Infectious Causes of Upper Airway Obstruction in Children
Authors: Ronald M. Perkin, MD, MA, FAAP, FCCM, Professor and Chairman, Department of Pediatrics, The Brody School of Medicine at East Carolina University, Greenville, NC; James D. Swift, MD, FAAP, Medical Director, Sunrise Children’s Hospital, Las Vegas, NV.
Peer Reviewers: James E. Colletti, MD, Senior Associate Consultant, Department of Emergency Medicine, Department of Pediatrics, The Mayo Clinic, Rochester, MN; Kenneth H. Butler, DO, Associate Residency Director, Emergency Medicine Residency Program, University of Maryland School of Medicine, Baltimore.
Of the many conditions seen by those caring for acutely ill infants and children, none can be so frightening, and yet rewarding to treat, as severe upper airway obstruction. The severe consequences of an incorrectly managed child mandate that these children be approached rapidly with a structured plan for securing and maintaining a patent airway.—The Editor
Why Are Children at Greater Risk?
Children are particularly susceptible to conditions affecting the upper airway for two basic reasons: the small size of the conducting air passages and the dynamics of air flow. The narrowest portion of an infant or child’s airway is the cricoid ring—about 3.5 mm in diameter in a newborn. In addition, the supportive tissues of the extrathoracic airway are more compliant in children and, therefore, are more liable to collapse with increased inspiratory effort. This leads to further airway narrowing, which increases resistance to airflow.
Laminar flow occurs along smooth straight tubes. Under these conditions, resistance to airflow is proportional to the length of the tube and inversely related to the fourth power of the radius; i.e., a narrow tube is much more resistant than a wide one.
If the radius of the airway was halved, it would require 32 times the pressure drop (and a significant increase in the work of breathing) to maintain the same airway flow.1 Small degrees of obstruction, therefore, cause an increase in respiratory effort, decrease in volume exchanged, and increased transit times in either inspiration or expiration, depending on the level of obstruction. Extrathoracic airway obstruction is aggravated by the physiological collapse of the airways during inspiration, while intrathoracic obstruction is aggravated by airway compression during forced expiration.2,3
The resistance to flow through an airway also is related to two physical characteristics of gas: viscosity and density. In a smooth airway where the flow is laminar, gas viscosity determines flow. With an abrupt narrowing of an airway, as occurs in subglottic inflammation, the flow is turbulent and density is the property determining airflow.1,2
Another important characteristic of turbulent flow which differs from laminar flow is that the pressure gradient required to produce a given gas flow rate is proportional to the square of the gas flow rate. Since resistance to airflow is defined as pressure gradient divided by flow rate, resistance is not constant in turbulent flow as it is in laminar flow, but rises in proportion to the flow rate. This is a crucial clinical point: When a child with acute airway obstruction becomes distressed or agitated, and increases gas flow, resistance to airflow increases. Put simply, the harder a child with acute airway obstruction tries to breathe, the harder it is for him or her to breathe.
Turbulent flow often is audible and invariably is present when obstruction to gas flow is a problem. The nature of the sound transmitted and its timing in the respiratory cycle further may assist in diagnosing the level of obstruction. In general, obstruction of the softer, boggy tissues of the oropharynx and nasopharynx are transmitted as snores, while vocalizations from the supraglottic area commonly are muffled. Obstruction at the vocal cord and subglottic level causes the classical dysphonic inspiratory stridor typical of viral croup.3 Bronchotracheal obstruction causes a predominantly expiratory wheeze and a whistle.
Careful observation of the respiratory effort, nature of the retractions, and evaluation of timing and pitch of breath sounds may assist in assessing the severity and location of obstruction.
Viral Croup
Croup is a common respiratory illness in children. The illness commonly is manifested in young children by a hoarse voice; dry, barking cough; inspiratory stridor; and a variable amount of respiratory distress that develops over a brief period of time.
Definition and Terminology. The term "croup syndrome" refers to a group of diseases that varies in anatomic involvement and etiologic agents and includes laryngotracheitis, spasmodic croup, bacterial tracheitis, laryngotracheobronchitis, and laryngotracheobronchopneumonitis.4-6
Acute viral infection is the most common cause of croup, but bacterial and atypical agents also have been identified. It generally is accepted that acute laryngotracheitis and spasmodic croup are caused by viral agents alone, whereas both bacterial and viral agents may be responsible for causing disease further down the respiratory tract, such as laryngotracheobronchitis and laryngotra-cheobronchopneumonitis. Bacterial tracheitis, also known as mem-branous or bacterial croup, involves infection with bacteria such as Staphylococcus aureus, Haemophilus influenzae, and Coryne-bacterium diphtheriae and will be discussed later in this review.
Epidemiology. Croup accounts for about 15% of respiratory tract disease seen in pediatric practice.4,5 Viral croup is primarily a disease of children between 1 and 6 years of age, with a mean age of 18 months. In the United States, its peak incidence is about 5 cases per 100 children during the second year of life. Although most cases occur during the late fall and winter, croup can manifest throughout the year.
Etiology. Parainfluenza viruses (types 1, 2, and 3) account for more than 65% of croup cases, with parainfluenza virus types 1 and 2 responsible for the majority of illnesses and outbreaks.4-6
Other viruses associated with this disease include influenza A and B, adenovirus, respiratory syncytial virus (RSV), and measles. The most severe laryngotracheitis has been noted in association with influenza A viral infections.6
Pathogenesis. As with most respiratory infections, viral infection in acute laryngotracheitis, laryngotracheobronchitis and laryngotracheobronchopneumonitis begins in the nasopharynx and spreads to the respiratory epithelium of the larynx and trachea. Diffuse inflammation, erythema, and edema develop in the tracheal walls, and the mobility of the vocal cords becomes im-paired. The portion of the trachea below the larynx (subglottic trachea) is the narrowest part of a child’s upper airway. This area is surrounded by firm cartilage, and any swelling in that region encroaches on the airway and can restrict airflow significantly. This airway narrowing leads to audible inspiratory stridor, and the vocal cord swelling results in a hoarse voice.
With disease progression, the tracheal lumen becomes further obstructed by fibrinous exudate and pseudomembranes. Histologic sections of the larynx and trachea reveal marked edema, with cellular infiltration of histiocytes, lymphocytes, plasma cells, and polymorphonuclear leukocytes.
Further extension of the disease from the trachea into the bronchi and alveoli results in laryngotracheobronchitis and laryngotracheobronchopneumonitis, respectively. However, the progressive obstructive disease at this level is usually the result of secondary bacterial involvement.4
In spasmodic croup, histology of the subglottic tissues shows noninflammatory edema. Accordingly, it is presumed that there is no direct viral involvement of the tracheal epithelium in this entity and that the obstruction is due to the sudden occurrence of noninflammatory edema within the submucosa of the subglottic trachea. Although there is an association with the same viruses that cause acute laryngotracheitis, the reason for this sudden edema is unknown. It has been suggested that spasmodic croup represents more of an allergic reaction to viral antigens than direct infection.
