Presentation, Assessment, and Management of Acute Myocarditis in Infants and Children
Presentation, Assessment, and Management of Acute Myocarditis in Infants and Children
Authors: Andora Bass, MD, Assistant Professor of Pediatrics, Department of Pediatrics, Division of Pediatric Critical Care Medicine, Brody School of Medicine at East Carolina University; David Fairbrother, MD, Assistant Professor of Pediatrics, Department of Pediatrics, Division of Pediatric Cardiology, Brody School of Medicine at East Carolina University; Ronald M. Perkin, MD, MA, Professor and Chairman, Department of Pediatrics, Brody School of Medicine at East Carolina University
Peer reviewer: Dante Allen Pappano, MD, MPH, Attending Physician, East Tennessee Children's Hospital, Knoxville, TN.
During a busy viral season clinicians can become complacent, with many children presenting with the same viral history and physical exam. However, early recognition of unusual complications or atypical presentations of viral infections are critical to minimize morbidity and mortality. For example, any child who wheezes should receive a thorough cardiac exam to make sure that the symptoms have a noncardiac origin.
Myocarditis may have a wide variety of etiologies and presentations and the clinician must have an awareness of this disease process to adequately screen patients. Although viral infections are the most common etiology, noninfectious etiologies also may occur. This article provides a review of the diverse presentation, assessment, and management of a child with myocarditis.
The Editor
Introduction
Myocarditis is an inflammatory process involving the myocardium that leads to cardiac dysfunction or even failure. It can be an elusive diagnosis; therefore, to identify potential victims it requires a high index of suspicion, careful attention to history, and a thorough physical exam. The complication and mortality rates for children with myocarditis are notable, making early recognition important and possibly even life-saving.
Etiology
Most cases of myocarditis are caused by viruses. The inflammatory process that ensues is the result of several mechanisms, including direct myocardial cell invasion, immunologic responses, or toxin production.1 Adenovirus is the most common viral etiology, and it leads to 55-60% of virus positive cases. Coxsackie viruses are associated with 30-35% of cases.2,3 Other viruses implicated in myocarditis are listed in Table 1.
 |
Globally, American trypanosomiasis (Chagas' disease) is another important infectious cause of cardiac failure. It primarily is found in Central America, South America, and Mexico. The symptoms of congestive heart failure present in the chronic phase of the illness and are thought to be related to autoimmune responses secondary to T cell activation.4
Other infectious causes of myocarditis are much less common. However, there have been cases associated with bacteria, rickettsia, fungi, mycobacteria, and parasites. Some bacteria cause a hypersensitivity reaction that leads to myocardial inflammation and dysfunction. Streptococcus pyogenes causes pancarditis and valvulitis by an immune-mediated inflammatory reaction. This process also has been reported with meningococcemia and typically occurs four to seven days after initial presentation.5 Exotoxin production that is associated with Corynebacterium diphtheriae and Shigella sonnei also has been reported to lead to direct myocardial damage.6
Myocarditis also can be the result of noninfectious etiologies, including medications. Since early discontinuation of the offending agent may decrease the sequelae, a thorough history must include information about medication exposures. Some medications cause direct cardiac toxicity and lead to myocardial dysfunction. (See Table 2.) The anthracycline chemotherapeutic drugs doxorubicin and daunorubicin are the most commonly implicated agents and can either cause symptoms during initial exposure or months to years later. Toxicity also has been associated with 5-Fluorouracil and is not dose related. It occurs in 1-2% of patients and carries a mortality rate of 10-15%.7 Drugs also can cause hypersensitivity reactions that initiate extensive cardiac inflammation (see Table 3), and hemorrhagic necrosis of cardiac cells has occurred with cyclophosphamide. This reaction also is not dose related and leads to acute congestive heart failure.8 Chronic use of ipecac (emetine) also is associated with myocardial toxicity and deaths due to inhibition of protein biosynthesis and subsequent interstitial edema and muscle necrosis.9
 |
Other causes of noninfectious myocarditis are autoimmune diseases (rheumatoid arthritis, ulcerative colitis, and most notably systemic lupus erythematosus), exogenous poisonings (ethanol, heavy metals, envenomation, and radiation therapy), and other inflammatory disease processes (Kawasaki disease, Takayasu's disease, and sarcoidosis). Kawasaki disease most commonly is associated with coronary vessel dilatation and aneurysmal formation; however, myocarditis also can occur and does not correlate with degree of coronary artery involvement.10
Epidemiology
The true incidence of myocarditis in the pediatric population is difficult to measure due to the wide variety of presentations and the lack of diagnostic protocols. A post-mortem analysis showed a prevalence of 3.5-5%,11 but the rate of clinically significant cases more closely approximates 0.1-0.6%.12 Patients of all ages may be affected, but the majority of cases occur in infants and teenagers; more than one-half of all cases are seen in the first year of life.1 The reason for this unusual bimodal age distribution is currently unclear.
