Acute Abdominal Pain in Special Populations, Part II: Elderly, Immunocompromised, and Pregnant Patients
Part II: Elderly, Immunocompromised, and Pregnant Patients
Authors: Larissa I. Velez, MD, Assistant Professor, Assistant Director of Emergency Medicine Residency Program, University of Texas Southwestern Medical Center, Dallas; Fernando L. Benitez, MD, Assistant Professor, Emergency Medicine, University of Texas Southwestern Medical Center, Dallas; and Salvador E. Villanueva, MD, Assistant Professor, Emergency Medicine, University of Puerto Rico.
Peer Reviewers: J. Stephan Stapczynski, MD, Chair, Emergency Medicine Department, Maricopa Medical Center, Phoenix, AZ; and O. John Ma, MD, Professor and Vice Chair of Emergency Medicine, Truman Medical Center, University of Missouri—Kansas City School of Medicine.
The first part of this series discussed abdominal pain in pediatric patients. This second and final part will cover abdominal pain in elderly, immunocompromised, and pregnant patients. — The Editor
The Older Patient
Those 65 years of age and older constitute the fastest-growing segment of the population, and currently comprise about 12% of the U.S. population.1 This means that abdominal pain in the elderly will be a commonplace occurrence in EDs. (See Table 1 for common causes of abdominal pain.) In the elderly, the history may be more difficult to obtain due to the higher frequency of problems with communication, such as hearing impairment, dementia, and stroke.1 The symptoms in this age group are less specific and often atypical—they tend to present with less pain, less systemic symptoms such as fever, and have fewer laboratory abnormalities such as leukocytosis.1,2 They also tend not to develop signs of peritonitis, namely involuntary guarding and rebound.2
Table 1. Common Causes of Acute Abdominal Pain
The distribution of diagnoses here differs from other segments of the population, and the disease often is more complicated due to co-morbid conditions.1,2 Furthermore, there often is a delay in presentation due to lack of transportation or fear of institutionalization.2 This leads to complicated disease upon presentation.
Van Geloven and colleagues studied 180 elderly patients presenting to the ED with abdominal pain. They constituted 5% of those with abdominal pain in the ED. However, these patients had an 86% admission rate; 27% of these required surgery, and the mortality rate was 17%.3 Although this is a European study, where the utilization of ancillary diagnostic tests such as computed tomography (CT) scan was low (11%), it highlights some of the challenges of diagnosing and managing abdominal pain in the elderly. A recent U.S. study documented a rate of surgery of 22.1% in those 65 years of age and older. The authors found that the presence of hypotension, abnormal abdominal radiographs, leukocytosis, abnormal bowel sounds, and advanced age correlated with an adverse outcome in those 65 and older.4
Bowel Obstruction. Bowel obstruction is a common cause of abdominal pain in the elderly and is the second most common cause for a surgical intervention in this age group.5
Large and Small Bowel Obstruction. Neoplasms are the most common cause of large bowel obstruction (LBO), followed by diverticulitis and volvulus. LBO symptoms typically develop over days. The patients present with abdominal distention, colicky abdominal pain, and obstipation. Vomiting may be absent. The diagnosis of LBO usually is confirmed by an acute abdominal series showing a distended colon with the typical haustral pattern (not crossing the full width of the bowel) and air-fluid levels with more than 20 mm of height difference when measured in the same bowel loop in cases of a mechanical obstruction.6
Adhesions are the most common cause of small bowel obstruction (SBO), followed by hernias and neoplasms.2,7 The patients complain of colicky intermittent abdominal pain. Six clinical variables have a high sensitivity and positive predictive value for the diagnosis of SBO: previous abdominal surgery, history of constipation, age older than 50 years, vomiting, abdominal distention, and increased bowel sounds ("tinkles and rushes").8
The diagnosis can be confirmed with an acute abdominal series showing distended bowel loops that are centrally located and show bands that traverse the entire bowel width (valvulae conniventes). (See Figure 1.) The CT scan will not only confirm the obstruction, but often will show the level of the obstruction and its possible cause.9
Figure 1. Small Bowel Obstruction
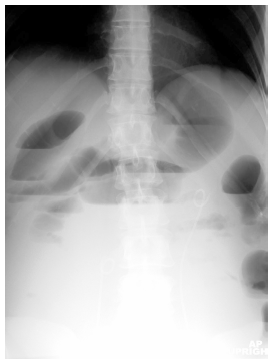
There are multiple distended small bowel loops
with the valvulae connivente. This points to an
SBO. The patient also has a low abdominal
mass and bilateral ureteral stents.
Volvulus. Gastric volvulus is more frequent in the elderly, followed by colonic volvulus. In the geriatric population, sigmoid volvulus is the most common type of colonic volvulus, accounting for 65-80% of cases. It has a 20-40% mortality rate.10 Cecal volvulus is the second most common type of volvulus, accounting for 15-20% of cases. Volvulus of the transverse colon and splenic flexure are rare and comprise only 2-5% of cases. The peak age for presentation is in the 50s, and typically the patients are either debilitated or institutionalized.11,12 Chronic constipation secondary to inactivity and use of neuropsychiatric drugs that alter bowel motility increase the likelihood of volvulus in these patients. The typical presentation is that of chronic constipation, followed by crampy low abdominal pain, progressive abdominal distention, and then vomiting and obstipation. On physical examination, there is marked distention and tympany to percussion. This distention can result in respiratory compromise. Peritonitis can be evident, and usually is due to bowel strangulation.
The diagnosis of sigmoid volvulus usually is confirmed by plain films of the abdomen, which show a single dilated loop of colon on the left half of the abdomen. (See Figure 2.) The barium enema will show the pathognomonic "bird’s beak" appearance.13
Figure 2. Volvulus
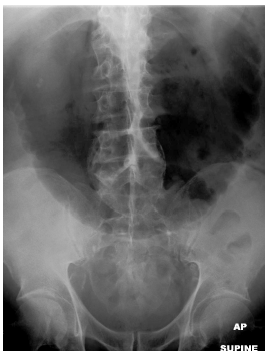
There is a distended large bowel loop in the
mid abdomen, with a coffee bean appearance.
A haustral pattern can be seen. This patient
had a sigmoid volvulus.
The management is mainly supportive, with early surgical consultation. If not strangulated, the sigmoid volvulus often can be reduced using a sigmoidoscope or a barium enema.5 Operative reduction is needed for those with a cecal volvulus, strangulation, or the rare perforation.14
Remember that neoplasms are the most common cause of LBO, and adhesions are the most common cause of SBO.
Biliary Tract Disease. Biliary disease is the most common indication for abdominal surgery in the elderly.15-18 The incidence of cholelithiasis is 20-50% in those older than 70 years, and it is more common in women.18,19 Cholelithiasis also is more common in certain ethnic groups, such as those of Mexican heritage and Native Americans. Besides the increased prevalence of gallstone disease in this group, the elderly also have more complications from acute cholecystitis, such as gangrene or perforation.
