Advances in Pediatric Ultrasound, Part 2
Advances in Pediatric Ultrasound
Part 2: Focused Applications for Cardiac, Abdominal, and Renal Complaints
Authors: Anthony J. Weekes, MD, RDMS, Associate Medical Director, Emergency Ultrasound Director, Montefiore Medical Center, Bronx, NY; and Resa E. Lewiss, MD, Director, Division of Emergency Ultrasound, St. Luke’s/Roosevelt Hospital Center, Columbia University College of Physicians and Surgeons, New York, NY.
Peer Reviewer: Michael Blaivas, MD, RDMS, Associate Professor of Emergency Medicine, Chief, Section of Emergency Ultrasound, Department of Emergency Medicine, Medical College of Georgia, Augusta.
Ultrasound (US) continues to evolve as a valuable adjunct to the clinical care of patients in the emergency department (ED). US has emerged as a safe, portable modality for the diagnosis or exclusion of many significant disease processes in children. The ability to avoid unnecessary exposure to ionizing radiation has become a priority, and the emergency physician (EP) plays a critical role in determining the ideal test for each patient. Understanding the strengths, limitations, and clinical implications of each focused US examination can help the EP to decide the best and most specific test for each patient. The first part of this two-part series addressed the use of US for complications of early pregnancy in adolescent females and for testicular complaints in pediatric and adolescent males. This issue focuses on the use, role, and limitations of US in the evaluation of cardiac, abdominal, and renal disease processes. —The Editor
Pediatric Cardiac Sonography
Pediatric echocardiography is a rapidly developing field. Fetal echocardiography, especially the transesophageal approach, can identify congenital cardiac abnormalities with improved resolution and accuracy. Three-dimensional (3-D) echocardiography adds a powerful tool to the pediatric cardiology and cardiothoracic specialties. Transthoracic echocardiography remains the most readily available, non-invasive, and least anxiety-provoking cardiac imaging modality that can be brought to the bedside of the pediatric patient. The use of bedside cardiac sonography in the ED by EPs is well documented in the adult population. In contrast, research regarding the use of cardiac US in the pediatric emergency medicine literature is very sparse. The purpose of pediatric cardiac sonography in the ED is not identification and specification of complex, congenital heart defects, but instead the identification of cardiac pathology that immediately affects clinical care. Potential clinical applications include: the evaluation of the presence or absence of a pericardial effusion, cardiac tamponade, or global wall motion /function, including the differentiation of subcategories of hypotensive states. Although adult cardiac emergencies are a routine part of most EPs’ practice, pediatric cardiac emergencies are uncommon.1 The focus of cardiac ultrasound in the ED includes: 1) identification of pericardial effusions and cardiac tamponade, and 2) assessing global cardiac wall function. In addition, the ultrasound screening evaluation of the older child/adolescent, especially the athlete, who presents with symptoms that may be sentinel events for sudden cardiac death is included.2
A pediatric transducer equipped with a small footprint will enable transmission between ribs. A frequency of 5.0 MHZ is suitable for the child or infant and 3.5 MHz for the average-sized adolescent. The main cardiac views are the parasternal and subxiphoid (subcostal) approaches in a supine infant or child.
The Parasternal View. The parasternal view can be a long axis view (PSLA) or a short axis view (PSSA).
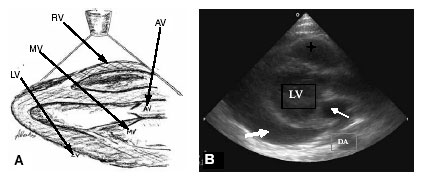
The PSLA view is obtained by placing the transducer near to, but not on, the left sternal edge. The marker should be pointing toward the patient’s right shoulder. Make slight rotations of the transducer until an elongated image appears of the heart. (See Figure 1, below.) The US image depicts the apex toward the left of the screen. The fibrous pericardium is echogenic (white) and surrounds the heart. Blood in the chambers is echolucent (black). The myocardium is of medium echogenicity. Structures closest to the transducer surface appear at the top of the monitor. During this part of the evaluation, observe the movement of the walls —they thicken during systole— toward the center of the chamber. Assess the global wall motion. Next, assess ventricular cavity size and change during systole as global assessment of cardiac ejection. On the PSLA view, the descending aorta is seen in cross section. The descending aorta is a very important landmark. It appears as an echolucent circle, posterior to the heart at the atrioventricular sulcus and the mitral valve.
|
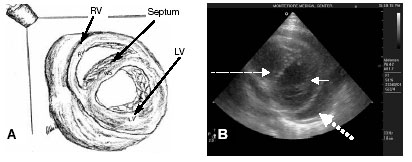
Convert from the PSLA to the PSSA view by slowly rotating the probe marker clockwise so that it points in the direction of the patient’s left shoulder. (See Figure 2, below.) Maintain full contact of the probe with the patient. PSSA views have multiple image options created by slight angulations of the transducer. Angling the transmitted US beam slowly between the patient’s right shoulder and the left hip provides cross-sectional cuts. The left ventricle normally has thicker walls and is draped anteriorly by the right ventricle. The right ventricle is thin-walled and has a semi-lunar appearance. The mitral valve traverses the left ventricular cavity. Its movement resembles that of a fish’s mouth. The aortic valve is seen in PSSA cuts. The aortic valve is centrally located in this view between the anterior right ventricular outflow tract and the posterior left-sided chambers.
|
The Subcostal View. Place the transducer at the subxiphoid area. Apply gentle pressure to the abdomen, then direct the beam just under the left ribcage and to the child’s left shoulder. Rotate the probe to get a four-chamber view of the heart. The apex is seen clearly in this view.
