Managing Acute Decompensated Heart Failure in the Emergency Department: Part II
Managing Acute Decompensated Heart Failure in the Emergency Department: Part II
Authors: Thomas A. Tallman, DO, FACEP, Associate Staff Physician, Department of Emergency Medicine, The Cleveland Clinic Foundation, Cleveland, OH; and W. Frank Peacock, MD, Director of Clinical Operations, Department of Emergency Medicine, The Cleveland Clinic Foundation, Cleveland, OH.
Peer Reviewers: John Sarko, MD, Clinical Attending Physician, Department of Emergency Medicine, Maricopa Medical Center, Phoenix, AZ; and Frank LoVecchio, MD, Research Director, Maricopa Medical Center, Department of Emergency Medicine, Phoenix, AZ.
The treatment of acutely decompensated heart failure (ADHF) has changed dramatically in the past decade. As discussed in Part I, there is a new understanding of the pathophysiology and a significant improvement in our ability to diagnose heart failure with BNP and NT-pro BNP testing. Treatment has also changed. Standard therapy with morphine has been replaced with nitroglycerin. Patients are now maintained on angiotensin-converting enzyme inhibitors (ACEIs) and beta-blockers. But few new treatments have been surrounded by more controversy than the use of a recombinant natriuretic peptide, nesiritide. In reality, it is difficult, if not impossible, to be impartial in the interpretation of the literature on this subject. Although the authors of this article attempt to present a balanced discussion of the use of nesiritide, it is unlikely that the reader will interpret it as such. Those who believe in the use of the drug likely will find its negative aspects are over-emphasized. Those who believe the drug is too expensive and potentially dangerous likely will focus on the favorable discussion. The final verdict on nesiritide is not yet clear.
—Sandra M. Schneider, MD, FACEP, editor
Management of Heart Failure in the Emergency Department
A key role is played by the emergency physician in the emergency department (ED) regarding evaluation and management of the HF patient. The ED physician must have a firm understanding of the hemodynamic classifications of ADHF. An early goal directed therapy (EGDT-HF) approach emphasizes aggressive treatment, because preliminary evaluations have shown that patients treated early tend to have shorter hospital stays and better outcomes than patients whose intervention is delayed.1,2
The aim of EGDT-HF is listed in Table 1. Care needs to focus on rapid initiation of proven therapies that improve patient symptom and cardiorespiratory status without placing the patient at risk for immediate (i.e., arrhythmia, hypotension, ischemia) or delayed (i.e., worsening renal insufficiency, toxicity) adverse events.3
| Table 1. Early Goal-Directed Therapy HF |
|
• Hemodynamic and respiratory improvement • Prompt relief of symptoms • Enhance decision-making in the ED with an emphasis on timely transition to inpatient care if indicated • Early initiation of therapy to facilitate hospital discharge • Avoidance of high resource utilization |
During patient assessment, start with oxygen, the ABCs (airway, breathing, circulation), and an electrocardiogram (ECG). Pharmacologic treatment is aimed at decreasing right-sided filling (preload) and increasing left-sided emptying. The latter is achieved by decreasing systemic vascular resistance (SVR, or afterload). Improving LV contractility (inotropic support) is necessary in some cases, but only in the presence of symptomatic hypotension. Inotropes, such as dobutamine and milrinone, increase myocardial contractility, but carry adverse side effects, and thus should be avoided when possible. Table 2 illustrates an overview of current treatment choices for ADHF in the ED.
| Table 2. Current Treatments for Acutely Decompensated Heart Failure |
|
Diuretics, vasodilators, and inotropic agents have been effective in the treatment of patients with heart failure, but only agents that are capable of antagonizing the neurohormonal response have demonstrated a beneficial effect on mortality.4 Neurohormonal antagonists include angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), beta-blockers, and aldosterone antagonists.
Patients with ADHF frequently present with worsened symptoms in a hypervolemic state. Therapy initially may target this excess fluid by incorporating diuretics along with vasodilators to help reduce the overloaded state. Treatment options for patients with HF are directed at the pathophysiological mechanisms that are stimulated in HF.5 Nitroglycerin (NTG) is a vasodilator and should be a first-line therapy. Administered sublingually, it is a rapid and effective treatment, and is followed by topical NTG or intravenous NTG.6 ACEIs and ARBs decrease afterload by interfering with the renin-angiotensin-aldosterone (RAAS), leading to peripheral vasodilatation. They also affect LV hypertrophy, remodeling, and renal blood flow. Heart failure leads to increased aldosterone production by the adrenal glands, which stimulates renal sodium retention and potassium excretion and promotes ventricular and vascular hypertrophy. ACEIs may be considered as second-line therapy after NTG.
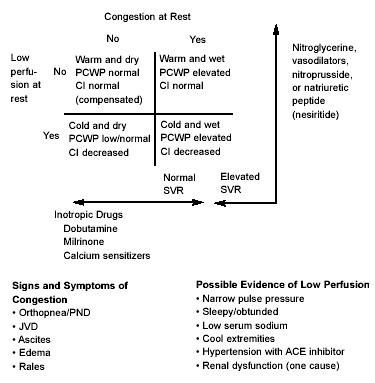
Referring back to hemodynamic profile assessment in Figure 1, if there is adequate blood pressure, vasodilators and diuretics are good treatment modalities. The cold and wet patient is fluid overloaded with diminished perfusion. Those with elevated SVR should improve with vasodilators and diuretics and, for symptomatic hypotension, the addition of a pressor agent must be considered. The cold and dry patient is poorly perfused. A fluid bolus will be required and, if unresponsive to this, inotropic support and pressors must be added. Most patients presenting to the ED with ADHF will be fluid overloaded and have adequate perfusion (warm and wet).
| Figure 1. Hemodynamic Profile Assessment for Patient Selection and Treatment |
|
Treatment for Acute Decompensated Heart Failure
Emergency treatment should start with assessing the vital signs and the ABCs. Patients who are extremely short of breath need an immediate airway assessment. This could lead to consideration of supplemental oxygen via nasal cannula or mask options. Persistent hypoxia in the continued dyspneic/tachypneic patient must provoke consideration for intubation or an airway adjunct such as BiPAP or CPAP.
