Acute Movement Disorders in Children: Emergency Department Presentation and Evaluation
Acute Movement Disorders in Children: Emergency Department Presentation and Evaluation
Authors: Ewen Wang, MD, Associate Professor of Surgery, Associate Director of Pediatric Emergency Medicine, Stanford University Medical School, Stanford, CA.
Kristin Fontes, MD, Stanford/Kaiser Emergency Medicine Residency Program, Stanford, CA.
Peer Reviewer: Ghazala Q. Sharieff, MD, FACEP, FAAEM, FAAP, Director of Pediatric Emergency Medicine, Palomar Pomerado Health System/California Emergency Physicians, Clinical Professor, University of California, San Diego.
Abnormal movements, outside of seizure and ataxia, are an uncommon chief complaint among children presenting to the emergency department. A working knowledge of movement disorder phenomenology, etiology, differential diagnosis, and associated potentially life-threatening conditions is essential for emergency physicians. Each movement disorder and its clinical considerations will be discussed in detail in the article.
Ann M. Dietrich, MD, Editor
Epidemiology
As a group, acute movement disorders (see Table 1) in previously healthy children are rare. Some epidemiologic data exist for the more common disorders such as tic disorders, dystonia, and tremor; however, the remaining disorders have not been studied in this context. Even in movement disorder specialty centers, many disorders (such as myoclonus and chorea) account for less than 10% of the patient population.1 While one is more likely to encounter abnormal movements due to seizure or ataxia in the emergency department, it is nonetheless important to know how to distinguish a true movement disorder.
Etiology
The potential etiologies of an acute movement disorder in a previously healthy and developmentally normal child are numerous, but can be divided into a few major categories: infectious, inflammatory, autoimmune, structural, toxic/metabolic/drug-induced (also known as extrapyramidal adverse effects), and psychogenic (as a diagnosis of exclusion). (see Table 2.)
Drug-induced movements are important to consider in the workup of a new movement disorder. Movement disorders are a known potential adverse effect to antipsychotic medications in particular, and the use of these medications among children has increased in recent years.2,3 The prevalence of youth using more than one psychotropic medication has also increased.4
Definition and Pathophysiology
Abnormal movements can result from an insult to components of the cerebral cortex, basal ganglia, cerebellum, or spinal cord.
The planning and execution of movements start in the motor cortex. In children, damage to the motor cortex may result in paresis (as in adults with stroke), but may also manifest as seizure, or abnormal paroxysmal activity resulting from excessive synchronous discharges from cortical neurons. Seizure is the most common cause for life-threatening abnormal movements in children, with an estimated 1% of children experiencing an afebrile seizure before the age of 14.1 The basal ganglia are the deep nuclei of the brain that control the direction, speed, and amplitude of movement. In general, neurons of the basal ganglia use GABA as a neurotransmitter, leading to a system of inhibition and disinhibition to control movement. The facilitation of movement is largely dominated by dopamine, which becomes important in conditions such as Parkinson's disease (in which a depletion of dopamine leads to bradykinesia). The coordination and precision of movements are among the many functions of the cerebellum. Children with cerebellar pathology usually present with ataxia, usually manifested as disturbed gait. The most common cause of ataxia in a child is acute cerebellar ataxia (accounting for up to 50% of cases), which is thought to result from antiviral or autoimmune antibodies reacting against cerebellar tissues after a viral infection such as varicella or Epstein-Barr virus.5 However, it remains a diagnosis of exclusion after other life-threatening causes are ruled out. (see Table 2.)
Pathology of any of the motor pathways in these structures can result in abnormal movements characterized as either excessive involuntary movements that interfere with normal functioning (hyperkinetic or dyskinetic) or suppression or slowing of voluntary movements (hypokinetic or bradykinetic).
Clinical Features
The evaluation of movement disorders requires both a detailed history and neurologic examination to document the movements (if they are occurring at the time of presentation) and to identify any other abnormalities.
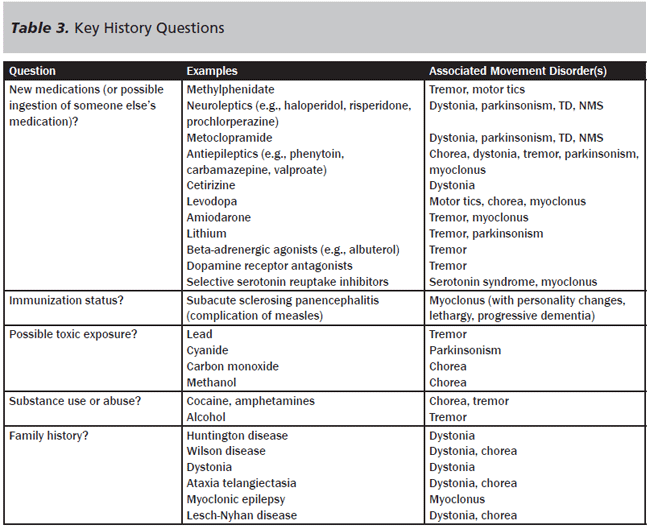
History. As in many pediatric histories, parents and caregivers can provide valuable information simply based on their prior observations of the child. For example, in the case of movements involving the dominant hand, one might observe the child switching to the non-dominant hand to perform routine tasks (such as eating or writing). The patient may describe an inability to control the movements or briefly suppress them, interference with normal activities, associated muscular weakness, and pain. Table 3 provides a list of important history questions that may shed light on the etiology of the abnormal movements. As with all pediatric patients, it is important to document past medical and surgical histories, family history, social history, medications, immunization status, and allergies.
