Rheumatoid Arthritis: An Emergency Physician’s Perspective
November 16, 2014
Reprints
This article is adapted from one that appears in the November 2014 issue of Primary Care Reports.
Executive Summary
-
Rheumatoid arthritis is a systemic polyarticular inflammatory arthritis that is intimately associated with autoantibodies, rheumatoid factor, and anti-CCP.
-
Early diagnosis and treatment is essential in preventing
complications and functional disability. -
The most commonly used tests are the serum RF, ACPA, ESR, and CRP. RF and anti-CCP antibodies are helpful in the diagnosis of RA and in prognostication.
-
Acute flares are commonly treated with high dose methylprednisolone, over a 2-4 week course. Patients who fail to respond, or who are already on steroids, may require admission to the hospital.
Introduction
Rheumatoid arthritis (RA) is a systemic polyarticular form of inflammatory arthritis of unclear etiology. It primarily involves the synovial joints and is commonly symmetric. Joint pain and swelling are most commonly seen in patients with this condition. The presence of autoantibodies such as rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPAs), which the anti-cyclic citrullinated peptide (CCP) falls under, are found in the majority of patients. Left untreated, there is a high risk of significant deformity and disability.
Over the past decade, much has been learned of the pathophysiology and treatment of RA. The development of disease-modifying antirheumatic drugs (DMARDs) such as methotrexate and biologic agents has changed the disease course. Due to the efficacy of these therapies, there has been a shift of emphasis toward diagnosing the disease much earlier. Multiple studies have shown that early intervention is critical for achieving optimal outcomes.1 Emergency physicians play an important role in initial referral of suspected cases.
This article will review the important features of RA, including its clinical manifestations and predisposing factors, and will present a summary of the differential diagnoses that mimic RA, and the work-up and treatment of the disease.
Epidemiology
The global prevalence of RA is approximately 0.24%.2 However, in the United States, the estimates are believed to be higher at 0.5-1.0%.3 In 2007, an estimated 1.5 million U.S. adults were affected by the disease, with prevalence of 9.8 per 1000 in women and 4.1 per 1000 in men.4 In general, women are affected two to three times more often than men.5 The average age of RA patients is 66.8 years.6
Risk Factors
The underlying etiology of RA is still unclear. However, researchers have identified several risk factors believed to affect disease initiation. For example, the complex interaction between genetic factors and environmental exposures may predispose a patient to having the disease.7
Cigarette smoking is a significant risk factor. A recent meta-analysis that included 4552 patients found that the risk of developing RA increased by 26% (relative risk = 1.26) among those who smoked 1-10 pack-years compared to never smokers. This risk doubled among those with more than 20 pack-years8 and is found to be highest among people with positive anti-CCP antibodies.
Infectious causes have been implicated through epidemiologic studies in the development of RA. One such pathogen is Porphyromonas gingivalis, the main etiologic agent for periodontitis. Patients with RA have been found to have a higher prevalence of periodontal disease (3.95%) compared to the general population (1%).9 P. gingivalis has the ability to express peptidyl arginine deiminase, an enzyme responsible for post-translational citrullination of arginine residues. The exposure to these citrullinated antigens predisposes one to develop anti-CCP antibodies, which are implicated in the pathogenesis of RA.10