Acute viral myocarditis is now implicated as one of the most common causes of sudden death in children without congenital heart disease. The reported incidences are 6%13 to 10%.14,15 Other studies have reviewed cases initially considered sudden infant death syndrome in which immunohistochemical and PCR analyses of myocardium indicated the presence of viral pathogens.16,17
Clinical Presentation
The clinical presentation of myocarditis is highly variable; it ranges from acute cardiovascular collapse to an insidious presentation with subtle cardiovascular signs.1 This spectrum includes the classifications of fulminant myocarditis, subacute or chronic myocarditis, and dilated cardiomyopathy. Each of these disease states correlates with a group of presenting features, histological features, and clinical outcomes as demonstrated in Table 4.2
Despite this variable presentation, most cases of acute viral myocarditis present with vague complaints and minor cardiovascular involvement. Patients may have tachycardia (out of proportion to degree of fever if present), mild hypotension, wheezing secondary to pulmonary edema, and altered perfusion with subsequent metabolic acidosis. History may reveal a nonspecific viral prodrome (e.g., fever, gastrointestinal complaints, coryza), syncope, chest pain (more common in adolescents), or symptoms of congestive heart failure (e.g., irritability, failure to thrive, poor feeding, or diaphoresis).2 Providers initially may think of diagnoses such as bronchiolitis, viral gastroenteritis, dehydration, colic, or asthma since they may present similarly and more commonly. Therefore, a high index of suspicion often is necessary to identify affected patients and proceed with further workup.
Diagnosis
The diagnosis of myocarditis requires suggestive historical and physical clues paired with further diagnostic evaluation, including an electrocardiogram (ECG), echocardiography, chest radiograph, cardiac troponin levels, endomyocardial biopsy, and viral studies. The first step, however, is to rule out other causes of cardiac dysfunction, such as structural abnormalities, coronary anomalies, pericardial effusion with tamponade, chronic hypertension, metabolic disorders (such as glycogen storage diseases), and chronic dysrhythmias.
ECG findings with acute myocarditis are variable but most typically show sinus tachycardia, low-voltage QRS in standard and precordial leads, and flattening or inversion of T waves in standard or left precordial leads.18 (See Figure 1.) Patients also may have ST segment elevation or acute dysrhythmias (e.g., supraventricular tachycardia, atrial ectopic tachycardia, premature beats, ventricular tachycardia (see Figures 2 and 3), and ventricular fibrillation). Additionally, myocarditis should be included in the differential diagnosis of a child whose electrocardiogram shows signs of acute myocardial ischemia or infarction.19 These ischemic-like findings in myocarditis presumably result from focal areas of inflammatory myocardial necrosis or inflammatory changes in the coronary arteries causing occlusion.