The Neurologic Examination in Children. The approach to the neurologic examination in children is similar to that in adults, but may be limited depending on the child's age and cooperation. In the case of a new movement disorder, observing the child (ideally, prior to entering the room and being seen by the child) is essential and can provide a wealth of additional information, including motor skills, gait, and the ability to perform voluntary movements. It is also useful to have the child perform a series of specific tasks to see if the movements are altered (either suppressed or enhanced); this may be helpful when evaluating a tremor, for example. While movement disorders can usually be distinguished from one another based on one or more movement components, including timing/frequency, speed, duration, amplitude, characteristics, and associated findings observed, note that a patient may present with more than one movement type. Refer to see Table 1 for distinguishing features of each movement disorder.
Hyperkinetic Movement Disorders
While few pediatric movement disorder epidemiologic studies exist, available data suggest that the hyperkinetic disorders are by far more common than hypokinetic, possibly accounting for greater than 90% of cases.6
Tics. Tics are repeated, individually recognizable (stereotyped), intermittent movements or movement fragments that are almost always briefly suppressible and are usually associated with awareness of an urge to perform the movement.4Tics are the most common of the hyperkinetic movement disorders, with a male predominance of 3:1 and a lifetime prevalence of 20% or greater in boys.7 They most frequently manifest in the first decade of life, with a median age of onset of 6 to 7 years.8 Motor tics can manifest as simple (one muscle group) or complex (multiple muscle groups), and are easily identified by their stereotyped and predictable nature (for example, intermittent shoulder shrugging). The most distinguishing feature, however, is the premonitory urge that patients describe prior to each tic. Acute onset motor tics usually do not lead to serious injuries or falls. Malignant Tourette syndrome, reported in about 5% of Tourette syndrome patients,22 involves a variable presentation of severe tics, suicidal ideation, and self-injurious behavior. A small number of patients may also present with psychotic symptoms, including hallucinations and paranoia.6
An interesting but controversial topic relating to acute onset tics/Tourette syndrome and/or obsessive compulsive disorder (OCD) symptoms is the pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection (PANDAS), whose presence in the medical literature and the media is increasing. While much debate exists about this condition, it may be beneficial to test a child with a new-onset OCD and/or tic disorder for an underlying group A streptococcal infection, which, if identified, requires treatment.23
Stereotypies. Stereotypies are repetitive, simple movements that can be voluntarily suppressed.4 Of the hyperkinetic movement disorders, stereotypies are the most benign. They are single, simple back-and-forth movements usually of the upper extremities, and are often precipitated by emotional excitement or stress.4 The child is usually able to perform normal activities. As with tics, children with a stereotypy can be distracted from performing the movement, and they can be voluntarily stopped. Unlike tics, there is no premonitory urge to perform the movement.
Stereotypies are somewhat common in healthy preschool-aged children17 and are also seen in children with autism spectrum disorders.24 One ominous condition that can present with stereotypies is Rett syndrome, a genetically based developmental regression that most commonly affects girls and is classically associated with a "hand-wringing" stereotypy.25 Although there are no established drug therapies, follow-up with a pediatric neurologist is recommended.
Dystonia. Dystonia involves involuntary sustained or intermittent muscle contractions causing twisting and repetitive movements, abnormal postures, or both.4 Dystonia in children is usually a primary disorder, most frequently seen in patients with dyskinetic cerebral palsy or a genetic mutation. Dystonic movements are commonly precipitated by attempts at voluntary movement (and can be task-specific, e.g., the patient has dystonic movement while walking forward but not backward) and usually do not persist during sleep. They can be distinguished from other movement disorders by their consistent pattern and by the sustained abnormal postures that result.
In a previously neurologically normal child, a secondary cause for dystonia must be evaluated. With a negative family history and an absence of signs of neurodegeneration (e.g., personality changes, dementia), the workup should focus on infectious and drug-induced causes. (see Table 2.)
Drug-induced dystonic reactions (DIDR) are well-documented in the literature, and most commonly result after starting a therapeutic dose of a neuroleptic26 (e.g., haloperidol) or antiemetic medication (e.g., ondansetron,27 metoclopramide28), although many other drug types have been associated.11,29 The majority of DIDRs occur within 5 days of starting a new drug.26,30
The typical presentation involves focal posturing of the head and neck, but one retrospective study found 7% of children with a DIDR presented with laryngospasm, an airway emergency.31 In addition to removing the offending agent, multiple sources recommend IV diphenhydramine in the acute management.13 Benzodiazepines, anticonvulsants, and antispasmodics (e.g., baclofen) may also be tried if the movements are not adequately controlled after multiple doses. A child with a history of drug-induced dystonia is at increased risk for a second episode; thus, take extreme caution when deciding whether to administer any agent that has been implicated in the development of acute dystonic reactions.
Status dystonicus, a condition of generalized, unremitting dystonia, is usually limited to patients with pre-existing dystonia and can be precipitated by infection or medication (initiation or discontinuation). The therapeutic approach is similar to that for acute dystonic reactions, but these patients require close monitoring in an intensive care unit setting while undergoing treatment.
Chorea. Chorea is an ongoing, random-appearing sequence of one or more discrete involuntary movements or movement fragments.4 While chorea is classically taught as a "dance-like" movement disorder involving all extremities, patients can also present with hemichorea. Chorea can also occur with athetosis, which is a slower, writhing movement limited to the extremities. The movements of chorea can be brief and jerky (as seen with Sydenham chorea) or slower and flowing (as seen in Huntington disease).
