Chemoprevention of Colon Cancer by Vitamin D and Calcium
By Lynn Keegan, RN, PhD, HNC, FAAN, and Gerald T. Keegan, MD, FACS
Part 2 of a 2-Part Series
In part one of this two-part series, the natural history and pathogenesis of colorectal cancer was explored in considerable detail. NSAIDs and turmeric were proposed as chemopreventive agents to reduce the risk of this disease. There is increasing clinical evidence suggesting that calcium is a chemopreventive agent, and there are theoretical reasons to suggest that vitamin D also may be helpful. This article will examine the chemoprotective effects of calcium and vitamin D for colorectal cancer.
Mechanisms of Action
Calcium. There is convincing laboratory evidence that calcium reduces the risk of colorectal cancer, but previous epidemiologic studies have reported somewhat inconsistent results. A recent large prospective study confirms that higher calcium intake is associated with a modestly reduced risk of distal colorectal cancer.1 The mechanisms for this chemoprevention have yet to be defined. Studies in China suggest the reactive oxygen species (ROS) play a crucial role in induced cell apoptosis. A number of studies have demonstrated previously that perturbed cellular calcium homeostasis has been implicated in apoptosis. The authors of this study further demonstrated that an ROS increase leading to cancer cell death was due to mitochondrial calcium overload and that calcium-dependent ROS was a signal molecule that could turn off cancer cell proliferation in human colonic tissue cell culture.2 The calcium-sensing receptor (CaR) may act as a switch to turn on and turn off cancer cells. For many cell types, including intestinal epithelial cells, by acting upon these receptors, extracellular calcium concentration can switch the cellular behavior from proliferation to terminal differentiation or quiescence. As cancer is predominantly a disease of disordered balance between proliferation, differentiation, and apoptosis, disruptions in the function of the CaR could contribute to the progression of neoplastic disease.3
Vitamin D. Biological effects of 1,25-dihydroxyvitamin D [1,25(OH)2D] are mediated through nuclear transcription factors known as vitamin D receptors (VDR).4 Upon entering the nucleus of a cell, 1,25(OH)2D binds with the VDR; this in turn binds with the retinoic acid X receptor (RXR). In the presence of 1,25(OH)2D, the VDR/RXR complex binds small sequences of DNA known as vitamin D response elements (VDREs), and initiates a cascade of molecular interactions that modulate the transcription of specific genes.5 A form of vitamin D, 1,25-dihydroxyvitamin D(3), has antimitotic, pro-differentiating, and pro-apoptotic activity in tumor cells. This form of vitamin D can be synthesized and degraded not only in the kidney but also extrarenally in intestinal cells. Studies in Vienna using real-time polymerase chain reaction, immunoblotting, and immunofluorescence in cancerous and normal tissue suggested that autocrine/paracrine antimitotic activity of 1,25-dihydroxyvitamin D(3) could prevent intestinal tumor formation. Well-differentiated tumors in early stages of malignant deterioration still showed VDRs enhanced over normal colonic tissue, whereas high-grade undifferentiated tumors had lost their receptor expression.6
There appears to be a relationship between oncogenes and VDRs. The ras is the most characterized oncogene in human cancer, and yet there are no effective therapeutics to selectively target this oncogene. Cell death in K-ras-activated colon cancer cells has been linked to the down-regulation of VDR by an AP-1-dependent mechanism. Forced VDR expression in K-ras-activated cells inhibits p38 activation-induced cell death, and inhibition of endogenous VDR protein expression in K-ras- disrupted cells increased the arsenite-induced toxicity. Analysis of an additional two human colon cancer cell lines with and without K-ras mutation also showed a K-ras- and VDR-dependent toxicity of MKK6. Hence, p38 pathway activation selectively induces cell death in K-ras-mutated human colon cancer cells by mechanisms involving the suppression of VDR activity. These findings, although not directly explaining the role of vitamin D, do indicate that apoptosis of the cancer cells is occurring through the regulation of the VDR.7
Epidemiological and preclinical data in Spain indicate that vitamin D and its most active metabolite 1alpha,25-dihydroxyvitamin D(3) [1alpha,25(OH)2D(3)] have anticancer activity. This substance, a nonhypercalcemic vitamin D(3) derivative, has been found to induce proliferation arrest and epithelial differentiation of human SW480-ADH colon cancer cells. The postulated mechanism of action is that 1alpha,25(OH)2D(3) causes a profound change in gene expression profile and may be effective in both colon cancer prevention and treatment.8 Many authors have concluded that dietary calcium, vitamin D, and folate can modulate and inhibit colon carcinogenesis. Supporting evidence has been obtained from a wide variety of preclinical experimental studies, epidemiological findings, and a few human clinical trials. Important molecular events and cellular actions of these micronutrients contributing to their tumor- modulating effects include a complex series of signaling events that affect the structural and functional organization of colon cells.9
Clinical Trials
Calcium. As mentioned above, there is laboratory evidence demonstrating that calcium reduces the risk of colorectal cancer. However, the clinical studies have been somewhat inconsistent.1 An extensive retrospective review of 1,346 patients using several different research engines concluded that although the evidence from two RCTs suggested that calcium supplementation might contribute to a moderate degree to the prevention of colorectal adenomatous polyps, this did not constitute sufficient evidence to recommend the general use of calcium supplements to prevent colorectal cancer.10
