Evaluation of the Child with a Limp
Authors: David S. Brancati, DO, Department of Emergency Medicine, St. Vincent Mercy Medical Center and Mercy Children’s Hospital, Toledo, OH; John Jewell, MD, Department of Emergency Medicine, St. Vincent Mercy Medical Center and Mercy Children’s Hospital, Toledo, OH; and Michael Omori, MD, Mercy Children’s Hospital Department of Pediatric Emergency Medicine; Faculty, St. Vincent Mercy Medical Center Emergency Medicine Residency Program.
Peer Reviewer: Andrew D. Perron, MD, FACEP, Residency Program Director, Department of Emergency Medicine, Maine Medical Center, Portland.
The evaluation and diagnosis of the child with a limp can be challenging for the emergency physician. Causes can range from life-threatening infections to problems as simple as ill-fitting footwear. Most children who limp, however, do not require urgent interventions and can be followed clinically once life- or limb-threatening causes have been ruled out.1,2 Often, a careful history and physical are all that are needed to narrow the differential; at other times, complex and invasive tests are needed to support the diagnosis. The authors review common causes for the acutely limping child, with special attention to those etiologies that need emergent or urgent intervention and referral. Additionally, the authors offer clinical and historical clues to help decipher the cause.—The Editor
Introduction
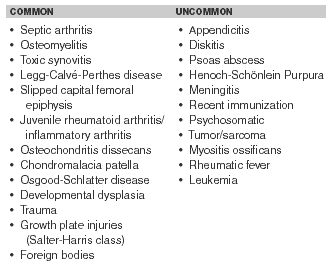
Evaluation of the limping child may be challenging to emergency physicians. Numerous barriers to establishing a definitive diagnosis may exist. History may be limited or unavailable, particularly in the younger age groups, and physical examination may be difficult due to age and apprehension. In addition to difficulties in obtaining an accurate history and localizing the source of limp in an uncooperative child, the lack of availability of definitive diagnostic studies can further complicate the evaluation.1-4 The differential diagnosis of the limping child also is quite broad. (See Table 1.) Most children, fortunately, present with non-emergent causes of limp and can be referred and followed as outpatients.1,2 By separating etiologies into traumatic and non-traumatic causes and focusing on common disease patterns, the physician can develop a simplified approach to establishing the correct diagnosis.
|
Physiology
A basic understanding of normal gait, gait patterns, and child development is clinically important for the emergency physician. Adult gait pattern, which usually is seen by 3 years of age, is divided into two major divisions—a swing portion and a stance portion.1 The stance portion further can be subdivided into heel-strike phase, flat foot phase, heel-rise phase, and toe-off phase; these describe the process of shifting weight to various portions of the foot.1 A limp is defined as any alteration of the normal gait cycle.5 Abnormalities in the swing portion of gait often point to difficulty with range of motion, whereas problems with the stance portion more likely indicate difficulty bearing weight. Two common pathologic gait patterns include antalgic and Trendelenburg gaits. An antalgic gait, which is the most common abnormal gait pattern, is caused by unilateral limb pain.1,2,4,6 An antalgic gait results in patients spending a greater portion of the gait cycle on the unaffected side.7 Antalgic gaits commonly are seen in patients with infectious arthritis, fractures, and even foreign bodies. A Trendelenburg gait is painless and a result of muscle weakness or instability.7 It is characterized by a pelvic tilt away from the affected side due to a relative weakness of the hip abductors.1 This pattern commonly is seen with slipped capital femoral epiphysis (SCFE), Legg-Calvé-Perthes Disease (LCPD), or developmental dysplasia of the hips (DDH).1,2
History and Physical
Although essential to determining the cause of a limp, the history and physical exam can be misleading, especially in younger children. Caregivers and patients should be questioned carefully for diagnostic clues, such as developmental accomplishments, fever, time-course, type of gait disturbance, exacerbating and relieving factors, history of trauma, and the presence or absence of pain. A history of fever may indicate the presence of an inflammatory condition or infectious process and usually accompanies septic arthritis, transient synovitis, and osteomyelitis.1,2 A recent growth spurt or history of obesity may increase the index of suspicion for SCFE. A high index of suspicion for trauma, a common cause of limp, always should be considered, regardless of history. During the physical exam, children should be completely undressed and ambulated through several gait cycles to help localize the cause of the limp. The bottom of the feet should be carefully inspected for foreign bodies. All joints should be observed in passive and active range of motion and assessed for the presence of warmth, tenderness, or effusions.
Atraumatic Limp
Introduction and Approach to the Patient. The pediatric patient who presents with a gait disturbance, but without a history of trauma, may represent a daunting challenge to even the most astute clinician. The extensive differential of non-traumatic pathologic processes that may present with pain in the lower extremities are numerous and can range from life-threatening problems, such as leukemia or osteogenic sarcoma, to limb-threatening causes, such as inflammatory or septic arthritis. Additionally, children with significant disease of the lower spine, such as diskitis, initially may present with a limp, as may patients with intra-abdominal or retroperitoneal pathology, such as appendicitis or iliopsoas abscess.
Fortunately, the most common causes of atraumatic lower extremity pathology often may be differentiated based on characteristic presentations and historical factors, such as age, gender, body habitus, polyarticular or monoarticular involvement, acuity of onset, and presence or absence of fever. In patients in whom the diagnosis is uncertain after examination, or when confirmation of clinical suspicion is needed, pertinent lab, radiographic, or ultrasonographic studies may be useful.
Inflammatory Disease. Transient Synovitis. Transient synovitis, also referred to as toxic synovitis, is the most common cause of hip pain and gait disturbance in children 3-10 years of age.8 It can occur in any age group, is more common in males, and, in the majority of cases, there is a history of an antecedent viral illness in the 1-2 week period prior to presentation.9 The exact etiology is unclear, although the male predisposition has led some authors to suggest that many cases are actually secondary to unreported trauma.10
Patients with transient synovitis usually present with relatively acute onset of pain in the hip or groin, or referred to the anterior thigh or knee. Not uncommonly, patients are more symptomatic in the morning, with some improvement as the day progresses.11 They typically demonstrate an antalgic gait, but are capable of weight-bearing. The affected extremity usually is not held in an abnormal position,9 although patients may demonstrate some modest limitation of range of motion, especially in abduction and internal rotation. Erythema, warmth, and joint swelling generally are not present. Large joints, especially the hip, commonly are affected.
