Abdominal Pain in Young Children: Intussusception and Midgut Volvulus
Abdominal Pain in Young Children: Intussusception and Midgut Volvulus
Author: Mark S. Mannenbach, MD, Assistant Professor, Department of Pediatric and Adolescent Medicine, Mayo Medical Center, Rochester, MN
Peer Reviewer: Ronald M. Perkin, MD,MA, Professor and Chairman, Department of Pediatrics, Brody School of Medicine at East Carolina University, Greenville, NC
Editor's Note: The child under 2 years of age is often the most difficult for the clinician to evaluate in the emergency setting. The inability of these children to localize pain and their accompanying stranger anxiety can often create barriers to an accurate and timely diagnosis. The relative infrequent occurrence of some of the more serious potential diagnoses can also lead to a false sense of security for many clinicians. Often the children with these more serious diagnoses have been seen by clinicians who have mistakenly felt that the child had a relatively simple problem such as constipation or a simple viral illness.
In a previous issue of Pediatric Emergency Medicine Reports (October 2005), the author specifically addressed the issues regarding appendicitis in the young child. This article reviews both intussusception and malrotation with midgut volvulus, as these diagnoses are not as common as appendicitis and are both serious, with the potential for catastrophic outcomes. The presenting clinical signs and symptoms as well as the available diagnostic modalities for these two diagnoses are reviewed.
— The Editor
Introduction
The young child who presents with vomiting with our without abdominal pain is often a challenge in an acute care setting. These children often have vague symptoms and might not even present with any apparent abdominal complaint, but instead present with an altered mental status. The differential diagnoses for these children is quite extensive and can include something as simple as otitis media to something as catastrophic as necrotic bowel secondary to malrotation with midgut volvulus.
With careful attention to the patient's history and physical exam, the clinician can focus on the more likely possibilities in this broad differential. A thorough history may require talking with several caregivers to understand the course of events leading up to the emergency department (ED) visit. Questions regarding how the patient has fed over the past several days and whether apparent pain preceded the onset of vomiting can be helpful in narrowing the possibilities. A thorough exam including a child's genitalia to look for possible hernias cannot be overemphasized. Repeated physical exams over time may be helpful to determine the location of apparent pain, pattern of pain and other problems. Often these exams are best accomplished with the child resting comfortably in the caregiver's lap or with the caregiver performing palpation of the child's abdomen.
When the clinician is unable to rule out the more serious problems after a thorough history has been taken and a complete physical exam has been done, utilization of consulting services is very important. Early involvement of colleagues in radiology and surgery can be extremely helpful to make decisions regarding a logical approach to evaluation of these young children with either abdominal pain or vomiting. The lack of availability of colleagues comfortable with these types of problems or diagnostic resources may necessitate the transfer of the child to another facility.
The clinician cannot forget to inform the caregivers of the reasons for their concerns. A frank discussion with the caregivers regarding the difficulty in establishing a diagnosis for these young children with abdominal pathology should take place early in the evaluation to gain their trust and to enlist their help in identifying any further information that could be helpful. If a young child with either abdominal pain or vomiting is discharged, indications for a repeat evaluation should be clearly outlined verbally and in written form. The clinician who evaluates the patient at a return visit should keep an open mind and again consider the complete differential, including the more serious possibilities.
Two specific diagnoses that are often difficult to make in the young child with abdominal pain with or without vomiting are intussusception and midgut volvulus with malrotation. Each of these conditions has unique features that the clinician must pay attention to in order to make a prompt diagnosis. At the same time, the reader will appreciate the considerable amount of overlap between these conditions and less serious diagnoses. The main message of this article is to emphasize the fact that the young child with abdominal pain or vomiting does not always have simple gastroenteritis.
Intussusception
A relatively common abdominal emergency in children younger than 2 years of age is intussusception. Intussusception occurs when a proximal segment of bowel invaginates or telescopes into a distal segment of bowel. Irish, et al. report that intussusception was first described by Barbette in 1674 and was first successfully treated surgically by Wilson in 1831.1
The idiopathic form of intussusception is typically found in infants between 6 and 9 months of age with a range of 3 months to 2 years of age. Ueki, et al. report an incidence of intussusception of 0.1–0.4% of live births.2 The condition is more common in males, with a ratio of 3:2. Several authors report on a seasonal incidence of this condition, with peaks in spring, summer, and the middle of winter1 and in the warmer months.3
There are also reports of a familial disposition to intussusception. Kurzbart, et al. report on two otherwise healthy families with five of 10 and two of two siblings, respectively, who presented with idiopathic ileocolic intussusception.4 The authors suggest that a greater disproportion between the size of the ileum and the ileocecal valve may account for this familial disposition, although this could not be documented.
The most common location for the development of idiopathic intussusception is at the junction of the terminal ileum and the colon leading to an ileocolic obstruction. Certainly, other locations are possible and include ileoileal, colocolic, and ileo-ileo-colic. Eighty percent of ileocolic intussusceptions have their apex in the ascending or transverse colon.5 The primary concern for any of these lesions is the development of bowel ischemia and potential bowel necrosis. Irish, et al. report as many as 10% of patients who undergo operative reduction of intussusception will require bowel resection due to bowel ischemia or necrosis.1
Inflammation and hypertrophy of Peyer's patches located in the bowel wall are the presumed lead point for most cases. The wall of the terminal ileum of infants has a rich supply of lymphoid tissue. DiFiore reports that in approximately 90% of cases, there is no obvious cause other than hyperplasia of this lymphoid tissue. A specific abnormality that serves to lead the proximal portion into the distal is found in only 5–10% of cases of intussusception. DiFiore goes on to report that before the age of 2 years, lead points occur in less than 4% of cases; after 2 years of age, a pathological lead point is found in one third of cases.5 Pathologic lead points are typically found in older children, with a Meckel's diverticulum being the most common abnormality.6 Other abnormal lead points include polyps, intestinal duplications, submucosal hemorrhage caused by Henoch-Schonlein purpura, hemangiomas, lymphomas, and lymphosarcomas. Meconium plug syndrome associated with cystic fibrosis can lead to intussusception in the neonatal population.
The use of antibiotics has been reported to be associated with primary idiopathic intussusception. In their case-control study, Spiro, et al. reported on 93 case patients with intussusception and 353 controls younger than 4 years of age who were seen in the ED of the Children's Hospital of Alabama between January 1, 1996 and April 30, 2001.7 Antibiotic use within 48 hours of diagnosis was found in 23 cases (25%) and 33 controls (9%) odds ratio, 4.15; 95% confidence interval [CI], 2.17 – 7.92; attributable risk, 18.7%. Cephalosporin use was associated with more than a 20-fold increased risk of intussusception.