Ascending cholangitis is rare before 70 years of age and carries a high mortality.20 It is a bacterial infection within an obstructed or poorly draining biliary system. The condition often is defined by the presence of Charcot’s triad: right upper quadrant abdominal pain, fever, and jaundice. Acute suppurative cholangitis has Reynold’s pentad, which adds mental confusion and hypotension to Charcot’s triad.5 It carries a 100% mortality if the biliary tree is not decompressed urgently. Another condition more commonly seen in the elderly is acalculous cholecystitis. Risk factors for acalculous cholecystitis are diabetes, parenteral nutrition, and the use of opiates. The mortality rate is 15%.21 Mirizzi’s syndrome is jaundice in acute cholecystitis due to ductal compression from an edematous gallbladder.21 Gallstone ileus is another cause of intestinal obstruction in the elderly. The condition has a 15% mortality rate.22 Gallstones cause about 50% of the cases of acute pancreatitis, what is termed gallstone pancreatitis. The condition has a higher mortality than alcoholic pancreatitis. Finally, bile peritonitis also is a complication of acute cholecystitis.2 The gallbladder ruptures, spilling bile in the peritoneum, which causes a diffuse inflammatory reaction.
Patients with biliary tract disease and acute cholecystitis will present with right upper quadrant pain, usually post-prandial, and vomiting. However, the clinical findings in the elderly do not correlate with the severity of the disease.18 Up to 25% of patients have no previous biliary symptoms. The majority of patients with cholecystitis are afebrile at presentation, and approximately 30-40% will fail to develop leukocytosis.
Ultrasound (US) is the preferred test to confirm the diagnosis of cholelithiasis and acute cholecystitis. In the hands of an experienced operator, its accuracy for detecting gallstones is 95-98%, but for ductal stones is only 20-55%.21 US is safe, non-invasive, accurate, rapid to perform, and easy to perform. US reliably shows gallbladder stones and sludge, with the characteristic acoustic shadowing. It also shows signs of acute cholecystitis, such as gallbladder wall thickening (greater than 3 mm), pericholecystic fluid, biliary tree dilatation, and gas in the biliary tree and gallbladder wall.23 The Murphy’s sign also can be reproduced using the US probe.24 The HIDA scan is better than the US at identifying acute cholecystitis and acalculous cholecystitis, but not as widely available.5,18 For those with biliary duct disease, endoscopic retrograde cholangiopancreatography (ERCP) is the gold standard. ERCP has the benefit of being both diagnostic and therapeutic. Magnetic resonance cholangiopancreatography (MRCP) also is used infrequently due to its low availability and high cost.
A biliary colic can be treated with analgesia, intravenous fluids, and antiemetics, while the diagnosis of any complication from cholelithiasis is excluded. For acute cholecystitis, antibiotics generally are recommended. Ampicillin (Principen) (4-6 g/d), ampicillin/sulbactam (Unasyn) (3 g IV/IM q6h), or piperacillin/tazobactam (Zosyn, Tazocin) (3.375 g IV q6h) often are used. For severe cases of acute cholecystitis, gentamicin (Garamycin) (3-5 mg/kg/d) with clindamycin (Cleocin, Dalacin C) (1.8-2.7 g/d) or metronidazole (Flagyl, Florazole ER, Trikacide) with a third-generation cephalosporin also will provide adequate coverage. Patients with ascending cholangitis and acute suppurative cholangitis require emergent biliary tree decompression.
Physicians should take care to recognize that acalculous cholecystitis is a disease of the elderly, and that the HIDA scan is a better diagnostic tool. An endoscopist for biliary tree decompression in cases of ascending cholangitis and acute suppurative cholangitis.
Acute Appendicitis. Appendicitis accounts for 5% of surgical abdomens in the elderly, and 7% of all appendectomies occur in those older than 60 years of age.5,17 Despite being so common, only 20% of the elderly present with the classic symptoms of anorexia, fever, right lower quadrant pain, and leukocytosis.5 Furthermore, many of these patients will present more than 24 hours from the onset of symptoms. The diagnosis carries a 3-15% mortality in this age group.2
Appendicitis complications are age-related. In the elderly, acute appendicitis has a 30-70% rupture rate.25,26 This is due, in part, to the fact that the blood supply to the appendix is more limited, the lumen is more narrow, and the wall more fibrotic and weak in elderly patients. In addition, the history often is atypical, the examination nonspecific, and the plain films and laboratories are normal.5 All these factors result in delayed diagnosis.15,17-19
Both US and CT scan are useful diagnostic tools for unclear cases.19 Once the diagnosis of acute appendicitis is established, surgical consultation is necessary.
Physicians should remember that only 20% of elderly patients with acute appendicitis have a typical presentation.
Diverticular Disease. The prevalence of diverticular disease increases with age. The prevalence of colonic diverticula is 30% in those older than 50 years, and 50% in those older than 70 years.18,19 About 30% of those with diverticulosis will at some point develop diverticulitis.5,15 As a general rule, the incidence of diverticulitis increases with the number of years with the disease.18
The most common presentation of diverticulitis is left lower quadrant abdominal pain, fever, and leukocytosis.5 A recent change in bowel habits also is common. Sometimes a mass is palpable in the left lower quadrant. There is pain on rectal examination, and about 50% of the patients will have heme-positive stools. The patients can have peritonitis if the diverticuli have ruptured. A second presentation is lower gastrointestinal bleeding. Diverticuli from the right colon, although less frequent, tend to bleed more, and the bleeding can be massive.18 Diverticular hemorrhage is the most common cause of major lower GI bleeding in the elderly.
The CT scan is the most helpful study to establish the diagnosis and show some of the complications.5,18,19,27
Mild cases can be treated as outpatients with a clear diet, analgesics, and antibiotics (trimethoprim-sulfamethoxazole [Bactrim, Septra, Sulfatrim] or ciprofloxacin [Cipro], and metronidazole).5 For those admitted, several antibiotic regimens have proven useful, including cefoxitin (Mefoxin), or gentamicin, and metronidazole. For those with diverticular bleeding, the management is aimed at assessing for hemodynamic instability and locating the source of the bleeding. Surgical consultation must be obtained for all.
In general, left-sided diverticuli become inflamed and cause pain, while right-sided diverticuli bleed.
Abdominal Aortic Aneurysm (AAA). Abdominal aortic aneurysms (AAA) are a disease of the elderly. The condition is associated with atherosclerosis, hypertension, and family history.5 An AAA can dissect or, more commonly, rupture. The mortality from rupture is very high, ranging 50-70%.28
The classic presentation is that of an elderly patient with sudden onset of abdominal, back, or flank pain.15 Hypotension can be present, but may be transient.29 If the condition starts as an aortic dissection, tearing chest pain may be the typical initial complaint; however, most cases are asymptomatic and are discovered incidentally.18 The physical examination is 100% sensitive and 95% specific when the enlarged pulsatile aorta is palpable.18 Unfortunately, this is not the case in many patients.
