Pediatric Hypertension: What Is It and When Does It Need to be Treated?
Pediatric Hypertension: What Is It and When Does It Need to be Treated?
Authors: Raemma Paredes Luck, MD, Assistant Professor, Pediatric Emergency Medicine, Temple University School of Medicine, Philadelphia; Patricia F. Campbell, MD, Assistant Professor, Pediatric Emergency Medicine, Temple University School of Medicine, Philadelphia.
Peer Reviewer: Larry B. Mellick, MD, MS, FAAP, FACEP, Medical Consultant, FBI Academy, Quantico, Virginia; Professor, Departments of Emergency Medicine and Pediatrics, Medical College of Georgia, Augusta
Hypertension is a very common disease affecting up to 20-25% of adults in the United States.1,2 In the past, hypertension has been considered a rare disease in children; however, in the last 10 years it has emerged as an important disease in pediatrics as well. Because hypertension is a risk factor for cerebrovascular disease, cardiovascular disease, as well as renal disease,3 the increase in pediatric hypertension is a concern because of its potential for long term sequelae. Epidemiologic studies have confirmed the link between obesity and hypertension;4 both are becoming significant health problems in children. The prevalence of obesity in adolescents has more than doubled since 1960,5 and currently 14% of 12- to 19-year-olds are overweight.6 The incidence of systolic hypertension is three times higher in obese adolescents compared with their normal weight counterparts.7 The association between hypertension and metabolic syndrome (obesity, insulin resistance, and dyslipidemia) is being recognized. Because blood pressure (BP) tends to increase with age, hypertension in children may result in significant public health problems as these patients mature into adulthood.8 This article reviews pediatric hypertension, highlighting the unique features and etiologies by age group.
The Editor
Definition
Defining hypertension in children is problematic. Blood pressure measurements are a continuum, with increasing risk for complications from the lower to higher levels.9 In the adult population, the definition of hypertension is based upon data focused on the long-term health sequelae of essential hypertension. Research in adults reveals that for every increase of 20 mm Hg in systolic blood pressure (SBP) and 10 mm Hg in diastolic blood pressure (DBP) starting at 115/75 mm Hg, the risk of cardiovascular disease doubles.10 Unlike adults, hypertension in pediatrics is defined based upon the normative distribution of blood pressure in healthy children. The National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents (NHBPEP), armed with recent data from the National Health and Nutrition Examination Survey (NHANES), recently published the Fourth Report on the Diagnosis, Evaluation, and Treatment of High Blood Pressure in Children and Adolescents. This report provides an update and recommendations to the clinician, incorporating the rapid advances in the detection, evaluation, and management of hypertension in the pediatric population.11
Normal blood pressure is defined as blood pressure that is less than 90th percentile for gender, age, and height. Prehypertension is defined as the average BP that is more than 90th percentile but less than 95th percentile. As in adults, adolescents with a BP reading that is 120/80 mm Hg or higher are considered prehypertensive. Hypertension is defined as the average systolic or diastolic blood pressure that is greater than or equal to the 95th percentile for gender, age, and height on at least three separate occasions. It is further divided into Stage 1 and Stage 2 to indicate severity (Table 1). This article will focus on hypertensive urgencies and hypertensive emergenciesthe two most common presentations of Stage 2 hypertension in the ED. For more information about the BP charts, please visit the official Web site: http://www.nhlbi.nih.gov/guidelines/hypertension/child_tbl.htm.
| Table 1. Classification of Hypertension in Pediatrics11 |
|
|
In infants younger than 1 year, hypertension is defined by SBP alone (Table 2).11,12 Because routine blood pressure measurement has not been recommended in this age range due to the technical difficulty in obtaining an accurate measurement13,14 and the low incidence of hypertension in healthy newborns, there is less normative data available.15
| Table 2. Blood Pressures in Infants12 |
|
|
These definitions for hypertension have been adopted for both primary and secondary hypertension. It is important to note that the data used in the BP tables were derived from epidemiological methods and, hence, may not reflect the actual physiologic effects of longstanding hypertension on the individual.16 Serious complications of secondary hypertension (i.e., encephalopathy) can occur at relatively low blood pressure values,17 underscoring the artificial nature of this definition.
It is important to confirm elevated BP readings on multiple occasions before diagnosing hypertension. BP variability is greater in children,18 and elevated BP levels tend to fall on subsequent measurement in this population.11 Thus, an isolated elevated BP reading is less likely to represent true pathology.18 Except in the situation of severe hypertension or evidence of end-organ damage, BP should be characterized over a period of time (weeks to months).11
Severe hypertension is categorized further to identify those patients who are at higher risk of immediate complications:
Hypertensive urgency is defined as severe hypertension that may be associated with nonspecific symptoms, but without evidence of target organ injury.11,12 This condition usually develops over a period of days and weeks.
