Management of Constipation in Adults
Author: Louis Kuritzky, MD, Clinical Assistant Professor, Department of Community and Family Medicine, University of Florida College of Medicine, Gainesville.
Peer Reviewer: Joseph Varon, MD, FACP, FCCP, FCCM, Professor, The University of Texas Health Science Center, St. Luke’s Episcopal Hospital, Houston, TX.
Clinicians should become confident in management of constipation for numerous reasons, not the least of which is the sheer magnitude of its prevalence in the ambulatory setting. Although the morbidity and mortality associated with other commonplace maladies like hypertension, heart disease, and diabetes typically command strong clinician focus, certainly patient preferences and priorities should help to guide the agenda of the clinical encounter. Hence, it is easy to envision that when addressed with a diversity of patient complaints that might include hyperlipidemia, hypertension, obesity, and constipation, it is indeed the latter that may be most compelling for the patient who is seeking relief. In contrast to other commonplace medical disorders in which clinicians possess a great deal of sophistication both for diagnosis and pathophysiologically targeted treatment (e.g., heart failure, diabetes), constipation management often is characterized by general, nonspecific techniques that lack the same intellectually sound underpinnings as would be applicable in management of most other clinical disorders. There appears to be a clinico-scientific discordance in regard to constipation, in that clinicians do not approach this commonplace and burdensome malady with the same scientific evidence base that is accorded essentially all other equally consequential health issues. This discordance results in management of constipation being fairly rudimentary, at best, much of the time. —The Editor
Constipation: Why Bother to Treat?
A complaint of constipation is responsible for as many as 2.5 million office visits annually in the United States1 and, fortunately, serious underlying pathology overall is quite uncommon. Because the etiologies of constipation most often are not serious, some clinicians have drawn the conclusion that constipation itself is not serious.2 Public expenditures on this malady, however, belie that assessment: Laxative sales amount to more than $500 million annually.1 Since most of the patient care for persons suffering constipation is provided by non-gastroenterologists,3 enhanced primary care skills are essential to better resolution of this important symptom.
Epidemiology
As much as 10% of the general U.S. population is affected by constipation, which is more prevalent in the elderly (25%).4 A recent systematic review of published literature has suggested that the prevalence of constipation approximates 12-19% in North American adults,5 although prevalence estimates range from 2-28%.6 Persons with diabetes are disproportionately represented, with as many as 60% reporting constipation.6 Similarly, almost half of patients with multiple sclerosis suffer consti-pation.7 A 1995 study examined the prevalence of constipation in persons older than 65 years in Olmstead County, Minnesota (n = 328); constipation was defined as one or more of the following: 1) straining; 2) hard stools; or 3) fewer than three bowel movements per week, all of which had to occur at least 25% of the time.8 In this study, 23% of respondents fulfilled the criteria for constipation; indeed, of those with constipation, 24% reported the need for digital self-facilitation of stool. Irrespective of age, the prevalence of constipation is inversely related to attained educational level.8
Definition
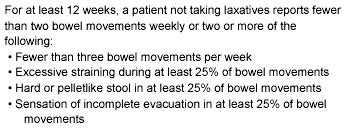
Though no consistent definition of constipation has held sway across the literature,2 patients and clinicians alike generally hold a similar impression of which symptoms and signs constitute constipation. Large population studies confirm that an adequate Western diet is associated with three or more bowel movements weekly, leading to the definition that two or fewer bowel movements weekly is consistent with a diagnosis of constipation.2 On the other hand, individuals who have a long-term consistent bowel pattern of fewer than three bowel movements weekly, but are not troubled by this frequency, and do not suffer difficulty in passage of stool, need not be labeled with the diagnosis.1 Hence, even a single weekly bowel movement need not be considered as constipation if not associated with adverse symptoms such as pain or bloating.1 The Rome Criteria (see Table 1) for constipation suggest that persons who have experienced for at least 12 months fewer than two bowel movements weekly OR two or more of the following: 1) fewer than three bowel movements per week; 2) excessive straining during at least 25% of bowel movements; 3) hard or pelletlike stool in at least 25% of bowel movements; or 4) sensation of incomplete evacuation in at least 25% of bowel movements fulfill the criteria for constipation.9 The author employs the following definition of constipation for clinical utility: "Any noteworthy departure from an established bowel pattern characterized by a persistent decrease in stool frequency, increase in stool hardness, or increase in straining at stool."
