Apparent Life-threatening Events
Authors: Ronald M. Perkin, MD, MA, Professor and Chairman, Department of Pediatrics, Brody School of Medicine at East Carolina University, Greenville, North Carolina; Elaine Cabinum-Foeller, MD, Assistant Professor of Pediatrics, Brody School of Medicine at East Carolina University, Greenville, North Carolina
Peer Reviewer: Robert Schafermeyer, MD, Associate Chairman, Department of Emergency Medicine, Carolina Medical Center, Charlotte, North Carolina
Infants with an apparent life-threatening event (ALTE) present for medical attention because an acute and unexpected incident has alarmed the caregivers. These frightening episodes of apnea and color change in infants have generated considerable concern in both professional and lay groups.
Although such episodes may occur in children older than 12 months, the majority of patients will be younger than 1 year and predominantly younger than 6 months.1,2 This age distribution and the fear engendered in the observer provide support to the possibility that such episodes, if undetected, may lead to the sudden and unexpected death of the infant. The hypothesis that these events might be premortem markers of the infant at risk for sudden infant death syndrome (SIDS) explains the large amount of attention, research, and clinical resources that have been focused on the problem of ALTEs in infants.2
This article reviews the presentation, recognition, diagnosis and ED management for children who present with an ALTE. — The Editor
Definition
The current definition of ALTE was established by the 1986 National Institutes of Health Consensus Development Conference on Infantile Apnea and Home Monitoring:3
"An episode that is frightening to the observer and is characterized by some combination of apnea (central or occasionally obstructive), color change (usually cyanotic or pallid, but occasionally erythematous or plethoric), marked change in muscle tone (usually marked limpness), choking, or gagging. In some cases the observer fears that the infant has died. Previously used terminology, such as aborted crib death’ or near-miss SIDS,’ should be abandoned because it implies a possibly misleading close association between this type of spell and SIDS."
Given the subjective nature of caregiver observations and interpretation, the initial medical problem for the health care practitioner is to determine which descriptions of the reported episode place the infant at increased risk for sudden death and future life-threatening episodes and which are a reflection of parental anxiety or of an acute nonrecurring problem. Health care providers, consistent with the recommendations of the conference, often make the assumption that reports of frightening episodes of apnea or respiratory difficulty are clinically of greater severity, and thus, more apt to be of medical significance (changes in skin color or muscle tone, or an episode that requires vigorous stimulation, mouth-to-mouth resuscitation, or cardiopulmonary resuscitation [CPR]).4 Conversely, many conclude that the reported episode is not medically important if not associated with these latter observations or not requiring some type of vigorous resuscitative intervention. However, these assumptions may not be accurate; studies have demonstrated that parental reports of an acute event are a poor reflection of an infant’s physiologic status.2,4-6 Until such a time when objective measures with adequate sensitivity and specificity exist to assist in making medical decisions (e.g., hospital admission, specialized clinical follow-up, home apnea monitoring), it may be necessary to rely primarily on clinical markers and expressions of parental concern that they had observed an episode sufficiently frightening to them to bring the infant to medical attention.4,7
An infant with an ALTE may present to health care personnel without clinical signs or symptoms of significant illness.7-10 The greatest difficulty for emergency physicians usually is deciding how to pursue a diagnosis when faced with infants who appear perfectly well. Most reports recommend a mandatory period of inpatient observation.8,9,11 Therefore, in evaluating this group of patients, the ED diagnostic evaluation has a limited role beyond the information provided by the history and physical examination.10
Incidence
Various estimates place the incidence of ALTEs in the general population at between 0.5% and 6% of all infants.2 The higher ranges of reported incidence (4-6%) come from studies in which parents were questioned as to whether their infant had ever turn-ed blue or stopped breathing. However, this mode of ascertainment tends to overestimate incidence.2,7,12 For a multitude of reasons, the true frequency and prevalence of ALTEs are unknown.12
Relationship to Sudden Infant Death
The most common category of death in infancy is that which is sudden and unexplained, despite a thorough autopsy, examination of the scene of death, and review of the case history (SIDS). Probably, the mechanisms that cause sudden and unexpected death in an infant are similar, in part, to some of the mechanisms that cause ALTEs. Whereas most studies report survival in in-fants who have had an ALTE, a small proportion of infants who suffer recurrent ALTEs progress to sudden and unexpected death.1 A small proportion of infants who die of SIDS have a history of a previous ALTE or apnea: 7% in the National Institute of Child Health and Development Cooperative Epidemiological Study of SIDS in the United States3 and 8.8% in an Australian series.13
Compared with SIDS victims, infants with ALTEs present earlier in life; they are 1 to 3 weeks younger than SIDS patients.12 Also, 82% of the ALTEs occurred between 8 a.m. and 8 p.m., when the caretakers are awake.12
Considered from a different, and perhaps more important, perspective, the incidence of sudden and unexpected death in ALTE infants appears to vary depending upon the initial presentation. Those infants whose frightening episode occurred during sleep and were perceived to require cardiopulmonary resuscitation have a 10-13 % risk of subsequent sudden unexpected death, even with the prescription of home monitoring devices.3,14 Infants who responded only to resuscitation and have a subsequent similar episode, who have sibling victims of SIDS, or who develop a seizure disorder during monitoring have a greater than 25% risk of dying.5 The precise risk for infants who experience less severe ALTEs has not been determined, but in many cases it may be no greater than the general population. Identification of a specific cause of ALTEs does not necessarily eliminate the risk of sudden and unexpected death.3
For these reasons, infants who suffer ALTEs have been considered a high-risk group for sudden death, and their study has been of particular interest. Addition of these data helps our understanding of the pathophysiology of events in living infants that may produce sudden death and complements the approaches from pathological and epidemiological studies.
Etiology
There are multiple identifiable causes of ALTEs; after evaluation approximately 50% of these infants will have a specific diagnosis.2,3,12,14,15 Symptoms reported by parents greatly contribute to establish most diagnoses.1,12 ALTE can be a symptom of many specific disorders, including gastroesophageal reflux, infection, seizures, airway abnormalities, hypoglycemia, or other metabolic problems, as well as impaired regulation of breathing during sleep and feeding. These episodes can occur during sleep, wakefulness, or feeding and are in infants who are generally of more than 37 weeks gestational age at the time of onset.3 Clinical management of such infants does not necessarily require an exhaustive list of investigations to find an abnormality, but rather a careful and focused clinical assessment of each case, commencing with history and examination and, usually, a period of inpatient observation.
Diagnosis
Infants often appear entirely normal by the time they reach medical attention following the ALTE. The most important initial diagnostic step is to obtain a careful history.
