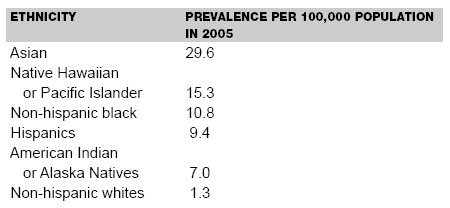Tuberculosis: A Primer for the Emergency Physician
Tuberculosis: A Primer for the Emergency Physician
Authors: Ewen Wang, MD, Assistant Professor, Division of Emergency Medicine, Stanford University, Stanford, CA; and Aparajita Sohoni, MD, Resident, Highland General Hospital—Alameda County Medical Center, Oakland, CA.
Peer Reviewers: Gregory A. Volturo, MD, FACEP, Professor of Emergency Medicine and Medicine, Vice Chairman, Department of Emergency Medicine, University of Massachusetts Medical School, Worcester, MA; and Gregory J. Moran, MD, FACEP, Clinical Professor of Medicine, David Geffen School of Medicine at University of California–Los Angeles, Department of Emergency Medicine and Division of Infectious Diseases, Olive View–UCLA Medical Center.
Tuberculosis (TB) is the second most common infectious cause of death in adults worldwide after HIV/AIDS.1 Approximately one-third of the world's population, or approximately 2 billion people, are infected with Mycobacterium tuberculosis.2 The global disease burden of tuberculosis continues to increase. The emergence of multi-drug resistant tuberculosis, the ongoing HIV epidemic, and societal issues such as poverty and homelessness influence the prevalence of this disease. While TB usually is a chronic disease and treatment is not usually initiated in the emergency department (ED), all emergency practitioners should have an understanding of the epidemiology, presentation, and treatment of TB.
Global Epidemiology
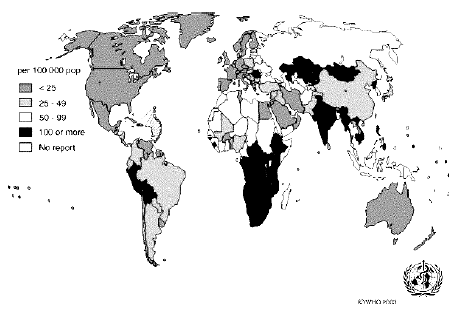
In 2000, the World Health Organization (WHO) estimated 8 to 9 million new cases of tuberculosis,1 with 95% of the disease burden occurs in developing countries.1 Sub-Saharan Africa has the highest incidence rate (290 per 100,000 population); Asian countries including India, Pakistan, China, Bangladesh, and Indonesia have the greatest number of cases.1 Most cases (5-6 million) occur in patients between 15 and 49 years old.1 (See Figure 1.) In 1993, the WHO declared tuberculosis a "global emergency," and estimated that nearly 70 million people would die from TB between 1997 and 2020 if current incidence levels were to continue.3,4
| Figure 1. Global Distribution of Tuberculosis, 2002 |
|
|
| Tuberculosis case notification rates by country in 2002. The designations employed and the presentation of material on this map do not imply the expression of any opinion whatsoever on the part of the World HealthOrganization concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries.White lines on maps represent approximate border lines for which there may not yet be full agreement. Reprinted with permission from: World Health Organization. Global tuberculosis control: Surveillance, planning, financing. WHO report 2004. DocumentWHO/HTM/TB/2004.331. Geneva (Switzerland): World Health Organization; 2004: 218, fig. 4. |
United States Epidemiology
Presently, approximately 15 million people are infected with Mycobacterium tuberculosis.5 The number of tuberculosis cases reported in the United States decreased nearly 75% from 1953 to 1985 primarily due to public health measures. From1985 to 1992, there was a sharp increase in the prevalence of tuberculosis,5 largely attributed to the rise in HIV-infected patients. HIV-infected patients likely account for 30-50% of the increase in cases of tuberculosis.2,6 With greater public health efforts, the annual incidence of TB has decreased since the peak in 1992.
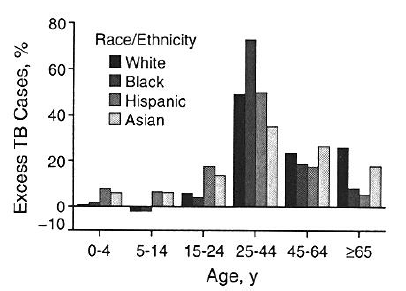
Between 1985 and 1990, the incidence of tuberculosis in Hispanic populations increased 55%, and the incidence in African-American populations increased 27%.5 (See Figure 2.) With the identification and rise in numbers of HIV-affected patients, the "face of the TB patient" changed dramatically to include younger patients with primary disease vs. older patients with reactivation disease. HIV has also dramatically influenced the reactivation of latent tuberculosis infection, with rates as high as 7-10% per year in an HIV-infected patient.8
| Figure 2. Disproportion of TB Cases in Minority Populations |
|
|
| Reprinted with permission from: Cantwell MF, Snider DE, Cauthen GM, et al. Epidemiology of tuberculosis in the United States, 1985-1992. JAMA 1994;272:535. |
Foreign-born individuals accounted for 53% of cases of tuberculosis in 2004,9,10 with Mexico, the Philippines, Vietnam, India, and China representing more than half of the foreign-born cases of TB in the United States in 2005.7 Almost one-half of the cases of tuberculosis among foreign-born individuals occur more than five years after arrival in the United States.9 (See Table 1.)
| Table 1. Prevalence per 100,000 Population of Tuberculosis by Ethnicity in 20057 |
|
|
Immunosuppressive agents such as TNF-alpha antagonists influence the host's resistance to granulomatous disease,11 in which the host's immune response walls off bacteria into granulomas. Chronic corticosteroid use at daily doses greater than 15 mg for greater than one month has been identified as a risk factor for TB.12 Current use of corticosteroids conferred an increased risk of development of tuberculosis with an odds ratio (OR) of 4.9 (confidence interval CI 2.9-8.3).13
Microbiology
Tuberculosis is caused by any member of the Mycobacterium tuberculosis complex, with Mycobacterium tuberculosis causing the majority of infections in humans. M. tuberculosis replicates slowly, taking 25-32 hours to divide.14,15 M. tuberculosis has no endotoxins or exotoxins, and thus does not generate any immediate host response to the infection.14,15 In an immunocompetent host, the numbers of tubercle bacilli will grow over 2-12 weeks until levels between 10,000 and 100,000 organisms are reached, at which point a cellular immune response manifested by a positive tuberculin skin test is produced.14,15 This slow growth, along with lack of host response to infection and subsequent microbial ability to survive intracellularly within macrophages, makes the bacillus difficult to eradicate with antibiotics while also enabling resistant organisms to develop if bacterial infection is incompletely treated.
Transmission
Tuberculosis is spread via airborne droplet transmission.1 Droplets are produced when patients with pulmonary or laryngeal tuberculosis speak, cough, or sneeze,1 or during sputum induction, aerosol treatments, or bronchoscopy.12 Aerosolized particles are capable of remaining in the air for minutes to hours. Droplets sized between 1 and 5 microns in diameter are best able to reach the pulmonary alveoli where they will replicate. The small size of these droplets also enables them to circulate within indoor air currents for long periods of time.16
Droplets deposited on intact skin are not infectious as the organisms are unable to invade tissue.12
Pathogenesis
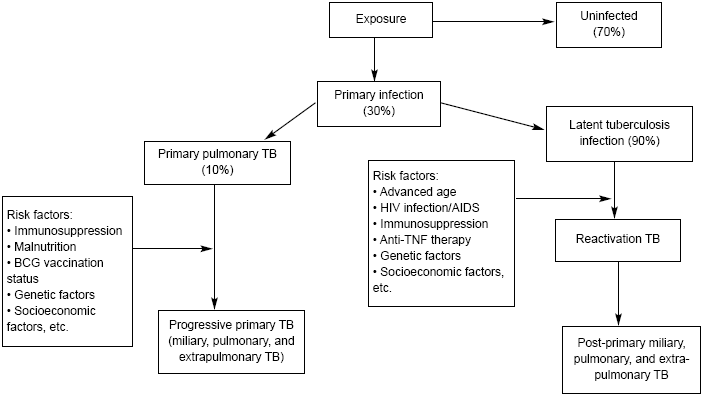
Following inhalation of a droplet and its deposition within a pulmonary alveolus, the tubercle bacillus is ingested by an alveolar macrophage, the host's natural defense mechanism.1 At this point, the host either does not become infected (70% of individuals), or develops a "primary infection" (30% of individuals).17 (See Figure 3.) The immediate clearance of the organism, or uninfected state, is mainly determined by the bacterial virulence and inherent bactericidal ability of the macrophage.1 The "primary infection" state refers to infection of the lungs classically appearing as a mid or lower lung parenchymal lesion or as hilar adenopathy.1
| Figure 3. Outcomes Following Exposure to Tuberculosis |
|
|
| Adapted from: Sharma SK, Mohan A, Sharma A, et al. Miliary tuberculosis: New insights into an old disease. Lancet Infect Dis 2005;5:415-430. |
Of the patients with a primary infection, 90% will contain the infection and develop latent tuberculosis infection or LTBI.17 In LTBI, activated T lymphocytes and macrophages form granulomas with M. tuberculosis at the center.1 M. tuberculosis is usually not viable or capable of replicating or spreading beyond the granuloma.1 In the absence of any immune compromise, viable bacteria contained within the granulomas do not cause active disease.1
In 10% of primary infected patients, the infection is not contained and the disease develops into progressive primary TB.17 During the development of progressive primary TB, factors such as immunosuppression, malnutrition, or age can predispose the patient to developing miliary TB.