A modified subcostal view provides information on the longitudinal axis of the proximal inferior vena cava (IVC) as it enters the heart. The beam is directed to the patient’s back with the marker to the head. Slight angling of the transducer to the right of the midline reveals the IVC. In a normovolemic patient with normal central venous pressures, the IVC can be observed to collapse to approximately half of its width with a patient’s deliberate sniff or with inspiration (a decrease in intrathoracic pressure improves venous return). Distended hepatic veins and a distended IVC support the concern of an increased central venous pressure.
Neonatal Cardiac Disease (Shock). Pediatric cardiac sonography may be very valuable in the first 30 days of life. Early recognition of significant cardiac lesions and differentiation of cardiac lesions from sepsis or pulmonary disease, allows the EP to accurately direct the medical care of these critical infants and expedite referral to the appropriate sub-specialist.
In the first few days of life, a patent ductus arteriosus (PDA) may maintain pulmonary and systemic vascular perfusion, despite the presence of obstructive left-sided cardiac lesions such as critical aortic stenosis and preductal coarctation of the aorta. Upon closure of the ductus arteriosus, the neonate will have signs of systemic hypoperfusion shock. The left atrium is under increased pressure and dilates. The foramen ovale directs left atrial blood flow to right-sided chambers, thus increasing the pulmonary blood flow and congestion.
The hypoplastic left heart syndrome also may present with congestive heart failure or signs of hypoperfusion. In these cases, echocardiography detects hypoplastic left ventricular walls and hypertrophied right-sided chambers.
Initial medical management is focused on reestablishing and maintaining the patency of the ductus arteriosus. This is accomplished in all cases with Prostaglandin E1 infusions. Additional required therapy may include: ventilator support and management, inotropic agents, and systemic vascular resistance reduction agents. Further definitive interventions may include surgical valvular repair and cardiac catheterization to the valve or area of coarctation.
Neonatal Cardiac Disease (Cyanosis). Newborns also may present with cyanosis and tachypnea, and differentiating between pulmonary and cardiac causes of hypoxia is extremely challenging. Pulmonary congestion on chest auscultation and cyanosis may exist in both scenarios. Accurate classification and delineation of clinically significant cardiac murmurs are difficult in a distressed tachypneic infant.
Right-sided heart failure signs such as cyanosis and tachypnea in the neonatal period can be secondary to pulmonary valve stenosis with an intact ventricular septum. Right ventricular hypertrophy with pulmonic valvular stenosis and an intact septal wall are the main echocardiographic features. This congenital cardiac condition may be treated successfully with Prostaglandin E1 infusions and balloon pulmonary valvulotomy. Atrial and ventricular defects can present weeks after birth depending upon the severity and location of the lesions.
Hypertrophic Cardiomyopathy. Cardiac sonography also may be a valuable screening tool for athletes who present with symptoms of chest discomfort (e.g., pain or tightness), lightheadedness, syncope, or palpitations. In patients with any of these clinical presentations, EPs should consider the possibility of an arrhythmia or a cardiac structural abnormality. In particular, a previous history of recurrent syncopal events, a family history of hypertrophic cardiomyopathy or sudden death, symptoms exacerbated by dehydration or exertion, or a loud systolic ejection murmur on physical examination should raise suspicion for hypertrophic cardiomyopathy (HCM). HCM occurs in 1 in 500 of the general population and accounts for approximately 40% of the cardiac disease in young athletes.
Left ventricular hypertrophy (LVH) is seen normally in the serious athlete, and septal and posterior wall hypertrophy occurs in male, but not female athletes. In most athletes, a wall thickness greater than 13 mm is a reliable indicator of hypertrophic cardiomyopathy. Unfortunately, idiopathic LVH accounts for 10% of sudden cardiac death cases.2 Patients found to have this condition need immediate referral to a cardiologist for a definitive diagnostic evaluation and determination of activity restrictions. (See Figure 3, below.)
|
Pericardial Effusion. Cardiac sonography may be used to exclude or confirm the diagnosis of pericardial effusion. Clinical scenarios where a pericardial effusion may be suspected include: suspected or known pericarditis, recent cardiac surgery, medical resuscitations of hypotensive patients, respiratory distress, pulseless electrical activity, or in the setting of penetrating thoracic trauma. Patients with malignancies, end stage renal disease, AIDS, and rheumatologic and autoimmune diseases also may build up large volumes of pericardial fluid.
Pericardial fluid accumulation may be clinically silent, or the symptoms may develop slowly and be subtle. The most common presenting symptom is chest discomfort. The enlarging pericardial sac also may affect adjacent structures and result in a variety of symptoms including dysphagia, cough, dyspnea, prolonged hiccups, hoarseness, nausea, and abdominal fullness. Subacute presentations may be as vague as anxiety, dyspnea, fatigue, and altered mental status.
The clinical findings associated with a pericardial effusion are difficult to appreciate even in optimal settings. Muffled heart sounds may be difficult to hear because of rapid, loud breath sounds. Heart sounds also may be transmitted clearly through the thin chest wall of the neonate or small child. Pericardial rubs and pulsus paradoxus are not specific physical examination findings of pericardial effusion. Beck’s triad of hypotension, muffled heart, and distended neck veins is considered a very late and non-specific finding. Distended neck veins may not ever occur in the hypovolemic patient.
Electrocardiogram findings of decreased QRS voltage and electrical alternans are not found commonly in patients with cardiac tamponade.