A clear description of the event should be noted, including who was present, what each person observed, and what their actions were. The timing of the event should be noted, including whether the infant was awake or asleep and what activity he or she was performing. It may be of value to determine how it was discovered that the event was occurring. It is not uncommon that parents/caregivers discover such cases by chance.
Record the infant’s position, whether there was movement, and how the infant was holding itself. Note the color of the infant (e.g., red, blue, purple, pale, gray, or white). Clarify whether color change has occurred on the face as a whole (central cyanosis), just around the eyes and mouth, or in the limbs. Record the presence of vomit or blood from the nose and mouth. Vomiting may reflect infection, gastroesophageal reflux, or excessive force from resuscitation attempts. Blood from the nose may be a marker for trauma and an event that was induced intentionally. Ask about the presence or absence of breathing movements; it may be difficult for observers to report this finding reliably. The child’s shallow breathing efforts may make it difficult to detect any chest wall movement. Even a nurse’s observations of breathing in infants receiving hospital neonatal care may correlate poorly with recorded breathing patterns.1,16,17
Note the actions of the observers as well as the response of the infant to these actions. Note the time scale for these actions and the recovery of the infant.
Obtain a general pediatric history of the mother’s pregnancy and delivery as well as the infant’s birth and neonatal progress. A history of feeding, weight gain, development, and recent minor symptoms should be noted, and obtain a detailed family medical history and social history. The latter should include who else is at home, other stresses and medical problems within the family, and any contact with psychiatric or social services.
The medical history also should be probed for evidence of previous frightening episodes, abnormal breathing patterns, feeding problems, seizures, prenatal insults, and other medical problems. A family history of apnea, infant deaths, seizures, or other cardiopulmonary problems should be explored.
A full clinical examination, with the infant completely undressed, should be performed. Record the height, weight, and head circumference of the infant and plot the results on a growth chart. Pay particular attention to examination of the upper airway, respiratory, and cardiovascular systems. Neurodevelopmental assessment is important because a number of neurological conditions may present with apnea. Funduscopy should be attempted to detect evidence of trauma. All infants should undergo an initial spot measurement of arterial oxygen saturation (SpO2); monitor SpO2 for a longer period in infants who were preterm or who may have a respiratory prodrome.
After taking a careful history and performing a physical examination, the physician is in a position to make an initial judgment about whether the event represents an ALTE. If not, usually no further diagnostic evaluation is required. If a physician believes that the infant did have an ALTE, then an aggressive approach to identifying the etiology of the event and instituting appropriate therapy is necessary (Figure 1).
Figure 1. Investigation of First ALTE
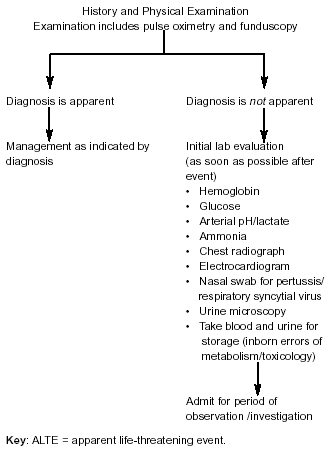
Hospital admission for protective monitoring, to facilitate the diagnostic evaluation, and for parent training is recommended.12,14,18 The diagnostic evaluation may include, but is not limited to, the items listed in Table 1. Although not every infant will require all of these tests, many of them often are performed before the episodes are termed "unexplained".
Table 1. Suggested Evaluation of an Infant with an ALTE

Management
Parents of an infant who has suffered an ALTE are usually anxious and may feel that their infant almost died. For this reason alone, it is probably appropriate to admit all infants who have suffered a first ALTE. This action allows time for a full assessment, a period of observation, and discussion with the family.
A recurrence rate for severe ALTEs as high as 68% has been reported; for all ALTEs, episodes are more likely to recur in the first few days after the event.1 These data provide further support for an initial hospital admission. Some ALTEs may be recurrent within a short period of time (hours to days). These subsequent events may be observed and a diagnosis made by clinical observation. In addition, clinical physiological monitoring or recording allows documentation of any further events, and their findings may indicate a diagnosis.1,19 Both respiratory illnesses, such as respiratory syncytial virus (RSV) and pertussis, and epileptic seizures may cluster in this way.1
No study has identified any single investigation as having a high positive predictive value for detecting an abnormality that will alter the outcome. Thus, there is much controversy as to which initial investigations should be undertaken in an infant presenting with a first ALTE. In most cases, it is not unreasonable to check serum sodium, potassium, and calcium levels, and order renal and liver function tests, complete blood count measurements, blood and urine cultures, throat/pharyngeal swabs, and chest x-rays. (See Figure 1 and Table 1.)
If the infant presents to the ED very soon after the event, an arterial pH measurement may help to provide documentation of the severity of the episode.20 Other markers of metabolic disturbance, such as blood glucose, lactate, and ammonia levels, also could be measured at this time. The patient should be observed carefully in the ED, particularly for the development of fever, or other signs of sepsis, and the clinician should have a low threshold for treating sepsis in infants younger than 4 weeks. Other tests that should be considered include nose swabs/pharyngeal aspirates for viral immunofluorescence, culture and pertussis culture; urine samples for microscopy, bacterial culture, and antibiotic sensitivities; specimens for urinary and blood metabolic screens;21 and urine and blood samples for toxicology screens. Parents/caregivers may not always provide complete information about medicine given to infants, some of which may have been administered inadvertently and some deliberately.
A majority of infants suffer a single event only and survive. Adverse outcomes are more likely with infants who have had recurrent events or where there is a family history of SIDS. In the absence of these, investigations should be kept to a minimum in an otherwise well infant.
Infants in whom a treatable cause of the ALTE can be identified are best managed by treating the specific etiology of the event. (See Specific Causes of ALTEs section.) Occasionally, the addition of home apnea-bradycardia monitoring may be required.3,14,18, 22-26
The diagnosis of apnea of infancy (AOI) is used when an identifiable cause for the ALTE cannot be found.3 Presently, there are no specific treatments for AOI; thus, home apnea-bradycardia monitoring for these infants often is recommended.3,14,18
Home monitors only alert the caregiver that a potential episode is occurring, and the caregiver must then respond to evaluate and/or terminate the episode and record the findings in a logbook or diary. Parents and caregivers must be trained in the proper operation of the monitor, a graded response to monitor alarms, and infant cardiopulmonary resuscitation. Thorough education of parents and psychosocial support of families using monitors are important for successful home apnea-bradycardia monitoring. Monitoring can be both a source of stress and a source of support and reassurance for parents.
