Anaphylaxis
Anaphylaxis
Introduction
Anaphylaxis is a rapid-onset and potentially life-threatening allergic reaction.1 The diagnosis is clinical and based on any one of three criteria. (See Table 1.) While dermatologic findings are present in up to 90% of cases, it is imperative to note that skin findings and hypotension/shock are not required for diagnosis in all cases.
Table 1: Clinical Criteria for Diagnosing Anaphylaxis
Anaphylaxis is highly likely when any one of the following 3 criteria are fulfilled:
1. Acute onset of an illness (minutes to several hours) with involvement of the skin, mucosal tissue, or both (e.g., generalized hives, pruritus, or flushing, swollen lips-tongue-uvula)
AND AT LEAST ONE OF THE FOLLOWING
a. Respiratory compromise (e.g., dyspnea, wheeze-broncospasm, stridor,
reduced PEF, hypoxemia)
b. Reduced BP or associated symptoms of end-organ dysfunction (e.g.,
hypotonia [collapse], syncope, incontinence)
2. Two or more of the following that occur rapidly after exposure to a likely allergen for that patient (minutes to several hours):
a. Involvement of the skin-mucosal tissue (e.g., generalized hives, itch-flush,
swollen lips-tongue-uvula)
b. Respiratory compromise (e.g., dyspnea, wheeze-broncospasm, stridor,
reduced PEF, hypoxemia)
c. Reduced BP or associated symptoms (e.g., hypotonia [collapse], syncope,
incontinence)
d. Persistent gastrointestinal symptoms (e.g., crampy abdominal pain,
vomiting)
3. Reduced BP after exposure to known allergen for that patient (minutes to several hours):
a. Infants and children: low systolic BP (age specific) or greater than 30%
decrease in systolic BP*
b. Adults: systolic BP of less than 90 mmHg or greater than 30% decrease
from that person's baseline
PEF = Peak expiratory flow; BP = blood pressure
* Low systolic blood pressure for children is defined as less than 70 mmHg from 1 month to 1 year, less than (70 mmHg + [2 x age]) from 1 to 10 years, and less than 90 mmHg from 11 to 17 years.
Reprinted with permission from: Sampson HA, Munoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: Summary report Second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. J Allergy Clin Immunology 2006;117:391-397.
A condition that has affected mankind since the beginning of history, anaphylaxis' earliest description is documented in Egyptian hieroglyphics showing a pharaoh's death from an insect sting in 2641 B.C. It was first documented in the medical literature in 1902, when a French scientist attempting to immunize dogs to jellyfish toxin found that some subjects had severe symptoms to small amounts of the injected toxin.2 The term "l'anaphylaxie" was subsequently coined from the Greek meaning "against protection."
Anaphylaxis is the medical equivalent of traumatic injuries; it affects all age groups and ethnicities. It presents in all medical specialties and settings, although a large proportion of cases will present to emergency services or the emergency department (ED) due to our ability to rapidly diagnose, resuscitate, and definitively treat affected patients.
Etiology
Major causes include medications, foods, and insect stings. Among medications, beta-lactams, and nonsteroidal anti-inflammatory drugs (NSAIDs) including aspirin predominate. Other common and novel medications include chemotherapeutic agents, loperamide, monoclonal antibodies (mAbs, cetuximab, infliximab, etc.), strepto- or urokinase, insulin, and folic-acid containing vitamins. Perioperative anaphylaxis can occur with neuromuscular-blocking agents, barbiturates, propofol (contains egg- and soy-based constituents), opiates, benzodiazepines, colloids (dextran, albumin, hetastarch), topical chlorhexidine or povidone-iodine, local anesthetics, heparin, and protamine. Alternative medicines and herbal medications should be considered, as they often contain multiple substances and are not regulated by the FDA. Vaccines rarely cause allergic reactions (1 event per million administrations, 235 million doses per year in United States). It is more common for diluents such as egg, gelatin, or dextran to cause a reaction.3-5
Among foods, peanuts, tree nuts (walnuts, pecans, pistachios, cashews), shellfish, fish, milk, eggs, and sesame are most prevalent. Hidden triggers (i.e., cross-processed foods made in the same factory as peanuts), additives (colorants, spices), contaminants (dust mites), and parasites also should be considered.
Stinging insect cases are most common from the order Hymenoptera. This includes the family Apidae (honeybees) as well as Vespidae (yellow jackets, hornets, wasps) and Formicidae (ants). Saliva from biting insects is known to cause anaphylaxis but this is less common.6
Iodinated radiocontrast dye is a rising non-immunologic cause, most likely due to the increase in imaging studies. Other non-IgE-mediated causes include exercise (either alone or commonly food trigger-dependent), temperature (hot or cold), ethanol, opiates, and vancomycin.
Latex use as an etiology has decreased in the medical setting due to the use of non-latex gloves and devices; however, drains and catheters may still be nosocomial etiologic agents. In the community setting, condoms, gloves, balloons, and pacifiers still result in anaphylaxis. Additionally, foods including avocado, banana, kiwi, papaya, potato, and tomato can cross-react with latex. Other less common etiologic agents include semen, inhaled pollen, and animal dander.
Risk Factors/Predisposing Conditions
Asthma and chronic obstructive pulmonary disease (COPD) are associated with acute bronchospasm leading to anaphylaxis-induced death.7 Mastiocytosis and other clonal mast cell disorders place patients at higher risk of severe and fatal reactions. Antihypertensives can interfere with the body's vasoconstrictive response, leading to more severe presentations. In particular, beta-blockers not only lead to potentiation of hypotension and increased bronchoconstriction but also interfere with treatment.