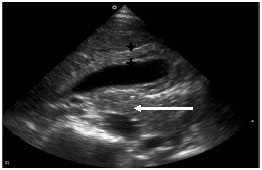
The sonographic appearance of a pericardial effusion is that of an echolucent area surrounding the heart. The pericardial space envelops the heart except at the AV sulcus. As mentioned above, the descending aorta on the PSLA view is an important landmark to the sonographer. It helps distinguish the echolucent pericardial fluid from the pleural fluid. Fluid in the pericardial space does not extend posterior to the descending aorta or left atrium. Any fluid seen posterior to the descending aorta is considered as pleural fluid. (See Figure 4, below.) The subcostal cardiac view offers the advantage of showing only the heart with the liver as the acoustic window. With the subcostal view there is no pleural reflection between the liver and the right-sided heart structures. Any fluid near the heart is pericardial or free abdominal fluid. Pericardial fluid will conform to the outline of the heart.
|
The presence of a pericardial effusion is a necessary, but not a sufficient condition for making the diagnosis of cardiac tamponade. The pericardial space normally holds up to 50 mL in the physically mature healthy adolescent. The pericardial sac can accommodate a gradual increase of pericardial fluid volume without significant alterations in intrapericardial pressure. Neonates can generate rapid heart rates to compensate for low, chamber-filling pressures. They may be able to maintain cardiac output even in the presence of cardiac tamponade. Rapid increases in pericardial volume, as in the setting of penetrating cardiac trauma, rapidly exceed the thinner-walled right atrial and ventricular end-diastolic wall pressures, and in this clinical scenario, small volumes of pericardial fluid can result in cardiac tamponade. Collapse of either right-sided chamber wall at end-diastole is diagnostic of cardiac tamponade.
Emergent pericardiocentesis or pericardial window placement is the definitive treatment.
Abdominal Sonography
Abdominal sonography in the pediatric ED is focused on the identification of three main intestinal abnormalities. Normally, the bowel is not the focus in abdominal sonography and may be a hindrance to other scanning goals such as aortic scanning. During the sonographic evaluation, a normal bowel displays peristaltic activity, and its walls are barely discernible. The internal substances, mixed with gas, can cast shadows that are not demarcated sharply. These are called "dirty shadows," which are in contrast to the "clean" shadows cast by stones and bone. The normal bowel is compressible with gentle transducer pressure on the abdomen.
Appendicitis. Appendicitis is the most common reason for abdominal surgery in children and presents a diagnostic challenge to even the most experienced clinician. Of children presenting to the pediatric ED with abdominal pain, 1-8% are diagnosed as having appendicitis.3 The younger the child, the more challenging the diagnosis, with significantly higher appendiceal rupture rates reported in pediatric patients younger than 3 years when compared with the adolescent age group.3,5 Fortunately, fewer than 5% of pediatric appendicitis cases occur in children younger than 5 years.
As the child grows, the appendix grows from an average length of 4.5 cm in infants to 9.5 cm in the adult. The shape also matures from a funnel shape to a more conical appearance with aging. Obstruction is less likely with the early, funnel shape.
The clinical presentation of children with appendicitis may lack specificity. Almost one-third of pediatric patients may not have a history of migration of pain from the periumbilical abdominal region to the right, lower quadrant, making the diagnosis more challenging. One out of every four patients with an eventual diagnosis of appendicitis was misdiagnosed initially due to misleading symptoms such as vomiting prior to abdominal pain, dysuria, upper respiratory symptoms, or diarrhea /constipation symptoms.5 The morbidity and mortality of a ruptured appendix is much higher than for surgical removal of an unruptured appendix.
Plain radiographs of the abdomen are rarely diagnostic, (an appendicolith in the right lower quadrant and/or a localized air fluid level suggesting a localized ileus may be helpful) and are insensitive and non-specific for appendicitis.
Computed axial tomography (CAT) scanning of the abdomen has been shown to be highly sensitive and specific for the diagnosis of appendicitis, although there continues to be concern about radiation doses from abdominal/pelvic CTs in children. Currently, centers vary in their use of oral, intravenous, or rectal contrast. The use of oral contrast may result in diagnostic delays and staff challenges, especially with an ill child. Centers that use non-enhanced or non-contrast CT scans have shown that the interpretation by attending radiologists produces sensitivity, specificity, and accuracy results of 95-100%, 98-100%, and 97- 99%, respectively. The residents’ non-enhanced CT interpretation of appendicitis showed a sensitivity of 63%, specificity of 96%, and an accuracy of 88%.6-9 In addition, use of the non-enhanced CT reduces time delays, and the test takes only several minutes to perform.
Graded compression US showed a sensitivity of 100%, specificity of 88%, and an accuracy of 91% in one series.8 Other studies have demonstrated greater variability in accuracy, with some studies supporting these results, and a single study showing less favorable sensitivities of 79%, specificities of 82%, negative predictive value of 88%, and positive predictive values of 65%.10-12
Graded compression US is performed using a linear transducer, although appendicitis has been visualized during the right adnexal views of pelvic transvaginal sonography.
The normal appendix is visualized uncommonly by sonography, and when visualized, is compressible with manual or tranducer pressure on the abdomen, and is usually fewer than 6 mm in thickness.13 Procedurally, the scan should be conducted with slow, but firm, pressure applied a few centimeters to the right of the umbilicus (displace underlying bowel), and then move down the abdomen at one-half centimeter increments until the right lower quadrant of the abdomen is scanned thoroughly. The other option is to allow the patient to place the transducer over the area of most pain/tenderness.
The inflamed appendix, in cross-section, has an edematous wall. The thick wall appears echolucent. A wall thickness greater than 3 mm (e.g., diameter more than 6 mm) is considered one of the main criteria for the sonographic diagnosis of appendicitis. A target sign consisting of five concentric layers is found in slightly more than 50% of cases of non-perforated appendicitis. There is usually no peristaltic movement. Compare this with the adjacent bowel. View the structure in longitudinal section. It should elongate into, and end as, a blind loop. There is central echogenicitiy from apposed inner mucosal lining. There may be a central echolucency representing fluid in the lumen of the appendix. A fecalith may add a central echogenic focus with posterior shadowing.