Table 1. Chronic Constipation: Rome Criteria9
Additionally, in this communication, the term functional constipation indicates constipation in the absence of demonstrable pathology. That functional constipation might respond to fiber supplementation, fluid enhancement, or activity augmentation need not imply that any of these modalities were necessarily deficient at baseline, but rather, that augmentation of any of these may have a positive impact upon efficiency of stool delivery.
Etiology
Most forms of constipation are idiopathic, and whatever underlying derangement is responsible, are not associated with long-term health detriment, notwithstanding the important effects upon quality of life sustained by sufferers.2 Of course, since constipation may be associated with important underlying disorders, including colon cancer, clinicians should be confident that no secondary etiology is present before embarking upon a therapeutic course.
Pathophysiology
Constipation is characterized under the pathophysiologic categories of delayed colonic transit (also called colonic inertia) or pelvic outlet delay.1 Approximately 1000 mL of fluid is presented to the colon daily. Active absorption is responsible for sodium absorption, which results in an electrical gradient that causes chloride ions to follow. The combined NaCl flow elicits an osmotic effect, which leads ultimately to absorption of up to 900 mL of fluid.10 (See Figure 1.)
When delayed colonic transit causes constipation, it may be subdivided categorically into causes that are idiopathic, medication-induced, and/or secondary to an underlying medical disorder.1 Commonly, constipation is associated with low or inadequate fiber in the diet. Although the mechanisms are ill defined, irritable bowel syndrome also may cause constipation. Among the well-recognized adverse effects of opioid analgesics, constipation is the only adverse effect to which tolerance does not appear to develop.
When colonic emptying is slowed, it follows that a greater degree of fluid absorption from the stool will occur, resulting in a harder, less moist stool. Delay in transit results in a decrease in the delivery of stool to the rectum, with a decline in rectal stool volume. This reduced stool volume may be insufficient to trigger the rectal evacuation mechanism, which results in still further delay in stooling and additional subsequent stool dehydration.
Anismus (also known as defecatory dyssynergy) is a less common etiology of chronic constipation. Failure to coordinate pelvic floor muscular contraction with anal sphincter activity is fundamental to the disorder.11 Under conditions of normal defecatory physiology, when rectal pressure increases to induce evacuation, anal sphincter relaxation should follow. In anismus, when studied with anal manometry, one can see aberrant patterns such as increased rectal pressure combined with a paradoxic increase in anal sphincter pressure; no increase in rectal pressure in combination with a paradoxic anal contraction; or an increase in rectal pressure accompanied by incomplete or absent anal sphincter relaxation.11 Management of dyssynergic defecation, which requires specialized testing for confirmation and subsequent treatment, is beyond the scope of this article.
Differential Diagnosis
Most constipation in adults is functional constipation. That is, there is no discernible secondary disorder responsible for the observed bowel function change. Because a diversity of underlying pathologies may produce constipation, and especially because the initial presentation of colon or rectal cancer may be a change in bowel function, clinicians must maintain a high level of vigilance for secondary causes prior to initiation of treatment, and especially in cases that do not promptly (i.e., within 1-2 weeks) respond to traditional therapeutic measures.
Though rare in the United States, worldwide the most common infectious cause of constipation is Chagas disease.12 In this disorder, Trypanozoma cruzi organisms penetrate the gastrointestinal (GI) muscular coat and result in destruction of the myenteric plexus of Auerbach. Ganglion cells are destroyed and are replaced with fibrosis, and constipation results from colonic atony. Although Chagas disease is very infrequent in the United States, this must be considered in the differential diagnosis of persons from geographically pertinent areas (most commonly South America).
The differential diagnosis of constipation is potentially extensive. (See Table 2.)
