Maximizing Survival from Out-of-Hospital Cardiac Arrest: Putting Effective Emergency Cardiac Care Into Practice
Maximizing Survival from Out-of-Hospital Cardiac Arrest: Putting Effective Emergency Cardiac Care Into Practice
Authors: Bentley J. Bobrow, MD, FACEP, Assistant Professor of Emergency Medicine, Department of Emergency Medicine, College of Medicine, Mayo Clinic, Scottsdale, AZ; Medical Director, Bureau of Emergency Medical Services and Trauma System, Arizona Department of Health Services, Phoenix, AZ; and Tom P. Aufderheide, MD, FACEP, FAHA, Professor of Emergency Medicine with Tenure, Associate Chair of Research Affairs, Department of Emergency Medicine, Medical College of Wisconsin, Milwaukee.
Peer Reviewer: William J. Brady, MD, Professor of Emergency Medicine and Medicine, Vice Chair of Emergency Medicine, Department of Emergency Medicine, University of Virginia, Charlottesville.
Like many emergency physicians my age, I have studied and performed resuscitation for many years. I learned mouth-to-mouth resuscitation in the Boy Scouts and CPR in medical school. I did chest compressions in the ED and on the wards. I helped with CPR studies in dogs. I've analyzed the outcome of paramedic CPR. I wrote a prehospital cardiac care manual. I've lectured on CPR and taught ACLS courses. And, despite this experience, I can remember only a few patients that I've "saved." Of course, it wasn't only me, but a whole team of professionals doing their best that produced the gratifyingly, albeit rare, neurologically intact outcome from cardiac arrest.
It has been with this background that I have long anticipated new approaches to cardiac arrest resuscitation. A new generation of physicians is reinvigorating the field of cardiac arrest research. I am grateful that two of the experts in this area have written this issue of EM Reports. After reading this, I anticipate you will place these principles into practice.
J. Stephan Stapczynski, MD, Editor
Introduction
Case scenario: Consider a 41-year-old male collapsing in the shower at a public pool immediately after finishing a daily swim workout. Fortunately, an off-duty paramedic sees him collapse and initiates cardiopulmonary resuscitation (CPR). A public access defibrillator is located, and two shocks are administered approximately 5 minutes after collapse. The victim regains return of spontaneous circulation (ROSC) but remains comatose after arrival in the emergency department (ED). In the ED, a 12-lead electrocardiogram (ECG) is diagnostic for an acute myocardial infarction (AMI). The ED initiates therapeutic hypothermia (TH), and the patient is taken to the cardiac catheterization laboratory where a 100% left anterior descending coronary artery occlusion is stented. The patient subsequently receives 24 hours of TH. Following re-warming, he regains consciousness and is without neurological deficit. An implantable cardiac defibrillator (ICD) is inserted by cardiology, and he returns home to his family, job, and pastime of competitive swimming.
This real-life scenario illustrates a common emergency with an uncommon outcome. By understanding the physiology of cardiac arrest, recognizing the hemodynamically significant components of CPR, and delivering optimal emergency cardiac care, healthcare providers can significantly increase the frequency of this outcome throughout our communities. While provocative research in resuscitation is being conducted, this communication focuses on putting current guidelines into practice to save lives.
Out-of-Hospital Cardiac Arrest
Cardiopulmonary arrest claims 350,000 to 450,000 lives per year and accounts for up to 10% of total mortality in the United States.1,2
Despite the scientifically established principle of "the chain of survival," including immediate activation of emergency medical services (EMS), early CPR, early defibrillation, and early advanced life support measures, neurologically intact survival from out-of-hospital cardiac arrest (OHCA) in the United States averages less than 6.4%.3
A 2005 study by Rea, et al. summarized the cardiac arrest experience of 35 communities, representing 9% of the U.S. population. These communities reported survival rates from ventricular fibrillation ranging from 3.3% to 40.5%a 12-fold difference.4
Why do the chances of survival vary so dramatically in different communities?
Variation in survival rates among communities has been attributed to differences in implementing each link in the chain of survival as described by the American Heart Association (AHA).5,6
While system factors (i.e., bystander CPR, availability of AEDs, time to first defibrillation, and EMS response time) remain difficult for individual emergency physicians (EPs) to modulate,7 new understanding of the importance of high-quality CPR, the interaction of CPR with defibrillation, and standardized post-cardiac arrest care has been shown to significantly improve survival.8
There are many questions yet to be answered on the best resuscitation techniques, however recent discoveries have been made in how to improve neurologically intact survival from OHCA. Established treatments for OHCA resuscitation demonstrate improved survival and can be consistently implemented now.
The purpose of this communication is to describe the physiology of CPR, identify interventions demonstrated to increase survival from adult cardiac arrest, and provide EPs with an approach to cardiac arrest that will improve outcomes by optimizing the delivery of emergency cardiac care.
The Physiology of Resuscitation from Ventricular Fibrillation Cardiac Arrest
The Three Phases. One of the many important concepts to come forward since publication of the 2000 American Heart Association Guidelines is the 3-phase, time-dependent concept of cardiac arrest due to ventricular fibrillation described by Weisfeldt and Becker.9 (See Figure 1.) The first phase, termed the electrical phase, lasts approximately 4 minutes. During this phase, the most important intervention is prompt defibrillation. The benefit of using an automated external defibrillator (AED) during this first phase has been shown in a wide variety of settings, including airplanes, airports, casinos, and in the community.10-14
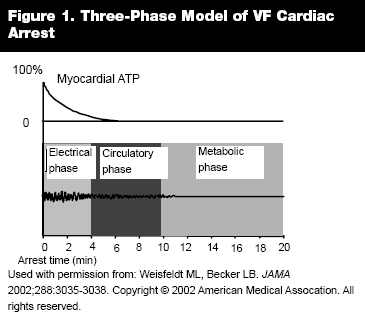 |
The second phase is the circulatory phase, which lasts approximately from minute 4 to10 of the arrest. During this time, generation of adequate cerebral and coronary perfusion pressure is critical to achieving neurologically normal survival. If adequate hemodynamics (through high-quality CPR) are not provided during this phase, the victim is much less likely to survive. The third phase (greater than approximately 10 minutes after circulatory arrest) is the metabolic phase for which current approaches offer little benefit and innovative new concepts are needed. An awareness of these 3 phases helps put into context some of the recent improvements in resuscitation technique.
Cardiopulmonary Resuscitation (CPR): Importance of CPR
The goal of CPR is to optimize blood flow to the vital organs, particularly the heart and brain, to maximize the chance of return of spontaneous circulation (ROSC) and a good neurological outcome. Effective CPR generates blood flow to vital organs until circulation is restored by defibrillation or other therapies. CPR has also been shown to prolong the duration of VF in OHCA victims,15,16 thus expanding the window of opportunity for successful defibrillation.