In one study, the appendix was not seen on US in up to 10% of the cases. Non-visualization of the appendix accounted for nearly all of the false negative studies.13 A ruptured appendix may be decompressed sufficiently to succumb to transducer pressure and identification. Peri-appendiceal fluid or purulence may be echolucent, and the central appendix can appear as an echogenic tube with a blunt end, emerging from the larger cecum. Edematous surrounding fatty tissue is echogenic. The application of color flow signal can be of some assistance. The absence of color flow signal over the appendix can be found in both the normal and abnormal appendix. Increased vascularity shows up as a ring of fire. This sign helps support the diagnosis of appendicitis. Sonographic features associated with a perforated appendix include: fluid within the non-compressible appendix; thickened bowel loops with minimal peristaltic activity; inhomogeneous pericecal/appendiceal echogenic mass; or echolucent fluid.13 A retrocecal appendix is beyond the purview of the linear transducer.
US may be used initially to rule in appendicitis in equivocal and high pretest probability cases. It has no radiation exposure, no IV contrast risks, and is non-invasive. Non-visualization of the appendix by sonography should not be interpreted as "negative for appendicitis" or as a "normal appendix." In one study, a normal appendix was visualized by sonography in only two out of 83 pediatric patients without appendicitis, whereas CT identified a normal appendix in 62 of 74 patients without appendicitis.14 Due to the range of sensitivities and specificities of US with appendicitis, the positive and negative likelihood ratios should be applied cautiously to the clinical pretest probability of appendicitis. The desired outcome in this clinical decision-making scenario is to move one’s post-test probability below the threshold for further diagnostic imaging or above the threshold for surgical treatment or admission.15 Regardless of the pretest probability of appendicitis, if the clinician wishes or feels compelled to rule out appendicitis, then a negative or equivocal US reading should prompt further testing. In the majority of cases this would include surgical consultation or a CT scan.
Positive sonographic findings for appendicitis should result in enough evidence in moderate to high pretest probability cases for surgical consultation for definitive management or a CT of the abdomen and pelvis. Ultrasound’s negative likelihood ratio range for detecting appendicitis does not support its use in dismissing the diagnosis of appendicitis at any level of pretest probability. In a prospective study of 139 pediatric patients with equivocal clinical findings of acute appendicitis, US was the first imaging modality in the protocol. It was followed by limited CT with rectal contrast if the US was indeterminate or negative. If the US reading was positive for appendicitis, the patient went to the operating room. This protocol resulted in a sensitivity of 94%, specificity of 94%, and accuracy of 94%. US should be used to confirm the diagnosis of appendicitis, but should not be used to exclude the diagnosis.14
Pyloric Stenosis. Hypertrophic pyloric stenosis (HPS) is the most common pediatric surgical cause of vomiting. This form of gastric outlet obstruction occurs in one out of every 250 births with a male to female ratio of 4:1. The diagnosis of HPS must be considered in the young infant who presents within the first 10 weeks of life with frequent dramatic post-prandial emesis. The peak age for presentation is 3-6 weeks. The emesis is classically forceful, non-bilious and occurs immediately at the end of feeding to within 30 minutes after. Early in the disease course, the infant is aggressively hungry after vomiting, but after several days may become lethargic from dehydration, electrolyte disturbances, and metabolic alkalosis.
Clinically, these children may present with varying degrees of dehydration, based upon the length of the illness and the severity of the symptoms. The infant with a palpable, olive-sized mass in the epigastric area can be diagnosed confidently with HPS, but the insensitivity of this physical examination finding usually prompts further diagnostic testing. A definitive diagnosis of HPS may be achieved by a skilled sonographer.
Just lateral to the midline of the abdomen, at the subxyphoid region, is where the enlarged pylorus is palpated usually. A linear 7 MHz or higher frequency probe is used to scan this region. The walls of the hypertrophied circular muscles of the pyloric sphincter appear echolucent and thick. The central mucosa is echo-genic. A wall thickness greater than 4 mm is diagnostic of pyloric stenosis. Several studies show 100% specificity and 86-92% sensitivity of the 4 mm muscle wall thickness criterion. Other parameters such as muscle length (e.g., greater than 2 mm) and channel length (e.g., greater than 17 mm) also are used.16-21
The immediate and crucial goals of medical management are correction of volume deficits and electrolyte abnormalities. The definitive care of the pediatric patient identified with HPS is surgical, with a pyloromyotomy providing a full correction of the condition.
Intussusception. Intussusception occurs when a proximal segment of bowel, usually the terminal ileum, invaginates into a distal segment of bowel pulling portions of mesentery with it. Incidence is estimated at 2.4 cases per 1,000 births.22 Although this condition occurs mainly in infancy, prior to the age of two years, it also may occur in older children, adolescents, and adults. In fewer than 10% of cases, intussusception may be initiated by an anatomical lead point such as polyposis or a neoplasm, but most commonly it is idiopathic. Older children have a higher incidence of an anatomic lead point. Closely associated with viral illnesses such as upper respiratory infections and gastrointestinal illnesses, the occurrence of intussusception peaks during the viral illness peaks of summer and winter.
Patients typically present with intermittent, episodic abdominal pain, vomiting, and a guaiac positive stool. In some cases, lethargy during the pain-free intervals may be the only clinical clue to the condition. Physical examination findings vary and may include a sausage-shaped, right-sided mass and abdominal tenderness.