Transient synovitis must be differentiated from more serious entities such as osteomyelitis, LCPD, and, especially, septic arthritis, which can pose the greatest challenge to the physician. As such, transient synovitis often is a diagnosis of exclusion. Transient synovitis, septic arthritis, and LCPD present in similar age groups, with complaints of hip pain and limp. Similarly, patients with transient synovitis may have an accompanying fever, but typically it is low-grade (less than 38° C), and these patients usually appear well.
Laboratory values in patients with transient synovitis usually are normal, although one occasionally may find a mild elevation of the erythrocyte sedimentation rate (ESR).12 Radiographs, which are helpful to exclude other conditions such as LCPD and osteomyelitis, should include anteroposterior (AP) and frog-leg views of the hips, and usually are unremarkable. Ultrasound may demonstrate a small effusion. Arthrocentesis usually demonstrates a normal glucose and WBC count between 20,000 and 50,000. Radionuclide scanning or magnetic resonance imaging (MRI) evaluation also may be undertaken to rule out infection or LCPD, and again, these typically are normal.
The disease is self-limited, and its anticipated course is 1-2 weeks in duration.8 Treatment is expectant and consists of bed rest and non-weight-bearing while significant pain persists, with gradual resumption of normal activities as symptoms subside. Non-steroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen (10 mg/kg orally every eight hours), may be used, or acetaminophen (10 mg/kg) may be given for symptomatic relief. Full recovery should occur in 2-3 weeks, and persistent or worsening symptoms should prompt a search for another cause of pain.
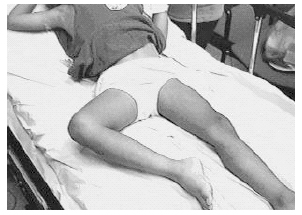
Septic Arthritis. Septic arthritis, also known as acute suppurative arthritis, is the inflammation of a joint space secondary to a bacterial infection. The most common organism isolated is Staphylcoccus aureus. Septic arthritis occurs more frequently in younger children, with a mean age of 3-6 years.12-16 Presenting symptoms in older children include an antalgic gait, a refusal to bear weight on the affected extremity, a tender and swollen joint and surrounding erythema. Neonates and infants may present with symptoms ranging from localized joint tenderness and muscle spasm to more general complains of irritability, decreased feedings, and dislike of being held.14 (See Figure 1.)
|
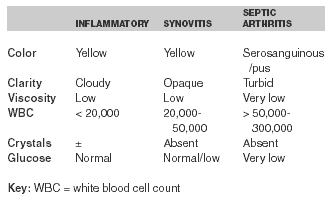
More than 90% of cases of septic arthritis affect the joints of the lower extremity, with the knee the most commonly involved joint.2 The high morbidity, including destruction of joint cartilage and bone with subsequent chronic arthritis in cases of missed or delayed treatment, make the emergency department (ED) physician’s role critical in correctly identifying septic arthritis and immediately beginning antibiotic therapy.14 The gold standard for diagnosis is identification of the organism on Gram stain and culture obtained by arthrocentesis. The diagnosis is highly suggested when aspirated synovial fluid shows pus, white blood cell (WBC) count greater than 50,000, elevated protein, and decreased glucose levels. (See Table 2.)
|
Kocher identified four independent clinical predictors of septic arthritis, based on multivariate analysis, including: 1) history of an oral temperature greater than 38.5º C; 2) non-weight bearing; 3) ESR greater than 40 mm/hr; and 4) serum WBC greater than 12,000 cells/mm3.15 Early septic arthritis, however, may show a paucity of symptoms and signs without overtly positive Gram stains. Additionally, inflammatory arthritis can show elevated WBC counts and inflammatory markers, thus mimicking septic arthritis. Del Beccaro et al demonstrated that an ESR greater than 20 and a temperature greater than 37.5º C, in the correct clinical scenario, correctly will identify 97% of patients with septic arthritis. Unfortunately, using this criterion, 47% of patients with an eventual diagnosis of toxic-synovitis would have undergone an unnecessary arthrocentesis to rule out septic arthritis.9 Regardless of presentation, the emergency physician must maintain a high index of suspicion for septic arthritis, as missing the diagnosis can lead to joint destruction and chronic morbidity.
Once the diagnosis of septic arthritis is entertained, timely administration of parenternal antibiotics is mandatory. Empiric intravenous broad-spectrum antibiotics, such as a third-generation cephalosporin and a penicillinase-resistant penicillin, is the usually recommended antibiotic regimen. In areas with a high prevalence of methicillin-resistant S. aureus (MRSA), vancomycin should be added to the treatment regimen. Once the pathogen is identified with susceptibilities, the spectrum can be narrowed. Ultimately, infected joints may need multiple aspirations and/or open surgical drainage with irrigations to reduce the pressure in the joint space. If left untreated, septic arthritis leads to joint destruction and adhesions, leaving the patient permanently disabled.
Juvenile Rheumatoid Arthritis. Juvenile rheumatoid arthritis (JRA) refers to a heterogeneous group of chronic inflammatory diseases involving the joints. By definition, the arthritis must present before the age of 16 years and involve at least one joint for more than six weeks.17 The prevalence of JRA in the United States is estimated at approximately 86 per 100,000 children.18
The disease is classified into sub-classes, based on the disease onset, as follows: 1) systemic; 2) pauciarticular (1-4 joints involved); and 3) polyarticular (five or more joints). Systemic-onset JRA provides the greatest challenge to the ED physician, because the diagnosis requires the exclusion of other infectious causes of joint pain. Children with systemic JRA classically have two fever spikes during the day that resolve spontaneously within 1-2 hours.17 This pattern of fevers, along with other systemic symptoms of generalized rash and serositis, can aid in the differentiation of system JRA from septic arthritis and osteomyelitis.19
Laboratory tests provide little assistance in diagnosing JRA. Rheumatoid factor (RF) and anti-nuclear antibody (ANA) routinely are ordered when JRA is suspected. RF has less than a 5% sensitivity for JRA,20 and 40% of children tested were positive for ANA,21 making both poor screening tests. X-rays should be obtained to look for joint destruction and to rule out other etiologies of joint pain.
