The Current Challenge of Venous Thromboembolism in the Hospitalized Patient: Optimizing Recognition, Evaluation, and Prophylaxis of At-Risk Patients - Part I
The Current Challenge of Venous Thromboembolism (VTE) in the Hospitalized Patient: Optimizing Recognition, Evaluation, and Prophylaxis of At-Risk Patients
Part I: Patient Identification, Risk Factor Assessment, and Diagnostic Strategies
Authors: Janet Poponick, MD, Attending Physician/Assistant Professor, MetroHealth Medical Center/Case Western Reserve University, Cleveland, OH; Gideon Bosker, MD, FACEP, Assistant Clinical Professor, Section of Emergency Services, Yale University School of Medicine; Associate Clinical Professor, Oregon Health Sciences University, Portland. Peer Reviewers: Kurt Kleinschmidt, MD, FACEP, Associate Professor, University of Texas Southwestern Medical Center, Dallas; Keith Wrenn, MD, FACP, FACEP, Vice Chairman, Emergency Medicine; Director, Emergency Medicine Residency Program, Vanderbilt University Medical Center, Nashville, TN.
Never before has there been a more heightened awareness of the problems associated with thrombosis prevention and management in the hospital environment. Although improvements have been made in the area of thrombosis management, there remains a widespread failure to screen, assess, diagnose, and/or prevent with pharmacologic intervention the growing number of patients at risk for venous thromboembolism (VTE). Put simply, VTE is a common disease that affects more than 2 million people each year, and may be responsible for up to 200,000 deaths annually. This condition is associated with a number of risk factors, including heart disease, respiratory failure, orthopedic surgery, malignancy, and other co-morbid conditions. With an incidence in the United States of 1 in 1000, VTE may present as deep venous thrombosis (DVT) or as pulmonary embolism (PE), which, according to some experts, is the most important preventable cause of hospital-associated death among medical inpatients.
Approximately 600,000 patients each year develop PE, and 60,000 deaths due to this disease are reported annually. Unfortunately, these statistics have not changed in three decades despite aggressive diagnostic evaluation, identification of risk factors (See Table 1), evidence-based treatment protocols, and improved prophylaxis, which remains under-utilized in hospitalized medical patients. Moreover, the prevalence of VTE actually may be on the rise due to the aging population and longer survival of cancer patients.
 |
The burden of VTE in hospitalized patients is especially significant, with certain medical conditions (e.g., congestive heart failure [CHF], respiratory failure, and systemic infection) being associated with an elevated risk for thromboembolic disease. DVT and PE are two manifestations of the same disease process, VTE. Of patients diagnosed with DVT, approximately 30% develop symptomatic PE. The survival rates for those with a diagnosis of VTE are poor. Moreover, the detection of PE frequently is elusive, with the diagnosis being made at autopsy in a large percentage of cases. More than one-third of all deaths from PE occur on the day of presentation. Consequently, emergency physicians and hospital-based practitioners must recognize patients who are at increased risk of VTE early in their presentations and initiate appropriate diagnostic evaluations and treatment plans.
From a clinical perspective, perhaps the most important issue is for hospital-based clinicians to recognize those patient subgroups that are at significant risk for acute DVT. Although numerous trials have identified specific medical disorders that, when they afflict acutely hospitalized, immobilized patients, increase the risk of acquiring acute DVT, there is no single patient profile that mandates medical prophylaxis against DVT. Rather, the clinician must weigh all of the relevant risk factors (e.g., respiratory status, cardiovascular function, patient age, etc.) and determine, based on clinical judgment, whether the risk of prophylaxis outweighs the risk of thrombosis.
With these clinical issues in clear focus, Part I of this two-part series will stress the importance of risk factor assessment and variable clinical presentations. A number of diagnostic modalities used for VTE diagnosis will be discussed, and the potential advantages and limitations of each will be outlined. Strategies for arriving at a pretest clinical probability based on history, physical examination, and risk factor assessment are emphasized, since establishing clinical probability of the disease is essential for guiding subsequent patient evaluation. Part II will address treatment and prevention.—The Editor
Introduction and Overview of the Problem
Autopsy studies confirm that clinicians simply are not doing enough to screen for or establish the diagnosis of VTE in medical and surgical patients. In one study, PE was thought to be the cause of death in 239 of 2388 autopsies performed (10%); 15% of these patients were younger than 60 years, and 68% did not have cancer.1 Of these patients, 83% had DVT in the legs at autopsy, of whom only 19% had symptoms of DVT before death. Only 3% of patients who had DVT at autopsy had undergone an investigation for such before death. Twenty-four percent of patients who died from PE had undergone surgery a mean of 6.9 days before. The investigators concluded that screening tests and/or risk stratification methodologies for DVT should be applied widely in the hospital population. In addition, this study demonstrated that PE is still a major cause of death in hospital patients despite advances in diagnosis and treatment, since a 10% incidence of hospital patients dying of PE is unacceptably high, inasmuch as PE is preventable and treatable.1
In another retrospective review, the incidence and patterns of fatal pulmonary embolism (FPE) observed in postoperative vs. non-postoperative patients was documented. A review of 14,667 necropsy reports for every year from 1965 to 1990 and 6,436 diagnostic venograms performed from 1976 to 1990 was undertaken at a single teaching hospital.2 A progressive reduction in the percentage of necropsies reporting FPE from 6.1 to 2.1% occurred during the 25-year period (chi2 tests for linear trend with time p < 0.00001). During the last decade studied (1980-1990), there was a significant reduction in the rate of venographically diagnosed postoperative DVT, from 49.9 to 24.7 per 100,000 population (p < 0.0001), which was in marked contrast to the constant rate of non-postoperative DVT. These findings suggested that the introduction of thromboprophylactic measures, in addition to changes in hospital practice, had a highly significant effect on the pattern of this serious but potentially avoidable disease.2
It was clear that within the accepted limitations of retrospective necroscopy and investigational studies, a highly statistically significant decrease in VTE was observed in the hospital population during a 25-year period. This was the good news. However, the authors also stressed that this decrease was most marked in VTE, both fatal pulmonary emboli and DVT, occurring postoperatively, and that there was only a relatively small decrease in FPE in medical patients. These results indicate that thromboprophylaxis, in its various forms, is effective in the hospital community in general. Moreover, a recommendation for the greater use of thromboprophylaxis in medically ill patients is supported by the data, which showed that the reduction in postoperative FPE has been more marked than non-postoperative FPE.2
VTE in Medical Patients. Hence, VTE remains a major cause of mortality and morbidity in hospitalized patients, despite the availability of effective prophylactic agents.3 Survival rates for patients with an established diagnosis of VTE are not encouraging, with only 64% surviving one year.4 Interestingly, studies demonstrate that the majority of patients who suffer an FPE have not undergone recent surgery, yet PE rarely is suspected as a cause of death in non-surgical patients and prophylaxis is used infrequently, despite consensus statement recommendations.5-8 The burden of VTE in non-surgical populations is significant, with certain medical conditions (e.g., CHF, respiratory failure, and systemic infection) associated with elevated risk for thromboembolic disease.8 Analysis of recent studies evaluating risk factors in individual patients will help clarify the need for and value of thromboprophylaxis in clearly defined groups of medical patients.