A study of 930 patients with a recent colorectal carcinoma was designed to examine the effect of calcium on different types of colorectal lesions. Patients were randomly assigned to 1,200 mg/d calcium carbonate or placebo. Follow-up colonoscopies were conducted approximately one and four years after the qualifying examination. The results suggested that calcium supplementation may have a more pronounced antineoplastic effect on advanced colorectal lesions than on other types of polyps.11
Vitamin D. Because of its hypercalcemic effect, few trials have studied the effect of high-dose vitamin D in chemoprevention of colon cancer. However, prospective cohort studies clearly have not found that total vitamin D intake is associated with significant reductions in colorectal cancer when other risk factors are taken into account.12-15 One five-year study of more than 120,000 people found that men with the highest vitamin D intakes had a risk of colorectal cancer that was 29% lower than men with the lowest vitamin D intakes.16 Vitamin D intake was not significantly associated with colorectal cancer risk in women. Serum 25(OH)D level, which reflects vitamin D intake and vitamin D synthesis, was inversely associated with the risk of potentially precancerous colorectal polyps.17
There are ongoing studies of the effect of nonhypercalcemic vitamin D. Data reveal that 1alpha,25(OH)2- D(3) causes a profound change in gene expression profiles and provide a basis for encouraging clinical studies using nonhypercalcemic vitamin D(3) derivatives for colon cancer prevention and treatment.8
Several authors have suggested that although the use of vitamin D is hampered by its hypercalcemic effects, the possibility exists that the level of local 1,25-dihy- droxyvitamin D levels and subsequent antitumor activity could be enhanced by the use of the phytoestrogens present in soy foods.18,19
Presumptive evidence of the role of vitamin D in colon cancer is suggested by the high incidence of the disease in populations living in areas of low sunlight exposure. This suggests 1,25-dihydroxyvitamin D3, an antimitotic prodifferentiating steroid hormone, as a potentially preventive factor since levels of the precursor 25-hydroxyvitamin D3 in serum are, to a major part, dependent upon sun exposure.
Studies in Vienna also have shown that during early tumor progression, expression of CYP27B1 and of the vitamin D receptor is upregulated. Therefore, the vitamin D system may function as a potent physiological defense against further tumor progression in cancer patients. Estrogenic substances and phytoestrogens present in soy food could, by increasing tumor tissue-located CYP27B1 activity and decreasing degradative CYP24 activity, augment tumor-localized 1,25-dihydroxyvitamin D3 levels and activity.19
Another alternative to cytotoxic drugs in the treatment of cancer is the use of differentiation therapy. Among compounds under scrutiny for this purpose is the physiologically active form of vitamin D(3), 1,25-dihydroxyvitamin D(3), and its chemically modified derivatives. As mentioned above, vitamin D(3) and its analogs increase the levels of serum calcium, and this effect, to some extent, precludes their use in cancer patients. A number of compounds have been shown to increase the differentiation-inducing and antiproliferative activities of vitamin D(3). Several agents include those that have differentiation-inducing activity of their own that is increased by combination with vitamin D(3). These agents include the retinoids and transforming growth factor-beta and plant-derived compounds and antioxidants. Turmeric, as reported in part one of this series, has been found to be one of those agents. Data reported from Israel illustrates that there are extensive, but largely unexplored, opportunities to develop combinatorial, differentiation-based approaches to chemoprevention and chemotherapy of human cancer.20
Adverse Effects
The use of vitamin D and calcium carries specific dangers. A study was performed at the Cleveland Clinic Cancer Center investigating the use of nutritional supplements in the general population and in cancer patients. Supplements are not perceived as medications and are presumed to be safe by cancer patients, who may be at risk for hypercalcemia. Many of the patients in this study developed symptomatic hypercalcemia, which the researchers suspected was caused by taking vitamin D, calcium, or shark cartilage supplements.21
Vitamin D toxicity (hypervitaminosis D) induces abnormally high serum calcium levels, which can result in bone loss, kidney stones, and calcification of organs like the heart and kidneys if untreated over a long period of time. Because the consequences of hypercalcemia can be profound, the Food and Nutrition Board established a conservative daily minimum of 2,000 IU/d for children and adults.22 Research published since 1997 suggests that the daily minimum for adults is too low and that vitamin D toxicity is very unlikely in healthy people at intake levels lower than 10,000 IU/d.23,24 As noted above, there are ongoing studies on the effects of non-hypercalcemic vitamin D.8
Dosage and Formulation
It appears that mean dietary intakes of calcium and vitamin D in the U.S. adult population are far below the adequate intake (AI) values recommended by the Food and Nutrition Board of the Institute of Medicine of the National Academies. Substantial segments of the American population have inadequate intakes and elevated risks of osteoporosis and colon cancer.