In the ED, the diagnosis of JRA rarely can be made definitively. The diagnosis should be considered only after exclusion of infectious causes of joint pain. First-line therapy for JRA consists of NSAIDs, and can be initiated in the ED.20,22,23 The use of oral steroids and other second-line therapies, such as intra-articular steroid injections or methotrexate, typically are administered only in consultation with a specialist because of potentially serious side effects. A patient diagnosed with JRA requires referral to a rheumatologist for long-term management.
Osteomyelitis. Osteomyelitis is an infection of the bone or bone marrow. The disease is classified by the length of time the infection has been present. Acute osteomyelitis is an infection of fewer than two weeks’ duration, and is the most common form in children. Sub-acute (2-6 weeks’ duration) and chronic (more than 6 weeks) more frequently affect the adult population.
Bones become infected by one of three mechanisms: 1) hematogenous spread; 2) infection from a contiguous source; or 3) direct inoculation from wounds, trauma, or surgery.24 Hematogenous seeding is the leading cause of acute osteomyelitis, and affects children more frequently due to the blood flow in growing bones. After approximately 1 year of age, diaphyseal blood vessels no longer cross the growth plate, and instead end in slow-moving pools of blood in the metaphysis.25 This area of relative stasis provides an ideal location for the implantation of hematogenous infections in long bones.
The incidence of childhood osteomyelitis is on the decline in developed countries, in large part because of the Haemophilus influenzae vaccine. Despite the decline, 1 in 1000 neonates and 1 in 5000 children younger than 13 years in the United States are afflicted with this disease each year. S. aureus is the most commonly identified pathogen, responsible for nearly half the single organism infections.25 Group B Streptococcus, S. pneumoniae, and gram-negative bacilli are other common pathogens. Several unique clinical scenarios are classically associated with otherwise uncommon organisms such as Salmonella infections in sickle cell patients, Pseudomonas aeruginosa infections in penetrating trauma, and Neisseria gonorrhoeae in newborns and sexually active teenagers.26
Diagnosing acute osteomyelitis in the ED can be challenging, especially differentiating between joint (septic arthritis) and bone involvement. Though often thought of as separate disease entities, recent studies show concurrent infections in 34-47.5% of cases.25,27 Patients with osteomyelitis often present with localized tenderness, overlying soft-tissue erythema, and warmth, as well as systemic symptoms such as fevers, irritability, and poor feeding (infants). Gait disturbances nearly always are present when the lower extremity (i.e., femur, tibia, and fibula) is affected.28
Laboratory studies have been shown to be of little assistance in making the diagnosis of osteomyelitis. The WBC count has been proven to not be a sensitive indicator of osteomyelitis, while ESR and C-reactive protein (CRP) are more sensitive, but not specific.24,29 Blood cultures and direct needle aspirates identify the bacterial pathogen in 50-75% of cases, respectively.29
The first step in imaging the affected area typically should be a plain radiograph. The classic signs of bone destruction and pereosteitis often are not evident until 10-14 days after disease onset, though early signs of soft-tissue swelling may be present. Radiographs also are useful to help exclude other diagnoses, including fractures and bone masses or tumors.30,31 Ultrasound examination may be useful if thickening and elevation of the periosteum are identified. Fluid collections may be identified and localized, and this may aid diagnostic and therapeutic aspirations.30
Bone scintigraphy (bone scan) identifies areas of inflammation, and is 82% sensitive in diagnosing osteomyelitis.26 Bone scans lack the specificity and ability to precisely delineate the extent of bone involvement. Although computed tomography (CT) and MRI scans are more specific, both are considered secondary tests.30,31
Once osteomyelitis is suspected, intravenous empiric antibiotics are required. Antibiotics should not be delayed for culture recovery, and should be administered parenterally. Empiric antibiotics should cover S. aureus, as well as Group A and B Streptococcus, gram-negative bacilli, and H. influenzae. This can be accomplished using a penicillinase-resistant penicillin and a third-generation cephalosporin.24 In regions of the country with high rates of MRSA, vancomycin should replace the penicillin. In sickle cell patients and patients with puncture wounds, where Salmonella and Pseudomonas are suspected, respectively, a quinolone must be added to the above regimen.22 All patients with osteomyelitis require admission to the hospital.
Disorders of the Hip. Legg-Calvé-Perthes Disease. LCPD is an idiopathic avascular necrosis of the capital femoral epiphysis (CFE), the ossification center of the femoral head. LCPD was first described in 1909 by Arthur Legg, Jacques Calvé, and George Perthes, who were working in pediatric tuberculosis hospitals in Boston, France, and Germany, respectively.32 The budding technology of roentgenography allowed them to identify patients who had been misdiagnosed with tuberculous arthritis, but who, in fact, suffered what Calvé termed "a pseudo-coxalgia."33
LCPD is seen in children ages 2-12 years, with the vast majority being 5-9 years of age. There is a 4:1 to 5:1 male predominance, and many of the patients are of small stature, with relative skeletal immaturity. There also is a reported association of LCPD with patients who demonstrate deficiencies in protein S and protein C and those with thrombophilia and hypofibrinolysis.9 The vascular insult may be secondary to arterial or venous thrombosis, with the latter being more common. Approximately 10-20% of patients will develop bilateral disease.
LCPD patients generally present with a history of several days of a painless limp, usually of the Trendelenburg type. Physical exam may demonstrate mild restriction of range of motion, mostly in abduction and internal rotation. Atrophy of the proximal thigh muscles may be noted in long-standing cases. Patients are non-toxic, and there usually are no symptoms of systemic disease, such as fever or rash.
