Treating Hypertension in the Emergency Department: First, Do No Harm, Part II
Authors: Lisa Freeman Grossheim, MD, FACEP, Assistant Professor, University of Texas Medical School at Houston, Department of Emergency Medicine; Monica Carvajal, MD, Resident Physician, Department of Emergency Medicine, University of Texas Medical School at Houston, and Heidi Knowles-Ely, MD, Resident Physician, Department of Emergency Medicine, University of Texas Medical School at Houston.
Peer Reviewers: Charles Emerman, MD, Professor and Chairman of Emergency Medicine, Case Western Reserve University, Cleveland, OH; and Richard O. Gray, MD, Assistant Professor of Emergency Medicine, Hennepin County Medical Center, Minneapolis, MN.
Part I of this series focused on hypertensive syndromes and clinical evaluation. This second and final part will cover antihypertensive medications and management of hypertension in specific disease processes.—The Editor
Antihypertensive Medications
Vasodilators. Nitroglycerin. Nitroglycerin is an arterial and venous vasodilator (primarily venous) that decreases blood pressure by decreasing venous return and cardiac output. It also is a coronary artery dilator. Heart rate usually increases modestly as a reflex to the fall in blood pressure, but paradoxical bradycardia can occur. Other adverse effects include headache and postural hypotension.
Nitroglycerin is indicated for high blood pressure associated with cardiac ischemia. Caution should be used in patients with increased intracranial pressure, uncorrected hypovolemia, constrictive pericarditis/pericardial tamponade, and concurrent use of erectile dysfunction medications such as sildenafil (Viagra) or tadalafil (Cialis).1
Tolerance to nitroglycerin therapy may occur, therefore resulting in poor response to high doses of nitroglycerin. Therefore in acute cardiac ischemia or decompensated heart failure that is unresponsive to nitroglycerin, other agents will need to be utilized.
There are many forms of nitroglycerin available. (See Table 1.) The most useful forms of nitroglycerin for use in the emergent setting are sublingual, aerosol, and injectable. The onset of action of the transdermal form is not rapid enough to be useful in the setting of acute chest pain or hypertension.
Table 1. Forms of Nitroglycerin

Nitroprusside. Nitroprusside is a direct vasodilator that, like nitroglycerin, affects both peripheral arteries and veins as well as the coronary arteries. It is given only by infusion, as the onset of action is fewer than five minutes, and the effects dissipate rapidly after stopping the medication.1 Nitroprusside is indicated for the immediate reduction of blood pressure in patients with a hypertensive emergency. As with all vasodilators, caution should be used in patients with increased intracranial pressure. Adverse effects include excessive hypotension and bradycardia or tachycardia. A well-known adverse effect is cyanide toxicity. This is more likely to occur in patients who are on high or prolonged doses or those with renal or hepatic insufficiency. Although unusual, cyanide toxicity can occur after only a few hours on nitroprusside. The diagnosis is not made via a cyanide level, as this is not readily available in the ED, but via clinical clues including bright red venous blood, confusion, air hunger, metabolic acidosis, and death. The usual starting dose of nitroprusside is 0.3 mcg/kg/min with titration upward. The average effective dose is 3 mcg/kg/min, with the maximum dose of 10 mcg/kg/min.1
Hydralazine. Hydralazine (Apresoline) is a peripheral vasodilator, primarily of the arterioles. It decreases peripheral vascular resistance but increases heart rate and cardiac output. Hydralazine primarily is used for treatment of blood pressure in preeclampsia and eclampsia. It has significant adverse effects, including headache, vomiting, diarrhea, and palpitations. It can be given orally or parenterally, with the oral route being preferred. The usual starting dose for all forms is 10 mg.
Minoxidil. Minoxidil (Loniten) is a peripheral vasodilator that is reserved for patients who do not respond adequately to maximum doses of a diuretic and two other antihypertensive agents. Minoxidil may produce serious adverse effects, such as pericardial effusion, pericardial tamponade, and exacerbation of angina. The concomitant use of a loop diuretic almost always is required to prevent fluid retention and congestive heart failure. The use of a beta-blocking agent almost always is required as well to prevent increased heart rate, cardiac output, and thus myocardial oxygen demand. Minoxidil is not an agent that should be started by the emergency physician unless it is in conjunction with the patient’s physician and the patient is to be admitted.1
Fenoldopam. Fenoldopam (Corlopam) is a vasodilator that is used via titratable infusion to treat hypertensive emergencies. It is a dopamine1 receptor agonist with a half-life of about five minutes. It generally is well-tolerated, although it can cause dose-related increase in intraocular pressure and heart rate as well as mild hypokalemia. It is safe in pregnancy. The concurrent use of beta-blocking agents with fenoldopam is contraindicated. It has similar efficacy as nitroprusside but is more expensive. The dose range is 0.1-0.6 mcg/kg/min infusion. No bolus is needed.1
Diuretics. Diuretics long have played a role in the treatment of hypertension, both in combination with other agents or as monotherapy. Elderly and African-American patients respond more readily to diuretics than do younger and Caucasian patients. The antihypertensive effects of diuretics are multiphasic. Initially the plasma volume decreases, as does cardiac output and systemic blood pressure.2,3 Later, the plasma volume returns to normal but the blood pressure remains low due to reduced peripheral vascular resistance.2
Diuretics are divided into four main classes—thiazide-type, loop, potassium-sparing, and aldosterone antagonists.