In patients with LTBI, the lifetime risk of re-activation is 10%.17 If reactivated, this infection is termed post-primary TB and presents with lesions at the apices of the lungs and little associated lymphadenopathy or caseating necrosis.12 Fifty percent of reactivations occur within the first 2 years after primary infection.17 However, in HIV-infected individuals, the risk of reactivation increases approximately 10% per year.17 During reactivation, lymphohematogenous spread is also possible, resulting in miliary TB.17 Immunosuppressive states conferring an increased risk of post-primary TB or reactivation TB include: HIV infection and AIDS, diabetes mellitus, malignant lymphoma, end stage renal disease (ESRD), alcohol abuse, or chronic corticosteroid use.17
TB and the Host
Immunocompetent Hosts. In immunocompetent hosts, 85% of reported tuberculosis cases involve only the lungs, while 15% involve both nonpulmonary and pulmonary sites, or only nonpulmonary sites.18
Immunocompromised Hosts. In patients with advanced HIV infection, 38% had pulmonary disease, 30% had extrapulmonary disease, while 32% had both pulmonary and extrapulmonary involvement.19 Worsening immune function confers an increased risk in the development of extrapulmonary tuberculosis.17 Most extrapulmonary cases of TB occur with CD4 counts less than 200/microliter, usually less than 100/microliter.17
In immunocompromised patients, the disease process is not only more extensive, but also more destructive.19 Caseating necrosis causes lesions capable of mechanically eroding into airways and rendering the host infectious to others.19
The immunocompromised nature of the host leads to an unreliable response to the tuberculin skin test. Thus, the tuberculin skin test, if negative, cannot rule out disease.
TB and the Pediatric Patient. In 2001, approximately 11% of the estimated 8.3 million new cases of TB worldwide occurred in children.20 In 2001, the United States reported 931 cases of TB in children, 5.8% of all tuberculosis cases that year.20 Half occurred in 3 states: California, Texas, and New York.20 In the former Soviet Union, Peru, and Sub-Saharan Africa, the rates of pediatric tuberculosis are increasing.20 Children who are carriers of tuberculosis represent the "reservoir of future disease," underlining the importance of understanding the pediatric burden of tuberculosis.20 Lastly, HIV-positive children are often exposed to HIV-positive parents who are at increased risk of developing active disease.
Pediatric patients are more likely than adults to progress to clinical disease and to develop extrapulmonary forms of tuberculosis.20 Presumably due to their immature cellular immunity, children younger than 5 years old are felt to be at a higher risk of developing active disease following infection.20 In children younger than 1 year of age, 43% of children exposed will develop active pulmonary disease.20 In children between 1 and 5 years of age, 24% will develop active pulmonary disease.20 In contrast, among adolescents 15% of those exposed will develop active pulmonary disease, and among adults only 5-10% will develop active disease.20 Children younger than 5 years old have also been found to develop progressive fulminant disease, especially miliary TB and TB meningitis, more frequently than older children or adults.20
Bacillus Calmette-Guerin (BCG) vaccine is a live attenuated vaccine derived from Mycobacterium bovis strains.21 While the BCG vaccine is not administered in the United States, it is administered to approximately 80% of infants world-wide.21 The efficacy of the BCG vaccine in the prevention of tuberculosis varies considerably, with a range of 0% to 80%.22 Theories for this dramatic difference in efficacy include questioning the quality of the BCG vaccine administered, the potential for waning immunity over time, and the patient's prior exposures to any similar Mycobacteria or other atypical bacteria.21
In vaccinated children, a meta-analysis of the BCG vaccine demonstrates a 75-86% efficacy rate against meningeal and miliary tuberculosis.23 In countries with a high prevalence of tuberculosis, studies have shown the BCG vaccine to be a cost-effective intervention for the prevention of miliary and meningeal tuberculosis in children.24
Major side effects of the BCG vaccine include lymphadenopathy, pustule formation, drainage, and scarring.25 Various case reports can be found in the literature of disseminated BCG infection in both immunosuppressed and immunocompetent individuals but the overall incidence is small.25-27
Children younger than 5 years old rarely can produce sputum for evaluation. Instead, gastric aspirates can be used. Gastric aspirates are smear-positive in fewer than 20%, and culture-positive in fewer than 50%, of children.20 The sputum specimens, when received, are rarely AFB smear positive. In children younger than 12 years of age, sputum smears are not positive in approximately 95% of cases.20 Since the WHO only counts smear-positive cases in compiling the epidemiology of tuberculosis, the global pediatric burden of tuberculosis is grossly underestimated.
TB and the Pregnant Patient. In general, pregnancy does not alter the management of tuberculosis to a great extent. Preventive therapy with INH and pyridoxine usually can be postponed until after delivery.28 This decision is made primarily because of a risk of increased hepatic toxicity in women, particularly minority women.29 INH does cross the placenta, but has no increased toxicity to the fetus.28 INH is also excreted in the breast milk, but at low concentrations, and therefore should not affect the maternal decision to breastfeed.30
Active tuberculosis in a pregnant patient must be treated immediately.28,31 Streptomycin should be avoided given its risk of ototoxicity. Pyrazinamide should also be avoided unless the patient is HIV-positive, in which case the benefits are thought to outweigh the risks of giving a drug with little data known about its risks for teratogenicity.32 Transplacental transmission of tuberculosis is rare, though transmission after birth is possible.33,34 Mothers should be screened for possible infectivity at the time of delivery.35
Presentations: Classic Pulmonary, Miliary, and Extrapulmonary
Pulmonary TB. Pulmonary tuberculosis presents with a constellation of systemic and pulmonary symptoms depending on the immune status of the patient.36 Primary TB can be asymptomatic or can manifest as a pneumonia with productive cough and fevers, while progressive primary disease may present with cough, hemoptysis, and weight loss.36 Post-primary disease usually presents as chronic cough with sputum, weight loss, and occasionally hemoptysis and pleuritic chest pain.36
Systemic Symptoms. Fever is classically thought of as a systemic symptom of tuberculosis, along with weight loss, night sweats, loss of appetite, and weakness.37 Studies have shown that fever has been observed in 37-80% of patients.37,38 In one study, 21% of patients had no fever at any point in the course of hospitalization for tuberculosis.38 Of the febrile patients, 34% became afebrile within one week of beginning treatment, while 64% became afebrile within two weeks of beginning treatment.38
Hematologically, tuberculosis has been associated with a variety of reactions ranging from a leukemoid reaction to leukopenia.39.40 In approximately 10% of patients, a slight increase in the leukocyte count and anemia is seen.39,40 An increase in monocytes and eosinophils may also be seen.39,40 The major electrolyte abnormality associated with tuberculosis is hyponatremia, which in a single series was found in 11% of patients.41 This is thought to be secondary to an antidiuretic hormone-like substance released from affected lung tissue in the setting of tuberculosis infection.42
Pulmonary Symptoms. Patients with pulmonary tuberculosis classically present with a cough. The cough is initially non-productive, but becomes productive of yellowish or yellow-greenish sputum as the illness progresses and tissue necrosis and inflammation occurs. Hemoptysis itself does not indicate active tuberculosis, but rather can result from one of multiple mechanisms including erosions of calcified lesions into airway lumens (broncholithiasis), bacterial or fungal infection in a cavity, rupture of a dilated vessel in a cavity wall (Rasmussen's aneurysm) causing massive hemoptysis, or from residual tuberculous bronchiectasis.43-45 Only 5-15% of cases of hemoptysis in the United States are felt to be due to tuberculosis.43-45 Hemoptysis is often of minimal amount and results in blood-streaked sputum.
Pleuritic pain may occur if there is inflammation of the lung parenchyma approximating a pleural surface, in the presence or absence of an effusion.36 Dyspnea is rare, but when it occurs is often in the setting of pleural effusions, pneumothorax, or extensive parenchymal involvement.36 Spontaneous pneumothorax secondary to tuberculosis can occur if either a cavity or an area of caseous necrosis erodes into the pleural space. The formation of a bronchopleural fistula can occur, necessitating chest tube placement for drainage and lung reinflation.46,47 In the United States, spontaneous pneumothorax associated with TB is very uncommon, with cases reported in less than 1% of hospitalized patients.46,47
Extensive pulmonary parenchymal involvement results from destruction of significant portions of one or both lungs, most often occurring in primary TB.48,49 Destruction can also follow years of chronic reactivation TB. Pulmonary gangrene, or the acute destruction of lung tissue, can result in the development of air-filled cysts that coalesce into cavities.50 Extreme dyspnea, respiratory failure, and death can ensue as the effects of the necrotic lung tissue augment.50
The physical exam findings of pulmonary tuberculosis are non-specific. Rales or bronchial breath sounds may be heard over areas of consolidation.49 Dullness or decreased fremitus may occur in areas of pleural thickening or pleural effusion.49 Hollow breath sounds over cavitations, also known as amphoric breath sounds, indicate likely significant disease but are often difficult to auscultate.49
Radiographic Changes. The classic chest x-ray for primary pulmonary TB is the finding of the Ghon complex on x-ray.36 The Ghon complex refers to the parenchymal focus of caseous necrosis—indicative of the area of initial bacillus infection—and the enlarged draining lymph nodes, as seen on gross specimen or on chest x-ray.36 It is important to remember that a significant percentage of patients with active disease to have no detectable radiographic abnormality on chest x-ray. Specifically, 10% of cases in a series reviewed from 1996 to 1997 had normal chest radiographs.51
In primary pulmonary TB, one study demonstrated that the most common abnormality seen on chest x-ray is hilar adenopathy, observed in 65% of patients.52 Hilar changes were seen within two months in all cases, starting as early as one week following skin test conversion.52 Hilar adenopathy can lead to right middle lobe collapse, presumably because the right middle lobe is surrounded by an increased number of lymph nodes.52 Also, the right middle lobe has a sharper branching angle, a smaller internal calibre, and a relatively longer length.52
Pulmonary infiltrates are seen in 27% of patients, with lower infiltrates observed in 33% of patients, and upper lobe infiltrates in 13%.52 Of adults with infiltrates, 43% also had pleural effusions.52 The infiltrates often present with ipsilateral hilar enlargement.52
In primary TB, pleural effusions may also be the only radiographic evidence of disease. One study demonstrated pleural effusions occurring as the sole feature in 5% of all primary TB cases.36