Epidemiology
Incidence/Prevalence. Misdiagnosis and underreporting make the true incidence and prevalence difficult to state. Many ED cases are not recognized or are coded as "acute allergic reaction," or "hypersensitivity reaction." This is particularly common with food-related etiologies.
Estimates of lifetime prevalence from all etiologies range from 0.05% to 2%, although rates as high as 15% have been stated. Of these, up to 2% may have fatal episodes. Anaphylactic incidence per year is estimated at 0.03% to 0.95%, and fatal cases at 0.002% annually (~1500 deaths per year in the United States).3,8-12
Among major causes of anaphylaxis, medications affect adults most often, with antibiotics and NSAIDs being most prevalent. Beta-lactam reactions occur at 5 per 10,000 patient courses of treatment, account for 22% of drug-related cases, and for up to 75% of drug-related fatal cases in the United States per year.8
More than one thousand Americans (0.0004%) experience food-related anaphylaxis each year. Almost all patients with food anaphylaxis have a known prior food allergy.13 Food-related anaphylaxis results in up to 50% of ED anaphylactic presentations. In the pediatric population, it is the most common anaphylactic etiology, affecting 2% of the population and causing up to 100 deaths per year in the United States. Peanuts and tree nuts are the most common triggers in children, and shellfish are the most common in adults.
Hymenoptera envenomation alone would require a dosage equivalent to 1500 stings in a 70 kg adult to be lethal in a nonallergic patient; however, due to anaphylactic reactions, upwards of 40 deaths occur per year.6
Scope/Population Distribution. Children and teenagers display a higher rate of anaphylaxis than adults. Young males up to 15 years of age receive the most epinephrine injections; thereafter, women receive more. There is no incidence difference between gender seen in elderly patients. Race and ethnicity are not contributory to frequency, although living in a rural area and having poor socioeconomic status are associated with lower rates of epinephrine usage in the medical setting.
Pathophysiology
Sensitization. The vast majority of anaphylactic episodes are immunologically mediated. The initial exposure to an allergen will not cause systemic symptoms. On presentation, the antigen will encounter antigen-presenting cells (APC) such as dendritic cells, macrophages, and B cells.
APCs internalize antigen and produce major histocompatibility complex II (MHCII) on their surface, which displays the allergen to CD4 TH2 type helper T cells. The T cells subsequently activate B cells to switch from generalized IgM to allergen-specific IgE antibody production. These IgE antibodies contain Fab (branch) and Fc (trunk) components. They circulate throughout the body and bind to Fc receptors on circulating basophils and tissue-bound mast cells.7
Activation. On subsequent allergen presentation, cell-bound IgE binds the allergen and activates these cells via calcium influx and tyrosine kinase activation. This leads to degranulation and release of pre-formed pro-inflammatory mediators. These include histamine, tryptase, chymase, and proteoglycans, as well as nitric oxide, various cytokines/chemokines, TNF-α, and arachidonic acid metabolites (prostaglandins, leukotrienes, platelet activating factor (PAF). (See Figure 1.)
Figure 1: Anaphylaxis Mediators
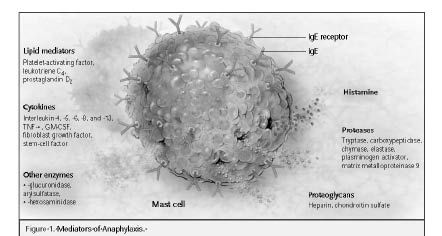
Reprinted with permission from: Burks AW. Factoring PAF in anaphylaxis. N Engl J Med 2008;358:79-81
Histamine binds H1 and H2 receptors resulting in urticaria, pruritus, flushing, rhinnorhea, headache, bronchospasm, hypotension, and tachycardia. Tryptase interacts with the complement and coagulation systems to cause hypotension, angioedema, and potential disseminated intravascular coagulation. Tryptase levels correlate with anaphylaxis severity except in food-induced episodes. Nitric oxide causes vasodilation and increased vascular permeability. Arachidonic acid metabolites enhance mast cell degranulation, increase vascular permeability and vasodilation, lead to bronchoconstriction, and may contribute to prolonged or biphasic anaphylactic episodes.
Variant Pathogenesis. The term anaphylactoid reaction is no longer used. Non-IgE-mediated anaphylactic reactions, such as those caused by radiocontrast media (RCM), are believed to occur when antigen directly interacts with the Fc component (as opposed to Fab component) of preformed antibodies on mast cells or basophils. Non-immune-mediated anaphylaxis to drugs may occur via direct complement activation (propofol, paclitaxel), direct mast cell/basophil activation (RCM, opiates, vancomycin, cold exposure), coagulation system activation (oversulfated chondroitin [OSCS] contaminated heparin), or bradykinin production (ACE inhibitors, OSCS heparin). Both non-IgE- and non-immune-mediated reactions are unique in that they may occur on first exposure to the inciting agent, although their treatment is identical to IgE-mediated reactions.3,14,15
Clinical Features
History. Anaphylaxis is under-recognized and undertreated. The history is the most important aspect of investigation into anaphylaxis. While chief complaint may vary, patients or EMS should be asked about any known exposure to a trigger and any previously known allergic reactions or anaphylactic events. Ask about recent diet, medication changes, body and clothes washing behavior, exposure to outdoor areas, and recent travel/weather changes. The patient's occupation may be important as a source of allergen presentation. The time course of the exposure, as well as the change in symptoms over time, should be noted.
Dermatologic symptoms are present in 80-90% of episodes, respiratory tract in up to 70%, GI and cardiovascular in up to 45% each, and CNS in up to 15%.1,3
Vital signs are highly variable. While tachycardia, tachypnea, and hypotension are classic for severe disease, they may not be present simultaneously or until late in the course.