Bystander CPR. Because early CPR increases the chance of survival from OHCA, the frequency of immediately initiated bystander CPR is critical to improving survival from cardiac arrest throughout a community. Bystander CPR doubles or triples survival rates from OHCA.17-19 Recent evidence also suggests that CPR may be particularly important in cases of prolonged cardiac arrest (i.e., an arrest duration of 4 to 5 minutes without treatment).20,21
The quality of bystander CPR is also related to survival.17,19,22 When Gallagher and colleagues studied the effect of quality bystander CPR (as assessed by trained pre-hospital personnel on arrival at the scene using American Heart Association Guidelines to determine adequate compressions and ventilations), they found that 4.6% of OHCA victims who received effective CPR survived compared to 1.4% of OHCA victims who received inadequate CPR (OR = 3.4; 95% CI, 1.1 to 12.1; P < .02).22
An analysis of 8,091 OHCA victims from 20 different communities in Ontario, Canada, demonstrated that the odds ratio for very good quality of life was 2.0 for patients who received citizen-initiated bystander CPR.23 The same study found that, of the 1-year survivors, 86% were in the best Cerebral Performance Category score class. This study helped dispel the common misperception that survivors from OHCA frequently survive in a chronic vegetative state.23
Holmberg et al analyzed the Swedish Cardiac Arrest Registry between 1990 and 1995. They found that of 9,877 patients, bystander CPR was attempted in 36% and contributed an odds ratio of 2.5 for survival.16
Similar to Europe, bystander CPR rates in the United States are low, and have been implicated as one of the reasons for poor survival. Vadeboncoeur and colleagues found an odds ratio for survival of 2.2 and a 37% overall bystander CPR rate in the state of Arizona between 2004 and 2006. However, when those with medical training as part of their vocation were excluded as rescuers, only 25% of OHCA victims in Arizona received layperson CPR.24
Possible reasons for the low rates of bystander CPR include a fear of performing CPR correctly, fear of causing harm to the victim, panic, reluctance to perform mouth-to-mouth breathing, and fear of transmitted diseases.25-31 The complexity of CPR for non-medical rescuers is another probable factor. Likely, the generally low rates of CPR are due to a combination of these factors.
Adult OHCA survival has been shown to be better when bystanders perform chest compression only CPR compared with no CPR.32 An observational study from the SOS-KANTO Group in Japan looked at 4,068 adult OHCAs, 439 (11%) received chest compression alone CPR, 712 (18%) received conventional CPR; and 2,917 (72%) received no CPR. The study showed that any resuscitation attempt was associated with a higher proportion of victims having favorable neurological outcomes than no resuscitation attempt (5.0% vs 2.2%, p < 0.0001). Additionally, on subgroup analysis, there was no evidence for any survival benefit from the addition of mouth-to-mouth ventilation.
Acknowledging all these factors and the need to increase the prevalence and quality of bystander CPR, the AHA recently changed its guidelines for bystander CPR and issued the advisory statement "Hands-Only (Compression-Only) Cardiopulmonary Resuscitation: A Call to Action for Bystander Response to Adults Who Experience Out-of-Hospital Sudden Cardiac Arrests."33 (See Table 1.) These new guidelines clearly emphasize the significance of chest compressions.
 |
Because of the importance of bystander CPR, EPs should support initiatives in their communities to increase the likelihood of a public trained and motivated to recognize sudden cardiac arrest, activate EMS, initiate high-quality CPR, and use an AED, if available.
Professional CPR. As with bystander CPR, the quality of CPR delivered by trained medical professionals has a significant impact on survival. Medical professionals inconsistently provide high-quality CPR. Wik and colleagues demonstrated this in recently retrained paramedics and nurse anesthetists during 176 adult OHCAs by continuously monitoring all chest compressions and ventilations using modified defibrillators programmed for CPR data collection. The primary outcome measure was adherence to the 2000 International Liaison Committee on Resuscitation Guidelines for CPR including a chest compression rate of 100/minute to 120/minute; depth of 1 ½ to 2 inches (38 to 52 mm); and a ventilation rate of 2 ventilations for every 15 compressions before intubation and 10-12/minute after intubation.
The resuscitation performance measured by the authors was dramatically different from that which is recommended. A mean of 11 (95% CI, 11-12) ventilations were given per minute. The mean compression depth was too shallow at 34 mm (95% CI, 33-35 mm) and only 28% (95% CI, 24%-32%) of the compressions had a depth of 38 mm to 52 mm as recommended in the Guidelines. One of the most important findings was that chest compressions were given only 51% of the available time during resuscitation.34
With a focus on minimizing interruptions to chest compressions and therefore maximizing myocardial and cerebral perfusion, pre-hospital providers in Arizona implemented a protocol termed Cardiocerebral Resuscitation (CCR) or Minimally Interrupted Cardiac Resuscitation (MICR) in 2004. The CCR protocol for pre-hospital providers includes an initial 200 uninterrupted chest compressions at 100 compressions per minute, rhythm analysis with a single shock when indicated, immediately followed by 200 post-shock chest compressions before any pulse check or rhythm reanalysis. Endotracheal intubation is delayed until after three cycles of chest compressions and rhythm analysis. One milligram intravenous epinephrine is administered as soon as possible during the protocol and again with each cycle of chest compressions and rhythm analysis. In a before-and-after analysis of 886 patients in two large metropolitan cities, survival-to-hospital discharge increased from 1.8% (4/218) before CCR training to 5.4% (36/668) after CCR training (odds ratio [OR], 3.0; 95% confidence interval [CI], 1.1-8.9). In the subgroup of 174 patients with witnessed cardiac arrest and ventricular fibrillation, survival increased from 4.7% (2/43) before CCR training to 17.6% (23/131) after CCR training (OR, 8.6; 95% CI, 1.8-42.0).
In an analysis of CCR protocol compliance involving 2,460 patients across the state of Arizona with OHCA, survival was significantly better among patients who received CCR than those who did not (9.1% [60/661] vs 3.8% [69/1799]; OR, 2.7; 95% CI, 1.9-4.1), as well as patients with witnessed ventricular fibrillation (28.4% [40/141] vs 11.9% [46/387]; OR, 3.4; 95% CI, 2.0-5.8). The authors concluded that survival-to-hospital discharge of patients with OHCA in Arizona increased after implementation of CCR as an alternate EMS protocol and that the results need to be confirmed in a randomized trial.35
Although the contribution of each individual component of the CCR protocol is difficult to quantify, minimizing interruptions to chest compressions is very likely an important factor in the results. This finding clearly adds further support to the 2005 AHA Guideline recommendation for medical professionals to minimize interruptions to chest compressions.