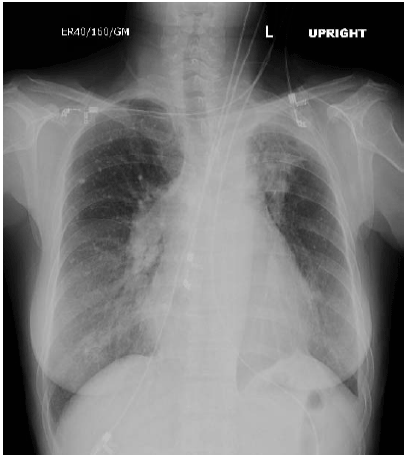
In reactivation tuberculosis, typically the apical-posterior segments of the upper lung fields are involved.36 (See Figure 4.) Studies demonstrate this pattern in 80-90% of patients, followed by involvement of the superior segment of the lower lobes, and the anterior segment of the upper lobes.53-56 A recent large series of adults demonstrated that 19-40% of patients with TB also had cavities with demonstrable air-fluid levels.53-56 Atypical presentations of reactivation TB include the presence of hilar adenopathy, at times complicated by right middle lobe collapse; lower lung field infiltrates or cavities; solitary nodules; or isolated pleural effusions.57-59
| Figure 4. Chest X-Ray Demonstrating Classic Left Upper Lobe (LUL) Reactivation TB in an Immigrant Patient with Positive PPD |
|
|
| (Used with permission from: Highland Hospital Radiology Teaching File) |
CT of the chest is more sensitive than chest x-ray in diagnosing tuberculosis, particularly in identifying smaller lesions in the lung apex. The classic CT finding is the "tree in bud" appearance of nodules and branching linear densities.60
Diagnosis of Pulmonary TB. Sample Collection. Sputum collection is key to the diagnosis of pulmonary TB. Three sputum samples on consecutive days is the general standard. Sputum can be induced with use of aerosolized sterile hypertonic saline.61 Patients should be in respiratory isolation during the induction of sputum because of the increased aerosolization that vigorous coughing produces.61 Gastric aspiration is also an option in children or in those patients who do not produce sputum even after aerosolized saline attempts.61
Sample Testing. A significant number of bacteria must be present to have a specimen stain positive on an acid-fast smear. One hundred four colony-forming units (CFU)/mL are necessary to have a positive smear.62 The Ziehl-Neelson stain is most commonly used and is recommended by the WHO. This smear is found to be positive in only 50-80% of patients with later culture-confirmed diagnoses of pulmonary tuberculosis.17,62 However, the smear's utility is added to by the finding that patients with smear-positive tuberculosis are more infectious than smear-negative cases, and also present with higher case-fatality rates.17
While developing countries may rely solely on the positive smear for diagnosis of tuberculosis, the United States relies on the growth of the organism in a sputum culture.61,62 The M. tuberculosis organism can take 6 weeks or longer to grow on solid culture media, however it may grow as fast as 7-21 days in liquid culture.61,62 Both media should be plated for growth of the organism and further sensitivity testing.61,62
Serologic Tests. In recent years, serum tests for the diagnosis of LTBI and active TB have become available. The QuantiFERON-TB Gold assay is FDA-approved for use in the United States and relies on ELISA-analysis of whole blood.63 These IFN-gamma assays have sensitivities ranging 80-95% in preliminary studies, with specificities of 95-100%.63 The principal strength of this type of test for detection of LTBI is that it has a higher specificity, less cross-reactivity with the BCG vaccine, and is more convenient as it only requires one patient visit.63 It also doesn't have the booster response that can occur with serial PPD tests.63 Given its recent introduction, further studies are needed for evaluating its efficacy in immunocompromised patients, patients with extrapulmonary TB, and in children.63 However, the CDC has recommended the use of this assay in all circumstances in which the PPD is being used.63
A serologic test for diagnosing active TB is the nucleic acid amplification (NAA) test that identifies nucleic acids of the TB complex and amplifies them.63 NAA tests have high specificity for pulmonary and extrapulmonary TB.63 However, these tests have low sensitivity, especially in smear-negative pulmonary and extrapulmonary TB cases.63 Thus, the current recommendation is that NAA tests should not replace culture or smears, but could be used to rule in, or confirm, a diagnosis of TB.63 They should be used as a confirmatory test in patients with a positive sputum smear, but not to rule out the diagnosis of TB.63
Miliary TB. Epidemiology. Miliary tuberculosis is a term that encompasses any disseminated manifestation or presentation of tuberculosis. Miliary TB represents 1-2% of all cases of tuberculosis, and 8% of cases of extrapulmonary tuberculosis in immunocompetent patients.17 In immunocompromised patients, miliary tuberculosis occurs with significantly higher rates, reported as high as 38% in HIV-positive individuals.17 Patients with any compromise of cell-mediated immunity are at increased risk of developing miliary tuberculosis.17 While initially the major age groups at higher risk of developing miliary tuberculosis were children and elderly adults, the rise of conditions causing immune compromise in young adults has altered the epidemiology of this form of TB.17 These conditions include: HIV infection, alcohol abuse, malignancy, connective tissue disease, renal failure, diabetes, pregnancy, corticosteroid use, and solid organ or bone marrow transplant patients.17 The mortality rate in children of miliary tuberculosis is 15-20%, while in adults it is 25-30%.17 The major predictors of a poor outcome are delays in diagnosing miliary tuberculosis and delays in initiating treatment.17
Pathogenesis. Miliary TB can occur either during primary progressive disease or as post-primary progressive disease.2 Miliary TB is caused by massive lymphohematogenous dissemination of Mycobacterium tuberculosis from a pulmonary or extrapulmonary focus, spreading primarily to highly vascular organs such as the lungs, bone, or brain.17 The initial nomenclature came from the appearance of the whitish nodule found on the surface of the affected lung, similar to millet seeds.17 This term is now used to refer to any disseminated tuberculosis infection, even if classic pathologic or radiographic evidence is lacking.
Presentation. Clinically, patients present with systemic symptoms such as fever, weight loss, and cough, along with signs and symptoms specific to the site of dissemination.17
Patients with pulmonary miliary TB can present with cough, dyspnea, or pleuritic chest pain.17 On exam, hypoxia and rales are common.17 Cases of Acute Respiratory Distress Syndrome (ARDS) have also been reported.17
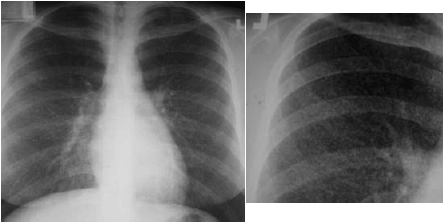
Diagnostic Imaging. On chest x-ray, a typical miliary pattern of pulmonary opacities measuring 2 mm or less may be seen.17 The reticulonodular pattern of miliary disease may be seen on either chest x-ray or CT.17 (See Figure 5.) These findings in the setting of a patient with concerning systemic symptoms strongly suggest a diagnosis of miliary TB. Similar chest CT findings can be seen in patients with infections due to Mycoplasma pneumoniae, Haemophilus influenzae, or in noninfectious disease processes such as lymphoma, sarcoidosis, or other pneumoconiosis.17
| Figure 5. Miliary TB on CXR |
|
|
| Used with permission from: Highland Hospital Radiology Teaching File. |
Diagnostic Tests. Patients with miliary tuberculosis are often unable to mount a sufficient immune response to cause PPD conversion.17 Diagnosis rests on sputum for smear and culture. Bronchoalvelolar lavage (BAL) can be considered if sputum cultures are not available or if clinical index is high and cultures are negative.17
Extrapulmonary TB: CNS Tuberculosis. Epidemiology. CNS infection with tuberculosis results in either meningitis or in the formation of an intracranial space-occupying lesion known as a tuberculoma.1 In the United States, the dominant form of CNS infection is meningitis with approximately 300-400 cases of tuberculous meningitis occurring per year.18,64 This represents approximately 1% of all clinical TB cases. Tuberculous meningitis is most commonly seen following primary infection in children or infants, or alternatively following reactivation disease in immunocompromised adults.1 The case fatality rate of tuberculous meningitis is 15-40% despite aggressive treatment.18,64 Early multi-drug therapy is key to the prevention of severe morbidity and mortality from this disease.
Pathogenesis. Meningitis develops after the activation of a tubercle either during primary progressive disease or during post-primary progressive disease. The activated tubercle ruptures and spills its tubercular protein into the subarachnoid space.65 The irritant effect of the protein in the subarachnoid space produces an inflammatory reaction that can manifest as either a proliferative arachnoiditis, intracranial vasculitis with subsequent thrombosis or infarction, or communicating hydrocephalus due to inflammatory changes in the basilar cisterns impeding cerebrospinal fluid (CSF) reabsorption.65,66
Presentation. Clinically, the signs and symptoms of tuberculous meningitis can be divided into three phases.67 In the prodromal phase, headache, low grade fever, malaise, and personality change are common. In the meningitic phase, classic meningismus, vomiting, confusion, severe headache, and potentially cranial nerve or other focal neurological deficits may be seen.67 The third phase, known as the paralytic phase, is the most dangerous as seizures, coma, hemiparesis, and complete confusion may occur.67 If considering meningeal TB, a fundoscopic exam should be performed in the ED to check for choroidal lesions.67
Diagnostic Imaging. CT scans with contrast of the head may show basilar meningeal enhancement.68 This finding, accompanied by any degree of hydrocephalus, strongly suggests a diagnosis of tuberculous meningitis.65,69 In the prodromal stage, the CT scan is normal in approximately 30% of patients.69 MRI is superior to CT, especially for evaluation of the basal ganglia, midbrain, and remainder of the brainstem.69 If possible, a non-contrast head CT should be obtained prior to the lumbar puncture to rule out significant hydrocephalus.69
Diagnostic Tests. Diagnosis is by CSF analysis, culture, and AFB smear.2 CSF fluid will most often show an elevated protein (100-500 mg/dL) and low glucose levels (less than 46 mg/dL in 80% of cases), with a lymphocytic pleocytosis (between 100 and 500 cells/microLiter).2,67,69,70 Early in the stage of disease, however, the cellular reaction may lack the leukocytosis, or present with a polymorphonuclear leukocyte (PMN) predominance. A PMN predominance may also occur after initiation of drug therapy for tuberculosis.2,67,69,70
Repeat CSF cultures are key to the diagnosis of TB meningitis.69 It is recommended that at least three lumbar punctures be performed at daily intervals to increase diagnostic yield.69 The repeat CSF specimens are of value even if antibiotic therapy has been initiated. In one series, 37% of cases were diagnosed based on an initial positive AFB smear.69 However, after four serial specimens, even after administration of anti-tuberculous drugs, the diagnostic yield increased to 87%.69 The yield on the CSF is dramatically improved if a large volume (10-15 cc) is removed, and if the last fluid from the lumbar puncture is used.67,69,71
Treatment. Treatment should be prompt and should not await the results of cultures or smears if the patient condition is critical or if there is strong suspicion for the disease, especially given that the yield of CSF AFB staining and culture is low. Often, treatment is initiated empirically in patients with CSF pleocytosis, elevated protein, and concomitant risk factors for TB without another diagnosis seeming more evident (see Treatment section).