Physical Exam and Presenting Signs/Symptoms. A rapid primary survey should be performed. The mnemonic ABCDE may be useful for prioritizing examination and treatment; Airway, Breathing, and Circulation are normal concerns, although in the anaphylactic patient the D stands for Disability/Neurologic status and Dermatologic examination. E represents Epinephrine, and while not an assessment, its primary role in treatment is imperative.
Anaphylaxis is a systemic phenomenon. In its most severe form, all of the following signs and symptoms culminate in circulatory or respiratory arrest. The emerging view is that instead of being a form of distributive shock, there is a combination of distributive shock from decreased vascular tone, hypovolemic shock from massive fluid extravasation and tissue edema leading to decreased intravascular volume, and cardiogenic shock from myocardial depression.7
Specific organ systems affected are as follows:
Dermatologic. Up to 90% of episodes display cutaneous findings. These include flushing, pruritus, urticaria (superficial layers), angioedema (deeper dermal layers), and piloerection. Be wary of patients who present without skin complaints or those who have recently taken H1-antihistamines as this may mask or decrease the appearance of skin findings.
Respiratory. Up to 70% of patients with anaphylaxis will display respiratory symptoms. These include rhinorrhea, sneezing, and laryngeal edema. Lower airway symptoms include cough, wheezing, dyspnea, and respiratory arrest.
Gastrointestinal. Nearly 40% of episodes involve oral pruritus/tingling, nausea and vomiting, cramping, diarrhea, or intestinal wall angioedema. GI symptoms tend to be more prominent in food-induced reactions. Higher dosages of ingested allergens and concomitant alcohol ingestion correlate with rapidity of onset and severity of symptoms.10,16
Cardiovascular. Cardiovascular signs/symptoms occur in 35% of anaphylaxis episodes.17 These include lightheadedness, chest pain, palpitations, tachycardia, hypotension, and widened pulse pressure. Up to 35% of intravascular fluid can extravasate within 10 minutes.1 There is an association between anaphylaxis and myocardial ischemia as well as conduction defects and arrhythmias. Mast cell degranulation within coronary plaques promotes plaque rupture and ischemia/infarction. Histamine has been shown to induce coronary artery vasoconstriction/spasm, and PAF decreases coronary blood flow.
Neurologic. Neurologic symptoms include anxiety, confusion, headache (common in exercise-induced anaphylaxis), syncope, or a sense of impending doom.
Other Systems. Ocular itching/tearing and erythema are common. Women may experience uterine cramping.
It must be re-emphasized that skin and mucosal symptoms may be absent in up to 20% of cases. This occurs particularly in food-related instances where GI symptoms may predominate as well as in the neonatal population.
Diagnostic Studies
While mediator levels, including serum histamine and tryptase levels, have been analyzed to confirm anaphylaxis, none are specific or predictive in a short enough time frame to be useful for diagnosis or to indicate need for treatment. Furthermore, tryptase is rarely elevated after food-induced reactions. Diagnostic studies have little to no role in the acute diagnosis or management of anaphylaxis.
Differential Diagnosis
Anaphylaxis should be differentiated from a less severe allergic reaction. As a rule, it is better to overestimate the severity of disease and favor definitive treatment if in doubt.
The differential includes generalized urticaria, asthma exacerbation, anxiety/panic attack, foreign body ingestion (pediatric population), vasovagal reactions, syncope, pulmonary embolus, seizure, stroke, myocardial infarction, and sepsis.
Food-associated non-anaphylactic illnesses as well as excess endogenous histamine syndromes are less common but include scrombroid, fish-related parasitic anisakiasis, echinococcus hyatid cyst rupture, mastiocytosis, carcinoid syndrome, VIPomas, and perimenopausal flushing.
More rare conditions include vocal cord dysfunction (clues may be not only inspiratory but also expiratory stridor, and little if any uvular edema), non-allergic hereditary or acquired angioedema (little to no associated urticaria or pruritus), spinal injury, dysautonomia, malignant hyperthermia, amniotic fluid embolus, and non-anaphylactic causes of shock (cardiogenic, hypovolemic, distributive, septic).
Lastly, medication reactions can mimic some or all anaphylactic presentations, most notably vancomycin-induced red man syndrome.3,18
Special Populations
Age or disease may mask symptoms and result in a delay in diagnosis.
Infants. Signs and symptoms including hypotension are difficult to recognize. In addition, normal conditions such as crying or feeding may result in flushing or dysphonia, emesis or loose stools.
Children. Patients with a history of atopy including asthma, eczema, and allergic rhinitis are at increased risk of anaphylaxis. Food allergens are common triggers in this population, especially eggs, milk, and soy (allergies that children often outgrow), as well as nuts and shellfish (allergies that are not outgrown). Vital signs initially can appear normal before sudden severe physiological changes occur. Teenagers' risk-taking behavior may lead them to be less rigorous in avoiding known triggers.14
Pregnant Women. Triggers are similar to those for non-pregnant women. The fetus itself is not a direct target, as maternal IgE cannot cross the placenta. Nevertheless, maternal hypoxemia and hypotension during anaphylactic episodes can result in fetal demise or CNS damage. During labor, beta-lactams are commonly given for group B streptococcal prophylaxis, potentially leading to a higher incidence of antibiotic-related reactions.3 In addition to standard treatment, left lateral decubitus positioning to prevent vena cava compression is warranted. Fetal monitoring is essential, with emergent caesarean section preparation made for a persistently non-reassuring tracing.19 Breast-feeding rarely is associated with anaphylaxis.
