Complications of Pregnancy: Part I
Complications of Pregnancy: Part I
Authors: Halleh Akbarnia, MD, FACEP, FAAEM, St. Francis Hospital, Evanston, IL; Teaching Faculty for Resurrection Emergency Department Residency Program, Evanston, IL.
Peer Reviewers: Catherine A. Marco, MD, FACEP, Professor, Department of Surgery, Division of Emergency Medicine, University of Toledo College of Medicine, Toledo, OH; and Elaine Thallner, MD, MS, Department of Emergency Medicine, Cleveland Clinic, Cleveland, OH.
In my emergency department (ED), we see a large number of pregnant patients, so you would think we would be good at evaluating them. But sometimes familiarity can lead to a sense of complacency, such as assuming that the vomiting and abdominal pain that brought the patient to the hospital are due to the pregnancy. It is important always to consider that the symptoms could just as easily be due to a medical complication of pregnancy as well as to the pregnancy itself.
These next two issues of Emergency Medicine Reports will cover many of the complications and problems that may cause the pregnant woman to come see you. Part I will focus on miscarriage, ectopic pregnancy, gestational trophoblastic disease, and venous thromboembolic states. Part II will discuss hypertensive disorders, amniotic fluid embolism, and late pregnancy bleeding.
J. Stephan Stapczynski, MD
Introduction
Treating a patient during the course of her pregnancy is a challenge for the physician. Symptoms are often identified as normal although they may be completely foreign to the patient. It is "normal" to feel nauseated and have emesis. It is "normal" to feel pain in the lower parts of the abdomen as the round ligaments stretch. It is "normal" to see some swelling in the legs as pregnancy progresses. The challenge for both the patient and her physician lies in determining when these symptoms are actually normal in an individual pregnancy and when they may signal a concern.
This review will briefly touch on miscarriages and ectopic pregnancies, but will concentrate more on those entities not typically discussed in the emergency medicine literature, such as gestational trophoblastic disease, as well as complications seen in the second half of pregnancy such as hypertensive disorders, venothromboembolic states, amniotic fluid embolism and late pregnancy bleeding. The objective of this review is to increase the physicians' ability to recognize diseases that are potentially life-threatening to both the mother and the fetus, and to understand how to initiate timely treatment plans and minimize legal risks.
Complications of Early Pregnancy
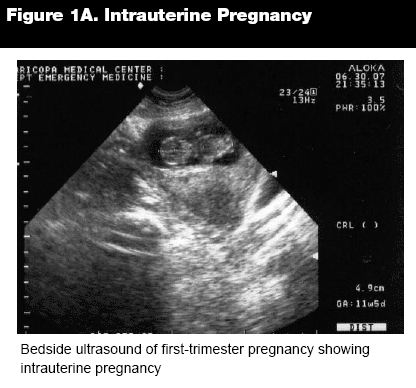
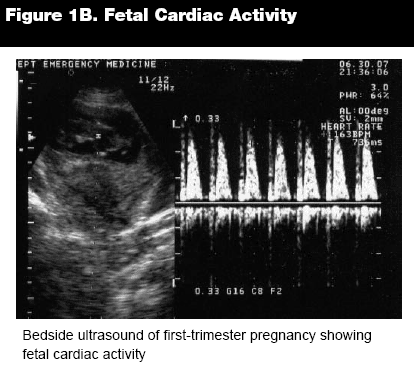
Approximately 25-30% of pregnancies experience first-trimester bleeding, so it is common for the patient to present to the ED, and sometimes the bleeding is accompanied by pain. Although ectopic pregnancy must be considered in all patients with these symptoms, more commonly the ED physician will encounter a patient undergoing threatened or actual miscarriage. For many patients a simple bedside examination, laboratory work including a beta human chorionic gonadotropin (beta-hCG) level, and a transabdominal or transvaginal ultrasound will answer the simple questions posed in these caseswhether the patient has an intrauterine pregnancy and whether the pregnancy is viable. (See Figures 1A and 1B.) Even after examination and these ancillary tests, many times the practitioner may still be left with uncertainty, however decisions still need to be made with the information that is available.