The best-described and most common acquired form is Sydenham chorea (SC), a major manifestation of acute rheumatic fever (ARF) according to the Jones criteria.14 Children with SC generally develop sudden-onset jerky movements involving the face and extremities and can also have muscular weakness (clinically demonstrated most easily as the "milkmaid's grip," or an inability to maintain a handgrip). Dysarthria, personality changes, and emotional lability are also common. SC is typically a later manifestation of ARF (with polyarthritis and carditis occurring earlier), developing within 1-6 months after a group A streptococcal infection.32 In the appropriate clinical setting, new-onset SC should prompt an evaluation for carditis.32,33 It has been demonstrated that children treated for ARF in the setting of SC who are free of carditis or valvulitis at diagnosis will be free of heart disease if there is no recurrence of ARF. Children with an audible murmur at diagnosis can expect resolution within the first year.33
In female patients of child-bearing age, a pregnancy test should be included in the diagnostic workup of new chorea. Chorea gravidarum has become a rare phenomenon since the advent of penicillin in the treatment of ARF. It usually begins in the first or early second trimester and resolves in the third trimester or shortly after birth.34 Today, it is most commonly seen in patients with systemic lupus erythematosus (SLE) and the antiphospholipid antibody syndrome (APS), disorders which are themselves associated with spontaneous abortion and other complications.35
Chorea occurs in about 10% of children with SLE. Neurologic manifestations are common in SLE, and are the first symptom in as many as one-third to two-thirds of children; the most common manifestation is seizure.36 Chorea is somewhat more variable (i.e., can occur before or after diagnosis of SLE) and seems to be more common in girls.37 CNS involvement conveys a worse prognosis.21
While cerebrovascular disease in children is very rare, it should be considered in the differential diagnosis of new chorea or hemichorea. Reported cases of chorea/hemichorea in children thought to be due to a vascular cause include moyamoya disease (a chronic, progressive cerebrovascular disease characterized by bilateral occlusion of the carotid and circle of Willis arteries, with resultant prominent collateral circulation resembling a "cloud of smoke" on angiography),38,39 post-pump chorea (a phenomenon limited to children who have undergone recent extracorporeal bypass, as in cardiac surgery),40 and diabetes mellitus. Cases of this movement disorder in diabetes have been reported for both diabetic ketoacidosis and nonketotic hyperglycemia.41 Fortunately, resolution of this movement disorder can be achieved with tight glucose control, although neuroleptic medication is also usually required. This is in contrast to ischemic or hemorrhagic stroke, in which the chorea persists despite adequate treatment.
Myoclonus. Myoclonus involves a sequence of repeated, often nonrhythmic, brief, shock-like jerks due to sudden involuntary contraction or relaxation of one or more muscles.4 Myoclonus occurs in all age groups in childhood, from benign neonatal sleep myoclonus to juvenile myoclonic epilepsy to essential myoclonus (which usually develops before age 20). While myoclonus can involve numerous body regions, the characteristic feature is the very rapid "shock-like" duration of each movement; this generally helps to distinguish myoclonus from other hyperkinetic movement disorders. However, myoclonus may be difficult to distinguish from tics and tremor, which can also be quite rapid. Tics generally do not persist during sleep, whereas myoclonus frequently does (and some forms of myoclonus occur only during sleep). Quite possibly the best distinguishing factors, however, are the premonitory urge and distractibility that are virtually unique to tics. Tremor is best distinguished by its rhythmicity.
In addition to epileptic and nonepileptic classes of myoclonus, there are also several subtypes named for the anatomic origin (cortical, subcortical, spinal) and precipitants of movement (spontaneous, reflex, action). Cases of febrile myoclonus have also been reported and, like simple febrile seizures, resolve with temperature correction.42 If it is an isolated and non-progressive symptom, myoclonus is usually benign. However, acute onset, progressive or symptomatic myoclonus in a previously healthy child tends to be an ominous sign and should prompt evaluation for a treatable cause.43
Myoclonus may occur after a cerebral anoxic event such as with drowning/near-drowning or cardiac arrest. Seizure precautions should be initiated for all patients with new symptomatic myoclonus, as it is frequently associated. Furthermore, in any patient with features of epileptic myoclonus (e.g., positive family history of seizures, myoclonus upon awakening, increasing frequency of myoclonic jerks, signs of encephalopathy), the workup should include an electroencephalogram (EEG). Benign myoclonus should have a normal EEG.
The acute management of symptomatic myoclonus should involve anticonvulsant therapy, whether or not the etiology is epileptic. Clonazepam (4-10 mg/day) and/or valproic acid (250-4200 mg/day) are effective options.44
A rare but important phenomenon worth mentioning is opsoclonus-myoclonus-ataxia syndrome (OMA, also known as "dancing eyes-dancing feet"). OMA is characterized by subacute limb and truncal myoclonus with involuntary and multi-directional saccades. Children with previously normal gait may develop an ataxia as well. The two most important etiologies to evaluate for children with new OMA are post-viral and paraneoplastic. Fifty percent of patients with OMA will have an underlying neuroblastoma.45 Any child presenting with OMA should, thus, have urgent imaging (CT or MRI of the chest/abdomen/pelvis) to further evaluate for neuroblastoma.46 Excluding a brain lesion with CT/MRI is also useful, and if acute infection is suspected, CSF analysis should be included in the workup; analysis may be normal or reveal a lymphocytic pleocytosis.47 Removal of a malignant mass alone does not result in complete resolution of the movement disorder, but most children benefit from immunologic therapy (e.g., steroids, IVIG).