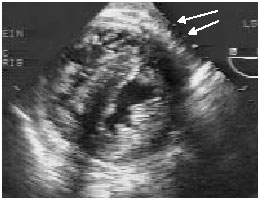
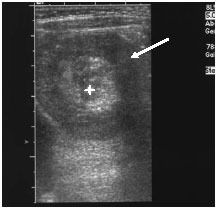
Abdominal radiography is not a reliable screening tool. Radiographs may be normal, show bowel obstruction, no stool in the distal colon, or very little intestinal gas. In some centers, the enema is the first option for its dual benefits of diagnostic imaging and therapeutic intervention. Others consider sonography to be an ideal non-invasive, screening diagnostic modality that limits radiation exposure to children. Using the high frequency linear probe will reveal a target or bull’s eye lesion in the tranverse scan plane. The hypoechoic rim represents the edematous bowel wall of the receiving end (i.e., intussuscipiens) infolded bowel loops. The central echogenicity represents the mucosal surfaces and muscular wall of the infolded bowel (i.e., intussusceptum). A transverse scan of the proximal segment of intussusception may have a central anechoic region (bull’s eye), giving it a target-like appearance. (See Figure 5, below.) On the longitudinal view, edematous bowel encasement is oval (i.e., it looks like a renal cortex), and the central echogenicity resembles that of a sinus of a kidney, hence the term "pseudo kidney" sign.
|
The sonographic identification of invaginated and edematous bowel wall in the pediatric patient suspected of intussusception can be treated rectally with the hydrostatic pressure of fluid or air. An enema alone may provide diagnostic and therapeutic benefits. Ultrasound guidance can be used also. Surgical involvement may be indicated if reduction attempts are unsuccessful.
Gallbladder Disease. Biliary tract disease and gallbladder abnormalities should be suspected in patients with upper abdominal pain and nausea and vomiting, or abdominal tenderness and jaundice.
A 3.5-5 MHz transabdominal probe should be used in the small child. If the patient is old enough and able to cooperate, ask the child to hold his/her breath. This will move the liver below the costal margin to provide an excellent acoustic window. Imaging also may be improved by rolling the patient onto his/her left side with the shoulders vertical. If this is not possible, then scan between the ribs, although this is a very limited scan area.
The non-contracted gallbladder (GB) is visualized with careful attention to key surrounding landmarks. It is usually inferior to the liver, anteromedial to the superior pole of the right kidney, and anterior to the echogenic walled portal vein. The main lobar fissure connects the GB to the right portal vein. The normal gallbladder has smooth echogenic walls, no internal echoes, and posterior acoustic enhancement. Normal variants include septations near the GB neck, Phrygian cap (e.g., folded fundus), Hartman’s pouch (e.g., folded neck), agenesis, or duplicate GB. The GB usually is contracted for a few hours after a meal. The normal cystic duct can be 1-2 cm in length and 1.8 mm in width. It is uncommon to visualize the cystic duct by sonography. The GB measures 3.0-3.2 cm in the infant, then matures to 7-10 cm at adulthood. The GB width is usually one-third the length. The GB should be viewed in both longitudinal and transverse planes. Posterior acoustic enhancement makes it difficult to measure the GB wall thickness at the posterior GB wall. Use only the anterior GB wall to assess or measure wall thickness. The posterior wall measurement is compromised by posterior acoustic enhancement artifact. Gallbladder wall thickness more than 4 mm is considered indicative of wall inflammation from a diversity of etiologies.
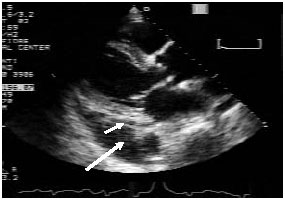
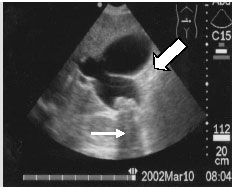
Cholelithiasis is uncommon in the preadolescent patient unless a predisposing, comorbid condition exists. Gallstones commonly are found in pediatric patients with sickle cell disease and may be found in the pediatric patient with hemolytic anemia, liver disease, Crohn’s disease, cystic fibrosis, total parenteral nutrition, and as an uncommon side consequence of certain drug use.23 If cholecystitis is suspected, the main sonographic features focus on the GB wall. The presence of gallstones is not sufficient evidence to state that gallstones cause the patient’s condition. Gallstones usually are echogenic structures on the inner wall of the GB. GB polyps are also echogenic. Polyps remain attached to the GB wall, whereas the majority of gallstones move to the gravity-dependent areas when the patient’s position is changed. Polyps do not produce shadows. When gallstones are 3 mm or larger, they produce posterior shadowing. The shadows produced by gallstones are "clean shadows" (i.e., sharp edges and free of internal echoes). Small, non-shadowing echogenic structures that move when the patient is repositioned are considered as stones. (See Figure 6, below.)
|
Another possible finding within the GB is sludge. Normal bile is completely echolucent. Sludge is considered modified bile. It has midlevel echogenicity (gray). It is viscous, does not produce shadows, is gravity-dependent, and conforms to the walls of the GB. Its clinical significance is unclear but has been noted to exist in situations of bile stasis and GB infection. It also has been noted to be a precursor of sludge balls, which have the ability to cause GB obstruction.
Cholecystitis —inflammation with or without infection of the GB wall— may be found in the absence of gallstones in the young patient. In contrast, acalculous cholecystitis is a rare occurrence in the adult patient. As the GB wall thickens, the inflammation may appear as an echolucent, middle-wall layer. (See Figure 7, below.) Advanced inflammation, extending throughout all the wall layers, can decrease the echogenicity of the entire GB wall. The GB may become distended if the cystic duct is obstructed completely. Wall thickness greater than 5 mm is considered abnormal. The ultrasonographic Murphy’s sign, although reported to have poor sensitivity and specificity, may provide additional information. Use the probe to apply pressure to the right, upper quadrant as the patient takes in a deep breath. If maximal tenderness is elicited when active GB compression is displayed on the monitor, then that is considered a positive Murphy’s sign. Nuclear medicine tests are more reliable than US for the diagnosis of acalculous cholecystitis. Choledochal cyst is a congenital dilation of the common bile duct. This cyst may cause intermittent pain, vomiting, and a palpable mass in the pediatric patient. The biliary obstruction may cause jaundice. US is not useful in confirming or dismissing a diagnosis of biliary atresia, an obstructive disorder that needs rapid diagnosis and surgical intervention in the neonatal patient.
