Geriatric Emergency Medicine Reports Supplement: Malignancy Complications in the Elderly
Author: Fitzgerald Alcindor, MD, FACEP, Assistant Director, Depart-ment of Emergency Medicine, North Shore University Hospital at Forest Hills, Queens, NY; Emergency Medicine Instructor, New York University School of Medicine Emergency Medicine Residency Program at North Shore University Hospital at Manhasset.
Peer Reviewer: Verena T. Valley, MD, Associate Professor, Department of Emergency Medicine, University of Mississippi Medical Center, Jackson.
Malignancy in any age group is a harbinger to potential complications. Complications of malignancy cross all age groups, although many features are common in both adult and geriatric patients. Cancer occurs more frequently in patients older than age 65, and ranks second to cardiovascular disease in mortality rate for that age group.1,2 Fifty percent of all cancers occur in patients older than age 65, and it is predicted that this figure will increase to 60% by the year 2010.2 Advanced age not only predisposes to cancer, but complications also can affect the elderly in unique ways compared to pediatric and adult populations. For example, the risk of thromboembolism is higher in the elderly; in the presence of malignancy, that risk is further increased in the older patient. Elders reached 13.1%3 of the U.S. population in the year 2000, and with advances in surgery, chemotherapy, radiation, and critical care, more elderly patients are seeking emergency department (ED) care for malignancy-related complications.—The Editor
Introduction
Malignancy complications in the elderly may present as subtly as altered mental status or as dramatically as seizures or frank bleeding. Emergency physicians should aggressively approach and manage elderly patients with an acute or an established malignancy-related complication; there is a 17% increase in cancer mortality in patients older than age 55, yet cancer mortality has declined in the younger population in recent years.1,2
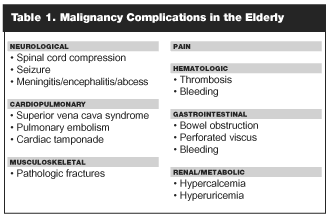
This article will review malignancy complications in the elderly from an organ system approach, outlining the physiological changes of aging and how they are related to the consequences of cancer when relevant. (See partial list in Table 1.) As many complications are similar in both adults and the elderly, features of disease, the differential diagnosis, and management strategies that are unique to the elderly will be emphasized. It should be noted that, although older age predisposes patients to cancer, cancer is not an inevitable consequence of normal aging.2
 |
Neurologic Complications in the Elderly
Brain function in the healthy older adult remains mostly unchanged with age.6 However, brain weight decreases with age, and functions such as memory, attention, and language variably decline in later years.7 Neurological manifestations of malignancy occur in 15-20% of cancer patients.8 Altered mental status is the most frequent neurological presentation and presents a challenge to the emergency physician, as the differential diagnosis is broad. The etiologies include cerebral herniation, central nervous system (CNS) infections, increased intracranial pressure, and toxic/metabolic disorders. Spinal cord compression (SCC) and seizures are common complications and warrant further examination.
Spinal Cord Compression. SCC is the second most common neurologic complication of malignancy second to brain metastases. It is found in 5% of cancer patients at autopsy.9 The lifetime risk of this potentially devastating event for a cancer patient is 1%.9 Most patients with SCC are known to have cancer. However, in 10% of SCC patients, it is the first sign of the presence of a neoplasm. Every year, 18,000-20,000 cases of SCC occur in the United States.10 The compression may be epidural, intramedullary, or extradural. The tumor initially spreads hematogeneously and leads to compression as its bulk increases. Anterior compression, the most common type, is secondary to posterior extension of a vertebral body mass into the epidural space (25%) or vertebral body collapse (75%), leading to vascular compromise of the spinal cord (edema, ischemia, and necrosis).11 SCC occurs most frequently with neoplasm of the breast (20.6%), neoplasm of the lung (17%), and lymphoma.3 The onset of cord impingement typically occurs earlier in lung carcinoma (within 3 months of diagnosis in 87% of patients) than in breast cancer (19 years after the initial diagnosis).3
Cord compression most commonly affects the thoracic spine (70%), followed by the lumbosacral region (20%) and the cervical spine (10%).12 SCC usually is a gradual process, but it also may be sudden. The most significant prognostic sign is the patient’s premorbid neurologic status. Patients who are ambulatory are likely to fare better than those with prior neurologic impairment.11 The most common initial symptom is localized or radicular pain (70-95%). The pain of cord compression typically is dull, persistent, and not relieved by rest, distinguishing it from the pain of a herniated disc. Movement, sneezing, and neck flexion tend to worsen the discomfort. The second most common complaint is weakness, which is seen in 80% of patients. Patients may complain of difficulty climbing stairs or rising from chairs. The physician should observe the patient walking. Paresthesias, ataxia, and later urinary frequency, urgency, urinary retention, impotence, and constipation are common. The emergency physician can pinpoint the level of dysfunction by obtaining a detailed history and performing a careful neurologic exam.
The differential diagnosis of back pain in the elderly ED patient includes a search for mechanical/degenerative, visceral, vascular, neurogenic, psychogenic, infectious, traumatic, metabolic, inflammatory, and neoplastic causes. In general, low back pain is benign in the majority of patients; it has a serious etiology, such as a malignancy, in 5% of patients ages 20 to 55 years, and in 20% of patients older than age 55.13
Plain spine radiographs are diagnostic of vertebral metastases in 72-83% of patients, and should be obtained as the initial study.11 However, they may be normal in up 28% of cases, especially in patients with lymphoma. Computed tomography (CT) and magnetic resonance imaging (MRI) are more sensitive for detecting posterior lesions than plain radiographs. CT is more sensitive than MRI for assessing vertebral stability and bone destruction. MRI has replaced myelography and is very useful for planning radiotherapy and surgery.11 Obtain a complete blood count (CBC), metabolic profile, erythrocyte sedimentation rate (ESR), and urinalysis to further define the etiology of low back pain in the elderly.
Patients with late signs such as localized weakness, bowel or bladder incontinence, and ataxia require emergent management and portend a poor prognosis. The emergency physician should administer a loading dose of dexamethasone of 10 mg IV. The patient’s clinical situation will dictate surgery, radiation therapy, or chemotherapy. Surgical candidates are individuals with spinal instability, unknown malignancy type, deteriorating neurologic function during radiotherapy, recurrent cord compression, or compression due to bone fragments. Nonsurgical candidates are eligible for emergent radiotherapy. A radiation therapy consult should be obtained in the ED. Chemotherapy is applied to patients who are not eligible for surgery or radiotherapy. Recent studies have shown that 80% of ambulatory patients retain the ability to walk, vs. 20-60% of patients who were paretic at the time of treatment.4,5
An area of controversy in the literature involves anterior cord compression management via laminectomy.16,17 Currently, laminectomy is applied to posterior lesions, while other techniques are used for anterior decompression and spinal stabilization.18
Another area of controversy has been the role of high-dose corticosteroids in the management of cord compression. Corticosteroids clearly are needed in the management of cord and nerve root compression, and animal model studies have demonstrated improvement in function.18-20 However, several clinical trials have not shown an advantage of high-dose corticosteroids over the standard dose.18
Seizures. The most common brain tumors are metastatic lesions to the brain (20-40%).2 Seizures in cancer patients mainly are associated with cranial metastases and less commonly are seen with primary brain tumors. Seizure, especially if focal, in any patient older than age 40 signals a mass lesion and possible malignancy until proven otherwise.21
Patients may present with a headache, in status epilepticus, or postictally. A history of cancer or epilepsy may not be readily available to the emergency physician. Issues of noncompliance, recent chemotherapy, and radiation should be raised during the initial evaluation. As in all emergencies, airway, breathing, and circulation should be addressed. Give oxygen, obtain a dextrose stick, and administer thiamine 100 mg IV. Look for papilledema and signs of head trauma. Draw a CBC, electrolytes, coagulation profile, anticonvulsant levels, and blood cultures as appropriate. The physician should obtain a contrast head CT to rule out any masses or abcesses after a non-contrast head CT has been performed.
ED management in these patients is similar to that of other patients with seizures. Rapid stabilization with benzodiazepines such as diazepam (0.2 mg/kg IV at 2 mg/min) or lorazepam (0.1 mg/kg IV at 1-2 mg/min) may be necessary. A longer-acting anticonvulsant such as phenytoin (15 mg/kg in the elderly) or fosphenytoin (15-20 PE/kg at 100-150 mg PE/min) also can be administered. The physician should provide a bolus for non-therapeutic anticonvulsant levels and adjust maintenance dosage as needed. Indications for admission include new diagnosis of cancer, status epilepticus or recurrent seizures, hemodynamic instability, and brain metastases with significant edema and CNS infections. Patients also should be referred for radiotherapy to decrease the size of metastases.