Tuberculous Lymphadenitis. Epidemiology. In the United States, lymphatic tuberculosis accounts for 31% of all cases of extrapulmonary tuberculosis.72 TB lymphadenitis presents with a median age of approximately 40 years, and the age of onset ranging from 20-40 years.73-76 Additionally, TB lymphadenopathy more commonly occurs in Asian Pacific Islanders and in women.72,77 Relapse rates of 3.5% are reported after treatment.78 Residual palpable lymphadenopathy may persist even after treatment.78
Pathogenesis. In non-HIV or otherwise immunocompromised patients, TB lymphadenopathy is often secondary to reactivation of disease at a site seeded hematogenously during primary infection. Cervical TB lymphadenitis is also possible secondary to primary tonsillar or adenoid infection.2,18,79
Presentation. In HIV-infected patients, TB lymphadenitis more frequently presents with systemic symptoms such as fever. In an immunocompetent individual, chronic nontender lymphadenopathy is observed with the absence of systemic symptoms.72-74 Scrofula, or the involvement of cervical lymph nodes, is found in 63-77% of cases.73,74 The anterior or posterior cervical triangles are more commonly involved, but submandibular and supraclavicular lymph nodes can also be affected.80-83 Multiple nodes at an affected site are often involved, but bilateral disease is uncommon. Infection of axillary, inguinal, mesenteric, mediastinal, and intramammary nodes has been reported.84-86 Mediastinal lymphadenopathy resulting in dysphagia, esophageal perforation, recurrent laryngeal nerve involvement with ensuing vocal cord paralysis, and pulmonary artery occlusion have also been observed.87-90 In the abdominal cavity, the periportal lymph nodes are more commonly involved, followed by mesenteric and peripancreatic lymph nodes.85 Lymph node compression of renal arteries has been reported to cause renovascular hypertension, while hepatic lymphadenitis has resulted in jaundice, portal vein thrombosis, and portal hypertension.91,92
Diagnostic Imaging. Radiographically, chest x-rays are often unremarkable in HIV-negative patients. In HIV-positive patients, the chest x-ray may show an abnormality such as evidence of active pulmonary TB or pleural thickening and fibrosis suggestive of prior TB disease.81,84,93 Ultrasound, CT with contrast, and MRI are newer imaging modalities for tuberculous lymphadenitis.
Diagnostic Tests. The standard for diagnosis of tuberculous lymphadenitis is fine needle aspiration (FNA), culture, and histology of an enlarged lymph node.80,82 FNA fluid should be sent for PCR testing (if available) in addition to culture, cytology, and microscopy. If excisional biopsy is performed, caseating granulomas suggest TB, but are not specific for this condition.80,82 Bronchoscopy or mediastinoscopy can also be performed.
Skeletal TB. Epidemiology. Tuberculous infection of the bones and joints accounts for 10-35% of cases of extrapulmonary tuberculosis.79,94-96 Of these cases, half are musculoskeletal disease involving the spine.97 Prognosis depends on the stage at which the antibiotic treatment is initiated and the clinical pattern of neurological involvement.
Pathogenesis. In the United States, bony TB more commonly occurs as part of reactivation disease. Primary tuberculosis infection seeds organisms to the bone and synovium.72,94 Vertebral bodies, due to their high blood flow, are vulnerable to this seeding even in adulthood. Bony TB can also occur from contiguous spread from an infected site. Rarely, an apical pulmonary focus of TB can invade the joint between the first and second cervical vertebrae, thereby causing atlantoaxial TB.98
Spinal TB, or Pott's disease, commonly affects the lumbar and lower thoracic region.99,100 Infection starts in the anteroinferior aspect of the vertebral body and then spreads along the anterior ligament to the adjacent vertebral body.12 Inflammatory bony destruction and necrosis can collapse the vertebral body and cause disc herniation.12 Severe bony deformities can compress the spinal cord.12 Possible complications of Pott's disease include development of a spinal or epidural abscess, cord compression, and resultant paraplegia known as Pott's paraplegia.101,102
The next most common skeletal tuberculosis conditions are tuberculous arthritis, which is usually monoarticular (classically affecting the hip or knee), and extraspinal tuberculous osteomyelitis.103 In tuberculous arthritis, granulomatous changes cause erosion of the cartilage and joint effusions. Classic arthritic changes including joint deformities can occur. In extraspinal tuberculous disease, almost any bone can be infected, from the sternum to the ribs to the symphysis pubis. Poncet's disease is a rare acute symmetric polyarthritis involving large and small joints and is associated with active pulmonary, extrapulmonary or miliary TB.104,105 In Poncet's disease, there is no evidence of active infection of the involved joints.106 This arthritis resolves after anti-tuberculosis drug therapy is initiated.
Presentation. Clinically, patients with spinal TB will present with focal pain, muscle spasm, and rigidity. As described in a classic text, the gait is "aldermanic" in nature, referring to the patient taking "short deliberate steps to avoid any jarring of the spine."107 The patient also maintains an upright erect posture for the same reason. Systemic symptoms are present in fewer than 40% of cases.107
Patients with articular TB will present with swelling, pain, and loss of function in the affected joint over a period of weeks to months.94 The hip joint is most frequently affected.94 "Night cries" often are reported, which refers to the pain accompanying unguarded movement of the joint at night, when relaxation of the muscle spasm has occurred.94 Systemic symptoms occur in only 30% of patients with tuberculous arthritis.108
Patients with extraspinal TB usually present with pain over the affected bony site, however presentations of painless masses that have been found to contain tuberculosis are also documented.109-110
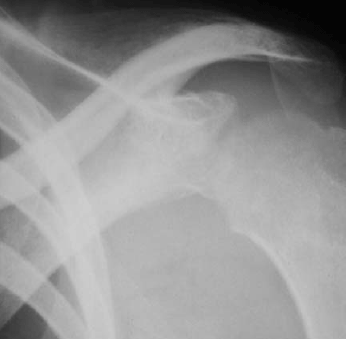
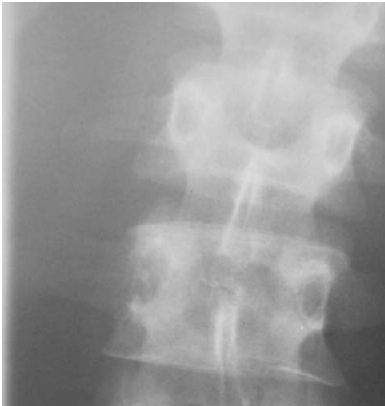
Diagnostic Imaging. Radiographically, there are no pathognomonic changes associated with musculoskeletal TB. Evidence of soft-tissue swelling, osteopenia, and bony destruction can be seen, followed by soft-tissue calcification, structural collapse, or sclerotic changes with later disease.100,107,111 Disc spaces can be obliterated. Osteolytic lesions in the absence of disc space involvement can be seen. In tuberculous arthritis, Phemister's triad consisting of osteopenia, peripherally located osseous erosions, and gradual narrowing of the disc space can be seen.100,107,111 MRI of the affected limb can be valuable in delineating soft-tissue extension and spinal cord compression, while CT of the affected limb is useful for determining extent of involvement.112 (See Figures 6, 7.)
| Figure 6. Tuberculous Arthritis in the Left Shoulder, Evidenced by Bony Changes |
|
| Used with permission from: Highland Hospital Radiology Teaching File. |
| Figure 7. Tuberculosis of the Spine as Seen on Plain Film |
|
| Used with permission from: Highland Hospital Radiology Teaching File. |
Diagnostic Tests. Needle aspiration, biopsy, and culture of the infected area is the standard for diagnosis.12 Synovial biopsy and culture for TB is also recommended.12
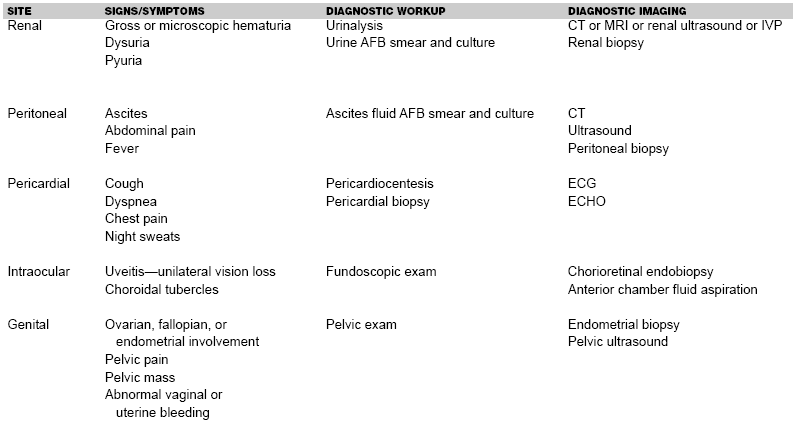
Other Extrapulmonary TB Manifestations. TB can infect almost every organ system of the body. (See Table 2.)
| Table 2. Uncommon Extrapulmonary Manifestations of TB, Presenting Signs and Symptoms, and Initial Recommended Diagnostic Workup and Imaging113-152 |
|
|
ED Management
ED Management of a Positive PPD. The PPD is an advanced version of the initial tuberculin test and is performed by precipitating protein components from culture filtrate of M. tuberculosis.