US is simple, safe, and highly reliable in the diagnosis of AAA.5,18,30,31 It is the test of choice for those with hemodynamic instability. The aorta is next to the inferior vena cava, and is recognized by its thicker wall, presence of pulsations, and lack of collapse with Valsalva’s maneuver. An aortic aneurysm with a diameter greater than 5-6 cm is considered at risk for rupture within 24 hours in patients who present with abdominal pain.24 US will give details about the internal and external diameter of the aorta, can distinguish the proximal and distal extent of the aneurysm, and can detect retroperitoneal fluid from a leaking aneurysm.24 The CT scan is useful in those who are hemodynamically stable.5,15 Aortography is considered the gold standard but can be used only in stable patients. In some cases, the diagnosis will be made by a kidney, ureter, and bladder film (KUB).19
Patients with a suspected ruptured or rapidly expanding AAA should be placed in a monitored bed, given oxygen, and have two large bore IVs started. At least four units of packed red blood cells must be requested. Any coagulopathy should be corrected. Immediate surgical consultation must be obtained. The rare case with a rapidly expanding, unruptured AAA with hypertension requires reduction of the tensile forces to the fragile vessel wall. This reduction of the rate/pressure product can be accomplished with IV beta adrenergic blockers. Esmolol (Brevibloc) (loading dose: 250-500 mcg/kg/min for 1 minute, then a maintenance infusion of 50 mcg/kg/min) or labetalol (Normodyne, Trandate) (20 mg IV q10min until blood pressure is controlled, then infused at 2 mg/min) are some of the agents that can be used. In the cases where an aneurysm with a diameter greater than 4 cm is found incidentally, all cases should be admitted for surgical repair.32
The most common misdiagnosis of a ruptured AAA is renal colic. It is estimated that abdominal aortic aneurysms have a misdiagnosis rate of 16-31%, making them a common cause of litigation in emergency medicine.29,33
Remember that the hypotension in a ruptured AAA can be transient. The most common misdiagnosis of a ruptured AAA is a renal colic.
Mesenteric Ischemia. Mesenteric ischemia is a disease of those older than 50 years of age. There are four different etiologies for mesenteric ischemia. Mesenteric emboli account for 50-55% of the cases and are associated with atrial fibrillation.34 Mesenteric arterial thrombosis and mesenteric venous thrombosis is the etiology in about 25% of cases. Finally, about 20% are termed non-occlusive mesenteric ischemia and are due to low-flow states.18,19 Suspect mesenteric ischemia in those with heart disease, hypovolemia, or hypotension of any cause.
There are no reliable screening tests for mesenteric ischemia, and the symptoms are non-specific.34 For these reasons, the condition often has delayed diagnosis. The mortality rate for these diagnostic delays is about 50-90%.15,35
The median age of presentation is 70 years, and most often it occurs in women and in those with history of cardiac disease. The pain often is described as out of proportion to the physical examination findings.15 Unfortunately, up to 25% may not even have abdominal pain. The examination otherwise usually is benign until late in the disease, where frank peritonitis develops from intestinal necrosis.
In the embolic form, usually there is no prior history of symptoms. Patients present with acute abdominal pain and GI emptying (diarrhea or vomiting).18,34 In contrast, patients with mesenteric artery thrombosis often have a prior history of recurrent abdominal pain (often called intestinal angina) and weight loss. The pain typically is post-prandial. Mortality in the thrombotic form can be as high as 85% due to a more proximal obstruction than with embolism. Risk factors for mesenteric thrombosis are severe atherosclerosis, increasing age, and hypertension. Mesenteric venous thrombosis presents similarly to artery thrombosis. It is seen as a complication of trauma, inflammatory intra-abdominal processes, hypercoagulable states, and venous stasis.
Non-occlusive mesenteric ischemia is due to vasoconstriction, not obstruction. Patients at risk are those with low-flow states and those in vasoactive medications. It thus usually is an intensive care unit (ICU) problem.
Patients with mesenteric ischemia often have leukocytosis, but a normal WBC count does not exclude the diagnosis. Metabolic acidosis and increased amylase can be seen in up to 50% of the patients, but the findings also are non-specific.15 An elevated lactate level is very sensitive, but has a poor specificity. 5,36,37 A normal lactate level, however, should not be used to exclude the diagnosis of mesenteric ischemia. Lactate is a non-specific marker for ischemia that becomes elevated later in the course of disease.
Plain films often are normal, but help exclude other causes of abdominal pain, such as obstruction and perforation.18 Since the CT scan is so widely available and utilized in the evaluation of abdominal pain, sometimes the diagnosis of mesenteric ischemia will be obtained with a CT.5 However, a normal CT scan does not exclude the presence of mesenteric ischemia.36 Angiography is the best test, and still is considered the gold standard.2,5 Angiography also can be therapeutic.5 However, in patients with shock or in vasopressors, the angiogram may have false results. Diagnosis by laparotomy in preferred in these specific cases.36
The treatment of a suspected mesenteric ischemia is geared at volume resuscitation and prompt attempts at confirming the diagnosis. Angiography can be both diagnostic and therapeutic. If there are signs of bowel necrosis, a surgical intervention is needed.
There are no reliable screening tests for mesenteric ischemia. One pitfall with this condition is considering mesenteric ischemia only in those with pain out of proportion to physical examination findings.
Other Conditions. Peptic ulcer disease (PUD) is common in the elderly. Gastric ulcers are more common than duodenal ulcers in this segment of the population.38 These ulcers can bleed or perforate. Peptic ulcers perforate in 5-10% of the geriatric patients. It is estimated that the plain films will show free air under the diaphragm in only 40-60% of the cases with a perforation.19,39
Constipation is common in elderly patients.19 Medications, low fiber diets, and insufficient fluid intake result in constipation. Importantly, constipation is not a cause of significant abdominal pain. Patients complain of fullness or discomfort, but true pain should point to a more severe problem and must not be ignored.
Elder abuse and neglect should be considered in patients with non-specific, difficult-to-explain, or recurrent abdominal pain. The history and physical examination may suggest abuse, but the diagnosis often is hard to obtain.
Non-abdominal Causes of Abdominal Pain. As in the pediatric patients, there are multiple diagnoses unrelated to GI pathology that can present with abdominal pain. Whenever evaluating the elderly with abdominal pain, also consider cardiopulmonary problems such as pneumonia, pneumothorax, acute pulmonary embolism, congestive heart failure, acute myocardial infarction, pericarditis, and endocarditis.
