Considerations in the Diagnosis and Emergency Management of Pediatric Tachycardias
Considerations in the Diagnosis and Emergency Management of Pediatric Tachycardias
While rhythm disturbance may be a common presenting complaint among adult emergency department (ED) patients, the incidence of cardiac dysrhythmia among pediatric patients is relatively low. In one retrospective review, primary cardiac arrhythmias were identified in 13.9 per 100,000 pediatric ED visits.1 The incidence of these dysrhythmias peaked during infancy and then again in adolescence.1 Cardiac dysrhythmias in children may be due to primary conduction abnormalities or may occur in the setting of structural heart disease, metabolic derangements from toxic ingestions, or infections. Supraventricular tachycardias (SVT) represent the most common pediatric dysrhythmias in adolescents (an estimated 63% of all documented tachycardias).1 After a brief review of initial emergency management of dysrhythmia, the authors will emphasize important pediatric ECG parameters and how they differ from adults.
Ann M. Dietrich, MD, Editor
Introduction and Epidemiology
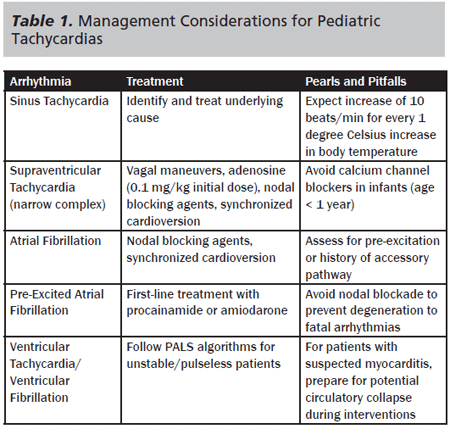
Although pediatric dysrhythmias are relatively rare, pathophysiology, presentation, and management can differ from adults in significant ways. This article focuses on the unique diagnostic and management considerations for pediatric dysrhythmias. Table 1 provides an overview of the incidence of pediatric tachycardias and introduces pearls and pitfalls for the practicing emergency provider who cares for both adults and children. After a brief review of initial emergency management of dysrhythmia, we will emphasize important pediatric ECG parameters and how they differ from adults. We then focus on common dysrhythmias in children, with an emphasis on presentation, management, and disposition. We also discuss unique management considerations in children presenting with myocarditis.
Initial Presentation
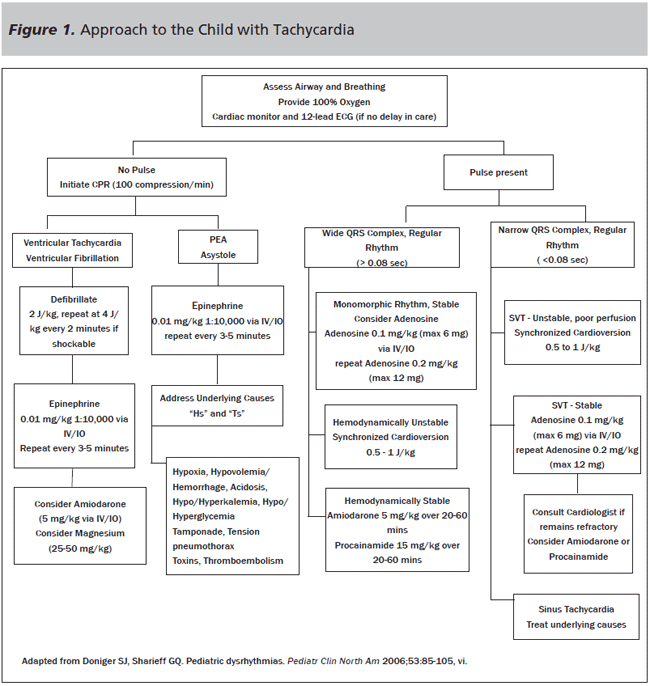
Initial assessment and management of the child presenting with dysrhythmia should begin with evaluation of the child's airway, breathing, and circulation. Emergency providers should be familiar with Pediatric Advanced Life Support (PALS) diagnostic algorithms for patients presenting in extremis with unstable or absent vital signs. (See Figure 1.) Presentations of bradycardia and cardiac arrest in children are more commonly due to progression of respiratory failure (versus primary cardiac disease in adults), so assessment for hypoxia and adequate ventilation is an important initial management priority.2,3 Assessment of respiratory status, capillary refill, and mental status may help to identify signs of cardiogenic decompensation requiring immediate intervention. Frequent reassessment of airway, breathing, circulation, and disability should be standard practice for any patient with suspected dysrhythmia, as a patient's stability may change abruptly.