In the United States, the PPD is employed mainly in the annual screening of asymptomatic high-risk individuals. High-risk individuals include those with close contact with persons with active TB (including health care workers, prison guards, laboratory personnel), as well as those with a medical condition that may confer a heightened risk of developing TB.153 These conditions include immunosuppressive conditions including diabetes mellitus, long-term steroid therapy, malignancies, hemodialysis, solid organ transplant recipients, as well as any state of malnutrition.12,153 Residents of long-term care facilities, either correctional or nursing facilities, as well as low-income, injection drug using, or homeless patients should also be screened regularly.
A PPD may be appropriate in patients who have either had a known exposure to tuberculosis in a close contact or relative, or in the case of recent immigration or refugee status from a TB-endemic country.12
If a person has been infected with M. tuberculosis, he will mount a cell-mediated delayed type hypersensitivity (DTH) response, mediated by T lymphocytes, to the purified protein.1 T cells and other inflammatory cells infiltrate the area of the PPD placement, producing maximal induration at 48-72 hours following placement.1 The patient's time from exposure should be 2-8 weeks to mount an effective PPD conversion response.1
A PPD should be read 48-72 hours after placement to detect the maximal immune reaction. However, a positive PPD at 96 hours or more can still be recorded as positive.1 A negative PPD at greater than 72 hours should not be trusted as a true negative.11
Most individuals who have received the BCG vaccine will have a 3-19 mm reaction to the tuberculin skin test at 2-3 months following the administration of the vaccine.24 This reaction will diminish over time and will usually not persist beyond 10 years from the date of vaccination.24
A chest x-ray should be obtained in all patients with a positive PPD to rule out active disease. If the chest radiograph is negative, and the patient is asymptomatic, the patient should be referred to a primary medical doctor for initiation of antibiotic therapy. If the patient has HIV risk factors, testing for HIV is also recommended. If the chest x-ray suggests active TB, or the patient has symptoms suggestive of TB, the patient should be admitted to confirm the diagnosis, treatment should be initiated, and the patients should be isolated until he is no longer infectious to others.153
This false negative PPD reaction can be secondary to: cytokines initiated during active disease that inhibit the delayed hypersensitivity immune response; anergy due to another condition causing immune compromise; severe TB (i.e., disseminated TB); other viral infection; other corticosteroid therapy; or poor nutrition.154
| Table 3. Correlation of PPD Reaction in Millimeters with Patient Risk Category12,153 |
|
|
ED Management of Suspected Active TB. Patients present with symptoms of pulmonary disease (i.e., dyspnea, cough, hemoptysis) only 33% of the time, while medical, but nonpulmonary (i.e., chest pain, abdominal pain, or vomiting) complaints, are present in 41% of cases. Still other studies have examined a range of screening questionnaires to identify and isolate patients with possible active pulmonary TB.155 Predictably, protocols with the broadest criteria do result in identification of a higher percentage of patients with active pulmonary TB, but use resources such as respiratory isolation rooms with greater frequency than is feasible at most hospitals.156 Thus, the issue of appropriate, effective, and cost-effective screening of patients with active pulmonary TB needs further attention and investigation.
The involvement of the ED practitioner with the management of suspected active tuberculosis begins with a thorough screening effort. In adults, this screening is done by evaluating a focused review of systems (ROS), ideally at the triage station. An assessment of risk factors should be performed by the primary provider. On ROS, concerning symptoms are history of cough, fever, night sweats, and weight loss.
Patients with such symptoms should immediately be given a surgical mask to wear and be placed in a negative pressure isolation room.2 If a negative pressure room is unavailable, as is often the case at crowded EDs, at the bare minimum a mask should be placed on the patient and on other patients sharing a room or hallway.2 Another option is to have the patient wait outside, if their condition and the weather permit. The TB risk factors usually are elicited after a more in-depth discussion with the patient and include: a recent contact with a TB patient; a history of TB; HIV disease; homelessness; shelter or skilled nursing facility or other group/congregational residence; IVDU (intravenous drug use); alcohol use/abuse; having been born in a foreign country; renal failure; diabetes mellitus.157
ED Management: The following management algorithm should be applied:
1. Respiratory isolation in a negative pressure room to prevent spread of organisms outside of the room.2 If patients have to leave the room for imaging, they should wear a surgical mask during transport.2
2. Imaging as appropriate. Any time extrapulmonary TB is suspected, pulmonary TB must be ruled out. Therefore, obtain screening chest radiographs and/or CT scans to rule out pulmonary involvement as part of the workup for extrapulmonary TB.
3. Physicians and nurses caring for the patient should wear N95 disposable respirators/masks.2
4. Initiate antibiotic therapy in cases where severe conditions such as miliary or meningeal TB are suspected. Unless resistant organisms are suspected or known, a standard first-line regimen should be used.2
5. In cases where patients are boarded in the ED for a long period of time, sputum for smear and other diagnostic cultures should be pursued. Serologic studies, as available, should also be sent.
6. Admit the patient for a full workup of extrapulmonary TB and/or workup of pulmonary TB. Treatment can be initiated following a positive smear, or even prior to results of the smear if the clinical and radiographic evidence is high.2 Ideally, all anti-tuberculous therapy should be started only in conjunction with infectious disease consultation, especially in patients who are HIV-positive, for whom the anti-retroviral medication must be coordinated with the anti-tuberculous treatment.2
7. All cases of tuberculosis must be reported to the Public Health Department.2
8. Health care workers who have known exposure to a patient with active TB should be PPD tested within a few months of exposure. There is no role for antibiotic prophylaxis for TB.
Treatment
Treatment is usually started after a definitive diagnosis of TB. Isolation is recommended until at least 2 weeks of therapy have been completed; however, if a patient has multiple drug resistant (MDR) TB, isolation should only end when the AFB smears are negative. For pulmonary TB, the complete regimen of therapy should utilize four drugs for the initial two months, followed by 18 weeks of continuation therapy with a modified regimen of isoniazid and rifampin. Patients with cavitary lesions at the time of diagnosis, positive sputum cultures at the end of the first two months of treatment, or those who did not receive pyrazinamide initially, should have 31 weeks of continuation therapy instead of the usual 18 weeks.
Pulmonary TB. Active Pulmonary TB. The treatment course for active pulmonary TB is divided into an "initiation phase" and a "continuation phase" of drug therapy.153 The initiation phase of treatment for active pulmonary TB is a four-drug regimen known as the RIPE regimen, of rifampin (RIF), isoniazid (INH), pyrazinamide (Pyr), and ethambutol (EMB).153 This regimen can be started before the results of cultures or smears are received, especially in patients for whom there is a high clinical suspicion for active tuberculosis or who are critically ill.153 The drug regimen should be modified after 2 months of initial treatment based on drug susceptibilities of the organism. The management of HIV-positive patients in the initiation phase of therapy does not differ from treatment of HIV-negative patients.153
Latent TB Infection. Latent TB infection should be managed with 9 months of daily INH therapy, with pyridoxine supplementation.158 No clinical trial has been done to compare 9 months of INH to 6 months of INH.158 Pyridoxine supplementation is given along with INH therapy because INH results in depletion of vitamin B6.159 Pyridoxine deficiency results in primarily a distal sensory neuropathy that is effectively prevented with daily supplementation.159 The management of LTBI is the same regardless of HIV status.158
Extrapulmonary TB. All forms of extrapulmonary TB should be managed with an initial 6- to 9-month regimen of anti-tuberculous therapy, provided that the initial regimen includes isoniazid and rifampin.160 In the first two months, the RIPE regimen (defined above) should be given.2 After the first two months, if cultures demonstrate adequate susceptibility, only the INH and RIF should be continued for the remaining 4-7 months.2
In cases of CNS tuberculosis, extended therapy for 9-12 months is recommended, though not supported by any randomized clinical trial.2,160 Extended therapy frequently is given in cases of bony or joint infection with tuberculosis or in cases of drug resistance.2 Steroids are included in the treatment of tuberculous meningitis.161
Close consultation with an infectious disease specialist for all treatment regimens in HIV-positive patients is recommended due to potential interactions between anti-retroviral and anti-tuberculous drugs.160 However, the current recommendation is that the treatment of extrapulmonary TB in HIV-positive patients is the same as in HIV-negative patients.160
Multi-Drug Resistant Tuberculosis (MDR-TB). MDR-TB refers to strains of Mycobacterium tuberculosis that are resistant to isoniazid and rifampin.162 MDR-TB is a global problem with an estimated prevalence of 1.1% in newly diagnosed patients.162 The development of MDR-TB is multi-factorial. Incomplete and inadequate treatment is felt to be the primary factor enabling the development of MDR-TB.162 Genetic factors of the organism, patient non-compliance, poor infection control in hospitals, and poorly supervised treatment programs are other factors contributing to the problem.162
Within the United States, the prevalence of drug-resistant tuberculosis is overall decreasing due to greater awareness, a more standardized approach to treatment (i.e., with multiple drugs, sputum for culture and sensitivities) programs to improve compliance, and infection control.163 However, despite the overall decrease, there are still areas of the nation where MDR-TB is a significant threat. Specifically, in California the percentage of MDR-TB cases (1.4%) has not changed between 1994 and 2003.163 These figures are evidence for the need for ongoing awareness regarding MDR-TB within the United States and the importance of initiating and ensuring compliance with adequate treatment regimens for tuberculosis.
Treatment Complications
Hepatitis. Use of INH, Rif, or Pyr can cause drug-induced hepatitis at any point during treatment.164 The drug-induced hepatitis may be asymptomatic or symptomatic. In asymptomatic patients, this is defined as having a serum aspartate transaminase (AST) level of 5 times the upper limit of normal.153 In a symptomatic patient (most commonly presenting with abdominal pain, nausea, vomiting), the condition is defined as having an AST of 3 times the upper limit of normal.153 If a patient presents to the ED with a significant elevation of AST, the medication should be discontinued.
The discontinuation of the hepatotoxic drug and the initiation of a new regimen ideally should be done in consultation with the patient's primary medical doctor; however, the discontinuation of the offending agent can be initiated by the ED provider.
If the emergency physician is unable to contact the primary physician, it is prudent to stop all anti-tuberculous drugs. The patient should follow up with his or her doctor within one week to have the LFTs rechecked. Reintroduction or redesign of their anti-TB regimen can then be done by the primary medical doctor.
