Heat and Moisture Exchangers and Ventilator-Associated Pneumonia
Heat and Moisture Exchangers and Ventilator-Associated Pneumonia
Abstract & Commentary
Synopsis: This meta-analysis of published trials comparing HMEs and heated humidifiers found a significant reduction in VAP when they were used, particularly among patients who required more than 7 days in the ICU.
Source: Kola A, et al. Intensive Care Med. 2005; 31(1):5-11.
Kola and associates in Hannover, Germany, carried out a systematic review of randomized controlled trials examining the effects on the incidence of ventilator associated pneumonia (VAP) by using a heat and moisture exchanger (HME) rather than a heated humidifier to condition inspired gases during mechanical ventilation. They sought reports of clinical trials published between 1990 and 2003 using Medline and the bibliographies of individual reports. They also searched the Cochrane Central Register of Controlled Trials. Published trials were included if they used HMEs in the treatment group and heated water humidifiers in the control group, provided specific definitions for pneumonia, and reported its incidence. Meta-analysis was then performed on the results of the identified trials.
Of 197 articles initially identified, Kola et al found 8 that met all their a priori inclusion criteria. These 8 trials included a total of 1368 patients, 693 managed with HMEs and 675 who had heated humidifiers. Only one individual trial reported a significant difference in VAP rate when comparing HMEs to heated humidification.1 However, pooling the results from all the studies revealed a reduction in the overall risk ratio of VAP in the HME group (0.69; 95% CI, 0.51-0.94). A test for homogeneity showed no evidence of significant heterogeneity in the pooled results, so that considering all 8 studies as one large cohort for the purposes of meta-analysis appeared justified.
Kola et al performed 2 subanalyses of their data, assessing the effect of duration of mechanical ventilation and of how pneumonia was defined. Only in studies with a duration of mechanical ventilation of 7 days or longer was there a significant difference in VAP incidence with the 2 humidification systems: for ventilation > 7 days the risk ratio of VAP was 0.57 (95% CI, 0.38-0.83), as compared to 0.99 (0.59-1.62) for ventilation < 7 days. The significant overall reduction in VAP incidence with HME use was found in the 4 studies using a clinical definition for VAP (RR, 0.64; 95% CI, 0.44-0.92), but not in the 4 studies using a microbiologically confirmed diagnosis (RR, 0.83; 95% CI, 0.49-1.42).
Comment by David J. Pierson MD
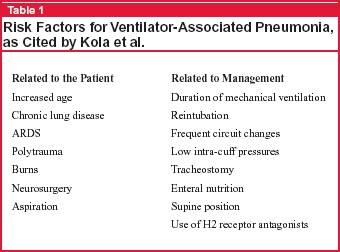
Next to urinary tract infection, VAP is the second most common ICU-acquired infection. It accounts for increased morbidity and mortality as well as enormous increases in the costs of ICU management. Risk factors for acquiring VAP, as listed by the authors of this paper, relate both to the patient and to management in the ICU, as shown in Table 1, below. Because of the magnitude of the problem of VAP, anything clinicians can do to decrease its incidence would seem worthwhile, especially if the intervention is easy to implement and inexpensive in comparison with alternatives.
For several reasons, HMEs are attractive substitutes for the traditional heated humidifiers used to warm and moisten inspired gases during mechanical ventilation. HMEs are passive and incorporated into the ventilator circuit, and do not require frequent circuit interruption (which increases the risk for VAP). HMEs reduce costs, most prominently because they require less attention from respiratory therapists and other personnel. However, HMEs cannot be used in all patients. Kola et al list 4 contraindications, as shown in Table 2, below.

Because they are essentially filters through which the patient’s inhaled and exhaled breath must pass, circumstances must be avoided in which they may become clogged. Thus, the presence of copious, tenacious, or grossly bloody secretions is a contraindication to their use. Even in the absence of obstruction from secretions, HMEs add resistance to the circuit. As a result, they could interfere with spontaneous breathing during partial ventilatory support, and might add unacceptable work of breathing in patients with severe underlying lung disease. Patients with obstructive lung disease were excluded from most of the studies included in the meta-analysis by Kola et al. HMEs could also impair the efficiency of the ventilator at high minute ventilations, and they are less effective under these circumstances. For this reason, at the institution at which I practice, HMEs are not used in patients with minute ventilations exceeding 15 L/min.
The possible effect of HMEs on the incidence of VAP has been examined previously with meta-analysis. Cook and colleagues looked at several aspects of airway management, including HMEs, in an article published in 1998.2 They concluded that, although the study of Kirton et al1 was the only randomized trial published to date to have a statistically positive overall result, the use of HMEs probably was associated with lower rates of VAP. The present meta-analysis included 4 additional trials not examined in the earlier review, and its findings make the case for a favorable effect of HMEs on VAP more strongly.
The fact that HMEs reduced the incidence of VAP only in studies using a clinical definition of pneumonia, and not when microbiological confirmation was used, is troubling. How best to diagnose VAP remains a hotly debated issue, but most authorities are not only leaning toward microbiological confirmation but also increasingly relying on quantitative cultures from bronchoalveolar lavage (BAL) or mini-BAL catheter.3 More studies, using precise and standardized definitions for VAP, will be required before this issue can be settled more objectively.
The advantages of HMEs, at least with respect to the incidence of VAP, apply mainly to patients requiring prolonged mechanical ventilation, and they cannot be used in some patient groups. However, it seems justified to use HMEs instead of heated humidifiers whenever feasible.
References
1. Kirton OC, et al. Chest. 1997;112:1055-1059.
2. Cook D, et al. JAMA. 1998;279:781-787.
3. Chastre J, et al. Am J Respir Crit Care Med. 2002;165(7):867-903.
David J. Pierson, MD, Pulmonary and Critical Care Medicine Harborview Medical Center University of Washington, is Editor for Critical Care Alert.
This meta-analysis of published trials comparing HMEs and heated humidifiers found a significant reduction in VAP when they were used, particularly among patients who required more than 7 days in the ICU.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