Socioeconomic and occupational exposures may present as risk factors for RA. According to a nationwide study in Sweden, an increased standardized incidence ratio of 1.4-1.8 was found among male miners and quarry workers compared to other occupations.11 Silica exposure among male smokers also has been linked to the development of RA.12
Clinical Manifestations and Classification Criteria
In most cases, RA presents insidiously and can develop over weeks to months before the diagnosis is confirmed. The predominant symptoms are pain, stiffness, and swelling of the peripheral joints.13
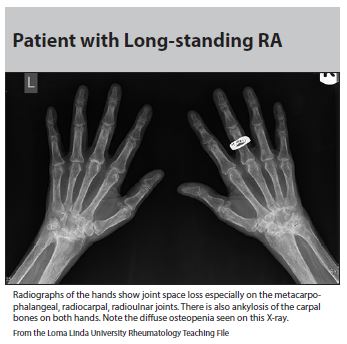
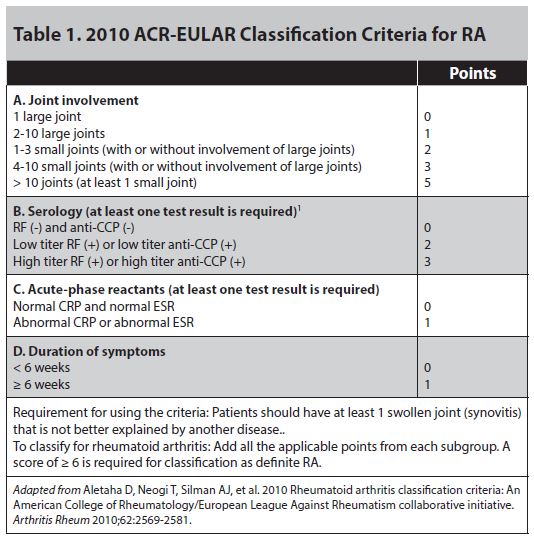
It should be noted that the symptoms of morning stiffness and the finding of joint swelling should preferably be present for at least 6 weeks. There are several clinical entities that can mimic a typical RA presentation, especially early in the disease process (see differential diagnosis section). Most infectious mimics of RA are self-limited and resolve in around 6 weeks. Some patients with early or very early presentations of RA may be difficult to diagnose, as they lack the characteristic complications such as rheumatoid nodules and radiographic changes (periarticular osteopenia and bone erosions, see Figures 1 and 2). The ACR and the European League Against Rheumatism (EULAR) for the classification of RA in 201014 reflect the evolving knowledge in the pathogenesis and treatment of RA and the need to identify the patients who have the disease early. The 2010 ACR/EULAR classification criteria are presented in Table 1.
Using these criteria, a patient is classified as having RA if he or she has at least 6 points (out of possible 10). However, the patient needs to have at least one joint affected with definite signs of clinical synovitis (swelling, not just arthralgia/joint pain) that is not better classified by another disease.
Differential Diagnosis
Several clinical entities share features with RA, so they must be excluded prior to committing to the diagnosis.
Viral Infections
Rubella. In children, the first manifestation usually is the characteristic maculopapular rash, which is pruritic and occurs 14-17 days after exposure. The rash may start on the face and later spread to the lower half of the body. Lymphadenopathy in the head and neck also are common in children. Infection in adults tends to be of longer duration.15 Arthritis is more common in adults and usually involves the knees, wrist, and fingers. Arthralgias and arthritis are not considered complications of rubella but are an integral part of the disease presentation.16
Parvovirus B19. Parvovirus B19 infects only humans. The symptoms vary depending on age and overall health.17 In children, it presents as a mild febrile illness with a rash (erythema infectiousum). In adults, swollen and painful joints are common. The arthritis is usually symmetric, involving the small joints of the hands (especially the proximal interphalangeal and metacarpophalangeal joints), wrists, knees, and feet.18 The difference between RA and parvovirus B19 infection is that 75% of patients with the infection eventually will develop a rash and the arthritis is self-limiting. In a majority of patients, the disease resolves in 2 weeks.18
Hepatitis B. Infection with hepatitis B virus (HBV) can result in extrahepatic manifestations. A serum-sickness like prodrome is seen in approximately one-third of patients acquiring HBV and presents with arthritis and rash. This prodrome spontaneously resolves at the onset of clinical hepatitis with few significant sequelae.19 Joint destruction is rare.