Table 2. Constipation Differential Diagnosis2

In the typical primary care setting, the disorders most commonly associated with constipation are diabetes, hypothyroidism, and adverse effects of medications. In diabetes, autonomic neuropathy of the colon is felt to be etiologic in constipation.13 Constipation may be the first indicator of hypothyroidism, but rarely presents in isolation from other more commonplace signs of hypothyroidism, such as fatigue, cold intolerance, and changes in hair and skin.13 Although opioids are the agents that routinely are expected to have the adverse effect of constipation (and this appears to be a class effect), the calcium channel blocker verapamil also may cause constipation (incidence 7.5%).13 All medications with anticholinergic effects may induce constipation, such as antihistamines, antidepressants, and medications for urinary incontinence like tolteridine (Detrol), oxybutynin (Ditropan, Oxytrol), trospium (Sanctura), and solifenacin (Vesicare). The anticholinergic effects of antihistamines generally are absent in latest-generation agents, such as loratadine (Claritin), desloratadine (Clarinex), and fexofenadine (Allegra). Other medications that commonly may produce constipation are listed in the Table 3. Concerningly, chronic use of stimulant laxatives, in particular anthraquinone or bisacodyl based treatments, ultimately may result in constipation.13
Table 3. Constipation-Inducing Medications

Fortunately, most systemic disorders that are associated with constipation also induce organ changes outside the GI system, and hence will be discovered by their extra-gastrointestinal symptoms.1 In other words, although numerous systemic disorders include constipation as part of their clinical constellation, it is uncommon for constipation to be the sole presenting complaint.
Diagnosis
Expert consensus generally advises that persons with new onset constipation receive an appropriate history, physical examination, and colonic evaluation, to include barium enema or colonoscopy, with consideration of colonic transit and anorectal function to follow if symptoms are refractory.1,14
Despite the consistent recommendation by experts that physical examination be performed, it rarely is helpful unless one detects an abdominal mass.1 Investigation of colonic integrity following a complaint of new onset constipation in an adult, in the absence of an obvious precipitating factor (such as opioid or anticholinergic), is similar to that for onset of rectal bleeding: Full colonic evaluation (e.g., colonoscopy) is necessary to rule out neoplasm or other obstructing lesion. Barium enema is an alternative investigation, and similarly might show obstructing neoplasm, strictures, Hirschsprung’s megacolon, spasm indicative of laxative abuse, or absent haustral markings as seen in chronic laxative use or atonic megacolon.1 Hence, it is advisable to suggest to all adult patients with unprovoked new onset constipation that they undergo colonic evaluation. In the absence of signs of intestinal obstruction, provisional therapeutic management may be instituted pending full definition of colonic integrity. On the other hand, most adults can identify a change in activity, hydration, diet, medication, or some other factor to which alterations in bowel function readily are attributable. In circumstances where an appropriate explanation for constipation is discerned, no further evaluation is necessary, and treatment may be begun expeditiously. The initial evaluation should include a targeted history to rule out secondary causes. The physical examination should include a rectal examination to rule out anal or rectal cancer and fissure, and a demonstration of the neurologic integrity of the perineum.
If constipation symptoms persist despite normal initial studies, measurement of colonic transit may be appropriate. Commonly, a three-day colonic transit study with radioopaque markers is performed; if markers remain intracolonic for more than 70 hours, the result is indicative of colonic inertia.2
In contrast to algorithms for disorders like gastroesophageal reflux disease (GERD), in which alarm symptoms are well defined, less literature guidance is available for constipation. It is suggested that any of the following symptoms should function as alarm symptoms, indicating that even with successful constipation resolution, evaluation for underlying serious pathology must be done: rectal bleeding, weight loss, fever, palpable abdominal mass, signs of obstruction, or a sustained, unprecipitated departure from the typical bowel pattern consisting of either a reduced bowel frequency, stool hardness, stool caliber, or facility in passing stool. (See Table 4.)
