Winter Viral Illness in Infants and Children
Winter Viral Illness in Infants and Children
Author: Aleta B. Bonner, DVM, MD, MSPH, Assistant Professor, Emergency Medicine and Pediatrics, Director of Research, Emergency Medicine, Scott and White Hospital, The Texas A& M University System Health Sciences Center College of Medicine, Temple, Texas.
Peer Reviewer: Alfred Sacchetti, MD, Chief, Emergency Services, Our Lady of Lourdes Medical Center, Camden, New Jersey
Emergency department (ED) resources are continually strained due to overcrowding. This intensifies during the winter months because of epidemics of viral illness that affect all ages. The very young and elderly populations are at increased risk for significant morbidity and mortality related to viral disease, and as a consequence have higher rates of hospitalization than other age groups. Due to the overlap in symptoms and examination findings in children with viral and bacterial disease, strategies must be determined that differentiate these infections in the most cost-effective manner. The diagnostic studies required and subsequent treatment options vary widely based upon the type of pathogen suspected and the age of the individual.
The author presents a review of the most commonly encountered winter viral pathogens that focuses on clinically relevant symptoms and physical examination findings, followed by diagnostic testing and treatment options. Additionally, the newly discovered human metapneumovirus (hMPV) is described. The author presents a rational approach to identification of common viral syndromes and their treatment that will prepare the ED physician to confidently assess and treat children during the winter viral season.
The Editor
Introduction
Winter weather causes people to stay indoors for extended periods of time, thus increasing close contact of children and adults. Schools and daycare centers are the perfect environment for spread of both viral and bacterial disease, especially through direct contact and/or aerosol transmission of respiratory secretions. Spread of bacterial pathogens that cause serious infections frequently occur in these settings and result in significant morbidity and mortality, especially in infants and younger children. Fortunately, the epidemiology of childhood illness has changed dramatically during the past two decades due to the advent of immunizations for Hemophilus influenza type B, varicella, and Streptococcus pneumoniae. Since the implementation of these routine immunizations, the rate of serious bacterial infections has decreased four to five fold, thus resulting in less stringent guidelines for the evaluation and treatment of febrile infants.
To provide optimal, cost-effective patient care, a thorough understanding of the epidemiology, diagnosis, and treatment of common winter respiratory and gastrointestinal viruses is crucial for today’s emergency physician.
The Burden of Winter Viral Illness in Children
Viruses that are commonly associated with epidemics of winter respiratory illness in children include parainfluenza (PIV) types 1-3, respiratory syncytial virus (RSV), human metapneumovirus (hMPV), influenza virus types A and B, and adenovirus. The overlapping seasonal occurrence of these viruses is illustrated in Figure 1. All of these viruses cause upper respiratory tract illness, varying degrees of lower respiratory tract illness, and frequently present as a primary febrile illness. In addition, rotavirus is a significant enteric pathogen that is responsible for the majority of viral gastroenteritis occurring in infants and children during the winter months. Table 1 provides a concise overview of these viruses.
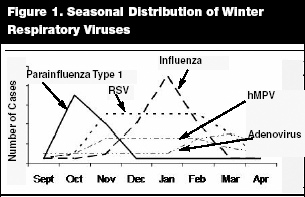
Respiratory viruses are common pathogens identified in children younger than 5 years who are hospitalized for febrile or acute respiratory illness. Respiratory viruses were isolated from 61% of children hospitalized during a prospective one-year study.1 Another surveillance study of children younger than 5 years with influenza-like illness demonstrated a 21-33% influenza positive rate and a 20-41% RSV positive rate during three consecutive winters.2
Two population-based surveillance networks currently monitor pediatric hospitalizations due to influenza. During the 2004-2005 influenza season, influenza-associated pediatric hospitalizations in children 4 years and younger were 3.1-7.0 per 10,000.3 Similar findings have been noted in children younger than 5 years, with rates of influenza-related hospitalization equivalent to those for persons 50-64 years of age.4 Several studies have demonstrated children with high-risk conditions (e.g., cardiopulmonary disease, autoimmune disease, renal disease, or cancer) have significantly higher rates of hospitalization than healthy children of the same age. Excess hospitalization in these groups is highest in children younger than 6 months and sequentially decreases with age.5,6
Winter viral illness constitutes a tremendous burden on outpatient medical care and resources. Neuzil and colleagues documented that for every 100 children younger than 15 years, there was an annual average of 6 to 15 excess physician visits and 3 to 9 courses of antibiotics due to influenza-related illness. Children 6-12 months of age accounted for the highest proportion of the increase in outpatient visits. During the winter months, 10-30% of excess antibiotic prescriptions occurred during times when influenza was present in the community, with the highest numbers of excess prescriptions in children 3-5 (26%) and 5-15 (30%) years of age.5 In addition, high-risk children have a two-fold increase in outpatient visits as compared with healthy children during the presence of influenza in the community.6
Influenza in school-age children had the following effects per 100 study subjects: school absenteeism (63 missed days), days of work missed by parents (20 days), and secondary spread of illness (22 illnesses in family members).7 In another outpatient study, acute otitis media was a complication of influenza in almost 40% of children younger than 3 years. They also reported an annual influenza rate of 179 cases/1000 children, which resulted in an average of 3.2 days of work missed by parents.8
Since October 2004, death associated with influenza infection is a nationally reportable condition in children 18 years and younger. This was prompted by an unusual influenza season in 2003-2004, during which 152 pediatric deaths were attributed to influenza-confirmed illness. During the 2004-2005 influenza season, 36 pediatric deaths related to influenza were reported to the Centers for Disease Control and Prevention (CDC).3
Influenza is associated with three times more deaths than RSV on average when considering infants through the elderly population as a whole. However, morality related to RSV is higher than that of influenza for children younger than 1 year, with approximately 500 RSV-related deaths annually.9 RSV and influenza were responsible for 15-33% of all respiratory-related hospitalizations in children younger than 5 years with chronic lung disease, and antibiotics were administered in 20-24% of these cases.10
Inappropriate antibiotic prescriptions for viral related illness have become a topic of paramount importance for the CDC, primarily due to significant increases in antibiotic resistant pathogens. Acute otitis media (AOM) is the most common indication for antibiotic prescribing, with approximately 5 million children having at least one episode per year. AOM results in approximately 30 million physician visits and more than 10 million antibiotic prescriptions per year.11 Although AOM is considered primarily to be due to bacterial pathogens, bacteria cannot be isolated from approximately 30% of all cases of AOM.12 Viruses and their contribution to AOM are receiving more attention, primarily due to improved isolation techniques.
