Drug Criteria & Outcomes: Telithromycin (Ketek) Formulary Evaluation
Drug Criteria & Outcomes
Telithromycin (Ketek) Formulary Evaluation
Part 2: Clinical Trials, Adverse Events, Drug Interactions, and Recommendation
By Jennifer Herring,
PharmD Candidate
Harrison School of Pharmacy
Auburn (AL) University
Clinical Studies
Definitions used in study descriptions:
- Modified intent-to-treat (mITT) — All patients who were randomized and received at least one dose of study medication.
- Per protocol population (PPc) — Patients who completed therapy without major protocol violation and/or an indeterminate response.
- Bacteriologic mITT (bmITT) — Patients in mITT group with causative pathogen isolated at pretherapy/entry visit.
- Per protocol population for analysis of bacteriological outcome (PPb) — Patients in PPc group with a causative pathogen isolated at pre therapy/entry.
- Pre-therapy/entry — Day 1.
- On-therapy — Days 3-5.
- End of therapy — Days 11-13.
- Post-therapy/Test of Cure (TOC) — Days 17-24.
- Late post-therapy — Days 31-45.
Acute bacterial exacerbation of chronic bronchitis (AECB)
A multicenter, double-blind study evaluating the efficacy and safety of oral telithromycin 800 mg daily for five days vs. amoxicillin/clavulanic acid (AMC) 500 mg/125 mg tid for 10 days for the treatment of AECB found that telithromycin is an effective and well-tolerated alternative to AMC. Clinical cure rates for telithromycin at TOC and late post-therapy were 86.1% and 78.1%, respectively. For AMC, cure rates were 82.1% (TOC) and 75.0% (late post-therapy). Both treatments were well tolerated with similar incidences of adverse drug reactions. The most common adverse events included diarrhea, dyspepsia, and vomiting (telithromycin 13.3%, AMC 25.0%). Clinically noteworthy abnormal laboratory values observed in both groups (telithromycin 11.3%, AMC 15.6%) were increased prothrombin time and international normalized ratio (INR), and decreased creatinine clearance.
Telithromycin 800 mg once daily for five days also has been compared to cefuroxime axetil and clarithromycin, each at 500 mg bid for 10 days. Similar results with efficacy and safety were noted in each of these trials as well. Again, diarrhea and nausea were the most common adverse events. Similar results were found in the mITT group for AMC; results for the cefuroxime axetil and clarithromycin mITT groups were not available. Of interest, the trial vs. clarithromycin was not published, possibly because telithromycin did not perform as well as hoped against a similar drug.
Acute bacterial sinusitis (ABS)
Telithromycin, five- and 10-day treatment, was shown to be equally efficacious in the treatment of acute bacterial sinusitis (ABS) vs. AMC in a randomized, double-blind, three-arm, parallel-group, multicenter trial. The primary efficacy analysis (TOC) showed that both telithromycin and AMC achieved a clinical cure in approximately 75% of patients, and at late post therapy 70%. The most common pathogens isolated were S. pneumoniae and H. influenzae, accounting for 55% of the total. Cure rates for those patients who underwent sinus puncture were 85.7%, 85.7%, and 80% for the five-day telithromycin, 10-day telithromycin, and AMC, respectively. Safety and tolerability also were equivalent between the groups, with 44% of the patients experiencing one or more treatment-emergent adverse events possibly related to study medication. Diarrhea and nausea were the most common adverse events.
Telithromycin also was compared to cefuroxime axetil for the treatment of ABS. Clinical cure rates at the post-therapy/TOC visit in the PPc patients demonstrated clinical equivalence: 85.2% and 83% for telithromycin and cefuroxime, respectively. These results were supported by the mITT groups (80.4% for telithromycin and 72.4% for cefuroxime axetil), where indeterminate outcomes were considered treatment failures. Discontinuation of treatment due to adverse events was comparable between the groups at 2% for telithromycin and 1.7% for those patients receiving cefuroxime axetil. Diarrhea and nausea were the most common adverse events.
S. pneumoniae and H. influenzae are the pathogens most often implicated in ABS, accounting for 27%-70% worldwide. Within the PPb group of the cefuroxime axetil study, telithromycin successfully eradicated 89.7% and 81.3% of these pathogens, respectively, whereas cefuroxime axetil eradication rates were 100% and 85.7%, respectively.
Furthermore, in patients with penicillin- or erythromycin-resistant isolates of S. pneumoniae, telithromycin achieved a clinical cure rate of 83.3% vs. cefuroxime axetil, which achieved 100.0%. Clinical cure rates for isolates that were penicillin- and erythromycin-resistant remained at 100% for cefuroxime axetil. However, they fell to 66.7% in the telithromycin-treated isolates. However, sample size of these populations was small (24 total patients).