Clinical Presentation. The classic presenting symptoms for the child with intussusception include: 1) intermittent and severe abdominal pain, 2) vomiting, and 3) bloody stools. The child often cries or screams during the episodes of pain, which may last for up to 30 minutes. Children often draw up their legs to their chest during these episodes. Vomiting of undigested food often is seen first followed by bilious emesis. Early in the course of the illness, the stools may appear normal and only later become dark red and mucoid, resembling "currant jelly." Blood in the stool is a sign of intestinal ischemia and mucosal sloughing.
Between these episodes of pain, the child may have a normal appearance and activity level, but alternating periods of lethargy may also be found. Pumberger, et al. report on a series of 13 infants who were ultimately found to have intra-abdominal pathology but who initially presented with an altered level of consciousness.8 These children had a variety of underlying problems, including intussusception, incarcerated inguinal hernia, and volvulus. The authors report that seven of 120 children were treated for intussusception after first being hospitalized for neurologic symptoms. Five of these seven children even underwent a lumbar puncture during their evaluation. In some of these cases, 24 hours elapsed before abdominal symptoms became evident and ultrasound (US) evaluation confirmed the correct diagnosis.
The etiology for the lethargy found in some patients with intussusception is not clearly understood. Venous occlusion, edema, and ischemia in the intussuscepted bowel may produce endotoxin or other mediators from the bowel to the bloodstream. Tenenbein and Wiseman do report the successful reversal of altered mental status in an infant with intussusception after the child was given naloxone.9 The reversal of symptoms in this child using an opioid antagonist supports the theory that release of endogenous opioid may be the cause of the lethargy seen in some patients with intussusception.
The classic triad of symptoms is only found in 20% of patients with intussusception.10 DiFiore (1999) reports that by changing the triad to include pain, vomiting, and gross or occult blood the average incidence increases to 32% of patients seen.5 Luks, et al. found that 86% of all cases presented with colicky abdominal pain, 64% had vomiting, 39% had hematochezia, 26% had a palpable abdominal mass, and 19% had some degree of lethargy or somnolence.11 Stringer, et al. report slightly different findings in their review of the literature with the following mean occurrence of symptoms and signs: abdominal pain, 83%; vomiting, 85%; blood in stool (occult or gross), 53%; and abdominal mass, 63%.12 The finding of a sausage-shaped mass in the right upper quadrant with absence of bowel in the right lower quadrant is known as Dance's sign and considered pathognomonic of intussusception. This physical finding is reportedly found in up to 85% of cases.6
Unfortunately, infants with intussusception can also present with non-specific and apparently minor symptoms until precipitous deterioration occurs. Byard and Simpson reported on two such cases in infants after their 35-year study of autopsy files of the Department of Histopathology at the Women's and Children's Hospital in Adelaide, Australia.13 The infants were 5 and 6 months of age and reportedly had "mild upper respiratory tract infection" symptoms for two days and five days, respectively. The children were found to have ileoileal and ileocecal intussusception with infarcted ileum at the time of autopsy. Neither of the two children had abnormal lead points as a cause of their intussusceptions. Ng'walali, et al. report a case of fatal intussusception in a 7-month-old infant.14 The child had no apparent medical problems by history and had no known abdominal pain or vomiting. Ileoileal intussusceptions were found at two sites in the ileum which showed blackish color changes due to compromised blood supply. The intestinal lumen for both proximal and distal portions of the lesions showed total obstruction. No other significant abnormalities were found at autopsy. Histology of the lesions demonstrated ischemic necrosis of the intestine associated with vascular congestion and focal hyperplasia of Peyer's patches.
Testing for the presence of blood is an important part of the evaluation of the infant or young child with vomiting or more nonspecific signs or symptoms supportive of intussusception. The clinician should specifically ask about the appearance of the child's stools as the parents may not see this as an important aspect of the child's problems. Losek and Fiete highlight the value of testing stool for occult blood when making the diagnosis of intussusception.15 The authors' retrospective review found that of the 96 children who had a barium enema study completed for a suspicion of intussusception over a four-year period, the diagnosis was confirmed in 57 patients. Of these 57 patients, 29 did not have history or physical findings of gross blood per rectum. Stool was tested in 16 of these 29 patients and 12 (75%) were positive. In comparison, for the patients without intussusception and without history or physical exam findings of gross blood per rectum, 15 of 33 had stool tested for occult blood and three were positive. Stool with occult blood was found to be significantly associated with intussusception (P < 0.002). The only other clinical factor significantly associated with intussusception was abdominal mass (P < 0.02). All of these patients were felt to have idiopathic intussusception. Although not a statistically significant association, the need for surgical reduction and bowel resection occurred more frequently in those patients who presented with gross blood per rectum than those who did not (69% vs 50%). The authors reported that they believed testing stool for occult blood may prevent the delay in the diagnosis of intussusception and therefore decrease the need for surgical reduction, especially in those patients who present with nonspecific irritability or vomiting.
Klein, et al. performed a retrospective cohort study of children seen in the ED at Children's Hospital and Regional Medical Center in Seattle, WA.16 Charts were reviewed for children who underwent contrast enema for the possible diagnosis and treatment of intussusception. The study covered a four-and-a-half-year period from January 1995 through July 1999. Charts were reviewed and history and physical examination findings were recorded on standardized data collection sheets. Abdominal exam findings specifically recorded included location of tenderness if present, presence or absence of distention, and presence or absence of a mass. The type of contrast enema performed (air vs barium vs a combination of both), the need for hospital admission, the need for surgical resection, and the final discharge diagnosis were recorded. A standardized radiograph evaluation form was used to indicate presence of soft tissue mass, presence of air fluid levels, obstruction, and stool and air pattern in the colon. Stool and air patterns were categorized as sparse/none, some, or a lot. Radiograph interpretations were calculated to be the average of both reviewers' readings. Characteristics of patients with intussusception were compared to patients without intussusception using chi-square test. The average age of the patients in each group was 1.2 years. Of 215 children who underwent contrast enema, 58 (27%) were positive for intussusception (41 male and 17 female) and 157 were negative for intussusception (91 male and 66 female). In a univariate analysis, patients with intussusception were more likely to have a history of vomiting (P = 0.02), abdominal pain (P = 0.1), rectal bleeding (P = 0.003), and physical exam findings of abdominal mass (P < 0.001), abdominal tenderness (P = 0.02) and guaiac positive stool (P = 0.004). The plain radiograph finding of the absence of stool in the ascending colon was also found to be predictive of the presence of intussusception (P < 0.05). However, the authors were unable to identify all patients with the diagnosis of intussusception. The sensitivity calculated for their prediction model was 100% (95% CI, 90% – 100%) and specificity of 7.1% (95% CI, 3 – 15%). However, they concluded that up to 10% of patients with intussusception would be missed using this same model.