Table 4. Constipation Alarm Symptoms
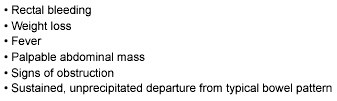
Radiographic Studies
Unless used to seek serious secondary pathology, radiography rarely is needed for management of functional constipation. However, when clarification of colonic transit is desired, probably the most commonly utilized test is demonstration of colonic motility by means of ingesting radiopaque markers. Radiopaque marker testing might be considered for patients with severe or refractory constipation. One type of colonic transit testing is performed by having the patient ingest a single capsule that contains 24 radiopaque polyvinyl chloride O-rings sized 1 mm x 4.5 mm (this capsule size is swallowed readily, about the size of typical antihistamine cough-cold remedy capsules). The patient is advised not to utilize any laxatives, enemas, or suppositories for the next five days. On day five after swallowing the capsule, an abdominal flat plate x-ray is obtained. Interpretation of results is as follows:
a) At least 80% of markers expelled = normal colonic transit;
b) Less than 80% expulsion, with markers distributed around the colon = colonic hypomotility (colonic inertia);
c) Retained markers collected around rectum or rectosigmoid = functional outlet delay (e.g., anismus).
The manufacturer suggests that if six or more markers are retained at the five-day interval, the patient should use a bulk-forming laxative daily for 7-14 days, followed by ingestion of another capsule to confirm the status of colonic function.
Treatment
A small portion of persons with constipation respond to simple reassurance and education, in that some patients are concerned with bowel movement infrequency that does not meet the definition of constipation. Some individuals will respond to improved measures of stool hygiene: ensuring adequate exercise, appropriate evacuation posture, a dedicated time for evacuation, and avoidance of chronic use of stimulant laxatives.15 These steps probably should be suggested for all persons who suffer functional constipation. If a secondary etiology is present, that should be addressed to see if the etiology can be resolved, i.e., correction of hypothyroidism may restore normal bowel function. Since restoration of euthyroidism may take weeks or even months, it is still appropriate to provide laxatives or cathartics until hypothyroidism is controlled.
Sometimes, a secondary etiology of constipation cannot be resolved. For instance, diabetic autonomic colonic neuropathy may result in constipation, but better control of diabetes has not been demonstrated to favorably alter GI motility in persons with established neuropathy. Nonetheless, identification of the likely underlying etiology may assist in therapeutic choices and motivate better management of the secondary disorder.
It is suggested that for persons with chronic constipation, behavioral treatment should include setting aside a preferred time for evacuation, preferably 5-10 minutes postprandial, to take advantage of the gastrocolic reflex.1 Establishing a consistent bowel regimen initially may require an induced timed stool, as with a lukewarm tap-water enema or suppository (e.g., bisacodyl).1
Expert opinion often recommends increases in fiber and hydration as foundation treatment for constipation. In the author’s experience, this rarely is fruitful because the lay public generally already is aware of the role of fiber in bowel function. Additionally, it is difficult for the clinician to make an accurate appraisal of baseline or post-intervention fiber consumption so that, at best, fiber supplementation often is a fairly gross maneuver, rather than well titrated. Similarly, although physicians commonly suggest enhanced hydration, especially if patients complain of hard, dry stools, it is distinctly uncommon that a clear demonstration of any degree of dehydration or decreased intravascular volume is made.
Nonetheless, tradition holds that ensuring adequacy of fiber and fluid intake is a reasonable initial step. The state of hydration could be assessed simply by a urine specific gravity, aiming for 1.015 or less. For persons whose urine specific gravity is greater than 1.015, suggest progressive increases in fluid, but be specific: Since fluid intake should be at least 1200 mL daily (40 ounces), begin with an intake of at least 48 ounces daily, which is a convenient amount for patients to measure. Patients whose stools remain dry at this level of hydration should increase fluid intake until stools are more moist, or until urine specific gravity is less than 1.010. Choice of fluid does not appear critical, though the impact of some fluids may be enhanced by choosing fruit juices (especially prune juice) with inherent laxative effect, osmotically active sugars, and fiber content.