It may be difficult to distinguish true apnea or bradycardia alarms from loose lead alarms or alarms for nonsignificant events. Documented monitoring, with event recorders built into the monitors, provide objective recordings of apnea and bradycardia alarms, and may be helpful in making these distinctions as to whether the infant actually was having an apnea or bradycardia during a given alarm.27 In addition, it provides information regarding compliance with monitor use, because the length of time the monitor was turned on each day is recorded. In a large consecutive series of referred monitored patients, utilizing event recordings, only 8% of 14,131 events were caused by true apnea or bradycardia.27 Of those true events, 70% were triggered by apnea and 30% by bradycardia. The true events occurred in 48% of patients. Loose lead alarms accounted for 69% of all events and false alarms for 23%.27 However, documented monitoring also may help confirm the caregiver’s observations regarding the clinical significance of episodes. In the usual clinical pattern, a true alarm will decrease in both frequency and severity with time. Infants whose alarms become more frequent or severe, those infants with multiple alarms requiring intervention, and those infants who continue to have true alarms after 6-8 months of monitoring require further diagnostic evaluation, including home event recordings with oximetry or overnight polysomnography.28-30 With severe episodes, these infants may require hospitalization for observation and further diagnostic evaluation. Sometimes the character of the events may change, suggesting the presence or development of other clinical problems, such as a seizure disorder, which also requires specific evaluation.
The criteria for monitor discontinuation should be based on the infant’s clinical condition. Clinical experience and the literature support monitor discontinuation when infants with ALTEs have had 2 to 3 months without significant alarms or apnea.3 Additionally, assessing the infant’s ability to tolerate stress (e.g., immunizations, illnesses) during this time is advisable.14
Specific Causes of ALTEs
Infections. Apnea can be the initial presenting symptom in some infants with respiratory syncytial virus (RSV) infections.31-34 Although the apnea can be life-threatening, episodes usually are of short duration and occur during the first week of illness. Apnea in infants with RSV bronchiolitis occurs most commonly in young infants (younger than 3 months) with a history of premature birth and apnea of prematurity.31,33 The apnea occurs primarily in quiet sleep and is not obstructive. The exact pathophysiology of apnea in RSV infection remains unclear. Lindgren and colleagues suggested that RSV might alter the sensitivity of the laryngeal chemoreceptors leading to laryngeal chemoreflex and central apnea.35 Infants with bronchiolitis also may develop apnea from increased work of breathing, respiratory muscle fatigue, and hypoxemia.32
Apnea also may complicate other infections including meningitis, encephalitis, bacterial sepsis, botulism, pertussis, and other respiratory infections.32
Discussions of the differential diagnosis in children presenting with an ALTE often do not mention urinary tract infection (UTI); a causal link between UTI and ALTEs has been made.36 Therefore, it is suggested that clinicians consider obtaining a urine culture in infants with an ALTE.
Seizure-induced ALTEs. Seizures can be associated with apnea and hypoxemia. Seizures have been shown to occur in 4-15% of infants with ALTEs; such infants have a particularly high risk of sudden death.3,12,37 However, it is often difficult to prove whether seizures resulted from or caused an ALTE. Usually, the possibility of seizure-induced events are considered on clinical grounds (e.g., clustering of attacks, facial/eye movements, or an increase in muscle tone); however, apnea may occur as the only clinical manifestation of seizures in neonates, infants, and young children.38-40
ALTEs that result from a seizure disorder frequently follow a characteristic pattern when carefully studied. First there is a change in the electroencephalogram (EEG), followed by one or more pauses in breathing movements, and ultimately a decrease is oxygen saturation in the presence of sinus tachycardia.38
EEG is a recommended procedure in the investigation of ALTE, to exclude central nervous system disorders, even in the absence of clinical clues.37
Airway Obstruction. Investigators using overnight poly-somnography have found that following an ALTE, infants have increased obstructive and mixed apneas compared with non-risk groups, and some infants who have had ALTEs later die of SIDS.15,41-44 The obstructed breaths occurred mainly in rapid eye movement sleep and were accompanied by drops in heart rate and oxygen saturation.41 The patients who later died of SIDS also were noted to have more frequent episodes of regurgitation after feeding and to move less during sleep.41 Such findings are in agreement with reports suggesting that although obstructive apneas are rare in normal infants,45 they may play a role in ALTEs and in SIDS.15,41-44 Obstructive apneas may be related to craniofacial malformations, gastroesophageal reflux, bulky arytenoid apparatus, central nervous system dysfunction, or vascular malformations.15,46,47
Recent evidence suggests that obstructive sleep apnea (OSA) in adults and sudden unexpected infant death/ALTE in their biologic relatives appear to be related.48-50 Familial factors influencing this association may include the degree of the predilection for OSA, liability for respiratory illness or allergy, dimensions of the oral-pharyngeal airway, and ventilatory response to hypoxia.48 These findings suggest that SIDS and ALTEs are related to OSA.49 Polysomnography is required to establish the diagnosis of obstructive apnea, as medical histories often are misleading.46
Breath-holding Spell (BHS). Cyanotic breath-holding episodes have been appreciated as a medical problem in pediatrics for many years. They are a common phenomena: Approximately 3% of all children manifest these episodes sometime during their early childhood; more than 25% begin before 6 months of age.51
There are two main clinical features of cyanotic breath-holding episodes: a) The first is a prolonged expiratory apnea, and b) the second is the rapid development of central cyanosis, reflecting severe hypoxemia.52
Episodes begin usually in response to a sudden painful or unpleasant stimulus with the infant or child becoming rapidly cyanotic (usually within 5 seconds of the onset of the event) and holding his/her breath. The characteristics of the breath-holding spell are that of prolonged expiratory efforts without inspiratory efforts. However, on some occasions, the cyanosis may occur despite continued breathing.53
Although the episodes often are precipitated by a cry or an attempt to cry; during the episodes the patient usually remains silent. As the apnea and cyanosis progress, the patient may develop an opisthotonic posture, usually at around 10 to 20 seconds into the attack. Loss of consciousness usually occurs within 30 seconds and may be followed by a generalized seizure.54 Recovery is heralded by a gasp, which often appears to be simulated by a sudden shock to the patient, such as the application of cold water, blowing in the face, or an attempt at mouth-to-mouth resuscitation. On recovery from an episode, the patient may remain drowsy, pale, and may sleep for several hours. Sometimes one attack will follow another within minutes with the succeeding episodes being more severe. These are frightening situations for parents, and many parents believe their child to be dying during such episodes. The application of mouth-to-mouth resuscitation by parents in this situation is not uncommon. In many patients with such cyanotic/apneic episodes, crying without apnea also is accompanied by central cyanosis.