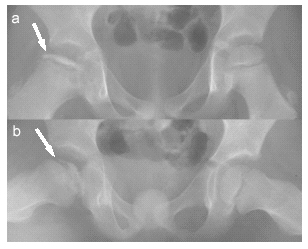
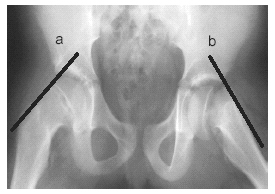
The diagnosis can be made or suggested radiographically. In patients with established disease, AP and frog-leg films of the hips and pelvis will demonstrate characteristic findings of compression deformity and increased density of the capital femoral epiphysis. (See Figure 2 a-b.) Early cases may require MRI or radionuclide scanning for detection, although there is no demonstrable treatment benefit to early diagnosis. There typically is no effusion, and arthrocentesis is not indicated, except in cases where septic arthritis is suspected.
|
The age of onset carries significant prognostic implications. Younger patients (usually younger than 6 years of age) are much less likely to sustain long-term sequelae. It is postulated that the higher proportion of cartilage in the less mature epiphysis retains a greater capacity for remodeling than the more ossified epiphysis of the older patient.32 Between the ages of 6 and 9 years, patients have a 35-40% risk of significant complications. Patients older than 10 years at onset, with a residual deformity of the femoral head, have nearly 100% incidence of degenerative osteoarthritis in later life.9 Radiologic findings that are prognostic are based on the degree of involvement of the lateral pillar (lateral one-third of the CFE). Patients with little or no involvement of the lateral pillar typically do well without treatment. Patients with lateral pillar height loss up to 50% have a less benign prognosis, and patients who demonstrate a loss of height greater than 50% generally do poorly, with higher incidences of degenerative arthritis.32
Accordingly, orthopedic treatment decisions are based on factors including patient age at the time of onset, radiologic measures of the severity of disease, and the symptomatic expression of the deformity.34 Patients younger than 5-6 years, or those with minimal lateral pillar collapse, generally are treated conservatively, with only expectant observation. In this group, patients with mild to moderate symptoms may benefit from periods of bed rest and/or abduction stretching therapy in an attempt to ameliorate discomfort and maintain joint mobility. In older patients or those with more advanced disease, the use of casts or orthotics may be indicated, and surgical containment procedures may be of benefit in cases of severe disease. Expeditious orthopedic referral and symptomatic treatment are indicated.
Slipped Capital Femoral Epiphysis. SCFE is the most common non-traumatic cause of pathology and pain in the adolescent hip. In the United States, the incidence of SCFE varies between two and 10 cases per 100,000 population.35 It more commonly is seen in males, with gender predominance between 2:1 and 3:1. The age of onset usually is 9-16 years. Most patients with SCFE are overweight, with greater than half of the patients measuring above the 95th percentile for weight. Many of these patients also demonstrate delayed skeletal maturation. Conversely, a second group of patients at risk are tall, thin adolescents who have recently undergone a growth spurt. Estimates of the incidence of bilateral disease range from 21-80%.36 Most patients develop contralateral symptoms within 1.5 years of their initial diagnosis.36 Close monitoring of patients with unilateral disease, therefore, is warranted to ensure early detection of bilateral disease.
The exact etiology of SCFE is unclear, but it is postulated to be the result of excessive loading of the femoral head, either acutely or chronically, resulting in shear forces beyond the load range of the physeal cartilage. The subsequent abnormality is a displacement of the CFE posteriorly and inferiorly on the femoral neck metaphysis. SCFE has also been observed as a complication of endocrinopathies, such as hypothyroidism, pituitary disease, pseudohypoparathyroidism, and others.37
Patients with SCFE may present with complaints of either acute onset or chronic symptoms of pain in the hip, groin, knee, or anterior thigh. Some patients may give a history of an inciting event, such as a jump or a fall, while others note an insidious and more gradual onset, with or without an abrupt worsening of their symptoms. The onset history and radiographic findings determine the classification of patients into four groups.9 The first group are patients with preslip. These patients have symptoms and historical and physical findings consistent with SCFE, and a widening of the physis without evidence of displacement. These often are found in the contralateral hips of known SCFE patients who are being monitored for bilateral involvement, but these also can be seen in new patients with early disease. The second group has an acute SCFE and is diagnosed in cases with fewer than three weeks of symptoms and no antecedent complaints. Pain and limp usually begin abruptly, without a history of trauma. Symptoms usually are severe enough to render the patient incapable of weight bearing. The third group of patients have an acute-on-chronic slip and experience mild symptoms, usually of several months’ duration, with a sudden exacerbation or progression of discomfort as a result of a new slip superimposed on a chronic one. These patients similarly are incapacitated and often are unable to bear weight on the affected extremity. The fourth and most common group of patients with SCFE presents with a chronic SCFE. These patients usually are symptomatic for months, and displacement of the CFE has occurred gradually enough to maintain continuity with the femoral neck. The resulting symptoms are mild, and the patient usually adapts with a slightly antalgic gait and some degree of external rotation.
Radiographic evaluation should include AP pelvis, lateral hip, and frog-leg views. Findings on x-ray will reflect the category of disease. Preslip patients will demonstrate some physeal widening without CFE displacement. Acutely slipped hips will demonstrate displacement without significant remodeling of the CFE. Acute-on-chronic slips will reveal displacement as well as evidence of remodeling that, presumably, predated the slip. Chronic slips will demonstrate displacement, and callus formation may be visible, usually at the inferomedial aspect of the physis.
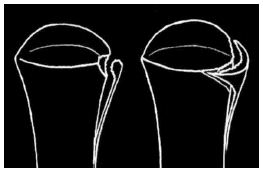
SCFE also may be classified on the basis of the degree of displacement. Mild cases slip less than one-third the diameter of the femoral neck, moderate cases are one-third to one-half the neck diameter, and severe cases exceed one-half the diameter. (See Figure 3.) Ultrasound, radionuclide bone scans, CT, and MRI may be helpful, but in general are not superior to plain films in establishing the diagnosis. Lab tests typically are normal and not necessary.
|
Treatment of SCFE is intended to prevent further displacement and to reduce the risk of complications and chronic sequelae. Currently, the most common treatment is closed reduction followed by epiphysiodesis, or surgical closure of the CFE,38 typically through fluoroscopically guided percutaneous placement of fixation screws through the femoral neck into, but not through, the CFE. Cases of more severe chronic slips often require an osteotomy at the physis, the base of the femoral neck, or at the trochanter to compensate for the irreversibly remodeled CFE.
The most common serious complication of SCFE is osteo-necrosis, which can affect all or part of the epiphysis. This is more common in patients who are pinned without reduction of the CFE. The second most common complication is chondrolysis, or degenerative change in the acetabular and femoral articular cartilages,39 which occurs in 3-7% of cases. The surgical treatment of osteo-necrosis may be undertaken early or may be delayed, sometimes for years, until the patient has reached adulthood.
