How to Read an Abdominal Computed Tomography Scan
How to Read an Abdominal Computed Tomography Scan
Authors: J. Christian Fox, MD, RDMS, Associate Professor of Emergency Medicine, University of California–Irvine School of Medicine, Irvine, CA; and Jeffrey J. Westin, MD candidate 2008, University of California–Irvine School of Medicine, Irvine, CA.
Peer Reviewers: Samuel Madoff, MD, Department of Imaging Sciences, University of Rochester Medical Center, Rochester, NY; and Chris Moore, MD, RDMS, FACEP, Assistant Professor, Department of Surgery, Section of Emergency Medicine, Yale University School of Medicine, New Haven, CT.
During my residency, our hospital received its first CT scan: a machine manufactured by EMI Ltd., formerly known before 1971 as Electric and Musical Industries, Ltd. The device was named an "EMI-Scanner," and we thought it was wonderful, not in small part due to our knowledge that EMI was the company that recorded and distributed the Beatles and other bands.
Looking back, the limitations of that machine seem mind boggling: it could only scan the brain, each slice took 2 minutes to acquire and process, and for a typical 15-slice head CT, the patient spent 30 minutes on the scan table! To hold the head still during the 2 minutes of scanning, a water filled donut fixed to the scanner enclosed the patient's head. This presented some unusual circumstances. I still have a copy of the radiologist's dictation that says "Cranial Computer Tomography: This examination was attempted on 8/3/76 without success because the patient's head size was too large to fit into the EMI brain scanner waterbag."
How much as changed. How many of your patients have a CT scan during their ED evaluation? Many hospitals report rates of 20% or more. A significant number of these scans are of the abdomen and pelvis. I believe it is important for the emergency physician to have the knowledge to view and interpret these images. We are often able to look at the scan and determine the abnormality before the radiologist has time. We sometimes identify abnormalities due to our clinical knowledge of the patient as opposed to the radiologist who may not have the same information. And viewing the images provides us a better understanding of the patient's condition.
Please enjoy this issue of Emergency Medicine Reports as the authors teach us how to read an abdominal CT scan.
J. Stephan Stapczynski, MD, Editor
Introduction
From its origins in the 1970s to today, computed tomography (CT) has evolved into one of the primary diagnostic tools of the abdomen, and it continues this ongoing technological progression every day. When first unveiled by Sir Godfrey Hounsfield in 1972, computed tomography was a revolution in radiological imaging. This revolution continued in the 1990s with the innovation of spiral (or helical) CT, which allowed for more continuous scanning and improved z-axis resolution.1,2 Spiral CT allowed for better three-dimensional imaging and diminished the disadvantages of previous two-dimensional, uniplanar scans by allowing for increased table speed and more distinct longitudinal resolution. This increase in table speed is valuable in a trauma setting, and also significantly reduces the amount of radiation exposure to the patient for any continuous area. The radiation exposure is further decreased as the pitch (pitch [p] = d/s, where d is table feed per rotation and s is slice thickness) is increased.2
Spiral CT works by obtaining a volumetric block of raw data in a spiral rather than a planar manner, with the patient moving in a continuous z-axis direction while the tube and detector array rotate around the patient. This continuous motion allows for a much larger volume to be covered, with less radiation exposure per volume covered when compared with sequential CT. The volumetric data images captured by the CT are described as voxels (the three-dimensional equivalent of 2D pixels, or picture elements). An additional method of increasing the speed of image acquisition is increasing the number of rows of detectors. With more parallel rows of detectors, the number of simultaneous readings can be increased without increasing the heat load of the tube, a limitation of early CT scanners.2 The fundamental principle in improving resolution and image quality is to utilize the smallest collimation and highest pitch possible.3 This goal is easier to achieve with multidetector CT arrays than with the standard spiral CT array. Accordingly, the advent of multidetector computed tomography (MDCT) has vastly improved imaging technology, especially as it relates to the representation of vascular anatomy.
Hounsfield Units
In order to differentiate between different types of fluid and tissue in the abdomen, it is important to understand the concept of Hounsfield units (HU) and how they are derived. Historically, the recreated images from CT scanners had a wide range of values of 12-bit digital data, measured in standard transform (ST). The inventor of the original CT, Sir Godfrey Hounsfield, developed a method to standardize the density measurements between different machines. Hounsfield's absolute density scale defined air as the minimum density, with a value of –1000 HU, and placed water as the benchmark of 0. The most dense material in the human body, bone, has an upper limit of +1000 HU. The raw data could be translated to HU by the equation HU = ST scale + offset (e.g. scale = 1.0, offset = -1000).2 This scale can be applied directly to the analysis of free fluid in the abdomen, especially as it relates to identifying blood. The appearance of blood in intra-abdominal hemorrhage can vary depending on the recency of the bleed, and this can help to determine if there is active bleeding or if clotting has occurred. Clotted blood has a heterogeneous appearance and is generally between 45 and 70 HU. Clotted blood tends to congregate close to the original hemorrhage site, producing the so-called "sentinel clot." Freely flowing blood, however, will have a less dense appearance, and typically ranges from 20 to 45 HU.4 Blood can also be identified by extravasation of contrast material, which can accumulate in the abdominal cavity or demonstrate sites of vascular disruption.2
Contrast Use
Abdominal CT scans can be done without the use of radiopaque contrast agents, termed a non-contrast enhanced CT or NECT. The advantage of NECT is that the intense radiodensity of these contrast agents can obscure areas of abnormality, like small renal or ureteral stones. Conversely, the use of a contrast enhanced CT, or CECT, can provide better distinction between tissues and various structures. Most abdominal CT scans are done with intravenous radiocontrast as this approach helps in the identification of inflammatory and neoplastic processes. Intravenous contrast agents use iodine as the radiopaque agent bound to either an organic (non-ionic) or ionic compound.
Barium sulfate, an insoluble powder suspended in water is a common radiocontrast used to fill the lumen of gastrointestinal structures during radiography. Many hospitals are using a water soluble iodine product (Diatrizoate Meglumine and Diatrizoate Sodium Solution) before CT scanning when visualization of the gastrointestinal lumen is desired. Both agents can be administered by mouth, nasogastic tube, or rectal enema, depending on the structures to be visualized.
 |
 |
 |
The routine use of both intravenous and oral contrast for abdominal CT scanning has been greatly debated. Because CT technology has dramatically improved, it is important to consider the costs versus the benefits of oral contrast using current machines. ED time studies typically find that the use of oral contrast adds 90 to 180 minutes of extra time until the CT scan is completed. Usually it is the clinicians wanting the study done quickly who order the CT scan without oral contrast. Conversely, radiology-based studies on CT helical scan accuracy find that oral contrast improves sensitivity and specificity for a variety of conditions, such as appendicitis, by 2-3%.5 It is often the radiologist who wants oral contrast to improve his or her interpretive accuracy.