Noninvasive Ventilation. All patients presenting to the ED with HF should receive supplemental oxygen. More aggressive adjuncts must be considered in patients with severe respiratory distress and dyspnea. In case of impending respiratory failure, endotracheal intubation is a consideration. However, noninvasive ventilatory techniques may be considered in selected patients.
The goal is to maintain the patency of the stiff fluid-filled alveoli and to prevent collapse during exhalation. By keeping alveoli open, the work of breathing is decreased. Other benefits include better oxygen and CO2 exchange, increased intrathoracic pressure, and increased cardiac output secondary to decreased preload and afterload. Such a therapeutic strategy must be considered early.
There are two noninvasive ventilatory techniques: continuous positive airway pressure (CPAP) and bi-level positive airway pressure (BiPAP).7 Patients must be awake and be able to cooperate with instructions. Patients who are obtunded, apneic, or near respiratory arrest must be excluded from consideration. Settings are determined by the individual patient, but consider a lower setting initially, such as an IPAP of 10 and an EPAP of 5, to allow the patient to tolerate and adjust. Then titrate settings upward in increments of 2-4 to achieve desired effects, based on patient comfort, persistent dyspnea, and blood gas monitoring.
Little evidence exists that suggests one technique is superior to the other, although some smaller studies suggest CPAP may be superior.8 CPAP maintains a constant pressure throughout all phases of the respiratory cycle. BiPAP allows for separate pressure and control in inhalation and exhalation and provides greater potential comfort.
Inotropic/Pressor Therapy. Following airway and breathing management, consider the blood pressure. Patients with persistent hypotension (systolic blood pressure < 85 mmHg) are candidates for pressor support. While generally discouraged in ADHF because of increased mortality risk, it is sometimes necessary to add inotropic and pressor agents despite deleterious effects such as ventricular ectopy. No single agent has been found to be clinically superior, but adrenergic agents may have a role in certain hemodynamic states. Adrenergic agents include the catecholamines dobutamine and dopamine, and the phosphodiesterase inhibitors milrinone and amrinone.
Dopamine may be preferable in patients with low blood pressure. This is because inodilators dobutamine and milrinone reduce SVR. Milrinone has even greater vasodilating unloading properties. Milrinone was evaluated for short-term use in acute exacerbations of CHF in the OPTIME-CHF trial and was not shown beneficial.9 The primary objective of the Prospective Randomized Evaluation of Cardiac Ectopy with Dobutamine or Nesiritide Therapy (PRECEDENT) study was to compare the effects of nesiritide vs. dobutamine on heart rate and ventricular arrhythmias during the first 24 hours of ADHF treatment.10 Nesiritide did not aggravate preexisting ventricular tachycardia or frequency of premature ventricular beats. Dobutamine significantly increased ventricular tachycardia events. Both agents were equally effective in improving signs and symptoms of heart failure.
There are drawbacks to the catecholamine class. In ADHF patients, circulating catecholamine levels are already running high, thus high doses of dopamine or dobutamine are needed for the effect on "burned out" receptors. This can result in tachycardia, tachydysrhythmias, and ischemia due to increased myocardial O2 consumption. Tolerance can develop rapidly, requiring escalating dosages and, thus, the potential to produce more adverse effects.11
The phosphodiesterase inhibitors (milrinone) function independently of adrenoreceptor activity and plasma catecholamine level. Tolerance is not likely to develop. These agents decrease preload and afterload as well as provide inotropic support. There are seven studies that compared milrinone to dobutamine in patients with severe ADHF. Increases in stroke volume (SV) and cardiac index (CI) were similar for both agents but milrinone demonstrated greater reduction in PCWP (preload) and SVR (afterload), no increase in myocardial O2 consumption, and less tachycardia.12-15
Overall, the routine use of inotropes as HF therapy is not indicated in either the short- or long-term setting. Inotropes are potentially appropriate as treatment for cardiogenic shock, diuretic/ACEI-refractory HF decompensation, or as a short-term bridge to definitive treatment, such as revascularization or cardiac transplantation. Milrinone provides preload and afterload reduction in addition to inotropic support. This is advantageous for patients with borderline hypotension. To summarize inotropes: because of their association with increased mortality in nearly every study performed (OPTIME, PRECEDENT, SURVIVE, etc), inotropes generally are discouraged for routine use in HF (see HFSA guidelines). In the fewer than 2% of HF patients presenting to the ED with symptomatic hypotension, defined as SBP < 85 mmHg, they may be considered.