These results are comparable to telithromycin cure rates for S. pneumoniae without regard to penicillin and erythromycin susceptibility. See Table 4, below for telithromycin cure rates.
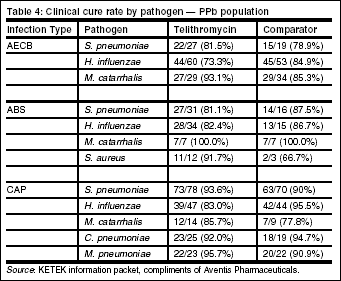
Community-acquired pneumonia (CAP)
In a randomized, double-blind, parallel-group clinical trial, telithromycin 800 mg once daily for seven to 10 days was not found to be inferior to trovafloxacin 200 mg once daily for seven to 10 days in the treatment of CAP. The study terminated prematurely in July 1999 when the FDA restricted use of trovafloxacin to hospitalized patients with life- or limb-threatening infection because of post-marketing reports of hepatic toxicity. Due to the early termination, the study was inadequately powered secondary to small sample size and equivalence could not be demonstrated.
At conclusion of the study, cure rates in the PPc population for the telithromycin and trovafloxacin groups were similar: 90% and 94.2%, respectively. The single significant difference between groups was number of smokers (44/104) randomized to receive trovafloxacin (vs. 25/100 in the telithromycin group). Clinical cure rates between the subgroups, however, were comparable at 95% and 97.3% for telithromycin and trovafloxacin, respectively. Atypical organisms (M. pneumoniae and C. pneumoniae) were causative agents in 31 patients in this study. Both telithromycin (14/15) and trova-floxacin (15/16) achieved excellent clinical cure in these patients.
Treatment-emergent adverse events reported during the study were typically mild. A statistically significant difference was noted in the occurrence of diarrhea, 18.5% for telithromycin and 6.4% for trovafloxacin. In terms of safety, abnormal liver function tests (2.8%), increased aspartate aminotransferase (1.9%), and decreased creatinine clearance (1.9%) were reported in the telithro-mycin group, whereas increased creatinine clearance (1.8%) was reported with trovafloxacin. Although this study was terminated early due to hepatotoxicity associated with trovafloxacin, higher incidence of abnormal liver function tests actually occurred in the telithromycin-treated group. Although the LFTs returned to normal range by the last drawn lab, it would be wise to limit the use of telithromycin in those patients at risk for hepatic dysfunction until further studies have confirmed its safety in this patient population.
Telithromycin 800 mg daily for seven to 10 days also was compared with amoxicillin 1,000 mg tid for 10 days and clarithromycin 500 mg bid for 10 days for the treatment of CAP in separate randomized, double-blind studies. Each study assessed more than 300 patients in whom the baseline demographic and prognostic characteristics were similar. Overall cure rates were comparable in the PPc groups in each study and supported by the mITT findings. Adverse events were predominantly gastrointestinal in nature and of mild or moderate intensity. Nausea and diarrhea occurred somewhat more frequently in the telithromycin group.
Clinically noteworthy abnormal laboratory values (more than three times the upper limit of normal) were noted in 4.5% of telithromycin patients and 5.9% of amoxicillin patients. All patients recovered without sequelae.
Adverse events
Like the macrolides, the principal adverse effects of telithromycin are associated with the gastrointestinal tract, including diarrhea and nausea. These adverse events are usually mild to moderate in severity. Headache and dizziness also were associated with telithromycin. More serious but less frequent side effects included abnormal liver function tests, pseudomembranous colitis, hepatitis with or without jaundice, and visual disturbances, some cases severe. Telithromycin is not recommended in patients with myasthenia gravis unless no other therapeutic options are available due to reported exacerbations of the disease.
The manufacturer also recommends that statins be discontinued during telithromycin therapy in those patients being treated with atorvastatin, lovastatin, and simvastatin due to metabolism by the CYP3A4 isoenzyme.
In addition, telithro-mycin has the potential to prolong the QTc interval. It would be wise to limit the use of telithromycin in those patients with ongoing, or who are at increased risk for, QTc interval prolongation, and in those who are being treated with other medications that may prolong the QTc interval.
Drug interactions
Telithromycin is metabolized by the CYP3A4 isoenzyme and is a strong inhibitor of the same (see Table 5, below). Additionally, telithromycin is thought to interact with the 2D6 and possibly the 1A2 isoenzymes. The manufacturer of telithromycin considers concomitant administration with cisapride or pimozide a contraindication due to QTc prolongation.