Patients with Neurologic/Psychiatric Conditions. Patients with disabilities, impaired sensorium, intoxication, or depression or other mental illness may have difficultly articulating their symptoms. Patients taking monoamine oxidase inhibitors or tricyclic antidepressants may have enhanced or prolonged epinephrine effects, as could those on stimulants including cocaine/methamphetamine or methylphenidate.17
Elderly. Concomitant conditions including COPD and cardiovascular disease and the medications used to treat them (beta-blockers, alpha blockers, etc.) place this population at higher risk of morbidity and mortality during anaphylaxis.
Fatal Anaphylaxis Cases. While rare, these episodes are more prone to present with initial respiratory rather than circulatory distress. Upper airway edema and bronchial obstruction are more common causes than overt circulatory collapse.17
Severe symptoms occur within 30 minutes of exposure in 84% of patients, and death occurs within an hour in 52% of these cases. A minority display urticaria (4%). Medications (especially beta-lactams) cause 50-75% of cases, while foods and insect stings cause almost 25% of cases.
The severity of previous reactions is not predictive, nor is the dose of the offending allergen. Asthma is a common co-morbid condition, as is cardiopulmonary disease in adults. Peri-operative anaphylaxis is associated with increased morbidity and mortality. Nearly all fatal cases involve patients who have had some form of prior allergic reaction during their lives. Patients tend to present at an older age (mean 59 years), although adolescents are at higher risk for food-related, adults for venom-induced, and the elderly for medication-associated etiologies. Gender, genetics, and race play an unclear role.5,9,12
Poor outcomes related to the health system include misdiagnosis, epinephrine underutilization, delay of epinephrine administration more than 30 minutes, and upright patient posture leading to "empty ventricle syndrome." Underdiagnosis is likely given the lack of definitive autopsy findings and diagnostic tests.9,20
Biphasic and Protracted Anaphylaxis. Uniphasic anaphylaxis is present in 80-90% of cases. Nevertheless, protracted and biphasic episodes occur in up to 20% of cases.7,21 Protracted anaphylaxis is defined as lasting hours to days without full resolution of symptoms. Biphasic anaphylaxis occurs when a response resolves for more than hour and is followed by recurrence of symptoms without subsequent allergen exposure.
It is thought that biphasic reactions occur because there is a second wave of mast cell degranulation, new synthesis and release of PAF and TNF-α, or uneven antigen absorption.21
Recurrent symptoms can vary in presentation and severity as compared to the first reaction, though they are typically less severe in nature. The average symptom-free episode is 10 hours, although episodes can occur in as soon as 2 hours and after a delay of up to 38 hours. Biphasic patients tend to take longer (> 2 hr) to resolve the initial episode, receive more epinephrine on initial presentation, and receive less epinephrine and fewer steroids during the recurrent presentation.22,23
Management
In addition to the usual management considerations of airway, breathing, and circulation, any skin contaminants or stingers should be removed and inciting IV medications stopped.11 Attention should then be paid to ongoing volume resuscitation and definitive pharmacological treatment.
Note that due to the unethical nature of randomizing patients with this disease to a non-treatment arm, all findings are based on case series, animal models, interpretation of underlying pathophysiology, and expert consensus.3,14
Epinephrine. Epinephrine administration is the single most important and effective action to be taken in a suspected anaphylactic episode.3,24-27 Whenever any of the three accepted criteria of anaphylaxis are met, epinephrine should be given immediately.1 (See Table 1.)
Alpha-1 receptor actions lead to vasoconstriction, increased blood pressure, and decreased mucosal edema. Beta-1 receptor activity results in increased inotropy and chronotropy. Beta-2 receptor agonism leads to bronchodilation. The cumulative effect is increased blood pressure and pulse rate, decreased mucosal edema and airway obstruction, and relief of urticaria.
Side effects at therapeutic dosages include anxiety, pallor, palpitations, tremor, and headache. While not desirable, they occur within 5-10 minutes, are typically mild in severity, and serve as indicators to the clinician that therapeutic intravascular concentration is achieved.
Nearly all adverse events and outcomes are a direct result of intravenous (IV) administration of epinephrine. Supratherapeutic dosage can occur in the ED setting and is most often due to confusion over solution concentration (1:1000 vs. 1:10,000), or too large or rapid an IV bolus dose. This can result in hypertension, pulmonary edema, intracranial hemorrhage, myocardial infarction, and ventricular arrhythmias.
Lack of response to epinephrine may occur from underdosing, hypovolemia or upright positioning resulting in low circulatory volume, rapid progression of disease, late administration, expired medication, or suboptimal route of administration.
Traditionally, the subcutaneous (SQ) route has been preferred for administration of epinephrine. In the last 15 years, however, a series of papers studying absorption pharmacokinetics first in rabbits, then in healthy children with a history of anaphylaxis, and finally in healthy young men was conducted. As epinephrine causes cutaneous vasoconstriction but skeletal muscle vasodilation, it was theorized that intramuscular (IM) administration is superior to the SQ route. These studies examined the difference in plasma epinephrine concentration using SQ, IM, and IV routes with appropriate saline controls. The results demonstrated that IM administration achieved significantly more rapid and higher peak epinephrine levels compared to the SQ route.14,28-30 No serious adverse effects occurred. Of note, the location of injection made a significant difference. Almost no difference was seen between SQ and IM when using the upper arm, but a large difference occurred when using the anterolateral thigh as the IM site.30 This is likely due to higher blood flow in the vastus lateralis muscle. Currently, the American Academy of Allergy, Asthma & Immunology (AAAAI), the American College of Allergy, Asthma & Immunology (ACAAI), the World Allergy Organization, and the National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network all recommend the anterolateral thigh as the preferred site of administration.1,8,24,25
IM administration should not be delayed. IM dosage of 1:1000 (1 mg/mL or 0.1%) epinephrine at a dose of 0.01 mg/kg is appropriate, up to a 0.5 mg maximum dose. (See Table 2.) This is given in the anterolateral thigh through the vastus lateralis muscle. Continuous cardiac, blood pressure, and O2 saturation should be monitored. If IV access is in place and there is no response to repeated q5-15 minute IM dosages, IV epinephrine may be used.