|
Common Bile Duct. The common bile duct (CBD) is important to assess. It is formed after the union of the cystic duct and the common hepatic duct. The CBD is joined eventually by the pancreatic duct before it enters the duodenum. It runs anterior to the portal vein and, for a part of its course, runs parallel to it on the longitudinal scan view. The probe is positioned at the right costal margin at the mid-clavicular line. The patient can be supine or turned 45° onto the left side. Keeping the probe in the transverse position, slowly angle from the left hip to the right shoulder until the portal vein’s echogenic walls are noted. Decreasing the depth setting of the machine provides more detailed images. Slight rotation of the probe will elongate the portal vein and its immediately anterior CBD. The normal echolucent bile duct lumen is narrower than the portal vein’s lumen. Slight rotation of the probe will obtain a transverse view of the portal vein and the hepatic artery and common bile duct lumina. It has the appearance of a silhouette of a round face with two small ears. The enlarged CBD will give an asymmetry to the ears. Measurements of the bile duct are made from inner wall to the inner wall perpendicular to the long axis of the CBD. Patients with symptoms determined to be due to cholelithiasis and/or cholecystitis should be admitted for pain control, observation, or antibiotic administration.
Pediatric Renal Sonography
Bedside US is useful in the pediatric patient presenting with acute urinary retention, renal colic, acute renal failure, acute pyelonephritis, possible renal abscess, or renal trauma. The focused application of emergency US in the pediatric patient presenting with flank pain is determination of the presence or absence of hydronephrosis.
The history should focus on factors that predispose to renal disease such as inborn errors of metabolism or other metabolic disorders, history of renal calculi, or urinary tract infections. Also, query patients or primary care providers regarding flank pain, trauma to the kidney, recent streptococcal infections, colicky abdominal pain, change in urinary output, urinary frequency, dysuria, retention, or hematuria in the child. A family history of renal disease, such as polycystic kidneys, calculi, or renal failure, also may help direct the diagnostic evaluation. On physical examination, pay particular attention to vital signs suggesting the distress of pain and abdominal or flank tenderness. Laboratory examination should include an examination of renal functioning, urinalysis, urine microscopy, and crystals.
Renal Sonography. Emergency renal sonography requires evaluation of both kidneys and the bladder in the longitudinal and transverse planes. Utilizing a low frequency probe (e.g., 3.5-5 MHz) and B-mode sonography, place the patient in the supine or lateral decubitus position. The kidneys are located anatomically in the retroperitoneum and are surrounded by adipose tissue. Both are bound supero-posteriorly by the dome of the diaphragm. Typically, the right kidney is imaged inferior to the costal margin in approximately the anterior axillary or mid-axillary line. The right kidney is slightly larger and more inferiorly displaced than the left. The right kidney is easier to image sonographically as the liver serves as an optimal acoustic window, while the left kidney is bound by the spleen, pancreas, stomach, and small and large intestines, creating more of a challenge. Identify the superior and inferior poles of the kidney with particular attention to the renal parenchyma (e.g., cortex and medulla) and the sinus into which the medullary pyramids/papillae drain to form the pelvis and travel with the renal artery and renal vein at the hilum. Scan in a longitudinal plan and obtain images of both the superior and inferior poles. Rotate the transducer 90° to obtain the images in the transverse plane. For the left kidney, place the transducer inferior to the costal margin at the level of the posterior axillary line. Scan the bladder in both planes with the transducer placed just superior to the pubic bone.24
A white, echogenic capsule—with less echogenic parenchyma than that of liver parenchyma— surrounds each kidney. The less echogenic and darker medullary pyramids merge to form the pelvis, which is black and hypoechoic, consistent with fluid-filled structures. The centrally located sinus appears white to gray in color, secondary to fat content. The bladder appears as an anechoic fluid-filled vessel surrounded by a thin, echogenic white line. On the transverse view, the ureteral jets marking urine flowing into the bladder from the ureters may be viewed using color flow Doppler.
Hydronephrosis appears as anechoic fluid-filled pockets prominently displayed in the central sinus. Hydronephrosis is graded as mild, moderate, or severe based upon the EP’s judgment performing the study. Occasionally, a calculus will be viewed within the kidney parenchyma. If hydronephrosis is viewed at the level of the kidney, evaluate for presence or absence of ureteral jets on bladder view. Absence of a ureteral jet on the affected kidney side is highly consistent with an obstructive calculus.
Commonly confused with hydronephrosis is prominent renal pyramids. This is due to the central location within the kidney and its similar sonographic appearance. To distinguish prominent pyramids from hydronephrosis, look for a triangular shape consistent with a pyramid, and look for intervening renal cortex.
If an abnormality of the kidney such as a duplicated collecting system, horseshoe kidney, a pelvic kidney, or congenital absence of a kidney, is recognized, order a formal US performed by a radiologist. Do not lose sight of the focused, clinically applicable question.