Diuretics. Diuretics are the mainstay in most patients being managed for HF. Loop diuretics such as furosemide (Lasix) reduce preload by removing total body fluid through diuresis. Furosemide 20-200 mg is administered intravenously to alleviate lung edema due to fluid overload, with the initial dose based on prior diuretic history.16 Subsequent dosing is determined by the diuretic response. This effect will have a 30-120 minute delay. Also, patients with chronic pulmonary edema may have a significantly reduced renal blood flow, approximately 20% of normal, due to increased afterload. The direct vasoactive effect (venodilation) has been proposed to reduce preload within 5-10 minutes. However, there are no convincing studies supporting an immediate effect.17
The decrease in preload via diuresis is a delayed effect. Diuretics also can sensitize patients to the hemodynamic effects of ACEIs by decreasing intravascular volume. Note that diuretics are not indicated as chronic monotherapy for ADHF. There is evidence to suggest that aggressive continuous IV infusion of furosemide can be administered safely and may shorten hospitalization. Howard and Dunn evaluated aggressive high-dose diuresis using continuous furosemide infusion in an elderly population with class IV HF.18
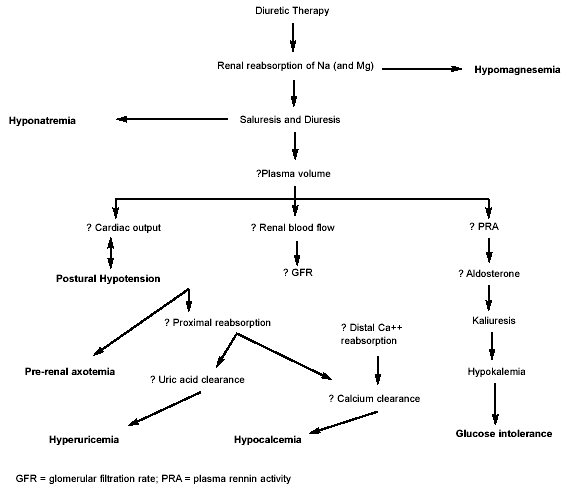
Complications from loop diuretics are well described. (See Figure 2.) Furosemide produces an initial activation of the sympathetic nervous system, which in turn increases heart rate, SVR, and myocardial oxygen demand. The result is decreased SV, CO, and tissue perfusion. Furosemide also produces initial activation of the renin-angiotensin system, not a favorable response in these patients. Loop diuretics also induce hypokalemia that can be quite severe in patients undergoing aggressive diuresis. Therefore, close monitoring or electrolytes and urinary response is needed. Finally, a widening QT interval on ECG may suggest hypocalcemia, hypokalemia, or hypomagnesemia, so check electrolytes in this event.
| Figure 2. Complication of Diuretic Therapy for Heart Failure |
|
Chronic diuretic use results in tachyphylaxis. Decreased perfusion of the kidney results in decreased delivery of loop diuretics. Decreased glomerular filtration rate results in decreased filtered load of sodium. Volume depletion activates plasma rennin activity and stimulates the sympathetic nervous system; elevated catecholamine levels lead to increased proximal tubule reabsorption of sodium as aldosterone stimulates increased distal reabsorption of sodium.
The two types of diuretic resistance are: 1) the "braking phenomenon" and 2) long-term tolerance. The braking phenomenon is the result of a decrease in response to a diuretic after the first dose has been administered. Long-term tolerance is the result of tubular hypertrophy compensating for salt loss. The end result in either case is diminished or lost diuretic response before resolution of edema has been achieved. Clinical presentation can include edema that persists despite compliance with diet and diuretic dose previously efficacious in maintaining euvolemia.
Vasodilators. Vasodilator therapy is an attempt to reduce preload and therefore capillary wedge pressure. If there is adequate blood pressure, vasodilators and diuretics are the treatment modalities. This is also the treatment option to consider initially in the patient with persistent hypertension.
The ADHERE registry has provided some information to guide ED therapy in patients at high risk for adverse outcomes.19 Outcomes based on the early use of intravenous vasoactive therapy, defined as any agent administered to obtain a hemodynamic affect, have been evaluated.20 Vasoactive agents included nitroglycerin, nesiritide, nitroprusside, dobutamine, dopamine, etc. This report included data from 46,599 patients analyzed to determine vasoactive usage. The comparison was between 4,096 patients who received IV vasoactive therapy in the ED (a mean of 1.1 hours after arrival) and 3,499 patients who received vasoactive therapy in the hospital (a mean of 22.2 hours after presentation). Outcomes between the two groups were significantly different. Early therapy was favored both statistically and clinically in parameters including mortality, ICU and overall length of stay, and use of invasive procedures.21
Nitroglycerin (NTG). NTG should be considered a first-line treatment choice for ADHF. Nitrates are given sublingually or intravenously for rapid, reliable preload as well as afterload reduction. There are multiple studies comparing NTG vs. morphine or furosemide for preload reduction, and NTG is clearly safer and faster.22 Moderate to higher doses (100-200 micrograms per minute) reduce SVR (afterload) as well, by maintaining or improving SV and CO. Another benefit of nitrates is the short half-life which limits the time of any adverse effects.
NTG is better and safer than morphine or furosemide to reduce preload. Sublingual (SL) NTG provides a rapid and effective means of initiating treatment, and can be followed by IV NTG if severe symptoms exist or if the patient is in extremis. Topical NTG can follow SL NTG for moderate symptoms. NTG can be used SL or intravenously in moderate to high doses (≥ 60 micrograms/minute). This is a very good agent for simultaneous preload and afterload reduction. Dosing can start at 10-20 micrograms/minute and can titrate quickly every 3-5 minutes to reach 200-300 micrograms/minute if needed to achieve desired effect.
Natriuretic Peptides. Nesiritide is a recombinant natriuretic peptide that causes vasodilation and increases renal blood flow and urine output. The result is rapid symptomatic and hemodynamic improvement in patients with ADHF.23 Nesiritide also exhibits lusitropic (relaxing) effects on cardiac muscle. Because of its vasodilation, nesiritide causes hypotension in a significant portion of patients. It is also quite costly.
Nesiritide has been shown to be capable of dose-related decreases in PCWP (preload) and SVR (afterload) as well as increases in CI.23 These are favorable effects in ADHF while also avoiding increases in heart rate or incidence of arrhythmias.
Nesiritide has been studied in 10 clinical studies. The Vasodilation in the Management of Acute HF (VMAC) study compared the addition of intravenous nesiritide or intravenous NTG to standard HF therapy.24 The VMAC study included "real world" ADHF patients; 84% were NYHA class III or IV, 21% had renal insufficiency (defined as serum creatinine > 2.0 mg/dL), 15% had preserved systolic function (ejection fraction > 40%), and 47% had diabetes. Administered as a 2 mcg/kg bolus followed by maintenance infusion of 0.01 mcg/kg/min, nesiritide resulted in decreased filling pressures and improvement in dyspnea compared to standard therapy (standard therapy could include IV diuretics, dobutamine, dopamine, and long-term therapies).
