Pill remains powerful force in contraception
Pill remains powerful force in contraception
Take a look at the last 10 patient charts in your outbox. If oral contraceptives (OCs) were prescribed, which ones were selected?
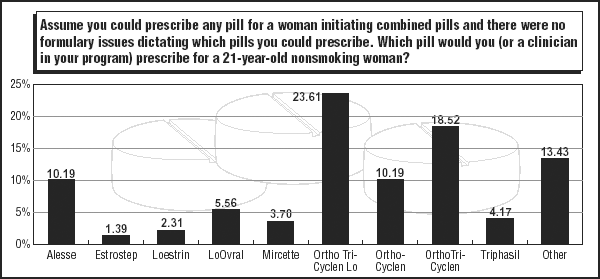
When it comes to prescriptions for younger women, about 24% of respondents to the 2004 Contraceptive Technology Update Contraception Survey say their No. 1 pill of choice is Ortho Tri-Cyclen Lo (Ortho-McNeil Pharmaceutical, Raritan, NJ). Ortho Tri-Cyclen Lo provides a daily dose of 25 mcg estrogen for 21 days and three doses of the progestin norgestimate (180 mcg daily/days 1-7; 215 mcg daily/days 8-14; 250 mcg daily/days 15-21). The last seven days contain no active ingredients. The pill won Food and Drug Administration (FDA) approval in 2002.
The higher-dose Ortho Tri-Cyclen, a 35-mcg ethinyl estradiol phasic pill also marketed by Ortho-McNeil, was named by 18.5% of survey respondents as their top nonformulary pill choice for a 21-year-old woman. While its numbers have dropped from its 22.6% ranking in the 2003 survey, the pill continues as the No. 1 formulary choice for young women. (See graphic on top formulary pills, below.)
As new contraceptive methods enter the marketplace, some family planning providers are seeing a dip in requests for OCs.
"There has been a decline in OC users in the last year," reports Carolyn Brown, RNP, nurse practitioner at Pinal County Health Dept., a public health facility in Florence, AZ. DMPA (Depot medroxyprogesterone acetate, marketed as Depo Provera, Pfizer, New York City) has become very popular, Brown says. Also, the intrauterine device is popular in certain populations, she adds.
However, some providers report that the pill is maintaining or even increasing in popularity. For example, pill prescriptions have increased at the student health center at New Mexico Institute of Mining and Technology, an undergraduate and graduate university in Socorro, reports Susan Lewark, CFNP. About 25% of patients now use OCs, she states.
How are pills used?
Ortho Tri-Cyclen was the first oral contraceptive to receive FDA approval for treatment of acne in women seeking contraception. Many clinicians have become used to requests for "the acne pill" following the 1997 FDA indication for the drug.
Other OCs now are being evaluated for possible treatment of acne.1 Two trials involving Alesse, a monophasic 20-mcg pill from Wyeth-Ayerst Laboratories, Philadelphia, showed total acne improvement of 23% to 40% compared with 9% to 23% with placebo.2,3
Researchers also are eyeing the use of Yasmin, a monophasic pill containing 3.0 mg drospirenone and 0.030 mg ethinyl estradiol, marketed by Berlex Laboratories of Montville, NJ. Findings from a recent study indicate the pill may be effective in reducing acne lesions.4
Pills for older women?
When it comes to pills for older women, which OCs are selected? Survey respondents continue to name Alesse. About 37% of respondents named the pill as their first nonformulary choice for a 42-year-old nonsmoking woman; about 43% listed it as their No. 1 choice in 2003. (See graphic on pill choices for older women, below.)
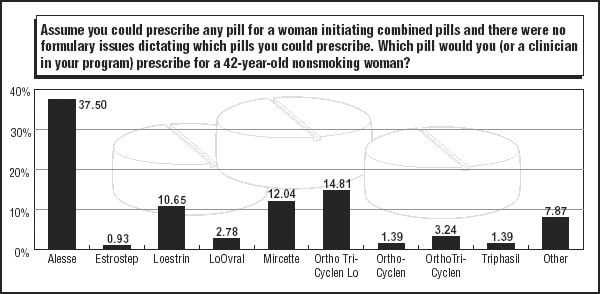
Ortho Tri-Cyclen Lo moved to the No. 2 spot in the category; about 15% named the pill in their 2004 responses. Loestrin, a monophasic 20-mcg pill from Pfizer of New York City, held the secondary position in 2003 with about 16%; its numbers fell to about 11% in 2004.
Extended regimen OK?
Clinicians are beginning to integrate the first FDA-approved extended regimen OC into their practices. Seasonale, marketed by Barr Laboratories of Pomona, NY, is a 91-day regimen taken daily as 84 active tablets of 0.15 mg levonorgestrel/0.03 mg ethinyl estradiol, followed by seven inactive tablets. It is designed to reduce the number of periods from 13 to four per year.
Since its launch in November 2003, more than 260,000 prescriptions have been filled for the drug, according to Barr Labs. About 15% of 2004 CTU survey respondents say they have written prescriptions for the drug in the last six months.
Yasmin also is being evaluated for use in an extended regimen. In a prospective observational six-month study in Germany, cycle control, premenstrual symptoms and general well-being were compared in 1,433 women taking Yasmin in either an extended regimen (63-126 days) or a conventional regimen.5
Fluid retention was reduced in 49% of women on the extended regimen compared to 34% of women on the conventional regimen; 50% of those on extended cycle noted a reduction in breast tenderness, compared to 40% on the short-cycle regimen.
One-third of the women on either regimen noticed an improvement in their skin condition.5
"I have observed few progestational side effects on Yasmin," says Joe Childress, MD, an obstetrician/gynecologist in private practice in San Antonio.
"I like drospirenone, especially for continuous dosing."
References
1. Haider A, Shaw JC. Treatment of acne vulgaris. JAMA 2004; 292:726-735.
2. Thiboutot D, Archer DF, Lemay A, et al. A randomized, controlled trial of a low-dose contraceptive containing 20 microg of ethinyl estradiol and 100 microg of levonorgestrel for acne treatment. Fertil Steril 2001; 76:461-468.
3. Leyden J, Shalita A, Hordinsky M, et al. Efficacy of a low-dose oral contraceptive containing 20 microg of ethinyl estradiol and 100 microg of levonorgestrel for the treatment of moderate acne: A randomized, placebo-controlled trial. J Am Acad Dermatol 2002; 47:399-409.
4. Van Vloten WA, van Haselen CW, van Zuuren EJ, et al. The effect of 2 combined oral contraceptives containing either drospirenone or cyproterone acetate on acne and seborrhea. Cutis 2002; 69(4 Suppl):2-15.
5. Sillem M. Yasmin and the extended regimen: Current experience. Presented at the Eighth Congress of the European Contraception Society. Edinburgh, Scotland; June 2004.
Take a look at the last 10 patient charts in your outbox. If oral contraceptives (OCs) were prescribed, which ones were selected? When it comes to prescriptions for younger women, about 24% of respondents to the 2004 Contraceptive Technology Update Contraception Survey say their No. 1 pill of choice is Ortho Tri-Cyclen Lo (Ortho-McNeil Pharmaceutical, Raritan, NJ).Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