Thiazide-type Diuretics. These include hydrochlorothiazide (Oretic), chlorothiazide (Diuril), methylclothiazide, and metolazone (Zaroxolyn). They interfere with sodium reabsorption in the early distal tubule. They are considered to be potassium-sparing agents. The antihypertensive effects are due to reduced blood volume and reduced cardiac output as well as some reduced vascular tone. They often are used to enhance the effectiveness of other antihypertensive drugs. These drugs generally are well-tolerated, but can cause hyperkalemia in patients with renal insufficiency, diabetes, or advanced age.22 Indications include edema associated with congestive heart failure, cirrhosis, or renal disease. These agents should not be used in patients who are anuric or are allergic to sulfa compounds. Adverse effects include hyponatremia, hypochloremic alkalosis, hyperkalemia, and hyperglycemia in diabetics. Hydrochlorothiazide often is found combined with other agents.
Loop Diuretics. Loop diuretics prevent sodium reabsorption in the distal loop of Henle. The agents are potent and have a rapid onset of action. They also are effective in patients with renal insufficiency. Furosemide (Lasix) and bumetanide (Bumex) are available parenterally and produce effective diuresis, especially in patients with pulmonary edema. Representative agents include furosemide, bumetanide, torsemide (Demadex), and ethacrynic acid (Edecrin).1,2
Potassium-sparing Diuretics. Potassium-sparing diuretics inhibit the sodium for potassium exchange in the distal tubule. Triamterene and amiloride (Midamor) are considered potassium-sparing diuretics. They are weak diuretics alone and often are used in combination with a thiazide.
Aldosterone Antagonists. Aldosterone antagonists include spironolactone (Aldactone) and eplerenone (Inspra). They are potassium-conserving diuretics as well that exert their effects by blocking aldosterone receptors. Spironolactone’s use is limited by its endocrine effects, as it has a binding affinity for androgenic and progestogenic receptors. Eplerenone is a newer aldosterone antagonist that seems to have a lower incidence of those side effects due to a lesser affinity for those receptors. In early trials, it demonstrated significantly decreased blood pressure effects both as a single agent or in combination with other antihypertensives.4
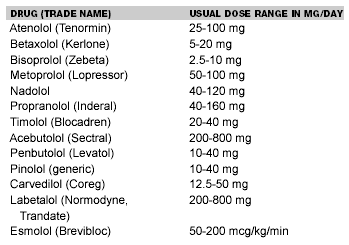
Beta-Blockers. Beta-blockers are first- or second-line agents for treatment of hypertension, either alone or in combination with diuretics. Commonly used beta-blockers include atenolol (Tenormin), metoprolol (Lopressor), propranolol (Inderal), esmolol (Brevibloc), and nadolol. Combined alpha- and beta-blockers include labetalol (Normodyne, Trandate) and carvedilol (Coreg).
The cardioselective and non-cardioselective beta-blockers are equally effective in the reduction of blood pressure. However, the cardioselective agents have lower risk of inducing bronchospasm. Most beta-blockers also have been shown to have anti-ischemic and anti-anginal properties and thus are recommended in patients with previous or ongoing myocardial infarction.5
Beta-blockers reduce blood pressure via multiple mechanisms, including reducing heart rate and cardiac output, blocking renin release, and blocking the production of angiotensin II and aldosterone.6 They generally are well-tolerated, with the most commonly cited side effects including dizziness, orthostatic hypotension, nausea and gastrointestinal effects, and fatigue.1 Contraindications to the use of beta-blockers include reactive airway disease, decompensated congestive heart failure, second-degree or higher heart block, severe bradycardia, or hypotension. Labetalol and carvedilol are combined alpha- and beta-blockers. Esmolol is an ultra-short acting beta-blocker that is used via continuous infusion. It is useful because the effects diminish rapidly when the infusion is stopped, thereby allowing any medication-induced hypotension to reverse. Beta-blockade is useful in patients with pheochromocytoma who have tachycardia, palpitations, and catecholamine-induced arrythmias. Beta-blockade must not be initiated prior to alpha-blockade because this may result in severe hypertension as a result of unopposed alpha stimulation from the circulating catecholamines.7 (See Table 2.)