The American Academy of Pediatrics (AAP) estimates that more judicious use of antibiotics for AOM could result in a reduction of 3 million antibiotic prescriptions per year.11 The CDC has initiated numerous measures to reduce unnecessary antibiotic prescribing as a result of the dramatic increase in antimicrobial resistance. Noteworthy is the "Get Smart: Know When Antibiotics Work" campaign that is targeted at educating parents about appropriate antibiotic use.13
In addition to the above-mentioned respiratory viruses, the enteric pathogen rotavirus is prevalent worldwide and causes significant morbidity and mortality in children younger than 5 years. The prevalence of hospitalization due to rotavirus is difficult to determine due to inaccuracies in the coding of diagnoses; many children with rotavirus are coded as having vomiting, diarrhea, or acute gastroenteritis, and rotavirus is not specified.14 Parashar and colleagues estimated that rotavirus causes approximately 125 million cases of gastroenteritis annually in children younger than 5 years. These result in more than 25 million outpatient visits, 2 million hospitalizations, and approximately 450,000 deaths per year.15
Etiology, Clinical Picture, and Diagnosis
Parainfluenza viruses. Parainfluenza viruses (PIV) are single-stranded RNA viruses and are classified as paramyxoviruses. There are four distinct types: 1-4; however, infection due to type 4 is not frequently identified. Parainfluenza viruses (PIV-1, 2, and 3) are estimated to cause up to one third of all lower respiratory tract infections in infants and pre-school children. These viruses account for significant morbidity, especially in younger infants and preschool-aged children. The maximum estimates of hospitalizations due to these viruses are: 29,000/year for PIV-1, 15,000/year for PIV-2, and 52,000/year for PIV-3.16 The incidence of disease and patterns of infection for the different parainfluenza types are predictable by the ages affected and the season of the year.
Parainfluenza viruses are most well known for producing the clinical syndrome known as croup and account for 50-60% of the viral isolates obtained from these patients. PIV-1 and PIV-2 are responsible for the majority of croup-related hospitalizations in the 2-6 year age group. PIV-1 is associated with epidemics of croup that have occurred predictably during the fall through mid-winter of odd-numbered years since the 1970s. Additional syndromes include bronchiolitis, pneumonia, and tracheobronchitis, all of which are more frequent in infants and are due primarily to infection with PIV-3. PIV-3 occurs sporadically, with peaks of activity during the spring through fall months. In children with the common cold, up to 10-15% of all viral isolates are parainfluenza types 1-3.
Parainfluenza viruses are readily spread through respiratory secretions via person-to-person contact and through contact with infected surfaces. Infected children shed large quantities of virus in their respiratory secretions, thus allowing for extremely efficient spread. Viral shedding is most profuse during the primary initial infection and continues for an average of 4-7 days for PIV-1 and up to 2-3 weeks for PIV-3. The average incubation period for the parainfluenza viruses is 2 to 4 days.17
The typical infection with parainfluenza virus begins with congestion, rhinorrhea, and cough, with subsequent development of AOM in up to one fourth of cases. Fever is present in approximately 75-80% of patients with PIV infection. Laryngotracheobronchitis croup is a common clinical syndrome that usually begins as a febrile upper respiratory infection (URI), with subsequent development of one or more of the following: a "seal-bark" cough, stridor, and/or respiratory distress. Significant stridor due to croup can be quite impressive and may distract from other key findings; especially lower respiratory tract involvement and hypoxemia that is frequently associated with pulmonary involvement. The differential diagnosis for younger pediatric patients with acute onset of stridor includes croup, bacterial tracheitis, foreign body aspiration, and epiglottitis.
The diagnosis of parainfluenza virus infection is typically made based upon the history and physical examination findings. Identification of parainfluenza virus is made by direct fluorescence antibody (DFA) test, viral culture, polymerase chain reaction (PCR) testing, or serology. Rapid, point-of-care testing is not commercially available, thus knowledge of key epidemiologic features such as the patient’s age, season of the year, and presence of the virus in the community are extremely helpful in establishing this diagnosis.