Kuppermann, et al. completed a retrospective cross-sectional study to identify predictors of intussusception in young children.17 A consecutive sample of children 1 month to 5 years of age on whom contrast enemas were performed because of suspected intussusception seen at an urban children's hospital from 1990 to 1995 served as the study population. Only those patients who had histories and physical examinations documented in the medical record prior to the contrast enema were considered eligible for study. Data from the physical examination documented closest in time prior to the enema were used in the analysis. Physical examinations performed by physicians-in-training were not analyzed. Sixty-eight (59%) of the 115 patients had intussusception. Univariate predictors of intussusception included male gender, age younger than 2 years, history of emesis, rectal bleeding, lethargy, abdominal mass, and a highly suggestive abdominal radiograph. The plain abdominal radiographs were interpreted by a single pediatric radiologist who was unaware of the contrast enema findings. The radiographs were categorized as 1) not suggestive of intussusception (normal bowel gas pattern and no signs of mass or obstruction), 2) moderately suggestive of intussusception (abnormal but nonspecific bowel gas pattern and no obvious mass or obstruction), or 3) highly suggestive of intussusception (soft tissue mass, evidence of bowel obstruction or a visible intussusceptum). In a multivariate analysis, the authors identified only four independent predictors (adjusted odds ratio; 95% CI): a highly suggestive abdominal radiograph (18.3; 4.0 – 83.1), rectal bleeding (17.3; 2.9 - 104.0), male gender (6.2; 1.2 – 32.3), and a history of emesis (13.4; 1.4 –126.0). The most important independent predictors were a highly suggestive abdominal radiograph and rectal bleeding. Fifty-one (80%) of 64 patients with this type of radiograph had intussusception. However, intussusception also occurred in 21% of patients without highly suggestive radiographs. The authors do report that a negative or only moderately suggestive radiograph does not exclude the diagnosis. Of the 71 patients with either a history or physical examination consistent with rectal bleeding, 52 (73%) had intussusception. A negative heme test did not exclude the diagnosis as eight (26%) of 31 patients documented to be negative for fecal blood on examination also had intussusception. The authors conclude that patients initially suspected of having intussusception but who have no evidence of rectal bleeding or highly suggestive radiographs have a low probability of having the diagnosis. However, the upper end of the 95% CI for this negative predictive value included a 16% false-negative rate, which highlights the limitations of the variables analyzed in the study and available to most clinicians caring for this patient population.
The clinician must also have a high index of suspicion when caring for newborns with abdominal distension or vomiting. Although not commonly found among neonates, Ueki, et al. does report on a series of 14 neonates diagnosed with intussusception between June 1974 and January 2001.2 They report that six of these infants presented on the first day of life and had either intestinal atresia or malrotation. The average gestational age and birthweight were 37 weeks (range 34 – 39 weeks) and 2720 grams (range 1940 – 3138 grams), respectively. The most common signs were abdominal distention (83%) and emesis (83%). Only one patient in this group had intestinal perforation with intussusception at the time of laparotomy. They also report that eight of these patients developed abdominal symptoms two to 24 days after birth. The average gestational age and birthweight were 39 weeks (range 30 – 42 weeks) and 2800 grams (range 1050 – 4400 grams), respectively. The most common findings in this group were abdominal distention (75%), bloody stool (75%), and emesis (75%).
Clinicians should be aware of several other situations that put children at increased risk for intussusception. Hughes, et al. report that of 251 gastrojejunostomy tubes inserted to aid with feeding difficulties, 40 (16%) intussusceptions occurred in 30 patients.18 The age range was 3 months to 17 years (mean 2.6 years). Eight recurrences were found in patients with a distal pigtail (n = 17), but only twice in patients without a distal pigtail (n = 18). deVries, et al. report on 11 pediatric patients who developed postoperative intussusception during a 16-year period.19 A variety of initial abdominal operations had been performed. None of the patients had extensive retroperitoneal dissection, previous radiation therapy, or previous chemotherapy. A patient whose initial operation was for intussusception was included only when the site of the intussusception was different from the original one and a lead point was absent. The presenting symptoms were abdominal distension (82%) and persistent bilious nasogastric (NG) tube production (73%). With only one exception, a diagnosis was not made before reoperation. The symptoms developed after a median period of five days after initial operation. Hamada, et al. report a case of postoperative intussusception caused by an appendiceal stump two days following an appendectomy performed during a right inguinal herniorrhaphy in a 10-month-old boy.20 At the time of laparoscopic reduction, the lead point of the ileo-colic intussusception was observed as the inverted appendiceal stump.
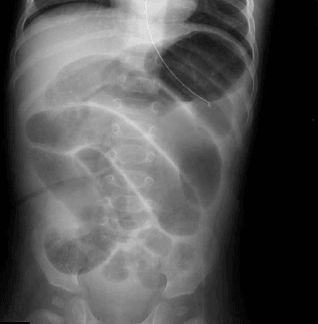
Diagnosis. Plain radiographs of the abdomen may be unremarkable early in the course of intussusception. The typical radiographic findings of a small bowel obstruction or the presence of free air are often seen in children with longer duration of symptoms. (See Figure 1.) Findings suggestive of the presence of intussusception include a soft tissue mass, absence of cecal gas and stool, and paucity of bowel gas. Exclusion of an ileocolic intussusception on abdominal radiographs is usually based upon the presence of gas and stool in the cecum. Daneman and Navarro report that in 45% of children under 5 years of age, the sigmoid colon is in the right lower quadrant of the abdomen and, therefore, a sigmoid filled with air and stool may be misinterpreted for the cecum.21 As mentioned earlier, Kuppermann, et al. found that intussusception occurred in 21% of patients without highly suggestive radiographs.17 John recommends obtaining radiographs to exclude any evidence of perforation prior to attempting any contrast reduction techniques.22 Sargent, et al. found that plain radiographs correctly identified only 45% of children with intussusception.23 They also found that the combination of three views of the abdomen enabled the diagnosis to be excluded in roughly 25% of their patients. The authors concluded that abdominal radio-graphs were often not helpful and that interobserver agreement of the interpretation of plain films in children with suspected intussusception is limited. If the diagnosis of intussusception cannot be made with the use of plain radiographs and clinical suspicion exists, another diagnostic test is indicated.