Cardiac Complications
With aging, the cardiovascular system undergoes atherosclerotic changes that result in arrhythmias, myocardial ischemia/infarction, and loss of distensibility of the heart muscle. The older heart cannot tolerate major fluid loads, especially postsurgically.22 Cancer complications, such as superior vena cava syndrome (SVCS) and pericardial tamponade, when superimposed on these expected physiologic changes, can result in significant morbidity.
Superior Vena Cava Syndrome. SVCS occurs when there is obstruction of the thin-walled, low-pressure superior vena cava. Pressures in the superior vena cava can reach 200-500 cm H2O in SVCS and can translate into a dramatic clinical presentation.18 The increased pressure is a gradual process but may occur rapidly depending on the presence or absence of tributaries to the superior vena cava. Kinking of a central venous catheter may cause rapid onset of SVCS.23 Currently, malignancy is the etiology in 85-95% of cases.24 Bronchogenic carcinoma of the lung alone accounts for 75%, lymphoma 15%, and metastatic lesions make up 7%.18,23 It is surprising that Hodgkin’s lymphoma rarely causes SVCS although it commonly affects the mediastinum. Breast cancer is the most frequent metastatic disease that causes SVCS (11%).18 Nonmalignant causes are the culprit in SVCS in 10-25% of cases today.18 These include central venous catheters, including Swan-Ganz catheters, and total parenteral nutrition lines, pacemakers, LeVeen shunts, and substernal goiter.
The diagnosis of SVCS often is straightforward due to typical symptoms such as dyspnea (63%), facial and head swelling (50%), cough (24%), arm swelling (18%), chest pain (15%), and dysphagia (9%).18 Physicians should search for distention of the upper body: neck (66%), facial edema (46%), plethora (19%), conjunctival suffusion, and cyanosis (9%). Stoke’s sign (tightness of the shirt collar) also may be observed. Headache, altered mental status, vertigo, stupor, and syncope may occur due to cerebral edema.23 In the differential diagnosis, physicians should consider congestive heart failure (CHF), pericardial tamponade, and nephrotic syndrome. The physiology of aging differentiates little in the pathophysiology of SVCS in adults and the elderly with cancer.
Chest radiography is a useful diagnostic tool in SVCS; it reveals a mass in most cases. In one study, a normal chest x-ray was found in only 16% of patients.24 Chest radiographic findings include superior mediastinal widening (64% of cases), pleural effusion (26%), right hilar mass (12%), bilateral diffuse infiltrates (7%), cardiomegaly (6%), calcified paratracheal nodes (5%), and anterior mediastinal mass (3%).18 CT of the chest details the surrounding structures such as superior vena cava tributaries, the bronchi, and the spinal cord. At this time, MRI can delineate the vascular anatomy and give clues to etiology.23 Contrast venography is controversial in the diagnostic approach to SVCS because it is thought to increase bleeding at the puncture site in the setting of venous hypertension.18 Radionuclide Technetium-99m venography can show patency and flow pattern, although the images are not as well defined as contrast venography. The key to the diagnosis of the underlying cancer is mediastinoscopy or bronchoscopic cytology, although SVCS is diagnosed 50% of the time by history, physical exam, or chest radiography before the underlying malignancy is discovered.18 In the presence of a pleural effusion, thoracentesis is diagnostic 71% of the time. Other diagnostic modalities include supraclavicular lymph node biopsy and bone marrow biopsy.24
Treatment of SVCS includes symptomatic relief and treatment of the causative malignancy. Institute temporizing measures such as oxygen and head-of-bed elevation in the ED. Diuretic therapy and reduced salt intake can be palliative, but may risk further thrombosis associated with dehydration. Such issues are not relevant to the ED management of SVCS. Some clinicians advocate steroids for respiratory compromise, but there is no proven benefit.25 Combination chemotherapy and/or radiation therapy is considered the treatment of choice of small cell carcinoma of the lung.26 Therapy involves a multidisciplinary approach including the patient, family, primary care physician, surgeon, radiation oncologist, and oncologist. However, recurrence of SVCS has been reported after initial treatment.26 For unknown reasons, some small studies of small cell carcinoma have found SVCS to be a good prognostic sign.27 Radiation therapy is the treatment of choice for SVCS induced by non-small cell carcinoma of the lung, and is the initial treatment if the diagnosis is not yet established and the patient’s condition is worsening. Chemotherapy is the first line of treatment for small-cell carcinoma of the lungs.23 The average survival rate for malignancy-induced SVCS is five months vs. nine years for non-malignant causes.28 Catheter-induced SVCS can be managed with streptokinase, urokinase, or recombinant tissue-type plasminogen activator.29,30 Heparin and coumadin may help reduce the extent of thrombosis. Removal of the catheter, along with anticoagulation, is another treatment choice. Percutaneous transluminal angioplasty has achieved a 93% patency rate for catheter-induced SVCS.31
The prognosis for SVCS depends on the tumor type and is better for lymphoma than bronchogenic carcinoma. The overall prognosis for patients with SVCS caused by non-small cell carcinoma of the lung is poor, with a survival of 17% at one year and 2% at two years.18 Other complications include recurrent obstruction, thrombosis, esophageal invasion, cardiac tamponade, vocal cord paralysis, and spinal cord compression. Rarely, bleeding, pulmonary edema, and stent migration have occurred.23
Cardiac Tamponade. Cardiac tamponade is a potentially lethal complication of malignancy due to malignant pericardial effusion. It can be associated with radiation-induced pericarditis and fibrosis. Impaired cardiac filling leads to decreased cardiac output. The fluid accumulation within the pericardial sac progresses to diastolic dysfunction and ultimately to cardiac decompensation. The elderly patient is particularly prone to cardiac tamponade, especially in the presence of underlying cardiac disease. Bronchogenic carcinoma, breast cancer, lymphoma, leukemia, melanoma, and sarcomas are the most common causes.21 The typical history includes dyspnea and substernal chest pain, which can be confused for a variety of entities including CHF, pneumonia, acute coronary syndrome, or pulmonary embolism. Additional complaints of hiccups, hoarseness, nausea, vomiting, and epigastric pain often can be elicited. The patient may appear confused, pale, or diaphoretic, and the initial impression may be one of myocardial infarction. The physical exam may reveal tachypnea, Beck’s triad (muffled heart sounds, distended neck veins, and hypotension), facial swelling, ascites, and hepatic enlargement. Muffled heart sounds may not always be easy to auscultate in the noisy ED setting. Pulsus paradoxicus is an important finding that typifies cardiac tamponade, but is not pathognomonic. Physicians should keep in mind other causes of pulsus paradoxicus, such as chronic obstructive pulmonary disease, asthma, pulmonary embolism, restrictive cardiomyopathy, infarction of the right ventricle, and cardiogenic shock. Electrical alternans on the electrocardiogram (ECG), is specific for tamponade, but is not seen often. Additional electrocardiographic findings may include sinus tachycardia, non-specific ST-T wave changes, and ST elevation. Chest x-ray in malignant cardiac tamponade shows cardiomegaly, mediastinal enlargement, and a prominent pulmonary hilum.
The definitive diagnosis of cardiac tamponade is via emergent echocardiography. In addition to pericardial fluid, bedside echocardiography may show diffuse hypokinesis and diastolic collapse of the right atrium, right ventricle, and ventricular outflow tract. Central venous pressure measurements show equalized pressures throughout the cardiac chamber. Emergent management includes rapid isotonic fluid infusion, close monitoring, oxygenation, and pericardiocentesis. For non-malignant cardiac tamponade, removal of 50-100 mL of fluid can yield dramatic hemodynamic improvement until more definitive measures such as a pericardial window or pericardiectomy can be performed in the operating room. In malignant cardiac tamponade, much more fluid needs to be removed for clinically significant improvement. The outlook for any patient with malignancy-induced cardiac tamponade is poor.