Table 2. Beta-Blockers9
Calcium Channel Blockers. Calcium channel blockers (CCBs) are a class of antihypertensive drugs known for their heterogeneity. The CCBs are divided into two chemically distinct groups, dihydropyridines (DHPs) and nondihydropyridines. Each group has its own distinct binding site, but similar mechanism of action. The approved DHPs include amlodipine (Norvasc), felodipine (Plendil), nicardipine (Cardene), and nifedipine (Adalat, Procardia). The DHPs are recognized for their effect on vascular calcium channels, rather than on the cardiac channels. The non-DHPs include diltiazem (Cardizem, Dilacor, Tiazac) and verapamil (Calan, Isoptin, Covera, Verelan). They have dual action on cardiac and vascular smooth calcium channels; however, their cardiac influence predominantly is responsible for decreased automaticity and inotropy. The mechanism by which they exert their effect is based on the inhibition of intracellular calcium responsible for maintaining smooth muscle tone and cardiac contractility.
The short-acting CCBs have half-lives of 1.5-7 hours. Their rapid delivery causes a reflex neurohormonal activation of the sympathetic nervous system that leads to increased heart rate, increased cardiac output, and increased plasma catecholamines and renin activity. With prolonged use and scheduled dosing, these effects are retarded. The long-acting CCBs have half-lives of 35-45 hours. Multiple early studies documented increased cardiovascular risk with short-acting CCBs; however, recent large clinical trials assert that long-acting CCBs are not associated with increased cardiovascular risk.8 Adverse effects include flushing, pedal edema, constipation, dizziness, headache, hypotension, and fatigue. Relative contraindications include congestive heart failure because of the negative inotropic effects, which can lead to decreased cardiac output and coronary flow, especially with non-DHPs. (See Table 3.)
Table 3. Calcium Channel Blockers

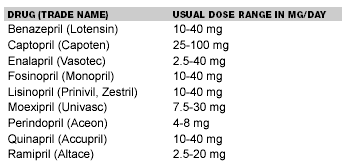
Angiotensin-converting Enzyme (ACE) Inhibitors. ACE inhibitors commonly are used in uncomplicated primary hypertension in white, young, or elderly individuals and patients with chronic congestive heart failure or a history of myocardial infarction. This drug is less effective in African-American patients due to their documented lower renin levels. The mechanism by which these drugs are successful in controlling blood pressure stems from the overstimulated renin-angiotensin-aldosterone system. These drugs block the angiotensin-converting enzyme that cleaves angiotensin I to form a potent vasoconstrictor angiotensin II. By inhibiting this action, not only is angiotensin II blocked from forming but also bradykinin inhibition is inactivated. Vasodilation results from lower levels of angiotensin II being available for vasoconstriction and the potent vasodilatory effect of bradykinin. Furthermore, angiotensin II levels are low such that secretion and activation of the aldosterone system is inhibited, resulting in less sodium and water retention.
Adverse effects include dry cough, rash, fever, altered taste, hyperkalemia, angioedema, first dose syncope, and leukopenia. Angioedema can occur even after the patient has been taking an ACE inhibitor for months. Relative contraindications include potassium supplementation or spironolactone, pregnancy, and renal artery stenosis.1,9 (See Table 4.)
Table 4. ACE Inhibitors9
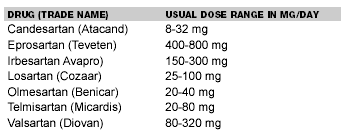
Angiotensin Receptor Blockers. The angiotensin receptor blockers are fairly new drugs used most commonly in patients who are unable to tolerate ACE inhibitors. The mechanism of action relies on the ability of effective blockade of the angiotensin type I receptor and not type II. As reviewed in the prior section, angiotensin I is converted to angiotensin II by angiotensin-converting enzyme. Angiotensin II acts on type I and type II receptors. The type I receptors are responsible for the known effects of angiotensin II, including vasoconstriction, increased sodium retention, suppression of renin secretion, increased endothelial secretion, increased vasopressin release, and initiation of sympathetic activity. The blood pressure effects are similar to those of ACE inhibitors. These drugs are more tolerable with fewer side effects. (See Table 5.)