Tardive Dyskinesia (TD). This condition involves a variable mixture of orofacial dyskinesia, athetosis, dystonia, chorea, tics, and facial grimacing that appears with a delayed onset after prolonged use of dopamine receptor blocking agents.48 Like neuroleptic malignant syndrome (discussed later), the risk of TD increases with increasing dose and duration of treatment. The incidence of TD in adults undergoing long-term second-generation antipsychotic treatment is less than 1%, and probably even lower in children and adolescents.49 In children, TD may also occur with withdrawal of an antipsychotic agent. A recent review demonstrated an increased risk of tardive dyskinesia in patients treated with risperidone for schizophrenia; the effect may be less prevalent among young children.50
Classically, TD presents as stereotyped movements of the lower face, most commonly the mouth and tongue.48 TD is rarely life-threatening, but closer observation and support may be required for patients with disabling movements (e.g., those that interfere with eating) or those with respiratory dyskinesias causing tachypnea or other signs of distress. Despite several studies evaluating pharmacologic therapy for TD, most have shown only slight to moderate benefit; the current focus, thus, is on prevention and early detection, although it may be helpful to consider a trial of benzodiazepine or anticholinergic medication to minimize the symptoms.51
Tremor. Tremor is the rhythmic oscillation of a body part, produced by alternating synchronous contractions of reciprocally innervated antagonistic muscles.4 Tremor is common in adults; up to one-third of adult patients with essential tremor report onset of the movement disorder in childhood or adolescence. The defining characteristic of tremor is a rhythmic pattern that distinguishes it from other rapid movements such as myoclonus and tics. Any body part can exhibit a tremor, but the vast majority are seen in the hands.52 Tremor is classified based on its associated movement triggers (rest, postural, action, intention). In children, the most common form is action tremor, which is further subdivided into physiologic and essential (familial).
Epidemiologic data for pediatric tremors are sparse, but available studies suggest that childhood-onset tremor is less common than adult-onset and is usually essential.53 Treatment with a beta adrenergic receptor antagonist, such as propranolol, may be effective for essential tremor in children (as it has been demonstrated in adults), although randomized trials are lacking.
A child with abrupt onset or rapidly progressive tremor should be evaluated for exposure to certain toxins or medications (such as antiepileptics, beta-agonists, stimulants, alcohol, or lead) and should undergo urgent evaluation with measurements of serum glucose, thyroid function tests, brain MRI (especially if focal neurologic deficits exist), and serum drug levels or urine toxicology screen (if applicable).
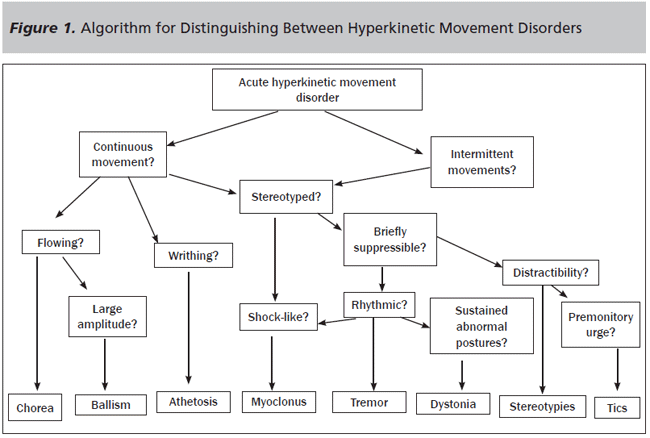
Summary. Figure 1 depicts a proposed manner in which one can distinguish between the hyperkinetic movement disorders, although mixed pictures may also occur and make precise identification more difficult.
Hypokinetic Movement Disorders
In contrast to the variety of hyperkinetic movement disorders, hypokinetic (or bradykinetic) disorders in children are limited to primary and secondary parkinsonism.
Parkinsonism. This condition is a syndrome of slowness of movement (bradykinesia), tremor in the hands or legs, rigidity of muscles, shuffling gait, and postural instability.4 Parkinsonism is exceedingly rare in children. Not surprisingly, then, few epidemiologic data are available. However, as with adults, parkinsonism can be a primary (e.g., Parkinson disease) or secondary. Parkinsonism classically presents with the "TRAP" signs and symptoms: tremor at rest, muscular rigidity, akinesia, postural instability. However, available case reports of parkinsonism in children suggest that a wider variety of presentations is possible, including non-motor complaints such as paucity of speech, mutism, autonomic dysfunction, and altered mental status.54 Patients can even present without the classic tremor in the akinetic-rigid subtype of parkinsonism.
Drugs are the most common etiology for new-onset parkinsonism in children. Reported precipitants are chemotherapeutics, antimicrobials, antipsychotics, and antiemetics (metoclopramide).55,56 Parkinsonism is much less commonly seen with structural brain lesions, infections, and hereditary syndromes (e.g. Wilson's disease, Huntington disease), but these should be considered in the workup if the appropriate clinical context exists. Regardless of the etiology of parkinsonism, the treatment of choice is levodopa (ideally prescribed by a pediatric neurologist).
Movement Disorders: Special Cases
Neuroleptic Malignant Syndrome (NMS). NMS is a syndrome of fever, rigidity, mental status change, autonomic dysfunction, and movement disorder resulting from exposure to dopamine receptor-blocking drugs.16 NMS is rare in adults (with incidence reports varying from 0.02-3%) and even more uncommon in children, with the literature for the latter being limited to case reports.57 It is a potentially fatal complication of treatment with antipsychotic medications such as haloperidol (with higher doses carrying an increased risk of developing NMS), and can have a variable presentation centered around hyperthermia, rigidity, and muscle damage.57 The pathogenesis involves central dopaminergic receptor blockage that occurs with use of these medications. Other risk factors include intramuscular administration and rapid increases in dose. The diagnostic criteria for NMS (based on the DSM-IV) include the development of severe muscle rigidity and elevated temperature associated with the use of neuroleptic medication with two or more of the following:
- diaphoresis
- dysphagia
- tremor
- incontinence
- changes in level of consciousness ranging from confusion to coma
- mutism
- tachycardia
- elevated or labile blood pressure
- leukocytosis
- laboratory evidence of muscle injury (e.g., elevated serum creatinine kinase).