Symptoms will depend on the circulatory status and duration of the dysrhythmia, and will vary according to age group. For example, in neonates, poor feeding, irritability, and sweating during feeding may be the only signs of heart failure secondary to tachyarrhythmias. Infants may not develop specific symptoms until congestive heart failure begins to manifest.4 Older children may be able to communicate the sensation of palpitations or dizziness.5,6 Children often have difficultly describing palpitations and therefore may complain of abdominal or chest pain. Dizziness or lightheadedness is common, but syncope resulting from SVT is rare and should be treated seriously. Syncope with no prodrome or during exercise is concerning and a cardiac cause should be considered. Occasionally, children and adolescents presenting with an apparent first-time seizure episode may actually have had a syncopal episode with tremor or convulsion that was mistaken for a seizure. A screening ECG should strongly be considered for all patients presenting with a first-time seizure without an obvious cause, such as febrile seizures, or in the case of an unusual history of present illness, such as seizure with exercise.
In toddlers as well as in adolescents, careful assessment for possible ingestion or intentional overdose is important. History of congenital heart disease, which is beyond the scope of this article, widens the differential diagnosis of the rhythm type greatly and complicates the treatment. Family history of dysrhythmia, syncope, or sudden cardiac death can offer clues to the presence of familial conditions. Similarly, the presence of findings often associated with congenital cardiac conduction disorders (such as long QT syndrome) may provide clues to underlying risks of arrhythmia; these may include, for example, congenital deafness or limb anomalies.
Vital signs should include an accurate temperature, as fever is perhaps the most common cause of a sinus tachycardia. Palpation of pulses in all four limbs could aid in detection of congenital heart disease such as aortic stenosis in a young child. Note that low blood pressure is a late finding in children. In addition to cardiac and pulmonary auscultation, assessment of the liver edge (greater than 1-2 cm below the costal margin) may reveal subtle signs of cardiac failure.
ECG Testing
The 12-lead ECG is the most essential diagnostic test for cardiac dysrhythmia, and it is important for emergency providers to be familiar with key differences in ECG interpretation among children. While there are many approaches to interpreting pediatric ECG, Evans et al have described a four-step method to reading a pediatric ECG: determining rate and rhythm; evaluating intervals; evaluating the QRS and P wave axes; and assessment for right and left ventricular hypertrophy.7 The pediatric ECG differs from the adult ECG in three fundamental ways. First, normal ranges of heart rate change with age. Second, the QRS axis in the newborn is rightward and, over the course of the first years of life, becomes leftward, as in adults. Third, the parameters for "normal" intervals change. For example, a widened QRS is > 0.9 ms in children compared to 100 ms in adults. Table 2 presents a summary of these specific age-related changes; note that these parameters provide the latest ranges but are constantly changing as studies are updated. As with any ECG interpretation in adults or children, if abnormalities are seen, comparisons with old ECGs should be made, if available.
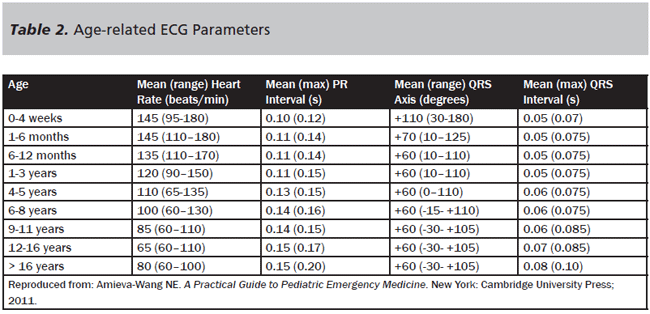
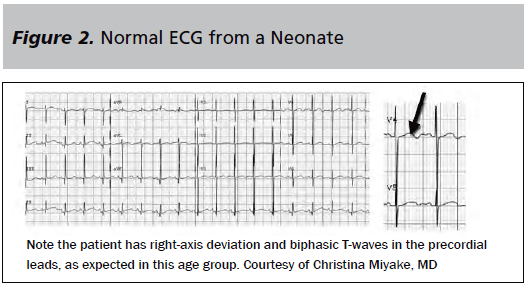
The heart rate varies from a mean normal range of 125 to 145 beats per minute in infancy to the normal adult mean of 80 beats per minute by adolescence; the change is related to changes in vagal tone that occur with age.8 QRS axis and interval will also change with age as the child's cardiac muscle mass decreases in the right ventricle and increases in the left ventricle. As a result, at very young ages children may have right-axis deviation, as shown in Figure 2, demonstrating a normal ECG from an infant. Left bundle-branch block in a child is always considered abnormal and is a marker of underlying pathologic disease. Lastly, the QTc interval, calculated based on rate, can vary but should warrant further evaluation if the QTc is greater than 450 msec.
The Common Dysrhythmias and Their Specific Management
Rhythm abnormalities may be categorized in several ways. A common approach is to categorize the abnormality based upon their effect on the child's pulse slow, fast, or absent.2,6,9,10 A second approach is to organize the rhythm disturbance according to the location of the conduction abnormality: nodal abnormalities, supraventricular/atrial rhythms, junctional, and ventricular rhythms.11 Regardless of the method of classification, both pulse rate and consideration of the etiology of the conduction abnormality are important in assessing the child's status and in determining the appropriate intervention.