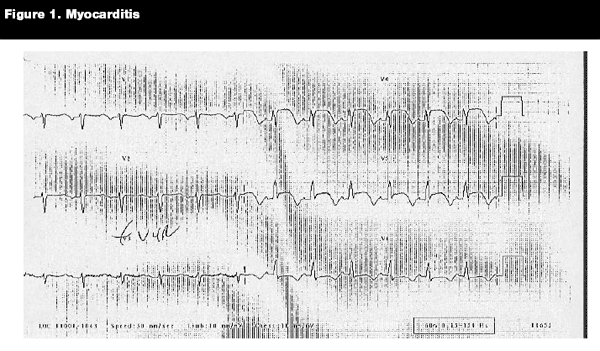 |
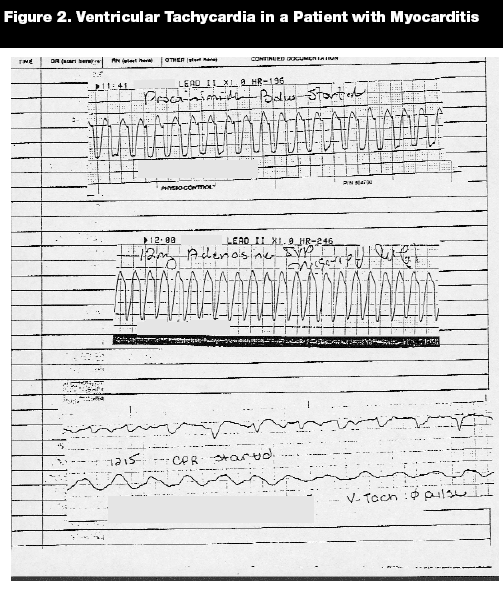 |
 |
A chest radiograph may be normal or may reveal an enlarged cardiac silhouette, pulmonary congestion, pleural effusion, or interstitial infiltrates. (See Figure 4.)
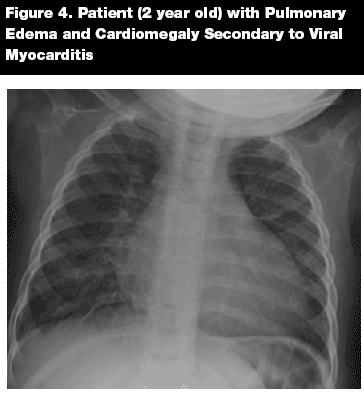 |
An echocardiogram characteristically shows enlarged ventricles and reduced shortening or ejection fractions with or without mitral or tricuspid regurgitation.19 Less commonly, there also may be findings of pulmonary hypertension (secondary to increased left atrial pressure) and intracardiac thrombi.
Some laboratory studies may be helpful in the evaluation of myocarditis. Cardiac troponin, a serum marker of myocardial injury, is elevated in patients with suspected acute viral myocarditis,20 as is the MB isoenzyme of creatine kinase. One pediatric study evaluating cardiac troponin found that a cutoff level of 0.052 ng/mL was able to distinguish acute viral myocarditis (diagnosed by endomyocardial biopsy) from chronic dilated cardiomyopathy and from congestive heart failure secondary to a large ventriculoseptal defect.21 Other less specific laboratory abnormalities include elevated white blood cell count22 or erythrocyte sedimentation rate.23 Studies to discover a potential viral etiology also should be considered, including nasal and rectal viral cultures and identification of known pathogens by polymerase chain reaction (PCR), immunoglobulin antibody assays, or viral titers.
The gold standard for diagnosis and classification of myocarditis is by endomyocardial biopsy. The Dallas criteria (see Table 5), which are widely accepted, define and classify myocarditis by histological features. PCR and ribonucleic acid hybridization techniques also are used to detect viral genetic material in the myocardium from these samples.3 However, there are known limitations. Patients may be too unstable for an endomyocardial biopsy procedure during the most opportune time to detect histologic or molecular biologic changes. There also may be a sampling error, procedural complications, or variations in pathologic interpretation.24
 |
Management
Just as diagnosing myocarditis can be elusive, there is no established, evidence-based protocol for managing these patients. More often than not, the interventions are supportive in nature rather than following recommendations from evidenced-based medicine. In general, the supportive measures utilized are driven by the clinical objectives at that time and usually include augmenting a low cardiac output state. The mainstays include improving cardiac output and hemodynamic function by maximizing ventricular function while minimizing myocardial oxygen consumption. (See Table 6.)
 |
Inotropic Support. Pharmacotherapeutic agents often are utilized in patients with myocarditis to improve cardiac function. The beta-adrenergic agonists (dopamine, dobutamine, and epinephrine) are used to augment cardiac output and improve myocardial contractility.25-28 The effects of these drugs are dose related and their use may have detrimental adverse reactions, including: 1) arrhythmogenesis; 2) excessive chronotropy (increased heart rate with resultant poor ventricular filling); 3) increased myocardial oxygen consumption; 4) down regulation of beta-adrenergic receptors; and 5) increased afterload (pulmonary and/or systemic hypertension).