The previously mentioned PRECEDENT study looked at 255 randomized patients and the effect of nesiritide or dobutamine on ventricular arrhythmias in the treatment of ADHF. Nesiritide reduced or had a neutral impact on ventricular ectopy while dobutamine was associated with increased arrhythmias and increased heart rate.10
With the increased use of Observation Units (OU) for the treatment of patients with ADHF, the use of nesiritide in the OU setting has been studied in the Prospective Randomized Outcomes Study of Acutely Decompensated Congestive Heart Failure Treated Initially in Outpatients with Natrecor (PROACTION) trial. This was a randomized, double-blinded, placebo-controlled pilot study of 250 patients. Patients were treated with standard therapy at the discretion of the treating physician and received either nesiritide or placebo. Neither group was treated with nitroglycerin (considered a first-line treatment). Hypotension was more common in the nesiritide group and that led to a slight increase in adverse events over standard treatment (45% vs 37%; p = 0.235). There was a slight decrease in the need for hospitalization after observation in the nesiritide group (49% vs 555; p = 0.436). More patients who received standard therapy were readmitted within 20 days (23% vs 10%; p = 0.058). No renal function data were reported. Mortality within 30 days was 4% with nesiritide and 1% with standard therapy (p = 0.213).25
Two meta-analyses26,27 have reported a trend toward increased mortality and worsening renal function with nesiritide in ADHF patients. The Sackner-Bernstein mortality report is a selective analysis of only a portion of existing data.27 However, these adverse trends raise questions that warrant further investigation with a well-designed randomized control trial.
An expert panel of cardiology and heart failure clinicians chaired by Eugene Braunwald, MD, met in June 2005 to review and assess data associated with the use of nesiritide after questions of safety were raised.28 After an in-depth discussion of substantial additional analyses of existing data, the panel made the conclusions and recommendations in Table 3.
| Table 3. Braunwald Panel Report |
|
1. Use of nesiritide should be limited to patients presenting to the hospital with acutely decompensated HF who have dyspnea at rest. 2. Physicians considering the use of nesiritide should consider: its efficacy in reducing dyspnea, possible risks of the drug, availability of alternate therapies to relieve symptoms. 3. Nesiritide should not be used: to replace diuretics to enhance diuresis, for intermittent outpatient infusion, for scheduled repetitive use, to improve renal function. 4. The manufacturer (Scios) should inform physicians about acceptable nesiritide use. |
Nitroprusside–Vasodilation. Some patients with extremely elevated blood pressure or SVR require additional afterload reduction (beyond NTG). Nitroprusside (NTP) is a potent vasodilator that relaxes both arterioles and venous smooth muscle. This decrease in preload and afterload can improve CO and SV. An intravenous infusion starts at 0.1-0.2 microgram/kg/minute with titrated increases every 5 minutes.
There are potential adverse effects. Blood pressure can drop precipitously and must be monitored closely. This generally requires arterial pressure monitoring. Nitroprusside or any vasodilator can theoretically precipitate a "coronary steal" syndrome that can result from impaired flow to an area of a fixed coronary lesion. Patients with renal failure are at greater risk for thiocyanate toxicity, and reflex tachycardia can result from the vasodilation and in turn affect myocardial oxygen consumption.
Morphine. Morphine sulfate is considered an acceptable treatment choice for patients presenting in ADHF, and is a common addition to the NTG regimen for preload reduction. There is, however, surprisingly little evidence to support the use. Historically, morphine has been considered "perhaps the most important agent used in treatment of acute pulmonary edema."29 In fact, there is significant evidence that it can cause respiratory and myocardial depression at high doses. Its histamine-related side effects such as rash, urticaria, nausea, and vomiting can actually increase catecholamine release, which can increase afterload.17
The effect of preload reduction comes from studies done back in the 1970s in which morphine was noted to produce venodilation in the hands and forearms of patients with pulmonary edema.29 More recently, Peacock et al. performed a retrospective analysis using the ADHERE registry, stratifying patients by whether they received morphine at any time during their hospitalization.30 This is illustrated in Table 4. In ADHF, use of morphine was associated with longer hospitalizations, more frequent and longer ICU admissions, and a greater risk of mortality.
| Table 4. Overall Outcomes30 |
|
Morphine should not be considered as a first-line agent in the treatment of ADHF due to the absence of safety and efficacy data and because it has never been shown to improve outcomes in ADHF patients. Morphine can be considered to reduce patient anxiety and decrease the work of breathing, which in effect can theoretically diminish central sympathetic flow. Consider an initial dose of 2-4 mg over a three-minute period, to be repeated if necessary at five- to 15-minute intervals.
Angiotensin-Converting Enzyme Inhibitors (ACEIs). Initial activation of the RAAS is beneficial as a response to decreased cardiac output, but chronic activation leads to adverse hemodynamic consequences. Systemic vascular resistance increases in response to higher levels of angiotensin, and elevated aldosterone leads to sodium retention. The basis of most treatments shown to reduce mortality in chronic HF is the interruption of the RAAS. ACEIs block the pathway between angiotensin I and angiotensin II, which is a vasoconstrictor that also stimulates production of aldosterone, although some angiotensin II still is produced by ACE-independent pathways.
ACEIs reduce both arterial and venous resistance and lower the impedance to LV ejection of blood. Patients with symptomatic LV dysfunction and chronic heart failure derive a mortality reduction from ACEI therapy,31,32 and there are limited studies of severe chronic HF that demonstrate significant improvements in hemodynamics immediately after administration of an ACEI. These data suggest a potential ED role for acute administration of ACEIs.33-35 In a single small limited study, Hamilton, et al. performed a randomized double-blind controlled analysis in 48 patients with pulmonary edema.35 Each patient was treated with standard therapy consisting of NTG, furosemide, and morphine. In patients with systolic blood pressure (SBP) > 110, captopril 25 mg was given sublingually. A reduced dose of 12.5 mg SL was given if SBP was in the range of 90-110. They noted a clinically significant reduction in "stress scores" by 30 minutes as determined by four parameters: patient reported dyspnea, physician judgment of respiratory distress, diaphoresis, and level of bed elevation tolerance. Although more research is needed before ED use can be recommended, ACEIs provide a rapid reduction in afterload and preload, and a rapid reduction in subjective level of distress.