Systemic problems such as Adissonian crisis, hypercalcemia, intestinal tuberculosis, and acute porphyria also can cause abdominal pain. Infections such as Rocky Mountain spotted fever, tabes dorsalis, and herpes zoster need to be considered. Finally, acute angle closure glaucoma, muscle strain, rectus sheath hematoma, and drugs (phenothiazines, oral hypoglycemic agents, diuretics, non-steroidal anti-inflammatory drugs, erythromycin, azathioprine, estrogen, furosemide, sulfonamides, and tetracycline) should be thought of as potential causes of abdominal pain.2,15,19,40
The Immunocompromised Patient
Immunocompromised patients are another segment of the population with atypical presentations of disease. These patients may not be able to mount an immune response, including the development of leukocytosis and fever. They often present with hypothermia instead of fever as a sign of sepsis. The conditions seen in this patient group are either related to the immunocompromised state or the common conditions that occur in all patients but present differently in this group. Some of the patients considered in this segment are cancer patients, HIV positive patients, those with organ transplants, and those who are receiving steroids.41,42
Kaste and colleagues studied pediatric oncology patients with abdominal complaints.43 Hemorrhagic gastritis, peptic ulcer disease, and esophagitis can occur in children with cancer due to stress, radiation, chemotherapy, and graft vs. host disease (GVHD). The syndrome of graft vs. host disease starts 2-10 weeks after bone marrow transplantation and is characterized by dermatitis, enteritis, and liver disease. The condition is managed by increasing the dose of immunosuppressants.41 Typhlitis (also known as neutropenic enterocolitis or necrotizing enteropathy) occurs in neutropenic patients and is the most common cause of an acute abdomen in patients with leukemia.41 It is due to bacterial invasion of the colonic wall. The condition can mimic acute appendicitis. Plain films may demonstrate thickened loops of bowel, an obstructive pattern, or pneumatosis intestinalis.41 Either CT scan or US will reveal the diagnosis. The management of typhlitis consists of bowel rest, fluid resuscitation, and the use of broad spectrum antibiotics.41
Cancer patients also are at risk of bowel obstruction from masses. In addition to this, chemotherapeutic agents can induce an ileus, and radiation can cause delayed obstruction that typically occurs 6-24 months after treatment. Finally, surgeries in this patient population can predispose them to intussusception and adhesions, also leading to intestinal obstruction.
Some chemotherapeutic agents, such as L-asparaginase (Elspar, Kidrolase), azathioprine (Azasan, Imuran), and some corticosteroids can cause pancreatitis, which presents with upper quadrant abdominal pain.41 Cancer patients are at risk for urinary obstruction from external compression, and urinary infection can ensue due to the resultant stasis. All of these have the potential to cause abdominal pain in the adult patient with malignancy.43
Another group of immunocompromised patients with abdominal pain is HIV/AIDS patients.42 In a study of 458 HIV-positive patients admitted to a hospital, 15% had an admission diagnosis of abdominal pain.44 In 59% of the cases where the diagnosis was made pre-mortem, the most common diagnoses were non-Hodgkin’s lymphoma in 17%; pancreatitis in 12%; cyto-megalovirus (CMV) colitis or enteritis in 11%; sclerosing cholangitis in 8%; gastrointestinal (GI) Kaposi’s sarcoma in 5%; acute cholecystitis in 5%; cryptosporidial infection in 6%; and CMV gastritis/esophagitis in 6%. The causes of pain were directly related to the immunocompromised state in 65% of these cases.44 In this group of patients, an admission due to abdominal pain also was related to reduced survival. Indications for surgery in the HIV patients were similar to those in the non-HIV patients, namely obstruction, perforation, or peritonitis. The post-operative prognosis also was poor.12,45
Of note, drugs used for the treatment of HIV are a risk factor for pancreatitis. Didanosine (DDI) (Videx, ddI), hydroxyurea (Hydrea, Droxia), and pentamidine (Pentam, NebuPent, Pentacarinat) are three of those drugs.46-48
Typhlitis is the most common cause of an acute abdomen in patients with leukemia. Remember to consider hypothermia as a sign of sepsis in the immunocompromised.
The Pregnant Patient
Many physiologic changes occur during pregnancy. Physicians must be familiar with these to adequately diagnose disease in pregnancy, and differentiate it from normal maternal adaptations. The changes that are relevant to the evaluation of a pregnant patient with abdominal pain will be discussed.
Pregnant patients have relative hypervolemia, with intravascular volume increasing between 30-50%, while hemoglobin decreases slightly. Because of this increase in blood volume, pregnant women tolerate much larger volumes of blood loss than their non-pregnant counterparts before any significant changes in vital signs are noticeable.49 Cardiac output increases by 30-50%, while resting heart rate increases by 10-15 beats/minute.49 Both arterial blood pressure and peripheral vascular resistance decrease during pregnancy. Therefore, vital signs should be interpreted with caution in pregnant patients, so as not to be reassured falsely.
Blood viscosity decreases, and platelet numbers decrease, but this is counteracted by shortened coagulation times. The end result is normal hemostasis. Finally, normal white blood cell counts in pregnancy range from 10,000 to 14,000/mm3; therefore, leukocytosis is not a reliable marker of infection during pregnancy.49
Polymorphonuclear leukocyte function is depressed with respect to chemotaxis and adherence. This can lead to a predisposition to infections.
Acute Appendicitis. Acute appendicitis is the leading non-obstetrical reason for surgery during pregnancy.50 Its incidence is reported at 1 in 833-1400 live births.51-54 Although the incidence of appendicitis is not increased during pregnancy, several factors cause delays in its diagnosis, which can lead to increased complications for both the mother and the fetus.50 Ruptured appendices are 2-3 times more common in pregnancy.50,55 After 24 hours from symptom onset, the risk of perforation is high. Fetal loss occurs in 3-5% of appendicitis cases but, if perforated, the rate goes up to 20-30%.49,50,56 Additionally, pulmonary complications occur in 18% of cases after appendicitis in pregnancy. These pulmonary complications include the acute respiratory distress syndrome (ARDS), acute pulmonary edema, and pulmonary infiltrates.57
The location of the appendix varies with gestational age, although this common teaching has been put to question in a recent study, where this displacement of the appendix was only seen in a small portion of pregnant women.58 During the first trimester, it is located at McBurney’s point. At 20 weeks, it is displaced upward to the level of the iliac crest, and during the third trimester it is located above the iliac crest and below the costal margin.49 This displacement results in pain not necessarily located in the typical right lower quadrant. Additionally, guarding and rebound are less common in the second and third trimesters, since the abdominal musculature is displaced away from the inflamed appendix.50 Up to 80% of the patients may complain only of rectal pain and vaginal tenderness. Nausea and vomiting are common symptoms, and anorexia is found in 60-70% of patients.52,59 Fever is found in fewer than 20% of patients.60
Pyuria and hematuria further may confuse the picture and can be found in up to 20% of cases.59,60 This probably is due to the location of the inflamed appendix close to the right ureter and kidney. As stated above, white blood cell (WBC) counts normally increase during pregnancy, so the WBC count is not a good marker of acute appendicitis.49,54 One study looked at 29 pregnant women undergoing appendectomy. A WBC count greater than 10,000 had a sensitivity of 85% and a specificity of only 33%. A WBC count greater than 15,000 had a sensitivity of 50% and a specificity of 89%.61 Other studies, however, also concur in the lack of usefulness of the WBC count, reporting that a WBC count greater than 16,000 had sensitivity of only 60% and a specificity of only 5%.62,63 The presence of a left shift is somewhat helpful. On physical examination, Alder’s sign and Rovsing’s sign are good for distinguishing uterine from extrauterine pain.64
The US is useful, but sometimes the appendix can be difficult to visualize.65 A study of 45 pregnant patients with acute appendicitis found that US has a PPV of 94% and a NPV of 100%.65 The sonographic criteria for acute appendicitis are an uncompressible appendix of greater than 7mm of width.66 Since the risk of failing to diagnose appendicitis carries such a high fetal mortality, it often is advocated to use laparoscopy or laparotomy as a definitive diagnostic procedure in those cases where the appendix cannot be visualized fully with US. The rate of negative laparotomies can be as high as 40% in the second and third trimesters.55
Management in the ED involves adequate hydration, nothing per mouth, and early surgical consultation.