 |
 |
 |
Bleeding in Early Pregnancy
Spontaneous Abortion or Miscarriage.The medical term abortion refers to any process, intentional or spontaneous, in which pregnancy terminates prematurely, with death and expulsion or removal of the fetus. However, because of varied opinions about issues surrounding medically induced abortions, in recent years there has been discussion within the medical community to avoid using the term "abortion" in favor of the term more accepted by patients for spontaneous pregnancy loss, "miscarriage."1
Almost all miscarriages arise from an abnormality in the host, the mother, or the actual fertilized ovum. A recent meta-analysis found that approximately half of all miscarriages (spontaneous abortions) occur because of chromosomal abnormalities, more commonly autosomal trisomy.2 It is thought that overall, approximately one-third of pregnancies end before they are even recognized, that is, before the first missed menses. After the first menses missed, another 10% (although the range is wide for both numbers) are recognized as clinical miscarriages. Therefore, approximately half of all pregnancies actually progress after implantation is complete. Most commonly, the term miscarriage is used for fetal demise that occurs before 20 weeksafter 20 weeks, the miscarriage is termed premature birth. The complaint of vaginal bleeding itself is a presenting sign in a high percentage of associated miscarriages. Of the approximately 25-30% of women who experience some form of first-trimester bleeding, about half will go on to miscarry, although this number varies according to other factors. As patients increase in age and parity, the risk of miscarriage also will increase. Younger patients, below the age of 20, only have a 12% risk of miscarriage while patients older than 40 have an increased risk of 26%.3-6
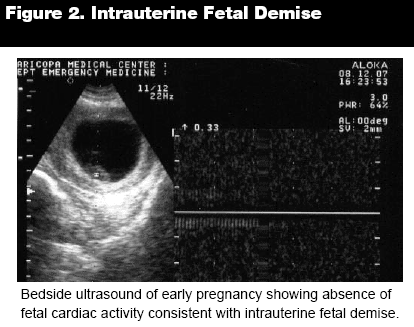
Missed Miscarriage, or Intrauterine Fetal Demise (IUFD). The term missed abortion, or missed miscarriage, is not used as widely anymorethis terminology has been modified to include the timing of fetal death. It is now known that fetal death can occur days or even weeks before symptoms are manifested or an ultrasound obtained. A beta-hCG level can help in determining time of demise.5,7 Early IUFD is defined as fetal demise noted before 20 weeks, intermediate IUFD represents demise between 20 and 27 weeks, and late IUFD is defined as death after 28 weeks gestation. This has also been termed first or second trimester fetal death when there is a fetus but no heart tones and at least a 15 mm crown-rump length. (See Figure 2.) Anembryonic gestation, or blighted ovum, could be part of the same clinical process. This happens in the first trimester when the fertilized egg attaches to the endometrium but the embryo never develops. In the majority of early miscarriages, fetal death happens well before it is clinically determined.8
 |
Threatened Miscarriage. With threatened miscarriage, the patient will have vaginal bleeding occurring typically before 12 weeks of gestation. Threatened miscarriage often has pelvic pain, typically described as cramping that may radiate to the low back. On pelvic examination, there may be blood in the vaginal vault, either clotted or liquid, and sometimes blood from the cervical os, but the os is closed when gently probed with the fingertip. With a threatened miscarriage, the pregnancy is still viable and fetal cardiac activity can be detected by transabdominal or transvaginal ultrasound. It was reported at one time that 35-50% of these patients would progress to complete miscarriage. Now, with the more frequent use of ultrasound, if the fetus is shown to have a heartbeat, there is an 85% chance that the fetus will progress to term.9
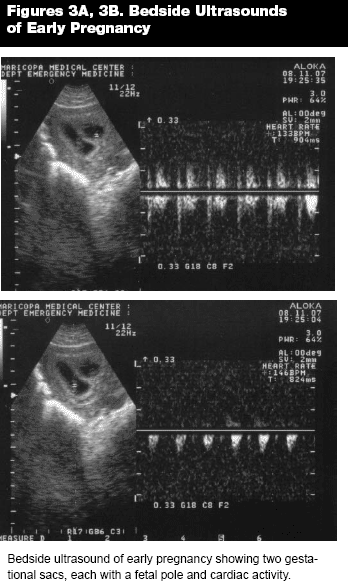
Ultrasound is extremely useful to evaluate patients with first trimester bleeding, both to localize an intrauterine pregnancy (e.g., exclude ectopic pregnancy) and determine viability. (See Figures 1A and 1B.) Ultrasound can also identify multiple gestations. (See Figures 3A and 3B.) Ultrasound has reduced ability to accurately localize a very early pregnancy. Studies indicate that a serum beta-HCG level of less than 1500 to 2500 IU/mL limits the accuracy of transvaginal ultrasound or a level of less than 3500 to 6000 IU/mL limits the accuracy of transabdominal ultrasound to localize pregnancy.10,11 If the ultrasound cannot localize a gestational sac, embryo, or yolk sac, the diagnosis of threatened miscarriage is suspected and the possibility of an ectopic pregnancy remains. Such patients should receive regular follow-up until the location of the pregnancy can be determined.
 |
In this age of ultrasound, there is little reason not to use this modality to help determine the location of a pregnancy, whether it is performed at the bedside by an emergency physician or obstetrician, or an ultrasound technician. With proper training and appropriate hospital credentialing, emergency physicians can perform bedside ultrasound for the assessment and evaluation of early pregnancy complications.14-16
For patients with threatened miscarriage, good discharge instructions from the ED and early visits to the obstetrician are important. In a large retrospective study, it was shown that more than half of patients with early ectopic pregnancies were discharged with the diagnosis of "threatened abortion."
Inevitable Miscarriage. With inevitable miscarriage, the cervical os is open, fetal cardiac activity has ceased, but products of conception have yet to be expelled. Usually, this is seen in the first 20 weeks of pregnancy. (See Figure 2.)
