2008 Salary Survey Results: Survey shows some wage growth, but experts say industry still lagging
2008 Salary Survey Results
Survey shows some wage growth, but experts say industry still lagging
More money needed to find, keep experienced staff
Salaries remain strong for readers of Clinical Trials Administrator, according to the 2008 Salary Survey.
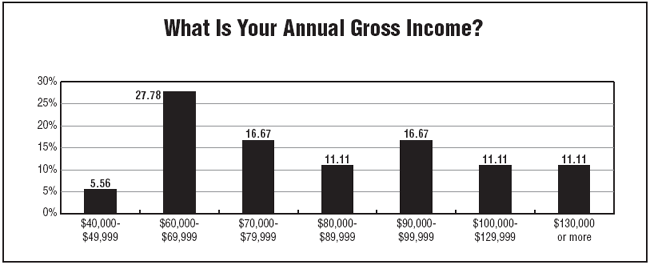
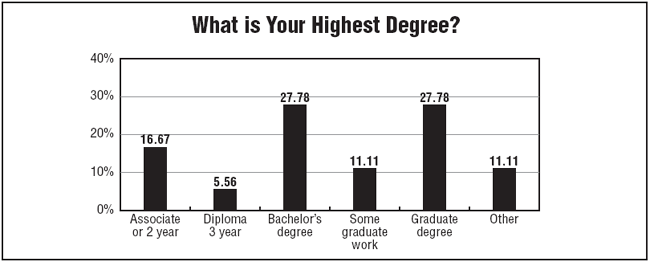
The survey found that more than 94% of respondents reported making $60,000 or more per year. And more than 22% reported making more $100,000 or more per year.
The salary findings are not surprising given the responsibilities and experience level of the survey respondents, notes Tamara Dowd Owen, RN, MSN, MBA, director of clinical trials at Pinehurst Medical Clinic in Pinehurst, NC.
"It's an age group trend," Owen says.
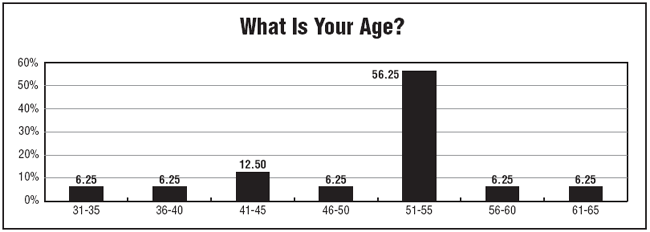
Survey respondents tend to be older, with nearly 94% reporting their ages to be 36 years or older, and nearly 70% reporting an age of 51 years or older. "Salaries tend to be a little higher if you're at a director level," Owen says. "Some monitors also are making between $80,000 and $90,000 a year."
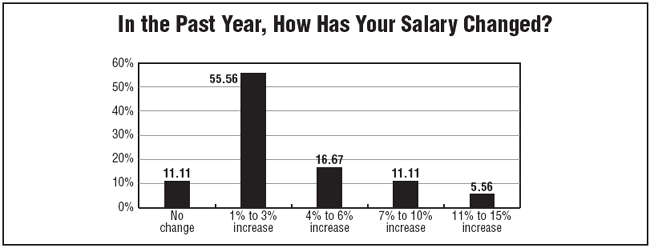
Also, the survey found that nearly 90% of respondents reported receiving a raise last year, with a little more than half reporting a modest 1% to 3% raise. Survey respondents tend to be an experienced and educated group of clinical research leaders.
Close to 95% of the people responding to CTA's salary survey have worked in health care for 13 or more years, with two-thirds reporting having had health care careers of 25 or more years.
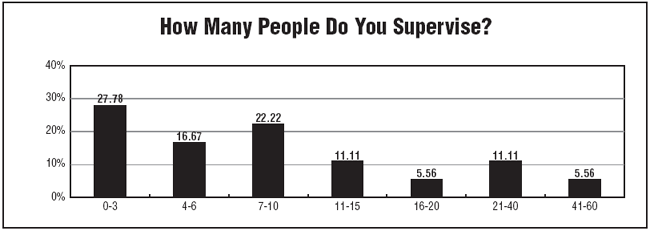
Survey respondents also reported having a supervisor role with close to three-quarters reporting having four or more employees to supervise. One-half of those surveyed said their staffing levels stayed the same last year, and a little more than one-fifth said they had lost staff, while more than one-quarter said they had gained staff.
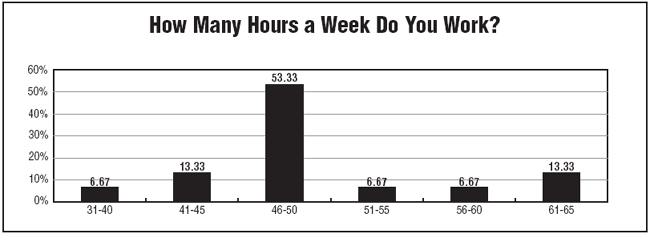
The clinical research workload for respondents meant long work weeks. According to survey results, nearly 94% of respondents work more than 40 hours per week, and a little more than half work between 46 and 50 hours per week, with one-quarter reporting having work weeks that are 51 hours or longer.