Radiological Imaging in the Pediatric Patient. There is no gold standard for evaluating hydronephrosis in the pediatric patient. Sonography is accepted as an initial study for establishing the diagnosing hydronephrosis. A combination of plain radiographs, intravenous pyelogram (IVP), voiding cystourethrography, and nuclear scintigraphy may be utilized to evaluate the hydronephrosis further. Although sonography does not provide information on kidney functioning or stone location, the EP can decide if there is hydronephrosis and how to proceed clinically in evaluating an individual patient. Sonography also may be useful for locating cysts and differentiating renal masses, but has limited applicability in the ED.24 US may be falsely negative if the patient is dehydrated, and falsely positive if the patient’s bladder is full, causing bilateral hydronephrosis. Repeat the scan after rehydration or voiding. Unilateral hydronephrosis, especially on the right side, may be seen in pregnant patients.24 The bedside US plus plain, abdominal radiograph (KUB) is considered as sensitive as intravenous pyelogram (IVP) in screening patients with suspected ureteral colic.24-27 Prospective data support the use of contrast-enhanced magnetic resonance imaging (MRI) for the evaluation of hydronephrosis in children, but neither is time nor cost effective for most EPs.28,29 Studies comparing sensitivity and specificity of US versus non-enhanced helical CT focus on the adult not pediatric population.30 Although IVP allows detection and location of renal calculi, its relative disadvantages include: placing an IV in an already agitated pediatric patient; awaiting laboratory results of renal function; a radiologist to administer the dye and perform the examination; and contraindications of pregnancy, contrast allergies, and renal insufficiency.
Renal Calculi. Pediatric and adolescent patients do present with renal calculi although the incidence is approximately 1/50 that of adults. Ninety percent of patients have hematuria, and 50% have preceding abdominal or flank pain.31,32 Renal calculi in the pediatric population commonly result from hypercalcuria and often are genetic.31,33 Infection-induced stones are more common in younger children, and are 80% more prevalent in boys. A small percentage of stones are secondary to cystinuria from hereditary disorders of amino acid metabolism and also secondary to uric acid with hyperuricemia or hyperuricosuria from increased purine synthesis.32 In North America, children and adolescents with metabolic disease account for approximately 50% of kidney stone presentations. In Europe, the majority of kidney stones are infection related, while in Southeast Asia, uric acid bladder stones are most prevalent. Unlike the male predominance in adults, kidney stones present equally in boys and girls.32,34 After the ED assessment described above, utilize bedside sonography to assess the kidneys expediently. Scan the bladder with color Doppler and evaluate for ureteral jets. If obstruction is suggested by sonography, urgent urologic consultation is recommended.
Acute Renal Failure. Renal sonography is the radiological adjunct study of choice to differentiate post-renal from intrarenal or pre-renal acute renal failure (ARF). Hydronephrosis is evident sonographically if the cause of ARF is obstructive in nature. In chronic renal failure, kidneys appear sonographically small, atrophic, and hyperechoic.
Renal Cysts. Sonographically, renal cysts are seen quite frequently. Do not mistake them for hydronephrosis. Cysts appear round or oval-shaped with a thin wall, a clear interface with the surrounding renal parenchyma, and are located eccentrally. Simple cysts contain clear fluid without internal echoes and demonstrate mild, posterior enhancement. Distinguish cysts from hydronephrosis by scanning to see that cysts do not connect centrally within the renal pelvis.24 Suspect polycystic kidney disease when sonographically large cysts distort the normal renal architecture. In polycystic kidney disease, patients typically present with hypertension, flank pain, and hematuria.
Acute Pyelonephritis or Renal Abscess. The sonographic appearance of acute pyelonephritis is typically that of a normal kidney. If the infection has progressed to an abscess, the EP may view an oval or round, heterogeneous-appearing mass filled with fluid, septations, or debris.24
Renal Mass. EPs are increasingly detecting renal masses as incidental findings when performing bedside US in the ED.35 The sonographic appearance of a renal mass is heterogeneous and varies in presentation from solid to cystic with variable echogenicity. Wilm’s tumor or nephroblastoma is the most common intra-abdominal malignancy in childhood. Most patients present before age 5, and US is the radiographic modality preferred for initial localization.36 Order a formal complete US performed by the department of radiology when a mass is viewed.
Acute Urinary Retention. The patient presenting with acute urinary retention, also may be evaluated with US. Initially, place the US probe over the bladder and obtain images in two planes. Estimate the size of the bladder in a small, medium, or large quantification. Image both kidneys to determine the presence of hydronephrosis. Based upon findings, place a urinary catheter for decompression or seek alternative causes. Most US machines have an automatic calculator setting for bladder volume measurement. Although of questionable accuracy, a rough estimate of bladder volume is obtained by multiplying length by width by height by 0.75.37
Renal Trauma. The use of sonography and typical findings in the pediatric patient presenting with renal trauma will be presented in a later article.
Summary and Pitfalls. Perform a limited, pediatric renal sonography when there is clinical suspicion of hydronephrosis. Order a formal, complete renal US for any incidental finding.


Conclusions
Ultrasound is a valuable diagnostic tool for the EP in the evaluation of cardiac, abdominal, and renal diseases. Its portability, safety, and accuracy suggest that this modality will become increasingly popular as a tool for screening pediatric patients for potentially serious disease processes. The EP should remain focused on the value of each test, the skills of the individual (i.e, EP or radiologist) performing the test, and associated limitations.
References
1. Lee C, Mason LJ. Pediatric cardiac emergencies. Anesthesiol Clin North Am 2001;19:287-308.
2. Basilico FC. Cardiovascular disease in athletes. Am J Sports Med 1999; 27:108-121.
3. Rothrock SG, Pagane J. Acute appendicitis in children: Emergency department diagnosis and management. Ann Emerg Med 2000;36: 39-51.
4.. Irish MS, Pearl RH, Caty MG, et al. The approach to common abdominal diagnosis in infants and children. Pediatr Clin North Am 1998;45:729-772.
5. Rothrock SG, Skeoch G, Rush JJ, et al. Clinical features of misdiagnosed appendicitis in children. Ann Emerg Med 1991;20:45-50.
6. Lowe LH, Draud KS, Hermanz-Schulman M, et al. Nonenhanced limited CT in children suspected of having appendicitis: Prospective comparison of attending and resident interpretations. Radiology 2001;221:755-759.
7. Lowe LH, Penney MW, Scheker LE, et al. Appendicolith revealed on CT in children with suspected appendicitis: How specific is it in the diagnosis of appendicitis? AJR Am J Roentgenol 2000;175:981-984.
