Drug Criteria & Outcomes: Iron Complex Evaluation
Drug Criteria & Outcomes: Iron Complex Evaluation
By Bradley Gilchrist, PharmD, Clinical Pharmacist; Nathan Holder, PharmD, Clinical Pharmacist; and Richard Cramer, PharmD, Drug Information Coordinator, Department of Pharmacy, Huntsville (AL) Hospital
Product Introduction
- Iron sucrose injection (Venofer) is dissociated by the reticuloendothelial system into iron and sucrose. The iron is transferred from the blood into iron stores that are located in the liver and bone marrow. The iron binds with ferritin and then is available for use by the body.
- Sodium ferric gluconate complex (Ferrlecit), referred to as iron gluconate, is a stable macro- molecular complex. The iron is more readily available for use because the complex does not have to be degraded.
- Iron dextran (DexFerrum/Infed) is absorbed after injection into the capillaries and the lym- phatic system. Circulating iron dextran is removed from the plasma by the reticuloen- dothelial system where the complex is split into iron and dextran. The iron binds to fer- ritin and then is available to replenish the depleted iron stores.
Indications
- Iron sucrose — treatment of iron deficiency in patients who receive chronic hemodialysis and supplemental erythropoietin therapy.
- Iron gluconate — treatment of iron deficiency in patients who receive chronic hemodialysis and supplemental erythropoietin therapy.
- Iron dextran — treatment of microcytic hypo- chromic anemia resulting from iron deficiency in which oral administration is infeasible or ineffective.
Contraindications
- Iron sucrose: Patients with evidence of iron overload, known hypersensitivity to iron sucrose or any of its inactive compounds, and anemia not caused by iron deficiency.
- Iron gluconate: Anemia not associated with iron deficiency, and known hypersensitivity to sodium ferric gluconate complex or any of its inactive compounds.
- Iron dextran: Hypersensitivity to iron dex- tran, all anemias that are not involved with iron deficiency, and hemochromatosis.
Dosage and administration
1. Iron sucrose
- Of three U.S. studies used to evaluate iron sucrose, two did not require patients to receive a test dose. The third required a test dose of 2.5 mL (50 mg) diluted in a 50 mL normal saline (NS) infusion over three to 10 minutes.
- The recommended dosage for repletion therapy for iron deficiency in hemodialysis patients is 5 mL iron sucrose (equivalent to 100 mg elemental iron) given by intravenous (IV) injection over five minutes or infusion over 15 minutes during the dialysis session. Doses can be given no more than three times per week. Give a minimum of 10 doses to achieve 1,000 mg elemental iron.
- Iron sucrose must be
injected directly into the dialysis line by slow injection or infusion.
— Slow IV injection: Iron sucrose may be administered directly into the dialy- sis line at a rate of 1 mL (20 mg iron) undiluted solution per minute (five minutes per vial) not exceeding one vial (100 mg) per injection.
— Infusion: Iron sucrose may be admin- istered by infusion (into the dialysis line for hemodialysis patients), which can reduce the risk of hypotension. Each vial must be diluted in 100 mL NS, immediately prior to infusion, and given over at least 15 minutes.
2. Iron gluconate
- Test dose currently is not recommended.
- Recommended dose for repletion of iron in patients on hemodialysis is 125 mg (10 mL) in 100 mL NS over one hour in hemodialy- sis. Most patients require a cumulative dose of 1 g elemental iron over approximately eight sequential dialysis treatments to achieve a favorable response.
- Iron gluconate may be administered either as an infusion or as a bolus or IV push at a rate no faster than 12.5 mg/min.
3. Iron dextran
- IV or intramuscular (IM) iron dextran test dose (given prior to initial dose).
- Dosages:
— Dose (mL) = 0.0442 (Desired hemoglobin (Hb)-Observed Hb) × lean body weight (LBW) + (0.26 × LBW).
— Iron replacement for blood loss: Replace- ment iron (mg) = blood loss (mL) x hematocrit.
- Test dose: Patients should be given a test dose of 0.5 mL for IV or IM iron dextran. The IV dose should be given gradually over 30 seconds. The manufacturer recommends that an hour should pass before administra- tion of the rest of the dose.
- IV bolus may be given at rate of < 50 mg/min or diluted in 250-1,000 mL NS and infused over one to six hours.
Note: Iron sucrose and iron gluconate cannot be given as total drug infusions (i.e., their doses must be separated over several visits).