Medication Side Effects. The drugs used for first-line treatment of tuberculosis can cause a range of side effects. Full review is beyond the scope of this paper. The most relevant and common reactions are covered here.
Gastrointestinal (GI) Symptoms. Nausea, vomiting, and decreased appetite can occur in the absence of elevated LFTs. All anti-tuberculosis drugs can cause GI upset.164 Taking the medication with food can alleviate the majority of non-hepatitis GI upset.
Rash. All anti-tuberculosis drugs can cause a rash, ranging from a minor pruritus to a petechial rash.164 Minor pruritus is treated with an antihistamine. A generalized erythematous rash with any mucous membrane involvement is more serious and all drugs should be stopped.164 If the rash is petechial in nature, the patient's platelet count should be checked to ensure the rash is not due to thrombocytopenia, likely induced by RIF.164 If the rash resolves after discontinuing the medications, the medications can be restarted one at a time, a few days apart.164 RIF should always be the first one to be restarted, then INH, and then EMB or PZA.164 If three drugs can be restarted without any rash recurring, the fourth drug should be held unless it is key to the therapy.164
Drug-Drug Interactions. Emergency medicine providers must be aware of some of the key interactions between anti-tuberculosis drugs and other drugs such as coumadin or anti-retrovirals. The rifamycins, including raifampin, rifabutin, and rifapentine, induce the cytochrome p450 system, and can thereby decrease the circulating concentrations of several other drugs such as coumadin164 and anti-retrovirals, specifically the protease inhibitors (PIs), and the non-nucleoside reverse transcriptase inhibitors (NNRTIs).164 INH also inhibits the p450 cytochrome system, and will increase the concentrations of phenytoin, carbamazepine, diazepam, acetaminophen, warfarin, or disulfiram.164 These drug-drug interactions should be considered when evaluating a patient on any of these medications.
Conclusion
Emergency medicine providers must be able to identify patients at risk of TB and understand the diagnostic tests needed, as well as the initial management of these patients. The clinical suspicion for both pulmonary and extrapulmonary manifestations of the disease must remain high, especially in the setting of the immunocompromised patient. Patients suspected of TB should be placed in respiratory isolation with health workers taking respiratory precautions as well. Patients should be admitted and diagnosis of both pulmonary and extrapulmonary TB should be aggressively pursued to prevent further transmission. Latent infections detected via the PPD test should be treated to prevent reactivation disease later in life. The pediatric patient should also be thoroughly evaluated for tuberculosis infection, because sputum smears and cultures are often negative. Treatment should be broad-spectrum initially and later tailored based on infectious disease specialist recommendations and culture and sensitivities. Patient and family education regarding this condition is key to the control of its spread. In the coming years, improved programs for testing, diagnosis, treatment, and follow-up of patients with tuberculosis will hopefully aid in the control of this epidemic.
References
1. Frieden TR, Sterling TR, Munsiff SS, et al. Tuberculosis. Lancet 2003;362:887-899.
2. Golden MP, Vikram HR. Extrapulmonary tuberculosis: An overview. Am Fam Physician 2005;72:1761-1768.
3. Raviglione MC, Snider DE Jr., Kochi A. Global epidemiology of tuberculosis. Morbidity and mortality of a worldwide epidemic. JAMA 1995;273:220-226.
4. Leung AN. Pulmonary tuberculosis: The essentials. Radiology 1999;210:307-322.
5. Cantwell MF, Snider DE Jr, Cauthen GM, et al. Epidemiology of tuberculosis in the United States, 1985 through 1992. JAMA 1994;272:535-539.
6. Murray JF. A century of tuberculosis. Am J Respir Crit Care Med 2004;169:1181-1186.
7. Trends in tuberculosis—United States, 2005. MMWR Morb Mortal Wkly Rep 2006;55:305-308.
8. Selwyn PA, Hartel D, Lewis VA, et al. A prospective study of the risk of tuberculosis among intravenous drug users with human immunodeficiency virus infection. N Engl J Med 1989;320:545-550.
9. McKenna MT, McCray E, Onorato I. The epidemiology of tuberculosis among foreign-born persons in the United States, 1986 to 1993. N Engl J Med 1995;332:1071-1076.
10. Talbot EA, Moore M, McCray E, et al. Tuberculosis among foreign-born persons in the United States, 1993-1998. JAMA 2000;284:2894-2900.
11. Latent Tuberculosis Infection-A guide for San Francisco Providers. Prepared by the San Francisco Department of Public Health, M.A.a.h.w.s.o.p.t.L.
12. Targeted tuberculin testing and treatment of latent tuberculosis infection. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. This is a Joint Statement of the American Thoracic Society (ATS) and the Centers for Disease Control and Prevention (CDC). This statement was endorsed by the Council of the Infectious Diseases Society of America. (IDSA), September 1999, and the sections of this statement. Am J Respir Crit Care Med 2000;161(4 Pt 2):S221-S247.
13. Jick SS, Lieberman ES, Rahman MU, et al. Glucocorticoid use, other associated factors, and the risk of tuberculosis. Arthritis Rheum 2006;55:19-26.
14. Dannenberg AM, Jr. Immune mechanisms in the pathogenesis of pulmonary tuberculosis. Rev Infect Dis 1989;11 Suppl 2:S369-S378.
15. Smith DW, Wiegeshaus EH. What animal models can teach us about the pathogenesis of tuberculosis in humans. Rev Infect Dis 1989;11 Suppl 2:S385-S393.
16. Riley RL. Airborne infection. Am J Med 1974;57:466-475.
17. Sharma SK, Mohan A, Sharma A, et al. Miliary tuberculosis: New insights into an old disease. Lancet Infect Dis 2005;5:415-430.
18. Farer LS, Lowell AM, Meador MP. Extrapulmonary tuberculosis in the United States. Am J Epidemiol 1979;109:205-217.
19. Small PM, Schecter GF, Goodman PC, et al. Treatment of tuberculosis in patients with advanced human immunodeficiency virus infection. N Engl J Med 1991;324:289-294.
20. Nelson LJ, Wells CD. Global epidemiology of childhood tuberculosis. Int J Tuberc Lung Dis 2004;8:636-647.
21. Skeiky YA, Sadoff JC. Advances in tuberculosis vaccine strategies. Nat Rev Microbiol 2006;4:469-476.
22. Clemens JD, Chuong JJ, Feinstein AR. The BCG controversy. A methodological and statistical reappraisal. JAMA 1983;249:2362-2369.
23. Rodrigues LC, Diwan VK, Wheeler JG. Protective effect of BCG against tuberculous meningitis and miliary tuberculosis: A meta-analysis. Int J Epidemiol 1993;22:1154-1158.
24. Trunz BB, Fine P, Dye C. Effect of BCG vaccination on childhood tuberculous meningitis and miliary tuberculosis worldwide: A meta-analysis and assessment of cost-effectiveness. Lancet 2006;367:1173-1180.
25. FitzGerald JM. Management of adverse reactions to bacille Calmette-Guerin vaccine. Clin Infect Dis 2000;31 Suppl 3:S75-S76.
26. Jouanguy E, Altare F, Lamhamedi S, et al. Interferon-gamma-receptor deficiency in an infant with fatal bacille Calmette-Guerin infection. N Engl J Med 1996;335:1956-1961.
27. Lallemant-Le Coeur S, Lallemant M, Cheynier D, et al. Bacillus Calmette-Guerin immunization in infants born to HIV-1-seropositive mothers. AIDS 1991;5:195-199.
28. Brost BC, Newman RB. The maternal and fetal effects of tuberculosis therapy. Obstet Gynecol Clin North Am 1997;24:659-673.
29. Snider DE Jr., Caras GJ. Isoniazid-associated hepatitis deaths: A review of available information. Am Rev Respir Dis 1992;145(2 Pt 1):494-497.
30. Snider DE Jr., Powell KE. Should women taking antituberculosis drugs breast-feed? Arch Intern Med 1984;144:589-590.
31. Snider DE Jr., Layde PM, Johnson MW, et al. Treatment of tuberculosis during pregnancy. Am Rev Respir Dis 1980;122:65-79.
32. Prevention and treatment of tuberculosis among patients infected with human immunodeficiency virus: Principles of therapy and revised recommendations. Centers for Disease Control and Prevention. MMWR Recomm Rep 1998;47(RR-20):1-58.
33. Ormerod P. Tuberculosis in pregnancy and the puerperium. Thorax 2001;56:494-499.
34. Jana N, Vasishta K, Saha SC, et al. Obstetrical outcomes among women with extrapulmonary tuberculosis. N Engl J Med 1999;341:645-649.
35. Adhikari M, Pillay T, Pillay DG. Tuberculosis in the newborn: An emerging disease. Pediatr Infect Dis J 1997;16:1108-1112.
36. Curvo-Semedo L, Teixeira L, Caseiro-Alves F. Tuberculosis of the chest. Eur J Radiol 2005;55:158-172.
37. Arango L, Brewin AW, Murray JF. The spectrum of tuberculosis as currently seen in a metropolitan hospital. Am Rev Respir Dis 1973;108:805-812.
38. Kiblawi SS, Jay SJ, Stonehill RB, et al. Fever response of patients on therapy for pulmonary tuberculosis. Am Rev Respir Dis 1981;123:20-24.
39. Cameron SJ. Tuberculosis and the blood—a special relationship? Tubercle 1974;55:55-72.
40. Corr WP Jr., Kyle RA, Bowie EJ. Hematologic changes in tuberculosis. Am J Med Sci 1964;248:709-714.
41. Chung DK, Hubbard WW. Hyponatremia in untreated active pulmonary tuberculosis. Am Rev Respir Dis 1969;99:595-597.
42. Vorherr H, Massry SG, Fallet R, et al. Antidiuretic principle in tuberculous lung tissue of a patient with pulmonary tuberculosis and hyponatremia. Ann Intern Med 1970;72:383-387.
43. Johnston H, Reisz G. Changing spectrum of hemoptysis. Underlying causes in 148 patients undergoing diagnostic flexible fiberoptic bronchoscopy. Arch Intern Med 1989;149:1666-1668.