Table 5. Angiotensin Antagonists9
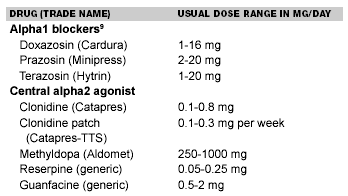
Anti-Adrenergic Agents (Alpha-Blockers). The alpha-blockers are a group of drugs that selectively antagonize the post-synaptic alpha1 receptors responsible for vasoconstriction. By blocking the alpha1 mediated constriction, there is a decrease in peripheral resistance with arteriolar and venous dilation. Since the presynaptic receptor is unblocked, the feedback loop of inhibition of norepinephrine release is intact. This action is responsible for less tachycardia, tolerance, and renin release. Alpha-blockers commonly are used in renal patients and those with obstructive prostatism since it relaxes smooth muscle of the bladder neck. Clonidine (Catapres) is a useful drug for emergency medicine because it has a short onset of action, lowers blood pressure predictably, and generally is well-tolerated. However, chronic clonidine users who abruptly discontinue its use can develop significant rebound hypertension. Its side effects include postural hypotension, reflex tachycardia, first dose syncope, dizziness, weakness, fatigue, and headache.1,9 (See Table 6.)
Table 6. Alpha1 Blockers and Central Alpha2 Agonists
Management of Hypertension in Specific Disease Processes
Hypertension and Ischemic Stroke. Elevated blood pressure at the time of ischemic stroke is common, probably due to a physiologic response that maintains adequate perfusion to the ischemic penumbra. Systemic hypertension also can be a reflection of the body’s attempt to maintain a cerebral perfusion pressure (CPP = mean arterial pressure-intracranial pressure) that becomes dependent on systolic blood pressure once the ischemic brain has lost its ability to autoregulate.10 Some authors believe that an increase in mean arterial pressure (MAP) in the setting of stroke might be protective. If this is the case, decreasing blood pressure might lead to further ischemic damage and stroke extension.11 The desire to do something to initiate secondary prevention without a full appreciation of the potential dangers associated with excessive blood pressure lowering can be detrimental.12
There is no general agreement on how blood pressure should be managed in the acute phase of ischemic stroke. Opinions range from not treating to treating if systolic blood pressure is greater than 220 mmHg or the diastolic blood pressure is greater than 120 mmHg.13,14
Elevated blood pressure in stroke is not considered an emergency requiring acute treatment unless associated with an acute MI or aortic dissection or if the patient is otherwise a candidate for thrombolytic therapy.10 The recommended blood pressure cutoff for patients receiving tissue plasminogen activator (TPA) is 180/110.15 However, TPA can be given if the blood pressure can be brought below 180/110 after one or two doses of medication.
A recent Cochrane review failed to find any evidence of a clinically meaningful benefit from any specific treatment for blood pressure in acute stoke.10 However, acute antihypertensive therapy was not associated with a worse outcome at three months among control patients in the large National Institute of Neurological Disorders and Stroke Recombinant Tissue Plasminogen Activator (NINDS-rTPA) stroke trial.11,16 Other studies are contradictory, noting that the lowering of elevated blood pressure in acute stroke can result in extension of the infarct and worsening stroke symptoms and has been shown to result in worse short- and long-term outcome.17-23
The latest American Heart Association guidelines recommend treatment of hypertension only if the systolic is greater than 220 mmHg or the diastolic is greater than 140 mmHg or if the MAP is greater than 130.10
If treatment of hypertension in acute stroke is deemed necessary, the ideal agent would be parenteral, easily titratable, have a short half-life, and have minimal effect on the cerebral vasculature.11 The drug that most closely fits this profile is labatelol. It can be given in IV increments of 10-20 mg every 10 minutes to a total dose of 300 mg. It easily can be switched over to oral therapy once the acute phase has passed. Direct vasodilators such as nitroprusside and nitroglycerin should be avoided, as they can cause increased intracranial pressure. Most oral agents should not be used in the acute setting as they are not easily amenable to titration. The overall goal blood pressure should be no lower than 185/110. Patients with persistent blood pressures greater than 185/110 should not receive thrombolytic therapy.24
Aortic Dissection. Chronic systemic hypertension is the most common factor predisposing to aortic dissection and is present in 62-78% of patients with aortic dissection.25-27 Dissection is thought to occur through aortic dilatation and high blood pressures superimposed on a structural weakness of the arterial wall.28 Pulsatile pressure extends the dissection by separating the layers of the arterial wall.11 Extension of the dissection is related to the aortic pulse wave and the MAP. The aortic pulse pressure is influenced by heart rate, contractility, and MAP.11 The goals are to reduce the force of the left ventricular contractions, to decrease the steepness of the rise of the aortic pulse wave, and to reduce the systemic arterial pressure to as low a level as possible without compromising vital organ perfusion.29 Intravenous antihypertensive therapy should be started on all patients except those that are hypotensive. The blood pressure should be lowered to a systolic of 100-120 mmHg. Dual agent therapy is preferred in most patients. A vasodilator such as nitroprusside or nicardipine will decrease the MAP but result in a reflex tachycardia. The tachycardia can be controlled with a beta-blocker such as labatelol.24
