Managing Acute Decompensated Heart Failure in the Emergency Department: Part I
Managing Acute Decompensated Heart Failure in the Emergency Department: Part I
Authors: Thomas A. Tallman, DO, FACEP, Associate Staff Physician, Department of Emergency Medicine, The Cleveland Clinic Foundation, Cleveland, OH; and W. Frank Peacock, MD, Director of Clinical Operations, Department of Emergency Medicine, The Cleveland Clinic Foundation, Cleveland, OH.
Peer Reviewers: John Sarko, MD, Clinical Attending Physician, Department of Emergency Medicine, Maricopa Medical Center, Phoenix, AZ; and Frank LoVecchio, MD, Research Director, Maricopa Medical Center, Department of Emergency Medicine, Phoenix, AZ.
Introduction
Heart failure (HF) continues to cause substantial morbidity and mortality in the United States, accounting for a higher portion of Medicare costs than any other disease.1 In fact, this disease is reaching epidemic proportions. HF is the leading cause of hospitalization in patients over the age of 65,2 and it is the most common reason for re-hospitalization in the same group.3 The rate of hospitalization for heart failure increased 165% from 1979 to 2000.2 In 2004 alone, there were more than 1 million heart failure hospitalizations in the United States, and another 1 million in Europe.3
To fully understand this epidemic, the impact of comorbid illnesses, patient demographics, and cost need to be explored. Within the general patient population exists a growing number of patients older than age 65. Unfortunately, HF is predominantly a disease of the elderly. The impact of HF is further compounded by improved survival following many previously fatal events, including myocardial infarction and chronic disease processes (e.g., hypertension).2 The demographics have changed and large databases show that clinical trials do not accurately identify the true HF population.
A more accurate reflection of the heart failure population is represented by the Acute Decompensated Heart Failure National Registry (ADHERE), a data set that currently contains information on more than 200,000 acute decompensated heart failure (ADHF) hospitalizations and, more importantly, can be used to determine what currently represents the population and standard care of ADHF. Limitations include that it is a self-reporting registry and therefore is not considered the gold standard. Having said this, Table 1 compares clinical trial patients with patients from the ADHERE database. Note that while randomized controlled trials provide data that suggest a therapy or test can work, registry data tell if a therapy or test does work (as real world issues can negate the most effective therapy). A comparison of patients enrolled in ADHF randomized controlled trials vs. ADHERE registry data reveals that in the registry, the average patient is much older, more often female, and more often burdened by co-existent disease.
| Table 1. Randomized Controlled Trials vs. ED Patients |
|
It is estimated that 80% of patients who present to the emergency department with acute decompensated heart failure (ADHF) will be hospitalized, and repeat admissions are common. In an analysis of 100,000 ADHF hospitalizations from ADHERE, nearly 25% of patients had a history of prior HF hospitalizations in the 6 months preceding their index admission.2 Information regarding process of care is also provided by this registry. Of the total patients admitted for ADHF, 78% presented through the ED, and 89% reported shortness of breath as the chief complaint. Once hospitalized, 79% of ADHF patients were placed to either telemetry (66%) or the ICU (13%). Although initially small in number, observation unit admissions increased approximately 100% between 2003 and 2004, and are expected to increase as the Center for Medicare and Medicaid Services has identified this as an area to increase.
Altogether, the costs for heart failure hospitalizations approximate $14.7 billion annually, and outpatient costs are estimated to equal this number.1 Among patients hospitalized with heart failure, almost 80% are receiving Medicare benefits, and these Medicare costs outweigh the costs for patients treated for acute myocardial infarction or any single cancer. Follow-up costs also remain high, and one study estimated that one-year follow-up costs of treating heart failure exceed those of initial hospitalization by 50%.4
When considering the progress made regarding treatment and understanding of myocardial infarction, it is unfortunate that no major United States cardiac or emergency medicine professional organization has published guidelines on the treatment of acute decompensated heart failure (ADHF).3 While many chronic heart failure management guidelines exist and are promoted, the European Society of Cardiology is the only organization to have produced guidelines for acute management. Recommendations for the management of pulmonary congestion in the setting of acute myocardial infarction were published in 2004 by the American College of Cardiology and the American Heart Association.5 The emergency department is the common gateway to the hospital for many of these patients. Thus, the burden is on the emergency physician to recognize ADHF early and initiate treatment well in advance of disposition to any inpatient hospital setting. An estimated 80% of ED visits for HF result in hospitalizations.
For the current standard of ADHF to represent optimal care, then treatment strategies must be successful in reducing the frequency of hospitalizations and ED visits as well as reducing the days spent in the hospital. In addressing this objective, the greatest success has come with developing a team approach to the patient with recurrent decompensated heart failure and with the advent of observation units.1 With a balanced team approach, careful discharge planning, and outpatient care, patient outcomes improve and visits to the emergency department decrease.