Remember to consider that a ruptured appendicitis increases fetal loss rates to 20-30%. The appendix may be displaced cephalad during pregnancy, and peritoneal signs are less common in the second and third trimesters.
Biliary Tract Disease. Cholecystitis is the second most common non-obstetrical surgical condition in pregnancy.49 Due to the surge in progesterone during pregnancy, the gallbladder does not contract effectively, and its emptying often is incomplete. There is bile stasis and concentration during pregnancy. This predisposes to stone formation.49 The presentation is similar to the non-pregnant state, with right upper quadrant abdominal pain, usually post-prandial.
The US is 96-98% sensitive and specific for cholelithiasis.49 Pregnant patients with symptomatic cholelithiasis should be evaluated by the surgeon prior to discharge from the ED. As in the non-pregnant counterparts, all patients with acute cholecystitis should be admitted.
The amylase and lipase may be elevated due to concomitant pancreatitis. When pancreatitis develops, fetal loss rates can be as high as 50%.67
Cholecystitis is the second most common non-obstetrical surgical condition during pregnancy, after acute appendicitis.
Ectopic Pregnancy (EP). Ectopic pregnancy is the leading cause for maternal death in the first trimester of pregnancy, and accounts for 10% of all maternal mortality. Its incidence has been increasing, with a prevalence of 1 in 100 pregnancies.68 Risk factors for EP are in vitro fertilization, previous pelvic inflammatory disease (PID), tubal ligation, use of an intrauterine device (IUD), and a previous ectopic pregnancy.69,70
The typical presentation of an EP is a patient with a delayed or missed menstrual period who has abdominal pain and vaginal bleeding. However, these findings are not sensitive or specific.71 The pain can be constant and severe, and radiate to the shoulder, but also can be crampy and intermittent. The presence of peritonitis, cervical motion tenderness, or lateral abdominal or pelvic tenderness increases the likelihood of an ectopic pregnancy.71
The most useful diagnostic tool is US. It will locate the gestation, give a gestational age, and assess fetal viability. Transabdominal US can detect a gestational sac as early as 5 weeks of gestation, a fetal pole by 5-6 weeks, and a fetal heart beat by 7-8 weeks of gestation. The findings using transvaginal US occur 0.5-1 week sooner.24 Transabdominal US will be indeterminate (unable to exclude an EP) in about 50% of cases of EP.72 Transvaginal US will decrease the rate of indeterminate studies to 18%.72 Ultrasound has a sensitivity of 87% and a specificity of 94% when compared to laparoscopic diagnosis.73 Other tests used to evaluate a suspected EP include serial quantitative human chorionic gonadotropin (HCG) levels, culdocentesis, and laparoscopy.74,75
Unstable patients should have emergent obstetrical consultation for surgery. The stable patient should have an obstetrical evaluation for operative (laparoscopy) or non-operative (methotrexate) management.
The US is the most useful diagnostic tool for EP. Remember to perform a pregnancy test on all women of childbearing age with abdominal pain.
Miscarriage. About one-third to one-half of all pregnancies result in a miscarriage.76 First-trimester spontaneous abortions are thus a common presenting complaint in the ED. Women present with crampy low abdominal pain and vaginal bleeding.
The ED evaluation should include establishing a length of gestation, Rh type of the mother, doing a pelvic examination, and obtaining an obstetrical consultation. Counseling is important, since about 50% of the threatened abortion cases seen in the ED will end up in a complete abortion.
Threatened abortion is characterized by bleeding through a closed cervical os. Inevitable abortion is defined by an open internal cervical os. Incomplete abortion is where the products of conception can be visualized at the os or in the vaginal canal. Dilatation and curettage may be necessary in these cases to remove any retained products of conception. Completed abortion is when all the products of conception have been expelled, the os is closed, and the uterus is contracted.
Ultrasound is the best study for fetal viability and fetal location. It is always important to exclude ectopic pregnancy in a pregnant patient with vaginal bleeding and adnexal pain.
Anti-D immune globulin (RhoGam) should be administered to all Rh negative mothers who present with a miscarriage and have vaginal bleeding. The dose is 50 mcg during the first trimester and 300 mcg thereafter.77
A potential pitfall is failing to provide RhoGam to pregnant women who are Rh negative and present with a miscarriage and vaginal bleeding.
Abruptio Placentae. Abruptio placentae is a separation of the normally implanted placenta from the uterine wall that can be seen during the second half of the pregnancy. It can be traumatic or spontaneous and can be acute or chronic.
Patients present with dark, scant vaginal bleeding and uterine tenderness. Uterine irritability may be present, as well as signs of maternal shock, fetal distress, and disseminated intravascular coagulation (DIC).
Ultrasound is the diagnostic modality of choice to evaluate fetal status and to locate the placenta to exclude the possibility of placenta previa or vasa previa. Still, it is rather insensitive, since fresh blood has a similar echogenicity as the placenta.78
The ED management of a placental abruption is geared at maternal stabilization and immediate obstetrical consultation.
Patients with abruptio placentae present with dark, scant vaginal bleeding and uterine tenderness.
Other Conditions. Urinary tract infections are common in pregnancy. Hydroureter and stasis predisposes to cystitis and pyelonephritis.49 HELLP Syndrome (Hemolysis, Elevated Liver enzymes, and Low Platelets) is a third-trimester complication. The condition is a variant of severe preeclampsia. Women present with right upper quadrant pain, nausea, and vomiting. The mental status generally is normal. These patients should be admitted to an intensive care unit after obstetrical consultation. Gastroesophageal reflux disease (GERD) complicates 30-70% of pregnancies. The surge in progesterone mediates relaxation of lower esophageal sphincter, which results in reflux of acid into the esophagus.49 Finally, preterm labor results is another cause of crampy low abdominal pain.
Unusual Causes of Abdominal Pain: The Zebras
The zebras are the diagnoses physicians rarely consider. These are the very unusual diagnoses that rarely are seen and, for that reason, can be missed.
Acute Adrenal Crisis. The adrenal gland produces glucocorticoids, mineralocorticoids, and androgens. Of these, cortisol and aldosterone are important. Cortisol stimulates gluconeogenesis, inhibits insulin secretion, and has an anti-inflammatory effect. Aldosterone’s main effect in the kidney results in the reabsorption of sodium and water. Acute adrenal insufficiency can occur due to damage to the adrenal gland from infiltration, infection, or hemorrhage or from disruption of the hypothalamic-pituitary-adrenal axis by the chronic use of steroids—and a sudden withdrawal from them.79,80 Patients with acute, severe illnesses also are at risk of developing acute adrenal insufficiency.