Clinical Presentation. Acute Laryngotracheitis. Acute laryngotracheitis typically starts with rhinorrhea, pharyngitis, a mild cough, and low-grade fever of a few days duration. However, after a short period, usually 12-48 hours, upper airway obstruction signs and symptoms are noted. The child develops a characteristic "barking" cough, hoarseness, and inspiratory stridor, with or without fever.
Physical examination reveals a child who has a hoarse voice, coryza, a normal or mildly inflamed pharynx, and a slightly increased respiratory rate. The speed of progression and degree of respiratory distress can vary substantially. Most cases are characterized by only the hoarseness and barky cough, with no other evidence of airway obstruction. These symptoms gradually normalize within 3-7 days. In other cases, an increasing severity of obstruction is evident and accompanied by increasing heart and respiratory rates, flaring of alar nasi and cyanosis with supra- and infraclavicular and sternal retractions. Affected children become restless and anxious with the development of progressive hypoxia and require close monitoring. The duration of illness in more severely affected children usually is 7-14 days. Children with previous airway surgeries, intubations or protracted newborn courses may have residual scar tissue which may predispose them to more severe croup and thus require a more careful assessment and conservative management strategy.
Spasmodic Croup. Spasmodic croup tends to occur at night in young children between 3 months and 3 years of age. Often, it is difficult at the onset to distinguish laryngotracheitis from spasmodic croup. The child may have cold symptoms and otherwise look well. Initially, the child awakens at night with sudden dyspnea, croupy cough, and inspiratory stridor. Fever is not present, and gentle reassurance and administration of moist air provide relief. The symptoms are the result of sudden subglottic edema, and the child can have repeat attacks on the same night and for the next three or four successive nights.6 Spasmodic croup may be differentiated from laryngotracheitis with endoscopic examination. The laryngeal mucosa appears pale and boggy in spasmodic croup and erythematous and inflamed in acute laryngotracheitis.
Much has been written about differentiating spasmodic croup from viral croup, but this differentiation often is of limited usefulness for clinicians.7,8
Differential Diagnosis. The most frequent serious differential diagnostic problem has been distinguishing acute epiglottitis from acute laryngotracheitis. Ascertaining the child’s immunization history is vital. Since the introduction of the H. influenzae type b vaccine in 1990 in the United States, cases of epiglottitis have declined markedly. However, because organisms other than H. influenzae occasionally can cause epiglottitis, this diagnosis cannot be forgotten.
In acute epiglottitis, the important differential points on clinical examination are lack of a croupy cough, drooling, toxic appearance, growing anxiety and apprehension, a sitting posture with the chin pushed forward and refusal to lie down. If the patient is completely stable and the posterior pharynx is examined, a cherry-red epiglottis would be present. In contrast, the child who has acute laryngotracheitis will have a barky cough, be comfortable supine, and be less apprehensive. On visual inspection, the epiglottis appears normal. Lateral neck and chest radiographs have been used to help make the diagnosis, but they usually are not recommended when epiglottitis is suspected because of the tenuous condition of these patients. When epiglottitis is suspected, the clinician must avoid agitating the patient, which could aggravate the child’s already compromised respiratory state, and should have equipment for intubation readily available. Classic radiographs of a child who has laryngotracheitis show the characteristic "steeple sign," or airway narrowing in the subglottic area; in epiglottitis, these films classically demonstrate the "thumb sign" of the swollen epiglottis. Radio-graphs, if performed, should be used as an adjunct to help confirm the diagnosis; clinical correlation is key to the diagnosis.
Other diagnoses for a child presenting with acute upper airway obstruction include foreign body aspiration and angioneurotic edema. Acute angioneurotic edema usually presents with other evidence of swelling of the face and neck. Laryngeal diphtheria, although rare these days, should be considered. Important information in this regard is the immunization history, clinical evidence of pharyngeal involvement, greater degree of hoarseness, and relative slowness of disease progression. Other conditions to consider include retropharyngeal or peritonsillar abscess, subglottic stenosis, infectious mononucleosis, and bacterial tracheitis.
Diagnosis. Croup is diagnosed primarily on clinical grounds. Radiographs of the neck have been used to confirm the diagnosis of laryngotracheitis and exclude other causes of the croup syndrome.6 The hallmark finding in croup is a tapered narrowing of the subglottic trachea (steeple or pencil sign) due to localized edema, as contrasted with the normally domed or shouldered configuration in the region.9 The narrowing of the trachea should be visible on both frontal and lateral views because the edema is circumferential. Be aware of a potential false-positive steeple sign on frontal view due to a normal sloping of the subglottic trachea when the vocal cords are relaxed and, therefore, separated.9 The lateral neck film may reveal overdistention (ballooning) of the hypopharynx during inspiration. The epiglottitis and the aryepiglottic folds appear normal.
However, these classic radiologic findings are present in only 50% of cases of viral croup, and many children who have croup will have normal findings on radiography. Furthermore, the steeple sign frequently is present in radiographs obtained from children who do not have croup, depending upon the phase of respiration.4
Therefore, because radiologic findings do not correlate well with clinical findings, the diagnosis of croup should rest on clinical grounds and not upon radiologic findings.6,10 The purpose of obtaining neck radiographs in a patient with suspected croup is not to document the diagnosis as much as it is to exclude other causes of upper airway obstruction that require intervention, such as a foreign body. These studies should be limited to children whose illnesses are atypical and whose respiratory status is stable.
Because laryngotracheitis is a disease of the upper airway, alveolar gas exchange usually is normal, and hypoxia and low oxygen saturation will be undetectable until a patient’s condition is severe. Most children who have laryngotracheitis or spasmodic croup have normal findings on pulse oximetry. Serial observations remain the most accurate methods of monitoring a child who has acute laryngotracheitis. Pulse oximetry may be more useful in patients who have laryngotracheobronchitis or laryngotracheobronchopneumonitis, which involves the lower airway.
Most children who have spasmodic croup or acute laryngotracheitis do not require intubation or direct visualization of the airway. However, a child whose illness is severe, who has signs of epiglottitis, or whose condition fails to follow the benign course of viral croup, direct airway visualization may be necessary. There is general agreement that the patient in whom complete obstruction of the airway is imminent requires laryngoscopy and intubation in a well-controlled environment, preferably under a predesignated protocol with anesthesia and otolaryngology part of the response team.
Assessing Severity of Viral Croup. While most children with croup have mild symptoms that will not progress, a subset of children may develop progressive airway obstruction and respiratory failure.3 Several clinical scoring systems have been proposed for assessing children with stridor.3,11-13 Most of these "croup scores" are based on five clinical signs: level of consciousness, cyanosis, stridor, air entry and retractions. Each sign is given an arbitrary range of numerical values to represent the gradiations of severity of that sign. While the Westley Score11 seems to be commonly used, no one particular scoring system has proven to be superior to any other. We suggest a modified Westley Score as provided in Table 1.