Compared with surgical populations, far fewer studies have reported the frequency of VTE in medical patients. However, those figures that are available suggest a moderate risk of DVT in general medical patients in the absence of prophylaxis, according to the risk categories (low, moderate, high) defined for surgical patients.9-11 It should be noted that much higher rates of VTE have been observed in specific groups which, accordingly, should be risk-stratified to receive thromboembolic prophylaxis when indicated.9,10,12-16 In this regard, a recent study reported that up to one in 20 hospitalized medical patients with multiple problems and severe immobility may suffer a fatal PE.17 It should be noted, however, that current management practices emphasizing extensive use of thrombolytics, unfractionated heparin (UFH), GP IIb/IIIa inhibitors, low molecular weight heparin (LMWH), and antiplatelet agents may contribute to a reduction in the incidence of VTE, including PE.
In light of the substantial burden of VTE in medical populations, current consensus statements on the prevention of VTE recommend assessment of all hospitalized patients, both medical and surgical, for thromboembolic risk and the use of appropriate prophylaxis. Specific prophylaxis recommendations have been made for patients with stroke and myocardial infarction (MI) by the American College of Chest Physicians (ACCP)18 and the International Consensus Conference.13 Prophylaxis also is recommended for other groups of medical patients with clinical risk factors for VTE. However, recommendations may vary among consensus documents. For example, recommendations by the United Kingdom-based second Thromboembolic Risk Factors (THRIFT II) Consensus Group are based on the individual level of thromboembolic risk assessed for each patient.19
Clinical approaches to evaluating patients with VTE vary among institutions, and approaches to such patients are debated in the medical literature. Currently, the ventilation-perfusion scan remains the initial (but not definitive) test for evaluating patients suspected of having PE. Compression ultrasonography is the initial procedure used for evaluating DVT. Although a number of adjunct and/or complementary tests shown to be useful in patient management are presented in this review, it should be stressed that no single test can be used alone to exclude the diagnosis. Rather, pre-test clinical probability of disease in conjunction with other diagnostic tests is the most outcome-effective method for evaluating patients suspected of having VTE.
VTE Risk Factors
Major risk factors for VTE include advancing age, venous stasis from any cause, and all causes of trauma, including childbirth. (See Table 1.)20 Thrombophilia, or hypercoagulable states, also predispose to VTE and may be classified as inherited or acquired. Inherited deficiencies of antithrombin, protein C, and protein S have been recognized for years. Among the most important inherited disorders predisposing to VTE is Factor V Leiden, which is a point mutation on Factor V in which an arginine is substituted for glutamine, causing coagulation factor resistance to degradation by protein C. This is the most common cause of thrombophilia and is identified in about 50% of individuals with recurrent VTE.21 It is estimated that this mutation occurs in 5% of the general population,22 and its presence not only doubles the risk of initial VTE,23 but is associated with a 40% risk of recurrence over an eight-year period.24
Prothrombin mutation recently has been recognized as another inherited cause of thrombophilia. This mutation causes higher plasma prothrombin levels and is associated with an increased propensity for forming thrombi through amplification of the coagulation system. The mutation occurs in 2-4% of the general population, and its prevalence is higher in southern Europe.23 These individuals have a 2-4 times increased risk of VTE. Moreover, among patients who have a recurrent VTE, this abnormality will be identified in 20%.23 Individuals with both prothrombin and Factor V mutations are at higher risk than those who have only one of the mutations.23,25
Malignancy is a well-recognized risk factor for VTE. All forms of cancer predispose a patient to thromboembolic disease; however, adenocarcinoma of the visceral organs is the most commonly recognized. Idiopathic VTE may be the first sign of an underlying malignancy: Such individuals have a 10-20% chance of developing cancer over a two-year period.23,26,27 Patients with both active malignancy as well as those with malignancy in remission are at increased risk for VTE.
Individuals also may have transient risk factors for VTE, including pregnancy, hormone replacement therapy, and prolonged immobilization or restricted mobility.20,23,28,29 Surgery is an important, common, and well-recognized transient risk factor, especially orthopedic and neurosurgical procedures. In the absence of prophylaxis, 45-70% of patients will develop DVT after a total knee or hip replacement surgery. Major trauma, especially involving the head, spinal cord, or pelvis, often is complicated by VTE. PE is the third most common cause of death in trauma patients who survive more than 24 hours. Almost 60% of major trauma patients will develop DVT.23
Travel and VTE. The risk of acquiring VTE posed by prolonged travel recently has received attention in both the medical literature and in the lay press. Prolonged travel requiring more than four hours duration is associated with an increased risk for VTE, even in patients without significant risk factors.30,31 In 1954, Homans first described several cases of travel-related DVT, including air travel.32 Since air travel is the most common mode of long-distance travel, "the economy class syndrome" has become a popular term to refer to travel-related VTE.
Unfortunately, most studies related to travel are small or poorly designed. Recently, a French study evaluated patients arriving at the airport who subsequently required urgent transport to the hospital.33 Of the 170 people transported, 56 (33%) were found to have PE. Only four of the 56 confirmed cases were considered to be at high risk for PE based on the presence of other medical problems or recent surgery. The risk for acquiring travel-related VTE is higher for those individuals who travel for more than six hours in duration or greater than 3500 miles. It should be stressed that this study only evaluated people requiring medical assistance upon arrival to the airport, and no data was available for those who may have developed VTE-related problems during the next few weeks.