In the majority of instances the cyanotic breath-holding episodes begin within the first year of life, with some starting within the first few days of life.54 In one report, the symptoms began at a median age of 7 weeks; 77% began at ages younger than 4 months.53
Central nervous system sympathetic activity in response to an environmental stimuli (e.g., stress) may produce the prolonged expiratory apnea through effects on the brainstem respiratory centers and right-to-left intrapulmonary shunting through effects on the pulmonary vasculature. These episodes are involuntary and reflexive and occur during active or full expiration.54 Other indications of an autonomic dysfunction can be derived from a literature review.53-56 Infants with BHSs have breathing disorders during sleep. Compared with a control group, infants with BHSs have obstructed breathing, snoring, and sweating during sleep.55
Breath-holding spells have been associated with several structural neurologic problems, including medullary tumor, bilateral abductor vocal cord paralysis, hydrocephalus, and Arnold Chiari malformation. An association with complex partial seizures has been described.57
Pallid BHSs are associated with severe bradycardia or asystole, whereas cyanotic BHSs are not.56 Pallid BHSs is a misnomer, for they are not breath-holding spells at all, but rather vasovagal syncope.
The evaluation of BHSs consists of a careful history; laboratory testing usually is not indicated unless accompanied by other findings from the neurologic examination. Treatment consists of parenteral reassurance. The spells are outgrown, usually without residua.
Gastrointestinal Causes of ALTEs
A high percentage of identifiable etiologies of ALTE are the result of a gastrointestinal (GI) problem.12 Investigation of a potential gastrointestinal cause for an ALTE begins with the history, which is often the most important part of the entire GI evaluation.
Was the episode related to feedings?
If there seems to be a temporal association between feeding and the events, it is important to differentiate distress during swallowing from distress after the infant finishes a meal. This may differentiate structural problems, such as H-type tracheoesophageal fistula (TEF) or laryngeal cleft, from gastroesophageal reflux (GER) induced events.
In the newborn period, feeding occasionally may be accompanied by symptoms such as coughing and choking, and signs such as cyanosis and apnea. These feeding-related problems may be life-threatening.58 An upper gastrointestinal series (UGI) should be the first radiological study in these patients; it will be diagnostic for H-type TEF or any other anatomical problem. However, in patients with a normal UGI and continued symptoms, a modified barium swallow should be performed to exclude swallowing dysfunction and aspiration.58
How long after feeding do infants with events develop symptoms?
Often, infants with GER have an associated delay in gastric emptying, which may manifest as regurgitation of milk many hours after it was ingested.59
Does vomiting occur, or was there evidence of regurgitation during the event?
Although half of all healthy infants between 2 and 8 months regurgitate two or more times each day, and 15% vomit three or more times daily, regurgitation leading to an ALTE is considered a red flag for pathological GER and requires further evaluation.60,61
Have there ever been any problems with weight gain?
Poor weight gain due to excessive vomiting or poor intake due to pain with eating can lead to growth problems, and also is a red flag for pathological GER, requiring further evaluation. However, certain central nervous system disorders (e.g., diencephalic syndrome) or metabolic diseases may manifest with poor weight gain and ALTEs.
Contrast radiography using barium generally is the best study to define the anatomy of the upper digestive tract in infants with ALTEs. Anatomic abnormalities of the esophagus, cardioesophageal junction, gastric outlet, and duodenal sweep easily are defined in this manner. More unusual anatomic causes of ALTE, such as gastric volvulus, can be identified with contrast radiography.62 However, because it is a static rather than dynamic test, it is neither sensitive nor specific in diagnosing GER, which is one potential GI-related cause of ALTE. Similarly, contrast radiography may be suggestive, but not diagnostic of esophageal motility disorders. These are commonly found in infants with birth defects, such as esophageal atresia post-repair, or in children with chromosomal abnormalities, such Trisomy 21 or Cornelia de Lange Syndrome. They also may be found as primary lesions as in achalasia. All of these motility disorders can present in infancy with ALTE, especially when regurgitation is noted, along with choking and obstructive apnea with cyanosis.
Gastroesophageal Reflux
Gastroesophageal reflux (GER) is characterized by the effortless passage of gastric contents into the lower esophagus. Reflux can be classified as physiologic, in which the infant remains free of clinical sequelae, pathologic reflux or gastroesophageal reflux disease (GERD), in which gastrointestinal, pulmonary, or neuropsychiatric complications are associated with intraesophageal acidification.60 A classification that particularly is useful to the clinician categorizes reflux by its expected natural history. Thus, infantile reflux, which results from a delay in the acquisition of normal upper gastrointestinal motility, is likely to resolve by the child’s first birthday. In contrast, childhood GER, although it may begin during infancy, appears to be a chronic disorder similar to reflux encountered in the adult population.
Infantile apnea remains the most controversial of the possible relationships between GER and pulmonary disease. Intraesophageal acidification results in two clinically distinct patterns of apnea. Awake apnea is characterized by a sudden staring or startled appearance within the hour after a feeding and often is preceded by a change in position.63 A history of choking, coughing, or vomiting is obtained for a minority of these infants. Spitzer and colleagues have confirmed the temporal relationship between intraesophageal acidification and awake apnea and the response of these infants to anti-reflux therapy.39 The majority of those children had some degree of obstructive apnea during the periods of GER.
Although there is general agreement that awake apnea is reflux related, there is less consensus regarding the relationship between sleep apnea or ALTE and GER. Attempts at confirming a temporal relationship between reflux and sleep apnea have produced variable results.60 Episodes of reflux occur more frequently in infants when awake than when asleep, although when episodes do occur during sleep, they tend to be of longer duration.64 The duration, and not the frequency, of these episodes during sleep may be an important determinate of both reflux associated respiratory disease and ALTE.64-66 Nocturnal reflux is uncommon, but when it occurs in infants, it is more apt to occur during active sleep than during quiet sleep. In infants, active (rapid eye movement) sleep is accompanied by reduced pulmonary oxygen reserves, depressed respiratory muscle responsiveness, and reduced upper esophageal sphincter pressure; therefore, reflux during sleep may occur at a time of increased vulnerability of the respiratory system.67 Children with prominent nocturnal cough and wheezing are especially likely to have nocturnal reflux. Additionally, nocturnal reflux predisposes to esophagitis because of the prolonged esophageal acid exposure that occurs during sleep.67
Another mechanism by which GER or pharyngeal incoordination causes apnea is by stimulation of chemoreceptors around the larynx that respond to a variety of stimuli, such as gastric acid and water; the reflex response comprises central apnea, bradycardia, and pallor due to central pooling of blood.2 In infants, a pattern of isolated bradycardia associated with irregular respiration while sleeping is highly suggestive of GER and should prompt further evaluation and treatment.68
Apnea may arise from either esophageal or laryngotracheal reflux events. This can occur from the presence of direct aspiration, altered gas exchange, or stimulation of the laryngeal chemoreceptor reflux.69 An acid solution instilled into the esophagus can provoke obstructive apnea, as well as central and mixed apnea, and is related to maturity.70
Many studies support the sequence of events in which GER provokes the apneic episode. In infants with severe GER, irregular breathing frequently is observed.70 Furthermore, conservative or surgical treatment of GER decreases the apneic events.