It should be noted that while rigidity must be present by definition, patients can present with hyperkinetic movements as well. It is crucial to evaluate and exclude CNS infection if there is concern for this based on history or physical exam, as it can present similarly. The differential diagnosis for NMS also includes heat stroke, malignant hyperthermia, and serotonin syndrome.
Depending on the severity of symptoms and vital sign instability, treatment will range from supportive care and observation to ICU-level monitoring. Like most presentations associated with an adverse drug effect, the offending agent should always be discontinued. The mainstays of treatment are hydration to prevent renal damage and sedation (e.g., with benzodiazepines) to control agitation. Hyperthermia can be managed with external cooling, levodopa, bromocriptine, or dantrolene. Dialysis can be utilized in severe cases of renal dysfunction, although the neuroleptic medications themselves are not usually dialyzable. Early recognition and treatment may help prevent complications and death due to cardiac arrest, pulmonary embolism, and sepsis, and while it appears the mortality rate has declined over time, there remains a high risk of morbidity.16 If the neuroleptic medication must be continued, it should be slowly titrated to the desired dose over a period of two or more weeks to reduce the risk of recurrence of NMS.
Serotonin Syndrome. Serotonin syndrome is a condition of mental status change, abnormal neuromuscular tone, and autonomic dysfunction resulting from exposure to serotonergic drugs.17
Classic serotonin syndrome (SS) is a triad of fever, altered mental status, and myoclonus, which emerges in the setting of exposure to serotonergic drugs, including antidepressant medications (selective serotonin reuptake inhibitors [SSRI], serotonin-norepinephrine reuptake inhibitors [SNRI], monoamine oxidase inhibitors [MAOI], tricyclic antidepressants [TCA], etc.), although several less obvious culprits have been reported, such as analgesics (triptans, opiates except morphine) and illicit drugs (MDMA, LSD, cocaine). SS in young children has been reported through accidental ingestion or even after taking an appropriate weight-based dose.58,59 The presentation can be quite variable in severity, from mild myoclonic jerks to a seizure-like state leading to rhabdomyolysis. (See Table 4.) SS is diagnosed clinically and may be difficult to distinguish from NMS. Although the onset of SS is generally over several days, with treatment it resolves within 24 hours. The creatinine kinase elevation seen with virtually every case of NMS is much less common (though not unheard of) in SS. Myoclonus and hyperreflexia are more characteristic of SS, though they are not always seen.

The mainstay of treatment of SS is discontinuation of the serotonergic agent. Further management is largely supportive and consists of close vital sign monitoring, fluid resuscitation, external cooling, and sedation with benzodiazepines.17 In cases of severe hyperthermia (temperature greater than 41.1 degrees Celsius), control of excess muscular activity with paralysis and endotracheal intubation is necessary. However, paralysis with succinylcholine should be avoided in these patients due to possible rhabdomyolysis; a non-depolarizing paralytic agent (such as rocuronium or vecuronium) should be used instead. In contrast to NMS, dantrolene is not recommended in the management of hyperthermia in SS (as no survival benefit has been demonstrated in SS).17 Treatment with a serotonin antagonist may also be considered in severe cases. Cyproheptadine use is supported by several case reports in the literature, although there are no randomized trials studying its efficacy and, thus, no FDA approval exists for its use in SS.60
Psychogenic Movement Disorders
Hyperkinetic or hypokinetic movement disorders associated with an underlying psychological disorder in which an organic cause for the movements is excluded are psychogenic movement disorders.
Psychogenic movement disorders can present with any movement type. While this diagnosis is difficult to make due to a lack of established criteria, certain warning signs should alert providers to a potential psychopathology (in a patient without focal neurologic deficits and an otherwise normal workup):
Abrupt onset with rapid progression;
Multiple movement types with an inconsistent pattern, variability (e.g. changing from one movement type to another);
Distractibility with performing tasks;
Disability out of proportion to the movement disorder.
Note that a child who presents with abnormal movements and personality changes should be evaluated for anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis, especially in the setting of a viral prodrome (e.g., headache, fever, myalgias). A prospective study of 52 children with acute movement disorders found that 5 patients who presented primarily with behavioral alterations also demonstrated chorea, dystonia, or parkinsonism. These patients were diagnosed by positive NMDAR antibodies in the CSF and serum.18
Clearly, a psychogenic movement disorder is a diagnosis of exclusion and is not easily made in the emergency department, where the often extensive workup for a movement disorder starts. However, an awareness of this phenomenon is important as patients may require psychiatric consultation and follow-up. One small study identified a multidisciplinary approach and family involvement in treatment as integral to effectively treating a psychogenic movement disorder.61
Differential Diagnosis and Workup
Perhaps one of the most important distinctions to make early is whether one is dealing with a potential seizure, even after completing a comprehensive history and physical. While it may be difficult to distinguish seizure from hyperkinetic movements simply based on phenomenology, there are some features that lend more weight to one or the other. First, alteration of mental status in the setting of abnormal movements usually indicates seizure, although an acute process (such as CNS infection causing myoclonus or chorea) may also present with abnormal mentation. In addition, the presence of a premonitory aura (such as sensory symptoms or nausea) is more indicative of seizure, as is a post-ictal state or Todd's paralysis.62
Hyperkinetic movements may also present similar to cerebellar pathology, causing a variety of ataxias. Cerebellar ataxia can usually be distinguished by abnormal coordination and balance testing (e.g., as seen with the "drunken sailor" gait characteristic of truncal ataxia). A positive Romberg's test is a strong indicator of sensory ataxia due to a cerebellar or dorsal column abnormality.63 A child with a movement disorder may appear to have abnormal cerebellar tests on examination, although this may be a result of inability to control the baseline movements.