One measure of successful ED treatment strategies is to evaluate chronic heart failure in the outpatient community setting. The ADHERE Disease Management Quality Initiative for Care Beginning in the Emergency Department Module (ADHERE ED DM) was initiated to provide insight into the treatment patterns and overall quality of disease management (DM) of ADHF in the emergency setting. This registry recorded more specific data on the emergency medicine encounter for ED patients in ADHF. The database offers insight into what currently represents the population and "standard of care" of ADHF treatment.6
Pathophysiology of Heart Failure
One of the most important challenges in the emergency department is to accurately diagnose heart failure. Patients may experience a period of latent or asymptomatic heart failure prior to ED presentation, and this makes it difficult to determine the etiology of their disease. Furthermore, this is primarily an elderly population with multiple comorbidities, and thus ADHF does not occur as a homogenous ED population. The combination of false-positive and false-negative diagnoses has been reported as high as 18.5% in the emergency setting.7-9
Understanding the pathophysiology of this disease is imperative to developing the approach to diagnosis and treatment. Although ADHERE data suggest that a greater number of patients with HF have hypertension than coronary artery disease, the presence of coronary artery disease is the underlying pathology in a significant number of patients.10,11 Cardiac markers such as troponin and creatinine kinase isoenzymes may be elevated in HF. Such elevation can predict prognosis.12,13 An increase in cardiac troponins has been associated with a poor prognosis in patients with ADHF and are related to the severity of HF.14 Thus measures to prevent the development of heart failure also center on resolving the risk factors associated with coronary artery disease, which include hypertension, obesity, smoking, diabetes, and dyslipidemia (though not immediate issues to address in the ED).15
ADHF, or cardiogenic pulmonary edema, is characterized by the transudation of excess fluid into the lungs secondary to increased left atrial pressure and subsequently pulmonary venous and capillary pressures. The net result is protein-poor fluid filtering across the pulmonary endothelium into the alveolar spaces. This leads to decreased diffusion capacity, hypoxia, and dyspnea. Cardiogenic pulmonary edema can manifest from either left atrial outflow impairment or left ventricular systolic or diastolic dysfunction. Despite different pathophysiologic initiators, symptoms at ED presentation initially will be identical, making treatment concerns supercede that of ejection fraction determination.
Left atrial outflow impairment can be acute or chronic. In chronically impaired left atrial outflow states (see Table 2), pulmonary edema precipitates from decreased left ventricular filling time as a result of rapid heart rate (HR). Acutely impaired left atrial outflow arises from increased left ventricular end-diastolic pressures, which lowers the ability of the left atrium to fill the left ventricle. Less common causes of impaired left atrial outflow include mitral stenosis and left atrial tumors.
| Table 2. Etiology of Cardiogenic Pulmonary Edema |
|
Pulmonary edema also can result from left ventricular systolic and diastolic dysfunction,15 left ventricular outflow obstruction, or left ventricular volume overload. Increased vascular volume can also precipitate pulmonary edema and can occur with pregnancy or increased salt intake. Reduced cardiac output as a result of a contractile deficit is the most common cause of cardiogenic pulmonary edema (CPE). The most common causes for systolic dysfunction are listed in Table 2. The impaired forward flow caused by systolic dysfunction activates the renin-angiotensin-aldosterone (RAAS) and sympathetic nervous system (SNS). This compensation leads to renal sodium and water retention and pulmonary edema.
Diastolic dysfunction is defined by an increase in ventricular stiffness, reduced compliance, and impaired ventricular filling during diastole. It can be induced by both acute and chronic disorders. It is further defined as increased left ventricular end diastolic pressure with a left ventricular ejection fraction greater than or equal to 40%. Diastolic dysfunction can exist with systolic dysfunction or can be an isolated process. Other factors that can lead to pulmonary edema during diastolic dysfunction include concurrent systolic dysfunction, reduced diastolic coronary blood flow, and arrhythmia. Echocardiography can be a useful tool in diagnosing diastolic dysfunction as the cause of an acute event and can identify normal ejection fraction ranges. Left ventricular volume overload can result from ventricular septal rupture, aortic insufficiency, and sodium retention due to renal disease. Acute aortic insufficiency is associated with congenital heart disease or may be the result of infective endocarditis, acute aortic dissection, lupus, or trauma. Left ventricular outflow obstruction can result from critical aortic stenosis, hypertrophic cardiomyopathy, or severe systemic hypertension.
To summarize, systolic dysfunction is due to pump failure, which typically results from ischemic cardiomyopathy, dilated cardiomyopathy (DCM), or valvular disease. Diastolic dysfunction is a factor in approximately one-third of ADHF cases and is associated with hypertension, hypertrophic cardiomyopathy, and infiltrative disease. High output causes of ADHF are less common, and are associated with anemia, hyperthyroidism, and pregnancy.16
Neurohormonal Imbalance in ADHF
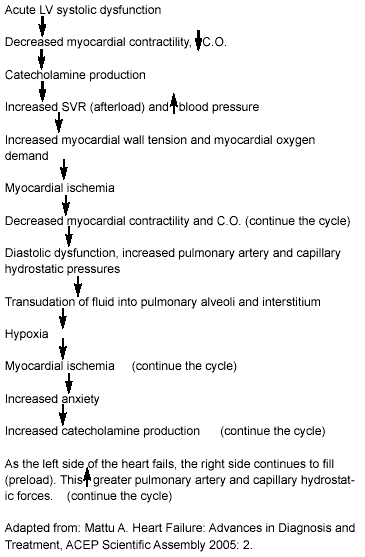
Left ventricular dysfunction results from myocardial injury. Changes occur in the peripheral circulation that also affect cardiac function and contribute to the symptoms of HF. Vasoconstrictors—those in the sympathetic nervous system and the RAAS, as well as arginine vasopressin and endothelin—become activated to increase afterload and preload by conserving sodium ions and water. Vasodilators—hormones in the natriuretic peptide system, as well as endothelin-derived relaxing factor and prostaglandins—help to unload the left ventricle and promote natriuretic action. In other words, the actions of atrial natriuretic peptides (ANP) and plasma brain natriuretic peptides (BNP) counteract the activation of the RAAS and sympathetic nervous system. Interventions, such as angiotensin-converting enzyme inhibitors (ACEIs) and beta blockers, have been developed to prevent ADHF by blocking the RAAS and SNS. Table 3 illustrates the physiologic effects of neurohormones and the counteraction of the natriuretic peptide system on the RAAS. Figure 1 illustrates the progression of heart failure following acute LV systolic dysfunction.