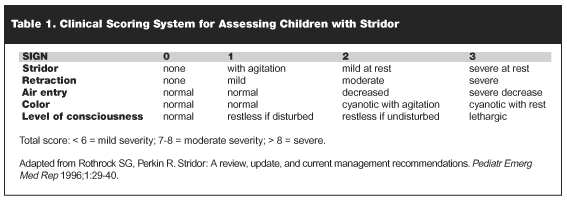 |
Croup scores are a relatively subjective measure of severity. Features suggesting a higher potential for respiratory failure have been identified, including age younger than 6 months, stridor at rest, cyanosis, decreased level of consciousness and hypercapnia.3,14,15 One study found that clinicians were only 33% sensitive in identifying hypoxemia by physical examination in this population of children.16 Hypoxemia generally indicates advanced disease and impending respiratory failure. Hypercapnia also has been found to be a predictor of clinical deterioration.3,15
Treatment. Mist Treatment. The mainstay of treatment for children who have croup is airway management. Since the 19th century, mist treatment has been used to treat croup symptoms. Cool mist is as effective as hot steam, and it avoids the risk of burns from hot water. Cool mist moistens airway secretions and soothes inflamed mucosa. Also, the humidity decreases the viscosity of tracheal mucus secretions.4 Animal studies have shown that the mist may activate mechanoreceptors in the larynx that produce a reflex slowing of respiratory flow rate.4 Young children best tolerate cool mist vapor delivered by aerosol while sitting on the parent’s lap. Although cool mist is viewed as a safe and simple method to relieve croup symptoms, the humidity can intensify bronchospasm in children who have croup with wheezing due to laryngotracheobronchitis or pneumonitis. These children should have a trial cool mist that is discontinued if the wheezing continues or worsens.
There are only a few studies that have tested the efficacy of moist air in patients with croup and, unfortunately, none of them has documented a significant benefit.6,7 Therefore, the use of humidified air for children with viral croup is a tradition without proven efficacy.17,18
Epinephrine. Often croup requires no other treatment beyond mist therapy, but occasionally pharmacotherapy also is necessary. Nebulized epinephrine has been used to treat severe croup symptoms for almost 30 years and has made tracheotomy for croup virtually nonexistent.6,19 Prior to the use of nebulized epinephrine, hospitalization of patients with viral croup was associated with tracheotomy rates between 2.9 and 13% and mortality rates between 0.09 and 2.7%.6
Racemic epinephrine is a 1:1 mixture of the d- and l- isomers of epinephrine. The mechanism of action is believed to be stimulation of alpha-adrenergic receptors with subsequent constriction of capillary arterioles. This results in fluid resorption instead of capillary leakage from interstitial space and a consequent decrease in laryngeal mucosal edema.6 Additional studies have shown that equal doses of only the l-isomer of epinephrine have the same beneficial effects as the racemic form.20
Although nebulized epinephrine may have a dramatic effect on croup symptoms, decreasing inspiratory stridor and intercostal retractions, common adverse reactions to both the racemic and l-isomer forms, including tachycardia, hypertension, and myocardial infarction, may limit their desirability in certain populations, such as children with significant congenital heart disease.6,21 In addition, the effect of the medication is brief (< 2 h), and as its activity diminishes, symptoms of croup can reappear (rebound phenomenon). Administration of nebulized epinephrine to children who had croup in the emergency department frequently led to hospital admission in the 1980s because of concern about the rebound phenomenon. Recent review and assessment of those recommendations suggest that it is safe to dismiss a child who has received nebulized epinephrine for croup from the emergency department after 3-4 hours of observation.22-25 For safe discharge the child should meet the following conditions: no stridor at rest; normal air entry; normal color; normal level of consciousness; and received one dose of dexamethasone orally or intramuscularly (IM).
Nebulized epinephrine should be utilized in children who have evidence of respiratory distress, children who appear ill with symptoms consistent with croup, children with previous airway procedures and/or croup and stridor at rest. A dose of 0.25-0.75 mL of 2.25% racemic epinephrine solution in 2.5 mL of normal saline can be given via nebulizer as often as every 20 minutes. If racemic epinephrine is not available, a 5-mL mixture of l-isomer epinephrine (1:1,000) and saline may be used.
Corticosteroids. After decades of debate about the potential benefits of systemic corticosteroids in children who have laryngotracheitis, there is now ample evidence to support their use.6,7 Clinical trials have demonstrated clear improvement in children who have viral laryngotracheitis treated with oral or parenteral steroids compared with those who received placebo. Clinical improvement, however, usually is not apparent until up to six hours after initiation of treatment.4
A review of the published literature during the past 10 years reveals a number of studies documenting the efficacy of corticosteroids in treating moderate, severe, and even mild presentations of croup. Additionally, two meta-analyses have been published—one in 1989 and another 1999.26,27
The majority of these studies are well-done trials (randomized, blinded, and placebo-controlled). They have differed in several respects: specific corticosteroids used, route of administration, dose of corticosteroids, and severity of croup. The most commonly used corticosteroids employed have been dexamethasone, given IM or orally, and budesonide administered by nebulization.
Beneficial effects of glucocorticoids evident from these studies include reductions in symptom severity (i.e., croup score); in the need for hospitalization after treatment in an emergency department; in the duration of hospitalization or time spent in an emergency department; in the necessity for admission to an intensive care unit; and in the need for further drug therapy (i.e., nebulized epinephrine). Recent commentaries, editorials, and review articles found in the literature all recommend corticosteroids for croup, even with mild presentations.28,29
No clinically significant adverse effects were reported in the majority of the studies described above, which is not surprising from the use of a single corticosteroid dose. In a study of nebulized dexamethasone, two neutropenic patients developed bacterial tracheitis, although it was unclear what role was played by dexamethasone.30
Dexamethasone has been evaluated the most extensively in published studies and has become the corticosteroid most often used in clinical practice. Dexamethasone is used because it has potent glucocorticoid (anti-inflammatory) activity as well as a long duration of action (> 48 hours). Earlier studies documented the beneficial effects of a single IM dose of 0.6 mg/kg (approximately equivalent to 4 mg/kg of prednisone in anti-inflammatory potency) in moderate to severe croup.7,31 In the mid-1990s, studies using oral dexamethasone (single dose of 0.6 mg/kg) began appearing in the literature.7,19,32 Oral administration of dexamethasone has the advantage of less distress of drug administration to the patient, who already is distressed, as compared to IM injection or nebulization with a face mask.32
Two studies further evaluated lower doses of dexamethasone, 0.15 mg/kg and 0.3 mg/kg. One study compared oral dexamethasone at doses of 0.25 mg/kg, 0.3 mg/kg, and 0.6 mg/kg in a randomized, double-blind manner, in the treatment of children hospitalized with croup.33 All doses were equivalent in their clinical benefit of reducing croup scores, the need for nebulized epinephrine, and duration of hospitalization.
More recently published studies have evaluated dexamethasone in milder forms of croup, using lower doses, and given orally.28,29 Geelhoed compared oral dexamethasone, 0.15 mg/kg, with placebo in a double-blind, randomized manner in 100 children with mild croup (croup score of 0.9/6; no stridor at rest).28 The number of children who returned to medical care with continued croup was significantly lower (P < 0.05) in those receiving dexamethasone. The number of children admitted to a hospital, or the duration of croup symptoms did not differ significantly between the groups.28
Dexamethasone also has been evaluated in a nebulized route of administration.30 This study was randomized, double-blind, and placebo-controlled, yet it included a relatively small number of patients and thus was somewhat limited in statistical power. These researchers found that dexamethasone, when given to children with moderate croup, resulted in improved croup symptoms at four hours, but it did not alter hospital admission rates four or 24 hours after treatment. The authors do not recommend using nebulized dexamethasone in the treatment of croup.
