemiology, clinical manifestations and diagnosis of tubo-ovarian abscess. Last edited 2/14/2012.
15. UpToDate.com. Genital Chlamydia trachomatis infections in women. Last edited 11/29/2011.
16. UpToDate.com. Genital Chlamydia trachomatis infections in men. Last edited 11/29/2011.
17. UpToDate.com. Epidemiology, pathogenesis and clinical manifestations of Neisseria gonorrhea infection. Last edited 5/6/2011.
18. UpToDate.com. Treatment of urogenital gonococcal infections. Last edited 3/20/2012.
19. Centers for Disease Control and Prevention. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5716a5.htm?s_cid=mm5716a5_e.
20. UpToDate.com. Epidemiology, clinical manifestations and diagnosis of genital herpes simplex infections. Last edited 5/10/2011.
21. UpToDate.com. Epidemiology and pathogenesis of dyslipidemia and cardiovascular disease in HIV infected patients. Last edited 5/20/2011.
22. UpToDate.com, Primary HIV-1 infection: Diagnosis and treatment. Last edited 6/14/2011.
23. Grant RM, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med 2010;363:2587-2599.
24. Landovitz RJ, Currier JS. Postexposure prophylaxis for HIV infection. N Engl J Med 2009;361:1768-1775.
25. UpToDate.com. Classification and epidemiology of anal cancer. Last edited 4/4/12.
26. UpToDate.com. Anal intraepithelial neoplasia: Diagnosis, screening, prevention and treatment. Last edited 3/23/2012.
27. UpToDate.com. Management and complications of tuboovarian abscess. Last edited 2/15/2012.
Sexually transmitted diseases (STDs) are frequently encountered in clinical medicine. According to the Centers for Disease Control, approximately 2.8 million infections with Chlamydia trachomatis and 700,000 infections with Neisseria gonorrhea occur annually in the United States.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