In contrast to the proposed mechanism of GER leading to apnea, other investigators have suggested a reversed order of events (i.e., apnea followed by GER).70,71 Arad-Cohen and colleagues examined the temporal relationship between GER and apnea in 21 infants with intermittent episodes of reflux and a history of ALTEs.70 They concluded that GER is frequent in infants with ALTEs, and obstructive and mixed apnea preceded the GER episode. The increased frequency of mixed and obstructive apnea in infants with ALTEs raises the question of whether the two phenomena coexist in this patient population as a result of maturational defect in the central control of the upper airway and the lower esophageal sphincter.71
The infant presenting with an ALTE and symptoms suggestive of GER is best evaluated with a 24-hour intraesophageal pH study. This test has been the gold standard in identifying pathological GER for more than 20 years. In using the 24-hour pH monitor to evaluate infants with ALTEs, clinicians have focused on correlating reflux events with changes in cardiorespiratory status. Events that mimic the described ALTE and that are preceded immediately by a reflux episode on the pH recording are said to be caused by GER. However, the changes that are noted in cardiorespiratory status may not replicate the symptoms that brought the infant to medical attention, and in fact, most often the ALTE is not repeated during the monitored period. This can make it very difficult to prove a cause-and-effect relationship between GER and the ALTE.
To identify infants with respiratory symptoms from GER, Jolley and colleagues evaluated the mean duration of reflux during sleep (ZMD score).72 They found that the ZMD score provided the best separation between two groups of patients: those with GER and respiratory symptoms caused by reflux and those with reflux and respiratory symptoms due to other factors. In children with reflux-related respiratory symptoms, 94% had a ZMD score of more than 4 minutes. Jolley and colleagues then examined their 10-year experience with 499 infants who had undergone extended esophageal pH monitoring.73 Of the 19 deaths found in the series, three were classified as SIDS, and two were in-hospital deaths caused by reflux-induced aspiration. All five of these infants had elevated ZMD scores when tested for GER, and the authors suggested that a cause-and-effect relationship exists between GER and sudden death in nearly 10% of infants with documented GER and elevated ZMD scores. Although they concluded that surgical management was the most effective and appropriate therapy in this specific pattern of GER, medical management generally has been helpful in most infants, allowing surgery to be saved for only the most refractory cases. When medical therapy is chosen, and a cause-and-effect relationship truly has been established between GER and ALTE, a home apnea/bradycardia monitor may be prescribed to alert the caretaker to further episodes when the infant is asleep or unattended.
When aspiration of foodstuffs is suspected, bronchioalveolar lavage (BAL) is a helpful tool in finding direct evidence of such an event.74 BAL is another procedure that can help support (but not prove) an exact GI-related etiology for the ALTE.
Inborn Errors of Metabolism
The rare association between inborn errors of fat oxidation and ALTE or with SIDS-like deaths has been described.21,75 An inborn error of metabolism is more likely to be associated with an ALTE if the initial ALTE is severe and if there is a family history of ALTE, consanguinity, seizure disorders, or SIDS.21 Inborn errors of fat oxidation may be apparent only during times of metabolic stress, such as fasting associated with an infectious illness, when the infant is forced to utilize fatty acids as substrates for energy production, rather than carbohydrates. The clinical presentation may include nonketotic-hypoglycemic attacks in a previously healthy infant, triggered by a physical stress or a period of fasting. Progression of symptoms may mimic an ALTE. Medium chain acyl-CoA dehydrogenase deficiency (MCADD) is the most common of these disorders, and the most likely to present as an ALTE. Inborn errors of beta-oxidation of fatty acids can cause up to 4% of severe ALTEs and up to 5% of cases of SIDS.21,75 Treatment of MCADD involves the avoidance of fasting, L-carnitine supplementation, a low-fat/high carbohydrate diet, and home apnea-bradycardia monitoring for infants with a history of ALTE.
In each infant with an ALTE, a blood NH4 should be considered as a screening test. A more thorough diagnostic evaluation is recommended for ALTE infants with: 1) an elevated serum NH4 level; 2) a positive family history for AOI, seizure disorders, SIDS, or other sudden infant deaths; 3) laboratory evidence of hypoglycemia, hyperammonemia, metabolic acidosis, elevated liver enzymes, or abnormal hemostasis; and/or 4) a positive patient history of unexplained failure to thrive, developmental delay, or seizures. More extensive laboratory evaluations would include blood and urine carnitine, urine nonvolatile organic acids, urine acylglycine, and blood and urine acylcarnitines measurements.
Nonaccidental Head Injury
The first reports of child abuse presenting as ALTEs occurred in 1979.76 Since then, multiple publications have documented an association between child abuse and ALTEs.
The link between ALTEs and abusive head injury was confirmed in 1995, when five patients admitted for evaluation of an ALTE were found to have shaken baby syndrome.77 The initial history and physical examination revealed no apparent cause, but the discovery of retinal hemorrhages in four of the infants and the development of focal seizures in the fifth patient prompted further evaluation that led to the diagnosis of child abuse.
In two recently reported series, one in 40 ALTE admissions were linked to abusive head injury.78-79 Because an infant who has sustained an abusive head injury may appear well on presentation, with no external signs of abuse,80,81 intentional head injury must be considered in a patient who has an ALTE unless an alternative cause is readily apparent. Therefore, unless physical examination and initial test results strongly suggest another cause, the clinician should consider taking steps to evaluate a possible inflicted injury, beginning with a dilated funduscopic examination and head injury studies.
Several forms of child abuse, other than inflicted head injury, have been implicated in ALTEs: intentional poisoning, intentional smothering, and Munchausen Syndrome by Proxy.79 Because all of these are difficult to diagnose, physicians must be vigilant to avoid overlooking instances of child abuse.