Budesonide is a potent synthetic corticosteroid that is commercially available as a metered-dose inhaler and as a nasal inhaler for the treatment of asthma and allergic disorders.34 Trials evaluating budesonide for croup have compared it to placebo, nebulized epinephrine, and dexamethasone in patients with mild, moderate, and severe croup.35-40
Compared with placebo, budesonide reduced croup symptom severity and allowed earlier discharge from an emergency department.35 Several studies have directly compared nebulized budesonide to dexamethasone (orally or IM) and generally have found them equally efficacious in reducing symptoms and duration of hospitalization. The beneficial action of nebulized budesonide occurs more rapidly.36-38 One study, however, found IM dexamethasone superior to budesonide in reducing symptoms (croup score) five hours after treatment. Treatment with either drug resulted in fewer hospitalizations as compared to placebo.39
A recent, larger study compared three therapies, oral dexamethasone alone, nebulized budesonide alone, or their combination, in the treatment of mild-moderate croup.40 Measures of croup score improvement, hospital admission rates, time spent in, or return visits to the emergency department were equal among all groups following treatment, and the authors concluded that oral dexamethasone should be the preferred treatment.
In conclusion, significant evidence now exists in the published literature to document the beneficial effects of corticosteroids in the treatment of croup, even in outpatients with milder presentations. (See Table 2.) Dexamethasone, given orally or IM, is effective, as is nebulized budesonide. Dexamethasone given orally may be the preferred treatment in most patients due to its ease of administration and low cost.6,40,41 Although limited evidence indicates that oral doses of dexamethasone as low as 0.15 mg/kg are effective, further research is needed prior to acceptance of this regimen.
 |
Helium-Oxygen Mixture. A mixture of helium and oxygen has been used to help manage children with severe viral croup.42-45
Although helium offers an additional tool in the treatment of various airway and pulmonary problems, it has no inherent therapeutic effect.42 As such, it can be used only as a temporizing agent to allow time for therapeutic agents to work or for the natural resolution of the disease.
To be effective, helium must be administered in concentrations of 60-80%.42 Helium concentrations less than 60% significantly blunt the density advantage.43,44 A mixture of 80% helium and 20% oxygen is ideal, and premixed tanks can be obtained.
Epiglottitis
Epiglottitis is a very serious infection of the epiglottis and supraglottic structures that results in acute airway obstruction and, if not managed correctly, death may occur.46-49 Since the development of the H. influenzae type B vaccine, this disease process has become unusual, but must be considered in a child with dyspnea and stridor. Although epiglottitis occurs mainly in children, it can occur at any age.46-48
Disease Mechanisms. Acute epiglottitis almost always is caused by H. influenzae type B (Hib), which can be cultured from direct epiglottic swabs or blood cultures. Other causative organisms are usually bacteria but occasionally viruses or Candida organisms.46,52-54
Direct invasion by Hib causes cellulitis with marked edema of the epiglottis, aryepiglottic folds, ventricular bands, and arytenoids. As edema increases, the epiglottis curls posteriorly and inferiorly. Infection of the supraglottic larynx may extend but does not usually reach the subglottis or the laryngeal lymphatic system. It is important to realize that in some cases the epiglottis itself may be only minimally involved but that the edema and inflammatory swelling of the uvula, aryepiglottic folds, false cords, and arytenoids causes equally severe obstruction. Supraglottitis is the more appropriate term to describe infection which involves all supraglottic structures.
Clinical Manifestations. Epiglottitis is rare; most commonly occurring in children younger than age 5 years, with an annual incidence that varies from region to region. With the introduction of Hib vaccination, it virtually has disappeared.55-59
Up to half of patients have preceding upper respiratory tract symptoms. The onset of epiglottitis typically is abrupt, with duration of symptoms usually fewer than 24 hours and early toxicity.
Children may present with a very sore throat, difficulty swallowing because of pain, respiratory distress, drooling, a choking sensation, irritability, restlessness, and anxiety. The temperature is high, usually between 38.8° and 40°C (101.8°-104°F). Sighing respirations, mild stridor, retractions, and mild tachypnea may occur. Less common symptoms include cough, delirium, lethargy, hoarseness or aphonia, vomiting, chills, anorexia, cervical adenopathy, wheezing, and hypotonia.
The child naturally assumes a posture that maximizes the diameter of the obstructed airway: sitting and leaning forward with hyperextension of the neck and protrusion of the chin.
Previous publications have demonstrated a downward trend in the age at presentation in epiglottitis; up to 36% of all cases of epiglottitis occur in children younger than 2 years of age.60-63 Analysis of these publications reveals that the history and presenting signs of infants younger than 2 years old are more variable, thus making distinction from other causes of acute upper airway obstruction more difficult. Signs and symptoms not routinely described in children with epiglottitis but often observed in infants with epiglottitis include the absence of fever, the presence of only low-grade fever, a history of antecedent urinary tract infection, a prominent "croupy" cough and a "non-toxic" appearance.60-63
Children with epiglottitis are at risk for total airway obstruction. The enlarged, inflamed supraglottic ring can progress to respiratory obstruction with unexpected suddenness. The exact mechanism is not clear, and the early view that the swollen epiglottis collapses into the laryngeal inlet may be incorrect. Epiglottitis progresses to death in about 7% of children who do not have secured airways.59 With accurate early recognition and elective intubation, the mortality rate should approach zero.
Complications are uncommon and may include exudative tonsillitis, cervical lymphadenitis, and otitis media. Evidence of pneumonia or atelectasis is sometimes seen on the chest radiograph. Meningitis, septic arthritis, and pericarditis occurring with epiglottitis are rare; routine lumbar puncture is unnecessary.
The diagnosis is often clear when a child has a classic presentation. However, it is sometimes difficult to differentiate epiglottitis from viral croup. The distinguishing features of epiglottitis include the absence of spontaneous cough and the presence of drooling and agitation. Toxicity, high fever, and sore throat also may occur with bacterial tracheitis, uvulitis, and retropharyngeal or parapharyngeal abscess. Nasopharyngeal diphtheria is rare now, but may mimic acute epiglottitis and is associated with serosanguinous discharge. Noninfectious causes mimicking epiglottitis include angioedema, a pharyngeal burn, and a foreign body that is in the valleculae or larynx or that penetrates the pharyngeal tissues.
Chronic epiglottic enlargement may be seen with neck radiotherapy for cancer, granulomatous lymphangitis, or lymphangiectasis and infection with the human immunodeficiency virus. The chronicity of symptoms makes these conditions easily distinguishable from acute epiglottitis. Similarly, congenital anomalies of the airway and laryngeal papillomatosis usually are quite distinct by the presence of recurrent or persistent symptoms.
Diagnosis. Investigations should be left until the airway is secured. The diagnosis is confirmed under direct visualization.46,64 Detection of the responsible organism is important for guiding antibiotic management. Direct culture of supraglottic tissues reveals the causative organism in the majority of patients. The blood culture is positive for Hib in about 70% of cases.