Hypertensive emergency is defined as severely elevated blood pressure associated with rapid, progressive damage to end organs such as the heart, kidney, brain, and large arteries.11,12 The absolute level of hypertension is not as important as the evidence of complications in other organ systems.12 Conditions that are more likely to cause hypertensive emergencies are listed in Table 3.
| Table 3. Life-threatening Hypertension11,12 |
|
|
Epidemiology of Pediatric Hypertension
The prevalence of sustained high BP in childhood is 1-2%.11,15 The incidence of hypertension in neonates varies from 0.2% in healthy newborns19 to 2.6% in patients discharged from the neonatal intensive care unit.20 Some children do not have sustained hypertension, but will have an exaggerated BP response to stressors such as exercise and mental challenge.21,25 This sympathetic nervous system reactivity is a risk for developing essential hypertension26 and can be used as an early physiologic marker.
Essential hypertension is being recognized with more frequency in children. In fact, most children with hypertension have essential hypertension, lacking another cause for their elevated BP. The rising prevalence and increasing severity of obesity is associated with an increasing prevalence of essential hypertension in the pediatric population.27 The metabolic syndrome, also associated with the increasing severity of obesity, is a constellation of cardiovascular risk factors that include insulin resistance, abnormal glucose tolerance, dyslipidemia, and high BP.28 The syndrome is present in much higher rates in hypertensive children than their normotensive counterparts28 and is present in 30% of overweight patients.29 The presence of the metabolic syndrome in a patient represents a higher risk for cardiovascular and metabolic diseases such as type 2 diabetes mellitus and atherosclerosis.30
Children who suffer from sleep apnea associated with hypoxia have elevated BP during wakefulness that is independent of their degree of obesity.31 Hence, a brief sleep history to identify sleep disorders should be obtained in a child who has hypertension.11
Secondary hypertension is more common in children than in adults.11 These children are usually younger, have much higher BP readings,15 and are more likely to have both systolic and diastolic hypertension. In contrast to the children with essential hypertension, children with secondary hypertension have an associated high morbidity and mortality. The prevalence of secondary hypertension in childhood is about 0.1%. Renal abnormalities account for approximately 60-70% of cases of secondary hypertension, with reflux nephropathy and obstructive uropathy the most common causes. Another 10% of cases are caused by renovascular disease. All other causes of secondary hypertension account for the remaining 20-30% of cases.15 The mechanism causing secondary hypertension varies depending upon the primary disease process, such as vascular obstruction in coarctation, volume overload in renal diseases and mineral corticoid excess, hyper-reninemia in renovascular disease, and catecholamine excess in pheochromocytoma.32 Table 4 shows the most common causes of hypertension by age group.
| Table 4. Most Common Causes of Hypertension in Children12,30,33,35 |
|
|
Pathophysiology of Pediatric Hypertension
Blood pressure is the result of the interaction of two main factors: cardiac output and peripheral vascular resistance. An increase in one factor while the other factor remains unchanged will result in an increase in blood pressure. The renin-angiotensin-aldosterone system plays an important role in the development of high blood pressure by affecting peripheral vascular resistance. Renin is an enzyme produced in the kidney that converts angiotensinogen to angiotensin I. Its production is affected by arteriolar perfusion of the kidneys and the sympathetic nervous system, among other things. Angiotensin I is converted rapidly to angiotensin II by the angiotensin-converting enzyme (ACE). Angiotensin II is a potent vasoconstrictor that stimulates the production of aldosterone, another powerful vasoconstrictor. Both of these substances increase the blood pressure by increasing vascular resistance. Other factors that influence blood pressure include the activity of the autonomic nervous system, renal regulation of sodium, and compliance of both resistance and capacitance vessels.12,33
The pathophysiologic mechanisms causing acute hypertensive emergencies are complex and not well understood. The initiating step is a sudden rise in BP and the activation of the renin-angiotensin-aldosterone system.12,33-35 The rate of rise in BP determines the clinical presentation. Organs that are at greatest risk for injury are the brain, heart, and the kidneys. An increase in afterload causes left ventricular dysfunction, an increase in oxygen demand, and decreased coronary blood flow. Hypertension causes decreased flow to the kidneys as manifested by a deterioration of renal function. Hypertensive encephalopathy is a constellation of signs and symptoms of severe hypertension due to hyperperfusion of the brain. Normally, brain perfusion is controlled by autoregulatory processes over a wide range of mean arterial pressure (MAP), usually between 60 and 125 mm Hg. In chronically hypertensive patients, this steady state perfusion is shifted to the right so that the cerebral blood flow is maintained at a higher MAP, usually from 80 to 160 mm Hg. As the MAP increases, cerebral vessels vasoconstrict to maintain blood flow and protect the brain. However, a rapid increase in mean arterial pressure can overwhelm the steady state autoregulation of cerebral blood flow. The loss of autoregulation and disruption of the blood brain barrier permits excessive blood flow to the brain. This leads to cerebral edema and a resultant increase in intracranial pressure, hemorrhage, and eventually death. The retinal changes usually mirror the changes in the brain as encephalopathy progresses. Eye findings include areas of arteriolar vaso-spasm and dilatation, hemorrhage, exudates, and papilledema. Symptoms of hypertensive encephalopathy include severe headache, vomiting, seizures, lethargy, confusion, or coma.12, 33-35 Beneath this simplified approach are other dynamic processes that are just now being elucidated. Although beyond the scope of this discussion, substances such as oxygen free radicals, prostacyclin and nitric oxide secretion of the endothelium, proinflammatory cytoxines, and their interaction with the renin-angiotensin-aldosterone system, and a host of other substances are changing the way we understand the development of hypertension and hypertensive emergencies.