Figure 3 describes an approach to classifying pediatric tachycardias and provides the basis for a discussion of the most common dysrhythmias in children. Once a tachycardia has been identified, we recommend differentiating between narrow QRS-complex or wide QRS-complex rhythms (again, recall that the normal QRS duration varies by age). SVT is the most common etiology of narrow-complex tachycardia in the pediatric population.
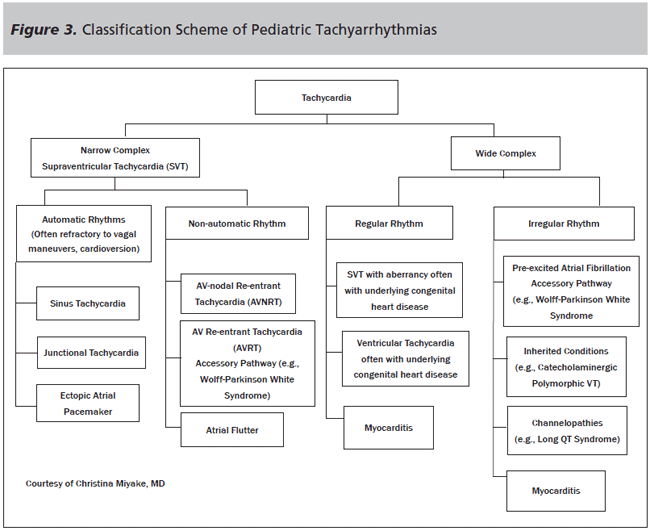
Wide-complex tachycardias suggest ventricular tachycardia in children, and should be treated as such. Generally speaking, SVT "with aberrancy" (i.e., wide-complex SVT) can occur in the pediatric population, but when a wide-complex tachycardia is identified, it is safest to assume a ventricular tachycardia. The emergency physician should differentiate wide-complex tachycardias by determining whether the rhythm is regular or irregular. (See Figure 3.) Irregular, wide-complex tachycardias, which are consistent with pre-excited atrial fibrillation, must be recognized and require unique management.
Supraventricular Tachycardia (SVT). SVT itself can be a somewhat nebulous term. Technically speaking, a supraventricular tachycardia refers to any tachyarrhythmia that arises from foci at or above the bundle of His, independent of a specific mechanism; thus, the term can be applied to a host of conduction abnormalities, including sinus tachycardia, re-entry tachycardias, atrial flutter, and ectopic atrial or junctional tachycardias.4,10,12 Understanding the underlying source of a narrow-complex dysrhythmia can increase the likelihood of successful treatment in the ED.
SVT may be "automatic," meaning that the SVT comes from an ectopic pacemaker focus that is continuously firing at rapid rates. Examples of these types of SVT include sinus tachycardia and ectopic atrial or junctional tachycardia. Junctional tachycardia and ectopic atrial tachycardias are relatively rare, accounting for less than 5% of SVT in children. While multifocal atrial tachycardias may be seen in adults, this specific rhythm disorder is exceedingly rare in the pediatric patient and is almost uniformly only seen in neonates. These automatic SVTs do not terminate with typical ED treatments such as vagal maneuvers, adenosine, or direct current cardioversion. (See Figure 3.) Alternatively, the rhythm may be re-entrant and not automated. If an otherwise healthy child presents with a narrow-complex SVT, there is a 90% chance that the rhythms will be due to AVNRT or an accessory pathway. These re-entrant rhythms, unlike automatic tachycardias, can be broken with vagal maneuvers, adenosine, or direct current cardioversion and are the most commonly encountered in the ED.10,12
Automatic Tachycardias: Sinus Tachycardia. Sinus tachycardia deserves special mention because it is the most common of pediatric dysrhythmias. Diagnostic parameters are adjusted for age, as reviewed above in Table 2. The rate of this rhythm will vary over time. On ECG, at high rates above 200 bpm, the p wave may appear absent, even in sinus tachycardia, because it becomes hidden within the preceding T wave.7,9,12 This rhythm arises from an automated focus and, in general, represents a physiologic response to a particular stimulus. There may be associated clinical signs to suggest secondary causes of sinus tachycardia, including simple psychosocial factors such as anxiety or pain. Fever in children is a very common cause of sinus tachycardia; in fact, among young children, one should find an expected increase of 10 bpm in pulse for every 1 degree Celsius increase in body temperature.13,14 Just as in adults, if the response is deemed to be an "appropriate" rate without causing significant end-organ effects (e.g., dizziness or hypotension), the underlying etiology should be determined and addressed.
Re-entrant Supraventricular Tachycardias (Non-automated Tachycardias). Re-entrant tachycardias represent the most common etiology for supraventricular tachycardia in children and are either due to an AV nodal re-entrant tachycardia (AVNRT) or an accessory pathway mediated atrioventricular re-entrant tachycardia (AVRT).