Munchausen Syndrome by Proxy/Factitious Illness by Proxy
Child abuse as a cause of ALTE is difficult to diagnose. The circumstances associated with child mistreatment may range from a sudden isolated loss of control by a parent to circumstances in which there is a long-standing catalog of premeditated and intentional acts of harm to the child. The latter form of abuse may involve injuries such as fractures of different ages, deliberate burns or scalds, pinch or human bite marks, and the induction of illness in the child.20 One particular symptom, namely recurrent apneic or cyanotic episodes, may have occurred as a result of intentional suffocation.19,20,82-88
Munchausen syndrome is a bizarre illness in which a patient either fabricates or actually causes symptoms that require medical intervention. First described by Asher in 1951, the name is derived from Baron von Munchausen, an 18th century mercenary who became known for fanciful and wildly embellished tales of his travels abroad.89 Munchausen syndrome by proxy (MSBP), first described in 1977 by Meadow, is a form of child abuse in which a parent, usually the mother, systematically fabricates information about her child’s health or intentionally makes the child ill.90,91 When a parent induces a life-threatening illness or fabricates an illness, resulting in invasive diagnostic, anesthetic, or surgical procedures for the child, the case becomes one of child abuse.92 The psychodynamics involved in these situations are quite different from those operative in the typical child-abusing parent.92 The parent who is causing the illness (most often the mother) displays model behavior, and therefore, the diagnosis of induced or factitious illness often is unsuspected and hence, delayed. In one review, the mean time for onset of signs and symptoms of illness to the diagnosis of MSBP was 14.9 months.93 Considering the 10% mortality associated with the syndrome, early awareness of the associated signs and symptoms by physicians is important.94
The most common presenting signs for MSBP are bleeding (44%), seizures (42%), central nervous system depression (19%), apnea (15%), diarrhea (11%), vomiting (10%), fever (10%), and rash (9%). The most common methods of fabricating illness are lying, poisoning, suffocating, specimen tampering, and chart falsifying.93-95
Apnea frequently has been described as a manifestation of MSBP, and conclusive proof of the parent’s role has been provided by the use of covert video surveillance (CVS).20,94 Techniques used by these mothers to asphyxiate their infants include covering the mouth or nose with one or both hands, a piece of cotton fabric, an article of the patient’s clothing, domestic plastic film, a pillow, holding the child tightly to their chest and also inserting the fingers into the back of the mouth.20,82,95 Infants struggle violently, but there often are no cutaneous markings. If present, bleeding from the nose or mouth in association with an ALTE distinguishes intentional suffocation from ALTE attributable to natural causes.20 The presence of erythema over the nose or face also should raise the possibility of intentional suffocation.
The presence of bleeding from the nose, mouth, or both and a family history of sudden death in childhood should dictate a full and forensic analysis of the family history, including information from social services and other child protection agencies, the police, EDs, and the family physician.20 There should be a low threshold for performing a skeletal survey, retinal examination, and brain imaging. Siblings also should be examined, and their records reviewed.
Deliberate human actions are likely to explain a small part of SIDS and ALTE events. The American Academy of Pediatrics estimates that fewer than 5% of apparent SIDS deaths are due to abuse.96 They occur, however, and must be considered, particularly in light of the implications for subsequent mortality and morbidity in siblings and other infants harmed by a repeat perpetrator.
Conclusion
Infants with ALTE form a heterogeneous entity, with a wide range of diagnostic possibilities. In approximately 50% of the cases, a specific medical or surgical cause can be found for the event. A systematic diagnostic evaluation of infants with an ALTE, together with a comprehensive treatment program, is necessary to increase the possibility of quality survival for these infants.
References
1. Samuels MP. Apparent life-threatening events: Pathogenesis and management. In: Loughlin GM, Carroll JL, Marcus CL (eds): Sleep and Breathing in Children. New York:Marcel Dekker, Inc.;2000:423-441.
2. Brooks JG. Apparent life-threatening events and apnea of infancy. Clini Perinatol 1992; 4:809-838.
3. National Institutes of Health Consensus Development Conference in Infantile Apnea and Home Monitoring, Sept. 29 to Oct. 1, 1986: Consensus statement. Pediatrics 1987; 79:292-299.
4. Steinschneider A, Richmond C, Ramaswamy V, et al. Clinical characteristics of an apparent life-threatening event (ALTE) and the subsequent occurrence of prolonged apnea or prolonged bradycardia. Clin Pediatr 1998; 37:223-230.
5. Oren J, Kelley D, Shannon DC. Identification of a high risk group for sudden infant death syndrome among infants who were resuscitated for sleep apnea. Pediatrics 1986; 77:495-499.
6. Steinschneider A, Santos V. Parental reports of apnea and bradycardia: temporal characteristics and accuracy. Pediatrics 1991; 88:1100-1105.
7. Stratton SJ, Taves A, Lewis RJ et al. Apparent life-threatening evens in infants: High risk in the out-of-hospital environment. Ann Emerg Med 2004; 43: 711-717.
8. Gray C, Davies F, Molyneux E. Apparent life-threatening events presenting to a pediatric emergency department. Pediatr Emerg Care 1999; 15:195-199.
9. Davies F, Gupton R. Apparent life-threatening events in infants presenting to an emergency department. Emerg Med J 2002; 19:11-16.
10. DePiero AD, Teach SJ, Chamberlain JM. ED evaluation of infants after an apparent life-threatening event. Am J Emerg Med 2004; 22:83-86.
11. Perkin RM, Swift JD, Baron H, et al. Apparent life-threatening events: Recognition, differentiation, and management. Pediatr Emerg Rep 1998;3:99-109.
12. Kahn A, Rebuffat E, Franco P, et al. Apparent life-threatening events and apnea of infancy. In: Beckerman RC, Brouillette RT, Hunt CE, eds. Respiratory control disorders in infants and children. New York:Williams and Wilkins; 1992:178-189.
13. Rahilly PM. The pneumographic and medical investigation of infants suffering apparent life-threatening episodes. J Paediatr Child Health 1991; 27: 349-353.
14. Brooks JG. Sudden infant death syndrome and apparent life-threatening events. In: Levin DL, Morris FL, eds. Essentials of Pediatric Intensive Care. New York: Churchill Livingstone;1997:111-115.
15. Ruggins NR, Milner AD. Site of upper airway obstruction in infants following an acute life-threatening event. Pediatrics 1993; 91:595-601.
16. Southall DP, Levitt GA, Richards JM, et al. Undetected episodes of prolonged apnea and bradycardia in preterm infants. Pediatrics 1983;72:541-551.
17. Muttit SC, Finer NN, Tierney AJ, et al. Neonatal apnea: diagnosis by nurse versus computer. Pediatrics 1988;82:713-720.
18. Spitzer AR, Gibson E. Home monitoring. Clin Perinatol 1992;19: 907-925.
19. Samuels MP, Poets CF, Noyes JP, et al. Diagnosis and management after life-threatening events in infants and young children who received cardiopulmonary resuscitation. Br Med J 1993;306:489-492.