| Table 3. Physiologic Effects of Neurohormones |
|
| Figure 1. Pathophysiology |
|
Natriuretic Peptides
Natriuretic peptides (NP) represent a series of proteins released as a result of volume stimulus. First studied in 1981 by Debold it has since become clear that natriuretic proteins exert a counter-balancing influence on many of the deleterious consequences of neurohormonal activation.16 ANP originates from the cardiac atria and is increased in response to atrial distension. BNP is released in response to increased ventricular stretch or pressure. Its endogenous concentration can be assayed in the forms of BNP and NT–pro BNP. There are multiple assays available for BNP and at least two assays available for the NT–pro BNP. Only BNP is available on a point-of-care testing platform. Physiologically BNP and NT–pro BNP are very different from each other, but both are helpful to confirm the diagnosis of heart failure, especially when the signs and symptoms are ambiguous. The upper limit cutoff of normal varies between the two tests and the assays used.
Once in circulation, BNP binds to natriuretic peptide receptors (NPRs) which are located on endothelial and smooth muscle cells. The physiologic actions of BNP occur when it binds to NPR A and B receptors, which causes an increase in cyclic GMP. In essence, the natriuretic proteins exert a counterbalancing influence on many of the deleterious consequences of neurohormonal activation. BNP is metabolized by three pathways: binding to the NPR-C receptor, cleavage via neutral endopeptidase, and to a lesser extent by renal elimination. NT-pro BNP relies predominantly on renal elimination.
Clinical Features
Presenting Symptoms. Using a symptom-based approach, ADHF is not a homogenous presentation to the ED, but can be divided into two distinct syndromes, defined by the nature of their presentation. The two presentations are manifested as volume overload or hemodynamic mismatch and can be distinguished by the acuity of their symptoms and their response to vasodilation.3
In patients with hemodynamic mismatch, function is relatively near their baseline until they suffer a precipitous decompensating event, at which time they present to the ED with severe respiratory distress. The precipitating event leads to a decrease in CO- e.g., MI, dysrhythmia. Fluid overload generally is limited and the response to vasodilation is remarkable. They can, in fact, return to being nearly asymptomatic within minutes of treatment with vasodilators despite little volume being removed. The underlying pathophysiology in this cohort is hypothesized to be excessive systemic vascular resistance resulting from diminished cardiac output.17
Acute pulmonary edema presents clinically as tachypnea, rales, and respiratory distress due to pulmonary congestion. Dyspnea, especially exertional dyspnea, is the most common symptom of HF.2 An increase in respiratory rate (> 16) often accompanies dyspnea, but is a nonspecific symptom seen with a wide variety of pulmonary, cardiac, chest wall, or even neurologic disorders. Other manifestations include orthopnea (breathing discomfort precipitated by lying supine) and paroxysmal nocturnal dyspnea or sudden onset of dyspnea at night. Heart failure could also manifest as fatigue, increased frequency of nocturnal dyspnea, and increased systemic venous pressure, which contributes to the GI complaints.
In the volume overload patient, the acuity of presentation is that of moderate respiratory distress, and complaints generally are driven by exertional limitations or the discomfort from excessive volume. Diuretics can remove large volumes from these patients, and they may have partial improvement with vasodilation, but their symptoms only improve with volume removal.
Symptoms of HF can be divided into those due to left-sided or right-sided heart failure. Symptoms of left-sided failure include dyspnea, orthopnea, paroxysmal nocturnal dyspnea (PND), nocturia, fatigue, and altered mental state. Symptoms of right-sided heart failure are the result of elevated systemic venous pressure and include peripheral edema and GI complaints such as anorexia and nausea. A physical finding of jugular venous distention (JVD) may be the most important exam finding for estimating volume status.2
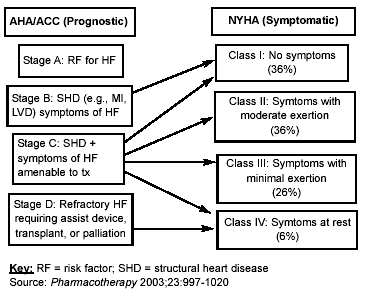
The New York Heart Association (NYHA) class severity for HF patients (see Figure 2) represents an historical standard for categorizing clinical severity of HF. NYHA class is directly proportional to survival. There is, however, no direct relationship between NYHA and ejection fraction (EF). New York Heart Association class does suffer from interobserver variability.18 Brain-type natriuretic peptide assay correlates are provided and likely provide a more objective measure. The mortality rate in NYHA class III patients is 15% to 20% per year. In class IV the mortality rate increases to 50% per year or more.
| Figure 2. Heart Failure Classification Systems |
|
The American College of Cardiology and the American Heart Association have published a classification system and includes patients who are in asymptomatic initial stages of HF. This system does recognize the fact that earlier intervention has the potential for greater morbidity and mortality benefits as compared to therapy provided in later stages.19,20
Differential Diagnoses
There are a number of clinical conditions that can mimic HF and the wrong diagnosis can delay optimal response to treatment or cause further decompensation. Shortness of breath is such a common presenting symptom that other conditions causing dyspnea need consideration. Table 4 summarizes the differential diagnosis for dyspnea in the clinical setting. For instance, impending pulmonary embolism (PE) can present with intense dyspnea but in the absence of rales or edema.
| Table 4. Heart Failure Differential Diagnosis for Dyspneic States |
|
It is imperative to be able to distinguish between two diagnoses such as pulmonary edema and PE due to the potential for rapid deterioration. In the patient with pulmonary edema, arterial blood pressure often is elevated and requires immediate therapy with vasodilators. Decreasing preload in a patient with PE may lead to deterioration.