Table 2: Appropriate Epinephrine Dose and Route for Different Indications
|
Indication |
Appropriate Epinephrine Dose per American Heart Association Guidelines |
|
Anaphylaxis |
0.3--0.5 mg of 1:1,000 concentration IM |
|
Anaphylactic shock |
0.1 mg of 1:10,000 concentration slow IV during 5 min |
|
Cardiac arrest |
1 mg of 1:10,000 concentration IV push |
|
Reprinted with permission from: Kanwar M, Irvin CB, Frank JJ, et al. Confusion about epinephrine dosing leading to iatrognic overdose: A life-threatening problem with a potential solution. Ann Emerg Med 2010;55:341-344. |
|
For refractory cases with persistent hypotension, intravenous epinephrine at a concentration of 0.01 mg/kg can be infused over 5-10 minutes. An epinephrine drip can be used with 0.1 mg (100 mcg) of epinephrine (either 0.1 mL of a 1:1000 [1 mg/mL] solution, or 1 mL of 1:10,000 [0.1 mg/mL] solution) in 10 mL of NS. This results in a 10 mcg/mL solution that can be run at 2-20 mcg/min infusion, titrating for hypotension and mentation.3,14,17
Fluid Resuscitation. Secondary only to epinephrine, prompt establishment of large-bore multiple-site IV access and aggressive IV fluid volume administration is potentially life-saving. One-liter boluses of isotonic normal saline are preferred and should be administered over 5-10 minutes. Up to 7 L may be required in severe cases.17 In the pediatric population 20 mL/kg boluses are appropriate up to 100 mL/kg. Concomitantly, the patient should be placed supine or in mild Trendelenburg position to maximize venous return.
H1 Blocking Agents. H1-antihistamines are second-line agents in the treatment of anaphylaxis. Their utility in acute treatment has not been established.32 Presently, only diphenhydramine is available in IV form. It may be given as a 50 mg IV dose (1 mg/kg for children). Many other agents are available in oral form and may be utilized for residual hive reduction after epinephrine treatment. These agents do not affect mucosal edema or prevent preformed mediator release once the anaphylactic episode has commenced. Their onset of action is 1-3 hours and they are not recommended as primary treatment. Side effects include drowsiness and possible confusion leading to under-recognition of symptoms.14,32,33 (See Table 3.)
Table 3: Adjunctive Medications for the Treatment of Anaphylaxis
|
Medication (example) |
H1-antihistamines* (oral, such as cetirizine; IV, such as diphenhydramine) |
H2-antihistamines* (ranitidine) |
β2-Adrenergic agonists* (salbutamol [albuterol]) |
Glucocorticoids* (oral, such as prednisone; IV, such as methylprednisolone) |
|
There are no randomized double-blind, placebo-controlled trials of any of these medications in the treatment of acute anaphylaxis episodes. The route of administration of H1-antihistamines and glucocorticoids depends on the severity of the anaphylaxis episode. Adapted from Simons FER. Anaphylaxis: Evidence-based long-term risk reduction in the community. Immunol Allergy Clin North Am 2007;27:231-248. *For use in anaphylaxis. Reprinted with permission from: Simons FER. Anaphylaxis: Recent advances in assessment and treatment. J Allergy Clin Immunology 2009;124:625-636. |
||||
|
Strength of recommendation* |
C |
C |
C |
C |
|
Pharmacologic effects |
At H1-receptor |
At H2-receptor ↓ Gastric acid secretion |
At β2-receptor |
↓ Late-phase allergic response to allergen |
|
↓ Itch (skin, mucus membranes) |
↓ Vascular permeability |
↑ Bronchodilation |
||
|
↓ Flush |
↓ Hypotension |
|||
|
↓ Hives |
↓ Flushing |
|||
|
↓ Sneezing |
↓ Headache |
|||
|
↓ Rhinorrhea |
↓ Tachycardia |
|||
|
↓ Chronotropic and inotropic activity |
||||
|
↓ Mucus production (airway) |
||||
|
Practical aspects |
↓ Itch and hives but not life-saving in anaphylaxis |
Small additive effect (10% or so) when used in conjunction with an H1-antihistamine for ↓ in vascular permeability, ↓ flushing, and ↓ hypotension |
↓ Wheeze, cough, and shortness of breath but do not ↓ upper airway obstruction or relieve hypotension and are not life-saving in anaphylaxis |
Effects take several hours; used to prevent biphasic or protracted anaphylaxis; however, there is no evidence from high-quality randomized controlled trials that this occurs. |
|
Potential adverse effects (usual doses) |
First-generation drugs cause sedation and impair cognitive function. |
Ranitidine: unlikely cimetidine: potentially causes hypotension if infused rapidly |
Tremor, tachycardia, dizziness, jitteriness |
Unlikely to occur during a short 1- to 3-day course |
|
Potential adverse effects (overdose) |
Coma, respiratory depression |
Unlikely |
Headache, hypokalemia |
Unlikely |
|
Comment |
Many different H1-antihistamines and different dose regimens are listed as adjunctive medications in anaphylaxis guidelines. |
Not mentioned in most anaphylaxis guidelines; an H2-antihistamine should not be used alone in anaphylaxis; if used, it should be given with an H1-antihistamine |
Deliver by nebulization and face mask. |
Different glucocorticoids and different dose regimens are used; these medications are unlikely to play a role in the initial minutes to hours of an anaphylactic episode. |
H2 Blocking Agents. Similar to H1 blocking agents, H2 antihistamines are second-line treatment. A single-center, double-blinded randomized controlled trial demonstrated benefit of H2 antihistamines in decreasing urticaria in the emergency setting. No effect was seen on erythema, edema, or vital signs. If given in the acute setting, they should be added to H1 antagonists. They are generally well tolerated with a favorable risk profile. Ranitidine may be given as a 50 mg (1 mg/kg in children) IV dose.33,34 (See Table 3.)