The LONFLIT studies are a group of prospective trials evaluating the effect of long-haul air travel (average flight duration of 12.4 hours) on VTE.34 Ultrasound of the legs was performed on each subject before and after travel. Those at extremely high risk due to medical diseases such as hypertension, diabetes, renal insufficiency, heart failure, or recent surgery were excluded. LONFLIT1 was designed to evaluate the incidence of DVT in low-risk subjects.34 Travelers in the low-risk group were compared to higher-risk travelers defined as those with a previous DVT, known coagulation disorder, severe limitation of mobility, neoplastic disorder, or large varicose veins. No DVT occurred in the low-risk group (355 subjects). However, the incidence of DVT for those in the high-risk group (389 subjects) was 4.8%.
In LONFLIT2, the high-risk traveler was evaluated further.34 The study group was instructed to wear elastic compression stockings, whereas the control group had no stockings. All subjects were instructed to stretch their legs and move about the cabin during the flight. For those in the control group, DVT occurred in 4.5% (422 subjects), whereas those in the study group had an incidence of 0.24% (411 subjects). Another recent study from the United Kingdom randomized a group of travelers into two groups: one wearing below-the-knee compression stockings and the other (control group) without stockings.35 The subjects were all 50 years or older without a history of VTE. The median duration of flight was 24 hours. DVT occurred in 12 of 116 subjects in the control group. The study group had no episode of DVT, but four developed superficial thrombophelibitis. Therefore, it appears that compression stockings reduce the incidence of DVT in long-haul flights.
The most recent study, LONFLIT3, evaluated various methods of preventing DVT in high-risk subjects, as defined in the previous studies.36 Three hundred travelers were randomized to receive no prophylaxis (control group), aspirin for three days prior to travel, or an LMWH such as enoxaparin 2-4 hours before travel. Compression ultrasound was performed before and after travel. In the control group (82 subjects), 4.8% developed DVT; in the aspirin group (82 subjects), 3.6% had DVT; and in the LMWH group (82 subjects), there was no episode of DVT, but one episode of superficial thrombophlebitis for an incidence of 0.6%. The results were significant in that aspirin prophylaxis reduced the incidence of DVT by 25%. However, the number of thrombotic events was 3.6% higher in the aspirin group when compared to the LMWH group.
Therefore, it appears that some form of prophylaxis is indicated in travelers who are at risk for VTE. For the general public, some airlines have begun warning about the risks of VTE and recommending basic measures such as moving about the cabin while in flight and avoiding alcohol, smoking, prolonged leg crossing, and constrictive clothing.
For those at high risk, administration of enoxaparin 40 mg subq can be recommended prior to long-distance (i.e., greater than seven hours of flight duration) air travel. Regardless of the strategy employed, physicians should consider prolonged travel to be a significant risk factor for VTE, and include among those at risk individuals who have completed travel weeks before presentation.
Clinical Evaluation, Assessment Strategies, and Confirming the Diagnosis
A detailed clinical history aimed at identifying VTE risk factors and a physical examination remain the most important procedures in the evaluation of the patient with suspected VTE. When the deep venous system of the leg is occluded by thrombus, a patient may develop calf pain, edema, venous distention, and pain on dorsiflexion of the foot, or a positive Homan’s sign, which is suggestive of DVT. Unfortunately, the classic symptoms and signs of DVT occur in fewer than one-third of patients.37,38 Therefore, clinical assessment is not, in itself, satisfactory for detecting the majority of cases of DVT. This is complicated by the fact that other problems, such as cellulitis, can masquerade as DVT. (See Table 2.)20
 |
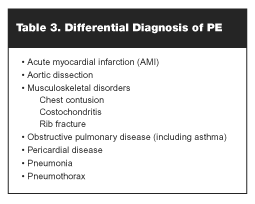
Similarly, symptoms caused by PE often are indistinguishable from those associated with other life-threatening disorders of the cardiorespiratory system. (See Table 3.) Almost without exception, PE is caused by embolization of thrombus from the deep venous system of the legs or arms. However, the clinical presentation is similar when air, fat, amniotic fluid, or thrombus reaches the respiratory vascular system.39 The presentation of PE is nonspecific, and symptoms may include dyspnea, anxiety, pleuritic chest pain, and hemoptysis. Sudden onset of such symptoms should raise suspicion for PE. In those with proven PE, fewer than one-third of patients also will have symptoms of DVT.38 Tachypnea remains the most common sign of PE, occurring in 70% of patients, while tachycardia is manifested in only one-third of patients.38 Syncope, sudden hypotension, or sudden death are signs of massive PE, but this suite of symptoms also may be attributed to cardiac tamponade, aortic dissection, or acute MI. (See Table 3.)37,39
 |
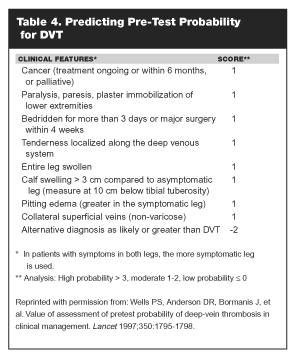
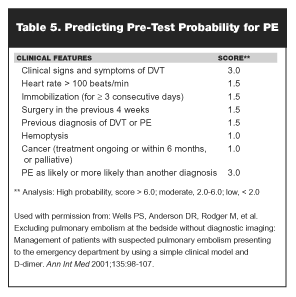
Assessment. Patient assessment and detection of DVT and PE present continuing challenges for hospital-based clinicians, including emergency physicians. In the prospective investigation of pulmonary embolism diagnosis (PIOPED) study, clinical assessment for the likelihood of disease was part of the process; however, no specific guidelines for arriving at the probability of disease were given.40 Recently, clinical models outlining probability of VTE have been published, all of which emphasize the importance of recognizing risk factors and clinical signs (See Tables 4 and 5) associated with venous thrombotic disorders.41,42 These models also stress the importance of assessing patients for other causes of signs and symptoms to differentiate patients with VTE from those with other conditions that present in a similar fashion. Routine tests such as the chest x-ray, electrocardiogram (ECG), and arterial blood gas (ABG) may be required to evaluate a heterogeneous patient population. If, based on findings from these modalities, another diagnosis is more likely than DVT or PE, the clinical probability of VTE is reduced.