While inotropic support with beta-adrenergic agonists are indicated for patients with severely compromised ventricular function and symptoms of low systemic oxygen delivery,25 no studies have specifically demonstrated the efficacy of these inotropes with viral myocarditis. In fact, lack of inotropic effect may occur in children with myocarditis since these medications require intact myocytes to improve contractility. Generalized cardiac dysfunction in viral myocarditis is the net result of myocyte injury and death from direct viral invasion and immune mediated cytopathic effects.29,30 However, the extent of myocyte injury and death is variable, which may explain the variability in cardiac function and response to inotropes seen in these patients.
The inability of beta-adrenergic drugs to improve contractility has been described in some patients, specifically in adults with acute and chronic dilated cardiomyopathies. The mechanisms by which this occurs include beta-adrenergic receptor down-regulation31 and tachycardia that then leads to increased myocardial oxygen demand and decreased left ventricular filling.32 The mechanisms explaining inotrope insensitivity described in these patients also may explain the poor contractile response to beta-adrenergic agents in patients with myocarditis. Dopamine and dobutamine specifically have been shown in some studies to increase myocardial oxygen demand without significantly improving contractility.33,34
Another concern with the use of beta-adrenergic agents is the potential for dysrhythmias. Patients with myocarditis are inherently at risk for rhythm and conduction abnormalities. Since these drugs are arrhythmogenic, they should be used cautiously in children with myocarditis.
Some physicians have recommended digoxin for use in patients with myocarditis. However, digoxin increases both the expression of proinflammatory cytokines and mortality in a murine model of viral myocarditis, which has lead investigators to conclude that digoxin should be used with caution in patients with myocarditis.35,36 Several authors suggest that digoxin may even be contraindicated in acute myocarditis.27,30,37
Afterload Reduction. Milrinone is a phosphodiesterase III inhibitor that combines inotropic activity with afterload reduction.38 It improves left ventricular relaxation (lusitropy), which results in increased stroke volume and cardiac output.39 The vasodilating effect of milrinone decreases systemic and pulmonary vascular resistance,40 which has therapeutic potential but means it should be used cautiously in patients who are hypotensive. This risk is reduced by starting a low-dose infusion without a loading dose. Unlike the beta-adrenergic agents, it maintains a favorable myocardial oxygen supply/demand ratio. Therefore, milrinone appears to be a good therapeutic choice in children with low cardiac output and high systemic vascular resistance.39,41
Diuresis. Patients with acute heart failure often will benefit from diuresis as they usually have excess total body water. In borrowing from the literature on adult heart failure, aggressive diuresis is considered to be one of the first therapies to institute in this setting.42 Although diuretic therapy does not seem to affect overall survival, its use is very effective in decreasing some of the symptoms associated with heart failure, in particular, respiratory distress due to pulmonary edema. Furosemide (Lasix) is particularly efficacious, and can be given orally, intravenously (IV), or by an intramuscular route if IV access is not available in an emergent situation.
Antiarrhythmic Therapy. In addition to the primary effects caused by the low cardiac output syndrome, secondary effects of an irritable myocardium mean that arrhythmia is an ever-present danger with acute myocarditis. Antiarrhythmic therapy usually is not started empirically, but is initiated once ectopy (usually ventricular in origin) is seen. Depending on the clinical scenario, beta blockers often are used as first-line agents with ventricular arrhythmias. They should be used with caution, however, as they can adversely affect myocardial performance. One exception is carvedilol. When started at low doses, it has been used in patients with chronic heart failure with limited success; it also provides some anti-arrhythmic effects due to its beta blockade.43 If cardiac function is significantly compromised and ventricular ectopy is present, amiodarone may be a more logical choice because it has less detrimental effect on ventricular function. However, it also should be used with caution, as rapid loading can result in both hypotension and arrhythmia.