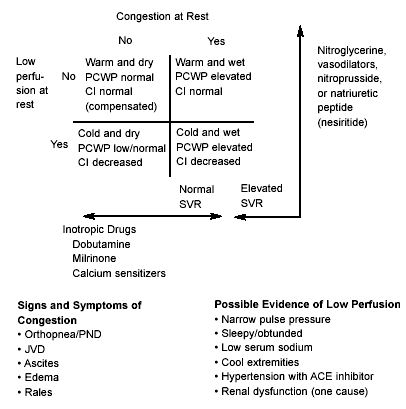
A useful approach to categorizing heart failure in the ED is by hemodynamic classification (see Figure 3). Patients can be placed into the appropriate hemodynamic category based on initial history and physical and then appropriate therapy can be selected.2 For example, the warm and dry patient is well compensated with no congestion at rest. Symptoms typically require minor medication adjustments and follow-up. The warm and wet patient is fluid overloaded and well perfused.
| Figure 3. Hemodynamic Profile Assessment for Patient Selection and Treatment |
|
Physical Exam and ADHF Diagnosis
Diagnosis of ADHF begins with a brief history and focused physical exam. A brief history should focus on risk factors, signs or symptoms of myocardial ischemia, arrhythmia, any potential infectious processes and, as appropriate, PE.21 Questions for the patient or family should include salt intake, medication compliance, and use of alcohol or drugs.
After addressing the airway, breathing, and circulation (ABCs) in treatment, the physical exam should focus on heart and lung sounds, especially murmurs that could indicate valve insufficiency or outflow tract obstruction. Such lesions warrant avoidance of aggressive therapies aimed at prompt reduction of preload and afterload.
Diagnosis of ADHF: How Good is the History and Physical Exam
Auscultation in the ED can be challenging, but should focus on the identification of heart sounds, murmurs, and the presence of rales. Rales may be heard in both lungs and represent the physical finding of pulmonary congestion.
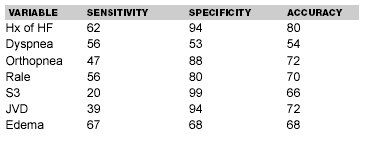
A third heart sound, if present, may be difficult to auscultate in the ED setting. It is due to decreased compliance of the left ventricle and most commonly is heard in patients with volume overload and tachycardia. An S3, when detected, is specific for elevated left ventricular pressure,22 low ejection fraction,23 and worse outcomes.24-26 Pulsus alternans may be present in severe cases and if identified is virtually diagnostic of severe advanced heart failure.27 (See Tables 5 and 6.)
| Table 5. Sensitivity of History and Physical Findings for an ADHF Diagnosis |
|
| Table 6. Findings of Physical Examination |
|
Hepatojugular reflux can occur in patients with mild heart failure and is manifested as JVD while compressing the liver. Right-sided heart failure leads to raised systemic venous pressure, resulting in JVD, peripheral edema, and ascites. JVD may be the most important exam finding for estimating volume status.2
Look for peripheral edema to accumulate in dependent parts of the body such as feet, ankles, and the pretibial regions of legs in ambulatory people. Presacral edema is more common in non-ambulatory patients. Ascites is a result of elevated pressure in hepatic veins and transudation of fluid into the peritoneal cavity. Keep in mind that misdiagnoses can be attributed to obesity, deconditioning, or another dyspneic condition. When limited to using only the history and physical exam, the diagnosis of HF frequently is in error.
Ancillary Testing
Objective tools can improve diagnostic accuracy. A right heart catheterization can yield great diagnostic value, but is not routinely performed in the ED. Echocardiography, the gold standard to measure ejection fraction and wall motion, also is not readily available, but reports of previous echocardiograms (ECGs) may be found in prior records.
Chest Radiography. A chest x-ray (CXR) should be obtained in all suspected ADHF patients but use caution in the interpretation. The key findings on the CXR indicative of HF are cardiomegaly and evidence of redistribution (cephalization) but, due to limited sensitivity, these findings alone cannot exclude or confirm HF or elevated filling pressure. Though incapable of excluding LV dysfunction, the CXR can help confirm other diagnoses, such as pneumonia. The key findings on the CXR indicative of HF are (in order of descending frequency): dilated upper lobe vessels, cardiomegaly, interstitial edema, enlarged pulmonary artery, pleural effusion, alveolar edema, prominent superior vena cava, and Kerley lines. The ED physician often encounters chronic HF patients in which the CXR does not identify patients with high pulmonary capillary wedge pressure (PCWP).28
Cardiomegaly can suggest an HF diagnosis, and a cardiothoracic ratio greater than 60% correlates with increased five-year mortality.28 However, the CXR has poor sensitivity for cardiomegaly (20% of cardiomegaly seen on echocardiography is missed on CXR).29 Pleural effusions are common in HF but often missed on CXR. A portable CXR done with the patient in the supine position further degrades the diagnostic accuracy to detect pleural effusions. Detection of pleural effusion if supine is 67% sensitive, 70% specific, and 67% accurate, as demonstrated in one study of 34 patients with pleural effusions proven by decubitus CXRs.30
Sensitivity is poor for HF findings with a portable radiograph. (See Table 7.) In cases of mild HF, only dilated upper lobe vessels were found in greater than 60% of patients.3 With severe HF, CXR findings occurred in at least 66% of patients, thus the frequency of CXR findings that confirm HF increases with severity of HF.
| Table 7. Chest Radiograph |
|
Electrocardiography. Though of limited diagnostic value, an ECG should be performed on every patient who presents with dyspnea and suspected HF. The ECG is a useful diagnostic tool in the assessment of HF patients because it can identify causal abnormalities, such as acute MI and acute ischemia, both of which would require CCU admission as well as a specific approach to treatment. An ECG also can assess cardiac rhythm, identify possible electrolyte disturbance (e.g., hyperkalemia), and may indicate a potential drug toxicity (e.g., junctional bradycardia due to digoxin toxicity).