 |
 |
Chest X-ray. The chest x-ray may be helpful by excluding the diagnosis of pneumonia and pneumothorax. However, it should be stressed that the patient with PE may demonstrate atelectasis, an elevated hemidiaphragm, or pleural effusion on chest x-ray. These findings are nonspecific and are insufficient for confirming the diagnosis of PE. The classical findings of Hamptom’s hump or Westermark sign (decreased vascularity) are seen infrequently. The chest x-ray even may be normal. In fact, a normal chest x-ray in a patient without underlying cardiopulmonary findings, and who presents with chest pain, dyspnea, and hypoxemia, increases the likelihood that PE is the underlying etiology for the clinical presentation.37,38
Electrocardiogram. The ECG is nonspecific and cannot confirm or exclude the diagnosis of PE. However, the ECG remains useful for ruling in or excluding the diagnosis of acute myocardial infarction or acute pericarditis. Sinus tachycardia is the most frequent ECG finding in PE. Other findings may include nonspecific ST-T wave changes, left or right axis deviation, and, rarely, signs of acute cor pulmonale with an S1 Q3 T3 pattern, right bundle-branch block, or P-wave pulmonale.43
Arterial Blood Gases. The ABG is a nonspecific but important test for evaluating patients suspected of having PE. Findings of hypoxemia, hypocapnea, and an elevated alveolar-arteriolar gradient raise the suspicion of PE, but these abnormal parameters also can be seen in pneumonia. Moreover, in young healthy patients, the ABG may be entirely normal, with one study demonstrating that 20% of those with proven PE had a normal ABG.38 One pitfall that has been identified in evaluating patients with suspected PE is relying too heavily on finding ABG abnormalities to confirm the diagnosis.44 Misinterpretation of ABG values also may compromise its diagnostic utility, especially in patients with chronic obstructive pulmonary disease (COPD) who manifest hypoxia with a normal carbon dioxide tension. On the one hand, these blood gas results may be the patient’s baseline ABG, or, alternatively, the "normal" pCO2 actually may represent new onset hyperventilation suggestive of PE. Comparing the ABG to baseline status is mandatory for accurate evaluation of COPD patients.
Non-Invasive Diagnostic Tests Specific for VTE
Historically, venography and pulmonary angiography have been the diagnostic "gold standards" for DVT and PE, respectively. To a great extent, these time-honored techniques have been replaced in clinical practice by noninvasive tests. Unfortunately, all noninvasive studies have limitations, and no single noninvasive test reliably excludes VTE in all patients. To overcome these pitfalls in diagnostic testing, a combination of noninvasive testing and clinical suspicion are utilized to evaluate the patient with suspected VTE.
Venous Ultrasonography. Compression ultrasonography of the common femoral vein and popliteal vein is the study most commonly used to evaluate patients with suspected DVT.37,45,46 It is highly sensitive (95%) and specific (96%) for symptomatic, proximal venous thrombosis located in the popliteal vein or in more proximal sites in the venous system of the leg.45 A non-compressible segment of vein accurately will diagnose DVT. Another advantage of this test is its ability to diagnose other pathology, including Baker’s cyst, abscess, or superficial thrombophlebitis. Any process that limits access to the leg limits the usefulness of the study. In this regard, lower extremity edema, obesity, tenderness, or plaster casts and splints can compromise utility of venous ultrasonography.37
The principal pitfall of the study is an over-reliance on a negative ultrasound, since the study does not accurately assess the calf veins. Most experts recommend serial ultrasounds to look for proximal extension of a thrombus that may occur in up to 25% of patients with calf DVT. The procedure is extended from the popliteal vein to the trifurcation of the calf veins. Multiple studies have shown that withholding anticoagulation at the time of an initial negative compression ultrasound is safe until a repeat study can be done in 5-7 days.45,46 In addition, patients with a documented history of DVT who present with recurrent symptoms in the same leg can be very difficult to assess. Abnormalities may be seen for up to one year in 50% of such patients. Therefore, a new noncompressible segment must be documented to accurately diagnose a recurrent DVT.47
Ventilation/perfusion (V/Q) scan. A nuclear medicine study, or V/Q scan, is the primary screening tool for patients with suspected PE.37,39 Its principal advantage is its simplicity and non-invasiveness. The test has been used for decades, and many radiologists have considerable experience with interpretation of these diagnostic scans. The most important investigation documenting the utility of V/Q scanning is the PIOPED study.40 In that study, 98% of patients with PE had an abnormality on the V/Q study. However, specificity was poor, with 72% of the study population having non-diagnostic studies. Among patients with angiographic documentation of PE, only 41% had a high-probability scan and 16% had a low-probability scan.40,48
Terminology in PIOPED was cumbersome and included four categories. Current terminology has been simplified to include three categories: normal or near-normal, high probability, and non-diagnostic.38,49 Normal studies or high probability studies, along with clinical assessment, help the clinician decide on management. Patients with high clinical pre-test probability with high probability V/Q scans should be treated for PE without further evaluation. Similarly, patients with low pre-test probability with normal or near-normal V/Q scans require no further evaluation but should be referred to their primary care providers for close follow-up. The large group with non-diagnostic studies is more challenging and generally requires further testing. (See Clinical Approach below.) A logistical problem with the V/Q scan is that many institutions do not have the ability to perform this test on a 24-hour basis.
Unfortunately, V/Q scanning is plagued by a number of pitfalls. For instance, bronchospasm in the absence of PE will lead to V/Q mismatching and, therefore, a patient with COPD or asthma presenting with chest pain and shortness of breath usually will have a non-diagnostic study. The most common cause of a V/Q mismatch is chronic or unresolved PE. Therefore, current recommendation is to obtain a repeat study three months after the initial episode to be used as the patient’s new baseline.49 Hopefully, this recommendation will augment diagnostic accuracy in patients who return to the hospital with recurrent symptoms. False negative studies and near-normal V/Q scans do occur in patients who recently were immobilized, had recent surgery or trauma, or had central venous instrumentation.49 With one or more of these risk factors, the possibility of PE is considerable and further testing becomes important when clinical suspicion is high. A large central or saddle embolus also may cause false negative V/Q scans, but such patients generally are critically ill, with the diagnosis being made by other modalities.