Gastrointestinal Complications
Gastrointestinal (GI) complications of malignancy in the elderly such as abdominal pain, obstruction, bleeding, perforation, and infection may manifest themselves similarly to nonmalignant disorders. Several factors may confound the diagnosis in the elderly, including: difficulty in obtaining a history, memory deficits, alteration in mental status, abnormal hearing and speech, and altered perception of pain. Abdominal pain is the most common complaint and is considered the best signal to acute gastrointestinal pathology in the elderly cancer patient. Evaluation demands the same broad differential as other common nonmalignant conditions. Constipation is a frequent cause of abdominal pain and may be due to chemotherapeutic agents such as vincristine, narcotics, anticholinergics, iron, low-fiber diet, immobilization, dehydration, and electrolyte abnormalities.32 Tumor extension into the nerve supply of the gut may lead to intestinal pseudoobstruction, commonly termed Ogilvie’s syndrome. Right upper quadrant pain may be the initial presentation of metastatic lesions to the liver. The pain of liver metastases may radiate to the shoulder and cause diaphragmatic irritation. It also may be the occult manifestation of venoocclusive disease in the postsurgical bone marrow transplant patient or occur after intense chemotherapy (4% to 22%).33 Such patients are jaundiced, have hepatomegaly, ascites, neutropenia, thrombocytopenia, and may display signs of encephalopathy. Hepatic vein obstruction (the Budd-Chiari syndrome), secondary to hepatoma, adrenal carcinoma, renal cell carcinoma, or thrombosis, may have a similar presentation. Lymphoma causing splenomegaly may be the cause of left upper quadrant pain and left shoulder pain (Kehr’s sign). Rectus sheath hematoma in a thrombocytopenic patient or a patient on warfarin may elicit abdominal wall pain. Back pain may be a manifestation of enlarged retroperitoneal adenopathy, retroperitoneal hematoma, or pancreatitis due to cytotoxic medications (i.e., L-asparaginase, 6-mercaptopurine, and corticosteroids). Corticosteroids are known culprits to delays in the diagnosis of abdominal complications. Physical findings such as fever and leukocytosis are likely to be masked, and these patients are at a higher risk of localized perforations. In summary, abdominal pain in the elder cancer patient should elicit a broad differential diagnosis, as the etiologies are protean.
Intestinal obstruction occurs in 59-100% of patients with recurrent malignant disease, and is the indication for laparotomy in 33% of cancer patients.18 Colorectal, ovarian, and gastric malignancies account for two-thirds of cancer-related intestinal obstruction. The sites of obstruction include small bowel (59%), colorectal (29%), both sites simultaneously (5%), and duodenum and stomach together (7%).34 The presentation of bowel obstruction does not differentiate malignant from nonmalignant causes of obstruction, crampy abdominal pain that is initially intermittent, then becomes more constant as the obstruction progresses, and is associated with nausea, vomiting (usually bilious), abdominal distention, and anorexia. The patient may look ill, dehydrated, and even cachectic. There may not be an abdominal scar as a remnant of prior surgery. The initial examination may reveal distention, localized tenderness (becoming diffuse with time), hyperactive bowel sounds. A mass may not be palpable. Important physical examination keys include searching for a mass, fecal impaction, or tenderness. Obtain a CBC, electrolytes, coagulation profile, urinalysis, a chest radiograph, and abdominal series. The patient should be hydrated with isotonic saline and have a nasogastric tube inserted. Colonoscopic decompression is needed when cecal distention is 12-14 cm. Conservative management with a nasogastric tube, bowel rest, hydration, serial abdominal exams (preferably by the same physician), and abdominal radiographs is generally indicated, since the majority of partial small bowel obstructions will relieve with this approach.18 However, in a few studies, conservative management in cancer patients yielded a 12-29% resolution rate within 3-9 days and a recurrence rate of 32-45% necessitating operative intervention.35,36 Surgery is indicated to relieve the symptoms and prevent bowel strangulation or gangrene if conservative management fails after three or more days, or if leukocytosis, fever, increased pain, distention, or peritonitis occur. Surgery is effective in 55-96% of cases. The recurrence rate of obstruction after surgical intervention is 9-33%. Morbidity and mortality rates are 31% and 16%, respectively. The mortality rate for emergency laparotomy is highest (38-54%) in patients ages 70 and older, whereas only 3-7% of the older surgical candidates die after an elective procedure.35,36 The mortality difference is most reflective of pre-morbid conditions. The extent of the operation and related treatment regimens should not depend on age alone, but rather should take into account the patient’s physiology, health status, and wishes.37
GI perforation is the indication for laparotomy in 33% of cancer patients. Lymphoma causes about 50% of GI perforations. The patient typically presents with abdominal pain, distention, fever, and peritoneal signs. Free air under the diaphragm is visualized about 50-75% of the time on an upright chest radiograph. The flat and upright abdominal films, along with a left lateral decubitus view, increase the sensitivity for free air detection. Sensitivity also can be increased by instilling 200 cc of air via a nasogastric tube prior to obtaining the radiograph. A CT scan of the abdomen should be obtained to confirm the diagnosis in equivocal cases when clinical suspicion is high. The absence of free air on plain films does not preclude perforation. Intravenous hydration, treatment of shock, repletion of electrolytes, broad-spectrum antibiotics, and immediate surgical consultation are the necessary ED steps.
Postoperative hemorrhage is the most common cause (67%) of bleeding in the cancer patient. According to endoscopic studies, malignancy is the culprit in 12-27% of GI bleeds in cancer patients, while gastritis, ulcer disease, and esophagitis (44-76%); and esophagitis and varices (11-17%) account for the rest.38 Other uncommon causes are Mallory-Weiss tears from vomiting due to chemotherapy, hemobilia due to hepatobiliary tumors or instrumentation, aortoduodenal fistula from a perforated metastastic tumor, and iatrogenic liver and spleen injuries after thoracentesis and percutaneous needle biopsy.39 Emergency management should include a rapid assessment of the ABCs (airway, breathing, and circulation) followed by intravenous fluid, packed red blood cells, and H2 antagonists as indicated. Coagulopathies should be corrected and any causative drugs such as aspirin, warfarin, heparin, nonsteroidal antiinflammatory agents, and alcohol discontinued. A surgical or gastroenterology consult for endoscopy should be obtained. A radionuclide-labeled erythrocyte scan or angiogram may be helpful if the endoscopy is inconclusive or if bleeding continues. Angiography is both diagnostic and therapeutic in that the bleeding site may be identified and gelfoam embolization may be performed. If the endoscopic work-up is inconclusive, an abdominopelvic CT to search for a retroperitoneal hematoma should be considered. Patients who remain unstable may require operative management.
Other GI complications of malignancy include abdominal abcesses and radiation enteritis. Abcesses are a common postoperative event and respond well to percutaneous drainage or surgery if there is no improvement with intravenous antibiotics within 24 hours. Anorectal infections are common and are managed in the standard fashion. Perirectal lesions that fail to heal need a biopsy to rule out carcinoma. Radiation enteropathy is an entity characterized by nausea, vomiting, intermittent crampy abdominal pain, weight loss, and watery or bloody diarrhea in a patient with a history of abdominal radiation.18 Treatment is supportive.
Renal/Metabolic Complications
With aging, there is a decrease in renal function and mass, glomerular filtration rate, and creatinine clearance, thereby affecting drug excretion and making the kidneys more susceptible to ischemia from anesthesia and major surgery.22 There also is decreased volume of distribution of water-soluble drugs.2
Hypercalcemia. Hypercalcemia represents the most frequent metabolic complication found in cancer patients. It is encountered in 20% of cancer patients.40 It most commonly is associated with multiple myeloma and breast cancer (40%), and is frequently seen in non-small cell carcinoma of the lung. It is an uncommon presentation in colon, prostate, and small cell carcinoma. The pathophysiology involves osteolysis from bony metastases and tumor production of parathyroid-like hormones, with the net effect being an elevated calcium level. Prostaglandins and other peptides also have been implicated. Any patient with an acute altered mental status may well be suffering from carcinoma-induced hypercalcemia. Other symptoms of hypercalcemia are vague and include anorexia, nausea, vomiting, constipation, fatigue, weakness, abdominal pain, polyuria, polydypsia, and memory loss. The clinical presentation is not directly related to the serum level. Many of these symptoms are so common in the elderly emergency patient that even the astute emergency physician may fail to associate them with hypercalcemia.