The ECG also offers prognostic information. In dilated cardiomyopathy, the presence of abnormal Q waves, QRS duration greater than 0.12 ms, or left bundle-branch-block predict an increased five-year mortality rate.31 The resting ECG is relatively insensitive and nonspecific in identifying severe underlying coronary disease (e.g., presence or absence of old Q waves).
A period of cardiac monitoring is essential for patients with HF. Continuous monitoring may be indicated by the clinical presentation (e.g., recent palpitations or near-syncope). With aggressive treatment such as repetitive diuretic dosing, an electrolyte abnormality may increase the possibility of arrhythmias. Initially, ECG monitoring may exclude cardiac arrhythmias as the cause of the current exacerbation.31-33
Heart Failure and Digital Heart Sound Detection for Rapid Diagnosis and Initial Treatment
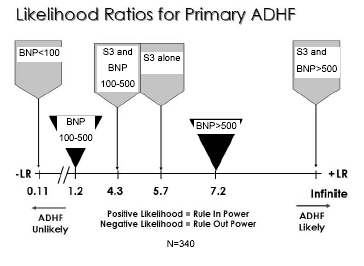
Phonocardiography is the measurement of heart sounds and can be used to help detect an S3 or an S4 that easily can be missed in the noisy ED environment. The third (S3) heart sound may be a normal finding in patients younger than 40 years, but in the patient at risk for HF, the S3 is 93-99% specific for positive diagnosis of ADHF. The presence of an S3 is highly correlated to LV systolic dysfunction, reduced ejection fraction, and elevated LV end diastolic pressure. In acute myocardial infarction (AMI), S3 usually indicates concomitant HF. In dialysis patients, S3 usually indicates fluid overload and elevated left ventricular end diastolic pressure (LVEDP). A third heart sound and JVD together carry a poor prognosis. Figure 4 illustrates likelihood ratios for primary ADHF.
| Figure 4. Sensitivity, Specificity, and +LR for an S3 in Predicting HF in Challenging Subgroups |
|
The fourth (S4) heart sound is an uncommon finding in young healthy subjects, but is nonspecific in the elderly, which is the population with HF. This highly correlates to LV dysfunction, elevated LVEDP and reduced LV compliance. An S4 may be present in ADHF but may be present in other conditions, such as valvular disorders, LVH, and hypertrophic obstructive cardiomyopathy. New devices allow digital detection of extra heart sounds, taken at the same time as the ECG via two acoustical sensors in the V3/V4 positions. Algorithms extract ECG and acoustical information simultaneously.
Laboratory Testing. Asymptomatic cardiac ischemia can precipitate acute HF or produce decompensation in previously stable HF patients. Troponin and creatinine kinase isoenzyme levels are predictors of adverse prognosis and risk in patients with HF.8,9 Because of their kinetics, a positive result may diagnose cardiac ischemia, but a negative result may not exclude it. As compared to AMI in non-HF patients, elevated cardiac necrosis markers in HF may not necessarily represent epicardial coronary artery occlusion.34 However, cardiac marker elevation is associated with an increased risk of adverse outcome.35-40 Perna et al. concluded that a cardiac troponin T (cTnT) level of 0.1 ng/mL or greater was associated with poor long-term survival and emerged as a powerful independent predictor of mortality in patients with acute cardiogenic pulmonary edema.41
Additional laboratory tests to consider include serum electrolytes. Serum magnesium levels should be ordered when either arrhythmias or severe hypokalemia is present.
Both sodium and potassium can be measurably altered by the activation of the RAAS or by diuretic treatment. Low serum sodium is not uncommon and is due to dilutional hyponatremia from expansion of extracellular fluid volume and correlates with a worse prognosis. The same can be said for worsening renal function. Elevated creatinine and liver function tests also are predictors of poor outcome. Inflammatory markers (e.g., C-reactive protein) appear to be elevated in CHF patients; however, their prognostic role requires further study.32
Natriuretic Peptides as a Diagnostic Tool
Brain-type natriuretic peptide levels correlate well with function and clinical presentation. In a study examining patients with and without heart failure, mean BNP levels were 38 in normal patients and 1076 pg/mL in those with heart failure.42 The extent of the BNP elevation also corresponds well to functional class. The NYHA classification is used to estimate severity of heart failure, with higher scores being directly proportional to increased mortality. When heart failure is graded by NYHA class, the BNP levels vary directly with severity.43,44
Endogenous BNP has a half-life of only 22 minutes.42 In comparison NT–pro BNP has a half-life of 1-2 hours. In the ED, BNP or NT-pro BNP can be used to diagnose heart failure. It is most helpful in those patients that are difficult to diagnose, such as in the combination of chronic obstructive pulmonary disease (COPD) and heart failure. In dyspnea resulting from isolated COPD, BNP generally is less than 100 pg/mL as compared to those whose symptoms are the result of heart failure where levels may exceed 1000 pg/mL.42