Hemodynamically Significant Components of CPR
Chest compressions create cerebral and myocardial blood flow primarily by the thoracic pump mechanism (increasing and decreasing intrathoracic pressure with each compression and decompression of the chest). Specifically, forward blood flow in the cerebral circulation occurs during the chest wall compression phase, while forward blood blow in the coronary circulation occurs during the chest wall decompression or relaxation phase. Adequate chest compressions increase the likelihood of defibrillation success. Sufficient blood flow generated by CPR is especially important if the first shock is delivered in the circulatory phase of VF cardiac arrest (approximately 4-10 minutes after collapse).21
Components of CPR known to affect hemodynamics include ventilation rate and duration, compression rate, compression depth, complete chest recoil, and hands-off time.31
Ventilation. There is an inverse relationship between intrathoracic pressure and coronary perfusion pressure and subsequent survival from cardiac arrest.36,37 Increased ventilation rates and ventilation duration cause increased intrathoracic pressure. Elevated intrathoracic pressure impedes venous blood return to the heart, thereby decreasing forward blood flow to the heart and brain during CPR.36-38 Conversely, generation of negative intrathoracic pressure on the upstroke of CPR augments venous blood return to the heart, significantly improving hemodynamics and outcome.39-42 This fundamental physiologic concept applies to states of shock and defines cardiopulmonary interactions during CPR.43
Aufderheide and colleagues performed a two-part clinical observational study to determine: 1) the incidence of hyperventilation during out-of-hospital CPR with the objective of quantifying the degree of excessive ventilation in humans; and 2) whether comparable excessive ventilation rates during CPR in animals significantly decrease coronary perfusion pressure and survival. The ventilation rates of 13 consecutive OHCA victims were documented to have an average ventilation rate of 30 breaths per minute (twice the 2000 AHA Guidelines-recommended rate). They found that survival in pigs ventilated at 12 breaths per minute (with 100% O2) was 6 of 7 (86%), compared with a survival rate of 1 of 7 (17%) in 2 groups of pigs at a rate of 30 breaths per minute (one group with 100% O2, another with 95% O2 and 5% CO2) (P < 0.001). Thus, pigs with higher mean intrathoracic pressure caused by excessive ventilation rate and duration had significantly lower coronary perfusion pressure and survival rates.
These data revealed that hyperventilation is common even by recently trained professionals. Also evident was the fact that any degree of hyperventilation is likely to have detrimental hemodynamic and survival consequences during low flow states such as CPR. Thus, rescuers should avoid excessive ventilation rates and duration during CPR and, following establishment of an advanced airway, provide a ventilation rate of 8-10 breaths/minute and a breath duration of no greater than 1 second/breath.31 (See Table 2.)
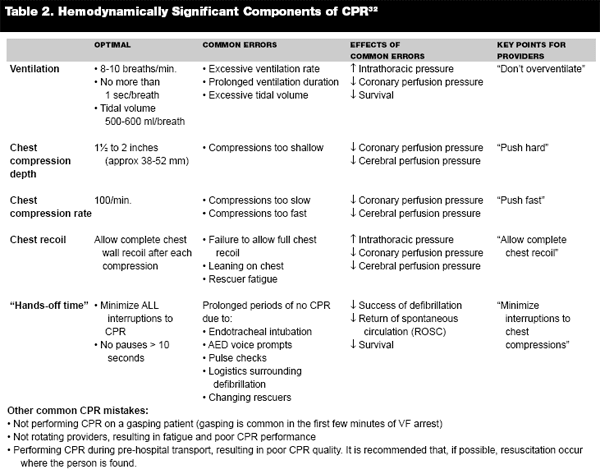 |
Chest Compression Rate. Inadequate rate of chest compressions commonly decreases the quality of CPR delivered during resuscitation. Chest compression rate should not be confused with the number of compressions provided per minute. For example, with a rate of 100/minute, each compression takes 0.6 seconds. With a chest compression to ventilation ratio of 30:2 (assuming 2 breaths delivered at 1 second/breath), 30 compressions (18 seconds) and 2 ventilations (2 seconds) are delivered in 20 seconds or a total of 90 compressions actually delivered/minute. Although it is possible to provide a chest compression rate that is too fast during manual CPR, it is much more common for compressions to be too slow and result in compromised hemodynamics. Therefore, it is crucial to maintain a chest compression rate of at least 100/minute to optimize hemodynamics during CPR. (See Table 2.)
Chest Compression Depth. One of the most common errors compromising hemodynamics during CPR is the rescuer's failure to press deep enough during chest compression. Studies in the out-of-hospital34 and in-hospital settings44 demonstrate that 40% of chest compressions are of insufficient depth.
Current 2005 AHA Guidelines state that the adult chest compression depth should be 1.5 to 2 inches (approximately 38-52 mm).32 (See Table 2.)
Complete Chest Recoil. Another significant factor in generating optimal forward blood flow during CPR is allowing the chest wall to fully recoil. New data have shown that a small vacuum (negative pressure) develops within the thorax each time the chest is allowed to fully recoil, facilitating venous blood return to the heart (preload).40,45 Conversely, when the chest is not allowed to completely recoil, continuous positive pressure occurs within the chest, increasing intrathoracic and intracranial pressure, compromising blood flow to the heart and brain.
In an animal study,45 as little as 1 cm of incomplete chest recoil significantly reduced coronary perfusion pressure. In addition, cerebral blood flow was significantly compromised. Furthermore, it is important to realize that excessive ventilations and incomplete chest recoil work synergistically to further compromise blood flow to the heart and brain compared with what would otherwise occur with just one of these errors in CPR performance.45
Incomplete chest recoil is common in clinical practice. During an observational study of EMS providers, incomplete chest wall recoil was visually observed at some time during resuscitative efforts in 6 of 13 (46%) consecutive adult OHCAs.46 This most often occurred during rescuer fatigue. Rescuers tend to lean on the chest during CPR when fatigued. Doing so increases intrathoracic pressure, inhibiting venous blood return to the heart and compromising hemodynamics. Permitting full release of the chest wall after each compression is essential to maximizing blood flow to the heart and brain and improving outcome from cardiac arrest. (See Table 2.)
"Hands-off Time." The recognition that interruptions to chest compressions are deleterious has led to the concept of "hands-off time" (the percent of time during resuscitation when chest compressions are not being performed). (See Figure 2.) Yu and colleagues studied the relationship between interrupting CPR and defibrillation attempts in a swine model. They found that delays exceeding 15 seconds decreased the success of defibrillation.47
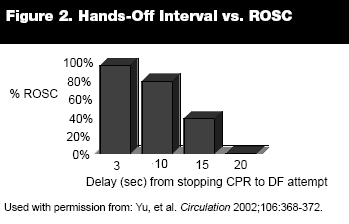 |
Human observational studies showed that professional healthcare providers interrupted chest compressions in 24% to 49% of total arrest time.34,44,48 Reasons for prolonged "hands-off time" include following AED verbal voice prompts, performing intubation, pulse checks, logistics surrounding defibrillation, and transporting patients. An aggressive but attainable goal for minimizing interruptions to chest compressions in clinical practice is 10% "hands-off time."