In addition to a serum calcium level, laboratory analysis should include electrolytes and renal function testing to look for renal insufficiency. An ECG should be obtained with emphasis on a shortened QT syndrome, dysrhythmias, and digitalis toxicity. Management includes ECG monitoring, saline hydration to correct dehydration and increase renal blood flow, and treatment of associated hypokalemia or hypomagnesemia. Furosemide 1-3 mg/kg may further enhance renal excretion of calcium after adequate urine output has been achieved. Avoid fluid overload in the frail elderly with poor cardiopulmonary reserve. To avoid hypovolemia and maintain fluid balance, the use of furosemide is limited to the patient who has been appropriately rehydrated (e.g., urine output of 100 mL/hr).
Acute therapy directed toward calcium excretion is now the standard of care. Bisphosphonates inhibit bone resorption by osteoclasts and have shown clinical efficacy. Examples include oral etidronate, clodronate, and intravenous pamidronate, as well as galium nitrate, an inhibitor of bone resorption (for resistant hypercalcemia).41,42 These drugs are not used in the ED. Calcitonin rapidly (onset of action 2-4 hours) increases calcium excretion by inhibiting bone resorption, however its peak action is at 48 hours and falls off afterwards. It is being used in combination with pamidronate and galium nitrate. Its role in the ED setting is not established. Plicamycin (mithramycin) is cytotoxic to osteoclasts; however, its 48-hour onset of action and associated thrombocytopenia and hepatic and renal toxicity preclude its use in the initial management of hypercalcemia. Aspirin and indomethacin act as inhibitors of prostaglandins to lower the serum calcium level. Corticosteroids are reserved for steroid-responsive malignancies such as breast carcinoma, myeloma, and lymphoma after oncologic consultation has been obtained. The ultimate treatment of malignancy-related hypercalcemia is the treatment of the underlying malignancy.
Most patients with serum calcium levels greater than 12 mg/dL do not respond to intravenous hydration and diuretics alone and often require treatment with a bisphosphonate or gallium nitrate. Calcitonin 8 U/kg may be injected IM every six hours for calcium level of 15 mg/dL associated with coma or cardiac arrythmias. Marked hypercalcemia associated with renal insufficiency requires hemodialysis. The patient should be admitted if the serum calcium is greater than 12 mg/dL, and is accompanied by nausea, vomiting, dehydration, altered mental status, renal insufficiency, cardiac arrhythmia, obstipation, and ileus, or if the patient lives alone or has difficulty accessing medical care.18
Hyperuricemia. Hyperuricemia is a malignancy complication found mostly after chemotherapy or radiation therapy of lymphomas and leukemias and myeloproliferative disorders. It also may be encountered with hyperparathyroidism, sarcoidosis, psoriasis, renal failure, and gout. Hyperuricemia also can occur as a chronic sequela of thiazides, ethacrynic acid, and furosemide. The underlying mechanism is either excess production or decreased excretion of uric acid, especially in the presence of underlying renal impairment, as may be present in the elderly. It may be an incidental finding, or it may be seen in an acutely oliguric patient after chemotherapy. Hyperuricemia may be asymptomatic. Affected patients are prone to acute renal failure secondary to intrarenal obstruction. Ultrasound or CT scans are useful to assess for obstructive uropathy. Patients also are at risk for acute hyperuricemic nephropathy, uric acid nephrolithiasis, and gouty nephropathy.
The key to management is to keep patients well hydrated (aim for daily urine output of 2 liters), ideally before the start of chemotherapy. Oral allopurinol 300-900 mg ideally should be started 48-72 hours before cancer therapy. Withdrawal of offending drugs should be instituted. Alkalinization of the urine to a pH above 7 is an additional step. Give colchicine 0.6 mg PO bid if a history of gouty arthritis is present. Further alkalinization can be obtained with acetozalamide 1 gram IV, a carbonic anhydrase inhibitor. Oliguria can be ameliorated with 12.5 gm of 20% mannitol. If the patient is still oliguric, dialysis is a last resort.18
The ED management of hyperuricemia is the same regardless of the etiology. Aggressive hydration is paramount, with careful attention to fluid overload in the frail elderly patient. The best treatment remains treatment of the underlying malignancy and prevention of this complication.
Tumor Lysis Syndrome. Tumor lysis syndrome is a constellation of malignancy-induced metabolic abnormalities resulting from the accumulation of intracellular products in the bloodstream. Hyperuricemia, hyperkalemia, hyperphosphotemia, and hypocalcemia are the end products. Such patients are at risk for dangerous arrhythmias (hyperkalemia), acute renal failure (hyperphosphotemia), muscle cramps, and tetany (hypocalcemia). The tumors most commonly responsible are bulky, highly proliferative malignancies that are very responsive to cytotoxic regimens. They include lymhomas, leu-kemias, and solid tumors.43,44 This syndrome also has been linked to potent myelosuppressive agents and other agents such as methotrexate, tamoxifen, interferon alpha, and cladribine. Early diagnosis and aggressive management are key. Hydrate patients at risk prior to initiating chemotherapy. Check and correct electrolytes, uric acid, phosphorus, and calcium as often as the clinical situation dictates for 3-4 days after receiving chemotherapy. ED management involves hydration, ECG monitoring, treating the presenting electrolyte abnormalities, and consultation with the patient’s oncologist.
Lactic Acidosis. Lactic acidosis is a rare metabolic complication of malignancy. It generally is associated with leukemia and lymphoma.45,46 Its onset usually signifies progression of a hematologic malignancy. Patients with solid tumors usually have hepatic metastases once lactic acidosis occurs. The clinical presentation may vary from vague symptoms such as tachycardia, weakness, nausea, and stupor to hyperventilation, hypotension, and elevated lactic acid. Differential diagnosis includes sepsis, mesenteric ischemia, and other causes of lactic acidosis. The prognosis is poor for patients with lactic acid levels greater than 4 mEq/L, and the outcome depends on treatment of the underlying cancer. Sodium bicarbonate therapy does not appear to affect survival.18
Hypoglycemia. Hypoglycemia in cancer is usually the result of insulin-producing islet cell tumors. The symptoms are the same as those seen in hypoglycemia of other etiologies: weakness, dizziness, diaphoresis, and nausea. Patients also may present with seizures, coma, and varying neurologic deficits. Malignancy-induced hypoglycemia may be derived from the secretion of insulin-like substances, excessive tumor utilization of glucose (by large tumors), decreased gluconeogenesis, or lack of counter-regulatory hormonal action such as growth hormone.47 Anti-tumor therapy should be aimed at the underlying malignancy, although little efficacy has been shown in halting the development of hypoglycemia. Mild hypoglycemia may be approached by increasing the frequency of meals; corticosteroids and intravenous glucose infusions may provide symptomatic relief in more severe cases.