| Figure 1. Plain Film Image of Bowel Obstruction |
|
| Plain films showing pattern consistent with small bowel obstruction in a 9-month-old child with vomiting |
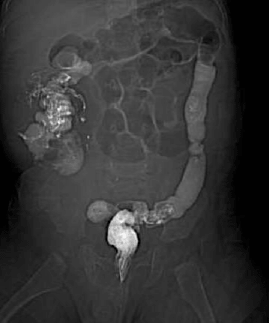
Contrast enema studies have been used for diagnosis and treatment of idiopathic intussusception for many years. (See Figure 2.) Risks of enema studies include relatively high radiation exposure, risk of perforation, and risk of barium peritonitis if barium is chosen for contrast medium. Royal reports a case of hypovolemic shock requiring resuscitation secondary to an air enema reduction of intussusception.24 The author reports that the technique used was as recommended by the American College of Radiology standards for intussusception reduction. He concluded that the patient had third spacing of fluids within the bowel lumen that was intrinsically related to the patient's intussusception with small bowel obstruction. He postulated that release of humoral agents such as cytokines or endotoxins were responsible for the patient's deterioration. He recommends that more data should be gathered on the fluid status of patients with intussusception prior to reduction technique and that an appropriate observation period of the patient following the enema reduction should occur.
| Figure 2. Contrast Enema in Patient with Intussusception |
|
| Contrast enema showing ileo-ileo-colic intussusception in an 8-month-old child |
Ng, et al. also report a serious complication after an attempt of air contrast reduction of an intussusception in a 9-month-old child.25 The patient was brought to an ED with a two-day history of abdominal pain, vomiting, and "currant jelly stool." He was diagnosed with an intussusception and an air enema was attempted. The procedure was performed in the radiology department without the use of sedation. During the procedure, fee intraperitoneal air was noted. Although insufflation pressure had been limited to less than 90 mmHg and insufflation was immediately stopped, the child's abdomen became extremely distended and tense. The child became tachypneic as well. The child was taken immediately to the operating room after becoming severely dyspneic and somnolent en route. Needle decompression was completed using a 14-gauge venous catheter in the upper abdomen. The abdomen deflated dramatically, the child regained a more normal pattern of respiration, and the child's mental status improved. A laparotomy followed and the intussusception was reduced manually. A perforation was found at the cecum just distal to the ileocecal valve. The child recovered without any sequela.
These cases reporting on complications that result from the performance of contrast enema studies highlight the need for pediatric preparedness for emergencies in the radiology department. Appropriate monitoring including airway equipment, monitoring devices, intravenous access, and trained personnel should be available whenever these interventions are made. Surgical personnel must also be readily available in the event of these complications.
Diagnostic contrast enema is still considered the quickest and most cost effective method of excluding or confirming the presence of intussusception. The main reasons given for its use include that this technique is less likely to miss the diagnosis, is well known to virtually all radiologists, and allows for timely therapeutic intervention. Air contrast enemas have been shown to be as effective as barium contrast enemas in terms of successful reduction.27 Air enema has been shown to be quicker and, therefore, leads to less radiation exposure. DiFiore reports that reduction rates with barium or water-soluble contrast were 50% to 85%, versus 81% to 91% success with air contrast reduction.6
However, the overlap of clinical signs and symptoms in children with other abdominal conditions that may mimic intussusception leads to unnecessary radiation exposure for those children without the diagnosis. US has offered a modality that allows for diagnosis without unnecessary radiation exposure. John reports that US is virtually 100% sensitive and specific for identification of intussusception.22 In 1987, Pracos, et al. also reported 100% accuracy of sonography for the diagnosis of intussusception.27 Verschelden, et al. reported a 100% negative predictive value in 83 patients in a prospective study and showed that none of their patients with a normal sonogram had an intussusception at contrast enema examination.28
The intussusception mass is usually fairly large with measurements greater than 5 cm. Most cases occur in the subhepatic region with evidence of displacement of adjacent bowel loops. The intussusception may demonstrate an outer hypoechoic region surrounding an echogenic center resulting in the "target" or "doughnut" appearance.29 Another pattern of imaging has been that of multiple concentric rings. On long axis scans, the hypoechoic layers on each side of the echogenic center may result in a pseudokidney appearance if the intussusception is curved or imaged obliquely. These signs should not be interpreted as pathognomonic as they can be seen in normal or pathologic intestinal loops. Bowel wall neoplasm, edema, or hematoma may cause a similar appearance with sonography. Stool or psoas muscle can be mistaken for an intussusception by an inexperienced operator.
The main concern regarding the use of sonography for the diagnosis is the feeling that the quality of the study and its interpretation are operator dependent. Reliance on this modality should only occur when those performing the exam have had adequate training and experience. Verschelden, et al. did show a 100% accuracy rate by third- and fourth-year radiology residents, but the residents in the study had a three- to five-month training period in sonography.28 Eshed, et al. completed a retrospective evaluation of the accuracy and performance of junior residents, senior residents, and staff radiologists over a four-year period.30 Of the 65 patients who had US and air enema, 37 (84%) were true positive and seven (16%) were false positive. Twenty-one patients had a negative US result, with 18 (86%) being true negative and three (14%) being false negative. Of the 86 patients who underwent US screening only, all were diagnosed as having a non-surgical condition after a period of observation in the emergency room. The total accuracy rate was 93%, sensitivity was 84%, specificity was 97%, positive predictive value was 93%, and the negative predictive value was 94%. Accuracy rate, sensitivity, and specificity were 92%, 85%, and 98% for staff radiologists; 94%, 75%, and 96% for senior residents; and 95%, 83%, and 97% for junior residents, respectively.
Navarro, et al. also report on 43 children with intussusception due to pathologic lead points between 1986 and 1999; 23 (66%) were depicted on sonography, five of seven (71%) on CT, three of 28 (11%) on air enema, and six of 15 (40%) on barium enema.31 US provided a specific diagnosis in nearly one-third of patients in their series.