Helical Computerized Tomography (CT). In most institutions, the V/Q scan continues to be the initial test of choice for evaluation of PE. However, helical CT is gaining acceptance as a first tier diagnostic modality as computer-based techniques are refined, and as radiologists gain more experience with CT. In outlying facilities, the CT scan may be the only choice available and, therefore, it is used as the initial study. In a cooperative patient who is able to do a single breathhold maneuver, the procedure can be performed quickly. A contrast bolus must be used; therefore, CT is contraindicated in those patients with allergies to contrast or in those who have renal insufficiency. Filling defects within contrast-filled vessels identify acute thrombi.48,49
Recent investigations have confirmed the utility of CT scan for diagnosing PE. One study compared the accuracy of helical CT with the V/Q scan.50 In this study, CT and V/Q scans were done on all subjects, with angiography reserved for those who had discordant results on CT and V/Q scans. The conclusion of this study was that helical CT scan was more sensitive than V/Q scan (87% and 65%, respectively). The specificities were essentially the same (95% and 94%, respectively).
Based on multiple studies evaluating helical CT scanning, the sensitivity and specificity for detecting central PE are 86-95% and 75-97%, respectively.49 However, helical CT is somewhat compromised for detection of small emboli in the peripheral pulmonary arterial system. When all pulmonary vessels are analyzed, including subsegmental vessels, sensitivity decreases to 77%.49 The accuracy of diagnosing PE with CT is decreased due to volume averaging in the right middle lobe and lingular vessels, which are oriented horizontal to the plane of the scan. However, improved technology, which features faster scans and smaller slices, has improved the visibility of small vessels.51
The main advantage of helical CT over other methods is that it is very accurate for diagnosing other pulmonary disorders that may account for the patient’s symptoms. In one study in which the CT was negative for PE, new diagnoses, including emphysema, cancer, pulmonary edema, aspiration pneumonia, and bronchiolitis, were confirmed.52 Most clinicians agree that a positive study is sufficient to diagnose PE; however, a negative CT scan should be interpreted with caution.49,53 Helical CT scans accurately may diagnose another cause for the patient’s symptoms, and in the context of low clinical suspicion can exclude the diagnosis of PE.52,54
D-dimer. This is a noninvasive blood test measuring fibrin degradation products. The test is useful as an adjunct for the diagnosis of VTE; however, it may be a better test for the diagnosis of PE due to the higher load of fibrin products. The test is performed using different methodology at different institutions, and may include the following options: latex agglutination (least sensitive); enzyme-linked immunosorbent assay (ELISA) techniques; and new rapid methods.38,55,56 The major problem with the use of D-dimer is that different techniques have been used in the reported studies, making it difficult to compare study results.37
The ELISA method is highly sensitive (99%) for VTE, when using a cutoff value of 500 mcg/L. A lower value essentially excludes VTE and recently has been used to eliminate further testing in patients with low clinical suspicion.56-59 However, ELISA testing is labor- and time-intensive. An elevated D-dimer is not very specific for VTE. Other conditions that may cause an elevated D-dimer include cancer, inflammation, infection, and aging due to multiple comorbid conditions.38,60
Several studies have used the rapid, point-of-care, whole-blood agglutination D-dimer test (SimpliRED). The reported sensitivity for this test ranges from 85-100%.61 However, one recent study conducted in the emergency department (ED) reported a sensitivity of only 65% and a negative predictive value of 81%. The investigators conclude that this test failed to exclude the diagnosis of VTE.62 Another study agreed that the SimpliRED D-dimer tests should not be used alone to exclude the diagnosis of VTE. In this study, 10% of patients with documented PE had a normal D-dimer.56
Two additional studies have used the SimpliRED test in combination with clinical suspicion and non-invasive studies.57,59 Taken together, these investigators applied diagnostic algorithms to more than 900 patients with suspected VTE. In this algorithm, no further diagnostic testing was performed for those with a negative D-dimer and low pre-test suspicion of disease. In three-month follow-up, only two in this low-risk group developed VTE. The negative predictive value was 99.5% (CI 99.1-100%).
Echocardiography. When patients with known PE undergo echocardiography, 40% will have abnormalities associated with the right ventricle. However, this is a nonspecific finding. Detecting a thrombus in the main pulmonary artery is a helpful but unusual finding. At this time, echocardiographic evaluation is not indicated for all patients, but may be a valuable bedside test for critically ill patients with hypotension to help exclude other diagnoses, such as MI, valvular disorders, aortic dissection, or cardiac tamponade.37,39
New Modalities. Combining CT pulmonary angiography with CT venography may become a very useful test in the future. Using rapid CT methods and one bolus of contrast, the pulmonary circulation as well as the venous system below the diaphragm can be visualized. One study evaluating five patients demonstrated the accuracy of diagnosis. Moreover, this approach permits visualization of the venous system for comparison later if new symptoms arise and for possible interventions, such as insertion of filters.63
Magnetic resonance angiography (MRA) has the advantage of screening for DVT and PE in one test. Although this is a developing technology, it is considered safe, rapid, and accurate for diagnosing VTE. However, MRA is expensive when compared to other tests, although it is less costly then a pulmonary angiogram. In the future, clinicians can expect MRA to emerge as another possible noninvasive study for VTE.64
Invasive Studies for Diagnosis of VTE
Venography. Contrast venography remains the gold standard for the diagnosis of DVT. This is a good method to assess calf veins. Findings consistent with DVT include an intraluminal filling defect in two views, or an abrupt cutoff in the contrast column in a patient without previous DVT. Venography rarely is used as the initial modality of choice due to the acceptable accuracy of ultrasonography. The test also is difficult to perform in patients with obesity or pedal edema. However, it still may have a role in evaluating patients with high clinical suspicion of DVT and a negative ultrasound, or in those with a non-diagnostic ultrasound who have had a previous DVT.37,38 If venography cannot be performed due to pedal edema or plaster casts, allergy to dye, or patient refusal, other options must be considered. Serial ultrasonography over 5-7 days, or immediate CT scan or MRA may be alternatives for diagnostic evaluation.45,46,63,64
Pulmonary Angiography. The gold standard for the diagnosis of PE remains pulmonary angiogram. However, the test is invasive and costly, and has been associated with contrast reactions, renal failure, and death in 0.2-0.5% of patients.37,38,40 Even though this is the standard test, intraobserver variability is a problem, especially with subsegmental vessels (only 66% agree in subsegmental vessels).37,40,65,66 Diagnosing PE improves with attention to subsegmental vessels by adding cineangiography, selective studies, or balloon-occlusion.65 Pulmonary angiography usually is performed when V/Q scan results and clinical probability are discordant, such as high probability V/Q scan and low clinical suspicion.