Evaluation of the Pediatric Patient with Elevated Blood Pressure
Measuring the Blood Pressure. The latest report from the NHBPEP recommends a BP measurement in patients 3 years and older presenting to a health care setting.11 In addition, children younger than 3 years with special conditions also need to have their BP checked (Table 5). During the triage process, a child with any of these conditions should have his/her BP measured, even if he/she has come to the ED with an unrelated complaint.
| Table 5. Special Conditions That Warrant A BP in Children Younger than 3 Years11 |
|
|
Accurate BP measurements are particularly problematic in children. Ideally, the patient should be relaxed, in a quiet room, at a comfortable temperature, and have a short period of rest (approximately 5 minutes) prior to the BP measurement. The patient should be sitting with his/her back supported, feet on the floor, and the right elbow supported so that the cubital fossa is at heart level.11 Unfortunately, these conditions rarely are met in the ED.
Blood pressure is a variable hemodynamic phenomenon that can be influenced by many factors that can cause elevations of 20-30 mm Hg. Anxiety can raise the BP due to the "fight or flight" phenomenon resulting in "white coat hypertension." The degree of reaction varies greatly from patient to patient and may be minimized or abolished by reassurance. These patients are only hypertensive in the medical environment.36
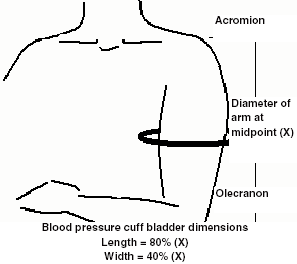
Accurate BP measurement requires an appropriately sized cuff (BPC) (Figure 1). The BPC bladder width-to-length ratio ideally should be 1:2. The width of the BPC bladder should encircle at least 40% of the arm measured midway between the olecranon and acromion. The length of the BPC bladder should cover at least 80% of the circumference of the arm. It is necessary to have several BPC sizes in the ED to accommodate patients from infancy to adulthood. Using a BPC with a bladder that is either too narrow or too short (undercuffing) will result in an artificially high BP reading, while using a BPC with a bladder that is either too wide or too long (overcuffing) may cause an artificially low BP reading. If the appropriate BPC size is not available, use the next larger size, even if it appears too large.11,37
| Figure 1. Sizing the Blood Pressure Cuff Bladder |
|
|
Measuring the BP by auscultation is the preferred method; all the normative data were obtained by this method. The mercury sphygmomanometer has been the gold standard device for measuring BP.11 However, oscillometric devices that measure BP are very popular in the ED because they are easy to use and minimize observer bias. It is the preferred method for measuring BP in newborns and young infants, and in situations requiring multiple, frequent BP measurements.11,33 Oscillometric devices measure the MAP and use algorithms to calculate systolic and diastolic values. Any elevated BP readings obtained by an oscillometric device must be confirmed by auscultation.11
History. The history should be geared toward identifying risk factors for secondary hypertension and co-morbidities in essential hypertension.
Birth history can be very significant. Premature neonates have unique risk factors. Steroids used in the treatment of chronic lung disease can cause elevated blood pressures for up to two weeks after the medication has been discontinued.39 Umbilical catheterization is complicated by thromboembolic events causing hypertension, usually presenting within two weeks of catheterization.41 Infants with apnea of prematurity may be treated with sympathomimetic medication. Independent of prematurity, a history of low birth weight is associated with hypertension.42,43 In addition, some factors in the prenatal environment may contribute to developing hypertension: For example, 50% of infants with a history of prenatal cocaine exposure develop hypertension by the age of 1 year.44
The medical history should be explored for evidence of renal disease both diagnosed and occult; renal pathology accounts for the majority of cases of secondary hypertension. Often the history may seem benign, but the patient can have significant renal scarring. Also, a history of congenital heart diseaseeven if it has been surgically repairedcan be significant. Most cases of coarctation of the aorta are diagnosed and repaired in the neonatal period; however, life expectancy remains reduced for these patients. Approximately one third will remain hypertensive or redevelop hypertension,3,45 and the hypertension is independent of any residual obstruction.46
The family history should explore inheritable causes of secondary hypertension as well as evidence to support the diagnosis of essential hypertension. Patients with tuberous sclerosis may have very subtle manifestations and remain undiagnosed. A family history of tuberous sclerosis is helpful, although most new cases are sporadic in nature.47 Neurofibromatosis has been associated with pheochromocytoma in multiple endocrine neoplasm Type 2b or Type 3;48 however, renal vascular lesions are much more common, and renal vascular stenosis is the most common etiology of hypertension in these patients.49
Many medications can affect blood pressure. Patients may not recognize over-the-counter medications or dietary supplements as medications because they have not been prescribed by a physician. Over-the-counter cold-and-cough preparations are a common source of sympathomimetic medications, including pseudoephedrine nasal sprays and drops that may artificially elevate BP. Always obtain a detailed medication history including dietary supplements, especially those which are purported to enhance athletic performance or induce weight loss.11 In addition to self-medication, older children and adolescents may abuse prescription drugs. Methylphenidate use increased 2.5 fold in the early 1990s.50 Nonmedical use of methylphenidate in high school seniors increased from 0.1% in 1992 to 2.8% in 1997.51 Most adolescents who are abusing methylphenidate were not prescribed the medication; they are abusing medication that has been diverted from therapeutic use.52 Another trend among school age children is the use of caffeine or caffeinated beverages. As little as 100 mg of caffeine can elevate the blood pressure in African American teenagers.53
The review of systems should include symptoms of adrenergic stimulation as well as symptoms of end-organ damage. Adolescent girls should be asked about their last menstrual cycle to determine if they are possibly pregnant.