Respiratory Support. When the patient is significantly compromised, respiratory failure often is not far behind. As a result, when acute myocarditis is suspected and a low cardiac output syndrome is present, preparation for mechanical ventilation should be made. Several trials have been conducted in the adult population evaluating both invasive and noninvasive ventilation in patients with respiratory distress associated with heart failure. Similar studies have not been conducted in the pediatric population.25 However, the physiology of positive pressure ventilation has been previously studied and the benefits include decreased afterload, decreased pulmonary edema, and resultant improved efficiency of gas exchange.44 Therefore, ventilatory support should be considered early in these patients to decrease oxygen consumption and improve cardiac output.
Immunomodulation. As a virus often is the inciting event for acute myocarditis, significant attention has been given to the use of therapies affecting the immune system in an effort to decrease the ensuing inflammatory response. These strategies have included steroids, immune globulin, and various immunosuppressive agents. Unfortunately, however, there are no large scale studies to compare the efficacy of these medications. Additionally, there does not appear to be a significant difference in outcome despite which approach is taken.45
Intravenous immune globulin (IVIG) has been used in the treatment of primary and secondary antibody deficiencies for more than 25 years.46 It is a safe preparation with no long-term side effects.46 There are currently six clinical indications for which IVIG have been licensed by the United States Food and Drug Administration (FDA).47 These can be summarized as follows: 1) treatment of primary immunodeficiencies; 2) prevention of bacterial infections in patients with hypogammaglobulinemia and recurrent bacterial infection caused by B-cell chronic lymphocytic leukemia; 3) prevention of coronary artery aneurysms in Kawasaki disease (KD); 4) prevention of infections, pneumonitis, and acute graft-versus-host disease after bone marrow transplantation; 5) reduction of serious bacterial infections in children with HIV; and 6) increase of platelet counts in idiopathic thrombocytopenia purpura to prevent or control bleeding.47 However, a number of other clinical benefits of IVIG treatment have been demonstrated. Many of these other uses result from its anti-inflammatory and immunomodulatory effects.46,47
Case reports suggest that patients with acute myocarditis benefit from high-dose IVIG.47 In one pediatric study, 21 children presenting with presumed myocarditis were treated with IVIG 2 g/kg over 24 hours after presentation.48 Twenty-five historical untreated patients were used as the control group. Improvement in the left ventricular shortening fraction favored the treatment group and significant differences existed in the treated group at the 12-month follow-up.48 Despite a common practice of IVIG administration among the Pediatric Cardiac Intensive Care Society members, there are no randomized, controlled studies in children to definitively support the use of IVIG in the management of acute viral myocarditis.45,49,50
Immune suppression is a more controversial topic that warrants further investigation. Multiple agents, including steroids, muromonab-CD3, and azathioprine, have been used in this arena. At present, several studies have indicated that these agents may be helpful in the adult population; however, their use in children has not been well described and is not recommended.48
Intuitively, the association between myocarditis and a potential viral etiology suggests that antiviral agents might have benefits. Several case reports document successful treatment of myocarditis with antiviral drugs.29,34 Some centers have used pleconaril (a drug that inhibits Coxsackie virus B) in the treatment of myocarditis; however, the timing of a patient's initial presentation most commonly occurs during the secondary immune response and autoimmunity stage when antiviral therapy is of limited or no therapeutic benefit.12
Mechanical Support
In some cases of acute myocarditis, circulatory failure may occur despite aggressive medical intervention. As a result, mechanical support may be necessary to provide adequate circulatory support while awaiting the recovery of usual cardiac function.50 Mechanical support should be considered early when patients require blood pressure support with high-dose dopamine or epinephrine.25 The best described model of mechanical support in children is extracorporeal membrane oxygenation (ECMO); however, experience with ventricular assist devices (VADs) is increasing. Although initially used as a bridge to transplant when medical measures were insufficient, ECMO (see Figure 5) and VADs are now being used in an increasingly supportive role while awaiting the return of adequate cardiac function.51 Although not without significant risk, such as stroke and embolization injury, the overall survival of patients requiring mechanical support for myocarditis may be as high as 60%.52 ECMO, which can utilize peripheral cannulation, may be the first line of mechanical therapy; however, it has limitations as a long-term means of support of myocardial function before the risk of complications significantly increases (usually 10-14 days). Once the decision is made that myocardial damage is irreversible, transplantation should be considered and the patient may be switched to an assist device that requires a ventriculotomy for cannula insertion.53 The decision to place these patients on mechanical support, although made when standard therapy is failing, will vary for each individual but eventually may be indicated for early initiation.