Congestive Heart Failure. Hypertension is a major cause of left ventricular failure, and control of high blood pressure slows the progression of heart failure and improves left ventricular function. The hypertension associated with acute pulmonary edema usually is the result of increased peripheral vascular resistance caused by elevated catecholamines. Occasionally, pulmonary edema occurs due to an abrupt rise in blood pressure that precipitates acute left ventricular failure (flash pulmonary edema) in a patient with no previous history of cardiomegaly.30 In addition to the standard therapy of oxygen and diuretics, vasodilators such as nitroprusside or nitroglycerin are used to decrease preload and afterload. Nesiritide is a recombinant B-type natriuretic peptide that also can be used to decrease preload and afterload as well as increased cardiac output. However, when compared to intravenous nitroglycerin, despite nesiritide producing a greater decrease in pulmonary capillary wedge pressure, there is no significant difference in dyspnea and global clinical status in decompensated congestive heart failure.31
If a patient requires mechanical ventilation to treat pulmonary edema, caution should be used in treating pre-intubation hypertension. Once an airway is secured and the patient is ventilating and oxygenating adequately, blood pressure may drop precipitously once the sympathetic stimulation of respiratory distress diminishes.
Cardiac Ischemia. Ischemic heart disease is the most common form of target end-organ damage associated with hypertension.32 The goal of blood pressure management in the setting of an acute coronary syndrome is to reduce myocardial oxygen demand and work while avoiding excessive blood pressure reduction that may worsen cardiac ischemia because coronary perfusion pressure depends on diastolic blood pressure.2 Reducing myocardial work by decreasing blood pressure and heart rate can reduce infarct size.11,33 In patients with acute coronary syndromes, hypertension initially should be treated with beta-blockers, ACE inhibitors, and nitrates.9
Nitroglycerin is a useful agent to treat hypertension in this setting because it is a coronary arterial dilator and reduces preload and afterload. Sublingual and intravenous nitroglycerin are the most effective forms used in acute coronary syndrome. Beta-blockade should be used during acute coronary syndrome as well, not only to control blood pressure, but to reduce myocardial oxygen demand. Labatelol or metoprolol are agents commonly used in this setting.
Pregnancy. Hypertension is an important cause of maternal and fetal morbidity and mortality and complicates 6-8% of all pregnancies.34 Hypertension in pregnancy is best divided into four categories: chronic hypertension; preeclampsia-eclampsia; preeclampsia superimposed on chronic hypertension; and gestational (transient) hypertension. Chronic hypertension is defined as elevated blood pressure (systolic blood pressure = 140 and/or diastolic blood pressure = 90 mmHg) that was either present prior to conception or was detected before the 20th week of gestation and does not resolve after delivery. Preeclampsia-eclampsia is a systemic syndrome characterized by hypertension occurring after the 20th week of gestation and usually is accompanied by proteinuria. Eclampsia is the convulsive phase of the disorder. Preeclampsia superimposed on chronic hypertension has a prognosis that is much worse than for either condition alone and can cause severe maternal and fetal complications.29 It can be difficult to distinguish superimposed preeclampsia from an exacerbation of chronic hypertension with underlying kidney disease. Nevertheless, it is better to err on the side of caution and overdiagnose preeclampsia rather than to miss it.35 Superimposed preeclampsia hypertension is highly likely in previously hypertensive women who have a new-onset of proteinuria, in women with hypertension and proteinuria before 20 weeks’ gestation, a sudden, precipitous increase in blood pressure (greater than 30 mmHg systolic or greater than 15 mmHg diastolic) in women with previously controlled hypertension; thrombocytopenia (fewer than 100,000 cells/mm3) or abnormal alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels.35 Gestational (transient) hypertension, a relatively benign condition, is elevated blood pressure occurring without proteinuria with onset late in pregnancy or the early puerperium that resolves within 12 weeks of delivery. The cause of preeclampsia is not known. Many consider the placenta to be the pathogenic focus for all manifestations of preeclampsia because delivery is the only definitive cure of this disease. Thus research has focused on the changes in the maternal blood vessels that supply blood to the placenta.34
The most commonly used drugs in treatment of preeclampsia/eclampsia include hydralazine and labetalol. Nitroprusside can be used if necessary. Magnesium sulfate is indicated as well, especially if seizures have occurred. Treatment should be done in conjunction with an obstetrician.