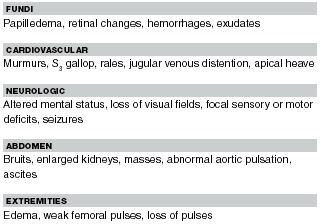
Physical Examination. The physical examination should focus upon signs of secondary hypertension or end-organ damage (Table 6). Patients with hypertensive urgency may be asymptomatic or may present with nonspecific symptoms such as headache, blurry vision, and vomiting.12,33 As noted in Table 6, patients with hypertensive emergency may present with altered mental status, seizures, left ventricular failure, oliguria, and focal neurologic findings.11,12,33 The examination should include a thorough cardiovascular assessment to look for heart murmurs, gallops, abdominal bruits, weak femoral pulses, and poor peripheral perfusion. Perform a thorough neurological assessment concentrating on subtle cranial nerve deficits and visual field defects. A fundoscopic examination is essential. Do not forget to look for signs of toxidromes, especially in adolescents.
| Table 6. Physical Exam Findings Suggestive of End-Organ Damage11,54-59 |
|
|
Infants and children with hypertension may present in unusual ways. Infants with severe hypertension may present with apparent shock and congestive heart failure. These infants may have renal or cardiac pathology.54 Infants also may present with very nonspecific findings, such as irritability or failure to thrive. Bell's palsy may be an uncommon presenting symptom of pediatric hypertension. Keep a high index of suspicion for elevated blood pressure in the child with recurrent Bell's palsy or a history of episodes affecting alternating sides of the face.54
Autonomic dysfunction is common in Guillian-Barre syndrome, occurring in up to 67% of patients. Hypertension usually is present when the neurologic symptoms are at their worst, making the diagnosis evident.56 However, patients may present with autonomic dysfunction and develop characteristic symptoms of the disease afterward.57
Conversely, some children may present with hypertension unrelated to the apparent presenting disease process. For example, Henoch-Schonlein purpura may affect the kidneys resulting in hypertension. However, if the patient does not have laboratory findings consistent with renal dysfunction, they must have a more extensive evaluation to investigate other possible etiologies of their hypertension.58
Management of the Hypertensive Pediatric Patient
Once it has been determined that the elevated BP reading is accurate, a brief but focused history and examination should be performed. The goal is to quickly identify patients with end-organ damage who need more emergent management as well as to uncover possible underlying etiologies. If the patient is asymptomatic, the physician can do a more thorough history and physical examination as outlined in the previous section. Frequently, a child presents to the ED with conditions that can increase his/her blood pressure. Patients who are experiencing pain or anxiety have elevated BP while in the ED. Patients with painful conditions should be given oral or parenteral analgesic medications as judged by the examining physician. A child who is anxious usually can be reassured by their caretakers and health care providers. Hypertension, in these instances, is usually mild and transient, requiring no further therapy once the painful condition is treated and the anxiety allayed. The EP should classify the hypertension according to the new recommendations of the NHBPEP because the evaluation and management of the patient varies by stage (Table 1).
Mild Hypertension (Stage 1 Hypertension). Patients in the ED with an elevated BP in the mild range (95th to 99th percentile plus 5 mm Hg) need to have repeat measurements done over several weeks. Often the elevation of the BP is an incidental finding in the course of an ED visit for an unrelated complaint. As noted before, a patient cannot be labeled hypertensive unless the elevation of the BP is documented on three separate occasions, preferably in non-ED settings. The patient should be advised to follow up with his primary care provider for further evaluation within a month.11 However, if a patient has nonspecific symptoms from his/her elevated BP, he/she should have more urgent follow-up within a week or two. Because renal pathology is the most common cause of hypertension in the pediatric age group, the physician may order a urinalysis and urine culture during this visit.33 If the urinalysis is normal, further work-up, including other laboratory testing, radiographic testing, and pharmacologic therapy should be left to the primary care physician. An abnormal urinalysis warrants further investigation.