Management
As for all patients, emergency department management for a new movement disorder begins with assessing for stability of the airway, breathing, and circulation before proceeding with workup. In the context of movement disorders, patients with upper airway dystonia should be immediately assessed for emergent intubation while receiving positive pressure ventilation via bag-valve mask. This can be done while administering treatment (e.g. IV diphenhydramine) to reverse the dystonia (especially if a drug-induced cause is suspected). A patient with altered mental status should also be considered for intubation for airway protection. A child with SS or NMS may require intubation for hypoxic respiratory failure if there is concern for life-threatening pulmonary embolism (a known complication of these disorders). As stated previously, use of succinylcholine as a paralytic agent should be avoided in these patients due to possible rhabdomyolysis. Circulation can be maintained with intravenous crystalloids, particularly for those children with SS or NMS who may have rhabdomyolysis from severe muscle rigidity. Children who appear septic should be given several weight-based fluid boluses while obtaining blood cultures and administering broad-spectrum antibiotics. Consider vasopressors if fluid resuscitation fails to maintain adequate mean arterial pressure.
While movement disorders are diagnosed clinically, the suspected underlying cause may require laboratory and radiologic investigation. Table 5 lists a general differential diagnosis for each movement disorder and suggested workup, including studies that are widely available and can be obtained quickly in the emergency department. In most cases, brain imaging (computed tomography and/or magnetic resonance imaging) is a reasonable means to exclude structural pathology. Head computed tomography (CT) is readily available at many institutions and provides rapid results. However, certain lesions are better characterized using magnetic resonance imaging (MRI), which may require hospital admission to complete. It should be noted, however, that not every child with abnormal movements requires imaging in the emergency department. Patients with certain movement disorders, such as tics, are unlikely to have abnormal brain imaging.64
Children with abnormal movements who also present with fever or rigors should be evaluated for CNS infection and sepsis, including blood cultures (and subsequently empiric antibiotics if bacterial meningitis is suspected),65 CT brain, and lumbar puncture with CSF studies. Any child who presents with abnormal movements after head trauma should be risk-stratified for clinically important traumatic brain injury according to the validated prediction rules established by the PECARN group.66 It is also important to consider evaluation for non-accidental trauma in any child presenting with an acute neurological problem, especially in the setting of multiple injuries or inconsistent history. If one is unable to distinguish between a movement disorder and seizure, EEG (particularly with video monitoring) may be considered as part of the workup.
A child without altered mental status or constitutional signs/symptoms (such as fever or vomiting) who has an unremarkable history, is functioning normally in their daily activities, and who isn't at risk for injury due to their movements may be better candidates for outpatient follow-up and further evaluation.
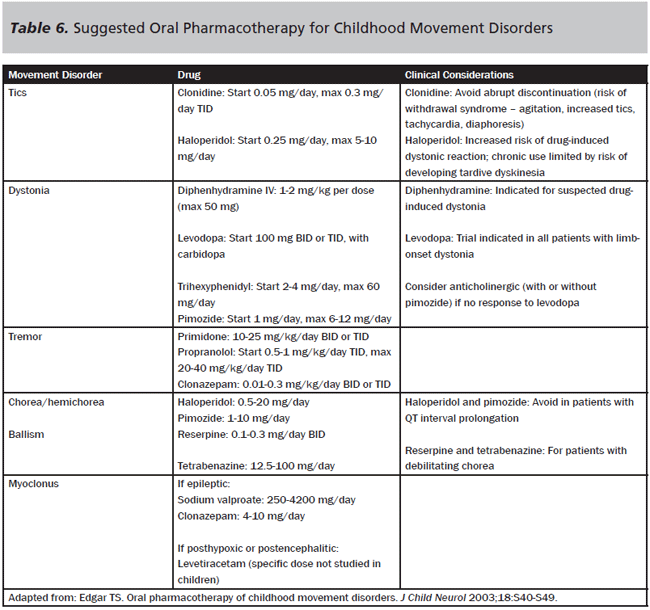
Once the movement disorder has been confidently diagnosed clinically, it is reasonable to begin therapy to minimize or stop the movements, particularly for patients who are injured or distressed. Table 6 lists some suggested drug therapies. If possible, however, the movements should be documented as best as possible prior to treatment (ideally, on video with consent). A pediatric neurologist should be consulted for specific drug recommendations prior to starting medication, as he or she will follow up with the patient after discharge.
Disposition
Most children with new movement disorders can safely be discharged home after a thorough evaluation. Certain situations may require longer ED observation or brief hospital admission, such as the need for further workup and control of symptoms. Admission is necessary in conditions for which airway monitoring is needed, for patients with disabling movements or those suffering physical injury due to the movement disorder, and for treatment of reversible and potentially harmful movement disorders (e.g., SS, NMS). In addition, children may benefit from consultation with a physical and/or occupational therapist in adaptive gait training and activities of daily living (ADLs) to function as independently as possible, including return to school.
Regardless of disposition, every child with a new movement disorder should have consultation and follow-up with a pediatric neurologist in addition to their primary physician, even when a reversible cause is identified, and particularly if drug therapy is initiated.
Reassurance and education are important interventions for patients and their families. While most movement disorders are easily distinguished from seizure on the basis of classic movement patterns and maintenance of consciousness, it is important to reassure parents and caregivers who may be most concerned about a seizure. Furthermore, the fact that most movement disorders in previously healthy children are not life-threatening and not permanent should be established, although the expected timeline of recovery will vary based on the specific movement disorder.