44. McGuinness G, Beacher JR, Harkin TJ, et al. Hemoptysis: Prospective high-resolution CT/bronchoscopic correlation. Chest 1994;105:1155-1162.
45. Conlan AA, Hurwitz SS, Krige L, et al. Massive hemoptysis. Review of 123 cases. J Thorac Cardiovasc Surg 1983;85:120-124.
46. Ihm HJ, Hankins JR, Miller JE, et al. Pneumothorax associated with pulmonary tuberculosis. J Thorac Cardiovasc Surg 1972;64:211-219.
47. Wilder RJ, Beacham EG, Ravitch MM. Spontaneous pneumothorax complicating cavitary tuberculosis. J Thorac Cardiovasc Surg 1962;43:561-573.
48. Bobrowitz ID, Rodescu D, Marcus H, et al. The destroyed tuberculous lung. Scand J Respir Dis 1974;55:82-88.
49. Palmer PE. Pulmonary tuberculosis—usual and unusual radiographic presentations. Semin Roentgenol 1979;14:204-243.
50. Khan FA, Rehman M, Marcus P, et al. Pulmonary gangrene occurring as a complication of pulmonary tuberculosis. Chest 1980;77:76-80.
51. Marciniuk DD, McNab BD, Martin WT, et al. Detection of pulmonary tuberculosis in patients with a normal chest radiograph. Chest 1999;115:445-452.
52. Poulsen A. Some clinical features of tuberculosis. Acta Tuberc Scand 1957;33:37-92; concl.
53. Barnes PF, Verdegem TD, Vachon LA, et al. Chest roentgenogram in pulmonary tuberculosis. New data on an old test. Chest 1988;94:316-320.
54. Poppius H, Thomander K. Segmentary distribution of cavities: A radiologic study of 500 consecutive cases of cavernous pulmonary tuberculosis. Ann Med Intern Fenn 1957;46:113-119.
55. Farman DP, Speir WA Jr. Initial roentgenographic manifestations of bacteriologically proven Mycobacterium tuberculosis. Typical or atypical? Chest 1986;89:75-77.
56. Lentino W, Jacobson HG, Poppel MH. Segmental localization of upper lobe tuberculosis: The rarity of anterior involvement. Am J Roentgenol Radium Ther Nucl Med 1957;77:1042-1047.
57. Choyke PL, Sostman HD, Certis AM, et al. Adult-onset pulmonary tuberculosis. Radiology 1983;148:357-362.
58. Miller WT, MacGregor RR. Tuberculosis: Frequency of unusual radiographic findings. AJR Am J Roentgenol 1978;130:867-875.
59. Woodring JH, Vandiviere HM, Fried AM, et al. Update: The radiographic features of pulmonary tuberculosis. AJR Am J Roentgenol 1986;146:497-506.
60. Im JG, Itoh H, Shim YS, et al. Pulmonary tuberculosis: CT findings—early active disease and sequential change with antituberculous therapy. Radiology 1993;186:653-660.
61. Shinnick TM, Good RC. Diagnostic mycobacteriology laboratory practices. Clin Infect Dis 1995;21:291-299.
62. Allen BW, Mitchison DA. Counts of viable tubercle bacilli in sputum related to smear and culture gradings. Med Lab Sci 1992;49:94-98.
63. Nahid P, Pai M, Hopewell PC. Advances in the diagnosis and treatment of tuberculosis. Proc Am Thorac Soc 2006;3:103-110.
64. Centers for Disease Control and Prevention. Tuberculosis morbidity—United States, 1997. MMWR Morb Mortal Wkly Rep 1998;47:253-257.
65. Sheller JR, Des Prez RM. CNS tuberculosis. Neurol Clin 1986;4:143-158.
66. Auerbach O. Tuberculous meningitis: Correlation of therapeutic results with the pathogenesis and pathologic changes. I. General considerations and pathogenesis. Am Rev Tuberc 1951;64:408-418.
67. Kennedy DH, Fallon RJ. Tuberculous meningitis. JAMA 1979;241:264-268.
68. Schoeman J, Hewlett R, Donald P. MR of childhood tuberculous meningitis. Neuroradiology 1988;30:473-477.
69. Leonard JM, Des Prez RM. Tuberculous meningitis. Infect Dis Clin North Am 1990;4:769-787.
70. Karandanis D, Shulman JA. Recent survey of infectious meningitis in adults: Review of laboratory findings in bacterial, tuberculous, and aseptic meningitis. South Med J 1976;69:449-457.
71. Udani PM, Dastur DK. Tuberculous encephalopathy with and without meningitis. Clinical features and pathological correlations. J Neurol Sci 1970;10:541-561.
72. Rieder HL, Snider DE Jr., Cauthen GM. Extrapulmonary tuberculosis in the United States. Am Rev Respir Dis 1990;141:347-351.
73. Geldmacher H, Taube C, Kroeger C, et al. Assessment of lymph node tuberculosis in northern Germany: A clinical review. Chest 2002;121:1177-1182.
74. Mert A, Tabak F, Ozaras R, et al. Tuberculous lymphadenopathy in adults: A review of 35 cases. Acta Chir Belg 2002;102:118-121.
75. Perlman DC, D'Amico R, Salomon N. Mycobacterial infections of the head and neck. Curr Infect Dis Rep 2001;3:233-241.
76. Wark P, Goldberg H, Ferson M, et al. Mycobacterial lymphadenitis in eastern Sydney. Aust N Z J Med 1998;28:453-458.
77. Dandapat MC, Mishra BM, Dash SP, et al. Peripheral lymph node tuberculosis: A review of 80 cases. Br J Surg 1990;77:911-912.
78. van Loenhout-Rooyackers JH, Laheij RJ, Richter C, et al. Shortening the duration of treatment for cervical tuberculous lymphadenitis. Eur Respir J 2000;15:192-195.
79. Fanning A. Tuberculosis: 6. Extrapulmonary disease. CMAJ 1999;160:1597-1603.
80. Artenstein AW, Kim JH, Williams WJ, et al. Isolated peripheral tuberculous lymphadenitis in adults: Current clinical and diagnostic issues. Clin Infect Dis 1995;20:876-882.
81. Lee KC, Tami TA, Lalwani AK, et al. Contemporary management of cervical tuberculosis. Laryngoscope 1992;102:60-64.
82. Shikhani AH, Hadi UM, Mufarrij AA, et al. Mycobacterial cervical lymphadenitis. Ear Nose Throat J 1989;68:660, 662-6666, 668-672.
83. Shriner KA, Mathisen GE, Goetz MB. Comparison of mycobacterial lymphadenitis among persons infected with human immunodeficiency virus and seronegative controls. Clin Infect Dis 1992;15:601-605.
84. Alvarez S, McCabe WR. Extrapulmonary tuberculosis revisited: A review of experience at Boston City and other hospitals. Medicine (Baltimore) 1984;63:25-55.
85. Shafer RW, Kim DS, Weiss JP, et al. Extrapulmonary tuberculosis in patients with human immunodeficiency virus infection. Medicine (Baltimore) 1991;70:384-397.
86. Yilmaz F, Yagmur Y, Uzunlar AK. Tuberculosis in an intramammary lymph node. Eur J Surg 2000;166:267-268.
87. Drake WM, Elkin SL, al-Kutoubi A, et al. Pulmonary artery occlusion by tuberculous mediastinal lymphadenopathy. Thorax 1997;52:301-302.
88. Ohtake M, Saito H, Okuno M, et al. Esophagomediastinal fistula as a complication of tuberculous mediastinal lymphadenitis. Intern Med 1996;35:984-986.
89. Popli MB. Dysphagia: A rare presentation of tuberculous mediastinal lymphadenitis. Australas Radiol 1998;42:143-145.
90. Rafay MA. Tuberculous lymphadenopathy of superior mediastinum causing vocal cord paralysis. Ann Thorac Surg 2000;70:2142-2143.
91. Caroli-Bosc FX, Conio M, Maes B, et al. Abdominal tuberculosis involving hepatic hilar lymph nodes. A cause of portal vein thrombosis and portal hypertension. J Clin Gastroenterol 1997;25:541-543.
92. Puri S, Khurana SB, Malhotra S. Tuberculous abdominal lymphadenopathy causing reversible renovascular hypertension. J Assoc Physicians India 2000;48:530-532.
93. Thompson MM, Underwood MJ, Sayers RD, et al. Peripheral tuberculous lymphadenopathy: A review of 67 cases. Br J Surg 1992;79:763-764.
94. Sharma SK, Mohan A. Extrapulmonary tuberculosis. Indian J Med Res 2004;120:316-353.
95. Teo HE, PehWC. Skeletal tuberculosis in children. Pediatr Radiol 2004;34:853-860.
96. Watts HG, Lifeso RM. Tuberculosis of bones and joints. J Bone Joint Surg Am 1996;78:288-298.
97. Vohra R, Kang HS, Dogra S, et al. Tuberculous osteomyelitis. J Bone Joint Surg Br 1997;79:562-566.
98. Lifeso R. Atlanto-axial tuberculosis in adults. J Bone Joint Surg Br 1987;69:183-187.
99. Lifeso RM, Weaver P, Harder EH. Tuberculous spondylitis in adults. J Bone Joint Surg Am 1985;67:1405-1413.
100. Weaver P, Lifeso RM. The radiological diagnosis of tuberculosis of the adult spine. Skeletal Radiol 1984;12:178-186.
101. Hsu LC, Leong JC. Tuberculosis of the lower cervical spine (C2 to C7). A report on 40 cases. J Bone Joint Surg Br 1984;66:1-5.
102. Nussbaum ES, Rockswold GL, Bergman TA, et al. Spinal tuberculosis: A diagnostic and management challenge. J Neurosurg 1995;83:243-247.
103. Kumar K, Saxena MB. Multifocal osteoarticular tuberculosis. Int Orthop 1988;12:135-138.
104. Isaacs AJ, Sturrock RD. Poncet's disease—fact or fiction? A re-appraisal of tuberculous rheumatism. Tubercle 1974;55:135-142.