Intracranial Hemorrhage. Damage to the brain as a target organ as a result of hypertension may manifest in many forms, including acute chronic hypertensive encephalopathy, lacunar infarction, intracerebral hemorrhage, subarachnoid hemorrhage, ischemic stroke, and transient ischemic attacks. The most devastating hypertensive-related organ damage in the brain manifests as intracerebral hemorrhage (ICH). ICH usually is caused by arterial bleeding into the brain parenchyma from small penetrating arteries. These vessels, affected by long-term hypertension, have reduced compliance and an increased likelihood of spontaneous rupture. The most frequent anatomic locations for hypertensive ICH include the putamen, globus pallidus, external capsule, subcortical white matter, thalamus, internal capsule, the cerebellum, and brain stem.36 Shortly after vessel rupture, a large hematoma forms with adjacent cytotoxic and vasogenic edema. The progression of the edema is directly related to patient morbidity and mortality. ICH can present either suddenly or with a gradual onset. Findings are related to the anatomic location and size of the ICH. Universal symptoms include headache, nausea, vomiting, decreased consciousness, elevated blood pressure, and focal neurological deficits. Noncontrast CT remains the most rapid, reliable, and readily available neuroimaging technique in most EDs.36 It has a sensitivity of 100% in diagnosing ICH. Fresh blood appears hyperdense (white) on CT scan. The CT of the brain also provides information on the location, size/grade, presence of ventricular blood, surrounding edema, mass affect, hydrocephalus, or midline shift. The management of these patients includes vigilant airway surveillance and early intervention, blood pressure control, and intracranial pressure reduction (medical or surgical). Commonly held dogma dictates that blood pressure control in the setting of brain hemorrhage should be more aggressive than for patients with ischemic strokes so as to decrease the risk of continued bleeding. However, the relationship between rebleeding and systemic blood pressure is unproven, and overly aggressive lowering of blood pressure might decrease cerebral perfusion pressure and worsening brain injury.11,37 The American Heart Association/ American Stroke Association (AHA/ASA) guidelines recommend similar blood pressure control as in ischemic strokes with a MAP goal of 130 mmHg. Useful agents include nicardipine, labatelol, esmolol, and enalapril.
Renal Failure. The kidney is both the target and the cause of hypertension syndromes. Chronic hypertension is a common cause of end-stage renal disease. The kidneys typically are atrophic and there is mild proteinuria. In contrast, malignant hypertension results in enlarged kidneys and severe proteinuria. Severe hypertension in young patients should raise suspicion for intrinsic renal disease.
Acute renal failure associated with a severe elevation of blood pressure is considered to be a hypertensive emergency and should be treated aggressively. This is an uncommon presentation, however. Poorly controlled blood pressure that is associated with chronic renal failure is the more common scenario. The overall goal of hypertension treatment in patients with chronic renal disease is to slow progression of the renal disease. This often requires several medications. The rapidity of reduction in blood pressure is dependent on the severity of the associated symptoms, as with all hypertension syndromes. Too rapid reduction to a normal is associated with worsening renal failure. Suggested agents include beta-blockers, calcium channel blockers, nitroprusside, nitroglycerin, and clonidine. ACE inhibitors are used in chronically hypertensive patients with renal insufficiency, and caution should be used in patients who have hyperkalemia and acute uremia. If a patient has undiagnosed bilateral renal artery stenosis, use of an ACE inhibitor may precipitate worsening renal failure and severe hyperkalemia.11
Disposition
The Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC) guidelines are designed for primary care providers with no specific emphasis on the role of emergency physicians in the treatment of newly diagnosed or poorly controlled hypertension. However, the guidelines still can be applied to patients in the ED with special consideration for arrangement of follow-up and monitoring on an outpatient basis. Initial treatment strategies must include lifestyle modifications, including dietary changes, tobacco cessation, weight loss, increased physical activity, and decreased alcohol intake.
If the patient is considered likely to be non-compliant or unable to follow up for blood pressure monitoring, it would be acceptable to place this patient in a short-term observation unit for treatment and monitoring. Patients with hypertensive emergencies should be admitted to an intensive care unit (ICU) setting. Patients with severely elevated blood pressure can be admitted to at least a short stay unit or monitored unit for gradual reduction of blood pressure and initiation of oral antihypertensive therapy. Many asymptomatic patients with high blood pressure that is discovered incidentally can be discharged safely from the ED, with or without medication as long as adequate follow-up is emphasized.