Vascular/Hematologic Complications
It should be emphasized that hematopoiesis, unlike most organ systems in the elderly, essentially remains the same as one ages. The volume of packed red cells, total leukocytes, neutrophils, lymphocytes, platelets, and the iron turnover rate in the healthy older person is no different from that of the young adult .48 However, the incidence of anemia increases in the elderly patient with concurrent illness, implying that illness causes a dysregulation of hematopoiesis.49
Thrombosis. Malignancy and older age are known risk factors for thrombosis. Other risk factors that are common to older patients include immobility, tobacco use, diabetes, and CHF. Mucin-producing tumors of the gastrointestinal tract and carcinomas of the lung, breast, ovary, and primary brain are particularly prone to thrombosis. Because of its prevalence, lung cancer is the tumor most frequently associated with thromboembolic disease. Malignancy creates a hypercoagulable state. The classic pathogenetic factors still play a critical role today, including: abnormalities in blood flow, blood composition, and blood vessel wall, as advocated by Virchow. Thromboembolism has been mentioned as one of the earliest signs of occult malignancy.50 The mediators of the hypercoagulable state include tumor cell procoagulants, procoagulant activities induced by host response to tumor, platelet aggregation, and endothelial cell activity. The presence of fibrin in the vicinity of a tumor is felt to be a nidus for tumor growth, metastasis, and a barrier to host humoral defenses. Thrombocytosis is encountered 30-60% of cancer patients, yet the absolute platelet count is not the underlying mechanism for thrombosis. It is thought to be induced by increased platelet reactivity via clonal abnormalities or tumor cell-platelet interactions.51
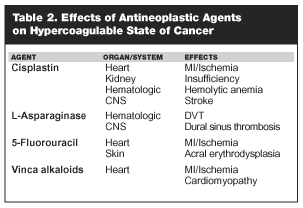
Certainly, surgery accompanied by immobilization is a major setting for thrombosis. The hypercoagulable state induced by malignancy also is maintained by the antineoplastic agents used to treat the various malignancies. For example, myocardial ischemia/infarction secondary to vasospasm and/or thrombosis has been observed with the vinca alkaloids, 5-fuorouracil, cisplastin, etoposide, and bleomycin in combination with vinblastine or cisplastin.52 Cerebral vascular accidents can occur due to cisplastin and L-asparaginase. L-asparaginase has been implicated in dural sinus thrombosis, and has been linked to deep venous thrombosis (DVT). The underlying mechanism is depletion of plasma asparagines leading to inhibition of protein synthesis (primarily a reduction of antithrombin III). High-dose chemotherapy after bone marrow transplant increases the risk of thrombosis. After bone marrow transplant, low levels of proteins C and S and antithrombin III have been observed in one study.53 However, it is uncertain whether a low protein C level can be used as a reliable marker for possible venoocclusive disease. Other agents implicated include tamoxifen (antiestrogen with weak estrogenic properties) and corticosteroids.53 Several studies have shown a higher risk for occult malignancy in patients with idiopathic DVT compared with patients with secondary DVT (DVT due to known risk factors/etiologies).50,54
Thrombosis occurs in 15% of cancer patients. The diagnosis of thromboembolic disease in the elderly cancer patient is the same as for the general population. Knowledge of the risk factors for thrombosis is essential. A thorough physical exam focusing on edema, venous cords, varicose veins, and tenderness is in order. The diagnostic approach also is the same as for noncancer patients. Although ascending venography is considered the criterion-standard and is reliable for both proximal and distal thrombosis, its use is limited because of patient discomfort, lack of portability, risk of contrast reaction, and precipitation of thrombosis. For these reasons, noninvasive tests are advocated as initial studies. Impedance plethysmography, Doppler ultrasound, real-time (B-mode) ultrasonography, and duplex scanning have a higher sensitivity for proximal than for distal DVT.55 If suspicion remains high, subsequent venography or a repeat non-invasive test (e.g., duplex scan) in 2-3 days may be indicated. MRI has shown similar accuracy to venous Doppler and venography in the assessment of DVT, but it is not widely used.55
Upper extremity thrombosis due to venous obstruction is seen more frequently in the cancer patient as a result of vascular injuries from intravenous catheters and the caustic effects of chemotherapy. Forty percent of Hickman catheters ultimately lead to venous thrombosis.56 Subclavian thrombosis is seen in 16% of completely implanted catheters and is manifested clinically by swelling and pain of the upper extremity, neck, shoulder, and chest wall. A triple-lumen catheter carries a higher risk of occlusion than a double-lumen catheter. Left-sided catheters tend to cause more complications, and a higher rate of venous obstruction has been observed when a catheter is placed above the level of the third thoracic vertebra.56 Large mediastinal tumors or bulky axillary lymphadenopathy also can cause venous obstruction. Diagnosis of upper extremity thrombosis can be made with either non-invasive studies or venography. A rare entity, the Trousseau syndrome, should be considered when a patient presents with recurrent migratory thrombosis of the superficial veins of the chest wall or the arms.52
Pulmonary embolism is another complication in elderly cancer patients. It is the complication of a DVT in 70% of patients. Again, a targeted history examining risk factors is indicated. Importantly, signs and symptoms may be nonspecific and a high level of suspicion is required. The work-up is the same as that used for the general population. Although pulmonary angiogram is the standard criterion, it usually is obtained if noninvasive tests and a ventilation/perfusion (V/Q) scan are nondiagnostic and clinical suspicion remains high. Spiral CT is another tool that can demonstrate a pulmonary embolism (PE) and is gaining popularity.51
Arterial thrombosis is much less common than its venous counterpart. It is primarily due to nonbacterial thrombotic endocarditis (NBTE) and is related to mucin-producing adenocarcinomas. The presenting signs are a direct result of arterial embolization, which can lead to end-organ failure. The brain, heart, spleen, and kidneys are the sites most commonly affected.51 Arterial thrombosis in the central nervous system can cause transient ischemic attacks, headaches, visual changes, and seizures, while peripheral arterial thrombosis results in digital ischemia or infarction.
The therapy for venoocclusive disease in the elderly cancer patient differs little from that used for the general population. Heparin, followed by warfarin, can be used. Low-molecular weight heparin currently is gaining wider use. Surgical patients with cancer need DVT prophylaxis. Those patients who have in-dwelling catheters may receive a 1 mg daily warfarin dose. An inferior vana caval filter (e.g., Greenfield filter) may be indicated in those with recurrent thrombosis while on warfarin, or if there is a contraindication to anticoagulation. One author has noted that resistance to anticoagulation is a sign of occult malignancy.50
Acral erythrodysplasia (painful erythema of the palms and soles) may result from standard or high-dose arabinoside, hydroxyurea, and prolonged use of 5-fluorouracil or doxorubicin.
Cancer patients also may complain of intense burning of the hands and feet, warmth, and erythema of the skin, especially when exposed to heat. This spectrum of signs and symptoms, called erythromelalgia, is reported to be very responsive to aspirin. It may due to a paraneoplastic syndrome in solid tumors, essential thrombocythenia, or polycythemia vera.57
Hyperviscosity Syndrome. Impairment of blood flow, massive leukocytosis, and delay in the clearance of clotting factors in cancer patients may predispose them to the hyperviscosity syndrome. The pathology extends to all organs. It is characterized by a variety of symptoms ranging from altered mental status, vertigo, headache, visual disturbances, seizures, bleeding, and cardiopulmonary dysfunction, to renal impairment and signs of thrombosis. This syndrome is most commonly secondary to Waldenstrom macroglobulinemia, followed by multiple myeloma.40 Various types of leukemias, cryoglobulinemia, polycythemia vera, and sickle cell disease account as other causes. The diagnostic approach should include electrolytes and coagulation profiles. A bedside test of relative serum viscosity has been described.58 Management includes hydration, diuresis, and emergent plasmapheresis and phlebotomy for severe cases, followed by initiation of chemotherapy.
Thrombocytopenia. Malignancy-induced thrombocytopenia is the most common reason for platelet transfusion. Bone marrow replacement by malignant cells, chemotherapy, radiation therapy, and infections (i.e., bacterial, viral, fungal, protozoal infections) are common etiologies. Infection-induced thrombocytopenia, however, usually does not cause major bleeding.55 Leukemia patients are at particular risk for thrombocytopenia and associated bleeding. There is increasing support for prophylactic platelet transfusion in leukemic patients with a platelet counts of 5000-10,000.59
Cancer patients also are at increased risk for heparin-induced thrombocytopenia, which typically occurs during the first several days of heparin administration. This occurs in patients on both full-dose heparin and from heparin flushes of venous access. The etiology is heparin-dependent IgG antibodies. Drug-induced thrombocytopenia should be followed by discontinuation of the responsible agent. Platelet counts usually begin to improve within 2-3 days. For patients requiring prolonged anticoagulation, warfarin should be instituted. Severe cases of thrombocytopenia associated with bleeding can further be managed by the use of intravenous IgG or Rh immune globulin (anti-D).60
Musculoskeletal Complications
Osteopenia in the elderly predisposes them to fractures of the long bones. The cancer-afflicted elderly patient is at further risk for fracture and spinal instability due to local metastases and tumor-induced osteopenia. Pathologic fractures can occur from seemingly trivial trauma. Long bones, especially the subtrochanteric region, are common sites of occurrence. The management of pathologic fractures includes rapid immobilization, analgesia with opioid narcotics, intravenous hydration, and prompt orthopedic and oncologic consultations.
Infectious Complications
The decline in the immune competence of the elderly predisposes them to a higher rate of infection.1 This section will concentrate on fever in the neutropenic cancer patient.
Fever is a common presentation in the elderly cancer patient. Its presence may be a marker to an underlying infection. Infection is a leading cause of death in the cancer patient (50-75%), and has a mortality rate of 20-50% if not promptly treated.63 Other etiologies include the underlying malignancy, cytokines, transfusion, thromboembolism, atelectasis, or drug therapy. One helpful distinction is that tumor-induced fever does not usually produce chills or rigors and the patient does not appear toxic, as compared to infectious etiologies of fever.63
A key parameter in the assessment of cancer patients with fever is the absolute neutrophil count (ANC), with neutropenia defined as an ANC less than 500/mm3. The duration of neutropenia along with certain risk factors dictates the likelihood of an infectious complication. High-risk groups include those with neutropenia of more than seven days duration, underlying malignancy, chemotherapy, altered cellular and humoral immunity, recent bone marrow transplantation, and the elderly and those with an indwelling catheter.64 Prolonged neutropenia is associated with potentially fatal infections from resistant bacteria, Candida species, and Asperigillus species. Bacteria, such as the coagulase-negative staphalococci, have become the dominant etiologies in the United States, secondary to increased indwelling catheter use.64 Staphylococcus aureus and Streptococci and Enterococci account for one-half of gram-positive isolates.64 Nonimmunized patients who are splenectomized because of Hodgkin’s disease are prone to Streptococcus pneumoniae, Neisseria meningitidis, and Haemophilus influenzae.