A Systematic Clinical Approach to VTE Assessment and Diagnostic Confirmation
Several diagnostic algorithms exist in the literature to facilitate evaluation of VTE. The most important phase of the assessment is the initial clinical evaluation and formulation of a pre-test clinical probability. (See Tables 4 and 5.) No one test will safely exclude VTE. Therefore, the approach to the patient centers on pre-test suspicion of disease, and attempts to exclude other conditions that are in the differential diagnosis. (See Tables 2 and 3.)
Using compression ultrasound and the pre-test probability model (See Table 4), patients suspected of having DVT can be managed effectively using non-invasive tests, with only a few individuals requiring invasive studies. (See Insert.)41,67 As a rule, venography is indicated only when the results of an ultrasound and pre-test clinical probability are discordant. In other words, patients with an abnormal ultrasound and low clinical probability of DVT, or alternatively, those with a normal ultrasound in association with a high clinical probability for VTE, should be evaluated further. A study conducted in the ED using this scheme reduced costs significantly.63 If the patient had a moderate or high clinical suspicion of DVT after normal radiology hours, treatment was initiated and the test was performed the next day. Combining the two studies that used such an algorithm, only five of 800 patients diagnosed as not having DVT developed VTE in the three-month follow-up period.41,67
Pulmonary Embolism. The algorithm for suspected PE also is based on pre-test clinical probability and selected tests. The standard has been to use the V/Q scan as the initial study. The results of the V/Q scan in conjunction with pre-test probability (See Table 5) should guide further evaluation. (See Insert.)42 It is accepted that a normal or near-normal scan excludes PE, and a high probability scan is sufficient for diagnosis and treatment of PE. However, for those patients with discordant results, most clinicians feel that further evaluation is necessary. The patient with a high probability V/Q scan and low pre-test suspicion of disease will require further evaluation with CT scan or selective angiography to be certain of the etiology of their symptoms. Also, a normal V/Q scan in the patient with a very high pre-test suspicion of PE should be evaluated further with close follow-up and additional diagnostic testing such as compression ultrasound of legs, CT scan, angiography, or further cardiac evaluation.
The problem arises in the non-diagnostic category. Fifty percent of V/Q scans are non-diagnostic. If angiography was performed on all patients with non-diagnostic scans, only 25% would be diagnosed with PE.40,42
Accordingly, the algorithm for a non-diagnostic V/Q scan continues with bilateral leg ultrasonography, which will diagnose DVT in some patients, who will require treatment for VTE. If the ultrasound is normal, pre-test clinical probability is used to decide on further testing. A high clinical pre-test probability group would require angiography. In the setting of low clinical probability and normal ultrasound of the legs, PE can be excluded from the diagnosis. The moderate probability group should be evaluated further with serial ultrasound or angiography, or even helical CT.42,59
In an attempt to refine the algorithm further, other investigators have added D-dimer using an ELISA method to reduce the number of unnecessary tests.59 In this large study, a D-dimer less than 500 mcg/L was used to exclude the diagnosis of VTE in 31%, and no further evaluation was done. (See Figure 1.) For those with abnormal D-dimer, ultrasound of the legs was performed, and in those with a normal study, further evaluation was done. For the PE group, a non-diagnostic V/Q scan and low pre-test probability excluded the diagnosis of PE (12%). Those with non-diagnostic scans and moderate or high pre-test probability went on to angiography. Overall, an appropriate diagnosis was made in 94% of the study population by using pre-test probability and noninvasive studies. All patients who had PE or DVT excluded were followed for a three-month period, and found to have 1.8% risk of developing VTE.58
 |
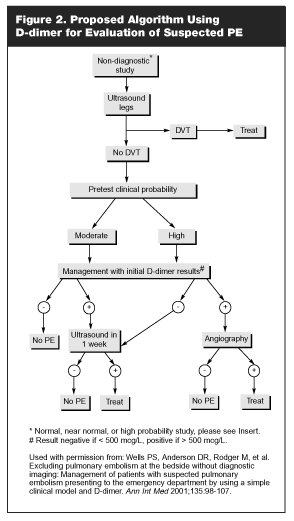
The results of D-dimer also can be added to further guide evaluation later in the PE algorithm. (See Insert.) Figure 2 demonstrates D-dimer being used in the group of patients with a non-diagnostic V/Q scan, negative ultrasound of the legs, and moderate and high pre-test clinical probability. In this study, only 0.1% developed VTE in the three-month follow-up period.59
 |
With helical CT becoming more available, it is gaining acceptance as the first diagnostic modality for those with suspected PE.38 If the CT is read as normal, or no PE, then pre-test probability is used to determine the need for angiography. While this approach would seem cost-effective and reasonable, it has not yet been validated in larger outcome studies. However, this approach with helical CT may be useful in patients who present with other reasons for their symptoms, such as COPD or emphysema. It remains to be seen how helical CT will be used in the evaluation of PE.
Finally, the patient presenting with symptoms of major PE requires immediate evaluation and treatment. The hypotensive patient often is too ill to be moved for multiple tests. These patients benefit from bedside echocardiography primarily to exclude other causes of hypotension. Next, angiography is undertaken to quickly evaluate the anatomy and start definitive treatment.37 Helical CT also would be accurate in the diagnosis of major PE. Its utility in such cases remains to be validated.