In atypical cases, where the patient is completely stable, lateral radiographic views of the soft tissues of the neck may be obtained with the patient’s airway carefully monitored throughout the procedure. The best view of the anatomic structures of the upper airway is obtained with patient upright. In patients with epiglottitis, the hypopharynx is dilated, and the normal cervical lordosis may be replaced by a straight or kyphotic contour. The valleculae are narrowed and may be obliterated. There is marked enlargement of the epiglottis and thickening of the aryepiglottic folds. An obliquely imaged epiglottis may artifactually appear wide because both the left and right sides of the epiglottis are being imaged adjacent to each other. This should not be confused with a truly enlarged epiglottis. Epiglottitis is recognized on lateral neck film by the classic "thumb sign," which describes the rounded, thickened, edematous epiglottis, giving it the approximate shape and size of the dorsum of an adult’s thumb. (See Figure 1.)48
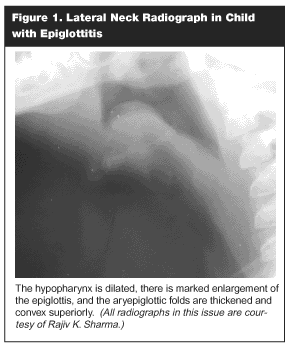 |
Objective criteria have been used to diagnosis epiglottitis in children on lateral neck films. These include the measurement of the following ratios: the epiglottic width to the third cervical vertebral body width (EW/CSW) of greater than 0.5 and epiglottic width to epiglottic height (EW/EH) of greater than 0.6.48
Management. Because of the high risk of complete airway obstruction, great care must be should be taken in managing a child with suspected epiglottitis. Once a clinician suspects this diagnosis, the child should be constantly attended by an individual skilled in airway management. Delays of 2-3 hours have proved fatal. Every effort should be made to reduce the time needed to secure a patient’s airway, preferably in the operating room under the direction of an airway management team (anesthesia and otolaryngology) and initiate antibiotic therapy. During the interval, unnecessary stress for the child should be prevented, the throat should not be examined, and radiographic confirmation usually is omitted.
The airways should be secured as early as possible after diagnosis. A large body of literature attests to the safety and efficacy of elective nasotracheal intubation, which is the treatment of choice. Optimally the child’s airway should be secured by the most skilled individual available, usually an anesthesiologist with an ENT surgeon immediately accessible if the child requires a tracheotomy. A short period of airway maintenance is usually all that is required. A nasotracheal tube that is 0.5 mm smaller than that predicted by the patient’s age is recommended. The criteria for extubation include being afebrile and swallowing comfortably. Repeat examination of the epiglottis and supraglottic structures by direct laryngoscopy or fiberoptic bronchoscopy is not normally necessary. Complications occurring after extubation may be laryngeal edema and subglottic granulations. Long-term complications of nasotracheal intubation are rare.
In about 10% of children with epiglottitis in whom there is severe airway obstruction, idiopathic pulmonary edema may occur before or after insertion of endotracheal tubes.65 The hypothetical mechanism is an increased pulmonary blood flow secondary to airway obstruction, causing markedly negative intra-pleural pressure with increased venous return to the right side of the heart and decreased left ventricular output. These changes increase the pulmonary microvascular pressure and produce a role in altering vascular permeability, but it is not a necessary prerequisite. Continuous positive airway pressure in intubated patients may decrease the occurrence of pulmonary edema.
Until the results of sensitivity tests are known, the child should be treated with an intravenous antibiotic to cover the majority of possible isolates. Increasing numbers of H. influenzae produce b-lactamase (up to one-third in some communities); therefore, initial treatment is usually a second-generation cepha-losporin such as cefuroxime or a third-generation cephalosporin such as cefotaxime or ceftriaxone. If the isolate is proved to be susceptible, ampicillin, a less expensive agent, may be substituted. If group A S. pyogenes is isolated form the airway, penicillin is the drug of choice. When S. aureus is isolated, a semisynthetic penicillinase-resistant penicillin or glycopeptide such as vancomycin should be used depending on sensitivity patterns. Erythromycin should be used for C. diphtheriae.
Although there have been some recommendations to use corticosteroids, no controlled data support their use; in fact, they may be hazardous because of the side effects.5 Therapy with inhaled epinephrine has not proven beneficial.
Bacterial Tracheitis
Bacterial tracheitis usually presents as severe upper airway obstruction, most often in a child who has had viral croup for several days.1 Alternative names are membranous laryngotracheobronchitis, pseudomembranous croup, and membranous croup.5,66
Disease Mechanisms. Direct bacterial infection of the tracheal mucosa is caused by a variety of organisms.66-72 S. aureus is the most common bacteria reported. Influenza virus, parainfluenza virus, and enterovirus have been isolated in children with bacterial tracheitis, suggesting that bacterial invasion may occur in an airway already inflamed by viral infection.72 Bacterial tracheitis is a recognized complication of measles.70 In a review of tracheal cultures obtained from patients with bacterial tracheitis, 40% contained anaerobic organisms and 75% of the cultures contained more than one organism.70 This suggests that, once injured (perhaps by an initial viral process), the tracheal mucosa becomes vulnerable to bacterial colonization, leading to opportunistic infection.68
The bacterial infection causes a diffuse inflammatory process of the larynx, trachea, and bronchi with mucopurulent exudate and semiadherent "membranes" within the trachea.66 These membranes contain numerous neutrophils and cellular debris, and cause major obstruction.
Clinical Manifestations. Bacterial tracheitis most commonly occurs in children in the age group vulnerable to viral croup. The initial clinical features are similar to those of viral croup, but there is a high fever (usually greater than 38.5°C), the child appears toxic, and there is severe airway obstruction. About half of cases have clinical or radiographic evidence of pneumonia. A rare complication is toxic shock syndrome.66
The differential diagnosis includes severe laryngotracheobronchitis (viral croup), laryngeal or tracheal foreign body aspiration, and epiglottitis. Bacterial tracheitis has a longer duration, a more typical barking cough than epiglottitis, and no drooling. Diphtheria once was a serious consideration as the most common cause of "membranous croup," which produces severe airway obstruction because of adherent membranes that separate from the airway wall with difficulty, causing bleeding.
Diagnosis. A lateral neck radiograph shows subglottic narrowing and often reveals findings of radiopaque material in the airway lumen (pseudomembrane).66,67,71 This may be confused with a foreign body.9,67
Endoscopy reveals thick mucopurulent material and sloughed epithelium.66 The epithelium forms a sheetlike pseudomembrane that separates easily from the airway wall without hemorrhage and sometimes extends from the trachea to the major bronchi.
Bacterial cultures of tracheal secretions reveal the organism(s). The results from blood cultures usually are negative. White cell counts may be high or normal.
Management. In a child suspected of having bacterial tracheitis, management should occur in a pediatric intensive care unit. Intubation usually is needed to relieve the airway obstruction, which takes days to resolve.66 Intermittent positive-pressure breathing is sometimes needed. Repeated suctioning is usually required because of the thick secretions and their tendency to form crusts, with intubation lasting 3-11 days. Meticulous care of the endotracheal tube, with adequate humidification and frequent suctioning, is imperative. Endotracheal tube plugging is a common complication. In extreme cases, reintubation with a fresh endotracheal tube and initiation of more aggressive pulmonary toilet is required. Sometimes, repeat endoscopic removal of the pseudo-membrane is required. Occasionally, tracheotomy is needed if endotracheal tube management of secretions proves too difficult.
