Palifermin and Mucositis Among Patients With Hematologic Malignancies: Closing a Portal of Entry for Bacteria?
Palifermin and Mucositis Among Patients With Hematologic Malignancies: Closing a Portal of Entry for Bacteria?
Abstract & Commentary
J. Peter Donnelly, PhD, Clinical Microbiologist, University Hospital, Nijmegen, The Netherlands, Section Editor, Microbiology, is Associate Editor for Infectious Disease Alert
Synopsis: Palifermin, a recombinant form of human keratinocyte growth factor, reduced the duration and severity of oral mucositis and the incidence of febrile neutropenia and bacteremia among patients given intensive chemotherapy and radiotherapy for hematologic cancers.
Source: Speilberger, et al. N Engl J Med. 2004;351:2590-2598.
Oral mucositis is a common complication of the intensive chemotherapy and radiotherapy given to patients to treat hematologic malignancies, and for which there has been no effective treatment. The development of mucositis is not fully understood, but is thought to arise from the damage caused by cytotoxic agents to the epithelia of the oral cavity and digestive tract as a whole. Through the years, a variety of remedies have been tried and tested but none has been found convincingly to prevent or ameliorate mucositis. Keratinocyte growth factor is a 28-kD protein of the family of fibroblast growth factors with keratinocyte stimulating activity, and palifermin is a truncated version with similar biologic activity to that of the native protein, but is more stable. The compound protected the epithelia of rodents exposed to chemotherapy, radiotherapy, and hematopoietic stem-cell transplantation and phase 1 trials indicated that doses of up to 80 µg/kg/day palifermin for 3 consecutive days were well tolerated. In this study, the ability of palifermin to decrease therapy was evaluated. In this large double-blind study the effect of palifermin on the development of oral mucositis was evaluated in 212 patients undergoing autologous stem-cell transplantation using the same myelotoxic regimen of fractionated total-body irradiation, etoposide and cyclophosphamide for lymphoma, leukemia or multiple myeloma. Palifermin at a dose of 60 µg/kg or placebo was given for 3 consecutive days immediately before starting and again for 3 consecutive days immediately after stopping myelotoxic therapy. Oral mucositis was evaluated daily form 8 days before until 28 days after transplantation.

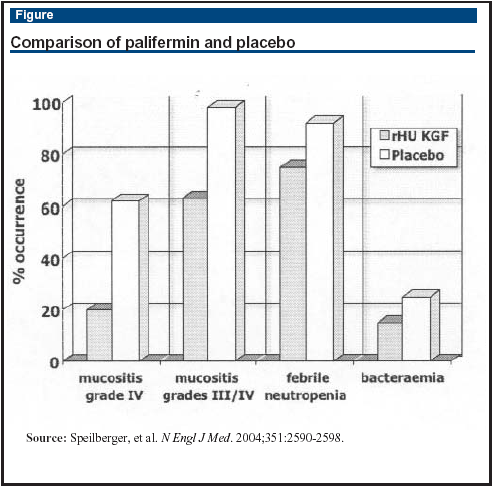
The incidence of World Health Organization (WHO) grade 4 oral mucositis for the palifermin and placebo groups, respectively, were 20 % vs 62% (P < 0.001), and the incidence of severe oral mucositis (WHO grade 3 or 4) was 63% and 98% (P < 0.001). Febrile neutropenia affected 75% in the palifermin group and 92% in the placebo group and bacteremia developed in 15% and 25%, respectively.
Patients in the palifermin reported less pain (area-under-the-curve score, 29.0 [range, 0 to 98] vs 46.8 [range, 0 to 110]; P < 0.001), and used less opioid analgesics (median, 212 mg of morphine equivalents [range, 0 to 9418] vs 535 mg of morphine equivalents [range, 0 to 9418] (P < 0.001). Palifermin was well tolerated, and the most commonly reported adverse reactions attributed to the drug were rash, pruritus, erythema, mouth and tongue thickness or discoloration, and taste alteration.
Comment by J. Peter Donnelly
Palifermin (Kepivance : www.kepivance.com) is the first drug product to be licensed for oral mucositis, mainly on the evidence supplied by this study. The drug is officially indicated "to decrease the incidence and duration of severe oral mucositis in patients with hematologic malignancies receiving myelotoxic therapy requiring hematopoietic stem cell support." In other words, those receiving myelotoxic therapy in preparation for an autologous stem cell transplant. There are no data available, as of yet, to broaden its use to include allogeneic hematopoietic stem cell transplant recipients or others treated solely with myelotoxic chemotherapy.
Apart from relieving the pain of mucositis, palifermin lowered the incidence of febrile neutropenia and even bacteremia. Unfortunately, we are not told anything of the etiology, but if this finding were to be reproduced, it would be very remarkable indeed, since the observed reduction of almost 50% is in the same order as that found for fluoroquinolone prophylaxis (Cruciani, et al. Clin Infect Dis. 1996;23:795-805). We are not told the antibiotic usage, but these drugs are not generally employed for this purpose in North America, as opposed to Europe. There are opportunistic microorganism encountered in blood cultures that are associated with mucositis (see Table), so it would have been relatively straightforward to have documented these bacteremias to test the hypothesis. Certainly, future studies should pay particular attention to these infectious complications.
Mucositis is a complex biological process thought to begin with an inflammatory phase in which pro-inflammatory cytokines are released, followed by epithelial cell death, which precedes ulceration, during which point, infection can occur. Therefore, if the reduction in bacteremia is genuine, it would lend further support to the notion that the offending organisms gain access through damaged tissue, which, if protected, as appears to be the case with palifermin, would no longer offer a portal of entry to the opportunists. It could also revolutionize the supportive care of these patients and dramatically lower costs brought about by the use of antimicrobial agents empirically. Therefore, besides opening up new horizons for treating mucositis palifermin might help pave the way to better understanding of how infections arise after myelotoxic therapy and how best to prevent them. At the very least it is hoped that the results of this study will provide greater impetus to conduct more specific studies to unravel the infectious complications associated with mucosal barrier injury in general and mucositis of the mouth and gut in particular to the benefit of patients and health-care systems alike.
Palifermin, a recombinant form of human keratinocyte growth factor, reduced the duration and severity of oral mucositis and the incidence of febrile neutropenia and bacteremia among patients given intensive chemotherapy and radiotherapy for hematologic cancers.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