Steroids. Corticosteroids are a second-line treatment for anaphylaxis. Their onset of action is 4-6 hours, and their role is not to prevent release of pre-formed mediators, but rather to prevent creation of newly synthesized mediators. This may assist in preventing prolonged or biphasic anaphylaxis, but has no effect on the acute course of anaphylactic reactions.1,3,14 Reasonable regimens include a one-time dose of either dexamethasone 10 mg IV or methylprednisolone 125 mg (1-2 mg/kg pediatric) IV.33 (See Table 3.)
Oxygen and Adrenergic Agonists. High-flow oxygen given via non-rebreather is appropriate for patients experiencing respiratory distress or hypoxemia. Beta-2 adrenergic agents can assist with bronchospasm, particularly in the asthmatic population. They have no hemodynamic effects and no impact on upper airway edema or obstruction. Inhaled beta-agonist nebulizer treatment with albuterol (2.5-5 mg in 3 mL NS) may be tried for wheezing.33
Glucagon. Theoretically, patients taking beta-blockers may have a blunted response to exogenous epinephrine administration. If these patients have refractory hypotension or bronchospasm, consideration may be given to activating a beta-receptor independent, adenylate cyclase-mediated mechanism of treatment by giving glucagon. The dosing is 1-5 mg over 5 minutes IV. Take care to monitor the airway as emesis is a known side effect.1
Disposition
While patients with prompt resolution of symptoms can be sent home after 6 hours, and those with cardiovascular collapse go to the ICU, a majority of patients fall somewhere in between. No prediction rule will apply to all patients. Consideration for prolonged observation (> 6 hours, up to 12 hours) or for admission include a severe initial presentation (including patients requiring repeated dosing of epinephrine, the need for IV fluid to correct hypotension, patients with laryngeal edema, or those with significant respiratory distress), delayed onset presentation (> 30 min), slow response to treatment, slow progression of symptoms, a history of biphasic anaphylaxis, or high-risk features such as asthma or cardiovascular disease.11,21
Provide thorough discharge instructions and appropriate prescriptions to discharged patients. Patients should not be allowed to leave without a reliable caretaker who can monitor them for signs of symptom recurrence during the first 24 hours after discharge.
Discharge Instructions
The mnemonic SAFE was developed as a guide for emergency department follow-up care. First, patients should Seek support including education resources and calling EMS for recurrence. Second, they should be encouraged to identify the Allergen and learn to avoid it. Third, Follow-up referral is essential with an allergy specialist, and lastly Epinephrine must be prescribed and taught to the patient for emergency use.17,35
In addition to avoiding a known food trigger, patients should be instructed to read food labels. Common hidden sources of food contamination include buffets, restaurants, bake sales, and unlabeled candies.
Those with medication reactions should be made aware of cross-reacting substances, such as the lesser but present risk of cephalosporin allergy in penicillin-allergic patients. Stinging insect-related anaphylaxis patients must be made aware of high-risk situations, including gardening, camping, and outdoor eating. Lastly, patients with less common triggers including exercise-induced anaphylaxis should be made aware of co-triggers including food, medication, ethanol, or cold weather. They should be instructed to carry an epinephrine auto-injector and a cell phone with them when exercising, as well as to exercise with other people if possible.
Additional measures include informing patients that symptoms can recur up to 3 days after the initial event, creating an emergency action plan (http://www.aaaai.org/members/resources/anaphylaxis_toolkit/action_plan.pdf), and encouraging patients to obtain medical identification bracelets.21,36
Prescriptions
The single most important action that can prevent life-threatening recurrence of anaphylaxis is to prescribe an epinephrine auto-injector. If one is present in your emergency department, it can be given to the patient. If not, 2 doses should be prescribed at the time of discharge, as up to 36% of patients will require a second dose.37
While under-prescribed up to 89% of the time, epinephrine is also under-taught when prescribed.38,39 Instruction is important because to be most effective epinephrine should be administered forcefully with a closed fist intramuscularly into the vastus lateralis portion of the midanterolateral thigh for a full 10 seconds. This can be done through clothing. Prompt use at the first sign of symptoms is essential, as delay is associated with fatal outcomes.
Appropriate dosing of epinephrine is 0.01 mg/kg of the 1 mg/mL (1:1000 dilution) solution up to a dosage of 0.5 mg, to be repeated every 5-15 minutes as needed. In the United States, 2 doses of epinephrine are available in auto-injectors, 0.3 mg and 0.15 mg. They are available in the Epipen (0.3 mg), Twinject (two doses of 0.3 mg), Epipen Jr (0.15 mg), and Twinject (0.15 mg x 2) devices.
These set dosages limit their appropriateness of dosage for use in patients less than 15 kg (0.15 mg leads to overdosage) or more than 30 kg (0.3 mg leads to under-dosage). They should still be utilized; however, patients should be informed of the dosing as well as the potential need for a second dose in patients > 30 kg. Parents of infants and children < 10 kg will need to be educated on how to properly dose their child using an ampule and needle syringe. Patients should be instructed to carry an auto-injector with them (some place one in the glove compartment of their car), use it at the onset of symptoms, and call 911 immediately after use.