It is beyond the scope of this article to discuss the appropriate use of intravenous and/or oral contrast. There are a variety of protocols that balance the clinician's need for speed and the radiologist's need for completeness and accuracy. Each institution is encouraged to develop an approach for the use of intravenous and/or gastrointestinal contrast according to the patient's suspected clinical condition for which CT scanning is being done.
Radiation Exposure Concerns
An important consideration in the use of CT is the radiation exposure to the patient, especially in pregnant patients and children. Radiation risks are difficult to quantify and predict, and most estimates are based on radiation exposures in the aftermath of the atomic fallout from Hiroshima, Nagasaki, and Chernobyl. In the studies associated with these events, the minimum absorbed dose associated with statistically significant cancer in children is somewhere between 100 and 200 mGy (1 rad = 10 mGy).6 By comparison, the approximate mean dose of a pediatric abdominal CT scan is 25 mSv (for X-rays, electrons and gamma rays, the absorbed dose [mGy or rad] is equivalent to the equivalent dose [mSv]).7,8
The advent of multislice CT has led to a significant improvement in the quality of pediatric CT scans, due to the increase in table speed, which leads to a decrease in artifact, refinement of contrast enhancement, and less need for sedation. However, this improvement in image quality comes at the cost of increased radiation doses delivered to the pediatric patients in question, due to less focused collimation, and it is suggested that tube currents and kilovolts be adjusted to reduce the dose of radiation delivered.9 In adults, the lowest dose significantly associated with radiogenic risk is higherapproximately 200 mSvand it cannot be demonstrated that there is any increased risk below 100 mSv.6,10 However, the fetus exposed in utero is considered to be more sensitive to radiation effects, and diagnostic studies in pregnant women can sometimes be delayed or avoided due to concerns about teratogenic and radiogenic effects. It should be noted, however, that the American College of Obstetricians and Gynecologists has declared, "Women should be counseled that x-ray exposure from a single diagnostic procedure does not result in harmful fetal effects. Specifically, exposure to less than 5 rad has not been associated with an increase in fetal anomalies or pregnancy loss."11 It is also notable that no single diagnostic procedure results in a dose greater than this limit, even direct CT scans of the abdomen and pelvis, and that scans of more distant body parts result in levels substantially below 5 rad. A single CT of the abdomen (10 slices) results in a mean estimated fetal dose of 2.6 rad (although some studies suggest an even lower mean estimated fetal dose of 8 mGy, or 0.8 rad), while a pelvic CT can produce a mean estimated dose of 2.5 rad.11,12 For scans of the chest and head, fetal doses are even lower, less than 0.100 rad and less than 0.050 rad, respectively. With such low fetal doses, even repeat scans of the head in trauma or stroke, or chest CT to rule out or follow pulmonary embolism in a pregnant patient pose relatively little risk to the fetus. At these doses, more than 100 head CT or greater than 50 chest CT could be performed before arriving at the 5 rad limit.11 Although concerns have been raised about the use of CT for the diagnosis of pulmonary embolism in pregnant patients, studies have demonstrated that helical chest CT results in less fetal radiation exposure than ventilation-perfusion (V/Q) scintigraphy.13 This is important to note, because a survey in the United Kingdom showed that more than 40% of physicians across all specialties were unaware of the differences in fetal dose exposure between V/Q scans and CT.14
Systematic Approach
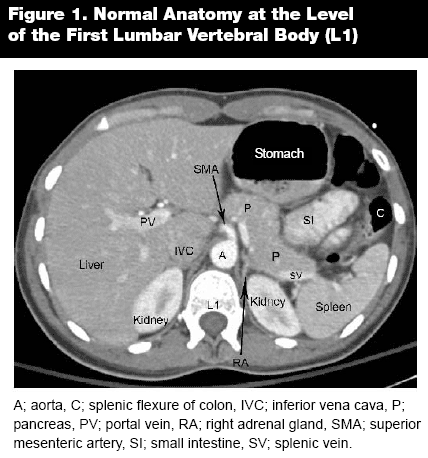
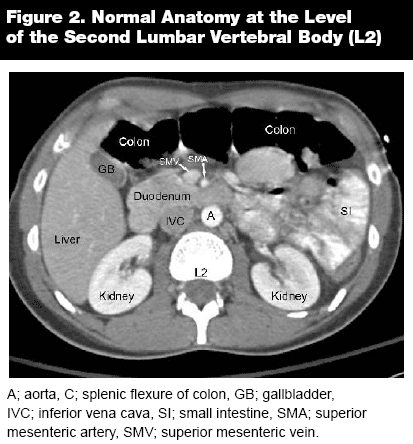
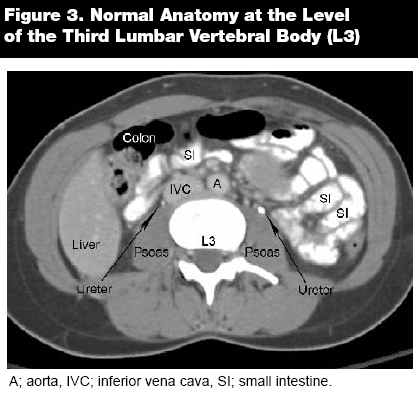
As in any other imaging analysis, it is important to take a systematic approach when reading abdominal computed tomography (CT). Always begin cranial and gradually move caudally. Likewise, assess structures from superficial to deep, first analyzing the tissues or abdominal wall and then progressing to the internal structures. For physicians with limited experience reading CT scans, it is best to begin by following one organ and tracking it through the entire sequence. With experience, the next step is to follow organs that lie in the same transverse plane, such as the liver and spleen, pancreas and adrenals, and the kidneys. As the CT tracks caudally, identify the appropriate anatomical landmarks, such as the celiac trunk, the superior mesenteric artery, the renal arteries, and the aortic bifurcation. Follow the major vessels to assure that the IVC and the aorta are intact and without major pathology.