Modulating fiber intake is a more complex issue. Suggesting fiber supplements generally will add bulk and liquid to the stool, but many individuals are intolerant of the taste of such supplements, or suffer a sensation of bloating or gaseousness when ingesting sufficient amounts of fiber to favorably affect constipation. Bulk-forming laxatives generally are composed of natural polysaccharides, synthetic polysaccharides, or cellulose derivatives. Increased fiber in the diet, whether dietary or supplementary, adds to stool volume and helps to adsorb water, increasing overall water content and bulk of the stool. Additionally, metabolism of fiber by colonic flora results in increased numbers of osmotically active metabolites.1 Even though fiber interventions are viewed as benign, they should not be used in the presence of colonic stricture or atonic colon, in that they may result in colonic obstruction. Overall, bulk-forming laxatives increase bowel movement frequency by a mean of 1.4 stools per week.1 Despite the variety of formats intended to enhance palatability (e.g, powder, biscuit, tablet), GI adverse effects often limit acceptance. Patients should be reassured that GI adverse effects of fiber generally decline with continued product use. Rather than suggesting a dramatic dietary change in fiber content (dietary or bulk-forming fiber laxatives), it is preferable to slowly advance fiber content, since gaseousness, bloating, and discomfort may occur during too-rapid dose escalation. Additionally, as fiber intake increases, so must fluid intake, lest a paradoxical worsening of constipation occur due to inadequate fluid supply. Clinicians should recommend a minimum of 2 liters per day of fluid intake to maintain adequate stool hydration during periods of increased fiber intake.15 A diversity of fiber supplements is available, with no clear demonstration of superiority of one agent over another for efficacy or safety. Hence, choice of fiber depends on clinician and patient preference, cost, palatability, and ease of administration. Sugar-Free Metamucil contains phenylalanine, which is contraindicated in phenylketonuria patients.
The role of dietary fiber in stool regulation has been a matter of substantial inquiry epidemiologically. Examination of stool weight among 1000 individuals of diverse ethnicities indicates marked variation.16 For instance, the aforementioned study by Burkitt found an average stool weight of 470 g among Ugandans, vs. 173 g in participants from the United Kingdom. In the Ugandan population at the time this study was performed (1972), in the absence of a Westernized diet (i.e., Ugandans had a high dietary fiber intake), constipation was in essence unheard of, and colonic transit times were inversely related to stool weight. Ethnic differences disappeared in Africans who adopted Western dietary patterns.15 Information such as this has formed the conceptual framework for the beneficial effects of high-fiber diets on stool evacuation function.
Dietary fiber may function in several ways to enhance stool evacuation. As mentioned earlier, the more rapid transit time of larger stool volume, as provided by fiber, provides more rapid attainment of critical mass to simulate ano-rectal responses to the presence of stool. Decreased colonic transit time also allows less time for fluid absorption, resulting in more retained stool moisture. Recommendations for the optimum amount of dietary fiber for Americans vary from 14.4 g/day to as high as 20-35 g/day.15 Traditionally, the primary categorizations of fiber are water soluble fiber and insoluble fiber. Water-soluble fiber (e.g., mucilages and pectin) has multiple GI effects, including formation of a gel in the stomach that promotes gastric distention and delays food absorption. Water-soluble fiber also has a favorable effect on lipid absorption, including cholesterol. Examples of water soluble fiber include apples, barley, beans, grapefruit, oats, peas, and potatoes. Insoluble fiber works primarily by absorbing water to enhance formation of soft, bulky stools, but has less impact on lipids. Typical examples of insoluble fiber include corn, fruits, nuts, and vegetables.
Functional constipation generally is treated with either laxatives or cathartics. The category laxative indicates agents of slower onset of action, typically 1-3 days, with a lesser degree of bowel evacuation; on the other hand, cathartics typically act within 6-12 hours and provide a greater degree of bowel evacuation.17
After appropriate education and stool hygiene management, it is appropriate to utilize some laxative medication, though there is no clear guidance for which laxative might be the intervention of choice. Interventions may include stool softeners, hyperosmolar agents, glycerin suppositories, enemas, and intermittent sparing use of stimulant laxatives.15
Laxative mechanisms of action often are poorly understood, and the potential for toxicity is underestimated.1 In contrast to other commonplace disorders in primary care, there are few head-to-head comparative data, and therapeutic choice most often is related to clinician or patient personal preference, rather than objective efficacy data.1 Similarly, product format, availability, cost, and ease of administration may be primary factors in medication choice. Since there are more than 700 products for constipation available in the United States alone,2 the clinician and the patient face a dizzying array of choices. Although any of the categories of laxatives is a potential reasonable first choice, probably the single agent that best combines efficacy, safety, tolerability, flexible dosing, and prompt response is polyethylene glycol (Miralax). Other agents will be discussed in detail prior to this category.