Severe Hypertension (Stage 2 Hypertension). Patients with severe hypertension often present to the ED in two recognizable syndromes: hypertensive urgency and hypertensive emergency; conditions that need to be evaluated by the physician immediately. The presence of signs and symptoms of end-organ damage determines the need for immediate therapy.11,33,35 Often the hypertension is chronic in nature and being treated. Patients with no history of preexisting hypertension, who present with hypertensive emergencies should be examined closely for head trauma, toxemia of pregnancy, acute drug ingestion, pheochromocytoma or glomerulonephritis.12 Keep in mind that the younger the child and the higher the BP, the more likely that the hypertension is due to a secondary cause.33
Hypertensive Urgency. Patients with hypertensive urgency have severe hypertension but with no life-threatening symptoms from end-organ damage. They usually present with nonspecific symptoms (e.g., headache, vomiting, abdominal pain, or blurry vision).11,12,33 For newly diagnosed patients with hypertensive urgencies, laboratory studies should include a complete blood count (CBC), an electrolyte panel, a blood urea nitrogen (BUN) measurement, a creatinine (Cr) measurement, a urinalysis, and a urine culture.33 Adolescent girls should have a urine pregnancy test to rule out preeclampsia. Urine drug screening is useful in cases where drug abuse is suspected as a cause of the hypertension. Further work-up depends upon other clues provided by the history and physical examination. A nephrologist should be consulted early in the management.
Because hypertensive urgency soon can progress to hypertensive emergency, the standard practice among many EPs is to treat these patients despite the lack of end-organ damage. The goal of therapy in a child or adolescent with hypertensive urgency is to reduce BP safely and slowly within a few hours or days to preserve organ function.35,62,63 Rapid reduction of the BP should be avoided because it can be harmful. Oral antihypertensive agents are usually sufficient to reduce the BP to normal.33 On rare occasions, parenteral therapy may be needed.
The EP has an array of pharmacological options to use in the patient with hypertensive urgency. The individual physician's experience with various antihypertensive agents, presumed underlying etiology, and comorbid conditions will often dictate which medication is used.12 Unlike adults, advances in drug therapy in the pediatric population has been hampered by lack of scientific data regarding efficacy, pharmacokinetics, and age-appropriate dosing of antihypertensive medications.64 Recent legislation has prompted industry sponsored trials specifically for children and as a result, newer data on drug therapy have emerged.11,64
Commonly used older antihypertensive agents are being replaced by newer and safer agents.64 One such drug is nifedipine, a short-acting calcium channel blocker that has been shown to reduce blood pressure by at least 30% after a single dose.65.66 Recent reports on adverse cardiac and neurologic sequelae caused by the rapid decrease in BP has led to the recommendation that other medications be used to treat hypertensive urgencies in adults.35,67,68 However, these complications have not been observed in children. Instead, a few cases of rebound hypertension causing neurologic sequelae have been reported.67 At the present time, nifedipine continues to be recommended by most nephrologists33,34,62,63 in the treatment of hypertensive urgencies. The drug is administered by having patients bite and swallow the capsule. Its onset of action is within 10 minutes, peaking in 60 minutes with a duration of 4-6 hours. Other drugs that are used include clonidine, captopril, and minoxidil (Table 7).
| Table 7. Drugs Used for Hypertensive Urgencies in Children11,12,31 |
|
|
If the patient has chronic hypertension and the blood pressure was lowered successfully after 4-6 hours of observation, he/she can be safely discharged home with close follow-up with a nephrologist. A patient needs admission if the underlying etiology of the severe hypertension is not known, ED management failed, or compliance and follow-up are problematic.
Hypertensive Emergencies. By definition, a child with a hypertensive emergency has severe hypertension and has symptoms resulting from end-organ ischemia.11,12,35,71,72 Fortunately, this is a rare event in the pediatric population. The most common clinical manifestations of end-organ damage are hypertensive retinopathy, encephalopathy, seizures, left ventricular hypertrophy, cranial nerve palsies, hemiplegia, and renal failure.33,35 As mentioned in the previous section, neonates may present with no symptoms, nonspecific symptoms, or congestive heart failure, seizures, and lethargy.35
As in any emergency, the ED physician should give careful attention and support to airway, breathing, circulation, and neurologic functions. A focused history and physical examination, including a fundoscopic examination, are important. The child should be placed on a cardiac monitor with frequent BP measurements and continuous pulse oximetry readings. An intravenous line (preferably two) should be immediately established, while samples for laboratory studies are obtained. Initial studies to assess end-organ dysfunction should include CBC, electrolytes measurement, BUN, creatinine, urinalysis, a urine culture, chest radiograph (CXR), and an electrocardiogram (ECG). An indwelling bladder catheter should be inserted, and an arterial line should be strongly considered. Similar to the patient with hypertensive urgency, a nephrologist should be consulted early to assist with evaluation and management.
The goals of treatment are to prevent complications of end-organ ischemia while preserving function and minimizing side effects of drug therapy.12,35 Parenteral antihypertensive drugs with short half-lives should be titrated carefully to therapeutic endpoints. Oral agents are not recommended because of rapid, yet unpredictable, lowering of the BP.35 The goal is a slow, but controlled, reduction in blood pressure by 25% during the first 6 to 8 hours with a gradual return to normal levels by 48 hours.11,35 A more rapid reduction may lead to hypoperfusion of the brain as autoregulatory mechanisms are not able to adjust. Again, the physician's preference for which drug to use is guided by his/her familiarity and previous experience with a specific agent and the possible underlying cause of the hypertension.