The greatest risk for the neutropenic febrile patient is gram-negative organisms, such as Pseudomonas aeruginosa, which can cause pneumonia and bacteremia. Neutropenic transplant patients on chemotherapy also are vulnerable to viruses, such as respiratory syncytial virus, adenovirus, parainfluenza virus, cytomegalovirus, and herpes simplex virus, as well as Pneumocystis carinii, mycobacteria, and fungi.
The evaluation of a febrile neutropenic patient takes on the same urgency as that of meningitis. Rapid history targeting risk factors and a thorough physical exam focusing on catheter site infection, and searching all sites for infection are necessary. In the majority of neutropenic patients, a site of infection is not found. The clinical evaluation should be followed by hematologic and coagulation, blood, urine, sputum, nasal, and pharyngeal, studies; and gentle rectal swabs; chest radiography; intravenous hydration; and prompt antibiotic therapy. Traditionally, broad spectrum antibiotic combination was advocated, but recent studies have evaluated monotherapy with ceftazidime alone.65,66 A recent study showed that carefully selected low-risk hospitalized neutropenic patients on chemotherapy fare well on oral ciprofloxacin and amoxicillin-clavulinate.67
Pain as a Complication of Cancer
Pain is a frequent companion to cancer. Nonmetastatic disease is associated with pain 15% of the time, whereas pain is seen in 74% of patients with metastases.1 Direct tumor invasion, such as in breast and prostate cancers, accounts for pain in 50% of patients with metastases, while nerve compression is the mechanism in the remainder. Cancer-related therapy causes pain in 25% of patients.
There are several issues related to pain management in the elderly. Normal aging is not significantly associated with impairment of pain sensation. The clinician’s lack of experience in pain assessment, the patient’s reluctance to report pain, and the cost of outpatient pain medications have contributed to the poor management of pain in the elderly cancer patient.61 Fear of addiction by patients and their families also is an important concern, but in one study of 12,000 patients treated with narcotics, only 0.1% became addicted.61 Management includes proper pain assessment (location, characteristics, radiation, co-related factors, and implications of the pain on the patient’s activities of daily living). Pain may result in sleep disorders, depression, and fear of cancer recurrence. A low-dose antidepressant and proper pain medications are in order. The physician should reassess the patient’s disease process and initiate the proper studies based on the physical exam. For example, increased low back pain may mean a collapsed vertebra with impending spinal cord compression, in which case an MRI is indicated. The absence of neurologic deficits does not preclude impending spinal cord compression.
One nonpharmacologic way of approaching pain is to treat the cancer itself. Examples include cancers that are responsive to chemotherapy or radiation (e.g., bone metastases). An exception is pancreatic cancer, which is not responsive to chemotherapy, but is helped by a celiac plexus block, for example. However, the toxicities of the treatments must be taken into account in the management of cancer pain.
The World Health Organization recently made some recommendations on managing pain: Pain relief must be a priority for the elderly cancer patient, and it must continue until relief is achieved.62 Additional modalities of pain control involve nerve blocks with chemicals or neuroablative methods, such as chordotomy (ablation of pain pathways in the spinal cord).
Toxicity of Chemotherapy
Unfortunately, the same chemotherapeutic agents used in the fight against cancer also are cytotoxic to normally dividing cells. (See Table 2.) The bone marrow is the most susceptible because its precursor cells have the most rapid turn over.32,68 Bone marrow suppression results in anemia, thrombocytopenia, and neutropenia. The agents least likely to suppress it include asparaginase, bleomycin, and vincristine. Chemotherapy may induce malignant cells to release procoagulant substances, along with patient risk factors, further increase the risk of thrombosis. Chemotherapy also causes thrombocytopenia, which may lead to bleeding complications that may result in thrombosis or disseminated intravascular coagulation. Hemolysis may be caused by melphalan, fluorouracil, and cisplatin.
 |
Gastrointestinal toxicity such as nausea and vomiting, diarrhea, and constipation are common. Extreme cases lead to dehydration, electrolyte imbalance, weakness, and weight loss. Acute nausea and vomiting usually take place within 24 hours after chemotherapy. The mechanism is unknown. The most emetogenic agents are cisplastin, cyclophosphamide, cytarabine, dacarbazine, actinomycin D, mechlorethamine, and streptozotocin. Serotonin, dopamine, and histamine blockers are helpful. The most effective therapy involves the 5-HT3 (serotonin) antagonists. Diarrhea and constipation are most commonly associated with fluorouracil and idarubicin.32 The causative agent must be stopped.
The mouth and throat also are susceptible to necrotic ulcers, mucositis, thrush, and herpes simplex in neutropenic patients on high-dose chemotherapy. Intensive oral hygiene is important to prevent hematogenous bacterial seeding. Oropharyngeal candidiasis may spread to the esophagus and vocal cords, causing dysphagia and dysphonia. Effective therapy for Candida and herpes simplex infections include flucanozole and acyclovir, respectively.
The antitumor antibiotics, anthracyclines (daunorubicin, doxorubicin, epirubicin, and idarubicin), and anthraquinones (mithramycin, mitomycin C, and Actinomycin D) are responsible for a dose-related cardiomyopathy and transient benign arrhythmias. The risk is increased in the elderly with underlying heart disease or prior mediastinal radiotherapy.32 Cyclophosphamide in high doses (>120 mg/kg) may result in fatal hemorrhagic myocardial necrosis within the first few days of treatment.32 A baseline echocardiogram often is obtained in this case to determine the patient’s ejection fraction prior to initiation of these therapies. These regimens are stopped if the ejection fraction is less than 50% or if it falls by 10% between treatments. The newer epirubicin and idarubicin are less toxic then the other anthracyclines. As with other dilated cardiomyopathies, treatment with angiotensin-converting enzyme (ACE) inhibitors may be indicated.
Asymptomatic elevation of liver enzymes is the most common liver abnormality secondary to chemotherapy. More serious entities involve hepatitis, cholestatic jaundice, and venoocclusive disease (which may occur with cyclophosphamide after bone marrow transplantation).32,33
Neurological manifestations of chemotherapy include peripheral neuropathy (paresthesias, weakness, loss of deep tendon reflexes), which can occur following treatment with vincristine. The elderly are prone to vinca alkaloid-induced autonomic neuropathy (vincristine, paclitaxel, and cisplatin) characterized by postural hypotension, severe abdominal pain, constipation, and paralytic ileus occurring within three days of treatment.56 Symptoms improve after discontinuing the agents. Cerebellar dysfunction may be seen with high-dose cytarabine and cisplatin. Alternate-day dosing of cytarabine has reduced this side effect. Intrathecal methotrexate has been linked to a stroke-like syndrome when used in combination with craniospinal irradiation for acute lymphocytic leukemia and non-Hodgkin’s lymphoma. Lower dosage appears to decrease the incidence of this complication.