The current Code of Federal Regulations, Title 21, sets standards for the optional addition of moderate amounts of calcium and vitamin D in the enrichment of cereal-grain products, a provision that essentially is not used. Some authors have proposed the addition of calcium and vitamin D to currently enriched cereal-grain products be mandated in the United States and believe this would result in an increase in mean daily dietary intakes in the United States of approximately 400 mg calcium and 50-200 IU vitamin D. These supplementary levels are quite low, certainly would not be detrimental, and possibly could help reduce the incidences of osteoporosis and colon cancer over time and improve overall health.
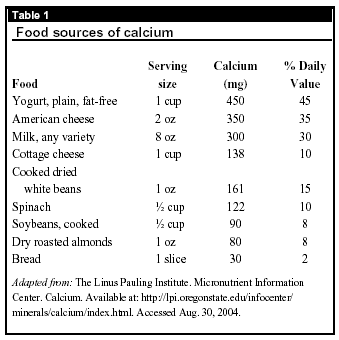
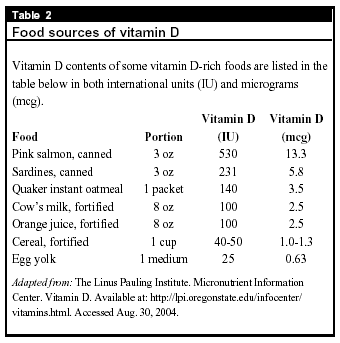
Adequate calcium intake in males and females older than age 50, the group most likely to develop colorectal cancer, is 1,200 mg/d. Postmenopausal females not receiving hormone replacement therapy should increase their intake to 1,500 mg/d. The need for vitamin D increases with age and should be in the range of 400-600 IU for adults older than age 50 and probably up to 800 IU in individuals at risk for osteoporosis. (See tables 1 and 2 for food sources of calcium and vitamin D.)
Although most young people receive enough daily sunshine to synthesize vitamin D, people who work indoors under fluorescent lighting from dawn to dusk probably have a diminished store of this vitamin. As people age, they have reduced capacity to synthesize vitamin D from sunlight exposure and may use sunscreen or protective clothing to prevent skin cancer and sun damage. People in northern climates should plan their sunlight time. As little as 5-10 minutes of sun exposure on arms and legs or face and arms three times weekly between 11 am and 2 pm during the spring, summer, and fall at 42 degrees latitude should provide a light-skinned individual with adequate vitamin D and allow for storage of any excess for use during the winter with minimal risk of skin damage.25
Conclusion
Although the clinical evidence to date is not definitive, calcium supplementation seems to contribute to the prevention of colorectal adenomatous polyps and has significant antineoplastic activity on advanced colorectal lesions. The role of vitamin D and its analogues as chemopreventive agents for colorectal cancer still remains in the realm of the theoretical.
Recommendation
Based on the available evidence, patients who are at high risk of colorectal cancer by virtue of detected colonic polyps found at the time of colonoscopy, family history, or other concomitant disease processes should be advised to use calcium supplementation. Moderate dietary supplementation with both calcium and vitamin D should be encouraged. Calcium citrate with vitamin D (containing 630 mg calcium and 400 IU vitamin D3) is probably the best non-food dietary calcium supplement and is the only form of calcium supplementation which is absorbed in the presence of the H2 blockers and proton pump inhibitors. It has the additional advantage of not causing gastric irritation and can be taken at any time, with or without meals.
Gerald T. Keegan, MD, is Emeritus Staff, Scott & White Clinic and Hospital, and former Professor of Surgery (Urology), Texas A&M University School of Medicine.