Relative contraindications to using ACEIs include orthostatic hypotension and elevated creatinine. A history of anaphylaxis is an absolute contraindication. Consider the likelihood that a patient has renal insufficiency if presenting to the ED in ADHF and chronic management does not include an ACEI.
Ultrafiltration in Heart Failure. This technique is provided in some hospital centers for ADHF patients with severe volume overload (> 15 lbs). Ultrafiltration requires central venous access and can remove fluid while maintaining intravascular fluid volume by filtering at the same rate as fluid is reabsorbed from the interstitium. Advantages include preservation of renal function, prevention of electrolyte imbalance, and a predictable and programmable amount of fluid removal. Furthermore, by not requiring diuretic use, the adverse effects of diuretics can be avoided.
The proposed sustained benefits of ultrafiltration include efficacy of sodium and water removal as compared to diuretics alone and reduced neurohormonal activation. It can provide a "diuretic holiday" allowing for increased diuretic responsiveness when resumed. Some hospital centers have established protocols for ADHF patients with volume overload and diuretic resistance.
Medications to Avoid in HF Patients
Nonsteroidal anti-inflammatory drugs (NSAIDs) should be avoided in patients with HF. Though not associated with a first occurrence of HF, it can cause worsening of existing HF.36-38 The suspected causal mechanism is an increase in afterload resulting from NSAID-induced systemic vasoconstriction. NSAID use also may blunt the renal effects of diuretics.
Potential interaction between NSAIDs and ACEIs can result in reduced renal perfusion and increased SVR. It is also recommended that calcium-channel blockers not be used for vasodilator therapy in patients with HF. ACEIs and beta-blockers are the preferential choices. Antiarrhythmics also have been shown to increase mortality.
Admission and Discharge Protocols
A significant component of disease management in HF patients concerns admission orders. Standardized orders provide consistent and complete care from the ED to the hospital floor, including medications, labs, ancillary test orders, and strict fluid management. In many ED settings, the patients can then spend 12-24 hours in the ED waiting for an available floor bed. Regardless of the hours spent in the ED prior to bed availability, the transition from the ED to the inpatient status must be seamless. These patients must receive appropriate treatment while care is transitioning from the ED physician to the admitting team.
Why standardized orders? They not only allow for objective data and objective findings for patients, but provide physicians with some autonomy and subjectivity because not every patient fits a protocol. Standing orders ensure guideline compliance from the literature while allowing for individual patient adjustments.
A patient discharge checklist may be helpful for patients being discharged from the ED or observation unit. This makes it easier for the physician and patient to follow a care plan. This also improves JCAHO compliance. This checklist is a method of ensuring those patients that meet criteria for specific interventions (i.e., medications, smoking cessation, cardiac rehabilitation) are given the appropriate medications and instructions upon discharge.
HF education is a critical component of disease management. HF is a complex disease process with a large social component, not only with follow-up, but also with dieting and medication compliance. A study in 1990 suggested that of the people readmitted to the hospital, almost half were due to diet and medication compliance alone.39
Unlike other acute disease processes such as pneumonia and pyelonephritis, ADHF exacerbations are treated until the subjects return to their baseline compensated state. The underlying disease process is never cured. Thus as a result, patient behavior after acute treatment may have a tremendous impact on the progression of the disease process and subsequent morbidity and mortality.40
Observation Units
Diagnostic workup and therapy targeted at ADHF, when provided in the ED observation unit, is associated with decreased ED revisits at one week and one month without an increase in mortality.91 Despite the recent advancements in therapeutic options that have improved the prognosis for many heart failure patients, ADHF episodes still occur. Timing to rapid symptomatic and hemodynamic stabilization is critical. (See Table 5.)
| Table 5. Improving Treatment of ADHF |
|
The final figure is for reference and can be applied to any ED setting. Figure 3 represents an algorithm to stratify patients to an observation unit. This algorithm was developed and currently is utilized to identify patients appropriate for the observation unit at the Cleveland Clinic.41 The entry criteria, exclusion criteria, and discharge criteria are identified here. This decision tree is the result of a coordinated effort with both the ED and the cardiology department. This diagnostic and therapeutic algorithm, along with aggressive monitoring by nursing staff, detailed patient education, cardiology consultation, and follow-up arrangements at time of discharge, comprise the complete observation unit treatment protocol.
Summary of the Treatment of Acute Decompensated Heart Failure
The ED remains the most important entry point for patients with HF. Short-term management decisions affect long-term outcome. Evidence suggests that rapid initiation of therapy shortens total and ICU length of stay (LOS). Collaboration between in-hospital and ED physicians is needed to optimize care of heart failure.
Refer back to Figure 1, which categorizes patients based on perfusion status and volume status. A majority of these patients are "warm and wet." Treatment strategy is based on risk—80% of patients will fall into intermediate risk, with 10% low risk, and 10% high risk. A majority of these patients do well because more than 50% of these admissions are due to medication noncompliance. The majority of these patients will do well with nitrates, furosemide, and adjustment of oral medications. The higher risk patient, however, needs early administration of IV vasodilator therapy.
Most patients are "warm and wet" and not in cardiogenic shock. High BUN, low systolic pressures, and high creatinine are an indication of higher disease severity. So treatment based on low, moderate, or critical severity in warm and wet patients depends on the ancillary data.
Chronic Management
Heart failure is not curable. Patients are maintained on a variety of medications to optimize their cardiac function and fluid status. Digoxin affects the Na+/K+ -ATPase pump in the myocardial cell, increasing contractility. Once the mainstay of treatment, most patients now are maintained without the use of digoxin. Diuretics decrease preload by stimulating natriuresis in the kidneys. Beta-blockers inhibit the sympathetic nervous system and adrenergic receptors. Although they once were considered contraindicated in heart failure, if patients can tolerate them, they can be very helpful. Aldosterone antagonists counteract the many effects of aldosterone. Inotropes such as dobutamine and milrinone increase myocardial contractility, and may be infused periodically as an outpatient. These agents slow heart rate, decrease BP, and have a direct beneficial effect on the myocardium, enhancing reverse remodeling.