H1 antihistamines can be prescribed once to twice daily for several days. If done, prescribing second-generation non-sedating ones such as cetirizine or loratidine should be considered. Twice-daily dosed H2 antihistamines are also appropriate, as are once-daily dosed steroids of prednisone 1 mg/kg up to 50 mg. A reasonable course is 3-7 days, as most episodes of recurrent anaphylaxis occur within 72 hours of the initial reaction.
Follow-up Care
Patients with anaphylactic reactions should be referred to an allergist for follow-up care. Skin testing can be performed after 3-4 weeks to allow for mast cell recovery and mediator synthesis. Another option is testing for serum levels of allergen-specific IgE. Patients with Hymenoptera venom-induced anaphylaxis are eligible for subcutaneous venom injections over a 3-5 year course. The risk of allergic reaction or anaphylaxis from this is low, with 88% of patients having no subsequent reaction to treatment.6 In children, up to 98% long-term protection rates may be accomplished.3
If patients are medically complex or have multiple medication allergies, desensitization should be considered. Immunotherapy does not have a role in food-induced anaphylaxis.40
Patients with vaccine-related reactions and negative skin tests may be given the vaccine but should be monitored for an hour afterward. If the offending vaccine was egg-derived, alternative vaccines may be available.
Additional Aspects
Epinephrine Use in the Cardiac Patient. Clinicians have lingering concerns over administering epinephrine in patients with known or suspected cardiac disease. There are no absolute contraindications to the administration of epinephrine in anaphylaxis.36 While overdosing of epinephrine has been associated with myocardial events, physicians should also be aware that untreated anaphylaxis can result in the same complications of infarction and arrhythmias by unmasking subclinical coronary artery disease.15
The heart is a target organ in anaphylaxis, undergoing increased myocardial demand and being influenced by endogenous epinephrine release. Furthermore, coronary plaques are at risk of rupture and thrombus formation due to intra-plaque mast cell degranulation and associated pre-formed mediator release (histamine, PAF, etc.) inducing coronary vasospasm. Exogenous epinephrine treatment can help to ameliorate these risks through beta actions that create increased coronary artery flow via augmented myocardial contractility and diastolic time.3 Epinephrine treatment should not be delayed in this population, although cardiac monitoring/evaluation should be considered.
Expired Epinephrine. Auto-injectors have a recommended one-year expiration date to ensure epinephrine bioactivity. As these devices are expensive, and as many patients forget to replace unused auto-injectors, concern occurs regarding the use of expired syringes. When evaluated 1-90 months after the expiration date, a bioavailability reduction of up to 49% was seen. It is recommended that expired auto-injectors be refilled yearly, especially if precipitants or discoloration are seen, or if extremes of temperature occurred during storage. Nevertheless, in an anaphylactic episode, an outdated auto-injector will retain some efficacy.36,38,41
Systems Issues: Dosing. In the ED setting, multiple concentrations of epinephrine are available. 1:1000 (1 mg/mL or 0.1%) as well 1:10,000 (0.1 mg/mL or 0.01%) cardiac dosed vials exist, as do multi-dose vials. Furthermore, the dosing of epinephrine for cardiac arrest (1 mg push of 1:10,000) is 10 times the recommended dose for anaphylaxis (0.1 mg of 1:10,000 as a 5-10 min push).42 (See Table 2.) Given the high density of decision making in critically ill patients, possible communication inadequacy between physicians and nurses, and the complicated dose calculations including decimals and ratios, the possibility of overdosing and selecting incorrect concentrations is present. Possible solutions include eliminating multi-dose vials and creating clearly colored and labeled pre-filled IM-specific syringes for emergency use in anaphylaxis.43
Areas for Future Research
The absence of reliable laboratory biomarkers for confirmation of clinical impression is an area of active research.44 Tests that can more accurately determine the inciting allergen in idiopathic presentations are needed, as are genetic and proteomic predictors of susceptibility, possibly including biomarker assays. Lastly, non-IgE- and non-immune-mediated anaphylaxis reactions are still poorly understood from pathophysiological and treatment perspectives.1
The majority of studies on anaphylaxis are single-center-based, so multi-center evaluations are needed. Observational studies that more accurately assess incidence and prevalence of anaphylaxis cases are also an area deserving of inquiry.
While randomized placebo-based trials of epinephrine are ethically impossible to conduct, there is the possibility of evaluating other aspects of anaphylaxis treatment including trials of antihistamines and glucocorticoids. In addition, epinephrine dosing (0.3 mg vs. 0.5 mg), type and volume of fluid resuscitation, vasopressor usage, leukotriene inhibitor, and cytokine antagonist actions are all areas for future progress. There is a concern that the standard auto-injector needle length of 1.43 cm may not penetrate deep enough into the muscle to be universally effective. Studies involving a 2.54 cm needle length are in process.15 Lastly, outpatient observational studies of prescribed medications and barriers to their implementation/usage could lead to improved understanding, treatment, and outcomes for patients.
Resources
Patients and parents desiring more information and education can be referred to the following sites:
Food Allergy and Anaphylaxis Network: www.foodallergy.org
American Academy of Allergy, Asthma, and Immunology (Anaphylaxis Emergency Action Plan): http://www.aaaai.org/members/resources/anaphylaxis_toolkit/action_plan.pdf
Auto-injectors: www.epipen.com, www.twinject.com
MedicAlert Jewelry: www.medicalert.org
Summary
Anaphylaxis is a rapid-onset, serious, and potentially life-threatening allergic reaction. It is a spectrum of disease and is not synonymous with skin findings or anaphylactic shock in all cases. It is an under-recognized and undertreated condition. Medications, foods, and insect stings are the most common inciting allergen triggers. The goal of care should be prompt identification and definitive treatment of patients with intramuscular epinephrine and volume resuscitation. Education, auto-injector prescription, and specialist referral are essential to prevent and treat future attacks.