Incomplete Miscarriage. A history of passing blood and parts of products of conception with a cervical os that is closed at time of examination is termed an incomplete miscarriage. Ultrasound is useful to detect retained products of conception, but the detection of echogenic material within the uterus can also be blood clots, infected or necrotic tissue, as well as placental material.17,18 In the past, these patients underwent dilatation and curettage, however, now it is likely that more conservative and expectant care works well with these patients who have a small amount of endometrial content determined by ultrasound.9 It has been shown in these women that there is an 82-96% rate of completion of miscarriage without further surgical or medical management.6,19-22 If the decision is made with the obstetricians to use misoprostol (Cytotec), it has been shown that using the intravaginal formulation works better than the parenteral one.23 The typical dose is 600 mg placed intravaginally. The patient should be instructed to rest and advised that she will experience some bleeding and cramping. Analgesics, such as nonsteroidal anti-inflammatory agents, should be given to help with the cramping.24
Complete Miscarriage. In a complete miscarriage, the patient is at less than 20 weeks gestation, all fetal tissue has been passed, and the cervical os is closed. It is important to note that many women may mistake the passage of clots for products of conception; therefore ultrasound will be important here as well. The routine use of ultrasound in women who were thought to have a complete miscarriage by clinical criteriaclosed cervical os and decreasing pain and bleeding after reported or observed passage of tissuehas found that up to 30% will have retained products of conception. (See Figures 4A and 4B.) A very early pregnancy, with a closed cervical os and no visible gestation sac by ultrasound may be mistaken for a complete miscarriage. Likewise, an ectopic pregnancy may be mistaken for an complete miscarriage.5,25
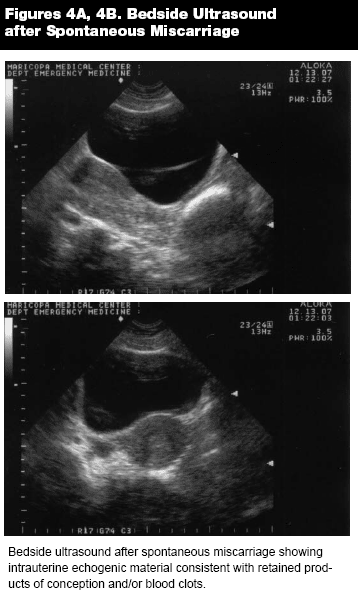 |
Septic Miscarriage. Infection of the uterine cavity after a complete or incomplete miscarriage is termed a septic abortion or miscarriage. Clinical symptoms include fever, typically above 101°F, chills, severe abdominal pain and cramping, and foul-smelling vaginal discharge. Sometimes there is prolonged heavy bleeding and low backache. Signs of a septic miscarriage include severe uterine tenderness and purulent exudate from the cervical os. The maternal systemic response to a septic miscarriage may include shock, with tachycardia and low blood pressure, and end-organ dysfunction, with confusion, dyspnea, and oliguria. These patients require aggressive hemodynamic support, prompt antibiotic administration, and urgent obstetric consultation.
Evaluating the Possibility of Miscarriage. During the first 20 weeks of pregnancy, a pelvic examination in the ED is the appropriate initial step to determine if there is bleeding, ascertain the quality and quantity of the bleeding, evaluate for tissue in the os or vaginal vault, and check for cervical dilation. If there is tissue, it should be sent for pathological examination. Laboratory tests should be obtained, including a blood type, Rh factor and antibody screen, a complete blood count if indicated, a quantitative beta-HCG, and a urinalysis to determine if a urinary tract infection is coexistent. Either transabdominal or transvaginal ultrasound should be done to localize pregnancy, determine fetal viability, and assess for retained products. To reiterate, an empty uterus on ultrasound in early stages of gestation may signal spontaneous miscarriage, however the diagnosis is never definitive until ectopic pregnancy is ruled out. Even if an intrauterine pregnancy is detected, the possibility of a coexistent ectopic pregnancy should not leave the practitioner's mind. The coexistence of intrauterine and ectopic pregnancies is termed a heterotopic pregnancy. The reported incidence is 1-3 per 10,000 pregnancies, but the rate can be as high as 1% in women who are using assisted reproductive techniques.26-29
Ectopic Pregnancy. The diagnosis of ectopic pregnancy must be considered in every female of childbearing years with abdominal pain that presents to the ED. Ectopic pregnancy is defined as a pregnancy that is implanted outside the uterus and one that can have devastating effects on the mother if not diagnosed.30 Typically occurring within early pregnancy between weeks 8-12, a female with an ectopic pregnancy can present in a multitude of ways. Between 1970 and 1992, studies based on CDC-derived data estimated the rate of ectopic pregnancies to have risen from 4.5 to 19.2 per 1000 reported pregnancies. Since 1992, as Zane et al have stated, it is difficult to obtain reliable estimates of the rate of ectopic pregnancy due to changes in the methods of diagnosis and options for management.31 But what was observed from a series of studies on the use of ultrasound in the emergency department in the 1990s and early part of this century was that ectopic pregnancy was found in between 6 and 16% of women who presented with abdominal pain and vaginal bleeding. Of note, up to 40% of ectopic pregnancies are missed during their first evaluation in the ED. It is the leading cause of death during the first trimester of pregnancy, seen typically during early pregnancy between weeks 8-12, and the second leading cause of maternal death during the entire pregnancy.5,31-35
In the patient who has taken measures to prevent getting pregnant, such as having an IUD or getting tubal ligation, the chance of pregnancy is extremely small. If the pregnancy does occur, however, the chance that the pregnancy would become an extrauterine one would be much higher. Other risk factors that have been reported but may not be as significant include history of spontaneous abortions in the past, increasing maternal age, smoking, multiple sexual partners, and cesarean delivery.30,36,37
Another condition the practitioner must be aware of is the increasing incidence of heterotopic pregnancies, or those that occur simultaneously in the uterus as well as extrauterine. The rate of these pregnancies has increased seven times over the initial estimates made 50 years ago, and can now be seen in as many as 1 in 4000 pregnant women. This rate goes up dramatically to 1:100 to 1:500 in those women who experience in vitro fertilization.28,29
As with miscarriages, clinical diagnosis is no longer enoughthe ultrasound is now becoming standard of care in diagnosis. It has been shown that with proper training, emergency physician-performed bedside ultrasound decreases stay, cost, and morbidity and is safe to do, with the goal being identification of an intrauterine pregnancy. (See Figures 5A and 5B.) Approximately 60-70% of the patients who receive an ultrasound are found to have an intrauterine pregnancy and can be sent home safely after the initial ED visit.38-40 In a retrospective study, 64% of patients who had an ultrasound suggestive of ectopic pregnancy were ultimately diagnosed with one.