Emergency physicians must maintain a high index of suspicion, a low threshold for obtaining appropriate radiographs, and a vigilant eye when interpreting those films. Patients in whom the diagnosis is made or strongly suspected should be referred emergently to an orthopedist, with appropriate symptomatic treatment initiated in the ED. Delayed or missed diagnosis carries the potential for an increased incidence of osteonecrosis and subsequent long-term sequelae.1
Developmental Dysplasia of the Hip. DDH is an uncommon cause of gait disturbance in the pediatric population, as it usually is detected on neonatal screening examinations. However, it may present in a young child who did not receive a complete evaluation as a newborn or infant.
Epidemiologically, more than half of these patients are first-born infants, and one-third to one-half were breech presentations. There is a 9:1 female predominance, and a 20% incidence of positive family history. The majority of these children will demonstrate generalized ligamentous laxity.40
Patients with previously undiagnosed DDH will present with an abnormal and non-painful (Trendelenburg) gait, usually described as "waddling." Other findings may include exaggerated lumbar lordosis, toe walking, or leg-length discrepancy.9
Physical screening examination may yield a positive Ortolani or Barlow test in newborns or neonates with DDH. The Ortolani test consists of holding the thigh flexed and in abduction with anterior traction applied to the thigh, leading to a palpable reduction of the dislocated hip. The Barlow test consists of axial loading of the thigh in the flexed and adducted position. If the hip dislocates, which readily is appreciated by the examiner, the test is considered positive.41 After 6 months of age, the Ortolani and Barlow tests are not applicable, and one may find only a positive Galleazzi sign. This test is performed with the patient in the supine position and requires the examiner to place the feet at the same level and compare the height of the knees. If the knees are not at the same level, one must infer a leg-length discrepancy, typically a shortening of the lower of the two sides. Note that this test will yield a false-negative result in the patient with bilateral disease.
Radiographic findings include abnormal alignment of the femoral head with respect to the acetabulum and pelvis. Treatment is variable, based on age at presentation and on whether the hip is dislocated, subluxed, or merely unstable (dislocatable). In cases of delayed diagnosis and progressive deformity, surgery often is necessary. Appropriate management in the ED includes timely orthopedic evaluation.
Disorders of the Knee. Patients who present with complaints of knee pain in the absence of trauma are common. The knee is a complex, weight-bearing joint given to a variety of developmental and structural problems that may disturb the gait. Fortunately, the most common of these problems usually occur in adolescents or teenagers who are able to localize their symptoms to the knee.
Osgood-Schlatter Disease. This is one of the most frequent causes of non-traumatic knee pain in children 10-15 years of age. It is slightly more common in males than females, particularly those who are athletic or very physically active, and often is bilateral.42 This condition is the result of repetitive traction of the quadriceps muscle on the patellar tendon at its insertion on the immature apophysis of the anterior tibial tubercle. Onset typically is gradual, although there may be a history of minor trauma to the area.
On physical examination, there is point tenderness and often swelling over the anterior tibial tubercle. Patients usually will have increased pain with extension of the knee against resistance or with passive flexion of the knee. Effusion, erythema, or warmth are uncommon, and their presence should prompt consideration of an alternate diagnosis.
Radiographs frequently demonstrate local soft-tissue swelling and a "ratty" appearance of the ossifying apophysis. The condition is self-limiting, and the patient and parents should be reassured that with rest and symptomatic treatment such as ice, NSAIDs, and acetaminophen, normal activities may be resumed when tolerated. This may be quickly, if improvement with NSAID therapy is sufficient, or may take up to 24 months following complete closure of the apophysis.9
Osteochondritis Dissecans. Osteochondritis dissecans may occur in other joints, but most commonly is found in the knee, usually involving the lateral surface of the medial femoral condyle. It results from an avascular insult to a small portion of the subchondral bone in this region, possibly due to repeated trauma from the tibial spine. This condition usually is seen in adolescents, particularly among athletic or very physically active patients. The history may be one of gradual and insidious onset, or, in 40-60% of cases, there may be a history of recent or antecedent trauma to the knee.43
Findings on physical examination may vary. With the knee in flexion, one may be able to directly palpate the involved area of the femoral condyle, eliciting point tenderness.9 Wilson’s sign may be positive and is specific for osteochondritic lesions of the medial femoral condyle. The sign is elicited when the knee is flexed to 90° and then slowly extended while the tibia is held in internal rotation. The sign is positive if the patient develops pain at approximately 30° of flexion, the point where the tibial spine contacts the medial femoral condyle.44 A small effusion may be present; however, one should not find signs of an acute inflammatory process such as erythema or warmth. The ligamentous structures of the knee are stable on examination. If actual avulsion of the avascular fragment has occurred, the clinician may elicit a history of locking of the knee or find evidence of it on examination.
Radiographs may be positive only on the tunnel view of the knee. A loose intra-articular foreign body may be present if actual avulsion of a fragment has occurred. MRI examination may further orthopedic assessment of the stability of the lesion and allow staging of the disease, but is not necessary for initial diagnosis.43
Treatment usually consists of NSAIDs for symptomatic relief, as well as rest and avoidance of activities that worsen symptoms. Arthroscopic intervention may be necessary in cases of intra-articular loose bodies.
Chondromalacia Patella (Patellofemoral Syndrome). The exact cause of chondromalacia patella is unknown, although abnormal tracking of the patella over the femoral condyles is a contributory factor.45 Patients may present with this condition at any age, but it is more common among adolescents and teenagers. Symptoms usually consist of dull, aching pain localized to the knee, and patients often will complain of discomfort behind the patella. The onset of symptoms may be gradual, or the complaints may arise after some direct trauma. Pain often is worse during activities such as climbing steps or riding a bicycle, or after prolonged periods of sitting with the knee in a flexed position, the so-called "movie sign."46
On examination, one may find tenderness of the medial posterior patella, and with application of posteriorly directed pressure applied to the patella with the knee in slight flexion. There also may be some crepitus palpable with flexion and extension of the knee.47 Radiographs may demonstrate some blurring of the periosteum of the posterior patellar surface. This best may be visualized on the sunrise view of the knee.
Treatment generally is expectant, with rest, NSAIDs, and quadriceps-strengthening physical therapy.