Hepatitis C. Joint swelling is the most common extrahepatic manifestation of hepatitis C virus (HCV) infection. The underlying etiology of this is unclear but is thought to be a localized inflammatory reaction directed toward viral invasion or deposition of cryoglobulin-induced immune complexes in the synovial fluid.20 HCV infection may present as symmetric arthritis, especially of the small joints. RF is generally positive in HCV infection but the anti-CCP is negative. Also, the arthritis in HCV infection is non-deforming and associated with less morning stiffness, and rheumatoid nodules are less likely than is the case in RA.20
Rheumatic Diseases. RA may be difficult to differentiate from other rheumatic diseases such as systemic lupus erythematosus (SLE), psoriatic arthritis (PsA), dermatomyositis, and mixed connective tissue disease. This is especially true during the first few months of the development of RA.1 Differentiating these entities is tricky, and is probably best left to the specialist. SLE may be accompanied by malar rash, oral ulcers, and positive serologic tests for antinuclear antibodies (ANA), anti-double-stranded DNA antibodies, and other autoantibodies. Dermatomyositis is associated with elevated serum creatine kinase and the characteristic rash on the face and neck along with proximal muscle weakness. The presence of psoriasis suggests a diagnosis of psoriatic arthritis (PsA). However, in about 20% of patients, the arthritis precedes the onset of the cutaneous lesions.21 A less common presentation of gouty arthritis can also be mistaken for RA, as the former may present as polyarticular synovitis. If gout is suspected, then joint aspiration with an assessment for presence of crystals is necessary.
Laboratory Testing
The most commonly used tests are the serum RF, ACPA, ESR, and CRP. RF and anti-CCP antibodies are helpful in the diagnosis of RA and in prognostication. The presence of these antibodies predicts which patients are at high risk of functional decline and radiographic progression. They are also known to be positive in approximately half of RA patients, even before the onset of clinical disease.22 On the other hand, ESR and CRP are most helpful in estimating disease activity.
Rheumatoid Factor. RFs are present in 75-80% of RA patients at some point during the course of the disease.23 The sensitivity of RF in RA is 69% and the specificity is 85%.24 The specificity is low, as it may be present in a variety of other diseases such as HCV, Sjögren’s syndrome, SLE, bacterial endocarditis, and tuberculosis.25
Anti-cyclic Citrullinated Peptides. Anti-CCP antibodies have roughly the same sensitivity as RF at 67%. However, anti-CCP has a higher specificity of 95%.24 Although specific, this antibody can also be found in other diseases such as SLE, Sjögren’s syndrome, and active tuberculosis. Anti-CCP is rarely positive in hepatitis C. This is in contrast to RF, which is fairly common in HCV infections. The combination of RF and anti-CCP positive tests increases the probability of true positives in the diagnosis of RA.26
Erythrocyte Sedimentation Rate. The ESR test has been used in a variety of settings; however, its utility lies mostly in following levels of inflammation. ESR values may correlate with disease activity in RA. The pitfall of ESR, unfortunately, is that this test is nonspecific. Various factors such as age, hemoglobin level, renal function, and presence of infection affect the test values. Thus, care should be taken in using this test.
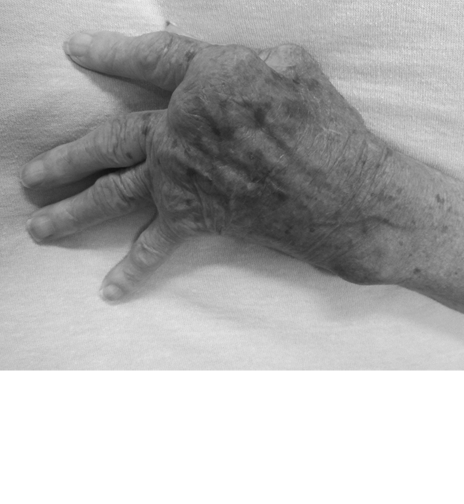
Her left hand shows complications of rheumatoid arthritis such as ulnar deviation of the digits and intrinsic muscle wasting. Heilman, James, MD. Rheumatoid Arthritis. Digital image. Wikipedia, 07 Aug. 2010. Web. 22 May 2014.