Abdominal Wall and Intraabdominal Fluid
Beginning superficially, check for hernias and other defects in the integrity of the abdominal walls. Assure that the entire abdominal wall is smooth and regular, and identify any irregularities. Also note any thickening of the peritoneum, and whether the thickening is smooth or nodular. Look for fluid collections within the abdominal cavity, and estimate the location and amount of fluid as closely as possible. The amount of fluid can be measured most simply by multiplying the volume of a single voxel (Xmm x Ymm x Zmm) by the number of voxels contained in the space occupied by the fluid. A finer approximation of the volume can be made with smaller voxel units, as there will be less rounding. However, in a scanner with less resolution or with insufficient slice thickness, approximating the shape of the structure may offer a more exact estimation of the volume than the discrete calculation.2 If there is any fluid collecting in the vicinity of the diaphragm, it is important to ascertain whether the fluid is intrathoracic or intra-abdominal by looking at slices above and below the diaphragm. Generally, pleural fluid is located outside the perimeter of the diaphragm, has a hazy border with peripheral structures, and appears to come in contact with the abdominal wall. Intra-abdominal fluid, on the other hand, is contained by the diaphragm, creates a sharp, distinct border with the diaphragm and surrounding organs, and is located medially. Intra-abdominal fluid accumulation can indicate ascites, hemorrhage, or intraperitoneal abscess, and is of clinical importance regardless of amount.4
It is vital to inspect the greater omentum for irregularities, as it often serves as a critical clue in the analysis of abdominal pathology. Nodularity or irregular densities in the greater omentum can indicate metastatic disease, and the position of the greater omentum can often point to a site of trauma or infection by adhering to injured or inflamed organs. Since it is in the transverse plane, it is often difficult to assess the integrity of the diaphragm on CT, but a scan in the correct plane can detect hiatal hernias or traumatic rupture of the diaphragm. Throughout the inspection, be alert for enlarged or abnormal lymph nodes, and note their location and size.
Liver and Biliary Tract
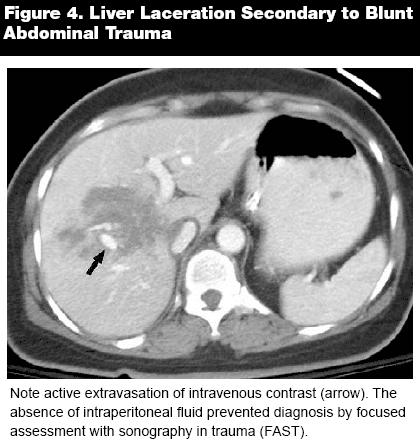
Moving caudally from the diaphragm, next survey the liver and spleen. When using intravenous contrast imaging, there are several important phases in assessing liver function and pathology. The early arterial phase occurs ~18-25 seconds after the injection of a bolus of contrast, and true to its name, is optimal for observing hepatic arterial angiography. Approximately 35-40 seconds post-contrast infusion, the late arterial (sometimes called portal venous inflow) phase begins, and this window is ideal for identifying and categorizing hypervascular tumors. Next, the portal venous, or parenchymal predominant phase begins at ~70 seconds. This is the standard contrast view, which will be used in the vast majority of abdominal CT scans. As indicated by the name, this phase maximally enhances the hepatic parenchyma. The parenchyma should appear homogenous and without notable lacerations or other lesions. Lacerations can be identified by active extravasation of intravenous contrast. (See Figure 4.) Sometimes an equilibrium (delayed) phase can be added to more precisely represent a suspicious hepatic mass. When assessing the liver, it is important to note the overall density, which is normally several HU greater than spleen or muscle. Hypodense areas generally indicate fatty infiltration, but may also occur in other settings, such as infection, malignancy, or hepatotoxicity secondary to toxic ingestion.2 In addition, evaluate whether the surface of the liver is well-defined, and identify any regions of nodularity or blurred borders.
 |
After scrutinizing the liver, the next contiguous structure is the gallbladder. The gallbladder is normally situated in the gallbladder fossa, a depression on the inferior surface of the liver. It is important to identify certain landmarks and specific anatomy of the gallbladder. The fundus is most distal to the common bile duct, and generally protrudes just below the inferior border of the liver. The gallbladder body is routinely in close proximity to the duodenum and hepatic flexure of the colon. The cystic duct is joined to the body of the gallbladder by the gallbladder neck, which is generally located near to the right hepatic portal vein. After leaving the gallbladder, the cystic duct, which is generally only 2-4 cm in length, merges with the common hepatic duct, forming the common bile duct. Completing the biliary tree, the common bile duct joins with the pancreatic duct and enters the duodenum through the ampulla of Vater, regulated by the sphincter of Oddi. Begin by examining the borders and wall of the gallbladder. The borders should appear sharp and distinct, and the wall should be thin and smooth. If the gallbladder wall is thickened, (see Figure 5) it can indicate a variety of pathologies, the most common of which are cholecystitis, hepatitis, or major organ failure. Similarly, nodularity or disruptions in the gallbladder wall can signify malignancy, perforation, or abscess. After scanning the walls, inspect the interior of the gallbladder for any abnormalities in lucency, which can indicate fluid collections or stones. Gallstone density can run the spectrum from hypodense to hyperdense, depending on the composition of the stone. Although CT is not as sensitive as ultrasound in revealing gallstones, calculi can be detected with relative accuracy.4
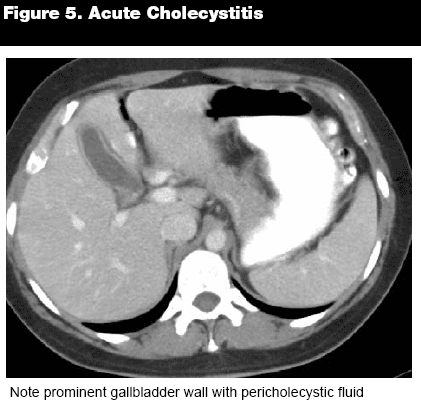 |
Spleen