Educating patients and families about the spectrum of etiologies for most movement disorders should occur early in the evaluation, which may take several hours in the ED. For patients discharged home, personalized discharge instructions are another way in which emergency physicians can educate about movement disorders, especially in the case of drug-induced syndromes to provide a reminder about offending agents and recommend notifying other healthcare providers about avoiding specific medications.
Additionally, it is important to address patients' and families' expectations about treatment of a movement disorder. If drug therapy is started in the emergency department, discharge instructions should contain detailed medication instructions and potential adverse effects (including the risk of developing a drug-induced dystonic reaction with a neuroleptic agent). If a reversible cause is found (e.g. drug-induced dystonia, acute rheumatic fever), patients may expect complete resolution of the abnormal movements but may not achieve this immediately. Sydenham chorea, even with appropriate antibiotic and immunomodulatory treatment, may take several months to fully resolve. Furthermore, children with a previously diagnosed movement disorder are at higher risk for recurrence. Appropriate follow-up is essential for long-term monitoring and management.
References
1. Fernández-Alvarez E. Prevalence of paediatric movement disorders. In: Thénenin M, ed. Paediatric Movement Disorders: Progress in Understanding. Montrouge, France: John Libbey Eurotext; 2005:1-18.
2. Alexander GC, et al. Increasing off-label use of antipsychotic medications in the United States, 1995-2008. Pharmacoepidemiology and Drug Safety 2011;20:177-184.
3. Olfson M, et al. Trends in antipsychotic drug use by very young, privately insured children. J Am Acad Child Adolesc Psychiatry 2010;49;1:13-23.
4. Comer JS, et al. National trends in child and adolescent psychotropic polypharmacy in office-based practice, 1996-2007. J Am Acad Child Adolesc Psychiatry 2010;49;10:1001-1010.
5. Helm M. Acute cerebellar ataxia. UpToDate 2012.
6. Fernandez-Alvarez E, Aicardi J. Movement Disorders in Children. London: High Holborn House. 2001:19.
7. Sanger TD, et al. Definition and classification of hyperkinetic movement disorders in childhood. Movement Disorders 2010;25;11:1538-1549.
8. Schlaggar BL, Mink JW. Movement disorders in children. Pediatrics in Review 2003;24:39-51.
9. Mahone EM, et al. Repetitive arm and hand movements (complex motor stereotypies) in children. J Pediatr 2004;145:391-395.
10. Jacobsen RC. Out-of-hospital lingual dystonia resulting in airway obstruction. Prehosp Emerg Care 2011;15:537-540.
11. Esen I, et al. Cetirizine-induced dystonic reaction in a 6-year-old boy. Pediatric Emerg Care 2008;24;9:627-628.
12. Jamuar SS, et al. Rhabdomyolysis, acute renal failure, and cardiac arrest secondary to status dystonicus in a child with glutaric aciduria type I. Molecular Genetics and Metabolism 2012;106:488-490.
13. Robottom BJ, Shulman LM, Weiner WJ. Drug-induced movement disorders: Emergencies and management. Neurol Clin 2012;30:309-320.
14. Gibofsky A, Zabriskie JB. Clinical manifestations and diagnosis of acute rheumatic fever. UpToDate 2010.
15. Przekop A, Sanger TD. Birth-related syndromes of athetosis and kernicterus. Handbook of Clinical Neurology 2011;100:387-395.
16. Wijdicks EFM. Neuroleptic malignant syndrome. UpToDate 2012.
17. Boyer EW. Serotonin syndrome. UpToDate 2012.
18. Dale RC, et al. A prospective study of acute movement disorders in children. Dev Med Child Neurol 2010;52:739-748.
19. Agrawal D. Approach to the child with acute ataxia. UpToDate 2012.
20. Wilfong A. Overview of the classification, etiology, and clinical features of pediatric seizures and epilepsy. UpToDate 2012. 22. Cheung MC, Shahed J, Jankovic J. Malignant Tourette Syndrome. Movement Disorders 2007;22;12:1743-1750.
21. Mink JW, Zinner SH. Movement disorders II: Chorea, dystonia, myoclonus, and tremor. Pediatrics in Review 2010;31:287-295.
22. Cheung MC, Shahaed J. Jankovic J. Malignant Tourette syndrome. Movement Disorders 2007;22;1743-1750.
23. Pinchichero ME. PANDAS: Pediatric autoimmune neuropsychiatric disorder associated with group A streptococci. UpToDate 2012.
24. Lewis MH, Bodfish JW. Repetitive behavior disorders in autism. Ment Retard Dev Disabil Res Rev 1998;4:80-89.
25. Goldman S, Temudo T. Hand stereotypies distinguish Rett syndrome from autism disorder. Movement Disorders 2012;27;8:1060-1062.
26. Swett Jr. CS. Drug-induced dystonia. Am J Psychiatry 1975;132;5:532-534.
27. Patel A, et al. Ondansetron-induced dystonia, hypoglycemia, and seizures in a child. Ann Pharmacotherapy 2011;45:e7.
28. Boulloche J, et al. Dystonic reactions with metoclopramide: Is there a risk population? Helv Paediatr Acta 1987;42;5-6:425-432.
29. Singh MK, et al. Acute dystonia associated with aripiprazole in a child. J Am Acad Child Adolesc Psychiatry 2007;46;3:306-307.