Pitfalls in Diagnosis
The major pitfalls of VTE evaluation are summarized in Table 6. Failure to consider pre-test clinical suspicion or changing the pre-test suspicion without sufficient evidence is a major pitfall. The models for pre-test probability have been proven helpful in the clinical assessment of VTE.40,41,59 The other broad category of pitfalls is over-reliance on any one physical examination finding or diagnostic test to safely exclude or make the diagnosis. All diagnostic tests discussed in the review are most useful as complimentary tests taken together with clinical probability of VTE. When history, physical examination, and risk factors are used to arrive at the pre-test clinical probability of disease, the astute clinician can determine an appropriate sequence for diagnostic testing and arrive at the correct diagnosis.
 |
References
1. Sandler DA, Martin JF. Autopsy proven pulmonary embolism in hospital patients: Are we detecting enough deep vein thrombosis? J R Soc Med 1989;82:203-205.
2. Cohen AT, Edmonson RA, Phillips MJ, et al. The changing pattern of venous thromboembolic disease. Haemostasis 1996;26:65-71.
3. Anderson FA, Wheeler HB, Goldberg RJ, et al. A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT study. Arch Intern Med 1991;151:933-938.
4. Prevention of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 1997;16:3-38.
5. Lewis HD, Davis JW, Archibald DG, et al. Protective effects of aspirin against acute myocardial infarction and death among men with unstable angina. Results of a Veteran Administration Cooperative Study. N Engl J Med 1983;309:396-403.
6. Goldhaber SZ, Savage DD, Garrison RI, et al. Risk factors for pulmonary embolism—the Framingham study. JAMA 1983;74: 1023-1028.
7. Anderson FA, Wheeler HB, Goldbert R, et al. Physician practices in the prevention of venous thromboembolism. Ann Intern Med 1991;115:591-595.
8. Olatidoye AG, Wu AH, Feng Y, et al. Prognostic role of troponin T versus troponin I in unstable angina pectoris for cardiac events with meta-analysis comparing published studies. Am J Cardiol 1998;81: 1405-1410.
9. Belch JJ, Lowe GDO, Ward AG, et al. Prevention of deep vein thrombosis in medical patients by low-dose heparin. Scott Med J 1981;26:115-117.
10. Cade JF. High risk of the critically ill for venous thromboembolism. Crit Care Med 1982;10:448-450.
11. Anderson GM, Hull E. The effect of dicoumarol upon the mortality and incidence of thromboembolic complication of congestive heart failure. Am Heart J 1950;39:697-702.
12. Salzman EW, Hirsh J. Prevention of venous thromboembolism. In: Coleman RW, Hirsh J, Marder VJ, et al. (eds.) Hemostasis and Thrombosis: Basic Principles and Clinical Practice. New York: Lippincott;1982:986.
13. Nicolaides AN, Bergquist D, Hull R, et al. Consensus statement. Prevention of venous thromboembolism. Int Angiol 1997;16:3-38.
14. Hirsh DR, Ingenito EP, Goldhaber SZ. Prevalence of deep venous thrombosis among patients in medical intensive care. JAMA 1995; 274:335-337.
15. Dekker E, Nurmohamed MT, Heijboer H, et al. Incidence of deep venous thrombosis (deep vein thrombosis) in high-risk intensive care patients. Thromb Haemost 1991;65:1348.
16. Diebold J, Lohrs U. Venous thrombosis and pulmonary embolism. A study of 5039 autopsies. Pathol Res Pract 1991;187:260-266.
17. Baglin TP, White K, Charles A. Fatal pulmonary embolism in hospitalized medical patients. J Clin Pathol 1997;50:609-610.
18. Clagett GP, Andersen FA, Heit JA, et al. Prevention of venous thromboembolism. Chest 1998;114(5 suppl):531S-560S.
19. Second Thromboembolic Risk Factors (THRIFT II) Consensus Group. Risk of and prophylaxis for venous thromboembolism in hospital patients. Phlebology 1998;13:87-97.
20. Hyers TM. Venous thromboembolism. Am J Respir Crit Care Med 1999;159:1-14.
21 Moser KM, Fedullo PF, LitteJohn JK, et al. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA 1994;271:223-225.
22. Middeldorp S, Meinardi JR, Koopman MMW, et al. A prospective study of asymptomatic carriers of the factor V Leiden mutation to determine the incidence of venous thromboembolism. Ann Intern Med 2001;135:322-327.
23. Martinelli I. Risk factors in venous thromboembolism. Thromb Haemost 2001;86:395-403.
24. Simioni P, Prandoni P, Lensing AWA, et al. The risk of recurrent venous thromboembolism in patients with An Arg506-->G1n mutation in the gene for factor V (factor V Leiden). N Engl J Med 1997; 336:399-403.
25. DeStefano V, Martinelli I, Mannuccio P, et al. The risk of recurrent deep venous thrombosis among heterozygous carriers of both factor V Leiden and the G20210A prothrombin mutation. N Engl J Med 1999;341:801-806.
26. Schulman S, Lindmarker P. Incidence of cancer after prophylaxis with warfarin against recurrent venous thromboembolism. N Engl J Med 2000;342:1953-1958.
27. Sorensen HT, Mellemkjar L, Steffensen FH, et al. The risk of a diagnosis of cancer after primary deep venous thrombosis or pulmonary embolism. N Engl J Med 1998;338:1169-1173.
28. Toglia MR, Weg JG. Venous thromboembolism during pregnancy. N Engl J Med 1996;335:108-114.
29. Varas-Lorenzo C, Garcia-Rodriguez LA, Cattaruzzi C, et al. Hormone replacement therapy and the risk of hospitalization for venous thromboembolism: A population-based study in southern Europe. Am J Epidemiol 1998;147:387-390.
30. Rege KP, Bevan DH, Chitolie A, et al. Risk factors and thrombosis after airline flight. Thromb Haemost 1999;81:995-996.
31. Ferrari E, Chevallier T, Chapelier A, et al. Travel as a risk factor for venous thromboembolic disease. Chest 1999;115:440-444.
32. Homans J. Thrombosis of the deep leg veins due to prolonged sitting. N Eng J Med 1954;250:148-149.
33. Lapostolle F, Surget V, Borron SW, et al. Severe pulmonary embolism associated with air travel. N Eng J Med 2001;345: 779-783.
34. Belcaro G, Geroulakos G, Nicolaides AN, et al. Venous thromboembolism from air travel: The LONFLIT study. Angiology 2001;52:369-374.