The spleen generally appears in the same plane as the liver, and can be evaluated simultaneously. Evaluation of the spleen can appear daunting, but it can be broken down into several key areas. Splenic volume can be computed quite accurately on CT, but this is often of limited clinical value, due to the fact that splenic size can be quite variable. Although the mean size is 150 cm3 in adults, there is a wide range, both within the population and even in a single patient, depending on age, nutritional and fluid status, and body habitus. Instead of using size and volume to assess splenomegaly, the contours of the spleen can often offer an alternative means of diagnosis. While a normal spleen generally has a concave visceral surface, in splenomegaly the surface often inverts and takes on a convex appearance. Furthermore, the surface of the spleen is often irregular and frequently has depressions and grooves on its surface that can be mistaken for lacerations. The principal method of differentiating between normal indentations and pathologic lesions is the presence or absence of perisplenic hemorrhage. In the case of splenic infarction, clearly defined, wedge-shaped, hypodense lesions will appear adjacent to the splenic capsule. (See Figure 6.) Of interest is the relative mobility of the spleen. Although the spleen is generally located in the left upper quadrant, a spleen located on the long mesentery may drift from its usual location and can be found in virtually any intraperitoneal position within the abdomen or pelvis. As a result, an ectopic spleen can be mistaken for a tumor or other mass if the reader does not recognize and account for the absence of the organ in its usual site.4
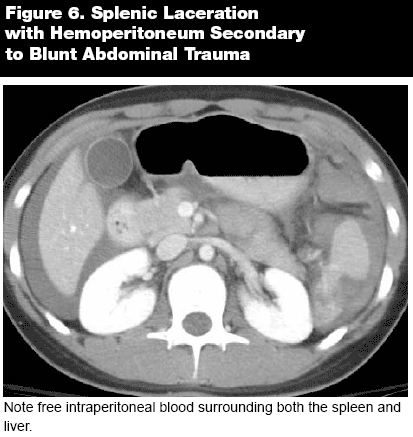 |
Pancreas and Adrenals
Proceeding caudally from the gallbladder, the pancreas and adrenals lie in approximately the same plane, and therefore can be analyzed simultaneously. To assess the pancreas, first determine if the borders are well-defined and the size appears to be proportionate to other abdominal organs. A lumpy or pitted border or a mass that appears ragged, uneven, heterogeneous, and/or hypoenhancing and/or obstructs the biliary tract is highly suspicious for malignancy. Pancreatic size is variable and is therefore difficult to assess independently, but in general an anteroposterior measurement > 3.4 cm or a craniocaudad measurement > 4.6 cm of the head of the pancreas (the most common site of pancreatic adenocarcinoma) is concerning.2,4
The most important indications for imaging of the pancreas are generally suspicion of tumor or acute pancreatitis. Although the same protocols employed to identify malignancy can be used to evaluate for acute pancreatitis, an acute abdomen protocol can also be utilized, and is generally adequate. If there is ductal involvement in pancreatic neoplasm, it can mimic acute or chronic pancreatitis in both its clinical and radiologic presentation (dilation of the pancreatic duct, calcification of pancreatic tissue), so it is important to differentiate between the two disease processes. Generally, pancreatitis will not demonstrate the peripancreatic inflammatory changes of the mesenteric fat, fluid collection around the pancreas, or lymphadenopathy that can be associated with pancreatic neoplasms. Destruction of retropancreatic fat may be seen in both carcinoma and chronic pancreatitis, but is generally not present in acute pancreatitis.4
Kidneys
Proceeding caudally from the pancreatic plane, the profile of the kidneys should appear smooth and elliptical, with an anteromedial concavity produced by the vascular pedicle. The normal renal parenchyma varies in attenuation according to the patient's level of hydration, generally ranging between 30 and 50 HU on NECT. Surrounded by the parenchyma is the renal sinus, which holds the renal vasculature, lymphatic ducts, pelvis, and calyces, and is filled by fatty tissue. Although the cortex and medulla are virtually indistinguishable on NECT, there is visible delineation of the cortex and medulla during the nephrogenic phase of renal contrast. The left renal vein passes anterior to the aorta and posterior to the superior mesenteric artery and vein on its path to the inferior vena cava, and it is discernibly longer than its counterpart. The right renal vein is shorter and follows a more direct course straight to the kidney.15 Once structural abnormalities have been assessed on NECT, contrast is often required for certain studies.
After the initial NECT images are acquired, there are several stages of renal contrast, beginning with the arterial or cortical phase (CP), which occurs between 25 and 80 seconds after contrast injection. This phase is useful for analyzing the renal vasculature and identifying obstruction or stenosis in the renal arteries and veins. The CP is especially useful for CT angiography (CTA) and the generation of three-dimensional (3D) images of the vasculature.2
The next stage, the nephrogenic phase (NP), takes place between 80 and 120 seconds following contrast administration. This phase generally is considered to be the best stage for identifying small renal masses. This is important, since the number of renal malignancies identified as incidental findings has increased dramatically in recent years. Some studies indicate that the use of CT and ultrasonography (US) has identified five times as many small cancers (< 3 cm) as before.2 The nephrogenic phase also demonstrates well renal injuries, such a lacerations and perinephric hemorrhage. (See Figure 7.)
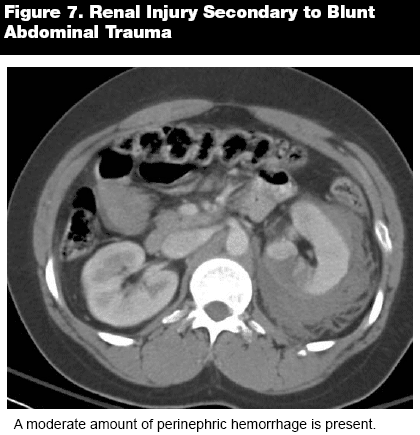 |
The final stage of contrast evaluation begins approximately 2-5 minutes after injection, and is known as the excretory phase (EP). During this phase, the calyces, infundibula, and renal pelvis can be assessed, and it is during this phase that calculi and tumors within the collecting system can be optimally identified.2 It is important to note if there is symmetric excretion of contrast or if there are filling defects causing asymmetry.