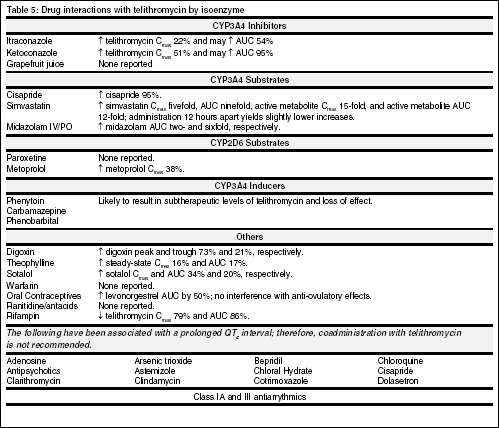
Recommendations
Telithromycin shows promise as a new agent for the empiric treatment of community-acquired respiratory tract infections (RTIs). It has shown significant activity against S. pneumoniae and also appears to be very efficacious against atypical pathogens. In addition, telithromycin neither selects for resistant strains nor induces MLSB resistance; it shows excellent concentration within respiratory tissue and the pharmacokinetics are conducive to once-daily dosing without regard to food.
Though telithromycin is a potentially valuable therapeutic tool for the treatment of community-acquired RTIs, it also has shown a greater propensity to cause serious adverse events vs. similar agents. Until these concerns are better addressed, telithromycin should probably be used as a second-line agent in those who are refractory to treatment, and in those who are intolerant of or have contraindications to traditional first-line agents.
Resources
- Abbott Laboratories. Biaxin [package insert]. North Chicago; 2004.
- Aubier M, Aldons PM, Leak A, et al. Telithromycin is as effective as amoxicillin/clavulanate in acute exacerbations of chronic bronchitis. Respir Med 2002;96:862-971.
- Aventis Pharmaceuticals. KETEK [information packet]. Bridgewater, NJ; 2004.
- Buchanan PP, Stephens TA, Leroy B. A comparison of the efficacy of telithromycin versus cefuroxime axetil in the treatment of acute bacterial maxillary sinusitis. Am J Rhinol 2003;17:369-377.
- Dipiro JT, Talbert RL, Yee GC, et al. Pharmacotherapy: A Pathophysiologic Approach. 5th ed. New York City: McGraw-Hill; 2002.
- Dunbar LM. Current issues in the management of bacterial respiratory tract disease: The challenge of antibacterial resistance. Am J Med Sci 2003;326:360-368.
- Dunbar LM, Hassman J, Tellier G. Efficacy and tolerability of once-daily oral telithromycin compared with clarithromycin for the treatment of community-acquired pneumonia in adults. Clin Ther 2004;26:48-62.
- GlaxoSmithKline. Amoxil [package insert]. Research Triangle Park, NC; 2004.
- GlaxoSmithKline. Augmentin [package insert]. Research Triangle Park, NC; 2004.
- GlaxoSmithKline. Ceftin [package insert]. Research Triangle Park, NC; 2004.
- Hagberg L, Torres A, Rensburg D, et al. Efficacy and tolerability of once-daily telithromycin compared with high-dose amoxicillin for treatment of community-acquired pneumonia. Infection 2002;30:378-386.
- Lutterman M, Tellier G, Lasko B, et al. Efficacy and tolerability of telithromycin for 5 or 10 days vs. Amoxicillin/ clavulanic acid for 10 days in acute maxillary sinusitis. Ear Nose Throat J 2003;82:576-590.
- Pullman J, Champlin J, Vrooman PS. Efficacy and tolerability of once-daily oral therapy with telithromycin compared with trovafloxacin for the treatment of community-acquired pneumonia in adults. Int J Clin Pract 2003;57:377-384.
- Roos K, Brunswig-Pitschner C, Kostrica R, et al. Efficacy and tolerability of once-daily therapy with telithromycin for 5 or 10 days for the treatment of acute maxillary sinusitis. Chemotherapy 2002;48:100-108.
- Shain CS, Amsden GW. Telithromycin: The first of the ketolides. Ann Pharmacother 2002;36:452-464.
- Telithromycin [monograph in electronic version]. MICROMEDEX Healthcare Series. Englewood, CO: MICROMEDEX; 2004.
- Pfizer. Trovan [package insert]. New York City; 2004.
- White RL. Antibiotic resistance: Where do ketolides fit? Pharmacotherapy 2002;22:18S-29S.
- Zervos MJ, Heyder AM, Leroy B. Oral telithromycin 800 mg once daily for 5 days versus cefuroxime axetil 500 mg twice daily for 10 days in adults with acute exacerbations of chronic bronchitis. J Int Med Res 2003;31:157-169.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