Mineral oil and docusate sodium (e.g., Colace) are two of the prototypes of emollient laxatives. The mechanical lubricating capacity of mineral oil is evident, but chronic use can result in reduced absorption of vitamins A, D, E, and K. It should be administered with caution to anyone with compromised swallowing capacity because of reports of potential for aspiration resulting in lipid pneumonia.2 Docusate sodium is reported to function by decreasing stool liquid surface tension, allowing for an increase in the aqueous-fat admixture with subsequent increase of water penetration into the stool; additionally, it increases colonic fluid and electrolyte secretion.2
The category of saline laxatives is so named because the active agents are all salts of cations, most commonly sodium and magnesium. Probably the two most familiar saline laxatives are Milk of Magnesia and Fleets Phosphosoda. Saline laxatives are poorly absorbed, resulting in an intracolonic hypertonic solution that draws water into the colonic lumen; saline laxatives also may stimulate cholecystokinin release.1 Although generally safe medications, they can have adverse events, including hypermagnesemia, hypocalcemia, hyperphosphatemia, and sodium overload, especially pertinent in the elderly or persons with tenuous fluid balance status. These electrolyte disarray consequences preclude use in patients with heart failure or compromised renal function.
Osmotic laxatives include unmetabolizable semisynthetic sugars (e.g., lactulose, sorbitol) and polyethylene glycol, a synthetic polyglycol. The mechanism of action is osmotic enhancement of fluid into the colon by non-digestible substances that are recovered unaltered in the stool. These agents may be used chronically. Polyethylene glycol generally is administered in an initial dose of 17 grams in 8 ounces of any liquid, anticipating a change in stool frequency and stool hydration within 48 hours. The dispensing canister of Miralax contains a marker in the cap to easily measure the 17 g dose. Subsequent dosing can be adjusted to provide a suitable stool frequency and malleability. As with any agent altering intestinal motility or colonic fluid transport, chronic use can result in electrolyte changes, so periodic electrolyte monitoring should be considered.18 Polyethylene glycol is tasteless, odorless, and dissolves fully into solution in the patient’s preferred beverage.
Lactulose (Kristalose) is a non-digestible disaccharide that results in osmotic fluid shifts. Additionally, bacterial action on lactulose breaks down the agent into lactic acid, formic acid, and acetic acid, acidifying the colonic contents, which also increases stool water content. Because lactulose includes a small amount of galactose, this product should not be administered to people who require a low galactose diet. Lactulose is administered as 10 g packets that should be dissolved in 4 ounces of water. Similar considerations for electrolyte monitoring as for polyethylene glycol are appropriate.19
The stimulant laxatives include cascara sagrada (peri-Colace), bisacodyl (Dulcolax), and senna (Senokot, ExLax). Their mechanism of action is primarily by means of altering mucosal electrolyte transport, which increases colonic motor activity. Traditional wisdom has suggested that chronic use might eventuate into dependency, but this is weakly established in the literature, and the possibility that some persons use progressively more stimulant laxative because of progressive pathology, rather than agent dependence, has never been ruled out.