Standard abdominal CECT are normally acquired during the late CP or early NP, which can make certain defects difficult to analyze. As a result, incidental findings or an unclear diagnosis on NECT or standard abdominal CECT may necessitate a dedicated renal CT at a later date.2
Although intravenous urography (IVU) and retrograde pyelography are commonly used for the evaluation of hematuria and flank pain, CT remains an important and often superior imaging modality in the kidneys, ureters, and bladder. Indeed, NECT or CT urography with intravenous contrast can arguably replace IVU for nearly all indications of renal, ureteral, and bladder imaging. One of the most common ureteral issues addressed by CT is urolithiasis, for which NECT is still the preferred imaging technique.4 In the past, IVU has been used quite frequently for diagnosis of urolithiasis, but in the last few years, the evidence has been mounting in support of NECT replacing IVU as the preferred definitive study. Studies have shown CT to have superior accuracy compared to IVU in diagnosing ureteral stones by both direct signs (98.8% vs. 79.3%, respectively) and the combination of direct and indirect signs (100% vs. 90.3%, respectively).16 In addition, patient management issues such as time management, use of contrast, and cost lean in the direction of CT over IVU. Although there is often little difference in the direct costs of the two studies, indirect costs are often lower with CT, due to decreased examination time (average in-room time of 23 minutes with CT, compared to 1 hour and 21 minutes with IVU), lack of contrast delivery, and the not inconsequential risk of adverse reactions (mild to moderate reactions seen in 5% of patients, often necessitating additional treatment and/or time in the emergency department), and the ability to forego preliminary KUB and US studies if necessary or desired. As a result, NECT is now preferred over IVU by many authors due to a higher diagnostic accuracy and an advantageous cost profile resulting from a more effective, quicker, less expensive, and less risky study.17 Of course, KUB and US are usually the least expensive and easily available studies, but CT can often provide the best definitive study in the case of equivocal radiographic or sonographic signs, or a negative US or KUB with strong clinical suspicion. Indeed, although ultrasonography can approach 96% sensitivity in ideal circumstances, it can be extremely operator dependent, and direct visualization is not always possible. In comparison, NECT can identify stones with a sensitivity and specificity of 98% to 100%, irrespective of size, location, and type of stone.18
Nearly all renal calculi are hyperdense on CT, and one of the best initial indications that a hyperdense focus is a stone that has passed into the ureter is the "rim sign." The rim sign is a ring of soft tissue enveloping a calcification within the ureter, signifying inflammation within the ureteral wall. (See Figure 8.) This sign can distinguish between a ureteral stone and a phlebolith, which can be mistaken for each other. Phleboliths lack the rim sign and often have a lucent center and a "comet tail."19 Although the rim sign is seen in more than 90% of small renal stones (< 4 mm diameter), other signs are tremendously important to note when assessing ureterolithiasis, especially signs of obstruction. In fact, when hydroureter is noted on CT and combined with either ipsilateral perinephric or periureteric stranding, the positive predictive value is 98% in detecting a stone. When neither of these signs are present, ureterolithiasis can be excluded with a negative predictive value of 93%. With those highly significant numbers, CT accuracy is considered to be vastly superior to the results seen with IVP or ultrasound.20
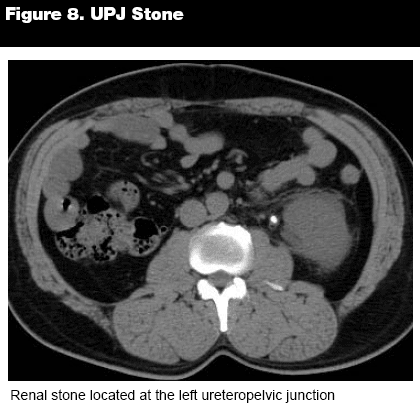 |
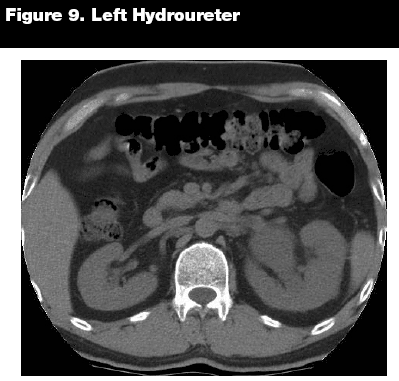
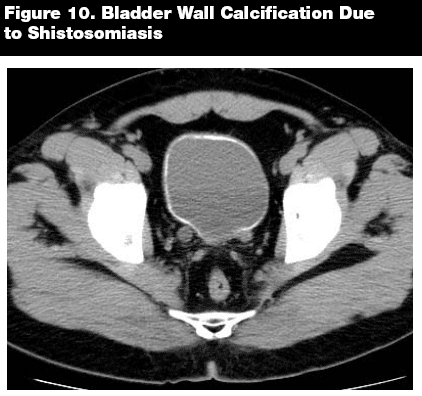
While assessing the ureters and bladder, it is important to look for not only obstruction, as with stones, but to evaluate the integrity of the ureteral and bladder walls. Ureteral dilations may be subtle, or they may be readily apparent. (See Figure 9.) The bladder wall should appear smooth and thin without dilations or calcifications. Bladder wall calcifications can be due to shistosomiasis, primary carcinoma of the bladder, alkaline encrustation cystitis, tuberculosis, cyclophosphamide-induced cystitis, and amyloidosis. (See Figure 10.)