C-reactive Protein. CRP is an acute phase reactant that is considered a sensitive indicator of inflammation. In low levels, it is present in atherosclerosis, diabetes mellitus, and obesity. In high levels, it may indicate bacterial infection or inflammation.1 In RA, this may be used in the same way as the ESR to approximate disease activity. However, up to 40% of RA patients at presentation have normal ESR or CRP.27
Treatment
Emergency physicians often initiate treatment of an acute flare. Most of these patients are already on a more definitive DMARD, or have been recently diagnosed. It may take 2-3 months for DMARD medications to become fully effective. In the meantime, NSAIDs and steroids are often used to bridge the patient and to treat acute flares. Methylprednisolone is often used in a short course of 1-4 weeks with a rapid taper. It is important to note that while NSAIDs may provide symptomatic relief, they do not prevent joint destruction.
The definitive treatment of RA includes DMARDs, as they are able to modify the disease, as opposed to simply treating symptoms such as inflammation and pain.28
Synthetic DMARDs include methotrexate, sulfasalazine, leflunomide, and hydroxychloroquine. Methotrexate has been considered the anchor and first-line drug for the treatment of RA. Various combinations of synthetic DMARDs that include methotrexate have been found efficacious and safe. More recently, the so-called triple therapy (methotrexate, sulfasalazine, and hydroxychloroquine) was found to be possibly as effective as the combination of methotrexate and etanercept, a biologic DMARD.29
Biologic DMARDs include medications that target specific cytokines, receptors, or cell surface molecules in the pathophysiology of RA. They can be classified according to their mechanism of action: 1) tumor necrosis factor (TNF)-alpha inhibitors include the medications adalimumab, certolizumab pegol, etanercept, infliximab, and golimumab; 2) rituximab is a B-cell depleting monoclonal antibody that acts by targeting the CD20 cell surface molecule; 3) abatacept is a selective co-stimulation modulator targeting the T-cell activation process; 4) anakinra is an interleukin-1 receptor antagonist; 5) tocilizumab is an interleukin-6 inhibitor; 6) tofacitinib, the most recently approved RA therapy, inhibits the Janus kinase pathway. These medications usually are given either through subcutaneous injection or via infusion (tofacitinib is the only oral agent). They are indicated for the treatment of RA either as monotherapy (except infliximab) or in combination with methotrexate.
The emergency physician is likely to see acute adverse effects related to therapy. Nearly all of the DMARDs cause liver and bone marrow toxicity. Most, even the biological agents, increase the risk of infection. Luckily, most adverse events occur in the first 2-3 months of treatment. Many vaccines are ineffective, but it appears that tetanus immunization can be safely administered and is effective. Most of these medications interact with a broad range of drugs, so appropriate drug-drug interaction databases should be consulted before prescribing medication.
Methotrexate is often a first-line drug for RA. Side effects include nausea, vomiting, diarrhea, and stomatitis. Serious side effects include bone marrow suppression, liver toxicity, and pneumonitis. Acute renal failure and Stevens-Johnson syndrome are reported. Use of folic acid decreases side effects without reducing efficacy.
Sulfasalazine causes nausea and vomiting and is often initiated in a tapered dose to avoid these side effects. Rashes and allergic reactions are seen, as well as headaches and alopecia. Patients with G6PD may develop hemolytic anemia. Other serious side effects include bone marrow suppression, liver/renal/pulmonary toxicity, and Stevens-Johnson syndrome.
Leflunomide primarily causes hepatic toxicity (at times fatal). In general it is well-tolerated.
Hydroxychloroquine is generally used for milder disease. Minor side effects include nausea, rash, photosensitivity, and diarrhea. The most serious side effect is ophthalmologic with reduced peripheral and night vision. Bone marrow suppression, tinnitus, central nervous system disturbances (ataxia, irritability, etc.), and hepatic toxicity can occur.