Specific Antihypertensive Medications
Table 8 shows the most common drugs used in the treatment of hypertensive emergencies in the pediatric population. Sodium nitroprusside, nicardipine, and labetalol are parenteral drugs that have been recommended as first-line medications because of their effectiveness, good safety profile, and limited toxicity.35
Sodium nitroprusside is a powerful direct vasodilator of both arteries and veins. It is, by far, the most effective parenteral drug used in hypertensive emergencies. It has a rapid onset and a brief duration of action, hence its use requires constant intensive monitoring of the patient. Because it is easily inactivated by light and needs to be given as a continuous infusion, it is cumbersome and time consuming to administer. Its utility in the ED is limited and has been mostly supplanted by other drugs.33,35 Sodium nitroprusside can cause cyanide toxicity if given for a prolonged period or if renal failure is present.11,35 It should not be given in pregnant adolescents.
Labetalol is a powerful alpha-1 and a nonspecific beta blocker that reduces blood pressure by decreasing peripheral resistance. The beta blockade is responsible for hyperglycemia, hyperkalemia, and diminished reflex tachycardia.33,69 It is effective for patients with pheochromocytoma, coarctation of the aorta, and in those with intrinsic renal disease. It is contraindicated in patients with asthma, insulin-dependent diabetes, or congestive heart failure.11,33,69 Labetalol is given as a bolus every 10 minutes or as an infusion.
Esmolol is another cardioselective beta blocker successfully used in patients with hypertensive crises after repair of congenital heart disease.69 Its side effects and contraindications are similar to other beta blockers.
Nicardipine is a drug that reduces peripheral vascular resistance by blocking calcium channels in vascular smooth muscle. It is the first drug in its class to be approved for intravenous use.73 Experience in children is increasing, and it has been used successfully in patients after repair of coarctation of the aorta and post transplantation.35,69,70
Enalaprilat is an angiotensin-converting enzyme (ACE) inhibitor and particularly effective in patients with high renin levels.73 It is contraindicated for patients with renal artery stenosis because it is excreted by the kidneys. Response to this drug is variable and dependent upon plasma renin activity. ACE inhibitors, in general, should be avoided in pregnant adolescents as teratogenic effects have been observed.35
Phentolamine is an alpha blocker indicated for conditions where there are high circulating catecholamines such as pheochromocytoma and drug-induced hypertension. Hydralazine is another direct vasodilator, yet its use has been gradually supplanted by other faster-acting and more potent drugs.33,69 It can cause reflex tachycardia, headaches, and fluid retention. It is primarily used in patients with preeclampsia or eclampsia.
Vitals signs, with special attention to the BP, should be obtained every 15 minutes with frequent assessment of neurologic status. The physician also should document serial examinations while the patient is in the ED. All patients with hypertensive emergency should be admitted to the intensive care for further management.12,33
Summary
Hypertension in the pediatric population is commonly encountered in the ED. Most cases are transient and need no further intervention. Mildly hypertensive and asymptomatic patients can be safely discharged for repeat BP measurements and close follow-up with their primary care physician. Severe hypertension can be life threatening and has serious sequelae if treatment is either inadequate or too aggressive. Patients with hypertensive urgency and emergency need to be evaluated and treated expeditiously while in the ED. The choice of drug depends upon the physician's experience, the presumed underlying condition, as well as the clinical presentation. Patients with hypertensive urgency whose blood pressure was lowered successfully can be safely discharged home with close follow-up. Patients with hypertensive emergency need to be treated promptly with parenteral antihypertensive agents and admitted to the intensive care unit for further management. Early consultation with a nephrologist is important (See Figure 2).
References
1. Victor R. Arterial hypertension. In: Goldman L, Ausiello D, eds. Cecil Textbook of Medicine, 22nd ed; St. Louis: WB Saunders; 2004.
2. Norwood VF. Hypertension. Pediatr Rev 2002;23:197-208.
3. Daniels SR. Repair of coarctation of the aorta and hypertension: Does age matter? Lancet 2001 Jul 14;358(9276):89.
4. Staessen J, Fagard R, Amery A. The relationship between body weight and blood pressure. J Hum Hypertens 1988;2:207-217.
5. Sorof J, Daniels S. Obesity hypertension in children: a problem of epidemic proportions. Hypertension 2002;40:441-447.
6. Flack JM, Peters R, Mehra VC. Hypertension in special populations. Cardiol Clin 2002;20:303-19.
7. Sorof JM, Poffenbarger T, Franco K. Isolated systolic hypertension, obesity, and hyperkinetic hemodynamic states in children. J Pediatr 2002;140:660-6.
8. Ingelfinger JR. Pediatric antecedents of adult cardiovascular disease – awareness and intervention. NEJM 2004;350:2123-2126.
9. Rocella EJ, Bowler AE, Horan M. Epidemiologic considerations in defining hypertension. Med Clin North Am 1987;71:785-801.
10. The seventh report of the joint national committee on detection, evaluation, and treatment of high blood pressure: The JNC7 report. JAMA 2003;289:2560-72.