BNP is able to predict clinical events in patients presenting to the emergency department. Levels greater than 480 pg/mL predicted a markedly increased risk of death or re-hospitalization from heart failure in the following one to six months, as compared to those patients with levels less than 230 pg/mL.43 Mortality also is predicted by BNP. Mortality in patients with BNPs greater than 73 pg/mL is markedly increased in the next 10-12 months, as compared to those with lower BNPs. Research indicates that admission status also correlates with BNP levels. In one study, heart failure patients required hospital admission with BNP levels greater than 700 pg/mL, while those able to be treated as an outpatients had BNPs less than 254 pg/mL.42-44
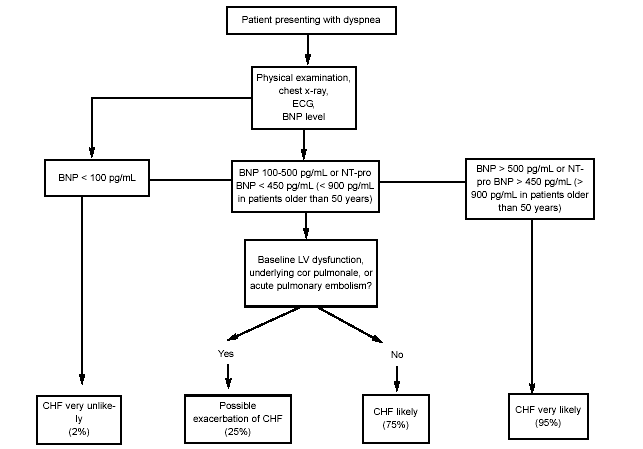
Figure 5 shows the recommended cutpoints of BNP as well as the general BNP reference guidelines. Current (BNP consensus panel)45 recommendations suggest the laboratory should perform BNP testing on a continuous 24-hour basis with a turn-around time of 60 minutes or less. If the BNP is less than 100 pg/mL, then HF is highly unlikely (negative predictive value is 90%). If the BNP is greater than 500 pg/mL, then HF is highly likely (PPV = 90%). If the BNP is 100-500 pg/mL, consider that the baseline BNP may be elevated due to a stable underlying dysfunction, which could include right ventricular failure from core pulmonale, acute pulmonary embolism, or renal failure. Brain-type natriuretic peptide also rises with age and is affected by gender, comorbidity, and drug use, and should not be used as the sole diagnostic tool in assessing patients for heart failure. It should be considered in addition to the clinical context presented by the patient history and physical examination, chest x-ray, ECG, and other laboratory assessments.
| Figure 5. Algorithm for Using BNP Levels in the ED to Diagnose CHF37,38 |
|
A number of clinical scenarios can alter BNP levels. BNP is altered with chronic renal insufficiency. As chronic kidney disease advances, a higher BNP cut-off point may be required. An upper limit of normal of approximately 200 pg/mL is reasonable for those with an estimated glomular filtration rate (GFR) less than 60 mL /min,42 In other non-heart failure cardiopulmonary diseases, BNP values may be increased. In approximately 20% of patients with pulmonary disease, BNP is elevated, which implies combined heart failure and lung disease, or cor pulmonale. In the setting of pulmonary embolism, BNP is elevated in one-third of cases and is associated with right ventricular pressure overload and higher mortality. Brain-type natriuretic peptide is not diagnostic for acute PE, however, and BNP levels can range from 100 to 500 pg/mL in cases of pulmonary hypertension and right ventricular pressure volume overload. Finally, BNP has been studied as a marker in patients with acute coronary syndromes. When used together, BNP and cardiac troponin levels provided a more effective tool for identifying patients at an increased risk for clinically important cardiac events related to acute coronary syndrome (ACS) and HF.46-48
There are situations when BNP levels are lower than expected clinically. In cases of preserved systolic function heart failure or diastolic myocardial dysfunction, BNP may be found to be approximately half as high as when compared to cases of systolic dysfunction. Therefore, BNP may or may not detect patients with diastolic dysfunction. Obese patients tend to have lower levels of BNP when measured for any given severity of heart failure. Physiologically, adipose tissue is related to the natriuretic clearance receptor, and obesity can interfere with the usual diagnostic approach to heart failure. In a study by Mehra, nearly 40% of obese patients had BNPs less than 100 pg/mL.49 Lastly, unexpectedly low levels can occur during the first one to two hours of flash pulmonary edema.50,51 Table 8 illustrates special circumstances in interpreting BNP levels. Figure 5 illustrates an algorithm for using BNP levels in the ED to diagnose ADHF. There is less literature on the use of NT-pro BNP. Elevation of NT-pro BNP above 450 pg/mL in patients younger than 50 years, or elevations above 900 pg/mL in patients older than 50 years are sensitive and specific for the diagnosis of HF.52
| Table 8. Special Circumstances in Interpreting BNP Levels |
|
Risk Stratification
This is a process well known in the setting of suspected acute coronary syndrome but is less well defined for ADHF. Risk stratification for ADHF can be divided into low- and high-risk predictors for adverse outcomes. Patients with a low risk of adverse outcomes can be considered for possible 23-hour observation admission. Longer hospitalization and aggressive therapy would be warranted for patients at high risk for an adverse event.
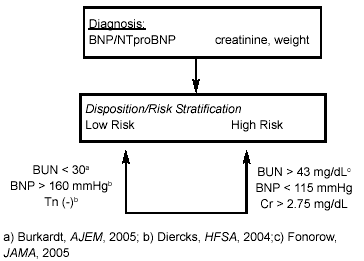
The low-risk population was evaluated in two studies. One study53 evaluated predictors of successful therapy in nearly 500 patients. Low risk was defined as discharge within 24 hours, and no death or rehospitalization within 30 days. Parameters predicting low risk include an initial negative troponin and systolic blood pressure (SBP) greater than 160 mmHg. Burkhardt54 used similar methods and was able to identify predictors of short stay failure. A measured BUN greater than 30 mg/dL at ED presentation was associated with an increased probability of hospitalization in excess of 24 hours and, therefore, identified patients inappropriate for observation unit therapy.