Nebulized epinephrine or corticosteroids do not relieve the acute airway obstruction. Intravenous antibiotics are vital, and should be directed initially against the four common pathogens until the results of tracheal cultures are known. The usual choice is oxacillin (150 mg/kg/day with four equal doses given every 6 hours) and a third-generation cephalosporin such as cefotaxime (150 mg/kg/day with four equal doses given every 6 hours) until the results of cultures and sensitivities are known.
Retropharyngeal Abscess
Retropharyngeal abscess (RPA) is a relatively uncommon infection of the space anterior to the prevertebral layer of the deep cervical fascia.73-78 This infection is most common in children younger than 3 or 4 years, because of the rich concentration of lymph nodes in this space. These lympathic chains begin to atrophy about the third or fourth year of life. Thus 50% of cases of RPA occur between 6 and 12 months of age, and the vast majority occur before age 6 years.74,76,78 The symptoms of RPA can be similar to those of acute epiglottitis; however, children with acute epiglottitis usually appear more toxic and progress to respiratory distress much more rapidly than those with RPA. The unique abnormal physical finding in older children and adults with RPA, which is an asymmetric bulge of the posterior pharyngeal wall when inflammation has progressed to phlegmon or frank abscess, may be difficult to appreciate in infants or toddlers.73,74,77
Retropharyngeal lymph node infection in children classically results from extension of oropharyngeal infections including pharyngitis, tonsillitis, and adenitis.74,75 The infection progresses through three stages: cellulitis, phlegmon, and abscess. Probably only the last stage requires surgical drainage. Contrast-enhanced CT is invaluable in delineating the stage of inflammation, but boundaries (incomplete vs complete ring enhancement) between phlegmon and abscess stages may be blurred. Trauma, often caused by a fall while holding a pencil or stick in the mouth, and dental infections are the usual underlying causes of RPA in older children and adults.73,74
Most symptoms and signs of RPA are identical to those of acute epiglottitis. They include fever, irritability, oropharyngeal pain, unusual positioning of the head and neck, and odynophagia. As the infection progresses, there may be refusal to swallow solids or liquids including saliva, which leads to pooling in the oropharynx and overflow drooling. The most suggestive physical signs of both diseases are hyperextension of the neck, torticollis, muffled voice, stridor, and other signs of upper airway obstruction.78 In several case series of patients with RPA, the classic physical sign of RPA, which is bulging of the posterior pharynx, was noted; however, this was present in fewer than 50% of infants.78 The major causative organisms are S. pyogenes, S. aureus, and oropharyngeal anaerobic bacteria.73,76
The differential diagnosis includes acute epiglottitis, foreign body aspiration, vertebral osteomyelitis, hematoma (particularly in boys with hemophilia), and lymphoma.
Diagnostic studies commonly include roentgenograms of the cervical area and CT scans. To minimize false-positive results, a lateral view roentgenogram may be helpful when obtained with the patient in sitting position with the neck hyperextended and during inspiration, if possible. (See Figure 2.) Normal buckling of upper cervical prevertebral soft tissues that occurs during flexion or expiration can simulate a retropharyngeal mass.78 A retropharyngeal space measured from the anterior aspect of the second cervical vertebral body to the soft tissues of the posterior pharyngeal wall greater than 7 mm (normal, 4-7 mm) or a retrotracheal space greater than 14 mm in a child (measured from the anterior/inferior aspect of C6 to the posterior pharyngeal wall) suggests a mass caused by phlegmon, pus, or blood.3,73,75,78
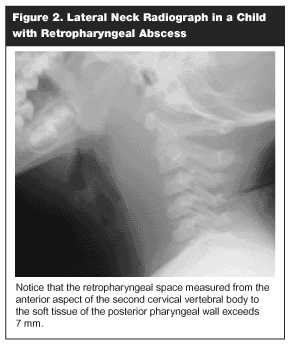 |
Another way to interpret abnormal thickening of the retropharyngeal soft tissue is the fact that in an infant or young child, the soft tissues between the posterior aspect of the aerated pharynx and the anterior aspect of the vertebral column should not exceed the anteroposterior diameter of the cervical bodies.48 However, in infants, it is common to see "pseudothickening" of the retropharyngeal soft tissues when the lateral radiograph is taken without the neck being well extended. Supportive evidence that there is true widening of the retropharyngeal soft tissues includes apex anterior convexity of the retropharyngeal soft tissues. The only radiographic feature that can differentiate abscess from cellulitis is the identification of gas within the retropharyngeal soft tissues.48 In suspicious cases, CT should be performed to define the extent of disease and help predict cases in which a drainable fluid collection is present.3,48 On CT, a low-attenuation, well-defined area with an enhancing rim is suspicious for a drainable fluid collection.
Management of RPA depends on the maturity of the infection and the degree of airway compromise. Cellulitis or a phlegmon requires only targeted antibiotics active against common oral facultative and anaerobic bacteria.74 Historically, the treatment of an abscess has been surgical drainage. However, several recent studies have shown that medical management alone can be curative for more than 50% of children with RPA (usually defined by CT).73,77 Security of the airway is of paramount importance. All patients should have a secure intravenous catheter placed and be given nothing by mouth. If a patient is stable and the benefit of avoiding a surgery outweighs the risk of complications from RPA, a trial of intravenous antibiotics for 24-48 hours can be initiated. If there is no improvement, either clinically or as determined by imaging, then the decision may be made to perform surgical drainage.
Suppurative complications of RPA include rupture of the abscess with aspiration, asphyxiation, or pneumonia; empyema; and mediastinitis.78 Vascular complications include thrombophlebitis of the internal jugular vein and erosion through the carotid artery sheath.3,48,73
Diphtheria
Pharyngeal diphtheria is now extremely rare in the United States. A single probable case was reported to the Centers for Disease Control and Prevention in 1998.79 The disease occurs primarily among unimmunized or poorly immunized members of socioeconomically disadvantaged groups.80 Of the 41 reported respiratory diphtheria cases in the United States from 1980 to 1995, none of the patients had been adequately vaccinated, and the four children who died following severe illness had not received any diphtheria immunizations.80 Appropriate vaccination protects against both severe disease and death.
The most notable physical finding is the grayish brown diphtheritic pseudomembrane, which may involve one or both tonsils or may extend widely to involve the nares, uvula, soft palate, pharynx, larynx, and tracheobronchial tree. Involvement of the latter structures can cause life-threatening respiratory obstruction. Removal of the membrane reveals a bleeding and edematous submucosa. Soft-tissue edema and prominent cervical and submental adenopathy may create a bull-neck appearance. The potent toxin elaborated by Corynebacterium diphtheriae may produce cardiac toxicity and neurotoxicity.84 The diagnosis, which may be strongly suspected on epidemiologic and clinical grounds, should be confirmed by culture of the pseudomembrane in Loeffler’s or tellurite selective medium. Pharyngeal diphtheria is treated with equine hyperimmune diphtheria antitoxin and penicillin or erythromycin.