Adalimumab carries a black box warning regarding serious infections, particularly TB and fungal infection. An increased risk of malignancy is reported in younger patients. Anaphylaxis is reported. Patients have been reported to develop a lupus-like syndrome, demyelinating disease, worsening heart failure, and bone marrow suppression.
Certolizumab pegol also carries a black box warning regarding an increased risk of severe infection and malignancy in younger patients. Like other medications in this category, bone marrow suppression and liver toxicity are reported.
Etanercept carries a similar black box warning for infection and malignancy. TB and fungal infections are particularly serious. Anaphlaxis, demyelinating disease, worsening heart failure, and bone marrow suppression are reported.
Infliximab again has the same black box warning etanercept. Angioedema, anaphylaxis, worsening heart failure, demyelinating disease, and Stevens-Johnson syndrome are reported.
Rituximab is most often used with methotrexate. Serious and at times fatal reactions occur, most often with the first dose. Patients are generally monitored for some period of time after the first infusion, but they may develop reactions up to 24 hours after the infusion. Patients present with hypotension, angioedema, bronchospasm, and severe cases of ARDS. Stevens-Johnson syndrome and other severe mucocutaneous reactions are reported. Progressive multi-focal leukoencephalopathy and renal failure are reported.
Abatacept is generally given IV or SQ. Patients with COPD may experience an acute exacerbation or develop pneumonia. Headache, nausea, and respiratory infections (and sepsis) are the most common side effects. Anaphylactoid reactions may occur up to 24 hours after administration. Malignancies are associated with treatment, especially in the elderly.
Tofacitinib carries a black box warning for increased risk of serious infections and increased risk of malignancy. Patients are at risk for bradycardia and gastric perforation.
Pregnancy. Most, if not all, of the treatments discussed above are teratogenic and classified as Class X for use in pregnant women. Women who become pregnant while on these medications should be advised to discontinue the drug immediately. There are limited data regarding the risk of pregnancy, but preterm labor, preeclampsia, and fetal growth retardation are reported.31
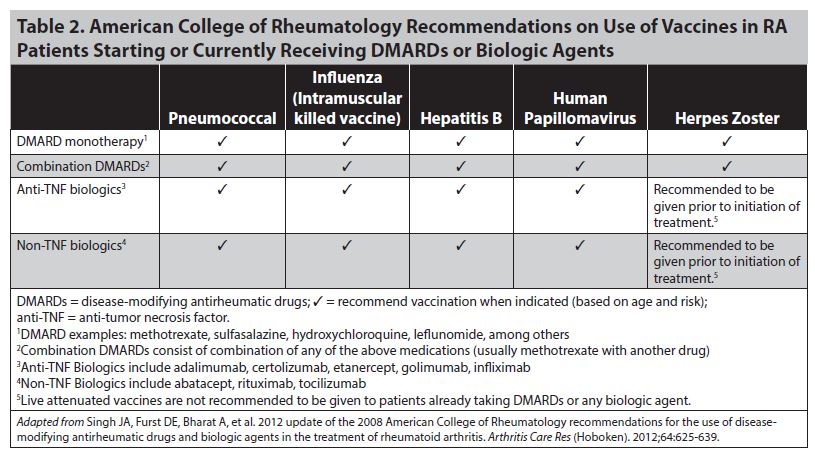
Vaccines. Because of the immunosuppressive effects of these medications, many vaccines are not effective. An important exception is tetanus. More importantly, live virus vaccines such as MMR and zoster should not be given to patients receiving anti-TNF medications. Table 2 gives the most recent recommendations for vaccine administration.