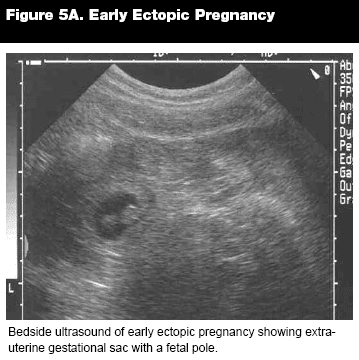 |
 |
It is also important to note that ectopic pregnancy has been diagnosed in patients with beta-hCGs less than 2000 IU/mL.41 Also, there are certain criteria that, if adhered to, have shown a huge reduction in the missed ectopic pregnancies done by ultrasound. In one study, of all patients diagnosed with ectopic pregnancy, 44% were found to have a serum beta-hCG less than 2000 IU/mL.42
Gestational Trophoblastic Disease. Gestational trophoblastic disease (GTD) actually comprises a group of several diseases that are characterized by disordered proliferation of chorionic villi. Unlike endometrial cancer, these tumors do not develop from the lining of the uterus; they start in the tissue that begins to develop immediately after conception, in the trophoblast, the layer of cells that surround the embryo. Early in development, the trophoblastic cells produce finger-like projections known as villi which will then attach to the lining of the uterus. With time, the trophoblastic layer will develop into the placenta.43,44
GTD are divided into four main types. Hydatidiform moles typically are benign tumors that are thought to occur in approximately 1 out of 1000 pregnancies in the United States. The other three forms of gestational trophoblastic diseasepersistent or invasive trophoblastic disease, choriocarcinoma, and placental-site trophoblastic tumorare either malignant or can become malignant and are seen in approximately 1 out of 20,000 to 40,000 pregnancies in the United States. All forms of GTD can be treated. The treatment, which may include chemotherapy and intensive oncologic management, can produce a complete cure in most cases.45-47
Hydatidiform Mole. The most common form of GTD is the hydatidiform mole, or molar pregnancy. The moles are actually villi that have become swollen with fluid (the term hyd meaning "water," and the fact that the moles are shaped like water droplets). There are two types of hydatidiform moles. If there is no fetal tissue, the pregnancy is termed a complete hydatidiform mole. It develops mostly because the sperm has fertilized an egg that contains no DNA. Approximately 20% of patients who have a complete mole may need more surgery or chemotherapy after the initial surgery to remove the mole. Rarely, an incomplete or partial hydatidiform mole occurs, which is one that includes two sperm fertilizing a normal egg. This will contain some fetal tissue, but also is mixed with the trophoblastic tissue. Therefore there is no viable fetus that is being formed and these will also require surgery and, at times, further treatment. Between these two categories, approximately 15% may develop into gestational trophoblastic neoplasias with persistence of molar tissue even after the pregnancy has been evacuated.46,48
Persistent or Invasive Gestational Trophoblastic Disease. Persistent gestational trophoblastic disease is the category of GTD that has not been cured by dilation and curettage. Many times, these are complete moles that persist and are invasive. This probably occurs because the surgery removes only the endometrium and does not remove the tumor that may be deeper in the myometrium. The risk of this type of invasive mole (formerly known as chorioadenoma destruens) is increased if the mother's age is greater than 40, if the mother has had a history of GTD in the past, if the uterus has become very large, or if there has been a lapse (greater than 4 months) between the time of the last menses and initiation of treatment. Of note, in about 15% of cases, the tumor can metastasize, with metastases in the lungs.45,46
Choriocarcinoma. Chorio refers to the chorion, one of the layers of the membrane surrounding the fetus. Approximately 1-3% of hydatidiform moles will progress to become choriocarcinomas, which accounts for about half of all of these malignant tumors. However, the other half do not start off as molar pregnancies. Twenty-five percent of all choriocarcinomas will develop after women have had a spontaneous miscarriage, or elective abortion or if they have had a tubal pregnancy. The other 25% will occur after normal pregnancy and delivery. If there is metastasis, it will most likely be spread to organs other than the uterus. Very rarely, choriocarcinomas can be seen developing in other parts of the body not related to pregnancy in both men and women. Most of the time these are mixed with other forms of malignancy forming a mixed germ tumor. These nongestational tumors have less favorable prognoses and tend not to respond to chemotherapy very well.46,47
Placental-Site Trophoblastic Tumor. The rarest form of GTD is the placental-site trophoblastic tumor which develops at the site of placental implantation. Most often, this tumor will develop after a normal pregnancy or abortion and will not spread to other sites in the body. These can also penetrate the myometrium, but unlike most forms of GTD, they are not sensitive to chemotherapy drugs and must be fully removed by surgery. Altogether, gestational trophoblastic tumors account for about 1% of all female reproductive system tumors.
Signs and Symptoms. GTD in the early stages is not clinically apparent. Although patients may have persistent hyperemesis gravidarum from high circulating levels of HCG (not all GTDs have high levels), the bleeding that can be associated with the tumors is what initially identifies them. This can be seen in almost all women (97%) with complete hydatidiform moles and less often with incomplete (partial) moles. Usually this bleeding is seen between weeks 6 and 16 of the pregnancy and, at times, the patient can identify tissue that may resemble grape-like clusters. Partial moles are typically diagnosed after what initially was thought to be an incomplete or missed miscarriage. After dilatation and curettage, the molar pregnancy is diagnosed.
In GTD, the absence of fetal heart tones is usually noted in the second trimester when evaluation for such takes place. If spontaneous miscarriage occurs, it usually happens during the second trimester and before 20 weeks. The patient and physician may notice the passage of grape-like hydrated vesicles. Uterine size is also larger than expected on examination by about 4 weeks in approximately 40% of patients. On occasion a patient may present with ovarian torsion.