20. Southall DP, Plunkett MCB, Banks MW, et al. Covert video recordings of life-threatening child abuse: Lessons for child protection. Pediatrics 1997;100: 735-760.
21. Arens R, Gozal D, Williams JC, et al. Recurrent apparent life-threatening events during infancy: A manifestation of inborn errors of metabolism. J Pediatr 1993;123:415-418.
22. American Academy of Pediatrics, Committee on Fetus and Newborn. Apnea, sudden infant death syndrome, and home monitoring. Pediatrics 2003;111: 914-917.
23. Ramanathan R, Corwin MJ, Hunt CE, et al. Cardiorespiratory events recorded on home monitors: Comparison of healthy infants with those at risk for SIDS. JAMA 2001;285:2199-2207.
24. Farrell PA, Weiner GM, Lemons JA. SIDS, ALTE, Apnea, and the use of home monitors. Pediatr Rev 2002;23:3-9.
25. Jobe AH. What do home monitors contribute to the SIDS problem? JAMA 2001;285:2244-2245.
26. Hunt CE, Corwin MJ, Lister G, et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. J Pediatr 1999;134:580-586.
27. Weese-Mayer DE, Brouillette RT, Morrow AS, et al. Assessing validity of infant monitor alarms with event recording. J Pediatr 1989;115:702-708.
28. Weese-Mayer DE, Morrow AS, Conway LP, et al. Assessing clinical significance of apnea exceeding fifteen seconds with event monitoring. J Pediatr 1990;117:568-574.
29. Silvestri JM, Weese-Mayer DE, Kenny AS, et al. Prolonged cardiorespiratory monitoring of children more than twelve months of age: characterization of events and approach to discontinuation. J Pediatr 1994;125:1-6.
30. Poets CF, Samuels MP, Noyes JP, et al. Home event recordings of oxygenation, breathing movements, and heart rate and rhythm in infants with recurrent life-threatening events. J Pediatr 1993;123:693-701.
31. Anas N, Boettrich C, Hall CB, et al. The association of apnea and respiratory syncytial virus infection in infants. J Pediatr 1982;101:65-68.
32. Panitch HB, Callahan CW, Schidlow D. Bronchiolitis in children. Clin Chest Med 1993;14:715-731.
33. Bruhn FW, Mokrohisky ST, McIntosh K. Apnea associated with respiratory syncytial virus infection in young infants. J Pediatr 1977;90:382.
34. Rayyan M, Naulaers G, Daniels H, et al. Characteristics of respiratory syncytial virus-related apnea in three infants. Acta Paediatr 2004;93:847-849.
35. Lindgren C, Jing L, Graham B, et al. Respiratory syncytial virus infection reinforces reflex apnea in young lambs. Pediatr Res 1991; 1:381-385.
36. Edwards KS, Gardner T, Altman RL, et al. Urinary tract infection presenting as an ALTE: Report of three cases. Clin Pediatr 2004;43:375-377.
37. Tirosh E, Jaffe M. Apparent life-threatening event: A neurologic perspective. J Child Neurol 1995;10:216-218.
38. Hewertson J, Poets CF, Samuels MP, et al. Epileptic seizure-induced hypoxemia in infants with apparent life-threatening events. Pediatrics 1994;94: 148-156.
39. Watanabe K, Hara K, Miyazaki S, et al. Apneic seizures in the newborn. Am J Dis Child 1982; 136:980-984.
40. Navelet Y, Wood C, Robieux I, et al. Seizures presenting as apnea. Arch Dis Child 1989; 64:357-359.
41. Kahn A, Groswasser J, Rebuffat E, et al. Sleep and cardiorespiratory characteristics of infants of sudden death: a prospective case control study. Sleep 1992; 15:287-292.
42. Kahn A, Blum D, Rebuffat E, et al. Polysomnographic studies of infants who subsequently died of sudden infant death syndrome. Pediatrics 1988; 82: 721-727.
43. Gulleminault C, Stoohs R, Skrobel A, et al. Upper airway resistance in infants at risk for sudden infant death syndrome. J Pediatr 1993; 122:881-886.
44. Kato I, Groswasser J, Franco P, et al. Developmental characteristics of apnea in infants who succumb to sudden infant death syndrome. Am J Respir Crit Care Med 2001;164:1464-1469.
45. Hoppenbrouwrs T, Hodjman JE, Cabal L. Obstructive apnea, associated patterns of movement, heart rate, and oxygenation in infants at low and increased risk for SIDS. Pediatr Pulm 1993;15:1-12.
46. Marcus CL, Loughlin GM. Obstructive sleep apnea in children. Seminars in Pediatr Neurol 1996;3:23-28.
47. Guilleminault C, Pelayo R, Leger D, et al. Apparent life-threatening events, facial dysmorphic and sleep-disordered breathing. Eur J Pediatr 2000;159: 444-449.
48. Tishler PV, Redline S, Ferrette V, et al. The association of sudden unexpected infant death with obstructive sleep apnea. Am J Resp Crit Care Med 1996; 153:1857-1863.
49. McNamara F, Sullivan CE. Obstructive sleep apnea in infants: Relation to family history of sudden infant death syndrome, apparent life-threatening events, and obstructive sleep apnea. J Pediatr 2000;136:318-325.
50. Guilleminault C, Pelayo R, Leger D, et al. Sleep-disordered breathing and upper-airway anomalies in first-degree relatives of ALTE children. Pediatr Res 2001;50:14-22.
51. Lombroso CT, Lerman P. Breath-holding spells (cyanotic and pallid infantile syncope). Pediatrics 1967;39:563-581.
52. Southall DP, Samuels MP, Poets CF. Prolonged expiratory apnea and intrapulmonary shunting. In Beckerman RC, Bronillette RT, Hunt CE,eds. Respiratory Control Disorders in Infants and Children. Baltimore:Williams & Wilkins:1992:242-250.
53. Southall DP, Samuels MP, Talbert DG. Recurrent cyanotic episodes with severe arterial hypoxemia and intrapulmonary shunting: a mechanism for sudden death. Arch Dis Child 1990; 65:953-961.
54. DiMario FJ. Breath-holding spells in childhood. Curr Probl Pediatr 1999; 29:288-299.
55. Kahn A, Rebuffat E, Sottiany M, et al. Brief airway obstructions during sleep in infants with breath-holding spells. J Pediatr 1990;117:188-193.
56. DiMario JF, Burleson JA. Autonomic nervous system function in severe breath-holding spells. Pediatr Neurol 1993;9:268-274.
57. Southall DP, Stebbens V, Abraham N, et al. Prolonged apnea with severe arterial hypoxemia resulting from complex partial seizures. Dev Med Child Neurol 1987;29:784-789.