This decision to start antihypertensive therapy from the ED is made by each physician individually, with the awareness that increasing numbers of patients are turning to the ED for their sole source of medical care, often due to lack of access to primary care providers. If the emergency physician decides to start antihypertensive therapy from the ED, some general guidelines can be applied. The key is to pick a medication that suits any underlying pathology and minimizes the chances of exacerbating it.2 It is reasonable to contact the physician that the patient is to follow-up with and discuss the choice of medication. A patient may resume a previously successful regimen as the simplest course of action. For patients previously diagnosed with hypertension that is poorly controlled, the dose of their current medication may be increased or an additional agent may be added. Monotherapy with most antihypertensive drugs effectively controls blood pressure in fewer than 50% of patients.2,38 The most common cause of resistant hypertension is volume overload, so adding a diuretic is a reasonable next step. Low-dose diuretics are highly effective in lowering blood pressure and often are used with other agents, such as beta-blockers, ACE inhibitors, CCBs, or angiotensin receptor blockers. Using such combinations may result in normotensive levels in up to 80% of patients, whereas monotherapy may be effective in only 40-50% of patients.23
For a new diagnosis of hypertension in a patient with no co-morbid illnesses, therapy with a thiazide-type diuretic alone is indicated for most patients. For patients with co-morbidities, certain agents are recommended over others, depending on the associated medical problem. The recommendations from the JC7 committee are summarized in Table 7.
Table 7. Compelling Indications for Individual Drug Classes9

Patients with renal disease often require multiple medications to control their blood pressure. Hypertension management in these patients is best done in conjunction with their nephrologist. Older, otherwise healthy patients can be started on a low-dose diuretic or long-acting dihydropyridine calcium channel blocker. African-American patients demonstrate reduced blood pressure responses to monotherapy with beta-blockers, ACE inhibitors, and angiotensin receptor blockers compared to diuretics or calcium channel blockers. They also have increased risk of angioedema associated with ACE inhibitor use.9
Summary
There is no one correct approach to management of each hypertension syndrome. Local practices vary, as do physicians’ opinions as to what constitutes a worrisome blood pressure. Many patients with elevated blood pressure can be discharged safely from the ED, with or without a prescription for an antihypertensive medication. Whatever course of action the physician chooses, the patient should understand the issues and importance of prompt follow-up and having the referral physician be involved in the management plan whenever possible.
References
1. Mosby’s Drug Consult, 2004 @ www.mdconsult.com/. Accessed 11-05-2004,
2. Moser M, Setaro J. Continued importance of diuretics and beta-adrenergic blockers in the management of hypertension. Med Clin North Am 2004;88:167-187.
3. Tarazi RC, Dustan HP, Frolich ED. Longterm thiazide therapy in essential hypertension: Evidence for persistent alteration in plasma volume and rennin activity. Circulation 1970;41:709-717.
4. Coleman CI, Reddy P, Song J, et al. Eplerenone: The first selective aldosterone receptor antagonist for the treatment of hypertention. Formulary 2002;37:54-524.
5. Gottlieb SS, McCarter RJ, Vogel RA. Effect of beta-blockade on mortality among high-risk and low-risk patients after myocardial infarction. N Engl J Med 1998;339:489-497.
6. Moser M, Setaro J. Continued importance of diuretics and beta-adrenergic blockers in the management of hypertension. Med Clin North Am 2004;88:167-187.
7. Braunwald E, Zipes DP, Libby P, eds. Heart Disease: A Textbook of Cardiovascular Medicine, 6th ed; Philadelphia: WB Saunders; 2001:941,968,972-992.
8. Eisenberg MJ, Brox A, Bestawros AN. Calcium channel blockers: An update. Am J Med 2004:116:35-43.
9. Chobanian AV, Bakris GL, Black HR, et al. The 7th report of the joint national committee on prevention, detection, evaluation and treatment of high blood pressure: The JNC 7 Report. JAMA 2003;289(19)L2560-2571.
10. Stead LG. Blood pressure control in acute stroke. Ann Emerg Med 2004;43:129-132.
11. Shayne PH, Pitts SR. Severely elevated blood pressure in the emergency department. Ann Emerg Med 2003;41:513-529.
12. Lindenauer PK, Mathew MC, Ntuli TS, et al. Use of antihypertensive agents in the management of patients with acute ischemic stroke. Neurology 2004;63:318-323.