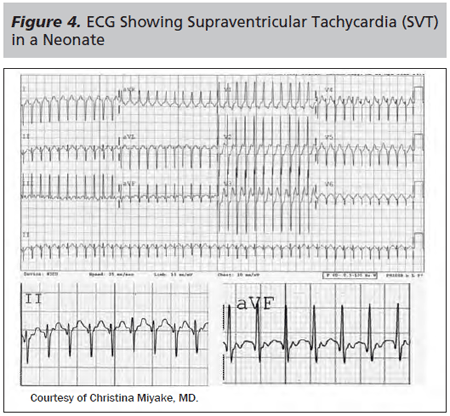
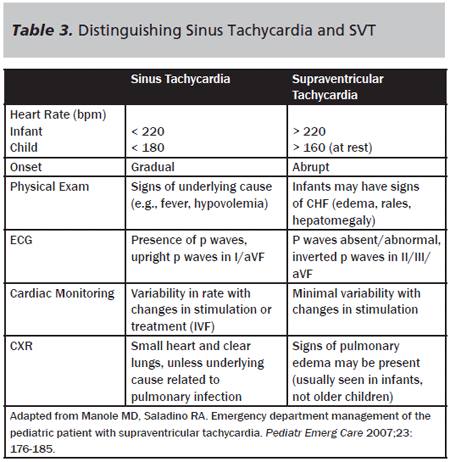
Atrioventricular Nodal Re-entrant Tachycardia (AVNRT). Differentiating the presence of sinus tachycardia and re-entrant SVT on ECG can be challenging. ECG characteristics include presence of a narrow QRS complex but absence of p waves. Figure 4 provides an example of an ECG showing SVT. Sinus tachycardia will show variability with physiologic or emotional stressors, such as exertion or crying, whereas re-entrant SVTs should show very little variation in rate. Table 3 provides distinguishing features between sinus tachycardia and SVT.
Treatment. Initial treatment of AVNRT or AVRT for most children is similar to that of adults. Typically in a stable patient, initial attempts should be directed at vagal maneuvers to elicit successful cardioversion to sinus rhythm. Among neonates and infants, the dive reflex is well-preserved and can be activated through placement of cold ice over the child's forehead. Placing ice into a plastic bag and holding this over the patient's forehead is a way to stimulate the reflex. Direct supraorbital pressure as a method to induce the diver's reflex is no longer routinely recommended, as there is risk for retinal detachment with this maneuver; similarly, carotid massage is not recommended in children as it has not been fully studied.12 In slightly older children who are able to cooperate with commands, valsalva maneuvers can be attempted.
Often, pharmacologic cardioversion with adenosine may be required. Adenosine is a naturally occurring nucleoside with an extremely short half-life of 6 to 10 seconds; physiologic effects on receptors in the sinus and AV nodal tissue result in slowed conduction through the AV node and may be seen about 10 seconds after administration.12 Administration and dosing are the same for children as well as adults, with a recommended initial dose of 0.1 mg/kg to a maximum of 6 mg. Just as with adults, the medication should be given by rapid IV push due to the short half-life and the need to deliver the medication to the coronary arteries for it to act. Thus, the adenosine should also be followed by a saline flush, and other methods such as elevating the limb may help. For rapid administration, two providers can administer the medication using a first syringe with the adenosine and a second saline flush syringe both pre-connected to the most proximal part of the IV tubing as possible. After the first provider pushes the adenosine, the second provider can immediately flush the line with the saline syringe. If initial attempts with adenosine are unsuccessful, additional doses of 0.2 mg/kg to a maximum of 12 mg may be attempted. For any unstable patient with SVT, synchronized electrical cardioversion should be performed with dosing according to PALS guidelines of 0.5 J/kg and then 1-2 J/kg on repeat attempts.3,12
During any maneuver, including ice, vasalva, adenosine, or DC cardioversion to terminate the rhythm, a 12-lead ECG or, at a minimum, the bedside telemetry should be run continuously, because much can be learned from the response to the maneuvers and termination if successful. If the rhythm converts from SVT to sinus tachycardia or normal sinus rhythm, then the likely etiology of SVT is a re-entrant circuit. If the rhythm does not convert, it may suggest a more automated focus. Adenosine can also be used to identify other underlying automated rhythms. For example, administration of adenosine to a patient seemingly in SVT may cause the rhythm to slow and reveal the presence of atrial flutter waves, suggesting flutter rather than AVNRT or AVRT as a cause. If the rhythm converts to sinus, obtain a post-conversion ECG and review it for signs of underlying or concealed accessory pathways (as will be discussed below).