Respiratory syncytial virus. Respiratory syncytial virus (RSV) is an enveloped RNA paramyxovirus. Two subtypes (A and B), as well as multiple strains exist, thus allowing for variation in virulence and infectivity. Almost all children have had at least one episode of infection by age 2 years, and reinfection throughout life is common. RSV is estimated to cause 51,000-82,000 hospitalizations annually, most commonly in infants and children at risk for a more severe illness course (See Table 2).18,19
| Table 2. Risk Factors for Severe RSV Disease |
 |
RSV epidemics occur annually during the winter through early springtime, although sporadic infection may occur throughout the year. Transmission of RSV occurs through respiratory droplets, contact with contaminated surfaces, and by self-inoculation with infected secretions. RSV can remain viable for hours on surfaces and one-half hour or more on the hands. Spread of RSV is prevalent among children in daycare with concurrent spread to household contacts, including adults. Nosocomial infections occur in hospitals and institutional settings such as nursing homes, primarily through transmission by health care personnel. The incubation period averages 4-6 days. Once infected, viral shedding occurs for an average of 3-8 days, but may persist for up to 3-4 weeks in younger infants.20
Symptoms of RSV infection in the very young or preterm infant may be nebulous and consist primarily of fussiness, lethargy, poor feeding, and/or episodes of apnea. In infants younger than 1 year, bronchiolitis and pneumonia are the most common clinical entities. Presenting symptoms may include any of the following: fever, URI, cough, tachypnea, nasal flaring, retractions, paradoxical abdominal breathing, and wheezing. A preliminary study by Attia determined that cough, wheezing, and retractions were independent predictors of RSV infection.21 Conclusive diagnosis of RSV infection may benefit families by predicting the disease course; symptoms generally worsen during the first 3-4 days and then gradually subside. Fortunately, most healthy infants do not require hospitalization and have complete resolution of symptoms within 1-2 weeks.
Significant RSV infection may serve as a predictor for future allergic mediated disease. Infants with RSV bronchiolitisespecially those requiring hospitalizationhave a higher recurrence of wheezing episodes during the following year. It is postulated that this occurs due to increased airway reactivity and/or pre-existing poor lung function.22 A prospective cohort of children hospitalized for bronchiolitis during the first year of life had significantly more allergic symptoms and asthma exacerbations as compared with matched controls at 13 years of age.23
Diagnosis of RSV may be made with rapid diagnostic tests, DFA testing, viral culture, and serology. Rapid testing and DFA testing produce reasonable results, with reported sensitivities in the 80-90% range. Culture of nasopharyngeal secretions is difficult due to the complex methodology required for viral isolation, as well as the labile nature of the virus. PCR testing has been used to detect RSV, but currently is not available commercially.20 Practitioners frequently rely upon the clinical presentation of an infant with bronchiolitis to make a diagnosis of RSV infection.
Human metapneumovirus. hMPV is a recently characterized viral pathogen that has been isolated from respiratory specimens in patients with upper and lower respiratory tract illness. The virus initially was isolated by van den Hoogen and colleagues from stored samples collected during a 20-year period from infants and children with documented respiratory illness and in whom no other viral or bacterial pathogen was previously isolated.24 Since that time, hMPV has been documented on multiple continents and is thought to have a worldwide distribution.25-27
Illness due to hMPV infection resembles that of RSV and probably accounts for a significant number of bronchiolitis cases in which no pathogen was previously isolated.28 RSV seroprevalence occurs more commonly in infants between the ages of 4 and 12 months as compared with hMPV. Seroprevalence of RSV is almost universal in children by the age of 5 years, whereas universal hMPV antibody detection does not occur until an average of 5-10 years of age. This indicates a predilection for occurrence of primary infection with hMPV at an older average age than does primary infection with RSV.29
Symptoms related to hMPV infection overlap with those of RSV and influenza. Fever is significantly more common in children with documented hMPV and influenza infections, whereas wheezing, bronchiolitis, or asthma exacerbation is more common in children with hMPV and RSV infections. There is a significant increase in respiratory illness within household contacts of children with documented hMPV and influenza infections. These children and their affected household contacts also require more medical attention and miss significantly more school and work days than those with RSV.30
Multiple studies have evaluated the incidence of dual infection with RSV and hMPV in children hospitalized due to bronchiolitis. Two of these studies documented co-infection with RSV and hMPV in 60-70% of children younger than 3 years with severe bronchiolitis who required intensive care support.31,32 Another study found that duel infection with hMPV and RSV in children younger than 2 years with bronchiolitis increased the relative risk (RR) of admission to the intensive care unit for mechanical ventilation by ten fold.33 A study by Lazer tested 23 children with severe RSV and 23 children with mild RSV for hMPV. None of these patient samples were positive for hMPV, which is in contrast to the studies noted above.34 Currently, isolation of hMPV requires PCR techniques that are performed only in specialized laboratories.
Influenza. Influenza viruses are orthomyxoviruses that are divided into three types (A, B, and C) based upon their unique antigenic characteristics. Epidemics of disease are due to infection with influenza types A and B, which typically have peak activity between December and March. Influenza A is subclassified on the basis of hemagglutinin (HA) and neuraminidase (NA) surface antigens. Antibodies produced against these antigens, especially HA, are important in providing immunity to these viruses. Major changes between HA subtypes or emergence of a new HA or NA subtype is called an antigenic shift. This is currently illustrated by the crossover and transmission of avian influenza to humans. Minor variations within the subtype are called antigenic drift and account for minimal variations in the viruses from year to year. Influenza B is not divided into subtypes, and changes in the virus occur more slowly, usually over several years.
Influenza is efficiently transmitted through respiratory droplets or via contact with contaminated surfaces. Epidemic activity within the community generally lasts 4-8 weeks. Presence of multiple influenza strains during a community outbreak may lead to a prolonged influenza season of three months duration or more. Influenza affects all ages, with the most significant morbidity and mortality occurring in the very young and the elderly. School-age children have the highest attack rates, often up to 40%.38 Lower respiratory tract involvement including croup, bronchiolitis, and pneumonia occurs in up to 25% of children.35 The incubation period for influenza is 1-3 days; patients are most contagious from 24 hours prior to onset of symptoms through the most symptomatic period of illness.