As recommended in the 2005 AHA Guidelines, a provider should not stop chest compressions for longer than 10 seconds.32 To achieve this goal, providers should be creative (e.g., intubating while chest compressions are being performed or first visualizing the vocal cords, getting ET tube in place, then stopping CPR for 10 seconds while placing ET tube in the trachea, then immediately starting CPR again). Another possible approach to decrease "hands-off time": performing chest compressions while the defibrillator charges. Never check for a pulse longer than 10 seconds. If you don't feel a pulse in 10 seconds, it isn't there. Continue CPR! (See Table 2.)
Impedance Threshold Device. The Impedance Threshold Device (ITD) is a small, 35-mL device that can be attached to any airway [e.g., face mask, Combitube® (Tyco-Kendall, Mansfield, MA), King LT ® (King Systems Corporation, Noblesville, IN), or endotracheal tube]. Each time the chest recoils during CPR, the device transiently blocks air from entering the lungs, creating an enhanced vacuum in the chest. This vacuum, created on the upstroke of CPR, not only facilitates venous blood return to the chest but also decreases intracranial pressure, thereby significantly improving blood flow to both the heart and brain on each subsequent compression. The ITD can be considered a hemodynamically significant component of CPR because its use has been shown to increase blood flow to the heart and brain,49,50 significantly raise blood pressure,42 and improve short-term survival after cardiac arrest.41,51 The first human trial randomized 230 adults after OHCA to receive standard CPR with either a sham or active ITD.41,42 In patients receiving femoral arterial blood pressure monitoring during CPR, systolic blood pressure was significantly increased with the active ITD versus the sham ITD: 85.1 ± 28.9 mmHg versus 42.9 ± 15.1 mmHg, respectively, P < 0.001.41 There were significantly increased ICU admissions in patients presenting in pulseless electrical activity (PEA) with use of active ITD, 19% vs. 52%, (P = 0.02).42 In a large EMS system in Staffordshire, England, survival to emergency department admission was significantly greater among cardiac arrest patients who received the ITD (61/181 [34%]) compared with historical controls (180/808 [22%]) (p < 0.01).51
Collectively these findings demonstrate that the ITD provides a novel means to significantly increase circulation during standard CPR and cardiac arrest. The 2005 American Heart Association Guidelines classified the ITD as a Class IIa device to increase circulation and ROSC.52
Rescuer Fatigue
Rescuer fatigue needs to be managed to maximize hemodynamics during CPR. Fatigue occurs rapidly (within the first two minutes of CPR) and significantly decreases the quality of CPR delivered. Furthermore, rescuers' performance of CPR is not accurately perceived or self-reported. Rescuers commonly overestimate the percentage of adequate compression depth, correct hand placement, and complete chest recoil.46
Ochoa and colleagues showed that for ED and intensive care doctors and nurses trained in CPR, the depth of chest compressions markedly declined after only 1 minute of CPR in 79.7% of cases. They found that this change in quality did not depend on gender, age, weight, height, or rescuer's profession, and it was not adequately perceived by the person who performed the chest compressions.53
In another study, a three-person team of professional rescuers optimized delivery of CPR by performing CPR in rotation at 1-minute intervals. The authors concluded that chest compressions performed for 1 minute, preferably by three rescuers in rotation, maintain a high level of effectiveness. Longer periods of chest compressions by an individual should be discouraged. While rotating rescuers every 1 minute during resuscitation may not be practical, a protocol employing a frequent change in rescuers, when possible, helps prevent fatigue and suboptimal CPR.54 The AHA recommends switching rescuers every 2 minutes to avoid fatigue and its detrimental effect on resuscitation.32
Monitoring the Quality of CPR During Resuscitation
Technology now exists for healthcare providers to receive feedback on the hemodynamically significant components of CPR in real time via devices that measure the rate, depth, and recoil of chest compressions, along with ventilation parameters. Devices such as the Real CPR Help® by ZOLL (ZOLL Medical Corporation, Chelmsford, MA) and QCPR® by Laerdal (Laerdal Medical Corporation, Stavanger, Norway) may contribute to improving the quality of CPR and subsequent hemodynamics.
Mechanical CPR
With clear evidence that the quality of CPR improves outcomes and that the delivery of manual CPR is flawed and inconsistent, the opportunity to provide mechanical CPR has been proposed for several years. There are several commercially available mechanical CPR devices that are currently FDA approved such as the LUCAS ® Chest Compression System (Jolife, Lund, Sweden), and the ZOLL AutoPulse ® (ZOLL Medical Corporation, Chelmsford, MA).
While mechanical CPR devices have been shown to improve short-term survival and hemodynamics in animals and humans,55 results on long-term survival have been mixed.
Results from the multi-center Assisted Prehospital International Resuscitation Research (ASPIRE) trial showed that survival at four hours was not significantly different between OHCA victims who received manual CPR and those receiving CPR with the ZOLL AutoPulse ®a portable device that automates and standardizes chest compressions by way of a load-distributing band (LDB) across the chest (26.4% and 24.7%, respectively). However, survival to hospital discharge was higher among the manual CPR group than the LDB-CPR group (9.9% vs. 5.8%, p = .04). During a planned interim analysis, the data and safety monitoring board found that the AutoPulse group showed worse neurological outcomes prompting them to end study enrollment early. The authors concluded that the worse outcomes in the LDB-CPR group were likely related to device design and implementation strategies.56
A second study, which tracked use of the AutoPulse® by the Richmond (VA) Ambulance Authority in a phased observational evaluation between January 2001 and March 2005, showed different results. It found that AutoPulse® patients showed higher rates of ROSC (34.5% vs. 20.2%), survival to hospital admission (20.9% AutoPulse® vs. 11.1% manual CPR, p = 0.0002), survival to hospital discharge (9.7% AutoPulse® vs. 2.9% manual CPR, p = 0.0001), and had no differences in neurological outcomes.57
An on-going international, multi-center, randomized controlled trial [the Circulation Improving Resuscitation Care (CIRC) Trial], of the AutoPulse® is intended to definitively answer the question of whether AutoPulse® CPR is superior to manual CPR for OHCA (http://www.circtrial.com/studyoverview.html).