Conclusion
The child’s airway is vulnerable to obstruction secondary to its anatomy and small size. Although there are many potential causes of upper airway obstruction, a few diagnoses predominate. Common airway infections can progress to critical airway obstruction and hypoxia, causing organ damage or death.
Individuals caring for children must be prepared to diagnose and treat airway emergencies expeditiously. Appropriate management of acute upper airway obstruction tests the organization of emergency care systems. Successful management of airway emergencies in children requires a team approach, including the skills of the primary physician and the staff of the emergency department, radiology department, and operating room staff. Management of these cases can be anticipated, and protocols can be established.
References
1. Cox PN. Current management of laryngotracheo bronchitis, bacterial tracheitis and epiglottitis. Intensive Care World 1993;10:8-12.
2. Perkin RM, van Stralen D. Pitfalls in pediatric airway management. Pediatric Emergency Medicine Reports 1997;2:91-102.
3. Rothrock SG, Perkin R. Stridor: A review, update, and current management recommendations. Pediatric Emergency Medicine Reports 1996;1:29-40.
4. Malhotra A, Krilov LR. Viral croup. Pediatrics in Review 2001;22:5-11.
5. Cherry JD. Croup (laryngitis, laryngotracheitis, spasmodic croup, laryngotracheo-bronchitis, bacterial tracheitis, and laryngotracheo bronchopneumonitis). In: Feign RD, Cherry JD, eds. Textbook of Pediatric Infectious Disease. Philadelphia: WB Saunders Co; 1998:228-241.
6. Kaditis AG, Wald ER. Viral croup: Current diagnosis and treatment. Pediatr Infect Dis J 1998;17:827-834.
7. Klassen TP. Croup: A current perspective. Pediatr Clin of North Am 1999;46: 1167-1178.
8. Skolnik NS. Treatment of croup: A critical review. Am J Dis Child 1989;142: 1045-1049.
9. Rencken I, Patton WL, Brasch RC. Airway obstruction in pediatric patients. Radiol Clin of North Am 1998;36:175-187.
10. Walner DL, Donnelly LF, Ouanounou S, et al. Utility of radiographs in the evaluation of pediatric upper airway obstruction. Am Otol Rhinol Laryngol 1999;108:378-383.
11. Westley CR, Cotton EK, Brooks JG. Nebulized racemic epinephrine by IPPB for the treatment of croup. Am J Dis Child 1978; 132:484-487.
12. Taussig LM, Castro O, Biandry PA, et al. Treatment of laryngotracheitis (croup): Use of intermittent positive pressure breathing and racemic epinephrine. Am J Dis Child 1975;129:790-795.
13. Steele DW, Stanucci KA, Wright RO, et al. Pulsus paradoxus: An objective measure of severity in croup. Am J Respir Crit Care Med 1998; 157:331-334.
14. Bank DE, Krug SE. New approaches to upper airway disease. Emerg Med Clin North Am 1995;13:473-487.
15. Fanconi S, Burger R, Maurer H, et al. Transcutaneous carbon dioxide pressure for monitoring patients with severe croup. J Pediatr 1990;117:701-705.
16. Mancker AJ, Petrack EM, Krug SE. Contribution of routine pulse oximetry to evaluation and management of patients with respiratory illness in a pediatric emergency department. Ann Emerg Med 1995;25:36-40.
17. Orlicek SL. Management of acute laryngotracheobronchitis. Pediatr Infect Dis J 1998;17:1164-1165.
18. Geelhoed GC. Croup. Pediatr Pulmonol 1997;23:370-374.
19. Prendergast M, Jones JS, Hartman D. Racemic epinephrine in the treatment of laryngotracheitis: Can we identify children for outpatient therapy? Am J Emerg Med 1994;12:613-616.
20. Waisman Y, Klein BL, Boenning DA, et al. Prospective randomized double-blind study comparing L-epinephrine and racemic epinephrine aerosols in the treatment of laryngotracheitis (croup). Pediatrics 1992;89:302-306.
21. Butte MJ, Nguyen BX, Hutchism TJ, et al. Pediatric myocardial infarction after racemic epinephrine administration. Pediatrics 1999;104:e9.
22. Klassen TP. Recent advances in the treatment of bronchiolitis and laryngitis. Pediatr Clin of North Am 1997;44:249-261.
23. Ledwith CA, Shea LM, Mauro RD. Safety and efficacy of nebulized racemic epinephrine in conjunction with oral dexamethasone and mist in the outpatient treatment of croup. Ann Emerg Med 1995;25:331-337.
24. Rizos JD, DiGravio BE, Sehl MJ, et al. The disposition of children with croup treated with racemic epinephrine and dexamethasone in the emergency department. J Emerg Med 1998;16:535-359.
25. Kunkel NC, Baker MD. Use of racemic epinephrine, dexamethasone, and mist in the outpatient management of croup. Pediatr Emerg Care 1996;12:156-159.
26. Kairys SW, Olmstead EM, O’Connor GT. Steroid treatment of laryngotracheitis: A meta-analysis of the evidence from randomized trials. Pediatrics 1989;83:683-693.
27. Ausejo M, Saenz A, Pham B, et al. The effectiveness of glucocorticoids in treating croup: Meta-analysis. BMJ 1999;319:595-600.
28. Geelhoed GC, Turner J, MacDonald WB. Efficacy of a small single dose of oral dexamethasone for outpatient croup: A double blind placebo controlled clinical trial. BMJ 1996;313:140-142.
29. Luria JW, Gonzalez-del-Rey JA, DiGiulio GA, et al. Effectiveness of oral or nebulized dexamethasone for children with mild croup. Arch Pediatr Adolesc Med 2001;155:1340-1345.
30. Johnson DW, Schuh S, Koren G, et al. Outpatient treatment of croup with nebulized dexamethasone. Arch Pediatr Adolesc Med 1996;150:349-355.
31. Cruz MN, Stewart G, Rosenberg N. Use of dexamethasone in the outpatient management of acute laryngotracheitis. Pediatrics 1995;96:220-223.
32. Rittichier KK, Ledwith CA. Outpatient treatment of moderate croup with dexamethasone: Intramuscular versus oral dosing. Pediatrics 2000;106:1344-1348.
33. Geelhoed GC, MacDonald WBG. Oral dexamethasone in the treatment of croup: 0.15 mg/kg versus 0.3 mg/kg versus 0.6 mg/kg. Pediatr Pulmonol 1995;20:362-368.
34. Szefler SJ. Pharmacodynamics and pharmacokinetics of budesonide: A new nebulized corticosteroid. J Allergy Clin Immunol 1999;104:S175-S183.
35. Klassen TP, Feldman ME, Walters LK, et al. Nebulized budesonide for children with mild-to-moderate croup. N Engl J Med 1994;331:285-289.
36. Fitzgerald D, Mellis C, Johnson M, et al. Nebulized budesonide is as effective as nebulized adrenaline on moderately severe croup. Pediatrics 1996;97:722-725.
37. Klassen T, Walters L, Feldman M, et al. The efficacy of nebulized budesonide in dexamethasone-treated outpatients with croup. Pediatrics 1996;97:463-466.