An adrenal crisis can present with abdominal or flank pain, vomiting, and hypovolemic shock that generally is unresponsive to fluids and vasopressors. The patients may be hypothermic or, less commonly, hyperthermic.80
Hyponatremia and hyperkalemia are the typical, but not diagnostic, laboratory findings. Hypoglycemia also can be seen. The patients also can be anemic, and the CBC often shows eosinophilia. The diagnosis is made by a serum adrenocorticotropic hormone (ACTH) stimulation test: A baseline cortisol level is drawn and 250 mcg of ACTH are injected. Cortisol levels are drawn 30 and 60 minutes after the ACTH injection. A cortisol increase of less than 7 mcg/dl is indicative of adrenal insufficiency.
The management largely is supportive, along with both glucocorticoid and mineralocorticoid supplementation. Dexamethasone (Decadron, Dexone, Dexasone, Hexadrol) 4-8 mg IV sometimes is used initially, because it does not interfere with the cortisol assay. Hydrocortisone (Cortef, Solu-Cortef) at 100 mg IV every 6 hours; however, is the drug of choice, since it has both glucocorticoid and mineralocorticoid activities. Always search for a precipitating cause.81
Porphyria. The porphyrias are a group of diseases affecting the metabolism of heme. They present with a rash, abdominal pain, constipation, neuropathies, and psychiatric changes. Acute intermittent porphyria (AIP), as the name implies, occurs with paroxysmal episodes of abdominal pain, followed by the psychiatric changes and neuropathies. The abdominal pain is severe and lasts days at a time. It often is described as colicky and can be accompanied by nausea and vomiting. Despite its severity, the physical examination findings are non-specific. The peripheral neuropathies from porphyria generally affect motor function and can mimic Guillain-Barré syndrome. Autonomic neuropathies and seizures also can be seen.
The diagnosis is confirmed by sending urinary porphobilinogen levels, a heme precursor that is elevated during AIP attacks. Coproporphyrin also may be elevated. Other laboratory abnormalities include hyponatremia and a mild leukocytosis. Plain films and CT scans often are normal.
The treatment of AIP is aimed at decreasing the synthesis of heme. This often is performed by inducing hyperglycemia (blood sugar greater than 400 mg/dl), giving hemin for injection (Panhematin) (4 mg/kg/day for 4 days) to those with severe attacks, narcotic analgesia, and treating seizures with gabapentin (Neurontin), since other anticonvulsants can worsen AIP.82,83
Familial Mediterranean Fever (FMF). Also called recurrent polyserositis, FMF causes recurrent peritonitis, pleuritis, and arthritis accompanied by fever. As the name implies, it commonly occurs in family groups and is more common in those of Mediterranean origin (Ashkenazi and Sephardic Jews, Armenians, Turks, and Arabs). It is caused by a genetic mutation that impairs the body’s ability to deactivate chemotactic factors that are formed in response to inflammation. Most of the attacks occur in those younger than 20 years of age. There is a male predominance for the disease.
The attacks are paroxysmal, with a sudden onset of high fever. Almost all patients will develop abdominal pain, which can progress to peritonitis. These patients often undergo appendectomies and cholecystectomies. Another commonly confused diagnosis with FMF is renal colic. Patients can have pleuritic chest pain, and pericarditis can occur, although pericardial effusions and tamponade are rare. Arthritis also occurs, resembling gout and most commonly involving the knees, wrists, and ankles. Less common symptoms include scrotal pain, pelvic pain, myalgias, and an erysipelas-like rash.84,85
Colchicine is the drug of choice for the treatment of FMF. Other treatments include nonsteroidal anti-inflammatory drugs for arthritis, and prednisone (Delta-Cortef, Prelone, Pediapred) for the myalgias.
Primary Epiploic Appendagitis. The epiploic appendages are small peritoneal pouches 0.5-5 cm long containing small vessels and fat. They are located on the antimesenteric serous surface of the colon.86 Patients with epiploic appendagitis present with non-specific abdominal pain, more commonly in the left lower quadrant, may have leukocytosis. On US, the inflamed epiploic appendages can be seen as a small, oval, hyperechoic, non-compressible mass with an occasional hypoechoic halo. The mass is adherent to the anterior abdominal wall and fixed during breathing.86,87 On CT scan, the mass is in contact with the serous surface of the adjacent colon and has the same attenuation as fat.86,87
Most of these appendages become inflamed after they torse or thrombose. Since the pain occurs more commonly in the left lower quadrant, the condition can be confused with diverticulitis. Primary epiploic appendagitis is considered a self-limited process, with the symptoms most often lasting between 3-7 days.86 In a reported case series, the patients with epiploic appendagitis constituted 7.1% of those having an US for left lower quadrant pain, making this a not-so-uncommon condition.86
Toxins. For hundreds of years, lead has been known to cause abdominal pain, which often is referred to as a lead colic. Most of the pediatric lead exposures are related to old, lead-containing paint in their homes. In adults, most of the exposure to lead is occupational—from smelting, soldering, battery manufacturing, and ceramic glazing. In addition to abdominal pain, patients with lead toxicity have anemia, basophilic stippling, neuropathies, hyperuricemia and gout (saturnine gout), proteinuria, encephalopathy, and hypertension. The diagnosis of lead toxicity is confirmed by a blood lead level.
Arsenic historically has been used as a homicidal agent. Patients with arsenic poisoning present with abdominal pain, vomiting, and profuse diarrhea similar to the rice water stools of cholera. They may have a metallic taste in their mouths and a garlicky odor to their breath. The laboratories often show anemia and basophilic stippling. The plain films may show metallic residues in the GI tract after an acute exposure. The diagnosis is best confirmed by a 24-hour urine collection for arsenic.
A working knowledge of the common and uncommon conditions that result in abdominal pain is essential for emergency physicians. In the specific patient populations discussed in this article, attention must be paid to subtle and unusual presentations. The use of laboratory and, more importantly, radiology studies will help exclude surgical or other life-threatening conditions. However, despite a physician’s best efforts at reaching a final diagnosis, one has to understand that almost half of the patients will leave the ED without a definitive diagnosis. In those cases, arranging follow-up care and providing detailed discharge instructions becomes extremely important.
Acknowledgements: The authors would like to acknowledge the assistance of Collin Goto, MD, and Patricia Santiago-Muñoz, MD, in the preparation of this manuscript.
References
1. Kamin RA, Norwicki TA, Courtney DS, et al. Pearls and pitfalls in the emergency department evaluation of abdominal pain. Emerg Med Clin North Am 2003;21:61-72, vi.
2. Kauvar DR. The geriatric acute abdomen. Clin Geriatr Med 1993;9: 547-558.
3. van Geloven AA, Biesheuvel TH, Luitse JS, et al. Hospital admissions of patients aged over 80 with acute abdominal complaints. Eur J Surg 2000; 166:866-871.
4. Marco CA, Schoenfeld CN, Keyl PM, et al. Abdominal pain in geriatric emergency patients: Variables associated with adverse outcomes. Acad Emerg Med 1998;5:1163-1168.