The diagnosis of hydatidiform mole is based on the classic "snowstorm" appearance seen sonographicallythe hydropic vesicles within the uterus. (See Figure 6.) In partial molar pregnancies, cystic changes are noted. In up to two-thirds of cases, the true diagnosis will be made on pathologic examination of aborted specimens. Complications of molar pregnancies include preeclampsia (which can occur before 24 weeks when typically it is seen in the third trimester), pulmonary embolization of trophoblastic cells, hyperthyroidism, hyperemesis gravidarum, and severe uterine bleeding.43,44,46,48
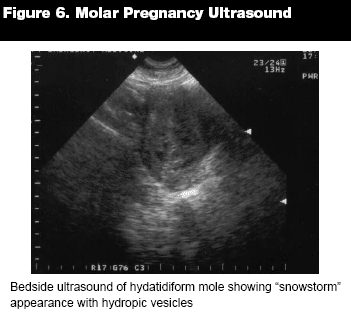 |
Treatment. GTD usually is treated with surgery, occasionally chemotherapy, and infrequently with radiation therapy. The initial treatment is usually dilatation and curettage ("D and C") to remove the hydatidiform mole. Oxytocin is sometimes given to produce contractions that help to expel uterine contents. Hysterectomy is the standard treatment for women with placental-site trophoblastic tumors, although D and C can be attempted as well. Of note, both D and C and hysterectomy will not remove the malignant cells that have already metastasized; therefore, post procedure, levels of HCG need to be monitored carefully. If they persist or rise, then chemotherapy may be the next option. This will occur in about 15% of patients with hydatidiform mole.43,44,46
The malignant forms of GTD (the invasive moles and choriocarcinomas) can develop sometimes after a complete mole is removed. They can also develop after normal pregnancy, ectopic pregnancy, or a miscarriage. Post-pregnancy GTD presents with recurrent or persistent vaginal bleeding. Physical examination shows that the uterus is not decreasing in size as expected (either remains enlarged or is growing in size) and there may be a cervical discharge consistent with endometritis. Metastases to other organs can occur, often the lungs, vagina, ovaries (luteomas), GI tract, and even the brain. Chemotherapy is usually effective for metastatic trophoblastic tumors.
A prognostic scoring system is used guide therapy in metastatic GTD. (See Table 2.) In low-risk patients (score 7 or less), a single agent such as methotrexate is typically used, often with leucovorin or folinic acid to reduce side effects. In higher-risk patients, a combination of several chemotherapy drugs including methotrexate, actinomycin-D, cyclophosphamide, etoposide, and vincristine are commonly used. If both surgery and chemotherapy are ineffective, radiation therapy can be used.43,44,46
 |
Rh Immunization. When an Rh-negative woman is exposed to an Rh-positive fetus, the woman's immune system may become stimulated to produce anti-Rh antibodies. Although throughout pregnancy, small amounts of fetal cells enter the material circulation, the mother's immune system is not triggered to respond until sufficiently large amounts of these cells are transferred, typically during with third trimester or at delivery. Approximately 15% of women will develop sensitization, and to prevent this, RhoGAM (anti-D immune globulin) is given at approximately 28 weeks to the Rh-negative mother and if the father's status is Rh-positive or unknown. This will protect the mother from spontaneous sensitization.
Other indications for RhoGAM include unexpected events that cause maternal transplacental bleeding, for example as a result of trauma, threatened or spontaneous miscarriage, ectopic pregnancies, and sometimes amniocentesis. If the patient is less than 12 weeks gestation, the recommended dose is 50 micrograms, and if she is above that, the full dose of 300 micrograms. If it is not known how far along the fetus is, the full dose of 300 micrograms can be given. This needs to be administered within 72 hours of the sensitization to prevent antibody development.49
Fetomaternal transfusion (transfer of fetal cells into the maternal circulation) can be detected by a variety of laboratory methods. The traditional method is the Kleihauer-Betke (K-B) stain test with manual cell counting, but a method using anti-HbF antibodies with flow cytometry has replaced the manual method in many laboratories. The reported sensitivity of the K-B method can be as low as approximately 0.1 mL of fetal blood in the maternal circulation. This corresponds to about 1 fetal cell per 50,000 maternal cells. The Kleihauer-Betke stain may occasionally underestimate the number of fetal RBCs present due to the fact that the fetus begins to synthesize hemoglobin A in the last trimester of pregnancy. Fetal cells, which had completed the switch to adult hemoglobin, would be counted as adult cells and the sensitivity of the K-B test to small volumes of fetomaternal transfusion is less. For these reasons, most consensus derived guidelines recommend that the physician have a low threshold to use RhoGAM in situations where Rh-positive fetal red blood cells could possibly be transfused into the Rh-negative mother, such as significant blunt abdominal trauma. A RhoGAM dose of 300 micrograms is considered sufficient to prevent maternal sensitization following exposures of up to 15 mL of fetal red blood cells (or 30 mL of whole blood).50
Recent analysis has noted that there is little evidence to support the routine use of RhoGAM in Rh-negative patients with first trimester bleeding due to miscarriage.51,52 Despite the lack of evidence of significant fetomaternal transfusion with first trimester bleeding, many guidelines and consensus statements routinely recommend RhoGAM for first trimester bleeding or miscarriage.
Venous Thromboembolic Disease. There is a five-fold increase in the incidence of venous thromboembolic disease in pregnancy, occurring in 0.5 to 3.0 per 1000 pregnancies and spread almost equally across all three trimesters.53 The diagnosis of deep venous thrombosis (DVT) or pulmonary embolism (PE) has undergone major changes in the last decade, with the creation of risk-stratification systems, D-dimer assays, and improved imaging modalities. Although, as discussed below, the application of these changes to the pregnant patient is uncertain.53,54
A sensitive (> 95%) D-dimer assay can be used to exclude thromboembolic disease in a patient judged to be low-risk (< 5% pre-test probability); a negative test in such a patient "rules-out" the diagnosis for most clinical purposes. Although a negative D-dimer assay is very useful in excluding DVT or PE in low-risk non-pregnant patients, there is uncertainty surrounding this approach in pregnant women. The first issue is that even in the absence of thromboembolism, D-dimer levels increase during pregnancy and remain elevated during the post partum period as well.53,55 The second issue is that there are multiple methods for measuring D-dimer levels, with a range of sensitivities and specificities for thromboembolism, 85-97% and 35-45%, respectively. The third issue is that the risk-stratification scoring systems have not been validated in a large population of pregnant women.24,56 Because of these factors, there is no consensus in the literature that a negative D-dimer test can be used to exclude DVT or PE in a low-risk pregnant patient.