Vasodilator therapy such as combination therapy with hydralazine and isosorbide dinitrate decreases afterload by counteracting peripheral vasoconstriction. The combination of hydralazine and isosorbide dinitrate, used long-term, was associated with a higher mortality rate than enalapril (25% vs 18% in 2 years) in the second VA Cooperative Heart Failure Trial (V-HeFT II).42 Selected agents that also block the alpha-adrenergic receptors can cause vasodilatation. Cardiac resynchronization therapy and biventricular pacing improves LV function and favors reverse remodeling. Nesiritide has been used to decrease preload by stimulating diuresis and decrease afterload by vasodilatation.
Angiotensin II Receptor Blockers. ARBs serve to antagonize the RAAS by preventing the effect of angiotensin at the receptor.
The ARBs' therapeutic effects are secondary to their ability to block the angiotensin receptor. Patients who have failed ACEIs due to secondary symptoms such as cough or due to angioedema are considered for ARB therapy. Though the efficacy of ARBs is similar to that of ACEIs, they are not superior and are not considered first-line agents for chronic HF.43
Beta-Blockers. Once contraindicated, beta-blockers counteract the harmful effects of the sympathetic nervous system and are currently advocated for chronic treatment in HF. Several studies suggest that the use of beta-blockers can reduce mortality and hospitalizations.44,45 All patients with HF should be considered for beta-blocker therapy, except in those situations listed in Table 6.
| Table 6. Contraindications to Beta-Blockers |
|
• Unstable hemodynamics, or congested and requiring IV diuresis • Severe bronchospastic airway disease • Symptomatic bradycardia • Advanced heart block • Acute vascular insufficiency or worsening claudication/rest pain • Class IV stable HF (therapy should be provided by HF specialist) • Inotropic therapy/cardiogenic shock • Severe conduction system disease (unless protected by pacemaker) |
Spironolactone. In chronic HF, aldosterone levels are elevated as a result of stimulation by angiotensin II and decreased hepatic clearance. Aldosterone not only causes more retention of salt and water by kidneys, but also has the deleterious effects in blood vessels and myocardium. Conversely, blocking its receptor with spironolactone or eplerenone has pleiotropic beneficial effects, improving endothelial function and reducing inflammation, collagen synthesis, and thrombosis.
Spironolactone, an aldosterone blocker, was used and evaluated in patients with NYHA III or IV HF in the Randomized Aldactone Evaluation Study. This study demonstrated a 30% reduction in mortality and hospitalization.46 Hyperkalemia is a potential side effect, thus spironolactone should be avoided in patients with renal insufficiency or hyperkalemia. Spironolactone is recommended in NYHA III or IV HF patients for chronic outpatient management.
Future Diagnostics
Thoracic electrical bioimpedance (TEB) is a new noninvasive technology that can provide information about cardiac output and contractility and thoracic fluid content.4 This technology was first developed by the National Aeronautics and Space Administration in the 1960s.47 The field of impedance cardiography (ICG) is the study of waveforms derived from TEB. Recently, ICG has been used in the ED to evaluate potentially unstable patients.
In patients presenting with shortness of breath as a primary complaint, the ED-IMPACT trial demonstrated that information obtained through ICG monitoring changed diagnosis and treatments 5.3% and 23.6% of the time, respectively.48 Information collected from ICG monitoring can help steer goal-directed therapy of ADHF and monitor results of therapeutic interventions or any changes in clinical condition.49
Future Therapeutics
Arginine Vasopressor Antagonists. Arginine Vasopressin (AVP) is a powerful vasoconstrictor and promotes water reabsorption by the kidneys. Elevated levels of AVP have been reported in chronic HF. Early trials of V1 receptor anatagonists have demonstrated a favorable hemodynamic response with an increase in urine output.50
Calcium Sensitizers (Levosimendan). Levosimendan is a new agent that sensitizes troponin C to calcium. This works independently of adrenoreceptor activity and plasma catecholamine levels. Compared with dobutamine in patients with severe low output CHF, levosimendan initially demonstrated more significant improvements in CO and PCWP and lower 180-day mortality (26% vs 38%).51 More recently, levosimendan was associated with similar mortality as related to dobutamine.
Aldosterone Blockers (Eplerenone). Eplerenone, a selective aldosterone blocker, was evaluated in the eplerenone post-acute myocardial infarction HF efficacy and survival study (patients with LV dysfunction).52 Patients who received eplerenone had reduced mortality, hospitalizations, stroke, arrhythmias, and HF.
Summary
Acute decompensated heart failure is a common presentation to the ED, and volumes of these patients are growing. The first step required for effective and accurate therapy is to make an accurate diagnosis in the ED.
• Traditional diagnostic modalities often are inaccurate. Add BNP or NT-pro BNP testing to the physical exam findings, CXR, and ECG, especially when the diagnosis is unclear.
• Treatment should be based on the concept of fluid redistribution; ADHF patients may be hypervolemic, euvolemic, or hypovolemic. ADHF does not equal volume overload.
• NTG should be the first-line agent and do not hesitate to titrate to effective doses. SL NTG is easy to use and start with, and IV NTG is a good single agent.
• There is preliminary data that suggests a potential ED role for the acute administration of ACEIs. In those studies, patients were treated with captopril 12.5 mg to 25 mg po or sublingual, or enalaprilat 1.25 IV. Onset of action was 15-30 minutes. Caution should be used when treating patients with a systolic blood pressure of less than 90 mmHg. While there is anecdotal evidence to support the use of ACEIs in acute heart failure, there needs to be more evidence it can be recommended as standard of care.53-56
• Furosemide is to be used after initiating preload and afterload reduction.
• Morphine—no good indication to continue using it in any ADHF algorithm. There is no proven benefit and certainly potential for harm. NTG is a better choice to reduce preload and benzodiazepines are better for anxiolysis.