Another agent that has shown promise in constipation refractory to initial treatments is tegaserod (Zelnorm). This agent is a partial agonist of the 5-HT4 receptor, and at a dose of 6 mg before breakfast and dinner, stimulates peristalsis.20 Tegaserod was approved for the management of chronic constipation in 2004. The putative mechanisms of action include: 1) activation of the enteric serotonin subtype 4 receptor, which enhances the peristaltic reflex in response to endogenous mucosal stimulation;21 and 2) increases in intestinal secretory responsivity.22 A recent randomized, double-blind placebo-controlled trial of tegaserod in patients with chronic constipation (n = 1,348) compared two dose strengths: 2 mg bid and 6 mg bid.21 Patients were long-term sufferers of constipation (mean = 19 years). Both doses of tegaserod were statistically more effective than placebo in producing at least one more complete spontaneous bowel movement weekly during the first four weeks of the 12 week trial (the primary efficacy endpoint). The responder rates for tegaserod 6 mg and 2 mg were similar (44.8% and 40.3%, respectively). Tegaserod was well tolerated, with an overall adverse effect profile similar to placebo. Diarrhea was more common with tegaserod than placebo (4.5-7.3% vs 3.8%), but resolved quickly, most commonly within 48 hours. There were no cases of disordered electrolyte balance. Only the 6 mg dose twice daily before meals is FDA approved. Because ischemic colitis has been reported with 5-HT4 receptor agonists, if diarrhea occurs while on tegaserod that does not promptly remit, or if it is associated with severe abdominal pain, the drug immediately should be discontinued, and the possibility of ischemic colitis should be evaluated. Tegaserod is indicated only for short-term use (4-6 weeks, with consideration for a second 4-6 weeks period).23 Tegaserod likely will remain a second-line agent for treatment of chronic constipation due to its expense. Additionally, it is not recommended for people older than 65 years.23
Of all of the adverse effects of opioid analgesia, constipation is the only one to which tolerance does not appear to develop. With increasing awareness and facility of primary care clinicians in reference to chronic pain management, it is anticipated that a need for stool modulation pharmacotherapy is necessary. All of the previously mentioned classes of agents are appropriate remedies. Recently, methylnaltrexone has shown promise in clinical trial populations. In one double-blind, placebo controlled, randomized trial, patients (n = 22) with constipation on methadone maintenance were treated with parenteral methyl-naltrexone, resulting in prompt laxation and no significant adverse GI effects save mild cramping.24 A follow-up trial with oral methylnatltrexone also showed favorable dose-related responses within 18 hours of administration.25 Although methylnaltrexone is not yet approved for clinical use, it is attractive for chronic opioid therapy because it does not cross the blood-brain barrier (which would induce drug withdrawal) and has very low plasma levels, suggesting that (at least with oral administration) the primary efficacy is in the gut. Adverse effects of some medications that have diarrhea as an acknowledged adverse effect may be capitalized upon to relieve constipation. For instance, misoprostol has been studied in severe chronic constipation, showing an improvement of bowel movements from a baseline of 2.2 per week up to 6.2 per week at a dose of misoprostol 1200 mcg/d.26 Similarly, because anticholinergic activity is felt to be the primary mechanism of opioid-induced constipation, it would be putatively attractive to provide cholinergic stimulation. To that end, a study of 2% pilocarpine solution (5 mg tid) has been reported in a case report to relieve morphine-induced constipation as well as urinary retention.27
When to Refer
Although many people will have persistent and recurring constipation problems that require interventions for resolution, only a very few persons will merit referral for gastroenterologic consultation. Since it is recommended that all healthy people older than 50 years undergo screening colonoscopy as one method of colon cancer screening, it is not unreasonable for clinicians to encourage this intervention when patients present with functional constipation at or beyond age 50. Patients who present with alarm symptoms, constipation that does not resolve promptly with appropriate interventions, or anyone in whom the clinician suspects pertinent underlying pathology merits consideration for referral.
Summary (see figure)
Because no one pharmacotherapeutic agent is appropriate for every patient, clinicians will need to familiarize themselves with different classes of agents and different members of each class. In the absence of serious underlying pathology, initial treatment can be from any of the classes, though the simplicity and flexibility of polyethylene glycol makes it an appropriate initial choice in many patients. If osmotic laxatives, emollients, and bulk-forming agents have failed to resolve constipation, either consultation should be considered or a trial of saline or stimulant laxatives employed. Alarm signs for serious underlying pathology in patients with constipation include blood, obstructive symptoms, weight loss, fever, a palpable abdominal mass, and unprecipitated persistent stool atypia (caliber or frequency). Clinicians should be confident that most patients with constipation can be satisfactorily managed in the primary care setting.
References
1. Cheskin LJ. Constipation and Diarrhea. In: Barker LR, Burton HR, Zieve PD, eds. Principles of Ambulatory Medicine, 5th ed. Baltimore:Williams & Wilkins; 1999: 498-503.
2. Etzkorn KP, Rodriguez L. Constipation. Rakel, Bope, eds. Conn’s Current Therapy. Philadelphia: WB Saunders; 2002.
3. DiPalma JA, Schiller L, Wald A, et al. New Paradigm in the Treatment of Constipation. Proceedings from a Symposium 2001. Digestive Disease Week, Atlanta: Monograph courtesy Braintree Labs.