Pitfalls. Children younger than 1 year of age have immature sarcoplasmic reticula, and use of calcium-channel blockers such as verapamil in infants is associated with hypotension, cardiovascular collapse, and death.4,12 Whereas amiodarone is commonly used to treat arrhythmias in adult populations, we recommend discussion with a pediatric cardiologist if considering use of this medication in a child. The use of IV amiodarone in infants has been associated with hypotension and death. The half-life of amiodarone is extremely long, sometimes remaining in the system for months, and initial administration of the medication may then affect the choice of anti-arrhythmics for long-term management of these patients. Additionally, the side effects of amiodarone, including thyroid and pulmonary effects, may be more consequential in children.
Disposition. Disposition of a patient presenting with SVT should be determined based on historical features, response to treatment, and clinical signs. The vast majority of patients with uncomplicated re-entrant SVT will likely respond to either vagal maneuvers or adenosine. For those patients with an established diagnosis of a re-entrant SVT and no signs or symptoms of other end-organ effects, safe discharge and outpatient follow-up can be considered.
Patients with new SVT requiring cardioversion or those with signs of ongoing cardiac instability should be admitted to a pediatric cardiologist in an intensive care setting. Refractory SVT requires urgent cardiology consultation and admission for further diagnostics and management. In addition, patients who have intercurrent illness, such as febrile illness or upper respiratory infections, may be at increased risk for recurrence. Therefore, in these children, especially those without reliable follow-up care, admission to pediatric cardiology for continued monitoring should be considered.
Accessory Pathways. The most common accessory pathways are atrioventricular, resulting in direct conduction outside of the usual AV-node and His-Purkinje system. Wolff-Parkinson-White (WPW) syndrome is an accessory pathway with bidirectional conduction and presents unique challenges in emergency management, as patients often present with narrow complex SVT, but can also present with life-threatening pre-excited atrial fibrillation.
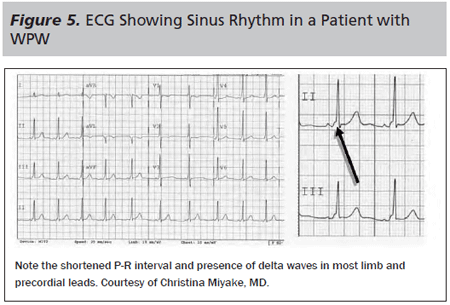
WPW is a syndrome characterized by ventricular pre-excitation through an accessory pathway directly from the atria to the ventricles, identified as a cause of fatal dysrhythmias and sudden death among young children and adolescents.15 The presence of a pre-excitation pattern will affect the choice of anti-arrhythmics. Children who develop a tachyarrhythmia due to electrical re-entry through the accessory pathway may present with a narrow complex SVT. Other children may be diagnosed simply on screening ECG. For patients with WPW who present in a sinus rhythm, the diagnosis is made by the presence of a delta wave and a shortened PR interval on the ECG, as demonstrated in Figure 5. The delta wave is characteristically present as a slurring of the upstroke at the start of the QRS complex, and electrically is accounted for by conduction antegrade (from atria to ventricles) via an accessory pathway separate from the AV node. Patients without septal q waves on lateral precordial ECG leads should raise suspicion for WPW.
In patients diagnosed with WPW, the incidence of reported arrhythmias ranges from 30% to 50%.6,15 When the antegrade conduction through the accessory pathway becomes continuous, the patient's sinus rhythm may become a sustained SVT. Most commonly, patients will present with a narrow QRS-complex tachycardia that is AV re-entrant (AVRT) as conduction occurs down the accessory pathway, then returns to the atria via the AV node. This type of orthodromic conduction is seen in approximately 95% of WPW patients who develop SVT.6 The ECG will characteristically show a narrow complex tachycardia with a regular R-R interval, occasionally but not always accompanied by a visible retrograde p wave after the QRS interval. If the conduction occurs down the AV node and back retrograde through the accessory pathway, known as antidromic conduction, a wide-complex tachycardia will result. Antidromic conduction may be difficult to distinguish from ventricular tachycardia.
Treatment. Any patient with an accessory pathway who presents with a narrow-complex SVT (or AVRT) and is stable may be treated with initial vagal maneuvers, and if not responsive, a trial of adenosine may be attempted. If still unresponsive, consultation with a pediatric cardiologist should be obtained and careful trial of other agents or synchronized electrical cardioversion may be pursued. After conversion to sinus rhythm, patients with WPW should not be discharged home on any AV nodal blocking agents, which include digoxin or calcium channel blockers.
Pitfall. Wide Complex, Irregular Tachycardias. Occasionally, patients with WPW may develop pre-excited atrial fibrillation. The rhythm will have wide QRS complexes, with an irregular R-R interval, and may have heart rates of 250-300 bpm. Atrial fibrillation with a rapidly conducting bypass tract may lead spontaneously to degeneration into ventricular fibrillation.