 |
 |
Stomach and Intestines
In imaging of the gastrointestinal tract, CT has taken a backseat for many years to endoscopy and barium studies. However, with the advent of spiral CT, it is emerging as a valuable addition to the clinician's diagnostic arsenal. Although endoscopy continues to be the most accurate method of diagnosing gastric carcinoma and mucosal diseases, spiral CT is superior in the assessment of intramural disease processes such as submucosal gastric masses, which may show minimal mucosal involvement.2,4 As such, in the routine evaluation of an abdominal CT, it is important to pay attention to the definition and thickness of the gastric walls. Abnormal thickening of the gastric wall or folds may signify underlying pathology, especially if the thickening is asymmetric or in the general proximity of an unidentified mass or abnormality. As in the analysis of other organs and tissues, it is also important to assess the definition and regularity of the wall contours and account for any aberrations. It is possible to diagnose gastritis or gastric ulcers in this manner with CECT, generally using water contrast. The attenuation of the submucosa of the gastric walls often will be reduced in gastritis, due to edema or inflammation, while the mucosa may be enhanced, causing a "target" or "halo" sign. In addition, there may be wall thickening and hypertrophy of the gastric folds. Wall thickening also may be seen in gastric ulcers, perhaps leading to a reduction in the intraluminal space. Ulcers may also penetrate nearby organs or lipid deposits, and free air may be present secondary to perforation.4
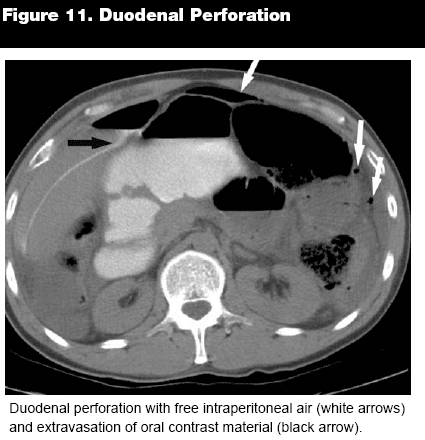
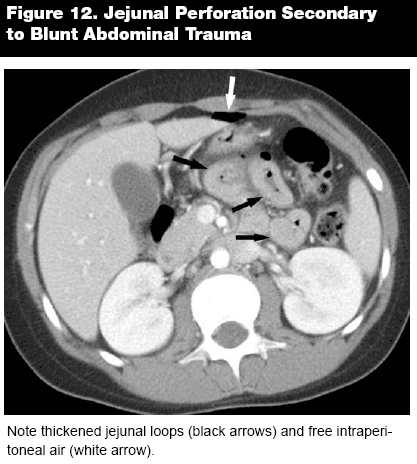
While CT is being used more often as a primary imaging technique for gastric pathologies, it is in the small bowel and proximal colon where spherical CT truly comes into its own. The area from the duodenum to the transverse colon is particularly difficult to image endoscopically, and recent improvements in contrast techniques have vastly improved CT visualization of the small intestine.2 Although small bowel follow through (SBFT) and barium studies still dominate in certain areas of diagnosis, CT is more suitable for identifying extraluminal disease and in the patient with a surgical abdomen.4 Even in areas where barium studies have certain advantages, such as the identification of intestinal ulcers and polyps, the improvements in spherical CT resolution have made it an increasingly useful tool. Ulcers and polyps can be identified with remarkable accuracy by pinpointing areas of intestinal wall enhancement on CECT. Enhancement of the section surrounding a lesion indicates an underlying pathology, whether it is inflammatory or neoplastic, and as an incidental finding can indicate the need for further study.2 Intestinal wall thickening or luminal constriction can be additional clues to duodenal ulceration and, as in the stomach, infiltration into surrounding fat and organs or free air can signal perforation.4 (See Figures 11 and 12.) Similar to analyzing other vessels and luminal organs, distention or stenosis usually signifies an underlying pathology, and the length of the intestine should be evaluated for continuity and consistent intraluminal area.
 |
 |
As with the rest of the gastrointestinal tract, inspection of the lumen and walls for distortion, distention, or changes in attenuation is important. Alterations in the regular pattern of the haustra or changes in the contour of the colonic wall, as well as distention of the colon with air or fluid should all be noted. Occasionally, air or blood can be visualized within the colonic wall, indicating ischemic bowel wall. Diverticula can be observed as outpouchings of the colonic wall containing air, contrast, or stool. On CECT, the previously mentioned halo or target sign can be observed when the inner and outer rings of the bowel wall are enhanced, while the middle ring shows minimal or no enhancement due to submucosal edema. Tapering or stenosis may also be apparent, as well as twisting of the mesentery.4
Diverticula are a fairly common finding in the elderly population, occurring in up to 80% of people by age 85. Diverticulosis, while not concerning in itself, can develop into diverticulitis in 10-35% of patients with documented diverticula, and up to 25% of these patients may need surgery.21 Diverticulitis can have classic clinical symptoms, including left lower quadrant pain and tenderness, fever, and leukocytosis, but it can also present with nonspecific symptoms, which may make imaging necessary.
Although barium enema was previously thought to be a reasonable alternative to CT, it is now generally accepted that this exam has key deficiencies in comparison to CT. In analyzing an abdominal CT for diverticulitis, several important signs have been identified, with varying degrees of sensitivity and specificity. In one study, 96% of patients showed bowel wall thickening (sensitivity 96%, specificity 91%) and 95% demonstrated fat stranding (sensitivity 96%, specificity 90%), and of course most (91%) patients revealed diverticula. These three signs are regarded by many authors to be the most common and reliable indicators in diagnosing diverticulitis, and generally occur between 70% and 100% of the time.
Other specific but less sensitive signs included fascial thickening (sensitivity 50%, specificity 100%), free fluid (sensitivity 45%, specificity 97%), inflamed diverticula (sensitivity 43%, specificity 100%), free air (sensitivity 30%, specificity 100%), and the "arrowhead" sign (sensitivity 16%, specificity 99%). Overall, thin-slice helical CT with colonic contrast has both a 99% sensitivity and specificity for diverticulitis.21
Appendix
During examination of the small and large bowels, the appendix should be assessed as a matter of routine in any patient with acute abdominal pain, especially with the classic symptoms of right lower quadrant pain, nausea, vomiting, and leukocytosis. In determining whether a CT scan is necessary for the diagnosis of acute appendicitis, some authors recommend using the Alvarado scores as a guideline. Patients with Alvarado scores of 3 or less can generally forgo CT scanning as the incidence of appendicitis in this population is < 5%.22 Conversely, for patients with an Alvarado score of 7 or greater have a greater than 70% incidence of acute appendicitis, so surgical consultation before CT scanning is recommended. It is with Alvarado scores of 4 to 6, where the incidence of acute appendicitis is about 30 to 40% where CT scanning is most useful.22 In patients with these equivocal scores, CT scans have a sensitivity for acute appendicitis of 90+% with a specificity of 95%.22
Overall accuracy of CT in the diagnosis of appendicitis varies, but is generally quite high. One larger review found a sensitivity of 96.5% and a specificity of 98%, with a positive predictive value of 94.5% and a negative predictive value of 98.8%, and an overall accuracy of 97.6%.23 Another study showed sensitivities of 88-100% and specificities of 91-99% for CT in detecting appendicitis.24 Studies in children have shown similar results, with sensitivities ranging from 95-100% and specificities from 93-100%, with an accuracy of between 94% and 99%. Interestingly, one of the smaller pediatric studies using CT without contrast had better sensitivity and specificity (both 100%) than the studies using IV, oral or rectal contrast, which showed sensitivities from 95-97% and specificities between 93% and 99% (with rectal contrast showing slightly better results than other contrast routes).25