Other Complications
Rheumatoid arthritis is a systemic disease. Rheumatoid nodules can develop on any surface but are most common on extensor surfaces or areas of chronic pressure. They are most common on the fingers, olecranon, proximal ulna, back of the heel, occipital and ischial tuberosity. Carpal tunnel syndrome may be seen due to nerve entrapment. Baker’s cysts are more common than in the general population. Cervical spine abnormalities are particularly problematic in the ED. Atlantoaxial instability or subluxation is the most common abnormality. Patients may also develop superior migration of the odontoid. These abnormalities may cause symptoms of cervical cord compression and make intubation of these patients potentially difficult. Patients may also develop subluxations anywhere along the spine but these are less common.
Because of systemic inflammation, patients are at higher risk for myocardial infarction, interstitial pulmonary fibrosis, and pericarditis. A generalized vasculitis may develop with skin lesions and organ damage. Keratoconjunctivitis is seen.
Many patients will develop an anemia of chronic disease. Some, however, will have a more serious disorder, such as Felty’s syndrome with neutropenia and splenomegaly.
Approach to the Patient
Rheumatoid arthritis should be suspected in any patient presenting with swollen and/or tender joints (i.e., polyarthritis). The history should focus on the number and location of painful and swollen joints, as well as the presence of morning stiffness. Medication history includes the use of analgesics, corticosteroids, or nonsteroidal anti-inflammatory agents and their effects on joint complaints. A personal history for the presence of symptoms compatible with psoriasis, spondyloarthropathies, inflammatory bowel disease, and SLE is important.
The physical examination should focus on the joints to assess for synovitis, joint limitation of motion, or any deformities. A quick search for any skin lesions (psoriatic rashes, malar or discoid rashes), rheumatoid nodules, or gout tophi is included in the examination.
Laboratory testing for ED purposes may include a CBC and possibly a sed rate and CRP. If there is a possible delay in seeking either primary or specialty care, consider drawing complete basic chemistries as well as RF and anti-CCP. Other evaluations that may be considered are hepatitis screen (HBsAg, anti-HBc, anti-HCV) and Tuberculin skin test.
Radiographic imaging of all the major joints can be obtained either in the ED or private office depending on local resources. This will assess for any damage to the joints caused by the inflammation as well as serve as a baseline for disease monitoring.
The most significant modifiable risk factor for development of RA is smoking. If the patient is considering smoking cessation, appropriate referrals are provided.
Conclusion
Rheumatoid arthritis is a polyarticular inflammatory arthritis that demands early diagnosis and treatment for improved patient outcomes.
References
1. Lard LR, Visser H, Speyer I, et al. Early versus delayed treatment in patients with recent-onset rheumatoid arthritis: Comparison of two cohorts who received different treatment strategies. Am J Med 2001; 111:446-451.
2. Cross M, Smith E, Hoy D, et al. The global burden of rheumatoid arthritis: Estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis 2014;73:1323-1330.
3. Silman AJ, Hochberg MC. Epidemiology of the Rheumatic Diseases. 2nd ed. New York: Oxford University Press; 2001.
4. Myasoedova E, Crowson CS, Kremers HM, Therneau TM, Gabriel SE. Is the incidence of rheumatoid arthritis rising? Results from Olmsted County, Minnesota, 1955-2007. Arthritis Rheum 2010;62:
1576-1582.
5. Schur P, Gabriel S, Crowson, C. Epidemiology of, risk factors for, and possible causes of rheumatoid arthritis. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. Accessed March 25, 2014..
6. Helmick CG, Felson DT, Lawrence RC, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum 2008;58:15-25.
7. Holoshitz J. The rheumatoid arthritis HLA-DRB1 shared epitope. Curr Opin Rheumatol 2010;22:293-298.
8. Di Giuseppe D, Discacciati A, Orsini N, Wolk A. Cigarette smoking and risk of rheumatoid arthritis: A dose-response meta-analysis. Arthritis Res Ther 2014;16:R61.
9. Mercado F, Marshall RI, Klestov AC, Bartold PM. Is there a relationship between rheumatoid arthritis and periodontal disease? J Clin Periodontol 2000;27:267-272.