Pulmonary complications of chemotherapy include pulmonary fibrosis and pulmonary edema. Pulmonary fibrosis following busulfan occurs within 3-4 years after treatment. Management includes steroids. Cyclophophamide also is associated with a pneumonitis leading to fibrosis. Melphalan (an alkylating agent) and carmustine (a nitrosurea) also have been linked to pulmonary fibrosis. Carmustine has been associated with pulmonary embolism, Wegener’s granulomatosis, adult respiratory distress syndrome, and pneumothorax.69 Five to eight percent of patients treated with methotrexate may develop pulmonary edema, pulmonary fibrosis, and hypersensitivity reactions. Headache, dyspnea, cough, and fever may be the presenting complaints.69 Continuous administration of cytarabine may result in a capillary leak syndrome and noncardiogenic pulmonary edema in 20% of patients within 2-20 days.32 Older patients are especially susceptible to hypersensitivity reactions and direct toxicity of antitumor antibiotics such as bleomycin, mitomycin, and doxorubicin. Steroids may be helpful.69
Renal toxicity is seen with cisplatin, mitomycin, methotrexate, and ifosfamide. This risk increases with the concurrent use of other nephrotoxic drugs. High-dose cyclophosphamide can lead to the syndrome of inappropriate secretion of antidiuretic hormone.32
Ocular toxicity is seen with busulfan, chlorambucil, cisplatin, cyclophosphamide, doxorubicin, methotrexate, and vincristine. Cataracts, diplopia, papilledema, conjunctivitis, and blurred vision are some of the ophtholmologic symptoms associated with common antineoplastic agents.32,68
Drug- and dose-dependent alopecia is the most frequent dermatologic manifestation of chemotherapy. Onset occurs within 7-10 days of treatment, and regrowth may begin within 1-2 months after completing treatment. This side effect carries its own psychologic sequelae. Treatment has not been shown to be effective.32,68
A paradoxidal effect of antineoplastic therapy is the occurrence of secondary malignancies. This is true concerning the anthracyclines, methotrexate, cytarabine, and cisplastin, among others. This potentially fatal side effect is dose and time-dependent. The most common secondary malignancies are acute myeloid leukemia and high-grade non-Hodgkin’s lymphoma. Myeolodysplastic syndrome is a risk factor. Older patients who have survived Hodgkin’s lymphoma are at greater risk.70,71
Conclusion
As the elderly population of the United States grows, the incidence of cancer is expected to rise almost proportionately.1 Malignancy complications in the elderly affect every organ system, and the symptoms often masquerade as common disorders. This is a further insult to the progressive systemic decline associated with normal aging. Altered mental status, seizures, chest pain, dyspnea, fever, hypercalcemia, abdominal pain, bleeding, fractures, thromboembolic disease, and various infections are all daily encounters in the ED. However, these common presentations become more challenging when they are accompanied by underlying malignancy in the elderly. As malignancy complications in the elderly become more commonplace in the ED, every emergency physician must consider occult malignancy in his or her differential diagnosis when approaching the elderly patient.
References
1. Kennedy BJ. Aging and cancer. In: Balducci L, Lyman GH, Ershler WB, eds. Comprehensive Geriatric Oncology. Australia: Harwood Academic Publishers; 1998:2.
2. Balducci L, Extermann M. Cancer And Age. In: Haskell CM, ed. Cancer Treatment. 5th ed. Philadelphia: W.B. Saunders Company; 2001:435-451.
3. McNamara RM. Demographics, aging, and emergency medical care. In: Sanders AB, ed. Emergency Care of the Elder Person. St. Louis, MO: Beverly Cracom Publications; 1996:3-9.
4. Balducci L, Extermann M. Cancer and aging—An evolving panorama. Hematol Oncol Clin North Am 2000;14:11.
5. Fox SA, Roetzheim RG, Kington RS. Barriers to cancer prevention in the older person. In: Balducci L, Lyman GH, Ershler WB. Comprehensive Geriatric Oncology. London: Harwood Academy; 1998:351-362.
6. Kenney RA. Physiology of aging. Clin Geriatr Med 1985;1:37-59.
7. Evans, R. Physiology of aging. In: Sanders AB, ed. Emergency Care of the Elder Person. St. Louis, MO: Beverly Cracom Publications; 1996:11-28.
8. Posner J. Neurologic complications of systemic cancer. Med Clin North Am 1979;63:783.
9. Posner JB. Management of central nervous system metastases. Semin Oncol 1977;4:81.
10. Siegal T, Siegal T. Current considerations in the management of neoplastic spinal cord compression. Spine 1989;14:223.
11. Byrne TN. Current concepts: Spinal cord compression from epidural metastases. N Eng J Med 1992;327:614.
12. Cloustin PD, DeAngelis LM, Posner JD. The spectrum of neurological disease in patients with systemic cancer. Ann Neurol 1992;31:268.
13. Deyo RA, Rainville J, Kent DL. What can the history and physical examination tell us about low back pain? JAMA 1992;268:760.
14. Maranzano E, Latini P. Effectiveness of radiation therapy without surgery in metastatic spinal cord compression: Final results from a prospective trial. Int J Radiat Oncol Biol Phys 1995;32:959.
15. Sioutos PJ, Arbit E, Meshulan CF, et al. Spinal metastases from solid tumors. Cancer 1995;76:1453.
16. Landmann C, Hunig R, Gratzl O. The role of laminectomy in the combined treatment of metastatic spinal cord compression. Int J Radiat Oncol Biol Phys 1992;24:627.
17. Young RF, Post EM, King GA. Treatment of spinal epidural metastases. Randomized prospective comparison of laminectomy and radiotherapy. J Neurosurg 1980;53:741.
18. Yahalom J. Oncologic emergencies. In: DeVita VT, Hellamnn S, Rosenberg SA, eds. Cancer: Principles and Practice of Oncology. 5th ed. Philadelphia:Lippincott-Raven Publishers;1997:2469-2475.
19. Ushio Y, Posner R, Posner JB, et al. Experimental spinal cord compression by epidural neoplasms. Neurology 1977;27:422.
20. Delatre JY, Arbit E, Thaler HT, et al. A dose response study of dexamethasone in a model of spinal cord compression caused by tumor. J Neurosurg 1989;70:920.
21. McCachren SS, Silberman HR. Approach to oncologic emergencies in the elderly. Clin Geriat Med 1987;3:575-586.
22. Kemeny MM, Busch-Devereaux E, Merriam LT, et al. Cancer surgery in the elderly. Hematol Oncol Clin North Am 2000;14:169-192.
23. Nishikubo C, Sadeghi A, Haskell CM. Superior vena cava syndrome. In: Haskell CM, ed. Cancer Treatment. 5th ed. Philadelphia: W.B. Saunders Co.; 2001:299-301.
24. Parish JM, Marsche RF, Dines DE, et al. Etiologic considerations in superior vena cava syndrome. Mayo Clin Proc 1981;56:407.
25. Lokich JJ, Goodman RL. Superior vena cava syndrome. JAMA 1975;231:58.
26. Maddox AM, Valdivieso M, Lukeman J, et al. Superior vena cava obstruction in small cell bronchogenic carcinoma. Cancer 1983;52:2165.
27. Wurschmidt F, Bunemann H, Heilmann HP. Small cell lung cancer with and without superior vena cava syndrome: A multivariate analysis of prognostic factors in 408 cases. Int J Radiat Oncol Biol Phys 1995;33:77.
28. Mahajan V, Strimlan V, Van Ordstrand HS, et al. Benign superior vena cava syndrome. Chest 1975;68:32.
29. Fine DG, Shepherd RF, Welch TJ. Thrombolytic therapy for superior vena cava syndrome (Letter). Lancet 1989;1:1200.
30. Grenberg S, Kosinski R, Daniels J. Treatment of superior vena cava syndrome with recombinant tissue-type plasminogen activator. Chest 1991; 99:1298.
31. Hennequin LM, Fade O, Fays JG, et al. Superior vena cava stent placement: Results with the Wallstent endoprosthesis. Radiology 1995;196:353.
32. Lowenthal RM, Eaton K. Toxicity of chemotherapy. Hematol Oncol Clin North Am 1996;10:967-990.
33. Ayash LJ, Hunt M, Antman K, et al. Hepatic venoocclusive disease in autologous bone marrow transplantation of solid tumors and lymphomas. J Clin Oncol 1990;8:1699.
34. Tang E, Davis J, Silberman H. Bowel obstruction in cancer patients. Arch Surg 1995;130:832.
35. Gallick HL, Weaver DW, Sachs RJ, et al. Intestinal obstruction in cancer patients. An assessment of risk factors and outcome. Am Surg 1986;52:434.
36. Butler JA, Cameron BL, Morrow M, et al. Small bowel obstruction in patients with a prior history of cancer. Am Surg 1991;162:624.
37. Sial SH, Catalano MF. Gastrointestinal disorders in the elderly. Gastroenterol Clin North Am 2001;30:567.
38. Shivshanker K, Chu DZJ, Stroehlein JR, et al. Gastrointestinal hemorrhage in the cancer patient. Gastrointest Endos 1983;29:273.