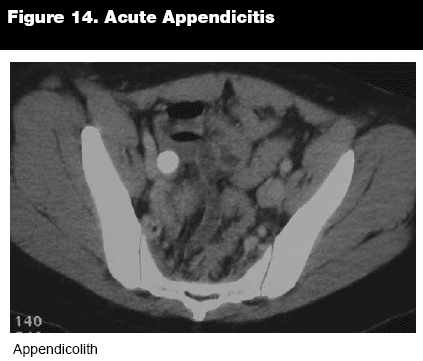
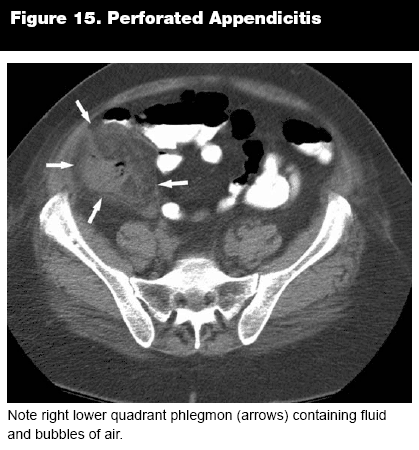
The appendix should be examined for dilation and fat stranding, (see Figure 12) and an appendicolith may be present, occasionally even as an incidental finding.4 (See Figure 14.) Once appendicitis has been diagnosed, it is often useful to determine if the appendix is perforated, to help guide therapy, including the possibility of nonsurgical treatment, and to calculate the likelihood of complications. Several signs have been identified that can indicate perforation on CT, with the most reliable predictor being extraluminal air (sensitivity 42.9%, specificity 96.9%). (See Figure 15.) Some signs, such as small-bowel dilatation (sensitivity 4.8%, specificity 95.4%) and abscess formation (sensitivity 9.5%, specificity 96.9%), are highly specific, but were observed in only a small number of patients in one study,26 although other studies have shown higher sensitivities (34%, 53%) with similar specificities (99%, 93%) for abscess and ileus.27 Other important signs include periappendiceal fluid collections (sensitivity 66.7%, specificity 73.8%), focal enhancement defects in the appendiceal wall (sensitivity 52.9%, specificity 55.4%), and either moderate or severe periappendiceal inflammatory stranding (sensitivity 76.2%, specificity 73.8%).26
 |
 |
Bowel Obstruction and Ischemia
Bowel obstruction and bowel ischemia are also issues pertinent to the emergency physician, and areas in which CT can be very helpful. In identifying bowel obstruction, CT is generally quite accurate, with sensitivities from 80-100%. Although peristalsis cannot be identified on CT, as it can on US, there are a few signs that are helpful in the recognition of obstruction, namely bowel caliber and the identification of a transitional zone. A small bowel caliber larger than 2.5 cm is deemed to be enlarged, and may indicate obstruction. (See Insert Figure 16.) In closed loop obstruction, several signs are regularly observed on CT, including C- or U-shaped loops of dilated bowel, adjacent collapsed loops, and "beak" or "whirl" signs. Adhesions, the most common cause of obstruction in the small bowel, can be identified as an enhancing band near the point of transition.28
Ischemia can be a result of obstruction, but it may also cause the obstruction in the first place. After ischemic injury, the bowel wall may appear hyper- or hypoattenuated on CT, and the degree of attenuation is diagnostically important. Other indirect signs, such as bowel wall thickening, localized fluid or hemorrhage, pneumatosis, and obscuration of the mesenteric vasculature, can also indicate ischemia.28 CT detection of bowel ischemia has sensitivities and specificities of 79.2% and 98.5%, respectively, with positive predictive value (PPV) of 90.5% and negative predictive value (NPV) of 98.2%.29
Aorta
Of course, one of the first uses of CT angiography (CTA) in the abdomen was visualizing the aorta and its branches. The retroperitoneal vessels are readily apparent on NECT, CECT, and CTA, and these modalities can be used to assess for abdominal aortic aneurysm (AAA), mycotic aneurysm, and ruptured AAA, as well as stenoses or thromboses of the tributary arteries. During the workup of abdominal or back pain, a NECT and CECT of the abdomen may be ordered, and the aorta should always be evaluated. On CECT, it is possible to measure both the lumen diameter and the entire circumference of the aneurysm. (See Insert Figures 17 and 18.) In many cases, CECT is avoided in the suspected AAA rupture, secondary to the high mortality rate and the patients' poor condition, but if the rupture is an unsuspected finding, highly enhanced fluid can be visualized within the abdominal cavity in the proximity of the aorta.19 Although sonography may detect AAA in asymptomatic patients, when a rupture is high on the differential, an NECT is the first choice for imaging. If hemorrhage is present, para-aortic fat streaking is a common finding, and fluid may be visualized in the retroperitoneum or the peritoneal cavity. (See Insert Figures 17 and 18.) When fluid is present in the abdominal space, the most likely cause is usually hemorrhage, and it is important to take note of the attenuation of the fluid, since this holds clues to the age of the bleed and the rate of hemorrhage. Isodense blood is generally fresh, indicating a rapid bleed and generally obviates emergency surgery. Fluid that appears as separate layers of differing attenuation may indicate a slower bleed as older, hyperdense layers of blood settle beneath the more recent, isodense strata.2 Aortic dissection can be identified by the detection of an intimal flap on NECT. (See Insert Figure 19.) The distinction between the true and false lumen requires the use of intravascular contrast.
Bone
Finally, the bone window of the abdomen is an important consideration in the abdominal CT. Assessment of the lumbar spine and pelvis is similar to other bone windows in that the vertebrae and pelvic bones should be scanned for overt fractures or anomalies, and density should be analyzed for areas of decreased mineralization or abnormal ossification. Degenerative and lytic lesions should be noted, as well as symmetry and proper orientation.
Conclusion
The use of CT in the emergency department is increasing almost daily due to the increased availability of CT scans on an emergent basis and the increased quality of diagnostic imaging, especially with the recent advances in multislice CT. With the profusion of diagnostic imaging technologies in the emergency physician's arsenal, it is important to consider when to rely on options that involve less radiation exposure and less cost and when to opt for CT. For many renal indications, CT is the imaging modality of choice, especially if IVU or sonography is inconclusive. For urinary stones, CT has a sensitivity of 96-100% and specificity of 94-99%,30 and for acute pyelonephritis (although usually diagnosed based on clinical symptoms, it can be confused with other disease processes and occasionally requires imaging), CT is much more sensitive than IVP.31 Abdominal aortic aneurysms can be diagnosed initially by ultrasound, but free blood and abdominal branches are more accurately detected by CT.32 In the gastrointestinal tract and biliary tree, the preferred study often varies by location and organ. For example, CT has a high sensitivity and specificity for detecting appendicitis,24 good positive and negative predictive values for bowel ischemia,29 and multislice CT has been shown to be the most dependable study to detect bowel perforation.33 On the other hand, although CT can diagnose unexpected findings of gallstones or cholecystitis, ultrasound or hepatobiliary iminodiacetic acid (HIDA) cholescintigraphy are the preferred methods of evaluation when acute cholecystitis is suspected.32 However, even while always taking into account radiation exposure risks and with precautions in children and pregnant patients (as mentioned above), for most abdominal indications, CT is the imaging modality of choice.