5. Hendrickson M, Naparst TR. Abdominal surgical emergencies in the elderly. Emerg Med Clin North Am 2003;21:937-969.
6. Harlow CL, Stears RL, Zeligman BE, et al. Diagnosis of bowel obstruction on plain abdominal radiographs: Significance of air-fluid levels at different heights in the same loop of bowel. AJR Am J Roentgenol 1993;161:291-295.
7. Beck DE, Opelka TG, Bailey HR, et al. Incidence of small-bowel obstruction and adhesiolysis after open colorectal and general surgery. Dis Colon Rectum 1999;42:241-248.
8. Bohner H, Yang Q, Franke C, et al. Simple data from history and physical examination help to exclude bowel obstruction and to avoid radiographic studies in patients with acute abdominal pain. Eur J Surg 1998;164:777-784.
9. Maglinte DD, Bender GN, Heitkamp DE, et al. The role of radiology in the diagnosis of small-bowel obstruction. AJR Am J Roentgenol 1997;168: 1171-1180.
10. Arnold GJ, Nance FC. Volvulus of the sigmoid colon. Ann Surg 1973;177: 527-537.
11. O’Mara CS, Wilson TH Jr., Stonesifer GL, et al. Cecal volvulus: Analysis of 50 patients with long-term follow-up. Curr Surg 1980;7:132-136.
12. Wilson SE, et al. Acquired immune deficiency syndrome (AIDS). Indications for abdominal surgery, pathology, and outcome. Ann Surg 1989;210: 428-433; discussion 433-434.
13. Frizelle FA, Wolff BG. Colonic volvulus. Adv Surg 1996;29:131-139.
14. Jones IT, Fazio VW. Colonic volvulus. Etiology and management. Dig Dis 1989;7:203-209.
15. Sanson TG, O’Keefe KP. Evaluation of abdominal pain in the elderly. Emerg Med Clin North Am 1996;14:615-627.
16. Telfer S, Fenyo G, Holt PR, et al. Acute abdominal pain in patients over 50 years of age. Scand J Gastroenterol Suppl 1988;144:47-50.
17. Fenyo G. Acute abdominal disease in the elderly: Experience from two series in Stockholm. Am J Surg 1982;143:751-754.
18. Balsano N, Cayten CG. Surgical emergencies of the abdomen. Emerg Med Clin North Am 1990;8:399-410.
19. Dang C, Aguilera P, Dang A, et al. Acute abdominal pain. Four classifications can guide assessment and management. Geriatrics 2002;57:30-32, 35-36, 41-42.
20. Chock E, Wolfe BM, Matolo NM. Acute suppurative cholangitis. Surg Clin North Am 1981;61:885-892.
21. Parks RW. Biliary tract emergencies. Hosp Med 2002;63:226-229.
22. Heuman R, Sjodahl R, Wetterfors J. Gallstone ileus: An analysis of 20 patients. World J Surg 1980;4:595-598.
23. Marincek B. Nontraumatic abdominal emergencies: Acute abdominal pain: Diagnostic strategies. Eur Radiol 2002;12:2136-2150.
24. Heller MB, Verdile VP. Ultrasonography in emergency medicine. Emerg Med Clin North Am 1992;10:27-46.
25. Owens BJ, Hamit HF. Appendicitis in the elderly. Ann Surg 1978;187: 392-396.
26. Lau WY, Fan ST, Yiu TF, et al. Acute appendicitis in the elderly. Surg Gynecol Obstet 1985;161:157-160.
27. Ferzoco LB, Raptopoulos V, Silen W. Acute diverticulitis. N Engl J Med 1998;338:1521-1526.
28. Johansen K, Kohler TR, Nicholls SC, et al. Ruptured abdominal aortic aneurysm: The Harborview experience. J Vasc Surg 1991;13:240-245; discussion 245-247.
29. Marston WA, Ahlquist R, Johnson G Jr., et al. Misdiagnosis of ruptured abdominal aortic aneurysms. J Vasc Surg 1992;16:17-22.
30. Blaivas M, Theodoro D. Frequency of incomplete abdominal aorta visualization by emergency department bedside ultrasound. Acad Emerg Med 2004; 11:103-105.
31. Kuhn M, Bonnin RC, Davey MJ, et al. Emergency department ultrasound scanning for abdominal aortic aneurysm: Accessible, accurate, and advantageous. Ann Emerg Med 2000;36:219-223.
32. Szilagyi DE, Elliott JP, Smith RF. Clinical fate of the patient with asymptomatic abdominal aortic aneurysm and unfit for surgical treatment. Arch Surg 1972;104:600-606.
33. Rose J, Civil I, Koelmeyer T, et al. Ruptured abdominal aortic aneurysms: Clinical presentation in Auckland 1993-1997. ANZ J Surg 2001;71:341-344.
34. Park WM, Gloviczki P, Cherry KJ, et al. Contemporary management of acute mesenteric ischemia: Factors associated with survival. J Vasc Surg 2002;35:445-452.
35. Mamode N, Pickford I, Leiberman P. Failure to improve outcome in acute mesenteric ischaemia: Seven-year review. Eur J Surg 1999;165:203-208.
36. Ruotolo RA, Evans SR. Mesenteric ischemia in the elderly. Clin Geriatr Med 1999;15:527-557.
37. Murray MJ, Gonze MD, Nowak LR, et al. Serum D(-)-lactate levels as an aid to diagnosing acute intestinal ischemia. Am J Surg 1994;167:575-578.
38. Narayanan M. Steinheber FU. The changing face of peptic ulcer in the elderly. Med Clin North Am 1976;60:1159-1172.
39. Kum CK, Chong YS, Koo CC, et al. Elderly patients with perforated peptic ulcers: Factors affecting morbidity and mortality. J R Coll Surg Edinb 1993; 38:344-347.
40. Purcell TB. Nonsurgical and extraperitoneal causes of abdominal pain. Emerg Med Clin North Am 1989;7:721-740.
41. Nylander WA Jr. The acute abdomen in the immunocompromised host. Surg Clin North Am 1988;68:457-470.
42. Wyatt SH, Fishman EK. The acute abdomen in individuals with AIDS. Radiol Clin North Am 1994;32:1023-1043.
43. Kaste SC, Rodriguez-Galindo C, Furman WL. Imaging pediatric oncologic emergencies of the abdomen. AJR Am J Roentgenol 1999;173:729-736.
44. Parente F, Cernuschi M, Antinori S, et al. Severe abdominal pain in patients with AIDS: Frequency, clinical aspects, causes, and outcome. Scand J Gastroenterol 1994;29:511-555.
45. Robinson G, Wilson SE, Williams RA. Surgery in patients with acquired immunodeficiency syndrome. Arch Surg 1987;122:170-175.
46. Barone JE, et al. Abdominal pain and anorectal disease in AIDS. Gastroenterol Clin North Am 1988;17:631-638.
47. Moore RD, Keruly JC, Chaisson RE. Incidence of pancreatitis in HIV-infected patients receiving nucleoside reverse transcriptase inhibitor drugs. Aids 2001;15:617-620.