39. Birkett DH. Gastrointestinal tract bleeding. Surg Clin North Am 1911;71:1259.
40. Pimentel L. Medical complications of oncologic disease. Emerg Med Clin North Am 1993;11:407.
41. Bertheault-Cvitkovic F, Armand J-P, Tubiana-Hulin M, et al. Randomized, double-blind comparison of pamidronate versus galium nitrate for acute control of cancer-related hypercalcemia. Proc 9th EORT/NCI Symposium on New Drugs in Cancer Therapy 1996:140.
42. Body JJ, Dumon JC. Treatment of tumour-induced hypercalcemia with the bisphosphonate pamidronate: Dose-response relationship and influence of tumour type. Ann Oncol 1994;5:359.
43. Arrambide K, Toto RD. Tumor lysis syndrome. Semin Nephrol 1993;13:273.
44. Fleming DR, Doukas MA. Acute tumor lysis syndrome in hematologic malignancies. Leuk Lymph 1992;8:315.
45. Madias NE. Lactic acidosis. Kidney Int 1986;29:752.
46. Sculier JP, Nicaise C, Klastersky J. Lactic acidosis: A metabolic complication of extensive metastatic cancer. Eur J Clin Oncol 1983;19:597.
47. Macaulay VM. Insulin-like growth factors and cancer. Br J Cancer 1992; 65:311.
48. Lipschitz DA, Udupa KB, Milton KY, et al. Effect of age on hematopoiesis in man. Blood 1984;63:502.
49. Baraldi-Junkins CA, Beck AC, Rothstein G. Hematopoiesis and cytokines —Relevance to cancer and aging. Hematol Oncol Clin North Am 2000;14: 45-61.
50. Wright IS. The pathogenesis and treatment of venous thrombosis. Circulation 1952;5:161.
51. Green Kb, Silverstein RL. Hypercoagulability in cancer. Hematol Oncol Clin Am 1996;10:499.
52. Rickles FR, Edwards RL. Hemostatic alterations in cancer patients. Cancer Metastasis Rev 1992;1:241.
53. Lipton A, Harvey HA, Hamilton RW. Venous thrombosis as a side effect of tamoxifen treatment. Cancer Treat Rep 1984;68:887.
54. Prandoni P, Lensing AWA, Buller HR, et al. Deep-vein thrombosis and the subsequent incidence of symptomatic malignant disease. N Eng J Med 1992;327:1128.
55. Goad KE, Gralnick HR. Coagulation disorders in cancer. Hematol Oncol Clin North Am 1996;10:457.
56. Eastridge BJ, Lefor AT. Complications of indwelling venous access devices in cancer patients. J Clin Oncol 1995;13:233.
57. Luzzatto G, Schafe AI. The prethrombotic state in cancer. Semin Oncol 1990;17:147.
58. Block KJ, Maki DG. Hyperviscosity syndrome associated with immuno-globulin abnormalities. Semin Hematol 1974;10:113.
59. Gmur J, Burger J, Schanz U, et al. Safety of stringent prophylactic platelet transfusion policy for patient with acute leukemia. Lancet 1991;338: 1223-1226.
60. Hackett T, Kelton JG, Powers P. Drug-induced platelet destruction. Semin Thromb Hemost 1982;8:116-137.
61. Porter J, Jick J. Addiction is rare in patients treated with narcotics. N Eng J Med 1980;302:123.
62. World Health Organization. Cancer pain relief and palliative care:Report of a WHO Expert Committee. Geneva, Switzerland: World Health Organization: 1990 Technical Report Series 804.
63. Emmanouilides C, Glaspy J. Opportunistic infections in oncologic patients. Hematol Oncol Clin North Am 1996;10:841.
64. Pizzo PA. Fever in immunocompromised patients. N Eng J Med 1999;341:893-899.
65. Hughes WT, Armstrong D, Bodey GP, et al. 1997 Guidelines for the use of antimicrobial agents in neutropenic patients with unexplained fever. Clin Infect Dis 1997;25:551-573.
66. Freifeld AG, Walsh T, Marshall D, et al. Monotherapy for fever and neutropenia in cancer patients: A randomized comparison of ceftazidime versus imipenem. J Clin Oncol 1995;13:165-176.
67. Freifeld A, Marchigiani D, Walsh T, et al. A double-blind comparison of empirical oral and intravenous antibiotic therapy for low-risk febrile patients with neutropenia during cancer chemotherapy. N Eng J Med 1999; 341:305-311.
68. Dorr RT, VonHoff DD. Cancer Chemotherapy Handbook. 2nd ed. Norwalk, CT: Appleton and Lange;1994.
69. Kreisman H, Wolkove N. Pulmonary toxicity of antineoplastic therapy. Semin Oncol 1992;19:508.
70. Travis LB, Curtis RE, Stovall M, et al. Risk of leukemia following treatment for non-Hodgkin’s lymphoma. J Natl Can Inst 1994;86:1450.
71. van Leeuwen FE, Chorus AM, van den Belt-Dusebout AW, et al. Leukemia risk following Hodgkin’s disease: Relation to cumulative dose of alkylating agents, treatment with teniposide combinations, number of episodes of chemotherapy and bone marrow damage. J Clin Oncol 1994;12:1063.
CME Objectives
Upon completing this program, participants will be able to:
- Quickly recognize or increase index of suspicion for specific conditions in the elderly patient;
- Understand the epidemiology, etiology, pathophysiology, and clinical features of the entity discussed;
- Perform necessary diagnostic tests correctly and take a meaningful patient history that will reveal the most important details about the particular medical problem discussed;
- Apply state-of-the-art therapeutic techniques (including the implications of the pharmaceutical therapy discussed) to patients with the particular medical problem discussed; and
- Provide patients with any necessary discharge instructions.
Physician CME Questions
1. Which of the following is true about the elderly?
A. Memory, attention, and language are unaffected by age.
B. Altered mental status is the most frequent neurological presentation in elderly cancer patients.
C. Cancer is an inevitable consequence of normal aging.
D. Cancer ranks ahead of cardiovascular disease in mortality in patients older than age 65.
E. Eighty percent of all cancers occur in patients older than age 65.
2. The most common location of spinal cord compression is:
A. the sacral region.
B. the cervical and lumbar levels, equally.
C. the lumbar region alone.
D. the thoracic region.
E. the cervical region alone.
3. Which of the following is true about superior vena cava syndrome (SVCS)?
A. It is most commonly associated with Hodgkins’ lymphoma.
B. SVCS is best diagnosed by laborarory tests.
C. SVCS may present with altered mental status.
D. There is no role for thrombolytics in the management of SVCS.
E. A salt-free diet markedly relieves the symptoms of SVCS.
4. A characteristic of cardiac tamponade in the elderly is:
A. It can be confused with congestive heart failure.
B. Pulsus paradoxicus is pathognonomonic.
C. Diuretics and nitrates constitute the best treatment.
D. Left ventricular pressure is markedly higher than right ventricular pressure.
5. Which of the following is true of neutropenic fever in the elderly on chemotherapy?
A. Neutropenia does not occur in the elderly, as they normally are neutropenic.
B. It may be the marker of an underlying infection.
C. A duration of more than 10 days portends a good prognosis for the elderly.
D. Neutropenia is defined as an absolute neutrophil count of 1200/mm3.
E. Most causes of neutropenia are discovered in the ED.
6. Which of the following is true about pain management in the elderly cancer patient?
A. Pain sensation is markedly lessened as one ages.
B. Cancer patients on narcotics have a high rate of proven addiction.
C. The clinician’s lack of experience in pain assessment may contribute negatively to its management.
D. The presence of pain is a sign of the end of life for the elderly.
E. The absence of neurological signs precludes the diagnosis of impending spinal cord compression.
7. Which of the following is true of hypercalcemia in the elderly cancer patient?
A. Serum calcium level is not directly related to the symptoms.
B. Patients are usually very well hydrated on presentation.
C. Furosemide is the first line of treatment.
D. ECG changes include prolonged QT and sine wave.
E. Hypercalcemic symptoms preclude treatment-induced fluid overload in the frail elderly.
8. Which of the following is true of abdominal pain in the elderly cancer patient?
A. Signs and symptoms may be masked in the patient on corticosteroids.
B. Right upper quadrant pain may be due to hepatic metastases.
C. The most common site of bowel obstruction is the small bowel.
D. Cecal distention of 12-14 cm on x-rays is an indication for colonoscopic decompression.
E. All of the above
9. Management of pulmonary fibrosis following busulfan includes steroids.
A. True
B. False
10. Drug- and dose-dependent alopecia is the most frequent dermatologic manifestation of chemotherapy.
A. True
B. False
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