It could be argued that because the D-dimer level rises during pregnancy, that a negative result with a sensitive test can reliably exclude thromboembolism in a pregnant patient.55,57 A recently published study of 149 pregnant women evaluated for possible DVT supports this concept.58 Using compression venous ultrasound as the gold standard, the authors found DVT in 13 (prevalence of 8.7%), and the red blood cell agglutination D-dimer assay (SimpliRED) had a sensitivity of 100% (CI, 77% to 100% [13 of 13 patients]) and specificity of 60% (CI, 52% to 68% [81 of 135]). The negative predictive value of 100% (CI, 95% to 100% [81 of 81]) supports the principle of using D-dimer to exclude DVT in pregnant women, but the relative large range in confidence intervals due to the small numbers of patients indicates that this approach should be used with caution until more clinical experience has been published.
The clinical diagnosis of DVT is unreliable, and is even more unreliable in pregnant women. Leg swelling and calf pain are common symptoms during pregnancy, particularly the third trimester. Leg swelling in pregnancy can be asymmetric, with the left leg more swollen than the right. Thus, objective tests for DVT are required before treatment. Compression ultrasound is the preferred test, it is non-invasive, does not entail radiation exposure, and can be repeated if necessary. The disadvantages of compression ultrasound is that the technique and experience of the operator influences the accuracy in diagnosing DVT. A limited study (2-point (common femoral and popliteal veins) and 3-point (common femoral and popliteal veins as well as the calf trifurcation) compression) is sometimes employed, it is recommended the entire proximal venous system to the trifurcation be imaged in symptomatic pregnant women. If the initial compression ultrasound study is negative, it is recommended that a repeat study be done in 2-3 days. If the initial ultrasound is equivocal, further imaging with magnetic resonance venography (MRV) or limited contrast venography with shielding of the abdomen is recommended. If there is suspicion of iliac vein thrombosis due to the symptom of back pain with swelling of the entire leg, MRV is the better study if the initial ultrasound was nondiagnostic.
Similar to DVT, physiologic changes during pregnancy may produce symptoms and signs, such as dyspnea and tachypnea, that overlap with finding in PE. While the overall sensitivity of at least one finding of dyspnea, tachypnea, and pleuritic chest pain for the diagnosis of PE is likely as high in pregnant as it is in non-pregnant patients (92%),59 the specificity is likely lower. The three imaging modalities used to evaluate the pregnant patient with a possible pulmonary embolus include chest CT with intravenous contrast using a protocol to visualize the pulmonary circulation for emboli (CT PE), radionuclide ventilation and perfusion lung scan (VQ) and compression venous ultrasound (CUS). There are several approaches that combine these studies. For example, one approach starts with CUS because the majority of symptomatic PE original in proximal venous system of the leg, so the finding of DVT by this test indicates the need for treatment.60 Another approach starts with a VQ scan, with the initiation of treatment if the result shows high probability, no treatment if the perfusion study is normal, and an alternative imaging study if the results are non-diagnostic.
In the recent years, more clinicians are using the CT PE in the pregnant patient because of the higher sensitivity when compared to CUS and the better specificity and lower fetal radiation dose when compared to VQ scan.619 If is possible to modify the CT PE protocol to reduce radiation exposure and a survey of practicing radiologists found a preference for using CT PE with this lower radiation dose than using VQ scan.62
Once DVT or PE is diagnosed, treatment should be initiated with either unfractionated heparin (UFH) or low molecular weight heparin (LMWH). Neither UFH or LMWH cross the placenta and both can be used in nursing mothers. While either are effective in treating DVT and PE, LMWH are emerging as the preferred agent because of the ease of use, lack of requirement for laboratory monitoring, and suggestion of lower incidence of side effects compared to UFH. A twice-daily dosing regimen of either enoxaparin or dalteparin is recommended. The initial dose is weight adjusted (e.g., enoxaparin 1 mg/kg twice daily) but it is unclear how to adjust the dose for subsequent weight changes that occur as pregnancy progresses. Conversely, treatment with UFH can be initiated at 80 units/kg intravenous bolus followed by an intravenous infusion of 18 unit/kg/hour with adjustments in the infusion rate guided by aPTT testing.63 The risk of hemorrhage is approximately 2% but is more easily managed due to the short duration of action of UFH compared to LMWH.63
The duration of treatment for a DVT or PE that occurs during pregnancy is typically for the duration and then continues for a period of past after delivery. Because warfarin derivatives cross the placenta and cause both teratogenicity and bleeding in the fetus, pregnant patients with DVT or PE are managed with prolonged heparin therapy. Management of anticoagulation around the time of delivery is a complex task and requires careful monitoring and balancing of risks.
References
1. Hutchon DJ, Cooper S. Terminology for early pregnancy loss must be changed. BMJ 1998;317:1081.
2. Goddijn M, Leschot NJ. Genetic aspects of miscarriage. Baillieres Best Pract Res Clin Obstet Gynaecol 2000;14:855-865.