48. Dutta SK, Ting CD, Lai LL. Study of prevalence, severity, and etiological factors associated with acute pancreatitis in patients infected with human immunodeficiency virus. Am J Gastroenterol 1997;92:2044-2048.
49. Stone K. Acute abdominal emergencies associated with pregnancy. Clin Obstet Gynecol 2002;45:553-561.
50. Musselman RC, Nunnelee JD, Ware DB. Appendicitis during pregnancy. Clin Excell Nurse Pract 1998;2:338-342.
51. Allen AE. Ultrasound investigation of pyloric stenosis. Radiogr Today 1988;54:49.
52. Horowitz MD, Gomez GA, Antiesteban R, et al. Acute appendicitis during pregnancy. Diagnosis and management. Arch Surg 1985;120:362-367.
53. Mazze RI, Kallen B. Appendectomy during pregnancy: A Swedish registry study of 778 cases. Obstet Gynecol 1991;77:835-840.
54. Mourad J, Elliott JP, Erickson C, et al. Appendicitis in pregnancy: New information that contradicts long-held clinical beliefs. Am J Obstet Gynecol 2000; 182:1027-1029.
55. Hee P, Viktrup L. The diagnosis of appendicitis during pregnancy and maternal and fetal outcome after appendectomy. Int J Gynaecol Obstet 1999;65: 129-135.
56. Firstenberg MS, Malangoni MA. Gastrointestinal surgery during pregnancy. Gastroenterol Clin North Am 1998;27:73-88.
57. de Veciana M, Towers CV, Major CA, et al. Pulmonary injury associated with appendicitis in pregnancy: Who is at risk? Am J Obstet Gynecol 1994; 171:1008-1013.
58. Hodjati H, Kazerooni T. Location of the appendix in the gravid patient: A re-evaluation of the established concept. Int J Gynaecol Obstet 2003;81:245-247.
59. Tamir IL, Bongard FS, Klein SR. Acute appendicitis in the pregnant patient. Am J Surg 1990;160:571-575; discussion 575-576.
60. Mahmoodian S. Appendicitis complicating pregnancy. South Med J 1992; 85:19-24.
61. Doberneck RC. Appendectomy during pregnancy. Am Surg 1985;51: 265-268.
62. Mackway-Jones K, Harrison M. Towards evidence based emergency medicine: Best BETS from the Manchester Royal Infirmary. Analgesia and assessment of abdominal pain. J Accid Emerg Med 2000;17:126-129.
63. Andersen B, Nielsen TF. Appendicitis in pregnancy: Diagnosis, management and complications. Acta Obstet Gynecol Scand 1999;78:758-762.
64. Alders N. A sign for differentiating uterine from extrauterine complications of pregnancy and puerperium. Br Med J 1951;4741:1194-1195.
65. Lim HK, Bae SH, Seo GS. Diagnosis of acute appendicitis in pregnant women: Value of sonography. AJR Am J Roentgenol 1992;159:539-542.
66. Barloon TJ, Brown BP, Abu-Yousef MM, et al. Sonography of acute appendicitis in pregnancy. Abdom Imaging 1995;20:149-151.
67. Braverman DZ, Johnson ML, Kern F Jr. Effects of pregnancy and contraceptive steroids on gallbladder function. N Engl J Med 1980;302:362-364.
68. Centers for Disease Control: Ectopic Pregnancy—United States, 1990-1992. MMWR 1995;44:46.
69. Skjeldestad FE, Hadgu A, Eriksson N. Epidemiology of repeat ectopic pregnancy: A population-based prospective cohort study. Obstet Gynecol 1998; 91:129-135.
70. Marchbanks PA, Annegers JF, Conlam CB, et al. Risk factors for ectopic pregnancy. A population-based study. JAMA 1988;259:1823-1827.
71. Dart RG, Kaplan B, Varaklis K. Predictive value of history and physical examination in patients with suspected ectopic pregnancy. Ann Emerg Med 1999;33:283-290.
72. Barnhart K, Mennuti MT, Benjamin I, et al. Prompt diagnosis of ectopic pregnancy in an emergency department setting. Obstet Gynecol 1994;84: 1010-1015.
73. Shalev E, Yarom I, Bustan M, et al. Transvaginal sonography as the ultimate diagnostic tool for the management of ectopic pregnancy: Experience with 840 cases. Fertil Steril 1998;69:62-65.
74. Chen PC, Sickler GK, Dubinsky TJ, et al. Sonographic detection of echogenic fluid and correlation with culdocentesis in the evaluation of ectopic pregnancy. AJR Am J Roentgenol 1998;170:1299-1302.
75. Kontoravdis A, Chryssikopoulos A, Hassiakos D, et al. The diagnostic value of laparoscopy in 2365 patients with acute and chronic pelvic pain. Int J Gynaecol Obstet 1996;52:243-248.
76. Wilcox AJ, Weinberg CR, O’Connor JF, et al et al. Incidence of early loss of pregnancy. N Engl J Med 1988;319:189-194.
77. Von Stein GA, Munsick RA, Stiver K, et al. Fetomaternal hemorrhage in threatened abortion. Obstet Gynecol 1992;79:383-386.
78. Sholl JS. Abruptio placentae: Clinical management in nonacute cases. Am J Obstet Gynecol 1987;156:40-51.
79. Koo DJ, Jackman D, Chaudry IH, et al. Adrenal insufficiency during the late stage of polymicrobial sepsis. Crit Care Med 2001;29:618-622.
80. Vella A, Nippoldt TB, Morris JC, 3rd. Adrenal hemorrhage: A 25-year experience at the Mayo Clinic. Mayo Clin Proc 2001;76:161-168.
81. Chin R. Adrenal crisis. Crit Care Clin 1991;7:23-42.
82. Kauppinen R, Mustajoki P. Prognosis of acute porphyria: Occurrence of acute attacks, precipitating factors, and associated diseases. Medicine (Baltimore) 1992;71:1-13.
83. Kalman DR, Bonkovsky HL. Management of acute attacks in the porphyrias. Clin Dermatol 1998;16:299-306.
84. Sohar E, Gafni J, Pras M, et al. Familial Mediterranean fever. A survey of 470 cases and review of the literature. Am J Med 1967;43:227-253.
85. Ben-Chetrit E, Levy M. Familial Mediterranean fever. Lancet 1998;351: 659-664.
86. Molla E, Ripolles T, Martinez MJ, et al. Primary epiploic appendagitis: US and CT findings. Eur Radiol 1998;8:435-438.
87. Rioux M, Langis P. Primary epiploic appendagitis: Clinical, US, and CT findings in 14 cases. Radiology 1994;191:523-526.
The first part of this series discussed abdominal pain in pediatric patients. This second and final part will cover abdominal pain in elderly, immunocompromised, and pregnant patients. Those 65 years of age and older constitute the fastest-growing segment of the population, and currently comprise about 12% of the U.S. population. This means that abdominal pain in the elderly will be a commonplace occurrence in EDs.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