13. Yatsu FM, Zivin J. Hypertension in acute ischemic stroke: Not to treat. Arch Neurol 1985;42:999-1000.
14. Spence JD, Del Maestro RF. Hypertension in acute ischemic stroke: Treat. Arch Neurol 1985;42:1000-1002.
15. Adams HP Jr, Adams RJ, Brott TG, et al. for the Stroke Council of the American Stroke Association. Guidelines for the early management of patients with ischemic stroke: a scientific statement from the stroke council of the American Stroke Association. Stroke 2003; 34:1056-1083.
16. Brott T, Lu M, Kothari R, et al. Hypertension and its treatment in the NINDS rt-PA stroke trial. Stroke 1998;29: 1504-1509.
17. Castillo J, Leira R, Garcia MM, et al. Blood pressure decrease during the acute phase of ischemic stroke is associated with brain injury and poor stroke outcome. Stroke 2004; 35:520-527.
18. Oliveria-Filho J, Silva SCS, Trabuco CC, et al. A detrimental effect of blood pressure reduction in the first 24 hours of acute stroke onset. Neurology 2003;61:1047-1051.
19. Fischberg GM, Lozano E, Rajamani K, et al. Stroke precipitated by moderate blood pressure reduction. J Emerg Med 2000;19:339-346.
20. Britton M, de Faire U, Helmers C. Hazards of therapy for excessive hypertension in acute stroke. Acta Med Scand 1980;207:253-257.
21. Lavin P. Management of hypertension in patients with acute stroke. Arch Intern Med 1986;14:66-68.
22. O’Connell JE, Gray C. Treating hypertension after stroke. BMJ 194;308:1523-1524.
23. Lisk DR, Grotta JC, Lami LM, et al. Should hypertension be treated after acute stoke? A randomized controlled trial using single photon emission computed tomography. Arch Neurol 1993;50:855-862.
24. Gray RO, Matthews JJ. Hypertension. In: Marx JA, Hockberger RS, Walls RM, et al, eds. Rosen’s Emergency Medicine, 5th ed. Mosby: St. Louis; 2002: 1158-1171.
25. Hagan PG, Nienaber CA, Isselacher EM, et al. The International Registry of Acute Aortic Dissection (IRAD): New insights into an old disease. JAMA 2000;283:897-903.
26. Spittell PC, Spittell JA Jr, Joyce JW, et al. Clinical features and differential diagnosis of aortic dissection: Experience with 236 cases (1980 through 1990). Mayo Clin Proc 1993; 68:642-651.
27. Hennessy TG, Smith D, McCann HA, et al. Thoracic aortic dissection or aneurysm: clinical presentation, diagnostic imaging and initial management in a tertiary referral center. Ir J Med Sci 1996;165:259-262.
28. Pretre R, Von Segesser LK. Aortic dissection. Lancet 1997;349:1461-1464.
29. Khan IA, Nair CK. Clinical, diagnostic and management perspectives of aortic dissection. Chest 2002;122:311-328.
30. Tintinalli JE, Kelen GD, Stapczynski JS, eds. Emergency Medicine: A Comprehensive Study Guide, 5th ed; New York: McGraw-Hill; 2000.
31. Publication Committee for the VMAC Investigators. Intravenous nesiritide vs. nitroglycerin for treatment of decompensated congestive heart failure: A randomized controlled trial. JAMA 2002;287:1531-1540.
32. Karras DJ, Wald DA, Harrigan RA, et al. Elevated blood pressure in an urban emergency department: Prevalence and patient characteristics. Acad Emerg Med 2001;8:559 (abstract).
33. First International Study of Infarct Survival Collaborative Group. Randomised trial of intravenous atenolol among 16,027 cases of suspected acute myocardial infarction: ISIS-1. Lancet 1986;2:57-66.
34. National Institute of Health. Working Group Report on high blood pressure in pregnancy, Bethesda: National Institutes of Health 2000; 2000. NIH Publication 00-3029.
35. Gregg AR. Hypertension in pregnancy. Obstet Gynecol Clin North Am 2004;31:223-241.
36. Panagos PD. Intracerebral hemorrhage. Emerg Med Clin North Am 2002;20:631-655.
37. Broderick JP, Adams HP Jr, Barsan W, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: A statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke 1999;30:905-915.
38. Victor R. Arterial Hypertension. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed; St. Louis: WB Saunders; 2004.
Part I of this series focused on hypertensive syndromes and clinical evaluation. This second and final part will cover antihypertensive medications and management of hypertension in specific disease processes.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