11. National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 2004;114:555-576.
12. Linakis JG. Hypertension. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine, 4th ed; Philadelphia: Lippincott Williams & Wilkins; 2000:309-315.
13. Watkinson M. Hypertension in the newborn baby. Arch Dis Child 2002;86:F78-81.
14. Zubrow A, Hulman S, Kushner H. Determinants of blood pressure in infants admitted to neonatal intensive care units: a prospective multicenter study. J Perinatol 1995;15:470-479.
15. Kilian K. Hypertension in neonates: causes and treatments. J Perinatal & Neonatal Nursing 2003;17:65-74.
16. Goonaskera CD, Dillon MJ. Measurement and interpretation of blood pressure. Arch Dis Childhood 2000;82:261-265.
17. Yap HK, Low PS, Lee BW. Factors influencing the development of hypertensive encephalopathy in acute glomerulonephritis. Child Nephrol Urol 1988;9:147-152.
18. De Swiet M, Dillon MJ, Little W. Measurement of blood pressure in children. Reccomendations of a working party of the British Hypertension Society. Brit Med J 1989;299:497.
19. Ingelfinger JR. Hypertension in the first year of life. In: Ingelfinger JR, ed. Pediatric Hypertension. Philadelphia: WB Saunders; 1982:229-40.
20. Friedman AL, Hustead VA. Hypertension in babies following discharge from a neonatal intensive care unit. Pediatr Neph 1987;1:30-4.
21. Jette A. Exaggerated blood pressure response to exercise in the detection of hypertension. J Cardiopulm Rehab 1988:8:171-177.
22. Benbassat J, Froom P. Blood pressure response to exercise as a predictor of hypertension. Arch Internal Med 1986;146:2053-2055.
23. Matthews CE, Pate RR, Jackson KL. Exaggerated blood pressure response to dynamic exercise and risk of future hypertension. J Clin Epid 1998;51:29-35.
24. Fredrikson M. Psychophysiological theories on sympathetic nervous system hyper-reactivity in the development of essential hypertension. Scand J Psychol 1991;32:254-275.
25. Matthews KA, Salomon K, Brady SS. Cardiovascular reactivity to stress predicts future blood pressure in adolescence. Psychosom Med 2003;65:410-415.
26. Thomas RJ, Liu K, Jacobs DR. Positional change in blood pressure and 8-year risk of hypertension: the CARDIA study. Mayo Clinic Proceedings 2003;78:951-958.
27. Boyd GS, Koenigsberg J, Falkner B, et al. Effect of Obesity and high blood pressure on plasma lipids in children and adolescents. Pediatrics 2005;116:442-446.
28. Weiss R, Dziura J, Burgert TS, et al. Obesity and the metabolic syndrome in children and adolescents. N Engl J Med 2004; 350:2362-74.
29. Cook S, Weitzman M, Avinger P. Prevalence of metabolic syndrome phenotype in adolescents: Findings from the third national health and nutrition survery, 1988-1994. Arch Ped Adol Med 2003;157:1-827.
30. Li S, Chen W, Srinivasan S, et al. Childhood cardiovascular risk factors and carotid vascular changes in childhood: the Bogalusa Heart Study. JAMA 2003;290:2271-2276.
31. Enright PL, Goodwin JL, Sherrill DL. Blood pressure elevation associated with sleep-related breathing disorder in a community sample of white and hispanic children: the Tucson Children's assessment of sleep apnea study. Arch Ped Adol Med 2003;157:901-904.
32. Kaplan RA, Hellerstein S, Alon U. Evaluation of the hypertensive child. Child Nephrol Urol 1992;12:106-12.
33. Kronan K and Norman ME. Renal and electrolyte emergencies. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine,4th ed;Philadelphia:Lippincott Williams & Wilkins;2000:835-841.
34. Vaughan CJ and Delanty N. Hypertensive Emergencies. Lancet 200; 356:411-17.
35. Adelman RD, Coppo R, Dillon MJ. The emergency management of severe hypertension. Pediatr Nephrol 2000; 14:422-427.
36. Beevers G, Lip GY, O'Brien E. Blood pressure measurement: part 1-sphygmomanometry: factors common to all techniques. Brit Med J 2001;322:981-985.
37. O'Brien E. A century of confusion: which bladder for accurate blood pressure measurement? J Hum Hypertension 1996;10:565-572.
38. Pappadis SL, Somers MJ. Hypertension in adolescents: a review of diagnosis and management. Curr Opin Pediatr 2003;15:370-378.
39. Smets K, Vanhaesebrouck P. Dexamethasone associated systemic hypertension in low birth weight babies with chronic lung disease. Eur J Pediatr 1996;155:573-5.
40. Flynn J. Neonatal hypertension: diagnosis and management. Ped Nephrol 2000;14:332-341.
41. Rajpoot D, Duel B, Thayer K. Medically resistant neonatal hypertension: revisiting surgical causes. J Perinatol 1999;19:582-583.
42. Huxley RR, Shiell AW, Law CM. The role of size at birth and post-natal catch-up growth in determining systolic blood pressure: a systemic review of the literature. J Hypertens 2000;18:815-831.