A high-risk population also can be identified. An analysis of more than 80,000 patients from the ADHERE registry revealed a BUN exceeding 43 mg/dL to be the single greatest acute mortality predictor.55 The mortality risk of nearly 10% compared to only 2.8% if the BUN was less than 43 mg/dL. The next most powerful mortality predictor was an SBP below 115 mmHg. Combined with a high BUN, mortality increased to approximately 15%. If serum creatinine is also more than 2.75 mg/dL, in-hospital mortality is greater than 22%. (See Figure 6.)
| Figure 6. Risk Stratification of Emergency Department Patients with Acute Dyspnea |
|
These parameters help identify patients appropriate for a 23-hour observation unit vs. those requiring more aggressive care and admission to a more critical bed. Patients directed to an observation unit can receive rapid emergency care for HF symptoms with close follow-up arranged at the time of discharge.
References
1. Peacock WF, Young J, Collins S, et al. Heart failure observation units: Optimizing care. Cardiology/Review Article. Ann Emerg Med 2006;47:22-33.
2. Peacock WF, Tiffany B. Cardiac Emergencies. Congestive Heart Failure: Demographics and Economics. The McGraw-Hill Companies, Inc. 2006;297.
3. Peacock WF. Heart failure in the emergency department. Critical Pathways in Emergency Medicine: A journal of evidence-based medicine. 2003;2:207-220.
4. Ansari M, Massie BM. Heart failure: How big is the problem? Who are the patients? What does the future hold? Am Heart J 2003;146:1-4.
5. Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction—executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation 2004;110:588.
6. Summers RL. Disease management of acute decompensated heart failure: The ADHERE emergency medicine module. Emergency diagnosis and treatment of ADHF. CME Monograph from ACEP 2005 Spring Congress Satellite Symposium. Mar 4, 2005; 33-38.
7. Peacock WF, Emerman CL. Preventing readmission in heart failure. Emerg Med October 2003. Available at: http://www.emedmag.com/html/pre/cov/covers/101503.asp. Accessed March 16, 2005.
8. Freda BJ, Peacock WF, Lindwell CJ, et al. Outcomes in heart failure patients with elevated cardiac markers of ischemia [abstract]. J Card Fail 2002;8(4suppl):570.
9. Peacock WF. Acute emergency department management of heart failure. Heart Fail Rev 2003;8:335-338.
10. Dao Q, Krishnaswamy P, Kazanegra R, et al. Utility of B-type natriuretic peptide in the diagosis of congestive heart failure in an urgent care setting. J Am Coll Cardiol 2001;37:379-385.
11. Maisel AS, Krishnaswamy P, Nowak RM, et al. Breathing Not Properly Multinational Study Investigators: Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med 2003;347:161-167.
12. Hut SA, Baker DW, Chin MH, et al. ACC/AHA guidelines for the evaluation and management of chronic heart failure in the adult; executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (committee to revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). J Am Coll Cardiol 2001;38:2101-2113.
13. Dei Cas L, Metra M, Nodaic S, et al. Prevention and management of heart failure in patients at risk. Am J Cardiol 2003;91(suppl 9A):10F-17F.
14. Potturi S, Ventura HO, Mulumundi M, et al. Cardiac troponin levels in heart failure. Cardiology in Review 2004;12:21-25.
15. Black HR. The burden of cardiovascular disease: Following the link from hypertension to myocardial infarction and heart failure. Am J Hypertens 2003;16(suppl 1):4S-6S.
16. Sharis PJ, Connor CP. Evidence Based Cardiology. Philadelphia: Lippincott Williams and Wilkins; 2003:290-291.
17. Maisel AS, Cairns CB, Kosowsky JM. Urgent management of acute decompensated heart failure: The role of the emergency physician. Clinician Vol.23, No. 2.
18. de Groote P, et al. Right ventricular ejection fraction is an independent predictor of survival in patients with moderate heart failure. J Am Coll Cardiol 1998;32:948-954.
19. Missov E, Mais J. A novel biochemical approach to congestive heart failure: Cardiac troponin T. Am Heart J 1999;138:95-99.
20. Glover DR, Littler WA. Factors influencing survival and mode of death in severe chronic ischemic cardiac failure. Br Heart J 1987;57:125-132.
21. Rame JE, Dries DL, Drazner MH. The prognostic value of the physical examination in patients with chronic heart failure. Congest Heart Fail 2003;9:170-175,178.
22. Patel R, Bushnell DL, Sobotka PA. Implications of an audible third heart sound I evaluation cardiac function. West J Med 1993;158:606-609.
23. Pina IL. The epidemiology, pathophysiology, and diagnosis of heart failure. Cardiology Review 2005;(22, suppl 8):4-5.
24. Drazner MH, Rome JE, Stevenson LW, et al. Prognostic importance of elevated jugular venous pressure and a third heart sound in patients with heart failure. N Engl J Med 2001;345:574-587.
25. Peacock WF, Freda BJ. The clinical challenge of heart failure: Comprehensive evidence-based management of the hospitalized patient with acute myocardial decompensation—diagnosis, risk stratification, and outcome effective treatment. Emerg Med Report 2002;23:81-92.
26. Spodick D, Quary V. Prevalence of the fourth heart sound by phonocardiography in the absence of cardiac disease. Am Heart J 1974;87:11-14.