Missing the presence of an irregular, wide-complex tachycardia can have dangerous consequences, as these patients are more likely to develop fatal rhythms such as ventricular fibrillation if conduction via the AV node is blocked. Whereas calcium channel blockers are often first-line treatments for adults with atrial fibrillation, among children and adults with WPW and pre-excited atrial fibrillation, AV nodal-blocking agents including adenosine, digoxin, and calcium channel blockers are contraindicated and should be strictly avoided. Instead, one can either attempt pharmacologic cardioversion using IV procainamide or amiodarone if the patient is stable or, if the patient is unstable, synchronized DC cardioversion should be performed.
Disposition. Patients presenting with these complex tachycardias or associated symptoms (for example, syncope) and found to have WPW based on ECG characteristics should be admitted for further evaluation and treatment. For patients who are found to have WPW and narrow-complex SVT with no history of syncope and who remain in a sinus rhythm, pediatric cardiology consultation is also recommended, although this can be performed in an outpatient setting. It is not unreasonable to consider a cardiology consult in the emergency room if the ED physician needs assistance in counseling the family, particularly in terms of sports restrictions once discharged and the need for long-term follow-up by a cardiologist.
It is important to fully document the abnormal rhythm in the patient's chart and to provide this information to pediatric cardiology consultants. Often, the cardiologist will want to review the abnormal ECG during follow-up visits and before pursuing long-term management options.
Ventricular Tachycardia (VT). Ventricular tachycardia in pediatrics is strictly defined as three or more consecutive ventricular beats on ECG. It is considered sustained if the rhythm lasts longer than 30 seconds, and this indicates likely need for intervention.10,16 ECG diagnosis of VT is characterized by the presence of a regular rhythm with wide QRS complex, as defined by age-appropriate criteria. (See Table 2.) Note, for example, that infants can have QRS of .08 and have VT. Monomorphic VT is described as the presence of uniform wide complexes with dissociation of the QRS from the p waves. Polymorphic VT may have varied QRS shape and width, as is seen with the classic torsades de pointes rhythms.9,16 VF is characterized by an ECG waveform that is chaotic and unpredictable with variable amplitude and direction and no identifiable P waves or QRS complexes. This rhythm stems from uncoordinated ventricular contraction. In both VF and VT, children who have underlying congenital or structural heart disease are predicted to have poor outcomes.2,16
While the diagnosis of VT may be more subtle due to age-related parameters, the management is similar to that of adults. However, the tragedies in pediatric populations lie either in not recognizing patient presentations that are associated with recurrent episodes of VT or missed diagnosis in patients presenting multiple times. Consider dysrhythmias in patients who have recurrent syncope with exercise or repeated visits for syncope. Patients may also reveal a family history of sudden cardiac death or even unexplained deaths at young ages. As previously discussed, syncope may be mistaken for seizure, especially in children who are febrile, and thus a screening ECG can help to avoid missing a cardiac etiology. Here, we will concentrate on two etiologies of VT: long QT syndrome and catecholaminergic polymorphic ventricular tachycardia.
Long QT Syndrome. Just as patients with pre-excitation syndromes are at increased risk for development of re-entrant rhythms, certain congenital conduction abnormalities can predispose patients to the development of VT. A prolonged QT interval carries an increased risk of torsades de pointes. The QT interval may be prolonged due to congenital long QT syndrome or due to an acquired long QT syndrome, such as from medication effects (i.e., antibiotics such as quinolones, antipsychotic medications, and even some anti-emetics such as ondansetron). Patients may present to the ED with the chief complaint of palpitations, but more commonly may present after syncope or seizure. Therefore, a screening ECG should be evaluated for possible QT prolongation, defined as a corrected QT interval greater than 450 msec (QTc = QT interval / √ preceding R – R interval).9,17
In patients for whom congenital long QT syndrome is suspected, pediatric cardiology consultation is warranted. These patients may benefit from either long-term management with beta-blockers or possible ICD placement.17 For patients who have acquired causes of a prolonged QT, the underlying condition should be addressed, either by discontinuing offending agents or correcting associated imbalances, such as hypocalcemia.
Catecholaminergic Polymorphic Ventricular Tachycardia (CPVT). A rarer but important cause of VT is catecholaminergic polymorphic ventricular tachycardia. While the overall incidence of CPVT is very low and definitive diagnosis can sometimes be difficult, the condition is important to consider, as the presence of CPVT portends poor clinical outcomes. Studies of various etiologies of VT have suggested that once diagnosed, CPVT carries a worse prognosis than other forms of VT, with an eight-year incidence of cardiac arrest of 58% in one untreated group18 and 10-year survival rates of 55% in a separate cohort of CPVT patients.19 The condition is thought to be due to a mutation in the ryanodine receptor gene and is associated with sudden onset of ventricular tachycardia developed in the setting of emotional or physical stress.