35. Scurr JH, Machin SJ, Balley-King S, et al. Frequency and prevention of symptomless deep-vein thrombosis in long-haul flights: A randomised trial. Lancet 2001;357:1485-1489.
36. Cesarone MR, Belcaro G, Nicolaides AN, et al. Venous thrombosis from air travel: The LONFLIT3 study—Prevention with aspirin vs low-molecular-weight heparin (LMWH) in high-risk subjects: A randomized trial. Angiology 2002;53:1-6.
37. American Thoracic Society. The diagnostic approach to acute venous thrombo-embolism. Am J Respir Crit Care Med 1999;160: 1043-1066.
38. Perrier A, Bounameaux H. Cost-effective diagnosis of deep vein thrombosis and pulmonary embolism. Thromb Haemost 2001:86; 475-487.
39. Goldhaber SZ. Pulmonary embolism. N Eng J Med 1998;339: 93-104.
40. Prospective investigation of pulmonary embolism diagnosis (PIOPED). Value of the ventilation/perfusion scan in acute pulmonary embolism.The PIOPED Investigators. JAMA 1990;263: 2753-2759.
41. Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet 1997;350:1795-1798.
42. Wells PS, Ginsberg JS, Anderson DR, et al. Use of a clinical model for safe management of patients with suspected pulmonary embolism. Ann Intern Med 1998;129:997-1005.
43. Ullman E, Brady WJ, Perron AD, et al. Electrocardiographic manifestations of pulmonary embolism. Am J Emerg Med 2001;19: 514-519.
44. Stein PD, Goldhaber SZ, Henry JW, et al. Arterial blood gas analysis in the assessment of suspected acute pulmonary embolism. Chest 1996;109:78-81.
45. Birdwell BG, Raskob GE, Whitsett TL, et al. The clinical validity of normal compression ultrasonography in outpatients suspected of having deep venous thrombosis. Ann Intern Med 1998;128:1-7.
46. Cogo A, Lensing AWA, Koopman MMW, et al. Compression ultrasonography for diagnostic management of patients with clinically suspected deep vein thrombosis: Prospective cohort study. BMJ 1998;316:17-20.
47. Kearon C, Ginsberg JS, Hirsh J. The role of venous ultrasonography in the diagnosis of suspected deep venous thrombosis and pulmonary embolism. Ann Intern Med 1998;129:1044-1049.
48. Maki DD, Gefter WB, Alavi A. Recent advances in pulmonary imaging. Chest 1999;116:1388-1402.
49. Worsley DF, Alavi A. Radionuclide imaging of acute pulmonary embolism. Radiol Clin North Am 2001;39:1035-1052.
50. Mayo JR, Remy-Jardin M, Muller NL, et al. Pulmonary embolism: Prospective comparison of spiral CT with ventilation-perfusion scintigraphy. Radiology 1997;205:447-452.
51. Remy-Jardin M, Remy J, Artaud D, et al. Peripheral pulmonary arteries: Optimization of the spiral CT acquisition protocol. Radiology 1997;204:157-163.
52. Garg K, Sieler H, Welsh CH, et al. Clinical validity of helical CT being interpreted as negative for pulmonary embolism: Implications for patient treatment. AJR 1999;172:1627-1631.
53. Perrier A, Howarth N, Didier D, et al. Performance of helical computed tomography in unselected outpatients with suspected pulmonary embolism. Ann Intern Med 2001;135:88-97.
54. Kim K, Muller NL, Mayo JR. Clinically suspected pulmonary embolism: Utility of spiral CT. Radiology 1999;210:693-697.
55. Bounameaux H, DeMoerloose P, Perrier A, et al. D-dimer testing in suspected venous thromboembolism: An update. QJ Med 1997; 90:437-442.
56. Ginsberg JS, Wells PS, Kearon C, et al. Sensitivity and specificity of a rapid whole-blood assay for D-dimer in the diagnosis of pulmonary embolism. Ann Intern Med 1998;129:1006-1011.
57. Kearon C, Ginsberg JS, Douketis J, et al. Management of suspected deep venous thrombosis in outpatients by using clinical assessment and D-dimer testing. Ann Intern Med 2001;135:108-111.
58. Perrier A, Desmarais S, Miron MJ, et al. Non-invasive diagnosis of venous thromboembolism in outpatients. Lancet 1999;353:190-195.
59. Wells PS, Anderson DR, Rodger M, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: Management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and D-dimer. Ann Inter Med 2001;135:98-107.
60. Tardy B, Tardy-Poncet B, Viallon A, et al. Evaluation of D-dimer ELISA test in elderly patients with suspected pulmonary embolism. Thromb Haemost 1998;79:38-41.
61. deGroot MR, van Marwijk Kooy M, Pouwels JG, et al. The use of a rapid D-dimer blood test in the diagnostic work-up for pulmonary embolism: A management study. Thromb Haemost 1999;82: 1588-1592.
62. Farrell S, Hayes T, Shaw M. A negative SimpliRED D-dimer assay result does note exclude the diagnosis of deep vein thrombosis or pulmonary embolus in emergency department patients. Ann Emerg Med 2000;35:121-125.
63. Loud PA, Grossman ZD, Klippenstein DL, et al. Combined CT venography and pulmonary angiography: A new diagnostic technique for suspected thromboembolic disease. AJR 1998;170: 951-954.
64. Meaney JFM, Weg JG, Chenevert TL, et al. Diagnosis of pulmonary embolism with magnetic resonance angiography. N Engl J Med 1997;336:1422-1427.
65. Stein PD, Henry JW, Gottschalk A. Reassessment of pulmonary angiography for the diagnosis of pulmonary embolism: Relation of interpreter agreement to the order of the involved pulmonary arterial branch. Radiology 1999;210:689-691.
66. Diffin DC, Leyendecker JR, Johnson SP, et al. Effect of anatomic distribution of pulmonary emboli on interobserver agreement in the interpretation of pulmonary angiography. AJR 1998;171:1085-1089
67. Anderson DR, Wells PS, Stiell I, et al. Thrombosis in the emergency department. Use of a clinical diagnosis model to safely avoid the need for urgent radiological investigation. Arch Intern Med 1999;159:477-482.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