These hard-working, highly-paid, well educated, and very experienced clinical research professionals are, perhaps, the very same directors and managers who are finding it difficult to come up with salary packages that will help them recruit and retain the best qualified clinical trial associates.
"Increasingly, it's difficult to find experienced staff," Owen says. "How do you lure those people?"
About 90% of the CT associate applicants to Pinehurst Medical Clinic are coming from a nursing background, Owen estimates.
The problem is that a research site might only be able to pay a new clinical research associate $35,000 per year, which makes it difficult to recruit the most capable and bright research field newcomers, particularly if they are accustomed to higher salaries, she adds.
"For us, it's case-by-case, and we have a little bit of latitude with regard to salaries," Owen says.
But there are times when the best qualified candidate is out of reach because of salary limitations, she notes.
"We had a job candidate from New Hampshire who was making $75,000 there and had tons of experience," Owen recalls. "We couldn't get her a salary that high — the best we could do was $65,000."
Although, the North Carolina site has a lower cost of living, the job applicant could not be convinced to take the pay cut, Owen says.
"We were at a place where we couldn't offer more, and she couldn't see how it would happen," she adds.
One of the problems for clinical trial sites is that they compete for employees with pharmaceutical companies and clinical research organizations that sometimes can pay more for experienced staff.
"From an academic perspective, we're constantly competing with industry for staff," says Ramesh Gunawardena, MBA, director of clinical trial operations in the clinical trial office of Beth Israel Deaconess Medical Center of Boston, MA.
New CR associates quickly find that the workload piles up quickly at a CR site, Gunawardena says.
"You're dealing with patients and running protocols, and at the same time you have all of these requirements for submitting forms to the IRB, sponsor, handling paperwork, IND safety reports, etc.," he explains. "The workload requirement is huge, and it's not on par with what the pay scale is."
Salaries are a drawback to finding good clinical trial employees because so many of these professionals come from health care backgrounds where they were making more money, says Elizabeth E. Hill, PhD, RN, an associate chief of staff for research at the VA Sierra Nevada Health Care System in Reno, NV. Hill previously was the director of the clinical research management program at Duke University School of Nursing in Durham, NC.
The ideal clinical trial managers are people who have been educated in clinical trial management after having some experience working in health care as a nurse or some other discipline, Hill says.
Duke University has a master's degree program in research for nurses or post-graduate certificate in research for people with master's degrees or doctorates.
Duke's model is the new trend of training research associates in an academic setting before they venture out into the CR world for on-the-job training.
Of course, these CR professionals start out their research careers at an advanced level when compared with those who enter at the bottom rung.
The Duke program doesn't address basic CR training, Hill notes.
Education and training are limited for CR staff at the one or two clinical trial level, she says.
"There is a real lack of education for people at the study coordinator level," Hill notes. "It's almost all on-the-job training."
It might have worked in the past to hire nurses with no research experience and expect them to learn about clinical research on the job, but Hill says this is no longer the best strategy.
"I feel like we're past this now," she says. "You need people with some background and education in research because someone could have a PhD and not keep up with it."
However, the CR industry will have to recognize that with the updated model of hiring people who have either CR experience or advanced education in clinical research, there comes the obligation of higher salaries, Hill says.
"I've had students [with experience as nurses and] with master's degrees in research who were offered salaries of $40,000 to $50,000 or less," Hill says. "And they could make more than that as a staff nurse somewhere with no other responsibilities than getting their work done each day."
One time, a research director called Hill to find a master's level research student who had extensive health care experience.
"He wanted to offer the person $55,000, and I said, 'This is not going to happen,'" Hill recalls.
Hill had another student with a doctorate, who earned a post-doctoral degree in clinical research management, and one research site offered him $50,000. "So he went somewhere else," she says.
Since CT directors often must hire employees who have very little or no CR experience, the key is to provide thorough training, Owen notes.
"We've gotten back to the basics," Owen says. "We review regulatory pieces, things that need to be filed, IRB submissions, protocol violations, policies on reporting adverse events, and other things that people should know."
While it sounds elementary, it's necessary to go over the basics when your staff is new to research, she adds.
"Those are the things we've never felt like we needed to tell people who were experienced," Owen says.
The CTA survey findings come in as the health care system — normally considered a safe haven in an economic storm – starts to feel the impact of the recession. For example, fewer patients are seeking hospital care while at the same time a growing proportion of patients need help paying for care, according to a recent report from the American Hospital Association (AHA). The report also noted that hospitals — which employ 5 million people nationwide — could be facing uncertain times as their financial health falters and ability to borrow funds for improving facilities and updating technology is squeezed.
The report is based on survey results from 736 hospitals and information from DATABANK, a web-based reporting system used in 30 states to track key hospital trends. Many hospitals are beginning to see the effects of the economic downturn, with more than 30% of survey respondents reporting a moderate to significant decline in patients seeking elective procedures, and nearly 40% of respondents reporting a drop in admissions overall. The majority of hospitals surveyed also noted an increase in the proportion of patients unable to pay for care. Uncompensated care was up 8% from July to September versus the same period last year, according to the report.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