 |
Outcomes
The long-term outcome for patients with acute myocarditis is somewhat encouraging, with survival near 70-80%.28,37 In patients who progress to the degree that they need mechanical support, many have an improvement in cardiac function to normal values.51
Transplantation
In some cases, myocardial damage is irreversible and progressive despite aggressive medical therapy and mechanical support. Cardiac transplantation becomes the treatment of last resort. In data culled from the worldwide adult experience, myocarditis is implicated in up to 10% of transplants.53 Initial results suggested that survival after transplant due to myocarditis was shorter than when performed for other causes, but more recent studies suggest that survival is similar regardless of the etiology. One exception to this is giant cell myocarditis, which also can be transmitted to the newly transplanted heart. If demonstrated by biopsy, these patients require more aggressive immunosuppressive therapy than typically is used in post-transplant patients.53
Cardiac transplantation carries its own risk of complications, including the constant risk of rejection (both acute and chronic), post-transplant coronary artery disease, malignancy, and hypertension.53 However, long-term survival in the current era of cardiac transplantation approaches 70% for patients with a "non-congenital" etiology.54 Although the majority of transplant recipients return to normal activity as compared with their peers, there appears to be psychosocial implications that are not fully explained54 and affect quality of life post-transplant.
Summary
Acute myocarditis presents a challenging picture, both in terms of diagnosis and treatment. Considering the possibility of myocarditis as the cause of a patient's presenting symptoms is an important first step to diagnosis. The etiologies are many, and the ability to identify the exact cause may be difficult. Current therapies generally are supportive rather than curative, and data reflecting the pediatric experience is limited. Research in immunosuppressive therapy and the use of assisted mechanical circulation is providing new avenues for treatment of these difficult patients. Fortunately, the majority of the pediatric patients diagnosed with myocarditis ultimately recover if prompt and aggressive treatment is instituted, allowing them to lead normal lives after the illness.
References
1. Deshpande J, Tobias J, Johns J. Inflammatory heart disease. In: Nichols D, Cameron D, eds. Critical Heart Disease in Infants and Children. 1st ed. New York: Mosby; 1995:942-947.
2. Shekerdemian L, Bohn D. Acute viral myocarditis: epidemiology and pathophysiology. Pediatr Crit Care Med 2006;7(6 Suppl):S2-S7.
3. Martin AB, Webber S, Fricker FJ, et al. Acute myocarditis. Rapid diagnosis by PCR in children. Circulation 1994;90:330-339.
4. Girones N, Carrasco-Marin E, Cuervo H, et al. Role of Trypanosoma cruzi autoreactive T cells in the generation of cardiac pathology. Ann N Y Acad Sci 2007;1107:434-444.
5. Bohm N. Adrenal, cutaneous and myocardial lesions in fulminating endotoxinemia (Waterhouse-Friderichsen syndrome). Pathol Res Pract 1982;174:92-105.
6. Rubenstein JS, Noah ZL, Zales VR, et al. Acute myocarditis associated with Shigella sonnei gastroenteritis. J Pediatr 1993;122:82-84.
7. Labianca R, Beretta G, Clerici M, et al. Cardiac toxicity of 5-fluoracil: a study on 1083 patients. Tumori 1982;68:505-510.
8. Mills BA, Roberts RW. Cyclophosphamide-induced cardiomyopathy: a report of two cases and review of the English literature. Cancer 1979;43:2223-2226.
9. Schiff R, Wurzel C, Brunson S, et al. Death due to chronic syrup of ipecac use in a patient with bulimia. Pediatrics 1986;78:412-416.