To date no mechanical CPR device has consistently been shown to be superior to standard manual CPR for out-of-hospital basic life support. The 2005 AHA Guidelines for Cardiopulmonary Resuscitation give LDB-CPR a Class IIb recommendation.52
Continual Quality Improvement
Continual quality improvement is critical to maximizing the success of out-of-hospital and in-hospital resuscitation programs. Standardized Utstein templates have been created to monitor and facilitate outcome measurements for out-of-hospital and in-hospital cardiac arrest.58-61
In the out-of-hospital setting, EMS directors should consider utilizing equipment capable of electronically monitoring CPR performance, establish a database of this information, and provide system-wide quality assurance feedback on the performance of CPR. (See Table 3.) This can be provided collectively (for the entire system) and individually following review of CPR performance after each cardiac arrest. CPR performance and patient outcome for the system can be monitored. It is also feasible for public health departments to track the incidence of OHCA and implement a system-wide quality assurance program.35
 |
In 2000 the American Heart Association established the National Registry of Cardiopulmonary Resuscitation (NRCPR) to aid participating health care facilities with systematic data collection on resuscitative efforts.62 The purpose of the registry is to develop a well-defined database to document resuscitation performance of hospitals over time. This information can establish the baseline performance of a hospital, target problem areas, and aid in the discovery of opportunities for enhancement in data collection and the resuscitation program in general. The registry is also the largest repository of information on in-hospital cardiopulmonary arrest. Information concerning the NRCPR, is available at its web site: www.nrcpr.org.
The Joint Commission for the Accreditation of Healthcare Organizations (JCAHO) has revised standards for individual in-hospital resuscitation capabilities to include evaluation of resuscitation policies, procedures, processes, protocols, equipment, staff training, and outcome review.63
Defibrillation
Defibrillation involves the administration of current through the chest to the heart to depolarize myocardial cells and abolish VF.
There are two categories of defibrillators (monophasic and biphasic) based on the waveform delivered. Lower-energy biphasic waveform shocks have equivalent or higher success for termination of VF than do monophasic waveform devices, which deliver escalating energy (200 J, 300 J, 360 J).64
EMS Defibrillation. The 2005 AHA Guidelines addressed two important questions regarding the interaction between CPR and defibrillation. The first was whether CPR should be provided prior to attempting defibrillation. Two clinical studies of adult OHCA showed improved survival when patients who had collapsed between 4 to 5 minutes or longer received a period of 90–180 seconds of CPR prior to attempted defibrillation.20,21 The Guidelines state that when the OHCA is not witnessed by EMS personnel (likely representing patients in the circulatory phase of VF arrest), providers may give 5 cycles of CPR prior to rhythm analysis and defibrillation (Class IIb).
The second question addressed the number of shocks to be delivered prior to resuming CPR after defibrillation. An observational study of OHCA revealed that health care providers performed chest compressions only 51% of total CPR time.34 Based in part on this study, the 2005 AHA Guidelines changed from a 3-stacked shock protocol to a 1-shock protocol in an attempt to reduce interruptions to chest compressions.31
Public Access Defibrillation. In addition to defibrillation performed by medical providers, it has been shown that automated external defibrillators (AEDs) are safe and effective when used by trained public-safety personnel who have a duty to respond to medical emergencies.65
The Public Access Defibrillation (PAD) Trial published in 2004 showed that defibrillation performed by trained volunteers (laypersons whose job description did not include the responsibility to respond to medical emergencies) in an organized system of 993 communities in 24 North American regions is safe and can increase the number of survivors after OHCA.65
However, the authors of the PAD trial cautioned against extrapolation of these results and commented that the effectiveness of widespread implementation of public AED programs would likely be moderate due to the fact that approximately 80 percent of arrests occur in the home.65
Home Defibrillation. Results from a study of home AED use were recently published and showed that for survivors of anterior-wall myocardial infarction who were not candidates for implantation of an ICD, access to a home AED did not significantly improve overall survival, as compared with reliance on conventional resuscitation methods.66 Clearly, further research is needed to identify what role AEDs may have in the home.
Post-Cardiac Arrest Care
Post-cardiac arrest syndrome is a distinctive and multifaceted combination of pathophysiological processes including: 1) post-cardiac arrest brain injury; 2) myocardial dysfunction; and 3) ischemia/reperfusion injury. Recent advances in the understanding of these pathophysiological processes have yielded treatments to help improve outcome from OHCA.
During the post-resuscitation phase, cardiac arrest patients are at high risk for multi-system organ failure. While a complete review of post-cardiac arrest care is beyond the scope of this report, some factors believed to impact survival include therapeutic hypothermia, percutaneous coronary intervention (PCI) when indicated, hemodynamic support, close glucose control, appropriate ventilation, as well as consideration of an ICD prior to discharge.
Therapeutic Hypothermia. Therapeutic hypothermia should be part of a standardized treatment strategy for comatose survivors of cardiac arrest.8,67-69 (See Table 4.)
 |
While TH is recommended by the 2005 AHA Guidelines as a Class IIa therapy for adult victims of VF OHCA,69 few patients in the United States currently receive it. A 2005 survey of 265 physicians (including intensivists, EPs, and cardiologists) revealed that 87% had never utilized TH for cardiac arrest patients.70 Reasons cited for non-use include: 49% felt that there were not enough data, 32% mentioned lack of incorporation of hypothermia into advanced cardiovascular life support (ACLS) protocols, and 28% felt that cooling methods were technically too difficult or too slow. Two randomized controlled trials comparing TH with normothermia were published in 2002. Both studies demonstrated improved neurological outcome and survival.71,72 Patients in these studies were cooled to 33ºC or the range of 32ºC to 34ºC for 12 to 24 hours, respectively.
Four studies with historical control groups showed a benefit after TH in comatose survivors of OHCA after non-VF arrest73 and all rhythm arrests.8,74,75 Additional observational studies also indicate possible benefit after cardiac arrest from other initial rhythms and in different settings.76,77
Therapeutic hypothermia is the only therapy given in the post-cardiac arrest setting that has been shown to improve neurological outcome. Numerous techniques are currently available to induce and maintain hypothermia, such as the topical application of ice, cold intravenous fluids, cooling blankets, intravenous cooling catheter, cooling vests, and ice water immersion. Each technique has advantages and disadvantages with regard to induction time, convenience, temperature accuracy, and cost. The ideal induction technique, target temperature, duration, and rewarming strategy have yet to be established. As the optimal method of TH evolves, more patients likely will receive and benefit from TH. More information on TH protocols can be found at: http://www.med.upenn.edu/resuscitation/hypothermia/protocols.shtml.
Percutaneous Coronary Intervention (PCI). Coronary artery disease (CAD) is found in the majority of OHCA patients,78-80 with acute myocardial infarction (AMI) being the most common identifiable cause of sudden cardiac death. It has been shown that ST-elevation and/or chest pain prior to collapse may be insensitive markers of acute coronary occlusion in post-cardiac arrest patients.81 Thus, a protocol advocating early post-cardiac arrest coronary angiography followed by PCI has been shown to be a successful approach after OHCA.81
Since acute coronary occlusion is an identified cause of OHCA, prospective studies are needed to determine if immediate coronary angiography should be carried out in all patients who regain ROSC.