58. Vazquez JL, Buonomo C. Feeding difficulties in the first days of life: Findings on upper gastrointestinal series and the role of the videofluoroscopic swallowing study. Pediatr Radiol 1999; 29:894-896.
59. Hillemeier AC, Lange R, et al. Delayed gastric emptying in infants with gastroesophageal reflux. J Pediatr 1981;98:190-193.
60. Glassman M, George D, Grill B. Gastroesophageal reflux in children: Clinical manifestations, diagnosis and therapy. Gastroenterol Clin North Am 1995; 24:71-98.
61. Ramenotsky ML, Leape LL. Continuous upper esophageal pH monitoring in infants and children with gastroesophageal reflux, pneumonia, and apneic spells. J Pediatr Surg 1981;16:374-378.
62. Honna T, Kamir Y, Yoshaki T. Idiopathic gastric volvulus in infancy and childhood. J Pediatr Surg 1990; 25:707-710.
63. Spizer, AR, Boyle JT, Tuchman DN, et al. Awake apnea associated with gastroesophagealrefluz: A specific clinical syndrome. J Pediatr 1984;104: 200-205.
64. See CC, Newman LJ, Berezin S, et al. Gastroesophageal reflux-induced hypoxemia in infant with apparent life-threatening events. AJDC 1989; 143:9951-954.
65. Booth IU. Silent gastroesophageal reflux: how much do we miss? Arch Dis Child 1992;67:1325-1327.
66. Halperm LM, Jolley SG, Tunnell WP, et al. The mean duration of gastroesophageal reflux during sleep as an indicator of respiratory symptoms from gastroesophageal reflux in children. J Pediatr Surg 1991; 26:686-692.
67. Putman PE, Ricker DH, Orenstein SR. Gastroesophageal reflux. In Beckerman RL, Brouillette RT, Hunt CE, eds. Respiratory Control Disorders in Infants and Children. Baltimore:Williams & Wilkins:1992:322-341.
68. Marcus CL, Hamer A. Significance of isolated bradycardia detected by home monitoring. J Pediatr 1999;135:321-326.
69. McGuirt WF. Gastroesophageal reflux and the upper airway. Pediatr Clin North Am 2003;50:487-502.
70. Arad-Cohen N, Cohen A, Tirosh E. The relationship between gastroesophageal reflux and apnea in infants. J Pediatr 2000;137:321-326.
71. Amin RS. Gastroesophageal reflux and infant apnea. J Pediatr 2000;137: 298-300.
72. Jolley SG, Herbst JJ, Johnson DG, et al. Esophageal pH monitoring during sleep identifies children with respiratory symptoms from gastroesophageal reflux. Gastro 1981;80:1501-1506.
73. Jolley SG, Halpern LM, Tunnell WP, et al. The risk of sudden infant death from gastroesophageal reflux. J Pediatr Surg 1991;26:691-696.
74. Nussbaum E, Maggi JL, Mathis R, et al. Association of lipid-laden alveolar macrophages and gastroesophageal reflux in children. J Pediatr 1987;110: 190-194.
75. Boles RG, Buck EA, Blitzer MG, et al. Retrospective biochemical screening of fatty acid oxidation disorders in postmortem livers of 418 cases of sudden death in the first year of life. J Pediatr 1998;132:924-933.
76. Berger D. Child abuse simulating "near-miss" sudden infant death syndrome. J Pediatr 1979;95:554-556.
77. Altman RL, Kutschner ML, Brand DA. The shaken-baby syndrome [letter]. N Engl J Med 1998;339:1329-1330.
78. Pitetti RD, Maftei F, Chang K, et al. Prevalence of retinal hemorrhages and child abuse in children who present with an apparent life-threatening event. Pediatrics 2002;110:557-562.
79. Altman RL, Brand DA, Forman S, et al. Abusive head injury as a cause of apparent life-threatening events in infancy. Arch Pediatr Adolesc Med 2003; 157:1011-1015.
80. Hymel KP. Traumatic intracranial injuries can be clinically silent. J Pediatr 2004;144:710-172.
81. Laskey AL, Holsti M, Runyan DK, et al. Occult head trauma in young suspected victims of physical abuse. J Pediatr 2004;144:719-722.
82. Meadow R. Suffocation, recurrent apnea, and sudden infant death. J Pediatr 1990; 17:351-357.
83. Samuels MP, McClaughlin W, Jacobson RR, et al. Fourteen cases of imposed upper airway obstruction. Arch Dis Child 1992;67:162-170.
84. McClure RS, Davis PM, Meadow SR, et al. Epidemiology of Munchausen syndrome by proxy, non-accidental poisoning, and non-accidental suffocation. Arch Dis Child 1996;75:57-61.
85. Mitchell I, Brummitt J, DeForest J, et al. Apnea and factitious illness (Munchausen Syndrome by Proxy). Pediatrics 1993; 92:810-814.
86. Hettler J. Munchausen syndrome by proxy. Pediatr Emerg Care 2002;18: 371-374.
87. Sheridan MS. Munchausen syndrome by proxy and apnea. Neonatal Intensive Care 2003;16:25.
88. Schreier H. Munchausen by proxy defined. Pediatrics 2002; 110:985-988.
89. Asher R. Munchausen’s syndrome. Lancet 1951;1:339-341.
90. Meadow R. Munchausen syndrome by proxy: the hinterland of child abuse. Lancet 1977; 2:343-345.
91. Bryk M, Siegal PT. My mother caused my illness: The story of a survivor of Munchausen by proxy syndrome. Pediatrics 1997;100:1-7.
92. Rosen CL, Frost JD, Bricker T, et al. Two siblings with recurrent cardiorespiratory arrest: Munchausen syndrome by proxy or child abuse? Pediatrics 1983;71:715-720.
93. Rosenberg D. Web of deceit: A literature review of Munchausen syndrome by proxy. Child Abuse Negl 1987;11:547.
94. Schreier H. Munchausen by proxy. Curr Prob Pediatr Adolesc Health Care 2004;34:126-143.
95. Sharif I. Munchausen syndrome by proxy. Pediatr Rev 2004;25:215-216.
96. AAP Committee on Child Abuse and Neglect. Distinguishing sudden infant death syndrome form child abuse fatalities. Pediatrics 1994; 94:124-127.
Infants with an apparent life-threatening event (ALTE) present for medical attention because an acute and unexpected incident has alarmed the caregivers. These frightening episodes of apnea and color change in infants have generated considerable concern in both professional and lay groups. This article reviews the presentation, recognition, diagnosis and ED management for children who present with an ALTE.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