• Nesiritide may be useful in the inpatient setting such as telemetry or the observation unit. There remains controversy over it effect on mortality and renal function. More studies are pending.
• Routine use of inotropes as HF therapy is not indicated in either short- or long-term setting. Milrinone is better than dobutamine from hemodynamic standpoint. Mortality rates are similar for all inotropes. The role of levosimendan will be based on future studies. Inotropes may be appropriate as palliative measure in patients with end-stage HF and/or hospice care.
• Consider early use of noninvasive positive pressure ventilation. It produces more rapid improvement, may avoid an intubation, and has been shown to decrease hospital lengths of stay and hospital costs.45
• Most patients who present to the ED with ADHF will require admission to telemetry or an observation unit.
• With proper patient selection by risk stratification, patients admitted to an observation unit are associated with decreased readmission rates with no proven increase in mortality.
• The greatest negative prognostic factors are renal dysfunction and low BP.
References
1. DiDomeniko RJ, Park HY, Southworth MR, et al. Guidelines for acute decompensated heart failure treatment. Ann Pharmacothr 2004;38:649-660.
2. Saltzberg MT. Beneficial effects of early initiation of vasoactive agents in patients with acute decompensated heart failure. Rev Cardiovasc Med 2004;5(suppl 4):17-27.
3. Blomkalns AL, Gibler WB. Emergency Diagnosis and Treatment of Acute Decompensated Heart Failure (ADHF). CME Monograph, Mar 2005:13-14.
4. Peacock WF, Tiffany B. Cardiac Emergencies. Congestive Heart Failure: Demographics and Economics. The McGraw-Hill Companies, Inc. 2006;297.
5. Silver MA, Maisel A, Yancy CW, et al. BNP Consensus Panel 2004: A clinical approach for the diagnostic, prognostic, screening, treatment monitoring, and therapeutic roles of natriuretic peptides in cardiovascular diseases. Congest Heart Fail 2004;10:53,1-36.
6. Johnson K. Boost nitroglycerin for cardiogenic PD. ACEP News 2005;(24;8); 1, 14.
7. Peacock WF, Emerman, Blicker JZ, et al. Noninvasive ventilation: Friend or foe of the failing heart. Acad Emerg Med 2005;12suppl1:55.
8. Pong D, Kernan SP, Cook DJ, et al. The effects of positive pressure airway support on mortality and the need for intubation in cardiogenic pulmonary edema. Chest 1998;114:1185-1192.
9. Cuffe MS, Califf RM, Adams KF Jr, et al., for the Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CNF) Investigators. Short-term intravenous milrinone for acute exacerbation of chronic heart failure: A randomized controlled trial. JAMA 2002;287:1541-1547.
10. Burger AJ, Horton DP, LeJemtel T, et al. Effect of nesiritide (B-type natriuretic peptide) and dobutamine on ventricular arrhythmias in the treatment of patients with acutely decompensated congestive heart failure: The PRECEDENT study. Am Heart J 2002;144:1102-1108.
11. Stevenson SW. Clinical use of inotropic therapy for heart failure: Looking backward of forward? Circulation 2003;108:367-372.
12. Shipley JB, Tolman D, Hostillo A, et al. Milrinone: Basic and clinical pharmacology and acute and chronic management. AM J Med Sci 1996;311:286-291.
13. Travill CM, Pugh S, Noble MI. The inotropic and hemodynamic effects of intravenous milrinone when reflex adrenergic stimulation is suppressed by beta-adrenergic blockade. Clin Ther 1994;16:783-792.
14. Varriale P, Ramaprasad S. Short-term intravenous milrinone for severe CHF: The good, bad, and not so good. Pharmacotherapy 1997;17:335-337.
15. Yamani MH, Haji SA, Starling RC, et al. Comparison of dobutamine-based and milrinone-based therapy for advanced decompensated congestive heart failure: Hemodynamic efficacy, clinical outcome, and economic impact. Am Heart J 2001;142:998-1007.
16. Sharis PJ, Connor CP. Evidence Based Cardiology. Philadelphia: Lippincott Williams & Wilkins; 2003:290-291.
17. Mattu A. Heart Failure: Advances in Diagnosis and Treatment. ACEP Scientific Assembly 2005, Washington, DC.
18. Howard P, Dunn M. Aggressive diuresis for severe heart failure in the elderly. Chest 2001;119:807-810.
19. Peacock WF. Heart failure in the emergency department. Critical Pathways in Cardiology: A Journal of Evidence-Based Medicine 2003;2:207-220.
20. Peacock WF, Emerman CL, Costanzo MR, et al. Early initiation of intravenous therapy improves heart failure outcomes: An analysis from the ADHERE registry database. Ann Emerg Med 2003;42:S26. Research Forum Abstract.
21. Peacock WF, Emerman CL, Costanzo MR, et al. Early indication of intravenous therapy improves heart failure outcomes: An analysis from the ADHERE Registry Database. Ann of Emerg Med 2003;42:526.
22. Beltrame JF, Zeitz CJ, Unger SA, et al. Nitrate therapy is an alternative to furosemide/morphine therapy in the management of acute cardiogenic pulmonary edema. J Card Fail 1995;4:271-279.
23. Colucci WS, Elkayam U, Horton DP, et al, for Nesiritide Study Group. Intravenous nesiritide, a natriuretic peptide, in the treatment of decompensated congestive heart failure. N Eng J Med 2000;343:246-253.
24. Publication Committee for the VMAC Investigators (Vasodilatation in the Management of Acute CHF). Intravenous nesiritide vs. nitroglycerin for treatment of decompensated congestive heart failure: A randomized controlled trial. JAMA 2002;287:1531-1540.
25. Peacock WF IV, Holland R, Gyarmathy R, et al. Observation unit treatment of heart failure with nesiritide: Results from the PROACTION trial. J Emerg Med 2005; 29:243-252.
26. Sackner-Bernstein JD, Skopicki WA, Aaronson KD. Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure. Circulation 2005;11:1487-1491.