Other Significant, Non-traumatic Causes of Gait Disturbance. As listed in Table 1, a wide variety of conditions not directly involving the lower extremity may present with a limp as the initial symptom. These include such significant problems as intervertebral diskitis,2 appendicitis, pelvic inflammatory disease, leukemia, and both malignant and benign bony tumors.
A discussion of these diagnoses is beyond the scope of this article; however, one always must be cognizant of the possibility that a child with a limp and no history of trauma warrants a careful and complete investigation before symptoms are ascribed to a benign process.
Traumatic Limp
Introduction and Approach to the Patient. A second broad category of acute limp results from traumatic injuries. Regardless of history, trauma always should be considered, particularly with younger children. The inability of the child to describe symptoms and localize pain makes pediatric trauma care a special challenge that requires experience and great patience on the part of the examining physician.48 Furthermore, injury patterns that are unique to pediatrics pose diagnostic dilemmas. The areas of immature pediatric skeletons composed of growth plates or physis represent the "weak link" in pediatric bone.37 Injuries in adults that often cause sprains with injuries to soft tissue and tendon more often result in injuries to the bone or growth plate in children. While a detailed discussion of pediatric trauma is beyond the scope of this article, a few classic patterns are described.
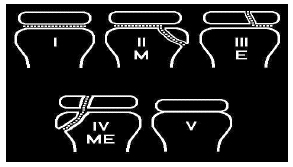
Salter-Harris Fracture Classification. Common in children, injuries of the growth plates or physis pose an increased morbidity and diagnostic challenge for the clinician. The Salter-Harris classification is used in the description and prognosis of injuries involving the growth plates of skeletally immature children. (See Figure 4.)
|
A Salter-Harris Type I fracture is a horizontal fracture through the physis or growth plate. Since no cortical or radio-opaque bone is involved, these injuries often are not visualized on radiographs and most are diagnosed clinically. They account for approximately 5% of physeal injuries.49 Radiographically, a non-displaced Salter-Harris Type I fracture will appear normal or near normal. Comparison views may help to identify subtle displacement, but do not alter treatment. Any child with tenderness at the physis should be splinted, with close follow-up regardless of radiographic findings. Salter-Harris Type I injuries are treated with closed reduction and casting for approximately four weeks.22 Overall, these injuries have an excellent prognosis.37 Salter-Harris Type II fractures involve the physis and metaphysis and do not involve the joint space. These are the most common Salter-Harris injuries seen in children and account for approximately 75% of growth plate injuries.49 Salter-Harris Type II fractures do not involve the joint space and, therefore, overall also have excellent prognosis.22 Salter-Harris Type II fractures are treated with closed reduction and short casting for four weeks.22
Type III injuries are intra-articular fractures involving the physis and epiphysis. These injuries account for approximately 10% of physeal injuries and usually occur in children with a partially closed physis.49 These injuries have a higher incidence of posttraumatic arthritis, but carry a good prognosis with proper anatomic correction.37 Therefore, they often require open reduction and internal fixation to avoid growth disturbances.22
Type IV injuries involve the physis, epiphysis, and metaphysis with an oblique fracture line. The fracture pattern is intra-articular and, therefore, can result in generally a worse prognosis than the previously mentioned three patterns. The relative incidence is approximately 10%.49 These intra-articular injuries often require open reduction and precise anatomic alignment to assure good outcomes.37,50
Salter-Harris Type V injuries are perhaps the most difficult to diagnose. These injuries are the result of crushing force to the physis. Radiographs of the involved growth plate often appear normal, though comparison views can be helpful in determining subtle differences. Although displacement of the epiphysis is rare, these injuries carry the poorest prognosis, with growth disturbances and premature cessation of bone growth.37 Fortunately, they are the rarest of the Salter-Harris injuries and represent approximately 1% of growth plate injuries.49
Knee Injuries. Cartilage and ligaments are far stronger than bone, and injuries that normally would result in knee sprains in adults are more likely to result in fractures or growth plate injuries in children. The results are several injury patterns uncommon in adults but that commonly are seen in children. Sprains of the knee are quite common in adolescents and adults, but are rare in children. Hyperextension injuries of the knee that usually result in anterior cruciate ligament tears in adults often result in tibial spine avulsions in children.51-53 Additionally, violent flexion against a contracted quadriceps muscle that would lead to patellar tendon tears in adults will lead to tibial tubercle avulsions in children.51 Tibial spine and tibial tubercle avulsions can be difficult to diagnose, and negative radiographs do not rule out these injuries. Children with significant pain and clinical effusions should be placed in a long leg splint or knee immobilizer with orthopedic follow up in 1-2 days.
Lower Leg. Toddler’s Fracture. Injuries of the lower leg are common in children, with the distal fibula and tibia being the most commonly injured sites.51,54,55 One fracture pattern, unique to pediatrics, requires mention because of the common confusion with child abuse.
Children ages 1-5 are at risk for a childhood accidental spiral tibial fracture otherwise known as "toddler’s fracture."26,51 Children with these injuries may present without a history of trauma, or may be too young to relay a history of minor trauma. The mechanism of injury is a twisting force, often the result of a young child learning to walk. Symptoms include tenderness, warmth, antalgic gait, or refusal to bear weight. These occult injuries are difficult to diagnose and may be seen only on follow-up radiographs or bone scan. The astute clinician must, therefore, be alert to the diagnosis. Fortunately, the toddler’s fracture is a stable fracture that often is treated conservatively with a long leg cast.26 While long-bone injuries in children of this age are uncommon and should raise the suspicion for abuse, the emergency clinician must be aware of this diagnosis so as not to unnecessarily pursue a child abuse workup.
Ankle Injuries. While ankle sprains are common in the adult and adolescent population, children seldom suffer from sprained ankles. The ankle commonly is injured in children; however, and accounts for approximately 5% of fractures and 15% of all growth plate injuries.51 The physis usually will separate or fracture before disruption or spraining of an adjacent strong or flexible ligament.52 The three lateral ankle ligaments—anterior talofibular, calcaneofibular, and posterior talofibular—are prone to rupture and tear in the adult, but give way to fractures in children. A Salter-Harris Type I fracture of the distal fibula is the most common ankle fracture seen in children. Ankle injuries with swelling and tenderness around the growth plate should be treated conservatively as occult fractures with splinting and close follow-up in 1-2 days. Distal fibula injuries heal well with few complications.56
Foreign Bodies. Often overlooked, the diagnosis of plantar foreign body in the nonverbal child should be considered.