If there are no facilities for immediate PCI, in-hospital thrombolysis is recommended for patients with ST-elevation who have not received pre-hospital thrombolysis.82,83 Coronary artery bypass grafting is indicated in the post-resuscitation phase for patients with left main stenosis or 3-vessel CAD if cardiac arrest was thought to be caused by ischemic heart disease and there is a reasonable chance of neurological recovery.82-85
The combination of early angiography/PCI with TH in 40 comatose patients resuscitated after OHCA caused by ST-segment elevation myocardial infarction (STEMI) resulted in a survival-to-discharge rate of 75% compared with 44% among patients who underwent PCI but not TH.84
It is technically feasible to bring patients to the cardiac catheterization laboratory during the hypothermia process, and the combination of early PCI and TH for patients who remain comatose after cardiac arrest caused by STEMI is an acceptable and promising approach.8,84
Patients resuscitated from OHCA and having ECG criteria for STEMI should undergo urgent coronary angiography with subsequent PCI, if indicated. In addition, given the high prevalence of acute coronary syndrome (ACS) in patients with OHCA and the limitations of ECG-based diagnosis, it is appropriate to consider immediate coronary angiography in all post-cardiac arrest patients in whom ACS is suspected. If PCI is not available, thrombolytic therapy is an appropriate alternative for post-cardiac arrest management of STEMI. Standard guidelines for management of ACS and CAD should be followed.
Hemodynamic Support. Hemodynamic instability commonly occurs after OHCA and manifests as dysrhythmias, hypotension, and low cardiac index. Managing hemodynamics is an important part of the post-cardiac arrest care. The first-line intervention for hypotension after OHCA is to maximize right-heart filling pressures by administration of intravenous fluids. One study showed that 3.5 to 6.5 L of intravenous crystalloid was required in the first 24 hours following ROSC to maintain right atrial pressures in the range of 8 to 13 mmHg. Inotropes and vasopressors should be considered if hypotension persists despite optimized preload.85
Post-Cardiac Arrest Continuum of Care. A smooth continuum of care and on-going collaboration between pre-hospital providers, EPs, cardiologists, and critical care intensivists is necessary to ensure a successful outcome post cardiac arrest. To improve consistency and quality of care, EPs must remain current with evidence-based protocols and coordinate their implementation. Given the complex nature of post-cardiac arrest care, there should be off-line agreements between pre-hospital providers and multidisciplinary hospital teams to develop strategies and clinical pathways to provide patients resuscitated from cardiac arrest optimal hospital-based therapies in a timely fashion. Acknowledging that not all hospitals have the resources to deliver these therapies, employing the regionalization of specialty care strategies analogous to the trauma center model may help assure that as many OHCA victims as possible receive high-quality standardized post-cardiac arrest care. While preliminary evidence suggests it would be safe to incorporate pre-hospital bypass protocol delivering patients to post-cardiac arrest centers, additional investigation into this practice is needed.86
Conclusion
Emergency physicians should implement and promote these proven interventions in their hospitals and throughout their communities. Our expanding insight into resuscitation provides the opportunity for significantly improving neurologically intact survival from cardiac arrest.
References
1. Callans DJ. Out-of-hospital cardiac arrestthe solution is shocking. N Engl J Med 2004;351:632-634.
2. Zheng ZJ, Croft JB, Giles WH, et al. Sudden cardiac death in the United States, 1989 to 1998. Circulation 2001;104:2158-2163.
3. Personal Communication, L. Cobb, Seattle Medic One, December 7, 2005.
4. Rea TD, Eisenberg MS, Sinibaldi G, et al. Incidence of EMS-treated out-of-hospital cardiac arrest in the United States. Resuscitation 2004;63:17-24.
5. Cummins RO, Ornato JP, Thies WH, et al. Improving survival from sudden cardiac arrest: the "chain of survival" concept. A statement for health professionals from the Advanced Cardiac Life Support Subcommittee and the Emergency Cardiac Care Committee, American Heart Association. Circulation 1991;83:1832-1847.
6. Guidelines 2000 for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. International Consensus on Science. Dallas; 2000.
7. De Maio VJ, Stiell IG, Spaite DW, et al. CPR-only survivors of out-of-hospital cardiac arrest: Implications for out-of-hospital care and cardiac arrest research methodology. Ann Emerg Med 2001;37:602-608.
8. Sunde K, Pytte M, Jacobsen D, et al. Implementation of a standardised treatment protocol for post resuscitation care after out-of-hospital cardiac arrest. Resuscitation 2007;73:29-39.
9. Weisfeldt ML, Becker LB. Resuscitation after cardiac arrest: A 3-phase time-sensitive model. JAMA 2002;288:3035-3038.
10. O'Rourke MF, Donaldson E, Geddes JS. An airline cardiac arrest program. Circulation 1997;96:2849-2853.
11. Page RL, Joglar JA, Kowal RC, et al. Use of automated external defibrillators by a U.S. airline. N Engl J Med 2000;343:1210-1216.
12. Caffrey SL, Willoughby PJ, Pepe PE, et al. Public use of automated external defibrillators. N Engl J Med 2002;347:1242-1247.
13. Valenzuela TD, Roe DJ, Nichol G, et al. Outcomes of rapid defibrillation by security officers after cardiac arrest in casinos. N Engl J Med 2000;343: 1206-1209.
14. Hallstrom AP, Ornato JP, Weisfeldt M, et al. Public-access defibrillation and survival after out-of-hospital cardiac arrest. N Engl J Med 2004;351:637-646.
15. Cummins RO, Eisenberg MS, Hallstrom AP, et al. Survival of out-of-hospital cardiac arrest with early initiation of cardiopulmonary resuscitation. Am J Emerg Med 1985;3:114-119.
16. Holmberg M, Holmberg S, Herlitz J. Effect of bystander cardiopulmonary resuscitation in out-of-hospital cardiac arrest patients in Sweden. Resuscitation 2000;47:59-70.
17. Van Hoeyweghen RJ, Bossaert LL, Mullie A, et al. Quality and efficiency of bystander CPR. Belgian Cerebral Resuscitation Study Group. Resuscitation 1993;26:47-52.
18. Abella BS, Aufderheide TP, Eigel B, et al. Reducing barriers for implementation of bystander-initiated cardiopulmonary resuscitation: a scientific statement from the American Heart Association for healthcare providers, policymakers, and community leaders regarding the effectiveness of cardiopulmonary resuscitation. Circulation 2008;117:704-709.
19. Wik L, Steen PA, Bircher NG. Quality of bystander cardiopulmonary resuscitation influences outcome after prehospital cardiac arrest. Resuscitation 1994;28:195-203.