Symptoms and findings associated with influenza infection frequently overlap with those of parainfluenza, RSV, and hMPV infection. Influenza classically produces sudden development of fever, chills, cough, headache, malaise, and myalgias. Respiratory symptoms become more prominent and include sore throat, nasal congestion, rhinorrhea, and worsening cough. Nausea, vomiting, and abdominal pain may be associated with influenza, particularly in younger children. Other clinical syndromes attributed to influenza infection in children include isolated acute febrile illness, a sepsis-like syndrome, croup, bronchiolitis, and pneumonia. Infection with influenza B has been associated with Reye syndrome and acute myositis.
Diagnosis is frequently made based upon history, clinical findings, and the knowledge that influenza is present within the community. Rapid diagnostic tests that can be performed in the point-of-care environment may be useful in providing a definitive diagnosis of influenza and also differentiate types A and B. These tests provide results within 10-15 minutes and are most useful when used within the first 48-72 hours of illness. During this time, viral shedding is significantly higher, and confirmation of influenza infection may be used to guide antiviral therapy. Rapid testing also may be used as an initial test to guide inpatient cohorting for infection control measures. Other methods of influenza detection include viral culture, DFA testing, and PCR testing.
Adenoviruses. Adenoviruses are double-stranded DNA viruses that have at least 51 different serotypes and cause primarily respiratory and gastrointestinal illness. All ages may be affected, but the highest incidence of infection is in children 6 months to 5 years of age.36 Participation in daycare increases the risk of developing infection. Adenoviral respiratory infections may occur year-round, but usually peak in late winter to early spring.
Transmission through respiratory spread is by aerosol, person-to-person contact, and via contact with surfaces contaminated with the virus. Acute gastroenteritis associated with adenovirus occurs year-round and does not demonstrate a peak period of activity. Spread of enteric adenovirus infection is by the fecal-oral route and occurs most frequently in children younger than 4 years. The incubation period for respiratory infection is 2-14 days and for gastrointestinal infection is 3-10 days.36
The spectrum of illness caused by adenovirus is the most diverse of the winter viruses. Although the primary site of infection is the upper respiratory tract, lower tract infection, conjunctivitis, nephritis, cystitis, hepatitis, and gastroenteritis may occur.36 Neurologic syndromes associated with adenovirus infection include aseptic meningitis, encephalitis, myelitis, and a Reye-like syndrome. Recent reports also attributed a syndrome of transient encephalopathy to adenovirus.37 The disease presentation of children with adenoviral infection may mimic Kawasaki Disease; both may have fever, conjunctivitis, mucocutaneous involvement, and an erythematous papular rash. Rocholl and colleagues detected adenovirus from 5 of 5 children who were hospitalized due to suspected Kawasaki Disease.38 Infants and immunocompromised patients are at increased risk for disseminated infection, which most frequently manifests as encephalitis, pneumonia, fulminant hepatitis, hemorrhagic cystitis, or gastroenteritis.39
Diagnosis of adenovirus infection is frequently made based upon clinical findings and clinical suspicion because there is no rapid diagnostic test for adenovirus. Laboratory confirmation of adenovirus includes viral culture, DFA testing, and PCR testing. Samples for viral isolation should be taken from the affected site(s): nasopharynx, lower respiratory tract, conjunctiva, or stool. Adenovirus is not cultured easily from the stool, thus antigen detection or direct visualization by electron microscopy is preferred. Adenovirus antigens also can be detected in bodily fluids by immunoassay techniques.16
Rotaviruses. Rotaviruses are double-stranded RNA viruses belonging to the Reoviridae family. They are the major cause of diarrheal illness worldwide; although they affect all ages, the highest prevalence is in children 4-24 months of age. Transmission is through fecal-oral spread or contact with contaminated surfaces and commonly occurs in daycare centers. The incubation period is 4-8 days, and symptoms usually persist for 3-8 days.40 Infection results in copious nonbloody diarrhea, often preceded by fever and vomiting.
A tentative diagnosis of rotavirus may be made on clinical grounds and the presence of the virus in the community. Stool samples usually will contain mucus but not blood or white blood cells. Definitive diagnosis is made in the laboratory by enzyme immunoassay or latex agglutination tests.
Management of Specific Clinical Disease Entities
Acute Otitis Media (AOM). Respiratory viruses frequently are isolated from the middle ear fluid of patients with AOM. McCormick and colleagues demonstrated that fullness/bulging of the tympanic membrane (TM) was the most important predictor for bacterial or bacterial/viral co-infection in children with AOM. Conversely, erythema of the TM in the absence of fullness/bulging was more likely to be predictive for either a negative culture or a virus as the sole isolate.41 A recent study indicated that children with nonsevere AOM can be managed with symptomatic care alone (e.g., analgesic administration/watchful waiting), and that 66% will have complete resolution of the AOM without requiring antibiotic therapy; parents of these children were as satisfied with their child’s care as the parents whose children received antibiotics at the initial visit.42 Implementation of these findings into daily practice could significantly reduce unnecessary antibiotic prescriptions for viral-related illness.
Croup. Laryngotracheobronchitis (croup) is a common respiratory syndrome primarily caused by infection with parainfluenza virus (50-60%), with the remainder of cases due to influenza, RSV, human metapneumovirus, adenovirus, and rhinovirus. During documented epidemics, influenza A may cause up to 65% of croup cases. The clinical course of children hospitalized with croup due to influenza is generally more severe than that of children with parainfluenza, as measured by length of hospitalization, readmission rate and use of pharmacologic therapy. Additionally, children with influenza-related croup have higher temperatures and were diagnosed with AOM and pneumonia more frequently than children with parainfluenza infection.43 This becomes an important factor in patient management because patients with influenza-related croup might derive additional benefit from antiviral therapy.