38. Husby J, Agertoft L, Mortensen S, et al. Treatment of croup with nebulized steroid (budesonide): A double blind, placebo controlled study. Arch Dis Child 1993;68:352-355.
39. Johnson DW, Jacobsen S, Edney PC, et al. A comparison of nebulized budesonide, intramuscular dexamethasone, and placebo for moderately severe croup. N Engl J Med 1998;339:498-503.
40. Klassen TP, Craig WR, Moher D, et al. Nebulized budesonide and oral dexamethasone for treatment of croup: A randomized controlled trial. JAMA 1998; 279:1629-1632.
41. Jaffe DM. The treatment of croup with glucocorticoids. N Engl J Med 1998; 339:553-555.
42. Tobias JD. Heliox in children with airway obstruction. Pediatr Emerg Care 1997;13:29-32.
43. Terregino CA, Nairn J, Chansky ME. The effect of heliox on croup: A pilot study. Acad Emerg Med 1998;5:1130-1133.
44. Smith SW, Biros M. Relief of imminent respiratory failure from upper airway obstruction by use of helium-oxygen: A case series and brief review. Acad Emerg Med 1999;6:953-956.
45. Weber JE, Chudnofsky CR, Younger JG, et al. A randomized comparison of helium-oxygen mixture and racemic epinephrine for the treatment of moderate to severe croup. Pediatrics 2001;107:e96.
46. Damm M, Eckel HE, Jungehulsing M, et al. Management of acute inflammatory childhood stridor. Otolaryngol Head Neck Surg 1999;121:633-638.
47. Chan ED, Hodges TN, Parsons PE. Sudden respiratory insufficiency in a previously healthy 47-year-old man. Chest 1997;112:1419-1422.
48. Sabella C, Fontanarosa PM, Pichichero M. Infectious causes of acute upper airway obstruction in infants and children: Epiglottitis and croup. Emerg Med Reports 1992;13:19-28.
49. Cressman WR, Myer III, CM. Diagnosis and management of croup and epiglottitis. Pediatr Clin of North Am 1994;41:265-276.
50. Milczuk H. Upper airway obstruction in children. New Horizons 1999;7: 326-334.
51. Berry FA, Yemen TA. Pediatric airway in health and disease. Pediatr Clin North Am 1994;41:153-180.
52. Lee TWR, Sandoe JAT. Epiglottitis caused by group C. streptococcus. Acta Pediatr 2001;90:1085.
53. Belfer RA. Group A ß-hemolytic streptococcal epiglottitis as a complication of varicella infection. Pediatr Emerg Care 1996;12:203-204.
54. Slack CL, Allen GC, Morrison JE, et al. Post-varicella epiglottitis and necrotizing fasciitis. Pediatrics 2000;105:e13.
55. Valdepena HG, Wald ER, Rose E, et al. Epiglottitis and Haemophilus influenzae immunization: The Pittsburgh Experience A five-year review. Pediatrics 1995;96:424-427.
56. Garpenholt O, Hugosson S, Fredlund H, et al. Epiglottitis in Sweden before and after introduction of vaccination against Haemophilus influenzae type b. Pediatr Infect Dis J 1999;18:490-493.
57. McCollough M. Progress toward eliminating Haemophilus influenzae type b among infants and children. Ann Emerg Med 1999;34:110-111.
58. Gorelick MH, Baker D. Epiglottitis in children, 1979 through 1992: Effects of Haemophilus influenzae type b immunization. Arch Pediatr Adolesc Med 1994; 148:47-50.
59. Kucera CM, Silverstein MD, Jacobson RM, et al. Epiglottitis in adults and children in Olmsted County, Minnesota, 1976 through 1990. Mayo Clin Proc 1996; 71:1155-1161.
60. Goldhagen JC. Supraglottitis in three young infants. Pediatr Emerg Care 1989; 5:175-177.
61. Blackstock D, Adderly RJ, Steward DJ. Epiglottitis in young infants. Anesthesiology 1987;97-100.
62. Brilli RS, Benzing G, Cotcamp DH. Epiglottitis in infants less than two years of age. Pediatr Emerg Care 1989; 5:16-21.
63. Losek J, Dewitz-Zink B, Melzer-Lange M, et al. Epiglottitis: Comparison of signs and symptoms in children less than 2 years old. Ann Emerg Med 1990; 19:55-58.
64. Mauro RD, Pool SR, Lockhart CH. Differentiation of epiglottitis from laryngotracheitis in the child with stridor. Am J Dis Child 1988;142:679-682.
65. Kallet RH, Daniel BM, Gropper M, et al. Acute pulmonary edema following upper airway obstruction. Respir Care 1998;43:476-480.
66. Gallagher PG, Myer III CM. An approach to the diagnosis and treatment of membranous laryngotracheo bronchitis in infants and children. Pediatr Emerg Care 1991;7:337-342.
67. Orenstein JB, Thomsen JR, Baker SB. Pneumococcal bacterial tracheitis. Am J Emerg Med 1991;9:243-245.
68. Lerner DL, Fontan JJP. Prevention and treatment of upper airway obstruction in infants and children. Curr Opin Pediatr 1998;10:265-270.
69. Fortenberry JD, Mariscalco M, Louis PT, et al. Severe laryngotracheobronchitis complicating measles. AJDC 1992;146:1040-1043.
70. Brook I. Aerobic and anaerobic microbiology of bacterial tracheitis in children. Pediatr Emerg Care 1997;13:16-18.
71. Bernstein T, Brilli R, Jacobs B. Is bacterial tracheitis changing? A 14-month experience in a pediatric intensive care unit. Clin Infect Dis 1998;27:458-462.
72. Donnelly BW, McMillan JA, Weiner LB. Bacterial tracheitis: Report of eight new cases and review. Rev Infect Dis 1990;12:729-735.
73. Lee SS, Schartz RH, Bahadori RS. Retropharyngeal abscess: Epiglottitis of the new millennium. J Pediatr 2001;138:435-437.
74. Gaglani MJ, Edwards MS. Clinical indicators of childhood retropharyngeal abscess. Am J Emerg Med 1995;13:333-336.
75. Toback S, Herr S. Retropharyngeal abscess in a toxic-appearing infant. Pediatr Emerg Care 2001;17:255-257.
76. Morrison JE, Pashley NRT. Retropharyngeal abscesses in children: A 10 year review. Pediatr Emerg Care 1988;4:9-11.
77. Lalakea ML, Messner AH. Retropharyngeal abscess management in children: Current practices. Otolaryngol Head Neck Surg 1999;121:398-405.
78. Goldenberg D, Golz A, Joachims HZ. Retropharyngeal abscess: A clinical review. J Laryngo Otol 1997;111:546-550.
79. Bisno AL. Acute pharyngitis. N Engl J Med 2001;344:250-211.
80. Bisgard KM, Hardy IR, et al. Respiratory diphtheria in the United States, 1980 through 1995. Am J Public Health 1998;88:787-791.
Individuals caring for children must be prepared to diagnose and treat airway emergencies expeditiously. Appropriate management of acute upper airway obstruction tests the organization of emergency care systems. Successful management of airway emergencies in children requires a team approach, including the skills of the primary physician and the staff of the emergency department, radiology department, and operating room staff.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