Patients may present to the ED after a syncopal episode that occurred after adrenaline release from either an emotional or exertional stressor. Occasionally, patients may identify a history of multiple syncopal episodes that were seemingly unrelated, but in retrospect occurred in the setting of emotional or physical stressors. Figure 6 provides an example of VT precipitated by an IV start in a patient with CPVT.
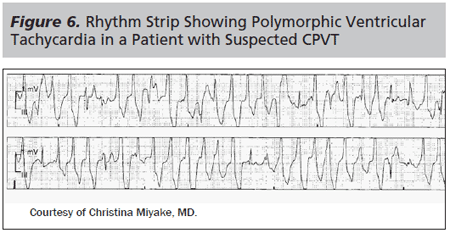
A history of syncope in a child younger than 5 years of age, syncope or arrhythmias during adrenergic or emotional stressors, or syncope with exertion should always raise suspicion of CPVT and warrant further investigation. Family history of unexplained sudden death or cardiac arrest is an important historical feature as, unfortunately, sudden cardiac death may be the only presentation for some patients.
The VT itself should be treated using standard PALS guidelines, including defibrillation for unstable patients and anti-arrhythmics. If CPVT is truly suspected, IV beta-blockers and sedation should be initiated. DC cardioversion should be attempted but may not always successfully terminate the VT and, in fact, may increase catecholamines that may trigger more arrhythmias. In general, however, once the patient degenerates into a hemodynamically unstable ventricular rhythm, DC cardioversion is effective. Any patient in whom CPVT is suspected should have urgent pediatric cardiology consultation and admission for monitoring and treatment.
Myocarditis. Myocarditis is a spectrum of disease that can occur due to infection (commonly viral etiologies) or other infiltrative processes, such as autoimmune diseases, and may range anywhere from an asymptomatic, subacute process to fulminant disease associated with cardiovascular collapse. Acute and fulminant forms of infective myocarditis are among the most lethal, and commonly cause arrhythmogenic events or cardiac arrest.19,20 Most patients with fulminant myocarditis and arrhythmia present with either ventricular tachycardia or heart block.
Diagnosis. Delayed diagnosis can be tragic for any patient with myocarditis, as late presentation increases the risk of fatal arrhythmias, heart failure, and circulatory collapse. Among children presenting with sudden cardiac death, myocarditis was implicated in 17% of cases in one study.21 While the definitive diagnosis is made based on myocardial biopsy, for the emergency physician, the diagnosis must be considered based on clinical history and may be supported by diagnostic testing. Myocarditis should be assumed in any patient presenting with new VT or heart block in the setting of preceding illness.
Often the course in older children includes reported chest pain, fatigue, and shortness of breath in the setting of recent viral symptoms. In older children, vague abdominal pain and nausea are commonly reported.22 However, nonverbal children may simply present nonspecifically with fatigue, dyspnea, or difficulty feeding. Physical findings include vital signs suggesting significant infection with possible signs of respiratory distress, including nasal flaring or retractions. The exam may also reveal epigastric or right upper quadrant tenderness that could be associated with liver congestion in the setting of CHF.
Laboratory testing to identify inflammatory markers, including C-reactive protein, brain natriuretic peptide, troponins, and liver function tests, may be elevated. A chest X-ray is recommended for any patient suspected of having myocarditis, as it can suggest the presence of cardiomegaly or potential pericardial effusion, reveal pulmonary edema, or suggest possible alternate etiologies of the patient's presentation. As bedside ultrasonography becomes more common in the ED, an echocardiogram should be obtained to evaluate for the presence of an effusion, dilated chambers, or myocardial dysfunction.
Treatment and Disposition. Those patients suspected of having myocarditis should be treated with utmost caution. Dysrhythmia and cardiovascular collapse can occur with any intervention because inflammation causes abnormal and unpredictable conduction of electricity through the heart muscle. Dilated cardiomyopathy is also associated with infective myocarditis and can be another cause of arrhythmia due to enlarged atria and abnormal conduction through the structurally damaged heart.21
Patients should have adequate IV access, including consideration of placing large, central catheters should the need for ECMO or other circulatory support arise. Both ventricular assist devices and extra-corporeal membrane oxygenation (ECMO) have been shown to have direct benefits while the muscle recovers or as a bridge to transplantation in patients with acute and fulminant myocarditis.23-25
Conclusion
The incidence of pediatric arrhythmias is relatively low, but missed diagnosis may have significant consequences for a child. Emergency providers should understand the specific challenges of treating rhythm disturbances in children, especially in cases where it differs from treatment of adult arrhythmias.
References
1. Sacchetti A, Moyer V, Baricella R, et al. Primary cardiac arrhythmias in children. Pediatr Emerg Care 1999;15(2):95-98.
2. Marx JA, Hockberger RS, Walls RM, et al. Rosen's Emergency Medicine: Concepts and Clinical Practice, 6th ed. Philadelphia: Mosby/Elsevier; 2006.