In addition, sonography has been used in several studies as a guide to air enema reduction in children with intussusception. Gu, et al. utilized this technique in 199 children.32 In phase I of the study, fluoroscopy was used to confirm that all 11 children underwent successful reduction using US-guided pneumatic reduction. In phase II, the success of the initial reduction was 95% in 188 children. In this second phase, following successful reduction, complete reduction was confirmed by clinical improvement of the child and repeat sonography one hour later. No recurrence was found in 92% of phase II patients. In 10 (5%) of the 188 phase II patients, initial reduction was unsuccessful. Fluoroscopically guided air reduction successfully reduced only three of these 10 failures. Yoon, et al. report their overall success rate of US-guided pneumatic reduction was 92% with no immediate recurrence.33 Their study group consisted of 49 consecutive patients (age range of 2 months to 7 years) who underwent 52 reductions of intussusception over a nine-month period. Of the two patients who had irreducible intussusceptions, one had residual ileoileal intussusception at the time of surgery and the other had an ileal polyp as a lead point. Perforation was found in two of 52 (4%) cases with one patient requiring right hemicolectomy due to bowel necrosis and a pinpoint perforation in the normal proximal transverse colon, and the other underwent manual reduction of an ileoileocolic intussusception with microrperforation noted in the proximal transverse colon. Crystal, et al. reported on their retrospective review of 83 consecutive children sonographically diagnosed with 101 cases of intussusception over a 40-month period.34 In 99 of these cases, sonographically guided hydrostatic reduction was attempted. In 88 of the 99 cases (89%), reduction was successful. No complications were found in their series. The success rate was significantly lower among patients whose intussusception was located in the left side of the abdomen (P < 0.01) or contained entrapped fluid (P < 0.02) or those in whom hydrostatic reduction was not performed by an experienced radiologist (P < 0.01).
In a prospective study comparing air reduction, barium reduction under fluoroscopy, and saline reduction under US guidance for treatment of 147 patients with intussusception, Hadidi and El Ahal found significantly different success rates for the varied approaches.35 Air reductions were performed by a pediatric surgeon experienced in the technique and were successful in 45 of 50 children (90%). Barium enema reduction and saline reduction under US were performed by a radiologist experienced in these techniques and were successful in 35 of 50 children (70%) and 32 of 47 (67%), respectively. The success rate difference between air reduction and the other two techniques was statistically significant (P < 0.01). There was no significant difference between the barium enema and saline enema under US techniques (P > 0.05). No perforations were found in the air enema group; three perforations were found in the barium enema; two perforations were found in the US group.
From these studies, it appears that the use of US offers many benefits in terms of both diagnosis and guidance for nonoperative treatment of children with intussusception. The decreased radiation risk to those patients with and without intussusception certainly adds to the benefits of choosing this modality for diagnosis. However, the studies quoted all appear to utilize experienced radiologists in the performance of the US imaging. It is unlikely that radiologists would always be available 24 hours a day at all U.S. medical centers. Hopefully, with enough time and experience, quality US imaging will be consistently available in more centers to allow this modality to be utilized.
Treatment. In a study of children with intussusception in Washington State from 1987 to 1996, Bratton, et al. reported that 53% of the 570 children had operative reduction and 20% had resection of bowel.36 Children with operative reduction did not differ from those with nonoperative care by median age or gender. However, children with operative care were significantly more likely to receive care in hospitals with smaller pediatric caseloads and to have a coexisting condition associated with intussusception. Sixty-four percent of children who received care in a large children's hospital had nonoperative reduction, compared with 36% of children who received care in hospitals with 0 to 3000 annual pediatric admissions and 24% of children who had care in hospitals with 3000 to 10,000 annual pediatric admissions. Median length of stay and charges were significantly less in the large children's hospital compared with other centers.
Regardless of the hospital size or setting, when the diagnosis of intussusception is entertained, attention should be given to the hydration and perfusion status of the patient. Appropriate fluid resuscitation should be administered and IV access should be established. A NG tube might be helpful to decompress the patient's stomach and also alleviate any abdominal distension that may interfere with breathing. As mentioned earlier, preparation for possible deterioration is an important part of the care of the patient with suspected intussusception. Early involvement of surgical personnel is essential. Mutual discussion between the radiologist and surgeon should lead to a plan of care for the individual patient with suspected intussusception. The only absolute contraindications to attempts to reduce an intussusception using an enema are the presence of free air or signs of peritoneal irritation.
Although standard practice remains immediate surgery if nonoperative reduction is unsuccessful, there have been several reports of delayed repeat enemas after initial nonoperative attempts have failed. Navarro, et al. performed a retrospective analysis of all intussusception cases seen at the Hospital for Sick Children, Toronto, Canada from May 1999 to December 2002.37 There were 219 intussusceptions among 163 children. Enema reduction was attempted in 211 (96%) and was successful in 90.2% of attempts. Delayed reduction attempts were used in 25 patients (15.3%) in 26 intussusceptions (12.3%) and were successful in 50% of cases. The authors concluded that delayed, repeated reduction attempts should be considered when the initial attempt manages to move the intussusceptum and the patient remains clinically stable.
Irish, et al. report that the overall mortality of intussusception is less than 1%.1 They also report that recurrence following nonoperative reduction is 5% to 10% and following surgical reduction is 1% to 4%. They report a higher failure rate of nonoperative reduction with the presence of the following: ileo-ileocolic location, rectal bleeding, failed reduction with barium at another institution, age of more than 2 years or less than 3 months, duration of symptoms for more than 24 hours, small bowel obstruction on plain radiograph, and dehydration of more than 5%. They also highlight longer term complications of surgery in that they report adhesive small bowel obstruction in up to 7% of operative cases and 0% in nonoperative cases. DiFiore reports that most recurrences develop within 1 to 36 months after reduction, with a peak incidence during the eighth month.5
Management after successful nonoperative reduction of intussusception generally has included admission to the hospital for observation with specific concerns for recurrence or early complications. Bajaj and Roback addressed this issue in their retrospective cohort study of children 0 to 18 years of age who underwent uncomplicated enema reduction for intussusception at the Children's Hospital in Denver, Colorado over a four-and-one-half-year period.38 Hospitalization versus ED observation management was compared for length of stay, incidence of recurrence, and adverse events. There were no significant differences between the groups in terms of age, gender, duration of symptoms, or historical or physical examination features. The decision to admit or observe early in the study period was often made by the consulting surgeon. Of the 123 patients identified with a diagnosis code for intussusception, 106 (86%) had an enema reduction attempted. Three patients had a normal enema exam. Eighty-three (80%) of the patients had a successful reduction. Of the 78 patients with no preexisting condition and complete medical records, 27 (35%) were hospitalized and 51 (65%) were observed in the ED. The mean length of hospitalization was 22.7 hours (range 10 – 50 hours) and the mean length of ED observation was 7.2 hours (range 0 – 21 hours). Eleven recurrences were observed in eight of these 78 patients (10%). Four patients in the hospitalized group and four patients in the ED observation group had recurrences (five hours to 10.9 months). Four of the eight patients had a recurrence within the first 48 hours. All first recurrences occurred after the patient had been discharged from the hospital or the ED observation unit. No adverse events occurred in any of the patients who had a successful initial reduction. The current practice at Children's Hospital of Denver is to care for all patients with successful enema reduction in the ED observation unit with the disposition decision left to the pediatric emergency medicine attending.