8. Lowe LH, Penney MW, Stein SM, et al. Unenhanced limited CT of the abdomen in the diagnosis of appendicitis in children: Comparison with sonography. AJR Am J Roentgenol 2001;176 :31-35.
9. Lowe LH, Perez R Jr, Scheker LE, et al. Appendicitis and alternate diagnoses in children: Findings on unenhanced limited helical CT. Pediatr Radiol 2001;31:569-577.
10. Puig S, Hormann M, Rebhandl W, et al. US as a primary diagnostic tool in relation to negative appendectomy: Six years experience. Radiology 2003;226:101-104.
11. Vazquez Ronco MA. Diagnostic yield of abdominal ultrasonography in acute appendicitis. An Pediatr (Barc) 2003.;58:556-561.
12. Poortman P, Lohle PN, Schoemaker CM, et al. Comparison of CT and sonography in the diagnosis of acute appendicitis: A blinded prospective study. AJR Am J Roentgenol 2003;181:1355-1359.
13. Hahn HB, Hoepner FU, Kalle T, et al. Sonography of acute appendicitis in children: 7 years experience. Pediatr Radiol 1998;28:147-151.
14. Garcia Pena BM, Mandl KD, Kraus SJ, et al. Ultrasonography and limited computed tomography in the diagnosis and management of appendicitis in children. JAMA 1999;282:1041-1046.
15. Gallagher EJ. Clinical utility of likelihood ratios. Ann Emerg Med 1998;31:391-397.
16. Blumhagen JD, Maclin L, Krauter D, et al. Sonographic diagnosis of hypertrophic pyloric stenosis. AJR Am J Roentgenol 1988;150: 1367-1370.
17. Blumhagen JD. The role of ultrasonography in the evaluation of vomiting in infants. Pediatr Radiol 1986;16:267-270.
18. Wilson DA, Vanhoutte JJ. The reliable sonographic diagnosis of hypertrophic pyloric stenosis. J Clin Ultrasound 1984;12:201-204.
19. Blumhagen, JD. Ultrasonography in diagnosis of hypertrophic pyloric stenosis. J Pediatr 1983;103:496-497.
20. Blumhagen JD, Noble HG. Muscle thickness in hypertrophic pyloric stenosis: Sonographic determination. AJR Am J Roentgenol 1983; 140:221-223.
21. Blumhagen JD, Coombs JB. Ultrasound in the diagnosis of hypertrophic pyloric stenosis. J Clin Ultrasound 1981;9:289-292.
22. Bisse, GS, Kirks DR. Intussusception in infants and children: Diagnosis and therapy. Radiology 1988;168 :141-145.
23. Maccherini M, Borlini G, Branchi M, et al. Ceftriaxone-induced cholelithiasis. Pediatr Med Chir, 1998;20:341-343.
24. Swadron S. Renal. In: Ma OJ, Mateer JR. eds. Emergency Ultrasound. New York: McGraw-Hill Companies, Inc.; 2003:197-220.
25. Henderson SO, Hoffner RJ, Aragona JL, et al. Bedside emergency department ultrasonography plus radiography of the kidneys, ureters, and bladder vs intravenous pyelography in the evaluation of suspected ureteral colic. Acad Emerg Med 1998; 5:666-671.
26. Dalla Palma L, Starul F, Bazzocchi M, et al. Ultrasonography and plain film versus intravenous urography in ureteric colic. Clin Radiol 1993;47:333-336.
27. Gorelik U, Ulish Y, Yagil Y. The use of standard imaging techniques and their diagnostic value in the workup of renal colic in the setting of intractable flank pain. Urology 1996; 47:637-642.
28. Perez-Brayfield MR, Kirsch AJ, Jones RA, et al. A prospective study comparing ultrasound, nuclear scintigraphy and dynamic contrast enhanced magnetic resonance imaging in the evaluation of hydronephrosis. J Urol, 2003;170:1330-1334.
29. Grattan-Smith JD, Perez-Bayfield MR, Jones RA, et al. MR imaging of kidneys: Functional evaluation using F-15 perfusion imaging. Pediatr Radiol 2003; 33:293-304.
30. Sheafor DH, Hertzberg BS, Freed KS, et al. Nonenhanced helical CT and US in the emergency evaluation of patients with renal colic: Prospective comparison. Radiology 2000;217:792-797.
31. Stapleton FB. Childhood stones. Endocrinol Metab Clin North Am 2002;31:1001-1015.
32. Cronan K. Renal and Electrolyte Emergencies. In: Fleischer GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine. New York: Lippincott Williams & Wilkins; 2000:811-858.
33. Ronnefarth G, Misselwitz J. Nephrocalcinosis in children: A retrospective survey. Members of the Arbeitsgemeinschaft fur padiatrische Nephrologie. Pediatr Nephrol 2000;14:1016-1021.
34. Polinsky MS, Kaiser BA, Baluarte HJ. Urolithiasis in childhood. Pediatr Clin North Am 1987; 34:683-710.
35. Mandavia DP, Pregerson B, Henderson, SO. Ultrasonography of flank pain in the emergency department: renal cell carcinoma as a diagnostic concern. J Emerg Med 2000;18:83-86.
36. Hogarty MD. Oncologic Emergencies. In: Fleischer GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine. New York: Lippincott Williams & Wilkins; 2000, 1168-1169.
37. Chan H. Noninvasive bladder volume measurement. J Neurosci Nurs 1993;2:309-312.
The first part of this two-part series addressed the use of US for complications of early pregnancy in adolescent females and for testicular complaints in pediatric and adolescent males. This issue focuses on the use, role, and limitations of US in the evaluation of cardiac, abdominal, and renal disease processes.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.