10. Hiraishi S, Yashiro K, Oguchi K, et al. Clinical course of cardiovascular involvement in the mucocutaneous lymph node syndrome. Relation between clinical signs of carditis and development of coronary arterial aneurysm. Am J Cardiol 1981;47:323-330.
11. Stevens PJ, Ground KE. Occurrence and significance of myocarditis in trauma. Aerosp Med 1970;41:776-780.
12. Wheeler DS, Kooy NW. A formidable challenge: the diagnosis and treatment of viral myocarditis in children. Crit Care Clin 2003;19:365-391.
13. Wren C, O'Sullivan JJ, Wright C. Sudden death in children and adolescents. Heart 2000; 83:410-413.
14. Basso C, Corrado D, Thiene G. Cardiovascular causes of sudden death in young individuals including athletes. Cardiol Rev 1999;7:127-135.
15. Wisten A, Forsberg H, Krantz P, et al. Sudden cardiac death in 15-35-year olds in Sweden in 1992-99. J Intern Med 2002;252:529-536.
16. Dettmeyer R, Schlamann M, Madea B. Immunohistochemical techniques improve the diagnosis of myocarditis in cases of suspected sudden infant death syndrome (SIDS). Forensic Sci Int 1999;105:83-94.
17. Dettmeyer R, Baasner A, Schlamann M, et al. Role of virus-induced myocardial affections in sudden infant death syndrome: a prospective postmortem study. Pediatr Res 2004;55:947-952.
18. Keith JD, Rowe RD, Vlad P. Heart Disease in Infancy and Childhood. 3rd ed. New York: Macmillan; 1978:928-931.
19. Checchia PA, Kulik TJ. Acute viral myocarditis: Diagnosis. Pediatr Crit Care Med 2006;7(6 Suppl):S8-S11.
20. Soongswang J, Durongpisitkul K, Ratanarapee S, et al. Cardiac troponin T: Its role in the diagnosis of clinically suspected acute myocarditis and chronic dilated cardiomyopathy in children. Pediatr Cardiol 2002;23:531-535.
21. Soongswang J, Durongpisitkul K, Nana A, et al. Cardiac troponin T: A marker in the diagnosis of acute myocarditis in children. Pediatr Cardiol 2005;26:45-49.
22. Gardiner AJ, Short D. Four faces of acute myopericarditis. Br Heart J 1973;35:433-442.
23. Hohn AR, Stanton RE. Myocarditis in children. Pediatr Rev 1987;9:83-88.
24. Mason JW, O'Connell JB. Clinical merit of endomyocardial biopsy. Circulation 1989;79:971-979.
25. Schwartz SM, Wessel DL. Medical cardiovascular support in acute viral myocarditis in children. Pediatr Crit Care Med 2006;(6 Suppl):S12-S16.
26. English RF, Janosky JE, Ettedgui JA, et al. Outcomes for children with acute myocarditis. Cardiol Young 2004;14:488-493.
27. Lee KJ, McCrindle BW, Bohn DJ et al. Clinical outcomes of acute myocarditis in childhood. Heart 1999;82:226-233.
28. McCarthy RE III, Boehmer JP, Hruban RH, et al. Long-term outcome of fulminant myocarditis as compared with acute (nonfulminant) myocarditis. N Engl J Med 2000;342:690-695.
29. Kawai C. From myocarditis to cardiomyopathy: mechanisms of inflammation and cell death: learning from the past for the future. Circulation 1999;99:1091-1100.
30. Lange LG, Schreiner GF. Immune mechanisms of cardiac disease. N Engl J Med 1994;330:1129-1135.
31. Merlet P, Dubois-Rande JL, Adnot S, et al. Myocardial beta-adrenergic desensitization and neuronal norephedrine uptake function in idiopathic dilated cardiomyopathy. J Cardiovasc Pharmacology 1992;19:10-16.
32. Brodde OE. Physiology and pharmacology of cardiovascular catecholamine receptors: implications for treatment of chronic heart failure. Am Heart J 1990;120(6 Pt 2):1565-1572.
33. Buser PT, Camancho SA. The effect of dobutamine on myocardial performance and high energy phosphate performance at different stages of heart failure in cardiomyopathic hamsters: a 31P MRS study. Am Heart J 1989;118:86-91.