27. Lee YC, Sutton FJ. Pulsus alternans in patients with congestive cardiomyopathy. Circulation 1982;65:1533-1534.
28. Chakko S, Woska D, Marinez H, et al. Clinical, radiographic, and hemodynamic correlations in chronic congestive heart failure: Conflicting results may lead to inappropriate care. Am J Med 1991;90:353-359.
29. Kono T, Suwa M, Ngnada H, et al. Clinical significance of normal cardiac silhouette in dilated cardiomyopathy—evaluation based upon echocardiography and MRI. Japanese Circulation Journal 1992;56:359-365.
30. Ruskin RA, Gurney JW, Thorsen MK, et al. Detection of pleural effusions on supine chest radiographs. AJR Am J Roentgenol 1987;148:681-683.
31. Koga Y, Wada T, Toshima H, et al. Prognostic significance of ECG findings in patients with dilated cardiomyopathy. Heart Ves 1993;8:37-41.
32. DeFerrari GM, Tavazzi L. The role of arrhythmias in the progression of heart failure. Eur J Heart Fail 1999;1:33-40.
33. Mathew J, Hunsberger S, Fleg J, et al. Incidence, predictive factors, and prognostic significance of supraventricular tachyarrhythmia in congestive heart failure. Chest 2000;118:914-922.
34. Del Carlo CH, O'Connor CM. Cardiac troponins in congestive heart failure. Am Heart J 1999;138:646-653.
35. Sato Y, Kita T, Takatsu Y, et al. Biochemical markers of myocyte injury in heart failure. Heart 2004;90:1110-1113.
36. Missou E, Calzolari C, Pau B. Circulating cardiac troponin I in severe congestive heart failure. Circulation 1997;96:2953-2958.
37. Setsuta K, Seino Y, Takahashi N, et al. Clinical significance of elevated levels of cardiac troponin T in patients with chronic heart failure. Am J Cardiol 1997; 84:608-611.
38. LaVecchia L, Mezzena G, Onetto R, et al. Detectable serum troponin I in patients with heart failure or non-myocardial ischemic origin. Am J Cardiol 1997;80:88-89.
39. Healey JS, Smith ST, Davies RF, et al. Prognostic value of serum troponin T and troponin I in patients with heart failure. J Card Fail 2000;6:234.
40. Perna ER, Cimbaro Canella JP, Macin SM, et al. Combined determination of natriuretic peptides and necrosis markers in decompensated heart failure: Should NT-pro BNP and troponin T be measured on admission, predischarge, or both? J Card Fail 2004;10:S102-S103.
41. Perna ER, Macin SM, Parras JI, et al. Cardiac troponin T levels are associated with poor short- and long-term prognosis in patients with acute cardiogenic pulmonary edema. Am Heart J 2002;143:814-820.
42. Peacock WF, Tallman TA. Critical Decisions in Emergency Medicine: B-Type Natriuretic Assay. 2006;20:13-22.
43. Silver MA, Maisel A, Yancy CW, et al. BNP Consensus Panel 2004; A Clinical Approach for the Diagnostic, Prognostic, Screening, Treatment Monitoring, and Therapeutic Roles of Natriuretic Peptides in Cardiovascular Diseases. CHF 2004;10:53, 1-36.
44. Mueller C, Scholer A, Laule-Killian K, et al. Use of B-type natriuretic peptide in the evaluation and management of acute dyspnea. N Engl J Med 2004;350:647-654.
45. Peacock WF. The evolving role of BNP in the diagnosis and treatment of CHF: A summary of the BNP Consensus Panel Report. Emergency Diagnosis and Treatment of ADHF. CME Monograph from ACEP 2005 Spring Congress: 16-26.
46. Kikenta K, Yasue H, Voshimura M, et al. Increased plasma levels of B-type naturetic peptide in patients with unstable angina. Am Heart J 1996;132:101-107.
47. Roongsritong C, Warraich I, Bradley C. Common causes of troponin elevations in the absence of scute myocardial infarction. Chest 2004;125:1877-1884.
48. Velazquez EJ, Pfeffer MA. Acute heart failure complicating acute coronary syndromes. A deadly intersection. Circulation 2004;109:440-442.
49. Mehra MR, Uber PA, Park M, et al. Obesity and suppressed B-type natriuretic peptide levels in heart failure. J Am Coll Cardiol 2004;43:1590-1595.
50. Silver MA, Maisel A, Yancy CW, et al. BNP Consensus Panel 2004: A clinical approach for the diagnostic, prognostic, screening, treatment, monitoring, and therapeutic roles of natriuretic peptides in cardiovascular diseases. Congest Heart Fail 2004;10:1-30.
51. Mueller C, Scholer A, Laule-Killian K, et al. Use of B-type natriuretic peptide in the evaluation and management of acute dyspnea. N Engl J Med 2004;350:347-654.
52. Januzza JL Jr, Camargo CA, Anwaruddin S, et al. The N terminal pro BNP investigation of dyspnea in the emergency department (PRIDE) study. Am J Cardiol 2005;95:948-954.
53. Dierck DB, Hisk JD, Peacock WF, et al. Identification of emergency department patients with decompensated heart failure at low risk for adverse events and prolonged hospitalization. J Cardiac Fail 2004;(Suppl) 10:5118.
54. Burkhardt J, Peacock WF, Emerman CL. Elevation in BUN predicts a lower discharge rate from the observation unit. Ann Emerg Med 2004;(Suppl)44(4):599.
55. Fonarow GC, Adams KF Jr, Abraham WT, et al. Risk statification for in-hospital mortality in acutely decompensated heart failure: Classification and regression tree analysis. JAMA 2005;293:572-580.
Heart failure (HF) continues to cause substantial morbidity and mortality in the United States, accounting for a higher portion of Medicare costs than any other disease.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.