Radiographs may be helpful in the diagnosis of foreign body, but cannot exclude the presence of all foreign bodies. Many types of small glass particles, wood, plant matter, and plastics can be missed on x-ray. Ultrasound may play an increasingly important role in the identification and removal, but is not always readily available.57,58 The astute clinician always should keep a high index of suspicion for foreign body and patients suspected of having retained foreign bodies should be given anticipatory guidance and follow-up in 1-2 days with a plastic, podiatric, or orthopedic surgeon. Tetanus should be updated and prophylactic antibiotics with gram-positive coverage may be prescribed in cases of deep, heavily contaminated wounds or for patients with underlying immunocompromised states. The use of antibiotics in otherwise healthy children with simple superficial foreign bodies remains controversial.
Child Abuse. With any pediatric traumatic injury, child abuse must be considered in the differential. Two-thirds of victims of physical abuse are younger than 3 years, and one-third are younger than 6 months.22 Certain subtle historical clues and injury patterns to the lower extremity should raise suspicion of abuse. For example, inconsistencies between historians or frequently changing histories from a single historian should alert the clinician to a possible abuse case. Likewise, inconsistencies between mechanism and injury, such as a spiral femur fracture in a non-ambulatory child, also should raise suspicion.
When evaluating the limping child, two particular fracture patterns may strongly suggest abuse. (See Figure 5.) The bucket-handle fracture, which is an avulsion fracture of the distal metaphysis of a long bone, is nearly pathognomonic for child abuse. This pattern is produced by extrinsic traumatic forces with a rotational component. Additionally, corner fractures also are highly suggestive of abuse and warrant further workup, including an ophthalmologic exam and skeletal survey. Femur fractures, which require high energy forces, always should raise suspicion of abuse. As many as 70% of children younger than 1 year of age with femur fractures are victims of abuse.51,59 All cases of suspected abuse require consultation and discussion with the primary physician as well as child protective services. Physicians are mandated by law to report all cases of suspected child abuse and neglect; however, reporting methods may vary by state.60
|
Conclusion
The orthopedic complaint of limp is common in the pediatric ED population and may pose many diagnostic dilemmas. Knowledge of normal gait problems and physiology are helpful in determining the cause. While the differential for the acutely limping child remains lengthy, an understanding of common injury and disease patterns and a simplified approach often will assist in determining the diagnosis. In those cases for which a diagnosis is uncertain, life- or limb-threatening causes should be ruled out, and patients should be referred for timely outpatient follow up. Whenever possible, the emergency physician should communicate with the primary or consulting physician to assure close follow up. Patients and families should be given clear discharge instructions, with anticipated course of disease and reasons to return to the ED.
References
1. Kim M, Karpas A. Orthopedic emergencies: The limping child. Clin Pediatr Emerg Med 2002;3:129-137.
2. Lawrence LL. The limping child. Emerg Med Clin North Am 1998;16:911-929.
3. Chung SM. Identifying the cause of acute limp in childhood. Some informal comments and observations. Clin Pediatr 1974;13:769-772.
4. Clark MC. The limping child: Meeting the challenges of an accurate assessment and diagnosis. Ped Emerg Med Reports 1997;2:123-134.
5. Gurr DE. Femur and hip. In: Marx MD, ed. Rosen’s Emergency Medicine Concepts and Clinical Practice. 5th ed. 2002:669-670.
6. Barkin RM, Barkin AZ. The limping child. J Emerg Med 1999;18:331-339.
7. Myers T, Thompson G. Imaging the child with a limp. Pediatric Clin North Am 1997;44:637-658.
8. Shetty AK, Gedalia A. Infectious arthritis. Rheumatic Dis Clin North Am 1998;24:287-304.
9. Thompson GH. The hip. In: Behrman RE, Kliegman RM, Jenson HB, eds. Nelson Textbook of Pediatrics, 16th ed. Philadelphia: W.B. Saunders; 2000:2055-2082.
10. Fink PC. Orthopedic disorders In: Barkin RM, ed. Pediatric Emergency Medicine Concepts and Clinical Practice, 2nd ed. Philadelphia: Mosby Yearbook; 1997:944-952.
11. Hyndman JC. Transient synovitis. In: Staheli LT. Pediatric Orthopedic Secrets. Philadelphia: Hanley and Belfus; 1997:261.
12. Del Beccaro MA, Champoux AN, Bockers T, et al. Septic arthritis versus transient synovitis of the hip: The value of screening laboratory tests. Ann Emerg Med 1992;21:1418-1422.
13. Levine MJ, McGuire KJ, McGowan KL, et al. Assessment of the test characteristics of C-reactive protein for septic arthritis in children. J Pediatr Orthoped 2003;23:373-377.
14. Bennett OM, Namnyak SS. Acute septic arthritis of the hip joint in infancy and childhood. Clin Orthoped 1992; 281:123-132.
15. Kocher MS, Zurakowski D, Kasser, JR. Differentiating between septic arthritis and transient synovitis of the hip in children: An evidence-based clinical prediction algorithm. J Bone Joint Surg 1999;81: 1662-1670.
16. Jung ST, Rowe SM, Moon ES, et al. Significance of laboratory and radiologic findings for differentiating between septic arthritis and transient synovitis of the hip. J Pediatr Orthopaed 2003;23:368-372.
17. Schneider R, Passo MH. Juvenile rheumatiod arthritis. Rheumatic Dis Clin North Am 2002;28:503-530.
18. Peterson LS. Juvenile rheumatoid arthritis in Rochester, Minnesota, 1960-1993. Arthritis Rheumatism 1996;39:1385-1390.
19. Malleson PN, Beauchamp RD, Esdaile JM. Rheumatology: Diagnosing musculoskeletal pain in children. Can Med Assoc J 2001;l65: 183-188.
20. Eichenfield AH, Athreya BH, Doughty RA, et al. Utility of rheumatoid factor in the diagnosis of juvenile rheumatoid arthritis. Pediatrics 1986;78:480-484.
21. McGhee JL, Burks FN, Sheckels JL, et al. Identifying children with chronic arthritis based on chief complaints: Absence of predictive value for musculoskeletal pain as an indicator of rheumatic disease in children. Pediatrics 2002;110:354-359.