3. Cunningham FG, et al, eds. Williams Obstetrics, 22nd ed. Norwalk, CT: Appleton & Lange; 2005. Chapter 9: Abortion.
4 Velez LI, Benitez F, Lowry JC, et al. Right lower quadrant pain in females. Emerg Med Rep 2007;28:165-180.
5. Houry DE, Abbott JT. Acute Complications of Pregnancy–Miscarriage. In: Marx JA, et al, eds. Rosen's Emergency Medicine: Concepts and Clinical Practice. 6th ed. Philadelphia, Mosby Elsevier; 2006:Chapter 177.
6. Blohm F, Friden B, Platz-Christensen JJ, et al. Expectant management of first-trimester miscarriage in clinical practice. Acta Obstet Gynecol Scand 2003;82:654-658.
7. Pridjian G, Moawad AH. Missed abortion: Still appropriate terminology? Am J Obstet Gynecol 1989;161:261.
8. Creinin MD, Schwartz JL, Guido RS, et al. Early pregnancy failure-current management concepts. Obstet Gynecol Surv 2001;56: 105-113.
9. Falco P, Milano V, Pilu G, et al. Sonography of pregnancies with first-trimester bleeding and a viable embryo: A study of prognostic indicators by logistic regression analysis. Ultrasound Obstet Gynecol 1996;7:165-169.
10. Seeber BE, Barnhart KT. Suspected ectopic pregnancy. Obstet Gynecol 2006;107(2 Pt 1):399-413.
11. Mukul LV, Teal SB. Current management of ectopic pregnancy. Obstet Gynecol Clin North Am 2007 Sep;34:403-419.
12. Bradley WG, Fiske CE, Filly RA. The double sac sign of early intrauterine pregnany: use in exclusion of ectopic pregnancy. Radiology 1982;143:223-226.
13. Ahmed AA, Tom BD, Calabrese P. Ectopic pregnancy diagnosis and the pseudo-sac. Fertil Steril 2004;81:1225-1228.
14. Stovall TG, Kellerman AL, Ling FW, et al. Emergency department diagnosis of ectopic pregnancy. Ann Emerg Med 1990;19: 1098-1103.
15. Wong SF, Lam MH, Ho LC. Transvaginal sonography in the detection of retained products of conception after first-trimester spontaneous abortion. J Clin Ultrasound 2002;30:428-432.
16. Ness E. Credentialing and Privileging in Emergency Ultrasound. ACEP Practices Resources in Ultrasound. www.acep.org. Accessed January 2008.
17. Hertzberg BS, Bowie JD. Ultrasound of the postpartum uterus. Prediction of retained placental tissue. J Ultrasound Med 1991;10: 451-456.
18. Mulic-Lutvica A, Axelsson O. Ultrasound finding of an echogenic mass in women with secondary postpartum hemorrhage is associated with retained placental tissue. Ultrasound Obstet Gynecol 2006; 28:312-319.
19. Rulin MC, Bonrstein SG, Campbell JD. The reliability of ultrasonography in the management of spontaneous abortion, clinically thought to be complete: A prospective study. Am J Obstet Gynecol 1993;168:12-15.
20. Sairam S, Khare M, Michailidis G, et al. The role of ultrasound in the expectant management of early pregnancy loss. Ultrasound Obstet Gynecol 2001;17:506-509.
21. Gronlund L, Gronlund AL, Clevin L, et al. Spontaneous abortion: Expectant management, medical treatment or surgical evacuation. Acta Obstet Gynecol Scand 2002;81:781-782.
22. Luise C, Jermy K, Collons WP, et al. Expectant management of incomplete, spontaneous first-trimester miscarriage: Outcome according to initial ultrasound criteria and value of follow-up visits. Ultrasound Obstet Gynecol 2002;19:580-582.
23. Pang MW, Lee TS, Chung TK. Incomplete miscarriage: A randomized controlled trial comparing oral with vaginal misoprostol for medical evacuation. Hum Reprod 2001;16:2283-2287.
24. Wells P, Hirsh J, Anderson D, et al. A simple clinical model for the diagnosis of deep-vein thrombosis combined with impedance plethysmography: Potential for an improvement in the diagnostic process. J Intern Med 1998;243:15-23.
25. Coppola PT, Coppola M. Vaginal bleeding in the first 20 weeks of pregnancy. Emerg Med Clin North Am 2003;21:667-677.
26. Griebel CP, Halvorsen J, Golemon TB, et al. Management of spontaneous abortion. Amer Fam Phys 2005;72:1243-1250.
27. Scroggins KM, Smucker WD, Krishen AE. Spontaneous pregnancy loss: Evaluation, management, and follow-up counseling. Prim Care 2000;27:153-167.
28. Varras M, Akrivis C, Hadjopoulos G, et al. Heterotopic pregnancy in a natural conception cycle presenting with tubal rupture: A case report and review of the literature. Eur J Obstet Gynecol Reprod Biol 2003;106:79-82.
29. Habana A, Dokras A, Giraldo JL, et al. Cornual heterotopic pregnancy: Contemporary management options. Am J Obstet Gynecol 2000;182:1264-1270.
30. Della-Giustina D, Denny M. Ectopic pregnancy. Emerg Med Clin North Am 2003;21:565-584.
31. Zane SB, Kieie BA Jr, Kendrick JS, et al. Surveillance in a time of changing health care practices: Estimating ectopic pregnancy incidence in the United States. Matern Child Health J 2002;6:227-236.
32. American College of Emergency Physicians Clinical Policies Subcommittee on early pregnancy: Clinical policy: Critical issues in the initial evaluation and management of patients presenting to the emergency department in early pregnancy. Ann Emerg Med 2003; 41:123-133.
33. Lazarus E. What's new in first trimester ultrasound. Radiol Clin North Am 2003;41:663-679.
34. Goldner TE, Lawson HW, Xia Z, et al. Surveillance for ectopic pregnancyUnited States, 1970-1989. MMWR CDC Surveill Summ 1993;42:73-85.