30. Ayd Jr. FJ. A survey of drug-induced extrapyramidal reactions. JAMA 1961;175;12:1054-1060.
31. Derinoz O, Caglar AA. Drug-induced movement disorders in children at paediatric emergency department: 'Dystonia.' Emerg Med J 2012 March 7 (Epub ahead of print).
32. Weiner SG, Normandin PA. Sydenham chorea: A case report and review of the literature. Pediatric Emerg Care 2007;23;1:20-24.
33. Panamonta M, et al. The relationship of carditis to the initial attack of Sydenham's chorea. International J Cardiology 2004;94:241-248.
34. Lee MJ, Hickenbottom S. Neurologic disorders complicating pregnancy. UpToDate 2011.
35. Robottom BJ, Weiner, WJ. Chorea gravidarum. Handbook of Clinical Neurology 2011;100:231-235.
36. Quintero-Del-Rio AI, Miller V. Neurologic symptoms in children with systemic lupus erythematosus. J Child Neurology 2000;15(12):803-807.
37. Parikh S, et al. Neurologic characteristics of childhood lupus erythematosus. Pediatr Neurol 1995;13:198-201.
38. Gonzalez-Alegre P, et al. Moyamoya-induced paroxysmal dyskinesia. Mov Disord 2003;18:1051-1056.
39. Suwanwela NC. Moyamoya disease: Etiology, clinical features, and diagnosis. UpToDate 2012.
40. Robinson RO, et al. Choreic syndrome after cardiac surgery. Arch Dis Child 1988;63:1466-1469.
41. Mihai CM, et al. Atypical onset of diabetes in a teenage girl: A case report. Cases Journal 2008;1:425.
42. Dooley JM, Hayden JD. Benign febrile myoclonus in childhood. Can J Neurol Sci 2004;31:504-505.
43. Pranzatelli MR. Myoclonus in childhood. Semin Pediatr Neurol 2003;10;1:41-51.
44. Edgar TS. Oral pharmacotherapy of childhood movement disorders. J Child Neurol 2003;18:S40-S49.
45. Russell HV, et al. Clinical presentation, diagnosis, and staging evaluation of neuroblastoma. UpToDate 2012.
46. Brunklaus A. Investigating neuroblastoma in childhood opsoclonus-myoclonus syndrome. Arch Dis Child 2011 Apr 3 (Epub ahead of print).
47. Dalmau J, Rosenfeld MR. Paraneoplastic syndromes causing movement disorders. Handbook of Clinical Neurology 2011;100:315-321.
48. Tarsy D. Tardive dyskinesia: Clinical features and diagnosis. UpToDate 2012.
49. Correll CU, Kane JM. One-year incidence rates of tardive dyskinesia in children and adolescents treated with second-generation antipsychotics: A systematic review. J Child Adol Psychopharm 2007;17;5:
647-655.
50. McDonagh M, et al. Drug class review: Atypical antipsychotic drugs: Final update 3 report. Portland (OR): Oregon Health & Science University; July 2010.
51. Tarsy D. Tardive dyskinesia: Prevention and treatment. UpToDate 2012.
52. Jankovic J, et al. Essential tremor among children. Pediatrics 2004;114;5:
1203-1205.
53. Ferrara J, Jankovic J. Epidemiology and management of essential tremor in children. Pediatr Drugs 2009;11;5:293-307.
54. Pranzatelli MR, et al. Clinical spectrum of secondary parkinsonism in childhood: A reversible disorder. Pediatric Neurology 1994;10;2:131-140.
55. Chutorian AM, et al. Toxic pediatric parkinsonism: Report of a child with metabolic studies and response to treatment.
J Child Neurol 2003;18:812-815.
56. Mott SH, et al. Encephalopathy with parkinsonian features in children following bone marrow transplantations and high-dose amphotericin B. Ann Neurol 1995;37:810-814.
57. Ty EB, Rothner DA. Neuroleptic malignant syndrome in children and adolescents. J Child Neurol 2001;16;3:157-163.
58. Marechal C, et al. Serotonin syndrome induced by tramadol intoxication in an 8-month-old infant. Pediatric Neurology 2011;44;1:72-74.
59. Phan H, et al. Serotonin syndrome following a single 50 mg dose of sertraline in a child. Clinical Toxicology 2008;46:845-849.
60. Graudins A, et al. Treatment of the serotonin syndrome with cyproheptadine.
J Emerg Med 1998;16;4:615-619.
61. Faust J, Soman TB. Psychogenic movement disorders in children: Characteristics and predictors of outcome. J Child Neurol (online):1-5. 2011.
62. Helms A, Shulman L. Chapter 12: Movement disorders that imitate epilepsy. In: Kaplan PW, Fisher RS. Imitators of Epilepsy. 2nd ed. New York: Demos Medical Publishing. 2005:163-184.
63. Subramony SH. Chapter 22: Ataxic disorders. In: Daroff: Bradley's Neurology in Clinical Practice. Philadelphia: Elsevier Saunders. 2012.
64. Scahill L, et al. Contemporary assessment and pharmacotherapy of Tourette syndrome. NeuroRx 2006;3:192-206.
65. Talan DA, et al. Role of empiric parenteral antibiotics prior to lumbar puncture in suspected bacterial meningitis: State of the art. Rev Infect Dis 1988;10;2:
365-376.
66. Kuppermann N, et al. Identification of children at very low risk of clinically-important brain injuries after head trauma: A prospective cohort study. Lancet 2009;374:1160-1170.
Abnormal movements, outside of seizure and ataxia, are an uncommon chief complaint among children presenting to the emergency department. A working knowledge of movement disorder phenomenology, etiology, differential diagnosis, and associated potentially life-threatening conditions is essential for emergency physicians. Each movement disorder and its clinical considerations will be discussed in detail in the article.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