Malrotation and Midgut Volvulus
Another surgical abdominal emergency that must be kept in mind when evaluating the young child is malrotation with midgut volvulus. The incidence of malrotation is estimated as 1 in 500 live births.1 Most children with this condition will present in their first year of life. Twenty-five to 40% of patients present within the first week of life, 50% within the first month, and 75% present prior to their first birthday.39 Volvulus occurs in 68% to 71% of neonatal malrotation cases39 and in 14% to 56% of non-neonatal cases.41 Males are at slightly higher risk than females.
Infants with other congenital anomalies may be at higher risk for the presence of malrotation, as well. Gastroschisis, omphalocele, and congenital diaphragmatic hernia are conditions that also involve rotational abnormalities and lack of fixation. Malrotation has been associated with intussusception, Meckel's diverticulum, and Hirschsprung's disease.1
Malrotation occurs when the normal rotation of the extracoelomic fetal intestine is interrupted. Lack of fixation of the small bowel results in obstructing, adhesive bands known as Ladd's bands. These abnormal bands form between the cecum and the right lateral abdominal wall causing compression of the duodenum and mechanical obstruction. A portion of the small bowel also becomes suspended on a stalk, which contains the superior mesenteric artery. If the small bowel twists around this stalk, a midgut volvulus and ischemia can follow. Prompt diagnosis is necessary to avoid the potential for development of bowel necrosis.
Clinical Presentation. The classic clinical presentation of midgut volvulus consists of sudden onset of vomiting in a previously healthy infant in the first month of life. The presence of bilious emesis has been called "the hallmark" of this condition, and reportedly occurs in 77% to 100% of cases.40,41 This presentation should trigger a prompt evaluation and immediate surgical consultation. Older patients may present with less clear cut symptoms with vague, intermittent abdominal pain along with vomiting.
The resultant obstruction which can occur with midgut volvulus is very proximal and abdominal distension is often not present. Torres and Ziegler report that a normal abdominal examination was found in 50% of their patients with malrotation and that another 32% had abdominal distension without tenderness.40 Once abdominal distension is seen, the patient most likely has already developed significant intestinal ischemia and peritonitis. A high index of suspicion is therefore necessary to make the correct diagnosis before a catastrophic event occurs.
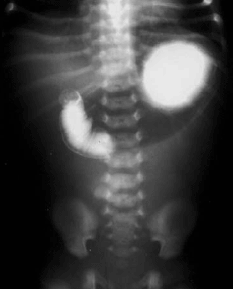
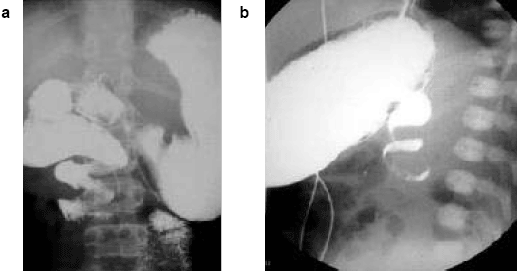
Diagnosis. Diagnosis is made with the use of contrast studies for the stable patient. Plain radiographs may show evidence of small bowel obstruction or proximal gastric or duodenal obstruction with little distal bowel gas. Contrast studies of either the upper or lower GI tract can be used to make the diagnosis. (See Figures 3–5.) When a malrotation is present, an upper GI series would outline an abnormal duodenal "C-sweep" with the duodenojejunal junction found to the right of the spine and a corkscrew tapering or ending of contrast. Irish, et al. report the specificity of the upper GI study to detect malrotation as 100% but a sensitivity for detecting midgut volvulus as only 54%.1 A contrast enema may reveal abnormal placement of the cecum into the upper abdomen, but the position of the cecum can be quite variable. Multiple dilated loops of small bowel may obscure this cecal displacement however. An upper GI series should be completed in those patients with a normal contrast enema in the setting of dilated loops of small bowel. Millar, et al. report that the presence of multiple air-fluid levels in dilated bowel loops are "ominous signs and usually indicate established gut infarction."42
| Figure 3. Malrotation with a C-shaped Sweep |
|
| Upper gastrointestinal series showing C-shaped sweep to the right of midline in 1-week-old child with bilious emesis |
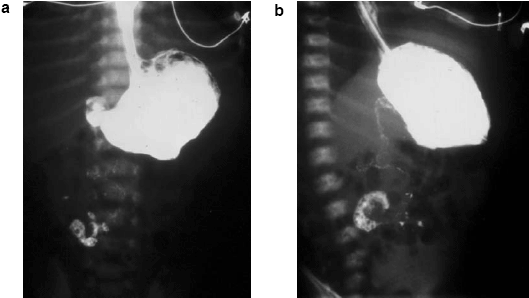
| Figure 4a-b. Corkscrew Appearance in Upper GI Series |
|
| Upper gastrointestinal series showing corkscrew appearance of contrast in 1-week-old child with bilious emesis. a: anteroposterior view; b: lateral view |
| Figure 5a-b. Contrast Cutoff in Upper GI Series |
|
| Upper gastrointestinal series showing abrupt end to passage of contrast material in 1-week-old child with bilious emesis. a: anteroposterior view; b: lateral view |
US findings in patients with midgut volvulus are also being reported in the literature. Sonographic views might reveal a fluid-filled, dilated proximal duodenum when malrotation is present. An abnormal relationship between the superior mesenteric artery and vein may be found when US is utilized for these patients. The superior mesenteric artery is normally found to the left of the vein. However, inversion of the superior mesenteric vessels has also been reported in patients with intussusception.43 Color Doppler US may reveal spiraling of the vessels called the "whirlpool sign."22 This sign is seen with a transverse scan just below the pancreatic head and appears as a circle of vascularity representing the superior mesenteric vein twisting around the superior mesenteric artery. In a review of a sonography computer database of a pediatric hospital over a 26-month period, Patino and Munden report on seven patients with a diagnosis of midgut volvulus on the basis of this whirlpool sign.44 These patients ranged in age from 1 day to 5½ years, with a mean age of 7.8 months. Most of these cases were not clinically suspected to have midgut volvulus. Their symptoms included projectile, nonbilious emesis, colicky abdominal pain with suspicion of intussusception, palpable abdominal masses, right lower quadrant pain with suspected appendicitis, and chronic diarrhea. Six of these seven cases of midgut volvulus were confirmed with upper GI studies and one patient was taken directly to surgery on the basis of the sonographic findings alone. One patient had an abdominal CT scan for continued suspicion of an abdominal mass. All seven cases were confirmed as midgut volvulus at surgery. All of the patients had an uneventful recovery and none had necrotic bowel at the time of surgery. These US examinations were done by a technician and then reviewed or rescanned by a board-certified pediatric radiology attending physician. As with any US technique, the widespread application of this approach certainly will take time to implement and would appear to be dependent on the experience and expertise of the person performing the examination.