Patients with croup have varying degrees of respiratory tract involvement, with the most significant inflammation and associated swelling occurring in the subglottic tissues. This results in a reduced diameter of the subglottic airway and accounts for production of the classic "seal bark" cough associated with croup. As the diameter of the airway narrows, airflow becomes more difficult and ultimately results in stridor and increased work of breathing (e.g., tachypnea, nasal flaring and retractions, and paradoxical "abdominal" breathing). Stridor with activity that subsides at rest is common in patients with mild croup. Moderate to severe cases of croup produce stridor even while at rest. Severe obstruction of the subglottic area may lead to respiratory failure and subsequent respiratory arrest.
Initial medical management of croup includes evaluation and correction of airway, breathing and/or circulatory abnormalities (ABCs). For patients with moderate to severe croup, obtain a baseline pulse oximetry measurement. If hypoxemia is present, treat with oxygen by facemask or nasal cannula, and monitor with continuous pulse oximetry. A randomized, controlled trial of aerosolized mist versus placebo in patients with moderate croup presenting to an ED revealed no significant difference in improvement of croup scores between the two groups.44 Nebulized epinephrine and administration of steroids are beneficial in the treatment of moderate to severe croup.45-47 More recently, oral dexamethasone has been proven to benefit patients who seek care for cases of mild croup.48 Recent work by Geelhoed demonstrated that co-administration of nebulized budesonide and oral dexamethasone did not provide additional benefit over oral dexamethasone alone in the treatment of croup.49 A summary of the medications used to treat croup is provided in Table 3.
| Table 3. Medications Used for the Treatment of Croup |
 |
Bronchiolitis. Bronchiolitis encompasses a spectrum of illness ranging from fairly mild symptoms to respiratory failure requiring mechanical ventilation. Symptoms are produced when viral infection of the lower respiratory tract results in inflammation and edema within the terminal bronchioles and alveoli. The resultant lower airways obstruction frequently causes cough, wheezing, and respiratory distress, which make it difficult to distinguish from asthma. Mucus plugging of the airways, in addition to primary pulmonary involvement may lead to inadequate oxygen exchange and the development of hypoxemia.
Infants with respiratory distress often have difficulty maintaining adequate oral intake and as a result become dehydrated. Common indications for hospitalization include dehydration requiring intravenous fluid therapy and/or hypoxemia requiring supplemental oxygen. Pulse oximetry readings and the perceived need for oxygen therapy appear to influence physicians’ decision making regarding admission of infants with bronchiolitis, while physician-risk attitudes and tolerance for diagnostic uncertainty are of no influence.50-52
Medical management of bronchiolitis with bronchodilators is commonplace, although their efficacy is limited to modest short-term improvement in patients with mild to moderate symptoms. However, these studies were underpowered to detect important measures such as hospital admission rates or length of stay.53-56 A large randomized study of infants hospitalized with bronchiolitis demonstrated no reduction in the length of stay for those receiving nebulized epinephrine versus placebo. In fact, infants requiring supplemental oxygen and intravenous fluids who received epinephrine actually had significantly longer hospitalizations as compared with the placebo group.57
Steroids also are used commonly in the treatment of bronchiolitis, although their use is controversial. The majority of evidence does not demonstrate clinical benefit, therefore their routine use in previously healthy infants and children is not recommended.55,58 Schuh and colleagues demonstrated benefit of a single 1-mg/kg oral dose of dexamethasone in children younger than 24 months with bronchiolitis who were treated in the ED.59 However, 58% of their study patients who received dexamethasone were febrile at enrollment. Fever reduction in the dexamethasone group could have decreased metabolic demands and minute ventilation, thus leading to a decrease in respiratory rate and retractions. In this case, the noted improvement would be due to the antipyretic effect and not the anti-inflammatory effect of dexamethasone. Until this question is answered, widespread use of dexamethasone in patients with bronchiolitis should not be done on the basis of this study.60 Another outpatient study evaluated dexamethasone use in conjunction with bronchodilator therapy. There were no differences between the groups until day 5, when the Respiratory Distress Assessment Instrument (RADI) scores were statisticallyalthough not clinicallysignificant between the dexamethasone and placebo treated groups.61 Other authors have demonstrated benefit when steroids were administered to bronchiolitis patients who were premature or required mechanical ventilation.62,63 Infants with pre-existing chronic lung disease often have airway hyper-reactivity that responds to treatment with steroids, therefore, this finding is not surprising and actually supports use in this subset of patients. Although the current best evidence does not support widespread use of bronchodilators or steroids for bronchiolitis, significant practice variation continues to exist in the treatment of bronchiolitis.64-66
Influenza. Influenza commonly produces an acute febrile illness associated with respiratory symptoms. Clinical predictors of influenza in preschool and older children include cough, headache, and pharyngitis.67 In addition to clinical findings, rapid diagnostic tests that can be performed at the point of care can be used to assist practitioners when the diagnosis is in doubt. Commercially available rapid influenza tests are summarized in Table 4. Several studies have documented physician decision making was influenced by results of the rapid influenza test.74-76 Antiviral medications effective for the treatment of influenza must be started within 2 days of the onset of symptoms. Table 5 summarizes use of these medications.