3. Kleinman ME, Chameides L, Schexnayder SM, et al. Part 14: Pediatric Advanced Life Support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122(18 Suppl 3): S876-908.
4. Schlechte EA, Boramanand N, Funk M. Supraventricular tachycardia in the pediatric primary care setting: Age-related presentation, diagnosis, and management. J Pediatr Health Care 2008;22(5): 289-299.
5. Kantoch MJ. Supraventricular tachycardia in children. Indian J Pediatr 2005;72(7):609-619.
6. Tintinalli JE, Stapczynski JS. Emergency Medicine: A Comprehensive Study Guide, 7th ed. New York: McGraw-Hill; 2011: http://www.accessemergencymedicine.com/resourceToc.aspx?resourceID=40. Accessed.
7. Evans WN, Acherman RJ, Mayman GA, et al. Simplified pediatric electrocardiogram interpretation. Clin Pediatr (Phila) 2010;49(4):363-372.
8. O'Connor M, McDaniel N, Brady WJ. The pediatric electrocardiogram. Part I: Age-related interpretation. Am J Emerg Med 2008;26(2):221-228.
9. Doniger SJ, Sharieff GQ. Pediatric dysrhythmias. Pediatr Clin North Am 2006;53(1):85-105, vi.
10. O'Connor M, McDaniel N, Brady WJ. The pediatric electrocardiogram part II: Dysrhythmias. Am J Emerg Med 2008;26(3):348-358.
11. Hay WW. Current Diagnosis and Treatment, Pediatrics, 19th ed. New York London: McGraw-Hill Medical; McGraw-Hill [distributor]; 2009.
12. Manole MD, Saladino RA. Emergency department management of the pediatric patient with supraventricular tachycardia. Pediatr Emerg Care 2007;23(3):176-185; quiz 186-179.
13. Davies P, Maconochie I. The relationship between body temperature, heart rate and respiratory rate in children. Emerg Med J 2009;26(9):641-643.
14. Hanna CM, Greenes DS. How much tachycardia in infants can be attributed to fever? Ann Emerg Med 2004;43(6): 699-705.
15. Santinelli V, Radinovic A, Manguso F, et al. The natural history of asymptomatic ventricular pre-excitation a long-term prospective follow-up study of 184 asymptomatic children. J Am Coll Cardiol 2009;53(3):275-280.
16. Biondi EA. Focus on diagnosis: Cardiac arrhythmias in children. Pediatr Rev 2010;31(9):375-379.
17. Delaney J, Mittal S, Sherrid MV. Current perspectives on congenital long QT syndrome. Anadolu Kardiyol Derg 2009;9 Suppl 2:3-11.
18. Hayashi M, Denjoy I, Extramiana F, et al. Incidence and risk factors of arrhythmic events in catecholaminergic polymorphic ventricular tachycardia. Circulation 2009;119(18):2426-2434.
19. Song MK, Baek JS, Kwon BS, et al. Clinical spectrum and prognostic factors of pediatric ventricular tachycardia. Circ J 2010;74(9):1951-1958.
20. Sharma JR, Sathanandam S, Rao SP, et al. Ventricular tachycardia in acute fulminant myocarditis: Medical management and follow-up. Pediatr Cardiol 2008;29(2):416-419.
21. Durani Y, Giordano K, Goudie BW. Myocarditis and pericarditis in children. Pediatr Clin North Am 2010;57(6): 1281-1303.
22. Amieva-Wang NE. A Practical Guide to Pediatric Emergency Medicine, 1st ed. New York: Cambridge University Press; 2011.
23. Duncan BW, Bohn DJ, Atz AM, et al. Mechanical circulatory support for the treatment of children with acute fulminant myocarditis. J Thorac Cardiovasc Surg 2001;122(3):440-448.
24. Wilmot I, Morales DL, Price JF, et al. Effectiveness of mechanical circulatory support in children with acute fulminant and persistent myocarditis. J Card Fail 2011;17(6):487-494.
25. Nahum E, Dagan O, Lev A, et al. Favorable outcome of pediatric fulminant myocarditis supported by extracorporeal membranous oxygenation. Pediatr Cardiol 2010;31(7):1059-1063.
While rhythm disturbance may be a common presenting complaint among adult emergency department (ED) patients, the incidence of cardiac dysrhythmia among pediatric patients is relatively low. In one retrospective review, primary cardiac arrhythmias were identified in 13.9 per 100,000 pediatric ED visits.1 The incidence of these dysrhythmias peaked during infancy and then again in adolescence.1 Cardiac dysrhythmias in children may be due to primary conduction abnormalities or may occur in the setting of structural heart disease, metabolic derangements from toxic ingestions, or infections. Supraventricular tachycardias (SVT) represent the most common pediatric dysrhythmias in adolescents (an estimated 63% of all documented tachycardias).1 After a brief review of initial emergency management of dysrhythmia, the authors will emphasize important pediatric ECG parameters and how they differ from adults.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