Sze, et al. report the benefit of sonography in the diagnosis of midgut volvulus in a term neonate.45 On the first day of life, the child had emesis. A distended stomach was noted on plain radiographs. The emesis became bilious and an upper GI series was attempted. The contrast study was nondiagnostic because the contrast agent did not empty from the stomach despite positional maneuvers and waiting several minutes. Sonography revealed pylorospasm and reversal of the normal relationship of the superior mesenteric artery and vein with the whirlpool appearance of the vessels. The authors also report an unusual truncated appearance of the proximal superior mesenteric artery near its origin from the aorta. The diagnosis of malrotation with midgut volvulus was confirmed at surgery. No ischemia or necrosis was found and the child was discharged in good condition.
The other imaging modality to consider in confirming the diagnosis of volvulus is computed tomography. Accuracy is certainly an important factor when making a decision on which imaging test to order, but potential radiation exposure is also a consideration. Ai, et al. reported on the use of spiral CT to diagnose this condition.46 The case report involved an infant male born at 32 weeks of gestation. The baby developed bilious emesis on day 4 of life and had an upper GI study performed on day 12. This study showed transient dilatation of the third part of the duodenum. The transit of the contrast medium was normal and no corkscrew of the duodenum was seen. The duodenojejunal junction was found to the left of midline overlying the pedicles on anteroposterior (AP) view and caudal to the duodenal bulb on both AP and lateral views. The position of the cecum was felt to be normal. There was still significant concern about the possibility of malrotation. An US study did not reveal any obstructing lesion at the duodenum and overlying bowel gas prevented clear demonstration of the superior mesenteric artery anatomy. A spiral CT was completed with the administration of oral gastrograffin contrast medium through a NG tube just prior to the study. Seven millimeter thickness non-enhanced and 3 mm thickness contrast-enhanced spiral CT of the upper abdomen was performed. A distended stomach and proximal duodenum was seen along with a "whirl-like" pattern of small bowel loops converging toward a central point at the horizontal duodenum. The superior mesenteric vein was clearly seen on the left of the superior mesenteric artery. Wrapping of the vein, small bowel, and mesentery around the superior mesenteric artery was noted. All of these findings were confirmed at the time of surgery that day. The authors report that upper GI studies are both sensitive and accurate for the diagnosis of malrotation, but they go on to report that the sensitivity of these studies to identify midgut volvulus is only 54%. They also estimate that the radiation dose of a spiral CT study without the noncontrast images would be less than that of a typical upper GI completed with two minutes of fluoroscopy, two spot films, and one over-table tube exposure.
Treatment. Treatment for patients with suspected malrotation with or without midgut volvulus must include prompt correction of fluid losses and frequent reassessment for the presence of shock. The placement of a NG tube may be helpful for the child with abdominal distension as this may lead to respiratory compromise for the young child. Administration of broad-spectrum antibiotics such as ampicillin, gentamicin, and clindamycin is indicated for children with any concern for bowel ischemia. Prompt surgical evaluation should be initiated for these patients.
Prompt diagnosis is indicated for these patients and morbidity and mortality is determined by the presence or absence of bowel necrosis. Irish, et al. reports a mortality in infants with malrotation from between 2.5% and 24 %.1 They go on to report that the presence of necrotic bowel at the time of laparotomy leads to a 25-fold greater mortality rate. Messineo, et al., in their study of 182 patients undergoing laparotomy for malrotation, found a 0.999 estimated survival in patients with 10% bowel necrosis, 0.920 with 50% necrosis, and 0.351 with 75% necrosis.10
Summary
The young child with intussusception or midgut volvulus with malrotation is often a challenge to the clinician. Delays in diagnosis can lead to significant morbidity and mortality. Consideration of these serious diagnoses must be made for every young child with symptoms of vomiting or apparent abdominal pain. A high index of suspicion along with special attention to each patient's history and physical exam will most likely lead to a more prompt diagnosis. Whenever these diagnoses cannot be ruled out with confidence based on the patient's history and physical exam, further evaluation utilizing the expertise of colleagues in radiology and surgery who are familiar with these conditions and their evaluation is indicated.
Acknowledgements. Special thanks to Judith Roberson for her diligent efforts at transcribing this manuscript, and to Dr. Kristen Thomas for providing the radiographic images.
References
1. Irish MS, Pearl RH, Caty MG, et al. The approach to common abdominal diagnoses in infants and children. Pediatr Clin North Am 1998;45:729-772.
2. Ueki I, Nakashima E, Kumagai M, et al. Intussusception in neonates: Analysis of 14 Japanese patients. J Paediatr Child Health 2004;40:388-391.
3. Velez LI, Benitez FL, Villanueva SE. Acute abdominal pain in special populations. Part I: Pediatric patients. Emerg Med Rep 2004;25:273-279.
4. Kurzbart E, Cohen Z, Yerushalmi B. Familial idiopathic intussusception: A report of two families. J Pediatr Surg 1999;34:493-494.
5. DiFiore JW. Intussusception. Semin Pediatr Surg 1999;8:241-220.
6. D'Agostino J. Common abdominal emergencies in children. Emerg Med Clin North Am 2002;20:139-153.
7. Spiro DM, Arnold DH, Barbone F. Association between antibiotic use and primary idiopathic intussusception. Arch Pediatr Adolesc Med 2003;157:54-59.