4. Fwong PWK, Kadakia S. How to deal with chronic constipation. Postgrad Med 1999;106:99-210.
5. Higgins PD, Johanson JF. Epidemiology of constipation in North America: A systematic review. Am J Gastroenterol 2004;99:750-759.
6. Talley NJ. Definitions, epidemiology, and impact of chronic constipation. Rev Gastroenterol Dis 2004;4(Suppl2):S3-S10.
7. Hinds JP, Eidelman BH, Wald A. Prevalence of bowel dysfunction in multiple sclerosis. A population survey. Gastroenterology 1990;98:1538-1542.
8. Ehrenpreis ED. Definitions and epidemiology of constipation. In: Wexner SD, Bartolo DCC, eds. Constipation Etiology, Evaluation & Management. Oxford: Butterworth-Heinemann Ltd; 1995:3-8.
9. Whitehead WE, Chaussade S, Corazziari E, et al. Report of an international workshop on management of constipation. Gastroenterol Intl 1991;4:99-113.
10. Guyton AC, Hall JE. Digestion and absorption in the GI tract. Textbook of Medical Physiology, 10th ed. Philadelphia: WB Saunders; 2000:754-763.
11. Jancin B. Biofeedback often effective in chronic constipation. (Report on a presentation by SSC Rao at the American College of Gastroenterology). Family Practice News 2001; p. 17.
12. Gemlo BT, Wong WD. Etiology of acquired colorectal disease. In: Wexner SD, Bartolo DCC, eds. Constipation Etiology Evaluation & Management. Oxford: Butterworth-Heinemann Ltd; 1995:3-8.
13. Castro DD, Cherry DA. Extracolonic causes of constipation. In: Wexner SD, Bartolo DCC, eds. Constipation Etiology Evaluation & Management. Oxford: Butterworth-Heinemann Ltd; 1995:23-30.
14. Hutchinson R, Kumar D. Colonic and small bowel transit studies. In: Wexner SD, Bartolo DCC, eds. Constipation Etiology Evaluation & Management. Oxford: Butterworth-Heinemann Ltd; 1995:52-63.
15. Jensen JE. Medical treatment of constipation. In: Wexner SD, Bartolo DCC, eds. Constipation Etiology Evaluation & Management. Oxford: Butterworth-Heinemann Ltd; 1995: 137-153.
16. Burkitt DP, Walker ARP, Painter NS. Effect of dietary fiber on stools and transit times and its role in the causation of disease. Lancet 1972;ii:1408-1412.
17. Locke GR 3rd, Pemberton JH, Phillips SE. American Gastroenterologic Association Technical Review on Constipation. Gastroenterology 2000;119:1766-1778.
18. Package Insert, Miralax, 2004 PDR.
19. Kristalose Package Insert, 2004 PDR.
20. Levinson MJ. Constipation. In: Rakel, Bope, eds. Conn’s Current Therapy. Philadelphia: WB Saunders; 2004: 24-26.
21. Johanson JF, Wald A, Tougas G, et al. Effect of tegaserod in chronic constipation: A randomized, double-blind, controlled trial. Clinical Gastroenterology and Hepatology 2004;2: 796-805.
22. Gershon MD. Serotonin and its implication for the management of irritable bowel syndrome. Rev Gastroenterol Disord 2003;3(Suppl 2):S25-S34.
23. Zelnorm prescribing information: Monthly Prescribing Reference 2004(October): page 185.
24. Yuan C, Foss JF, O’Connor M, Osinski J, et al. Methylnaltrexone for reversal of constipation due to chronic methadone use. JAMA 2000;283:367-372.
25. Yuan C, Foss JF. Oral Methylnaltrexone for opioid induced constipation. JAMA 2000;284:1383-1384.
26. Soffer EE. Misoprostol is effective treatment for patients with severe chronic constipation. Dig Dis Sci 1994;39: 929-933.
27. Mercadante S. Pilocarpine as an adjuvant to morphine therapy. Lancet 1998;351:338-339.
Clinicians should become confident in management of constipation for numerous reasons, not the least of which is the sheer magnitude of its prevalence in the ambulatory setting.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