34. Monard ES, Baim DB, Smith HS, et al. Milrinone, dobutamine, and nitroprusside: comparative effects in hemodynamics and myocardial energetics in patients with severe congestive heart failure. Circulation 1986;73(Suppl III):168.
35. Feldman AM, McNamara D. Myocarditis. N Engl J Med 2000;343:1388-1398.
36. Malsumori A, Igata H, Ono K, et al. High doses of digitalis increase the myocardial production of proinflammatory cytokines and worsen myocardial injury in viral myocarditis: a possible mechanism of digitalis toxicity. Jpn Circ J 1999;63:934-940.
37. Burch M. Heart failure in the young. Heart 2002;88:198-202.
38. Masse L, Antonacci M. Low cardiac output syndrome: identification and management. Crit Care Nurs Clin North Am 2005;17:375-383.
39. Wessel DL. Managing low cardiac output syndrome after congenital heart surgery. Crit Care Med 2001;29(10 Suppl):S220-S230.
40. Hoffman TM, Wernovsky G, Atz AM, et al. Prophylactic intravenous use of milrinone after cardiac operation in pediatrics (PRIMACORP) study. Prophylactic Intravenous Use of Milrinone After Cardiac Operation in Pediatrics. Am Heart J 2002;143:15-21.
41. Hoffman TM, Wernovsky G, Atz AM, et al. Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation 2003;107:996-1002.
42. Hunt SA, Abraham WT, Chin WH, et al. ACC/AHA 2005 Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure): developed in collaboration with the American College of Chest Physicians and the International Society for Heart and Lung Transplantation: endorsed by the Heart Rhythm Society. Circ 2005;112:e154-e235.
43. Bruns LA, Chrisant MK, Lamour JM, et al. Carvedilol as therapy in pediatric heart failure: an initial multicenter experience. J Pediatr 2001;138:505-511.
44. Naughton MT, Rahman MA, Hara K, et al. Effect of continuous positive airway pressure on intrathoracic and left ventricular transmural pressures in patients with congestive heart failure. Circulation 1995;91:1725-1731.
45. English RF, Janosky JE, Ettedgui JA, et al. Outcomes for children with acute myocarditis. Cardiol Young 2004;14:488-493.
46. Kazatchkinc MD, Kaveri SV. Immunomodulation of autoimmune and inflammatory diseases with intravenous immune globulin. N Engl J Med 2001;345:747-755.
47. Orange JS, Hossny EM, Weiler CR, et al. Use of intravenous immunoglobulin in human disease: a review of evidence by members of the Primary Immunodeficiency Committee of the American Academy of Allergy, Asthma and Immunology. J Allergy Clin Immunol 2006;117(4 Suppl):s525-s553.
48. Drucker NA, Colan SD, Lewis AB, et al. Gamma-globulin treatment of acute myocarditis in the pediatric population. Circulation 1994;89:252-257.
49. Feltes TF, Adatia I Immunotherapies for Acute Viral Myocarditis in the Pediatric Patient. Pediatr Crit Care Med 2006;7(6 Suppl): s17-s20.
50. Hia CP, Yip WC, Tai BC, et al. Immunosuppressive therapy in acute myocarditis: an 18 year systematic review. Arch Dis Child 2004;89:580-584.
51. Bohn D, Macrae D, Chang A. Acute viral myocarditis: mechanical circulatory support. Pediatr Criti Care Med 2006;7(6 Suppl):s21-s24.
52. The ELSO Registry: Cardiac Support. The Extracoporeal Life Support Organization, 2006.
53. Moloney ED, Egan JJ, Kelly P, et al. Transplantation for Myocarditis: A Controversy Revisited. J Heart Lung Transplant 2005;24:1103-1110.
54. Webber SA, McCurry K, Zeevi A. Heart and lung transplantation in children. Lancet 2006;368:53-69.
Myocarditis may have a wide variety of etiologies and presentations and the clinician must have an awareness of this disease process to adequately screen patients. Although viral infections are the most common etiology, noninfectious etiologies also may occur. This article provides a review of the diverse presentation, assessment, and management of a child with myocarditis.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