27. Sackner-Bernstein JD, Kowalski M, Fox M, et al. Short-term risk of death after treatment with nesiritide for decompensated heart failure. A pooled analysis of randomized controlled trials. JAMA 2005;293:1900-1905.
28. Scios web site. Panel of cardiology experts provides recommendations to Scios regarding Natrecor. June 13, 2005. Available at www.sciosinc.com/scios/pr_1118721302.
29. Peacock WF, Hollander JE, Diercks DB, et al. Morphine for acute decompensated heart failure: Valuable adjunct or historical remnant? Acad Emerg Med 2005;12(suppl 1):97.
30. Peacock WF, Hollander J, Diercks D, et al. Morphine and outcomes in acute decompensated heart failure: An ADHERE analysis. Publication pending. Personal communication.
31. [No authors listed.] Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The Consensus Trial Study Group. N Engl J Med 1987;316:1429-1435.
32. [No authors listed.] Effects of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med 1991;325:293-302.
33. Nande M, Erbel R, Meyer J. Sublingual administration of captopril versus nitroglycerin in patients with severe congestive heart failure. Int J Cardiol 1990;27:351-359.
34. Annane D, Bellissant E, Passord E, et al. Placebo-controlled randomized double-blind study of intravenous enalapril efficacy and safety in acute cardiogenic pulmonary edema. Circulation 1996;94:1316-1324.
35. Hamilton RJ, Carter WA, Gallagher EJ. Rapid improvement of acute pulmonary edema with sublingual captopril. Acad Emerg Med 1996;3:205-212.
36. Amabile CM, Spencer AP. Keeping your patient with heart failure safe: A review of potentially dangerous medications. Arch Intern Med 2004;164:709.
37. Feenstra J, Heerdink ER, Grobbee DE, et al. Association of nonsteroidal anti-inflammatory drugs with first occurrence of heart failure and with relapsing heart failure: The Rotterdam Study. Arch Intern Med 2002;162:265.
38. Mamdani M, Juurlink DN, Lee DS, et al. Cyclo-oxygenase-2 inhibitors versus non-selective nonsteroidal anti-inflammatory drugs and congestive heart failure outcomes in elderly patients: A population-based cohort study. Lancet 2004;363:1751.
39. Rich MW, Vinson JM, Sperry JC, et al. Early readmission of elderly patients with congestive heart failure. JAGS 1990;38:1290-1295.
40. Summers RL. Disease Management of Acute Decompensated Heart Failure: The ADHERE Emergency Medicine Module. Emerging Diagnosis and Treatment of ADHF. CME Monograph from ACEP 2005 Spring Congress Satellite Symposium. Emergency Medicine Cardiac Research and Education Group International Mar 2005:33-38.
41. Peacock WF, Young J, Collins S, et al. Heart failure observation units: Optimizing care. Cardiology/Review Article. Ann Emerg Med 2006;47:22-33.
42. Cohn JN, Johnson G, Ziesche S, et al. V-HeFT II. A comparison of enalapril with hydralazine-isosorbide dinitrate in the treatment of chronic congestive heart failure. N Engl J Med 1991;325:303-310.
43. Pitt B, Poole-Wilson PA, Segal R, et al. Effect of losartan compared with captopril on mortality in patients with symptomatic heart failure: Randomised trial—the Losartan Heart Failure Survival Study ELITE II. Lancet 2000;355:1582-1587.
44. [No authors listed.] The Cardiac Insufficiency Bisoprolol Study II (CIBIS II): A randomized trial. Lancet 1999;353:9-13.
45. [No authors listed.] Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL randomized intervention trial in congestive heart failure (MERIT-HF). Lancet 1999;353:2001-2007.
46. Pitt B, Zannad F, Remme J, et al. Randomized Aldactone Evaluation Study (RALES) Investigators. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med 1999;341:709-719.
47. Summers RL, Shoemaker W, Peacock WF, et al. Bench to bedside: Electrophysiologic and clinical principles on non-invasive hemodynamic monitoring using impedance cardiography. Acad Emerg Med 2003;10:669-680.
48. Peacock WF, Summers RL, Emerman CL. Bioimpedance monitoring changes therapy in dyspneic emergency department patients: The IMPACT Trial. Ann Emerg Med 2003;42:582.
49. Noble TJ, Harris ND, Markie AH, et al. Diuretic induced change in lung water assessed by electrical impedance tomography. Physiol Meas 2000;21:155-163.
50. Udelson JE, Smith WB, Hendrix GH, et al. Acute hemodynamic effects of conivaptan, a dual V(1A) and V(2) vasopressin receptor antagonist in patients with advanced heart failure. Circulation 2001;104:2417-2423.
51. Fullath F, Cleland JG, Just H, et al. Efficacy and safety of intravenous Levosimendan compared with dobutamine in severe low-output heart failure (the LIDD Study): A randomized double-blind trial. Lancet 2002;360:196-202.
52. Pitt B, Renme W, Zannad F, et al. Eplerenone, a selective aldosterine blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 2003;348:1309-1321.
53. Annane D, Bellissant E, Pussard E, et al. Placebo-controlled, randomized, double-blind study of intravenous enalaprilat efficacy and safety in acute cardiogenic pulmonary edema. Circulation 1996;94:1316-1324.
54. Hamilton RJ CW, Gallagher EJ. Rapid improvement of acute pulmonary edema with sublingual captopril. Acad Emerg Med 1996;3:205-212.
55. Barnett JC ZK, Touchon RC. Sublingual captopril in the treatment of acute heart failure. Curr Ther Res 1991;49:274-281.
56. Sacchetti A, Ramoska E, Moakes ME, et al. Effect of ED management on ICU use in acutepulmonary edema. A J Emerg Med 1999;17:571-574.
The treatment of acutely decompensated heart failure (ADHF) has changed dramatically in the past decade. As discussed in Part I, there is a new understanding of the pathophysiology and a significant improvement in our ability to diagnose heart failure with BNP and NT-pro BNP testing.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.