| Table 5. Antiviral Agents for the Treatment of Influenza in Children |
 |
Acute febrile illness is one of the most common problems in children younger than 2 years. All of the viruses described herein, as well as serious bacterial infections (SBIs) may have fever as the predominant finding. Recent studies indicate that the incidence of serious bacterial illness is significantly reduced when a documented viral infection is present. Infants who were classified as high risk by the Rochester criteria but had documented viral infection were noted to have SBI at rates comparable with low-risk infants.71 A study of infants and children 0-36 months of age with documented influenza demonstrated presence of bacteremia in 0.6%, urinary tract infection (UTI) in 1.8%, and pneumonia in 25.4%; versus the influenza negative group with bacteremia in 4.2%, UTI in 9.9%, and pneumonia in 41.9% of the subjects.75 Other studies have evaluated the incidence of SBI in infants with documented RSV infection. The incidence of bacteremia ranged from 0 to 1.1% and UTI from 1.1% to 5.7%, both significantly less than for the RSV negative infants.73--76
These studies indicated that a confirmed viral diagnosis decreases the risk of SBI and may be advantageous in tailoring the diagnostic workup in infants older than 1 month. However, there are no provisions within the current practice guidelines for infants with fever and no source as far as testing and treatment for viral illness, especially in infants younger than 1 month.
Dehydration secondary to acute gastroenteritis is a common problem especially during winter outbreaks of rotavirus infection. A meta-analysis of oral versus intravenous rehydration therapy clearly demonstrates that oral rehydration is as effective as intravenous rehydration. Enteral rehydration involving oral or nasogastric methods has fewer significant adverse events and shorter hospital length of stay than patients receiving intravenous rehydration.77 Arguments against using enteral rehydration in the ED primarily center on a perceived increase in the length of time required to perform oral rehydration verses intravenous rehydration. The AAP recommends oral rehydration over a period of 4 hours or less. A prospective case series of 549 children in an urban pediatric ED revealed an average treatment time of 5.4 hours for patients receiving intravenous rehydration. This study served to eliminate a perceived barrier against the routine use of oral rehydration.78
Conclusion
Viral illnesses continue to challenge EDs and the physicians who care for the patients. A comprehensive understanding of common viruses, diagnoses, and management improves ED care and may help facilitate flow in the department.
| Table 6. Internet Resources for Winter Viral Infections |
 |
References
1. Iwane MK, Edwards KM, Szilagyi PG et al. Population-based surveillance for hospitalizations associated with respiratory syncytial virus, influenza virus, and parainfluenza viruses among young children. Pediatrics 2004;113:1758-1164.
2. Zambon MC, Stockton JD, Clewley JP, et al. Contribution of influenza and respiratory syncytial virus to community cases of influenza-like illness: An observational study. Lancet 2001;358:1410-1416.
3. Update: Influenza activityUnited States and worldwide, 2004-05 season. MMWR Morb Mortal Wkly Rep 2005 July 1;54:631-634.
4. Thompson WW, Shay DK, Weintraub E et al. Influenza-associated hospitalizations in the United States. JAMA 2004 ;292:1333-1340.
5. Neuzil KM, Mellen BG, Wright PF, et al. The effect of influenza on hospitalizations, outpatient visits, and courses of antibiotics in children. N Engl J Med 2000;342:225-231.
6. O’Brien MA, Uyeki TM, Shay DK, et al. Incidence of outpatient visits and hospitalizations related to influenza in infants and young children. Pediatrics 2004;113:585-593.
7. Neuzil KM, Hohlbein C, Zhu Y. Illness among schoolchildren during influenza season: Effect on school absenteeism, parental absenteeism from work, and secondary illness in families. Arch Pediatr Adolesc Med 2002;156:986-991.
8. Heikkinen T, Silvennoinen H, Peltola V, et al. Burden of influenza in children in the community. J Infect Dis 2004 ;190:1369-1373.
9. Thompson WW, Shay DK, Weintraub E, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA 2003;289:179-186.
10. Griffin MR, Coffey CS, Neuzil KM, et al. Winter viruses: Influenza- and respiratory syncytial virus-related morbidity in chronic lung disease. Arch Intern Med 2002;162:1229-1236.
11. Amercican Academy of Pediatrics and American Academy of Family Physicians. Questions and Answers on Acute Otitis Media. American Academy of Pediatrics Web site;Available at: http://aap.org/advocacy/releases/aomqa.htm. Accessed: October 10, 2005.
12. Heikkinen T, Chonmaitree T. Importance of respiratory viruses in acute otitis media. Clin Microbiol Rev 2003;16:230-241.
13. Department of Health and Human Services Centers for Disease Control and Prevention. Snort. Sniffle. Sneeze. No Antibiotics Please! Get Smart: Know When Antibiotics Work. Available at: http://www.cdc.gov/drugresistance/community/snortsnifflesneezespot. Accessed: October 15, 2005.
14. Hsu V, Staat MA, Roberts N, et al. Use of active surveillance to validate international classification of diseases code estimates of rotavirus hospitalizations in children. Pediatrics 2005;115:78-82.
15. Parashar U, Hummelman EG, Bresee JS, et al. Global illness and deaths caused by rotavirus disease in children. Emerg Infect Dis 2003;9:565-572.
16. Counihan ME, Shay DK, Holman RC, et al. Human parainfluenza virus-associated hospitalizations among children less than five years of age in the United States. Pediatr Infect Dis J 2001;20:646-653.
17. Feigin RD, Cherry JD, Demmler GJ, et al. Parainfluenza Viruses. Textbook of Pediatric Infectious Disease. 5th ed. Philadelphia,PA: Saunders; 2004;2270-2283.
18. Centers of Disease Control and Prevention. Respiratory syncytial virus activityUnited States, 2003-2004. MMWR Morb Mortal Wkly Rep 2004;53:1159-1160.