References
1. Computed Tomography: Its History and Technology. Informational brochure. Siemens AG, Medical Solutions; Erlangen, Germany.
2. Terrier F, Grossholz M, Becker CD. Spiral CT of the Abdomen. Berlin: Springer-Verlag, 2000.
3. Harris J, Nelson R. Abdominal imaging with multidetector computed tomography. J Comput Assist Tomogr 2004;28:S17-S19.
4. Federle M, et al. Diagnostic Imaging: Abdomen. Altona: Amirsys, 2004.
5. Anderson BA, Salem L, Flum DR. A systematic review of whether oral contrast is necessary for the computed tomography diagnosis of appendicitis in adults. Am J Surg 2005;190:474-478.
6. Mossman K. Radiation Risks in Perspective. Boca Raton: Taylor & Francis, 2007:50-60.
7. Brenner DJ, Doll R, Goodhead DT, et al. Cancer risks attributable to low doses of ionizing radiation: Assessing what we really know. Proc Natl Acad Sci USA 2003;100:13761-13766.
8. Hirshfeld JW, et al. Clinical competence statement on physician knowledge to optimize patient safety and image quality in fluoroscopically guided invasive cardiovascular procedures: A report of the American College of Cardiology Foundation/American Heart Association/American College of Physicians Task Force on Clinical Competence and Training. Circulation 2005;111:511–532.
9. Silverman P. Multislice Computed Tomography: A Practical Approach to Clinical Protocols. Philadelphia: Lippincott Williams & Wilkins, 2002:
231-235.
10. Health Physics Society web site. Jacobus JP. Medical and dental patient issuesDiagnostic x-ray and CT. http://www.hps.org/publicinformation/ate/q5425.html. Accessed 12/18/2007.
11. Toppenberg K, Hill A, Miller D. Safety of radiographic imaging during pregnancy. Am Fam Physician 1999;59:1813-1819.
12. Lowe S. Diagnostic radiography in pregnancy: Risks and reality. Aust N Z J Obstet Gynaecol 2004;44:191-196.
13. Winer-Muram H, Boone JM, Brown HL, et al. Pulmonary embolism in pregnant patients: Fetal radiation doses with helical CT. Radiology 2002;224:487-492.
14. Groves AM, Yates SJ, Win T, et al. CT pulmonary angiography versus ventilation-perfusion scintigraphy in pregnancy: Implications from a UK survey of doctors' knowledge of radiation exposure. Radiology 2006;240:765-770.
15. Haaga J, Lanzieri C, Gilkeson R. CT and MR Imaging of the Whole Body. St. Louis: Mosby, 2003:1538-1541.
16. Wang LJ, Ng CJ, Chen JC, et al. Diagnosis of acute flank pain caused by ureteral stones: Value of combined direct and indirect signs on IVU and unenhanced helical CT. European Radiology 2004;14:1634-1640.
17. Pfister SA, Deckart A, Laschake S, et al. Unenhanced helical computed tomography vs intravenous urography in patients with acute flank pain: Accuracy and economic impact in a randomized prospective trial. Eur Radiol 2003;13:2513-2520.
18. Heidenreich A, Desgrandschamps F, Terrier F. Modern approach of diagnosis and management of acute flank pain: Review of all imaging modalities. European Urology 2002;41:351-362.
19. Ros M, Mortele K. CT and MRI of the Abdomen and Pelvis. Philadelphia: Lippincott Williams & Wilkins, 2007.
20. Zagoria R. Genitourinary Radiology. Philadelphia: Mosby, 2004:187-191.
21. Kircher MF, Rhea JT, Kihiczak D, et al. Frequency, sensitivity, and specificity of individual signs of diverticulitis on thin-section helical CT with colonic contrast material: Experience with 312 cases. AJR Am J Roentgenol 2002;
178:1313-1318.
22. McKay R, Shepherd J. The use of the clinical scoring system by Alvarado in the decision to perform computed tomography for acute appendicitis in the ED. Am J Emerg Med 2007;25:489-493.
23. Raman SS, Lu DS, Kadell BM, et al. Accuracy of nonfocused helical CT for the diagnosis of acute appendicitis: A 5-year review. AJR Am J Roentgenol 2002;178:1319-1325.
24. Choi D, Park H, Lee YR, et al. The most useful findings for diagnosing acute appendicitis on contrast-enhanced helical CT. Acta Radiol 2003;44:574-582.
25. Callahan M, Rodriguez D, Taylor G. CT of appendicitis in children. Radiology 2002;224:325-332.
26. Foley TA, Earnest F 4th, Nathan MA, et al. Differentiation of nonperforated from perforated appendicitis: Accuracy of CT diagnosis and relationship of CT findings to length of hospital stay. Radiology 2005;235:89-96.
27. Bixby SD, Lucey BC, Soto JA, et al. Perforated versus nonperforated acute appendicitis: Accuracy of multidetector CT detection. Radiology 2006;241:780-786.
28. Halligan S, Fenlon H. New Techniques in Gastrointestinal Imaging. New York: Marcel Dekker, 2004:245-251.
29. Wiesner W, Hauser A, Steinbrich W. Accuracy of multidetector row computed tomography for the diagnosis of acute bowel ischemia in a non-selected study population. Eur Radiol 2004;14:2347-2356.
30. Reiser M, Takahashi M, Modic M, Bruening R. Multislice CT. New York: Springer, 2001:32.
31. Tung G, Zagoria R, Mayo-Smith W. Case Review: Genitourinary Imaging. St. Louis, 2000:14.
32. Novelline R. Squire's Fundamentals of Radiology. Cambridge: Harvard University Press, 1997.
33. Pinto A, Scaglione M, Giovine S, et al. Comparison between the site of multislice CT signs of gastrointestinal perforation and the site of perforation detected at surgery in forty perforated patients. Radiol Med (Torino) 2004;108:208-217.
How many of your patients have a CT scan during their ED evaluation? Many hospitals report rates of 20% or more. A significant number of these scans are of the abdomen and pelvis. It is important for the emergency physician to have the knowledge to view and interpret these images.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