10. Rosenstein ED, Weissmann G, Greenwald RA. Porphyromonas gingivalis, periodontitis and rheumatoid arthritis. Med Hypotheses 2009;73:457-458.
11. Li X, Sundquist J, Sundquist K. Socioeconomic and occupational risk factors for rheumatoid arthritis: A nationwide study based on hospitalizations in Sweden. J Rheumatol 2008;35:986-991.
12. Stolt P, et al. Silica exposure among male current smokers is associated with a high risk of developing ACPA-positive rheumatoid arthritis. Ann Rheum Dis 2010;69:1072-1076.
13. Lee DM, Weinblatt ME. Rheumatoid arthritis. Lancet 2001;358:903-911.
14. Aletaha D, et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 2010;62:
2569-2581.
15. Edwards, MS. Rubella. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. Accessed March 24, 2014.
16. Epidemiology and Prevention of Vaccine-Preventable Diseases. Centers for Disease Control and Prevention, 07 May 2012. Available at: www.cdc.gov/vaccines/pubs/pinkbook/rubella.html. Accessed March 24, 2014.
17. About Parvovirus B19. Centers for Disease Control and Prevention, 14 Feb. 2012. Available at: www.cdc.gov/parvovirusB19/about-parvovirus.html. Accessed March 24, 2014.
18. Colmegna I, Alberts-Grill N. Parvovirus B19: Its role in chronic arthritis. Rheum Dis Clin North Am 2009;35:95-110.
19. Han SH. Extrahepatic manifestations of chronic hepatitis B. Clin Liver Dis 2004;8:403-418.
20. Kemmer NM, Sherman KE. Hepatitis C-related arthropathy: Diagnostic and treatment considerations. J Musculoskelet Med 2010;27:351-354.
21. Olivieri I, Padula A, D’Angelo S, Cutro MS. Psoriatic arthritis sine psoriasis. J Rheumatol Suppl 2009;83:28-29.
22. Nielen MM, van Schaardenburg D, Reesink HW, et al. Specific autoantibodies precede the symptoms of rheumatoid arthritis: A study of serial measurements in blood donors. Arthritis Rheum 2004;50:380-386.
23. Taylor PC, Maini RN. Clinically useful biologic markers in the diagnosis and assessment of outcome in rheumatoid arthritis. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. Accessed on March 31, 2014.
24. Nishimura K, Sugiyama D, Kogata Y, et al. Meta-analysis: Diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann Intern Med 2007;146:
797-808.
25. Dörner T, Egerer K, Feist E, Burmester GR. Rheumatoid factor revisited. Curr Opin Rheumatol 2004;16:246-253.
26. Sun J, Zhang Y, Liu L, Liu G. Diagnostic accuracy of combined tests of anti cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis: A meta-analysis. Clin Exp Rheumatol 2014;32:11-21.
27. Wolfe F, Michaud K. The clinical and research significance of the erythrocyte sedimentation rate. J Rheumatol 1994;21:1227-1237.
28. DMARD. Available at: www.dorlands.com. Accessed April 3, 2014.
29. O’Dell JR, Mikuls TR, Taylor TH, et al. Therapies for active rheumatoid arthritis after methotrexate failure. N Engl J Med 2013;369:307-318.
30. Saag KG, Teng GG, Patkar NM, et al. American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheumatic drugs in rheumatoid arthritis. Arthritis Rheum 2008;59:762-784.
31. Makol A, Wright K, Amin S. Rheumatoid arthritis and pregnancy: Safety considerations in pharmacological management. Drugs 2011;71:1973-1987.
Rheumatoid arthritis (RA) is a systemic polyarticular form of inflammatory arthritis of unclear etiology. It primarily involves the synovial joints and is commonly symmetric. Joint pain and swelling are most commonly seen in patients with this condition. The presence of autoantibodies such as rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPAs), which the anti-cyclic citrullinated peptide (CCP) falls under, are found in the majority of patients. Left untreated, there is a high risk of significant deformity and disability.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