Efficacy
- Iron sucrose — Iron sucrose has successfully raised hemoglobin and hematocrit in several small studies, mostly against placebo. One study comparing IV iron sucrose to oral fer- rous sulfate showed that patients who received IV iron sucrose had a better response than the oral iron group. Almost all studies took place in patients who were receiving maintenance hemodialysis.
- Iron gluconate — Iron gluconate has effec- tively raised hemoglobin and hematocrit lev- els vs. oral iron therapy. These studies were well-controlled large trials that included mostly iron-deficient hemodialysis patients. In one study, after one month, iron gluconate and oral iron increased hematocrit by 3.8% and 0.2% and hemoglobin by 1.3% and 0.4%, respectively.
- In the one study comparing iron sucrose and iron gluconate, the two iron products had rela- tively the same effect on hemoglobin in patients who also were on epoetin. This study used doses that were different from what is recom- mended by the manufacturer for both products.
- Iron dextran — Iron dextran has proven effec- tive in patients who are iron-deficient. It has been effective in various populations, not only those undergoing hemodialysis or who have end-stage renal disease.
Safety
The safety profile of iron dextran is dependent upon its administration (see Table 1, below). When given as a daily IM or IV injection (100 mg), the adverse effect profile is quite low. The IV bolus infusion is associated with a greater frequency of side effects. Serious side effects can be reduced with the administration of a test dose.
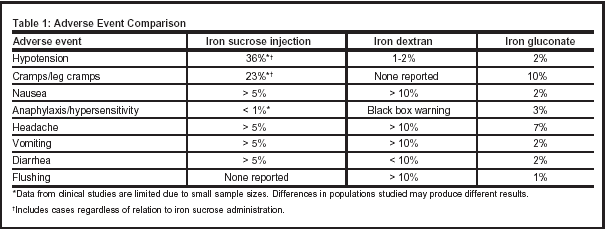
There has been recent concern that iron sucrose is more often associated with increased risk of infection. Recent studies have demonstrated the promotion of bacterial growth in iron-overloaded patients attributed to free, unbound iron in the serum. Presence of free iron in the serum has also been identified as a source of inhibition of neutrophil killing capacity. Other trials, however, have not been able to demonstrate an association between supersaturated transferrin or IV iron administration and risk of infection.
Drug/lab and drug/drug interactions
- Parenteral iron preparations should not be administered with oral iron preparations because the absorption of oral iron is decreased.
- Drug interactions with iron gluconate have not been studied.
- Iron dextran has a decreased effect when used with chloramphenicol.
- Iron dextran can cause falsely elevated serum bilirubin levels and falsely decreased serum calcium levels.
- Iron dextran can interfere with bone scan tests. After an infusion of iron dextran, bone scans have been reported to show a reduction of bony reuptake, marked renal activity, and excessive blood pool and soft-tissue accumulation.
- Do not mix parenteral iron preparations with other medications or add to parenteral nutri- tion solutions.
- Drug interactions would be expected to be similar among parenteral iron products.
Monitoring parameters
- Monitor hematologic parameters and iron indices (hemoglobin, hematocrit, transferrin saturation, and ferritin).
- Maintain transferrin saturation between 20% and 50%.
- Withhold iron therapy if ferritin level is greater than 800 ng/mL or if transferrin saturation is 50% or more.
- Serum iron values may be obtained 48 hours after IV dosing.
- Goals of therapy include a target hemoglobin level of 11.0-12.0 g/dL, hematocrit level of 33%-36%, transferrin saturation 20% or more, and ferritin 100 ng/mL or more.
Comparison
Iron dextran may be given as a total daily dose due to its slow release. Iron sucrose and iron gluconate cannot be given as a total daily dose because they release iron more rapidly. Iron sucrose and iron gluconate do not require a test dose (physician’s discretion), whereas iron dextran does require a test dose before administration. Iron gluconate, iron sucrose, and iron dextran each have had reports of anaphylactic reactions; iron dextran carries a black box warning as a result.
Iron gluconate is more expensive per dose than iron sucrose, but the amount of elemental iron in each dose of iron gluconate is greater than that in iron sucrose (see Table 2, below). Iron dextran is a relatively inexpensive iron product compared to the other two products ($215 per 1,000 mg dose).

Iron dextran has been used safely and efficaciously for years and can be administered as a single daily dose. Additionally, it is significantly less expensive than the two newer agents. However, iron dextran is associated with a slightly higher incidence of allergic reactions, including life-threatening anaphylaxis. When comparing the other two IV products, we also must examine the efficacy, cost, and nursing time. Iron gluconate is typically given as a series of eight 125 mg injections, while iron sucrose is given as a series of ten 100 mg injections. The increased number of injections required for iron sucrose could increase cost even more due to increased nursing time and extra patient visits. Also, iron gluconate appears to have more studies supporting its efficacy than iron sucrose.