105. Sood R, Wali JP, Handa R. Poncet's disease in a north Indian hospital. Trop Doct 1999;29:33-36.
106. Dall L, Long L, Stanford J. Poncet's disease: Tuberculous rheumatism. Rev Infect Dis 1989;11:105-107.
107. Chapman M, Murray RO, Stoker DJ. Tuberculosis of the bones and joints. Semin Roentgenol 1979;14:266-282.
108. Hodgson SP, Ormerod LP. Ten-year experience of bone and joint tuberculosis in Blackburn 1978-1987. J R Coll Surg Edinb 1990;35:259-262.
109. Frouge C, Miguel A, Cochan-Priollet B, et al. Breast mass due to rib tuberculosis. Eur J Radiol 1995;19:118-120.
110. Rubinstien EM, Lehmann T. Sternal osteomyelitis due to Mycobacterium tuberculosis following coronary artery bypass surgery. Clin Infect Dis 1996;23:202-203.
111. Yao DC, Sartoris DJ. Musculoskeletal tuberculosis. Radiol Clin North Am 1995;33:679-689.
112. Jung NY, Jee WH, Hak Y, et al. Discrimination of tuberculous spondylitis from pyogenic spondylitis on MRI. AJR Am J Roentgenol 2004;182:1405-1410.
113. Aclimandos WA, Kerr-Muir M. Tuberculous keratoconjunctivitis. Br J Ophthalmol 1992;76:175-176.
114. Aguado JM, Pons F, Casafont F, et al. Tuberculous peritonitis: A study comparing cirrhotic and noncirrhotic patients. J Clin Gastroenterol 1990;12:550-554.
115. al-Quorain AA, Facharzt, Satti MB, et al. Abdominal tuberculosis in Saudi Arabia: A clinicopathological study of 65 cases. Am J Gastroenterol 1993;88:75-79.
116. Beare NA, Kublin JG, Lewis DK, et al. Ocular disease in patients with tuberculosis and HIV presenting with fever in Africa. Br J Ophthalmol 2002;86:1076-1079.
117. Becker JA. Renal tuberculosis. Urol Radiol 1988;10:25-30.
118. Bloomfield SE, Mondino B, Gray GF. Scleral tuberculosis. Arch Ophthalmol 1976;94:954-956.
119. Bouza E, Merino P, Munoz P, et al. Ocular tuberculosis. A prospective study in a general hospital. Medicine (Baltimore) 1997;76:53-61.
120. Cameron J, Oesterie SN, Baldwin JC, et al. The etiologic spectrum of constrictive pericarditis. Am Heart J 1987;113(2 Pt 1):354-360.
121. Cangemi FE, Friedman AH, Josephberg R. Tuberculoma of the choroid. Ophthalmology 1980;87:252-258.
122. Chow KM, Chow VC, Hung LC, et al. Tuberculous peritonitis-associated mortality is high among patients waiting for the results of mycobacterial cultures of ascitic fluid samples. Clin Infect Dis 2002;35:409-413.
123. Chow KM, Chow VC, Szeto CC. Indication for peritoneal biopsy in tuberculous peritonitis. Am J Surg 2003;185:567-573.
124. Chung YM, Yeh TS, Shey SJ, et al. Macular subretinal neovascularization in choroidal tuberculosis. Ann Ophthalmol 1989;21:225-229.
125. Cook CD, Hainsworth M. Tuberculosis of the conjunctiva occurring in association with a neighbouring lupus vulgaris lesion. Br J Ophthalmol 1990;74:315-316.
126. Demir K, Okten A, Kaymakogh S, et al. Tuberculous peritonitis—reports of 26 cases, detailing diagnostic and therapeutic problems. Eur J Gastroenterol Hepatol 2001;13:581-585.
127. Fowler NO, Manitsas GT. Infectious pericarditis. Prog Cardiovasc Dis 1973; 16:323-336.
128. Jakubowski A, Elwood RK, Enarson DA. Clinical features of abdominal tuberculosis. J Infect Dis 1988;158:687-692.
129. Kollins SA, Hartman GW, Carr DT, et al. Roentgenographic findings in urinary tract tuberculosis. A 10 year review. Am J Roentgenol Radium Ther Nucl Med 1974;121:487-499.
130. Lattimer JK, Reilly RJ, Segawa A. The significance of the isolated positive urine culture in genitourinary tuberculosis. J Urol 1969;102:610-613.
131. Madhukar K, Bhide M, Prasad CE, et al. Tuberculosis of the lacrimal gland. J Trop Med Hyg 1991;94:150-151.
132. Marks LS, Poutasse EF. Hypertension from renal tuberculosis: Operative cure predicted by renal vein renin. J Urol 1973;109:149-151.
133. Marshall JB. Tuberculosis of the gastrointestinal tract and peritoneum. Am J Gastroenterol 1993;88:989-999.
134. McCaughan BC, Schaff HV, Piehler JM, et al. Early and late results of pericardiectomy for constrictive pericarditis. J Thorac Cardiovasc Surg 1985;89:340-350.
135. Mehta HJ, Talwalkar NC, Merchant MR, et al. Pattern of renal amyloidosis in western India. A study of 104 cases. J Assoc Physicians India 1990;38:407-410.
136. Mehta JB, Dutt A, Harvill L, et al. Epidemiology of extrapulmonary tuberculosis. A comparative analysis with pre-AIDS era. Chest 1991;99:1134-1138.
137. Mohan K, Prasad P, Banerjee AK, et al. Tubercular tarsitis. Indian J Ophthalmol 1985;33:115-116.
138. Okazawa N, Sekiya T, Tada S. Computed tomographic features of renal tuberculosis. Radiat Med 1985;3:209-213.
139. Ortbals DW, Avioli LV. Tuberculous pericarditis. Arch Intern Med 1979;139:231-234.
140. Perez Blazquez E, Montero Rodriguez M, Mendez Ramos MJ. Tuberculous choroiditis and acquired immunodeficiency syndrome. Ann Ophthalmol 1994;26:50-54.
141. Permanyer-Miralda G, Sagrista-Sauleda J, Soler-Soler J. Primary acute pericardial disease: A prospective series of 231 consecutive patients. Am J Cardiol 1985;56:623-630.
142. Pillai S, Malone TJ, Abad JC. Orbital tuberculosis. Ophthal Plast Reconstr Surg 1995;11:27-31.
143. Raina UK, Tuli D, Arora R, et al. Tubercular endophthalmitis simulating retinoblastoma. Am J Ophthalmol 2000;130:843-845.
144. Robertson R, Arnold CR. Constrictive pericarditis with particular reference to etiology. Circulation 1962;26:525-529.
145. Rosen PH, Spalton DJ, Graham EM. Intraocular tuberculosis. Eye 1990;4 (Pt 3):486-492.
146. Sagrista-Sauleda J, Merce J, Permanyer-Miralda G, et al. Clinical clues to the causes of large pericardial effusions. Am J Med 2000;109:95-101.
147. Saini JS, Mukherjee AK, Nadkarni N. Primary tuberculosis of the retina. Br J Ophthalmol 1986;70:533-535.
148. Sanai FM, Bzeizi KI. Systematic review: Tuberculous peritonitis—presenting features, diagnostic strategies and treatment. Aliment Pharmacol Ther 2005;22:685-700.
149. Simon HB, Weinstein AJ, Pasternak MS, et al. Genitourinary tuberculosis. Clinical features in a general hospital population. Am J Med 1977;63:410-420.
150. Tang LC, Cho HK, Wong Taam VC. Atypical presentation of female genital tract tuberculosis. Eur J Obstet Gynecol Reprod Biol 1984;17:355-363.
151. Tripathy SN, Tripathy SN. Infertility and pregnancy outcome in female genital tuberculosis. Int J Gynaecol Obstet 2002;76:159-163.
152. Wang HK, Hsueh PR, Hung OC, et al. Tuberculous peritonitis: Analysis of 35 cases. J Microbiol Immunol Infect 1998;31:113-118.
153. Potter B, Rindfleisch K, Kraus CK. Management of active tuberculosis. Am Fam Physician 2005;72:2225-2232.
154. Pouchot J, Grasland A, Collet C, et al. Reliability of tuberculin skin test measurement. Ann Intern Med 1997;126:210-214.
155. Sokolove PE, Lee BS, Krawczyk JA, et al. Implementation of an emergency department triage procedure for the detection and isolation of patients with active pulmonary tuberculosis. Ann Emerg Med 2000;35:327-336.
156. Gaeta TJ, Webheh W, Yazji M, et al. Respiratory isolation of patients with suspected pulmonary tuberculosis in an inner-city hospital. Acad Emerg Med 1997;4:138-141.
157. Kirsch TD, Chanmugam A, Keyl P, et al. Feasibility of an emergency department-based tuberculosis counseling and screening program. Acad Emerg Med 1999;6:224-231.
158. Dooley KE, Sterling TR. Treatment of latent tuberculosis infection: Challenges and prospects. Clin Chest Med 2005;26:313-326, vii.
159. Neuromuscular Disease Center web site. Washington University, St. Louis, MO. Toxic neuropathies: Clinical and pathological features. www.neuro.wustl.edu/neuromuscular/nother/toxic.htm#isoniazid.
160. Fuentes ZM, Caminero JA. [Controversies in the treatment of extrapulmonary tuberculosis]. Arch Bronconeumol 2006;42:194-201.
161. Thwaites GE, Nguyen DB, Nguyen HD, et al. Dexamethasone for the treatment of tuberculous meningitis in adolescents and adults. N Engl J Med 2004;351:1741-1751.
162. Sharma SK, Mohan A. Multidrug-resistant tuberculosis: A menace that threatens to destabilize tuberculosis control. Chest 2006;130:261-272.
163. Granich RM, Oh P, Lewis B, et al. Multidrug resistance among persons with tuberculosis in California, 1994-2003. JAMA 2005;293:2732-2739.
164. Blumberg HM, Burman WJ, Chaisson RE, et al. American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America: treatment of tuberculosis. Am J Respir Crit Care Med 2003;167:603-662.
Tuberculosis (TB) is the second most common infectious cause of death in adults worldwide after HIV/AIDS. Approximately one-third of the world's population, or approximately 2 billion people, are infected with Mycobacterium tuberculosis.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.