22. Tintinalli J, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill; 2002:1823-1832.
23. Leet AI, Skaggs DL. Evaluation of the acutely limping child. Am Fam Physician 2000;61:1011-1018.
24. Perron AD, Brady WJ, Miller MD. Orthopedic pitfalls in the ED: Osteomyelitis. Am J Emerg Med 2003;21:61-67.
25. Ibia EO, Imoisili M, Pikis A. Group A-hemolytic streptococcal osteomyelitis in children. Pediatrics 2003;112:22-26.
26. Mellick LB, Miller L, Egsieker E. Childhood accidental spiral tibial CAST fractures. Pediatr Emerg Care 1999;15:307-309.
27. Caksen H, Ozturk MK, Uzum K, et al. Septic arthritis in childhood. Pediatr Intern 2000;42:534-540.
28. Kim MK, Karpas A. Orthopedic emergencies: The limping child. Clin Pediatr Emerg Med 2002;3:129-137.
29. Gillespie WJ. Epidemiology of bone and joint infections. Infect Dis Clin North Am 1990;4:361-376.
30. Boutin RD, Brossmann J, Sartoris DJ, et al. Musculoskeletal imagining update, part II: Update on imaging of orthopedic infections. Orthoped Clin North Am 1998;29:41-66.
31. Della-Giustina K, Della-Giustina D. Emergency department treatment of orthopedic injuries. Emerg Med Clin North Am 1999; 17:895.
32. Herring JA. Legg-Calvé-Perthes Disease. In: Staheli LT. Pediatric Orthopedic Secrets. Philadelphia: Hanley and Belfus; 1997:269.
33. Calvé J. On a particular form of pseudo-coxalgia associated with a characteritisic deformity at the upper end of the femur. Clin Orthop 1980:150:4-7.
34. Grzegorzewski A. Treatment of the collapsed femoral head by containment in Legg-Calvé-Perthes Disease. J Pediatr Orthop 2000;23: 15-19.
35. Kallio PE, Mah ET, Foster BK, et al. Slipped capital femoral epiphysis: Incidence and clinical asessment of physeal instability. J Bone Joint Surg 1995;77B:752-755.
36. Canale ST. Slipped capital femoral epiphysis. In: Staheli LT. Pediatric Orthopedic Secrets. Hanley and Belfus; 1997: 271.
37. Loder RT, Hensinger RN, Alburger PD, et al. Slipped capital femoral epiphysis associated with endocrine disorders. J Pediatr Orthop 1995;15:349-356.
38. Aronsson DD, Loder RT. Treatment of the unstable (acute) slipped capital femoral epiphysis. Clin Orthop 1996;3:22-99.
39. Lubicky JP. Chondrolysis and avascular necrosis: Complications of slipped capital femoral epiphysis. J Pediatr Orthop 1996;5B:162.
40. Aronsson DD, Goldberg MJ, Kling TF Jr., et al. Developmental dysplasia of the hip. Pediatrics 1994;94:20.
41. MacEwen GD, Herman MJ. Developmental dysplasia of the hip. In: Staheli LT. Pediatric Orthopedic Secrets. Philadelphia: Hanley and Belfus; 1997:273.
42. Krause BL, Williams JP, Catterall A. The natural history of Osgood-Schlatter’s disease. J Pediatr Orthop 1990;10:65.
43. Robertson W, et al. Osteochondritis dissecans of the knee in children, Curr Opin Pediatr 2003;15:38-44.
44. Wilson JN. A diagnostic sign in osteochondritis dissecans of the knee. J Bone Joint Surg Am 1967;49:477-480.
45. Outerbridge RF. The etiology of chondromalacia patellae, 1961. Clin Orthop 2001;389:5-8.
46. Baskin MN. Injury-Knee. In: Fleischer GR. Textbook of Pediatric Emergency Medicine, 3rd ed. Philadelphia: Williams and Wilkins; 1993:280.
47. Ward WT. Orthopedics. In: Davis HW. Atlas of Pediatric Physical Diagnosis, 3rd ed. Philadelphia: Mosby and Wolfe; 1997:672
48. Strange GR, et al. The pediatric emergency medicine course: APLS, 3rd ed. Dallas: American College of Emergency Physicians;2000:59.
49. White N, Sty J. Radiological evaluation and classification of pediatric fractures. Clin Ped Emerg Med 2002;3:94-105.
50. Salter RB, Harris WR. Injuries involving the epiphyseal plate. J Bone Joint Surg 1963;45A:587-621.
51. Overly F, Steele DW. Common pediatric fractures and dislocations. Clin Pediatr Emerg Med 2002;3:106-117.
52. Beaty JH, Kumar A. Fracture about the knee in children. J Bone Joint Surg Am 1994;76:1870-1880.
53. Steele PM, Bush-Joseph C. Management of acute fractures around the knee, ankle, and foot. Clin Fam Pract 2002;4:661-705.
54. Bachman D, Santora S. Orthopedic trauma. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine, 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2000:1435-1478.
55. Dee R, Carrion W, Polisner S, et al. Fractures and dislocations in children. In: Dee R, Hurst LC, Gruber MA, et al, eds. Principles of Orthopaedic Practice New York: McGraw-Hill; 1997:565-614.
56. Beattie TF, Hendry M, McPhillips M, et al. Accident and emergency radiology. Comparison view radiographs are not routinely required. BMJ 1995;310:1605.
57. Hill, R. Ultrasound for the detection of foreign bodies in human tissue. Ann Emerg Med 1997;29:353-356.
58. Blankstein, A. Ultrasonography as a diagnostic modality and therapeutic adjuvant in the management of soft tissue foreign bodies in the lower extremities. Isr Med Assoc J 2001;3:411-413.
59. Gross RH, Stranger M. Causitive factors responsible for femoral fractures in infants and children. J Pediatr Orthop 1983:3:341-343.
60. Christian C. Assessment and evaluation of the physically abused child. Clin Fam Pract 2003;5:1-12.
The evaluation and diagnosis of the child with a limp can be challenging for the emergency physician. The authors review common causes for the acutely limping child, with special attention to those etiologies that need emergent or urgent intervention and referral. Additionally, the authors offer clinical and historical clues to help decipher the cause.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.