Recommendation
Iron sucrose and iron gluconate have both been used as alternative parenteral iron products at Huntsville Hospital for use in patients who are not able to receive iron dextran, such as those patients with a known allergy to it. Based on available data, iron gluconate and iron sucrose seem to be similar iron replacement products. The results found are not completely reliable due to the small sample sizes of each study.
Both iron sucrose and iron gluconate have been used without major administration problems in this hospital and the regional outpatient setting. Recently, iron gluconate has been used much more than iron sucrose in this region. Because of increased use of the product and other reasons discussed in this evaluation, iron gluconate is the hospital formulary agent and will be used in place of iron sucrose. Iron dextran will continue to be an IV iron formulary agent.
Resources
• American Regent Laboratories.
Venofer Formulary Monograph. Shirley, NY; 2003;1-32.
• American Regent Laboratories. Venofer Package Insert. Shirley, NY;
2003.
• Schein Pharmaceutical. InFeD Package Insert. Florham Park, NJ; 2003.
• Watson Pharma. Ferrlecit Package Insert. Florham Park, NJ; 2003.
• Iron sucrose. Drug Facts and Comparisons. St. Louis: Facts and Comparisons;
2001:36a-36b.
• Iron dextran. Drug Facts and Comparisons. St. Louis: Facts and Comparisons;
2000:33-35.
• Sodium ferric gluconate complex. Drug Facts and Comparisons. St. Louis:
Facts and Comparisons; 2000:35-36.
• Personal communication. Cindy Hall, Buyer. Huntsville (AL) Hospital System;
February 2004.
• Charytan C, Levin N, Al-Saloum M, et al. Efficacy and safety of iron sucrose
for iron deficiency in patients with dialysis associated anemia: North American
clinical trial. Clinical study report, 1999. Am J Kidney Dis 2001;37:300-307.
• Van Wyck DB, Cavallo G, Spinowitz BS, et al. Safety and efficacy of iron sucrose
in patients sensitive to iron dextran: North American clinical trial. Am
J Kidney Dis 2000;36:88-97.
• Silverberg DS, Blum M, Agbaria Z, et al. Intravenous ferric saccharate as
an iron supplement in dialysis patients. Nephron 1996;72:413-417.
• Van Zyl-Smit R, Moosa MR, Potgieter CD, et al. A multicentre study to investigate
the tolerance, safety, and efficacy of intravenous iron sucrose in hemodialysis
patients with anemia. Clinical study report, 1997 and data on file: American
Regent Laboratories; Shirley, NY. Abstract in Kidney Int 1999;55:2130.
• Macdougall IC, Chandler G, Elston O, et al. Beneficial effects of adopting
an aggressive intravenous iron policy in a hemodialysis unit. Am J Kidney
Dis 1999;34:S40-S48.
• Silverberg DS, Iaina A, Peer G, et al. Intravenous iron supplementation for
the treatment of the anemia of moderate to severe chronic renal failure patients
not receiving dialysis. Am J Kidney Dis 1996;27:234-238.
• Hussain R, Christi SH, Navqi S. Experience of iron saccharate supplementation
in haemodialysis patients treated with erythropoietin. Nephrology 1998;4:105-108.
• Iron dextran. Drug information handbook. Hudson (Cleveland): Lexi-Comp; 2000:643-644.
• Ferric gluconate. Drug information handbook. Hudson (Cleveland): Lexi-Comp;
2000: 688-689.
• Patruta SI, Edlinger R, Sunder-Plassman G, et al. Neutrophil impairment associated
with iron therapy in hemodialysis patients with functional iron deficiency.
J Am Soc Nephrol 1998;9:655-663.
• Parkkinen J, von Bonsdorff L, Peltonen S, et al. Catalytically active iron
and bacterial growth in serum of hemodialysis patients after IV iron-saccharate
administration. Nephrol Dial Transplant 2000;15:1827-1834.
• Deicher R, Ziai F, Cohen G, et al. High-dose parenteral iron sucrose depresses
neutrophil intracellular killing capacity. Kidney Intl 2003;64:728-736.
• Hoen B, Paul-Dauphin A, Hestin D, et al. EPIBACDIAL: A multicenter prospective
study of risk factors for bacteremia in chronic hemodialysis patients. J
Am Soc Nephrol 1998;9:869-876.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