References
1. Chia V, Newcomb PA. Calcium and colorectal cancer: Some questions remain. Nutr Rev 2004;62:115-120.
2. Wang H, et al. Both calcium and ROS as common signals mediate Na(2)SeO(3)-induced apoptosis in SW480 human colonic carcinoma cells. J Inorg Biochem 2003; 97:221-230.
3. Rodland KD. The role of the calcium-sensing receptor in cancer. Cell Calcium 2004;35:291-295.
4. Sutton AL, MacDonald PN. Vitamin D: More than a "bone-a-fide" hormone. Mol Endocrinol 2003;17: 777-791.
5. Norman AW. Vitamin D. In: Bowman BA, Russell RM, eds. Present Knowledge in Nutrition. 8th ed. Washington, DC: ILSI Press; 2001:146-155.
6. Bises G, et al. 25-hydroxyvitamin D3-1alpha-hydroxylase expression in normal and malignant human colon. J Histochem Cytochem 2004;52:985-989.
7. Qi X, et al. p38 MAPK activation selectively induces cell death in K-ras-mutated human colon cancer cells through regulation of vitamin D receptor. J Biol Chem 2004;279:22138-22144. Epub 2004 Mar 22.
8. Palmer HG, et al. Genetic signatures of differentiation induced by 1alpha,25-dihydroxyvitamin D3 in human colon cancer cells. Cancer Res 2003;63:7799-7806.
9. Lamprecht SA, Lipkin M. Chemoprevention of colon cancer by calcium, vitamin D and folate: Molecular mechanisms. Nat Rev Cancer 2003;3:601-614.
10. Weingarten MA, et al. Dietary calcium supplementation for preventing colorectal cancer and adenomatous polyps. Cochrane Database Syst Rev 2004;(1): CD003548.
11. Wallace K, et al. Effect of calcium supplementation on the risk of large bowel polyps. J Natl Cancer Inst 2004;96:893-894.
12. Terry P, et al. Dietary calcium and vitamin D intake and risk of colorectal cancer: A prospective cohort study in women. Nutr Cancer 2002;43:39-46.
13. Martinez ME, et al. Calcium, vitamin D, and the occurrence of colorectal cancer among women. J Natl Cancer Inst 1996;88:1375-1382.
14. Kearney J, et al. Calcium, vitamin D, and dairy foods and the occurrence of colon cancer in men. Am J Epidemiol 1996;143:907-917.
15. Bostick RM, et al. Relation of calcium, vitamin D, and dairy food intake to incidence of colon cancer among older women. The Iowa Women’s Health Study. Am J Epidemiol 1993;137:1302-1317.
16. McCullough ML, et al. Calcium, vitamin D, dairy products, and risk of colorectal cancer in the Cancer Prevention Study II Nutrition Cohort (United States). Cancer Causes Control 2003;14:1-12.
17. Peters U, et al. Vitamin D, calcium, and vitamin D receptor polymorphism in colorectal adenomas. Cancer Epidemiol Biomarkers Prev 2001;10:1267-1274.
18. Cross HS, et al. Regulation of extrarenal vitamin D metabolism as a tool for colon and prostate cancer prevention. Recent Results Cancer Res 2003;164:413-425.
19. Cross HS, et al. Regulation of extrarenal synthesis of 1,25-dihydroxyvitamin D3—relevance for colonic cancer prevention and therapy. Mol Aspects Med 2003;24: 459-465.
20. Danilenko M, Studzinski GP. Enhancement by other compounds of the anti-cancer activity of vitamin D(3) and its analogs. Exp Cell Res 2004;298:339-358.
21. Lagman R, Walsh D. Dangerous nutrition? Calcium, vitamin D, and shark cartilage nutritional supplements and cancer-related hypercalcemia. Support Care Cancer 2003;11:232-235. Epub 2003 Jan 15.
22. Food and Nutrition Board. Institute of Medicine. Vitamin D. Dietary Reference Intakes: Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington DC: National Academies Press; 1999:250-287.
23. Vieth R. Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr 1999;69:842-856.
24. Vieth R, et al. Efficacy and safety of vitamin D3 intake exceeding the lowest observed adverse effect level. Am J Clin Nutr 2001;73:288-294.
25. Holick MF. Vitamin D deficiency: What a pain it is. Mayo Clin Proc 2003;78:1457-1459.
Keegan L, Keegan GT. Chemoprevention of colon cancer by vitamin D and calcium. Altern Ther Women's Health 2005;7(1):1-5.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