43. Law CM, Barker DJ, Osmond C. Early growth and abdominal fatness in adult life. J Epid Comm Health 1992;46:184-186.
44. Horn P. Persistent hypertension after prenatal cocaine exposure. J Ped 1992;121:288-291.
45. Bald M, Neudorf U. Ambulatory blood pressure monitoring and coarctation of the aorta. J Ped 2001;138:610-611.
46. O'Sullivan JJ, Derrick G, Darnell R. Prevalence of hypertension in children after early repair of coarctation of the aorta: a coort study using casual and 24 hour blood pressure measurement. BMJ 2002;88:163-6.
47. Franz DN. Diagnosis and management of tuberous sclerosis complex. Semin Pediatr Neurol 1998;5:253-68.
48. Satake H, Inoue K, Kamada M. Malignant composite pheochromocytoma of the adrenal gland in a patient with Von Recklinghausen's disease. Clin Urol 2001;165:1199-1200.
49. Flynn MP, Buchanan JB. Neurofibromatosis, hypertension and renal artery aneurysms. South Med J 1980;73:618-20.
50. Safer DJ, Zito JM, Fine EM. Increased methylphenidate usage for attention deficit disorder in the 1990s. Pediatrics 1996;98:1084-1088.
51. Johnston LD, O'Mally PM, Bahman JG. Monitoring the Future National Survey Results on Drug Abuse 1975-2000, Vol 1:Secondary School Students (NIH publication 01-4924). Bethesda, MD: National Institute on Drug Abuse, 1-519.
52. Klein-Schwartz W, McGrath J. Poison Centers experience with methylphenidate abuse in pre-teens and adolescents. J Am Academ Child Adolesc Psych 2003;4:288-294.
53. Savoca MR, Evans CD, Wilson ME. The association of caffeinated beverages with blood pressure in adolescents. Arch Pediatr Adolesc 2004;158:473-7.
54. Hawkins K, Watson A, Rutter N. Neonatal hypertension and cardiac failure. Eur J Ped 1995;154:148-149.
55. Riordan M. Investigation and treatment of facial paralysis. Arch Dis Child 2001;84:286-288.
56. Singh NK, Jaiswal AK, Misra S. Assessment of autonomic dysfunction in Guillon-Barre syndrome its prognostic implications. Acta Neurol Scand 1987;75:101-105.
57. Labovitz AE, Mangurten HH. Guillain-Barre syndrome presenting with urinary retention and hypertension. Clin Pediatr 2004;43:659-61.
58. Harms MM, Rotteveel JJ, Kar NC. Recurrent alternating facial paralysis and malignant hypertension. Neuropediatrics 2000;31:318-320.
59. Aynaci FM, Sen Y. Peripheral facial paralysis as initial manifestation of hypertension in a child. Turkish J Pediatrics 2002;44:73-5.
61. Tirodker UH, Dabbagh S. Facial paralysis in childhood hypertension. J Pediatr Child Health 2001;37:193-4.
60. Drummond PM, Moghal NE, Coulthard MG. Hypertension in Henoch-Schonlein purpura with minimal urinary findings. Arch Dis Children 2001;84:163-164.
62. Egger DW, Deming DD, Hamada N, et al. Evaluation of the safety of short acting nifedipine in children with hypertension. Pediatr Nephrol 2002;17:35-40.
63. Blasnak RT, Savage JA, Ellis EN. The use of short-acting nifedipine in pediatric patients with hypertension. J Pediatr 2001;139:34-37.
64. Flynn JT. Pharmacologic management of childhood hypertension: current status, future challenges. AJH 2002;15:305-335.
65. Siegler RL, Brewer ED. Effect of sublingual or oral nifedipine in the treatment of hypertension. J Pediatr 1988;112:811-813.
66. Evans JHC, Shaw NJ, Brockleman JT. Sublingual nifedipine in acute severe hypertension. Arch Dis Child 1988; 63:975-977.
67. Leonard MB, Kosner SE, Feldman HI, Schulman SL. Adverse neurologic events associated with rebound hypertension after using short acting nifedipine in childhood hypertension. Pediatr Emerg Care 2001;17:435-437.
68. Flynn JT, Pasko DA. Calcium channel blockers: pharmacology and place in therapy of pediatric hypertension. Pediatr Nephrol 2000; 15:302-316
69. Porto I. Hypertensive emergencies in children. J Pediatr Health Care 2000;14:312-319.
70. Nehal US, Ingelfinger JR. Pediatric hypertension: Recent literature. Curr Opin Pediatr 2002;14:189-196.
71. American College of Obstetricians and Gynecologists, Committee on Obstetrics. Hypertension in Pregnancy. Washington DC: Technical Bulletin Number 219, January 1996.
72. Walker JJ. Pre-eclampsia. Lancet 2000;356:1260-1265.
Hypertension is a very common disease affecting up to 20-25% of adults in the United States. In the past, hypertension has been considered a rare disease in children; however, in the last 10 years it has emerged as an important disease in pediatrics as well. Because hypertension is a risk factor for cerebrovascular disease, cardiovascular disease, as well as renal disease, the increase in pediatric hypertension is a concern because of its potential for long term sequelae.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.