20. Cobb LA, Fahrenbruch CE, Walsh TR, et al. Influence of cardiopulmonary resuscitation prior to defibrillation in patients with out-of-hospital ventricular fibrillation. JAMA 1999;281:1182-1188.
21. Wik L, Hansen TB, Fylling F, et al. Delaying defibrillation to give basic cardiopulmonary resuscitation to patients with out-of-hospital ventricular fibrillation: a randomized trial. JAMA 2003;289:1389-1395.
22. Gallagher EJ, Lombardi G, Gennis P. Effectiveness of bystander cardiopulmonary resuscitation and survival following out-of-hospital cardiac arrest. JAMA 1995;274:1922-1925.
23. Stiell I, Nichol G, Wells G, et al. Health-related quality of life is better for cardiac arrest survivors who received citizen cardiopulmonary resuscitation. Circulation 2003;108:1939-1944.
24. Vadeboncoeur T, Bobrow BJ, Clark L, et al. The Save Hearts in Arizona Registry and Education (SHARE) program: Who is performing CPR and where are they doing it? Resuscitation 2007;75:68-75.
25. Swor R, Khan I, Domeier R, et al. CPR training and CPR performance: do CPR-trained bystanders perform CPR? Acad Emerg Med 2006;13:596-601.
26. Casper K, Murphy G, Weinstein C, et al. A comparison of cardiopulmonary resuscitation rates of strangers versus known bystanders. Prehosp Emerg Care 2003;7:299-302.
27. Locke CJ, Berg RA, Sanders AB, et al. Bystander cardiopulmonary resuscitation. Concerns about mouth-to-mouth contact. Arch Intern Med 1995;155:938-943.
28. Ornato JP, Hallagan LF, McMahan SB, et al. Attitudes of BCLS instructors about mouth-to-mouth resuscitation during the AIDS epidemic. Ann Emerg Med 1990;19:151-156.
29. Brenner BE, Kauffman J. Reluctance of internists and medical nurses to perform mouth-to-mouth resuscitation. Arch Intern Med 1993;153:1763-1769.
30. Brenner B, Stark B, Kauffman J. The reluctance of house staff to perform mouth-to-mouth resuscitation in the inpatient setting: What are the considerations? Resuscitation 1994;28:185-193.
31. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2005;112(24 Suppl):IV1-203.
32. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: Part 4: Adult Basic Life Support. Circulation 2005;112(24 Suppl):IV-19-IV-34.
33. Sayre MR, Berg RA, Cave DM, et al. Hands-only (compression-only) cardiopulmonary resuscitation: A call to action for bystander response to adults who experience out-of-hospital sudden cardiac arrest. A science advisory for the public from the American Heart Association Emergency Cardiovascular Care Committee. Circulation Mar. 31, 2008.
34. Wik L, Kramer-Johansen J, Myklebust H, et al. Quality of cardiopulmonary resuscitation during out-of-hospital cardiac arrest. JAMA 2005;293:299-304.
35. Bobrow BJ, Clark LL, Ewy GA, et al. Minimally interrupted cardiac resuscitation by emergency medical services for out-of-hospital cardiac arrest. JAMA 2008;299:1158-1165.
36. Aufderheide TP, Sigurdsson G, Pirrallo RG, et al. Hyperventilation-induced hypotension during cardiopulmonary resuscitation. Circulation 2004;109: 1960-1965.
37. Aufderheide TP, Lurie KG. Death by hyperventilation: A common and life-threatening problem during cardiopulmonary resuscitation. Crit Care Med 2004;32(9 Suppl):S345-351.
38. Heidenreich JW, Higdon TA, Kern KB, et al. Single-rescuer cardiopulmonary resuscitation: 'Two quick breaths'an oxymoron. Resuscitation 2004;62:283-289.
39. Yannopoulos D, Tang W, Roussos C, et al. Reducing ventilation frequency during cardiopulmonary resuscitation in a porcine model of cardiac arrest. Respir Care 2005;50:628-635.
40. Lurie KG, Zielinski T, McKnite S, et al. Use of an inspiratory impedance valve improves neurologically intact survival in a porcine model of ventricular fibrillation. Circulation 2002;105:124-129.
41. Aufderheide TP, Pirrallo RG, Provo TA, et al. Clinical evaluation of an inspiratory impedance threshold device during standard cardiopulmonary resuscitation in patients with out-of-hospital cardiac arrest. Crit Care Med 2005;33: 734-740.
42. Pirrallo RG, Aufderheide TP, Provo TA, et al. Effect of an inspiratory impedance threshold device on hemodynamics during conventional manual cardiopulmonary resuscitation. Resuscitation 2005;66:13-20.
43. Pepe PE, Raedler C, Lurie KG, et al. Emergency ventilatory management in hemorrhagic states: elemental or detrimental? J Trauma 2003;54:1048-1055; discussion 1055-1047.
44. Abella BS, Alvarado JP, Myklebust H, et al. Quality of cardiopulmonary resuscitation during in-hospital cardiac arrest. JAMA 2005;293:305-310.
45. Yannopoulos D, McKnite S, Aufderheide TP, et al. Effects of incomplete chest wall decompression during cardiopulmonary resuscitation on coronary and cerebral perfusion pressures in a porcine model of cardiac arrest. Resuscitation 2005;64:363-372.
46. Aufderheide TP, Pirrallo RG, Yannopoulos D, et al. Incomplete chest wall decompression: A clinical evaluation of CPR performance by EMS personnel and assessment of alternative manual chest compression-decompression techniques. Resuscitation 2005;64:353-362.
47. Yu T, Weil MH, Tang W, et al. Adverse outcomes of interrupted precordial compression during automated defibrillation. Circulation 2002;106:368-372.
48. Abella BS, Sandbo N, Vassilatos P, et al. Chest compression rates during cardiopulmonary resuscitation are suboptimal: A prospective study during in-hospital cardiac arrest. Circulation 2005;111:428-434.
49. Lurie KG, Voelckel WG, Zielinski T, et al. Improving standard cardiopulmonary resuscitation with an inspiratory impedance threshold valve in a porcine model of cardiac arrest. Anesth Analg 2001;93:649-655.
50. Langhelle A, Stromme T, Sunde K, et al. Inspiratory impedance threshold valve during CPR. Resuscitation 2002;52:39-48.
51. Thayne RC, Thomas DC, Neville JD, et al. Use of an impedance threshold device improves short-term outcomes following out-of-hospital cardiac arrest. Resuscitation 2005;67:103-108.
52. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: Part 6: CPR techniques and devices. Circulation 2005;112(24 Suppl):IV-47-IV-50.
53. Ochoa FJ, Ramalle-Gomara E, Lisa V, et al. The effect of rescuer fatigue on the quality of chest compressions. Resuscitation 1998;37:149-152.