19. Welliver RC. Review of epidemiology and clinical risk factors for severe respiratory syncytial virus RSV. infection. J Pediatr 2003; 143:S112-S117.
20. Respiratory syncytial virus. In: Pickering L, ed. Red Book: 2003 Report of the Committee on Infectious Diseases. American Academy of Pediatrics; 2003;523-528.
21. Attia MW, Friedman MJ. Clinical predictors of respiratory syncytial virus (RSV) in children. Acad Emerg Med 2003;10:440.
22. Cifuentes L, Caussade S, Villagran C, et al. Risk factors for recurrent wheezing following acute bronchiolitis: A 12-month follow-up. Pediatr Pulmonol 2003;36:316-321.
23. Sigurs N, Gustafsson PM, Bjarnason R, et al. Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am J Respir Crit Care Med 2005;171:137-141.
24. van den Hoogen BG, de Jong JC, Groen J, et al. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat Med 2001;7 6.:719-724.
25. Esper F, Boucher D, Weibel C, et al. Human metapneumovirus infection in the United States: Clinical manifestations associated with a newly emerging respiratory infection in children. Pediatrics 2003;111:1407-1410.
26. Williams JV, Harris PA, Tollefson SJ, et al. Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N Engl J Med 2004;350:443-450.
27. Noyola DE, puche-Solis AG, Herrera-Diaz A, et al. Human metapneumovirus infections in Mexico: Epidemiological and clinical characteristics. J Med Microbiol 2005;54:969-974.
28. Mullins JA, Erdman DD, Weinberg GA, et al. Human metapneumovirus infection among children hospitalized with acute respiratory illness. Emerg Infect Dis 2004;10:700-705.
29. Ebihara T, Endo R, Kikuta H, et al. Comparison of the seroprevalence of human metapneumovirus and human respiratory syncytial virus. J Med Virol 2004;72:304-306.
30. Bosis S, Esposito S, Niesters HG, et al. Impact of human metapneumovirus in childhood: Comparison with respiratory syncytial virus and influenza viruses. J Med Virol 2005;75:101-104.
31. Greensill J, McNamara PS, Dove W, et al. Human metapneumovirus in severe respiratory syncytial virus bronchiolitis. Emerg Infect Dis 2003;9:372-375.
32. Konig B, Konig W, Arnold R, et al. Prospective study of human metapneumovirus infection in children less than 3 years of age. J Clin Microbiol 2004;42:4632-4635.
33. Semple MG, Cowell A, Dove W, et al. Dual infection of infants by human metapneumovirus and human respiratory syncytial virus is strongly associated with severe bronchiolitis. J Infect Dis 2005;191: 382-386.
34. Lazar I, Weibel C, Dziura J, et al. Human metapneumovirus and severity of respiratory syncytial virus disease. Emerg Infect Dis 2004;10:1318-1320.
35. American Academy of Pediatrics. Influenza. In: Pickering LK, ed. Red Book: 2003 Report of the Committee on Infectious Diseases. 26th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2003:382-391.
36. American Academy of Pediatrics. Adenovirus infections. In: Pickering LK, ed. Red Book: 2003 Report of the Committee on Infectious Diseases. 26th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2003;190-192.
37. Straussberg R, Harel L, Levy Y, Amir J. A syndrome of transient encephalopathy associated with adenovirus infection. Pediatrics 2001;107:E69.
38. Rocholl C, Gerber K, Daly J, et al. Adenoviral infections in children: The impact of rapid diagnosis. Pediatrics 2004;113:e51-e56.
39. Krilov LR. Adenovirus infections in the immunocompromised host. Pediatr Infect Dis J 2005;24:555-556.
40. American Academy of Pediatrics. Rotavirus infections. In: Pickering LK, ed. Red Book: 2003 Report of the Committee on Infectious Diseases. 26th ed. Elk Grove Village, IL: American Academy of Pediatrics;2003:534-536.
41. McCormick DP, Lim-Melia E, Saeed K, et al. Otitis media: Can clinical findings predict bacterial or viral etiology? Pediatr Infect Dis J 2000;19:256-258.
42. McCormick DP, Chonmaitree T, Pittman C, et al. Nonsevere acute otitis media: A clinical trial comparing outcomes of watchful waiting versus immediate antibiotic treatment. Pediatrics 2005;115:1455-1465.
43. Peltola V, Heikkinen T, Ruuskanen O. Clinical courses of croup caused by influenza and parainfluenza viruses. Pediatr Infect Dis J 2002;2:76-78.
44. Neto GM, Kentab O, Klassen TP, et al. A randomized controlled trial of mist in the acute treatment of moderate croup. Acad Emerg Med 2002;9:873-879.
45. Donaldson D, Poleski D, Knipple E, et al. Intramuscular versus oral dexamethasone for the treatment of moderate-to-severe croup: A randomized, double-blind trial. Acad Emerg Med 2003;10:16-21.
46. Russell K, Wiebe N, Saenz A, et al. Glucocorticoids for croup. Cochrane Database Syst Rev 2004;1.
47. Waisman Y, Klein BL, Boenning DA, et al. Prospective randomized double-blind study comparing L-epinephrine and racemic epinephrine aerosols in the treatment of laryngotracheitis croup. Pediatrics 1992;89:302-306.
48. Bjornson CL, Klassen TP, Williamson J, et al. A randomized trial of a single dose of oral dexamethasone for mild croup. N Engl J Med 2004;351:1306-1313.
49. Geelhoed GC. Budesonide offers no advantage when added to oral dexamethasone in the treatment of croup. Pediatr Emerg Care 2005;21:359-362.