35. Centers for Disease Control and Prevention. Current trends ectopic pregnancyUnited States, 1990-1992. MMWR Morb Mortal Wkly Rep 1995;44:46-48.
36. Fylstra DL. Tubal pregnancy: A review of current diagnosis and treatment. Obstet Gynecol Surv 1998;53:320-328.
37. Barnhart K, Esposito M, Coutifaris C. An update on the medical treatment of ectopic pregnancy. Obstet Gynecol Clin North Am 2000;27:653-667.
38. Shih C. Effect of emergency physician-performed pelvic sonography on length of stay in the emergency department. Ann Emerg Med 1997;29:348-352.
39. Burgher SW, Tandy TK, Dawdy MR. Transvaginal ultrasonography by emergency physicians decreases patient time in the emergency department. Acad Emerg Med 1998;5:802-807.
40. Choi H, Blavias M, Lambert MJ. Gestational outcome in patients with first-trimester pregnancy complications and ultrasound-confirmed live intrauterine pregnancy. Acad Emerg Med 2000;7:200-203.
41. Adhikari S, Blaivas M, Lyon M. Diagnosis and management of ectopic pregnancy using bedside transvaginal ultrasonography in the ED: A 2-year experience. Am J Emerg Med 2007;25:591-596.
42. Flanders PC, Tayal V. Outcome analysis of an emergency physician pelvic ultrasound protocol to rule out ectopic pregnancy: A one-year review at an urban emergency department. Ultrasound Med Biol 2003;29:S188.
43. Goldstein DP, Berkowitz RS. Gestational trophoblastic disease. In: Abeloff MD, et al, eds. Clincal Oncology. Philadelphia, Pa: Elsevier, 2004.
44. Berkowitz RS, Goldstein DP. Gestational trophoblastic diseases. In: Kufe DW, et al, eds. Cancer Medicine. 6th ed. Hamilton, Ontario: BC Decker; 2003:1863-1869.
45. Muggia FM, Burke TW, Small W. Gestational trophoblastic disease. In: DeVita VT, et al, eds. Cancer: Principles and Practice of Oncology. Philadelphia, Pa: Lippincott Williams & Wilkins; 2005:1360-1363.
46. American Cancer Society web site; Gestational Trophoblastic Disease. Available at: http://www.cancer.org/docroot/ CRI/CRI_2_3x.asp?rnav=cridg&dt=49. Revised 5/16/2006. Accessed November 2007.
47. Copeland LJ, Landon MB. Malignant Diseases and Pregnancy. In:Gabbe SG, et al, eds. Obstetrics: Normal and Problem Pregnancies, 5th ed. Philadelphia, Churchhill Livingstone Elsevier 2007. Chapter 45.
48. Mungan T, Kusçu E, Dabakoglu T, et al. Hydatidiform mole: Clinical analysis of 310 patients. Int J Obstet Gynecol 1996;52:233.
49. Moise KJ. Management of rhesus alloimmunization in pregnancy. Obstet Gynecol 2002;100:600.
50. Balderston KD, Towers CV, Rumney PJ, et al. Is the incidence of fetal-to-maternal hemorrhage increased in patients with third-trimester bleeding? Am J Obstet Gynecol 2003;188:1615.
51. Hannafin B, Lovecchio F, Blackburn P. Do Rh-negative women with first trimester spontaneous abortions need Rh immune globulin? Am J Emerg Med 2006;24:487-489.
52. Jabara S, Barnhart KT. Is Rh immune globulin needed in early first-trimester abortion? A review. Am J Obstet Gynecol 2003;188:623-627.
53. Rosenberg VA, Lockwood CJ. Thromboembolism in pregnancy. Obstet Gynecol Clin North Am 2007;34:481-500.
54. Morse M. Establishing a normal range for D-dimer levels through pregnancy to aid in the diagnosis of pulmonary embolism and deep vein thrombosis. J Thromb Haemost 2004;2:1202-1204.
55. Greer IA. Prevention and management of venous thromboembolism in pregnancy. Clin Chest Med 2003;24:123-137.
56. Wells P, Anderson D, Rodger M, et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med 2003; 349:1227-1235.
57. Heim S, Schectman J, Siadaty M, et al. D-dimer testing for deep venous thrombosis: A metaanalysis. Clin Chem 2004;50:1136-1147.
58. Chan WS, Chunilal S, Lee A, et al. A red blood cell agglutination D-dimer test to exclude deep venous thrombosis in pregnancy. Ann Intern Med 2007;147:165-170.
59. Stein PD, Beemath A, Matta F, et al. Clinical characteristics of patients with acute pulmonary embolism: Data from PIOPED II. Am J Med 2007;120:871-879.
60. Martin S, Foley M. Intensive care in obstetrics: An evidence-based review. Am J Obstet Gynecol 2006;195:673-689.
61. Winer-Muram HT, Boone JM, Brown HL, et al. Pulmonary embolism in pregnant patients: Fetal radiation dose with helical CT. Radiology 2002;224:487-492.
62. Schuster ME , Fishman JE, Copeland JF, et al. Pulmonary embolism in pregnant patients: A survey of practices and policies for CT pulmonary angiography. AJR Am J Roentgenol 2003;181: 1495-1498.
63. Bates SM, Greer IA, Hirsh J, et al. Use of antithrombotic agents during pregnancy: The Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004;126(3 Suppl): 627S-644S.
These next two issues of Emergency Medicine Reports will cover many of the complications and problems that may cause the pregnant woman to come see you. Part I will focus on miscarriage, ectopic pregnancy, gestational trophoblastic disease, and venous thromboembolic states. Part II will discuss hypertensive disorders, amniotic fluid embolism, and late pregnancy bleeding.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