54. Huseyin TS, Matthews AJ, Wills P, et al. Improving the effectiveness of continuous closed chest compressions: An exploratory study. Resuscitation 2002;54:57-62.
55. Timerman S, Cardoso LF, Ramires JA, et al. Improved hemodynamic performance with a novel chest compression device during treatment of in-hospital cardiac arrest. Resuscitation 2004;61:273-280.
56. Hallstrom A, Rea TD, Sayre MR, et al. Manual chest compression vs use of an automated chest compression device during resuscitation following out-of-hospital cardiac arrest: a randomized trial. JAMA 2006;295:2620-2628.
57. Ong ME, Ornato JP, Edwards DP, et al. Use of an automated, load-distributing band chest compression device for out-of-hospital cardiac arrest resuscitation. JAMA 2006;295:2629-2637.
58. Cummins RO. The Utstein style for uniform reporting of data from out-of-hospital cardiac arrest. Ann Emerg Med 1993;22:37-40.
59. Zaritsky A, Nadkarni V, Hazinski MF, et al. Recommended guidelines for uniform reporting of pediatric advanced life support: the pediatric Utstein Style. A statement for healthcare professionals from a task force of the American Academy of Pediatrics, the American Heart Association, and the European Resuscitation Council. Writing Group. Circulation 1995;92: 2006-2020.
60. Cummins RO, Chamberlain D, Hazinski MF, et al. Recommended guidelines for reviewing, reporting, and conducting research on in-hospital resuscitation: The in-hospital 'Utstein style'. American Heart Association. Circulation 1997;95:2213-2239.
61. Jacobs I, Nadkarni V, Bahr J, et al. Cardiac arrest and cardiopulmonary resuscitation outcome reports: Update and simplification of the Utstein templates for resuscitation registries: A statement for healthcare professionals from a task force of the International Liaison Committee on Resuscitation (American Heart Association, European Resuscitation Council, Australian Resuscitation Council, New Zealand Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Councils of Southern Africa). Circulation 2004;110:3385-3397.
62. Peberdy MA, Kaye W, Ornato JP, et al. Cardiopulmonary resuscitation of adults in the hospital: a report of 14720 cardiac arrests from the National Registry of Cardiopulmonary Resuscitation. Resuscitation 2003;58:297-308.
63. In-hospital resuscitation requirements reinstated for hospitals. Jt Comm Perspect 1998;18:5.
64. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: Part 5: Electrical therapies: Automated external defibrillators, defibrillation, cardioversion, and pacing. Circulation 2005;112(24 Suppl):IV-35-IV-46.
65. The Public Access Defibrillation Trial I. Public-Access Defibrillation and Survival after Out-of-Hospital Cardiac Arrest. N Engl J Med 2004;351:637-646.
66. Bardy GH, Lee KL, Mark DB, et al. Home use of automated external defibrillators for sudden cardiac arrest. N Engl J Med 2008;358:1793-1804.
67. Nolan JP, Morley PT, Hoek TL, et al. Therapeutic hypothermia after cardiac arrest. An advisory statement by the Advancement Life Support Task Force of the International Liaison committee on Resuscitation. Resuscitation 2003; 57:231-235.
68. Soar J, Nolan JP. Mild hypothermia for post cardiac arrest syndrome. BMJ 2007;335:459-460.
69. 2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care: Part 7.5: Postresuscitation support. Circulation 2005;112(24 Suppl):IV-84-IV-88.
70. Abella BS, Rhee JW, Huang KN, et al. Induced hypothermia is underused after resuscitation from cardiac arrest: A current practice survey. Resuscitation 2005;64:181-186.
71. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med 2002;346:549-556.
72. Bernard SA, Gray TW, Buist MD, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med 2002;346:557-563.
73. Bernard SA, Jones BM, Horne MK. Clinical trial of induced hypothermia in comatose survivors of out-of-hospital cardiac arrest. Ann Emerg Med 1997; 30:146-153.
74. Oddo M, Schaller MD, Feihl F, et al. From evidence to clinical practice: Effective implementation of therapeutic hypothermia to improve patient outcome after cardiac arrest. Crit Care Med 2006;34:1865-1873.
75. Busch M, Soreide E, Lossius HM, et al. Rapid implementation of therapeutic hypothermia in comatose out-of-hospital cardiac arrest survivors. Acta Anaesthesiol Scand 2006;50:1277-1283.
76. Arrich J. Clinical application of mild therapeutic hypothermia after cardiac arrest. Crit Care Med 2007;35:1041-1047.
77. Holzer M, Mullner M, Sterz F, et al. Efficacy and safety of endovascular cooling after cardiac arrest: Cohort study and Bayesian approach. Stroke 2006;37:1792-1797.
78. Thom T, Haase N, Rosamond W, et al. Heart disease and stroke statistics2006 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2006;113:e85-151.
79. Pell JP, Sirel JM, Marsden AK, et al. Presentation, management, and outcome of out of hospital cardiopulmonary arrest: Comparison by underlying aetiology. Heart 2003;89:839-842.
80. Huikuri HV, Castellanos A, Myerburg RJ. Sudden death due to cardiac arrhythmias. N Engl J Med 2001;345:1473-1482.
81. Spaulding CM, Joly LM, Rosenberg A, et al. Immediate coronary angiography in survivors of out-of-hospital cardiac arrest. N Engl J Med 1997;336: 1629-1633.
82. Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarctionexecutive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to revise the 1999 guidelines for the management of patients with acute myocardial infarction). J Am Coll Cardiol 2004;44:671-719.
83. Richling N, Herkner H, Holzer M, et al. Thrombolytic therapy vs primary percutaneous intervention after ventricular fibrillation cardiac arrest due to acute ST-segment elevation myocardial infarction and its effect on outcome. Am J Emerg Med 2007;25:545-550.
84. Knafelj R, Radsel P, Ploj T, et al. Primary percutaneous coronary intervention and mild induced hypothermia in comatose survivors of ventricular fibrillation with ST-elevation acute myocardial infarction. Resuscitation 2007; 74:227-234.
85. Laurent I, Monchi M, Chiche JD, et al. Reversible myocardial dysfunction in survivors of out-of-hospital cardiac arrest. J Am Coll Cardiol 2002;40: 2110-2116.
86. Bobrow BJ, Vadeboncoeur TF, Spaite DW, et al. The impact of prehospital transport interval on survival in out-of-hospital cardiac arrest: Implications for regionalization of post-resuscitation care. Prehospital Emergency Care 2008;12:95-139.
A new generation of physicians is reinvigorating the field of cardiac arrest research. I am grateful that two of the experts in this area have written this issue of EM Reports. After reading this, I anticipate you will place these principles into practice.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