50. Baldwin RL, Green JW, Shaw JL, et al. Physician risk attitudes and hospitalization of infants with bronchiolitis. Acad Emerg Med 2005;12:142-146.
51. Mallory MD, Shay DK, Garrett J, et al. Bronchiolitis management preferences and the influence of pulse oximetry and respiratory rate on the decision to admit. Pediatrics 2003;111:e45-e51.
52. Schroeder AR, Marmor AK, Pantell RH, et al. Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med 2004;158:527-530.
53. Agency for Healthcare Research and Quality. Management of Bronchiolitis in Infants and Children. 2003. Report No. 69.
54. Kellner JD, Ohlsson A, Gadomski AM, et al. Bronchodilators for bronchiolitits. Cochrane Database Syst Rev 1999; 1.
55. King VJ, Viswanathan M, Bordley WC, et al. Pharmacologic treatment of bronchiolitis in infants and children: A systematic review. Arch Pediatr Adolesc Med 2004;8:127-137.
56. Mull CC, Scarfone RJ, Ferri LR, et al. A randomized trial of nebulized epinephrine vs albuterol in the emergency department treatment of bronchiolitis. Arch Pediatr Adolesc Med 2004;158:113-118.
57. Wainwright C, Altamirano L, Cheney M, et al. A multicenter, randomized, double-blind, controlled trial of nebulized epinephrine in infants with acute bronchiolitis. N Engl J Med 2003;349:27-35.
58. Patel H, Platt R, Lozano JM, et al. Glucocorticoids for acute viral bronchiolitis in infants and young children. Cochrane Database Syst Rev 2004;3.
59. Schuh S, Coates AL, Binnie R et al. Efficacy of oral dexamethasone in outpatients with acute bronchiolitis. J Pediatr 2002;140:27-32.
60. McBride JT. Dexamethasone and bronchiolitis: A new look at an old therapy? J Pediatr 2002;140:8-9.
61. Kuyucu S, Unal S, Kuyucu N, et al. Additive effects of dexamethasone in nebulized salbutamol or L-epinephrine treated infants with acute bronchiolitis. Pediatr Int 2004;46:539-544.
62. Bentur L, Shoseyov D, Feigenbaum D, et al. Dexamethasone inhalations in RSV bronchiolitis: A double-blind, placebo-controlled study. Acta Paediatr 2005;94:866-871.
63. van Woensel JB, van Aalderen WM, de Weerd W, et al. Dexamethasone for treatment of patients mechanically ventilated for lower respiratory tract infection caused by respiratory syncytial virus. Thorax 2003;58:383-387.
64. Christakis DA, Cowan CA, Garrison MM, et al. Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics 2005;115:878-884.
65. Mansbach JM, Emond JA, Camargo CA, Jr. Bronchiolitis in US emergency departments 1992 to 2000: Epidemiology and practice variation. Pediatr Emerg Care 2005;21:242-247.
66. Plint AC, Johnson DW, Wiebe N, et al. Practice variation among pediatric emergency departments in the treatment of bronchiolitis. Acad Emerg Med 2004;11:353-360.
67. Friedman MJ, Attia MW. Clinical predictors of influenza in children. Arch Pediatr Adolesc Med 2004;158:391-394.
68. Bonner AB, Monroe KW, Talley LI, et al. Impact of the rapid diagnosis of influenza on physician decision-making and patient management in the pediatric emergency department: Results of a randomized, prospective, controlled trial. Pediatrics 2003;112:363-367.
69. Esposito S, Marchisio P, Morelli P, et al. Effect of a rapid influenza diagnosis. Arch Dis Child 2003;88:525-526.
70. Sharma V, Dowd MD, Slaughter AJ, et al. Effect of rapid diagnosis of influenza virus type a on the emergency department management of febrile infants and toddlers. Arch Pediatr Adolesc Med 2002;156:41-43.
71 Byington CL, Enriquez FR, Hoff C et al. Serious bacterial infections in febrile infants 1 to 90 days old with and without viral infections. Pediatrics 2004;113:1662-1666.
72. Smitherman HF, Caviness AC, Macias CG. Retrospective review of serious bacterial infections in infants who are 0 to 36 months of age and have influenza A infection. Pediatrics 2005;115:710-718.
73. Bloomfield P, Dalton D, Karleka A, et al. Bacteraemia and antibiotic use in respiratory syncytial virus infections. Arch Dis Child 2004;89: 363-367.
74. Levine DA, Platt SL, Dayan PS et al. Risk of serious bacterial infection in young febrile infants with respiratory syncytial virus infections. Pediatrics 2004;113:1728-1734.
75. Melendez E, Harper MB. Utility of sepsis evaluation in infants 90 days of age or younger with fever and clinical bronchiolitis. Pediatr Infect Dis J 2003;22:1053-1056.
76. Titus MO, Wright SW. Prevalence of serious bacterial infections in febrile infants with respiratory syncytial virus infection. Pediatrics 2003;112:282-284.
77. Fonseca BK, Holdgate A, Craig JC. Enteral vs intravenous rehydration therapy for children with gastroenteritis: A meta-analysis of randomized controlled trials. Arch Pediatr Adolesc Med 2004;158:483-490.
78. Bender BJ, Ozuah PO. Intravenous rehydration for gastroenteritis: How long does it really take? Pediatr Emerg Care 2004;20:215-218.
Emergency department (ED) resources are continually strained due to overcrowding. This intensifies during the winter months because of epidemics of viral illness that affect all ages. The very young and elderly populations are at increased risk for significant morbidity and mortality related to viral disease, and as a consequence have higher rates of hospitalization than other age groups.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