8. Pumberger W, Dinhobl I, Dremsek P. Altered consciousness and lethargy from compromised intestinal blood flow in children. Am J Emerg Med 2004;22:307-309.
9. Tenenbein M, Wiseman NE. Early coma in intussusception: endogenous opioid induced? Pediatr Emerg Care 1987;3:22-23.
10. Messineo A, MacMillan JH, Palder SB, et al. Clinical factors affecting mortality in children with malrotation of the intestine. J Pediatr Surg 1992;27:1343.
11. Luks FI, Yazbeck S, Perreault G, et al. Changes in the presentation of intussusception. Am J Emerg Med 1992;10:574-576.
12. Stringer MD, Pablot SM, Brereton RJ. Paediatric intussusception. Br J Surg 1992;79:867-876.
13. Byard RW, Simpson A. Sudden death and intussusception in infancy and childhood – Autopsy considerations. Med Sci Law 2001;41:41-45.
14. Ng'walali PM, Yonemitsu K, Tsunenari S. Fatal intussusception in infancy: An experience in forensic autopsy. Leg Med (Tokyo) 2003;5:181-184.
15. Losek JD, Fiete RL. Intussusception and the diagnostic value of testing stool for occult blood. Am J Emerg Med 1991;9:1-3.
16. Klein EJ, Kapoor D, Shugerman RP. The diagnosis of intussusception. Clin Pediatr 2004;43:343-347.
17. Kuppermann N, O'Dea T, Pinckney L, et al. Predictors of intussusception in young children. Arch Pediatr Adolesc Med 2000;154:250-255.
18. Hughes U, Connolly B, Chait P, et al. Further report of small-bowel intussusceptions related to gastrojejunostomy tubes. Pediatr Radiol 2000;30:614-617.
19. de Vries S, Sleeboom C, Aronson DC. Postoperative intussusception in children. Br J Surg 1999;86:81-83.
20. Hamada Y, Fukunaga S, Takada K, et al. Postoperative intussusception after incidental appendectomy. Pediatr Surg Int 2002;18:284-286.
21. Daneman A, Navarro O. Intussusception. Part l: A review of diagnostic approaches. Pediatr Radiol 2003;33:79-85.
22. John SD. Imaging of acute abdominal emergencies in infants and children. Curr Probl Pediatr 2001;31:319-353.
23. Sargent MA, Babyn P, Alton DJ. Plain abdominal radiography in suspected intussusception: A reassessment. Pediatr Radiol 1994;24:17-20.
24. Royal SA. Hypovolemic shock after air reduction of intussusception. Pediatr Radiol 2001;31:184-186.
25. Ng E, Kim HB, Lillehei CW, et al. Life threatening tension pneumoperitoneum from intestinal perforation during air reduction of intussusception. Paediatr Anaesth 2002;12:798-800.
26. Rubi I, Vera R, Rubi SC, et al. Air reduction of intussusception. Eur J Pediatr Surg 2002;12:387-390.
27. Pracros JP, Tran-Minh VA, Morin DE, et al. Acute intestinal intussusception in children: Contribution of ultrasonography (145 cases). Ann Radiol 1987;30:525-530.
28. Verschelden P, Filiatraut D, Garel L, et al. Intussusception in children: Reliability of US in diagnosis – A prospective study. Radiology 1992;184:741-744.
29. Vasavada P. Ultrasound evaluation of acute abdominal emergencies in infants and children. Radiol Clin North Am 2004;42:445-456.
30. Eshed I, Gorenstein A, Serour F, et al. Intussusception in children: Can we rely on screening sonography performed by junior residents? Pediatr Radiol 2004;34:134-137.
31. Navarro O, Dugougeat F, Kornecki A, et al. The impact of imaging in the management of intussusception owing to pathologic lead points in children. A review of 43 cases. Pediatr Radiol 2000;30:594-603.
32. Gu L, Zhu H, Wang S, et al. Sonographic guidance of air enema for intussusception reduction in children. Pediatr Radiol 2000;30:339-342.
33. Yoon CH, Kim HJ, Goo HW. Intussusception in children: US-guided pneumatic reduction – Initial experience. Radiology 2001;218:85-88.
34. Crystal P, Hertzanu Y, Farber B, et al. Sonographically guided hydrostatic reduction of intussusception in children. J Clin Ultrasound 2002;30:343-348.
35. Hadidi AT, El Shal N. Childhood intussusception: a comparative study of nonsurgical management. J Pediatr Surg 1999;34:304-307.
36. Bratton SL, Haberkern CM, Waldhausen JHT, et al. Intussusception: Hospital size and risk of surgery. Pediatrics 2001;107:299-303.
37. Navarro OM, Daneman A, Chae A. Intussusception: the use of delayed, repeated reduction attempts and the management of intussusceptions due to pathologic lead points in pediatric patients. AJR Am J Roentgenol 2004;182:1169-1176.
38. Bajaj L, Roback MG. Postreduction management of intussusception in a children's hospital emergency department. Pediatrics 2003;112:1302-1307.
39. Weinberger E, Winters W. Abdominal pain and vomiting in infants and children: imaging evaluation. Comp Ther 1997;23:679-686.
40. Torres A, Ziegler M. Malrotation of the intestine. World J Surg 1993;17:326-331.
41. Seashore JH, Touloukian RJ. Midgut volvulus: An ever-present threat. Arch Pediatr Adolesc Med 1994;148:43.
42. Millar AJW, Cywes S. Malrotation and volvulus in infancy and childhood. Semin Pediatr Surg 2003;12:229-236.
43. Papadopoulou F, Efremidis SC, Raptopoulou A, et al. Distal ileocolic intussusception: Another cause of inversion of superior mesenteric vessels in infants. AJR Am J Roentgenol 1996;167:1243-1246.
44. Patino MO, Munden MM. Utility of the sonographic whirlpool sign in diagnosing midgut volvulus in patients with atypical clinical presentations. J Ultrasound Med 2004;23:397-401.
45. Sze RW, Guillerman RP, Krauter D, et al. A possible new ancillary sign for diagnosing midgut volvulus: The truncated superior mesenteric artery. J Ultrasound Med 2002;21:477-480.
46. Ai VHG, Lam WWM, Cheng W, et al. CT appearance of midgut volvulus with malrotation in a young infant. Clin Radiol 1999;54:687-698.
The young child who presents with vomiting with our without abdominal pain is often a challenge in an acute care setting.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.