References
1. Sampson HA, Munoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: Summary report-Second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. J Allergy Clin Immunol 2006;117:391-397.
2. Portier P, Richet C. De L'action anaphylactique de certain venins. CR Soc Biol 1902;54:170.
3. Simons, F. Estelle. Anaphylaxis. J Clin Allergy Immunol 2010;125:S161-181.
4. Kelso JM, Li JT, Nicklas RA, et al. Adverse reactions to vaccines. Ann Allergy 2009;103:S1-S16.
5. Ledford D, Feldweg A, Adkinson N. Perioperative anaphylaxis: Clinical manifestations, etiology, and diagnosis. Uptodate 2010;18.2: 1-17.
6. Freeman T. Hypersenstivity to Hymenoptera stings. N Engl J Med 2004;351:1978-84.
7. Kemp S. Pathophysiology of anaphylaxis. Uptodate.com. 2010, version 18.2;1-24.
8. Lieberman P, Camargo CA Jr, Bohlke K, et al. Epidemiology of anaphylaxis: Findings of the American College of Allergy, Asthma and Immunology Epidemiology of Anaphylaxis Work-ing Group. Ann Allergy Asthma Immunol 2006;97:596–602.
9. Bock S. Fatal anaphylaxis. Uptodate.com. 2010, version 18.2, 1-17.
10. Sampson H, Feldweg A, Sicherer S, et al. Food-induced anaphylaxis. Uptodate.com 2010. 18.2; 1-17.
11. Soar J, Pumphrey R, Cant A, et al. Emergency treatment of anaphylactic reactions Guidelines for healthcare providers. Resuscitation 2008;77:157-169.
12. Lane R, Bolte R. Pediatric anaphylaxis. Pediatric Emerg Care 2007;23:49-60.
13. Burks W, TePas E, Sicherer S. Clinical manifestations of food allergy: An overview. Uptodate.com 2010;18.2;1-15.
14. Davis J. Just red or near dead: Allergies or anaphylaxis? American College of Emergency Physicians Scientific Assembly 2010 Lecture. 9/29/10.
15. Simons FE. Anaphylaxis: Recent advances in assessment and treatment. J Allergy Clin Immunol 2009:124:625-635.
16. Sampson H. Peanut allergy. N Engl J Med 2002;346;1294-1299.
17. Simons FE, Feldweg A, Bochner B. Anaphylaxis: Rapid recognition and treatment. Uptodate.com 2010; 18.2:1-39.
18. Simons F, Feldweg A, Bochner B. Differential diagnosis of anaphylaxis in children and adults. Uptodate.com 2010. 18.2; 1-12.
19. Schatz M, Feldweg A, Barss V, et al. Anaphylaxis in pregnant and breastfeeding women. Uptodate.com 2010;18.2: 1-22.
20. Greenberger PA, Rotskoff BD, Lifschultz B. Fatal anaphylaxis: Postmortem findings and associated comorbid diseases. Ann Allergy Asthma Immunol 2007;98:252–257.
21. Lieberman PL, Feldweg A, Simons F. Biphasic and protracted anaphylaxis. Uptodate.com 2010;18.2; 1-12.
22. Ellis AK, Day JH. Incidence and characteristics of biphasic anaphylaxis: A prospective evaluation of 103 patients. Ann Allergy Asthma Immunol 2007;64:64-69.
23. Lieberman P. Biphasic anaphylactic reactions. Ann Allergy Asthma Immunol 2005;95:217-226.
24. Kemp SF, Lockey RF, Simons FER, and on behalf of the World Allergy Organization ad hoc Committee on Epinephrine in Anaphylaxis (2008). Epinephrine: The drug of choice for anaphylaxis. A statement of the World Allergy Organization. Allergy 2008;63:1061–1070.
25. Sheikh A, Shehata YA, Brown SGAet al. Adrenaline for the treatment of anaphylaxis. Cochrane Database Syst Rev. Allergy 2009;64:204–212.
26. Soar, et al. Emergency treatment of anaphylactic reactions Guidelines for healthcare providers. Resuscitation 2008;77:157-169.
27. Alrasbi M, Sheikh A. Comparison of international guidelines for the emergency medical management of anaphylaxis. Allergy 2007;62:838–841.
28. Estelle F, Simons R, Roberts JR, et al. Epinephrine absorption in children with a history of anaphylaxis. J Allergy Clin Immunol 1998;101:33-37.
29. Gu X, Simons FER, Simons KJ. Epinephrine absorption after different routes of administration in an animal model. Biopharmaceutics & Drug Disposition 1999;20:401–405.
30. Estelle F, Simons R, Xiaochen G, et al. Epinephrine absorption in adults: Intramuscular versus subcutaneous injection. J Allergy Clin Immunol 2001;108:871-873
31. Simons FE. Advances in H1-antihistamines. N Engl J Med 2004;351:2203-2217.
32. Sheikh A, Ten Broek V, Brown SG, et al. H1-antihistamines for the treatment of anaphylaxis: Cochrane Database Syst Rev 2007, Issue 1.
33. Lieberman P, Nicklas R, Oppenheimer J et al. The diagnosis and management of anaphylaxis: An updated practice parameter. J All Clin Immunol 2010;126:3;477-480e41.
34. Lin RY, Curry A, Pesola G. Improved outcomes inpatients with acute allergic syndromes who are treated with combined H1 and H2 antagonists. Ann Emerg Med 2000;36:462-468.
35. Lieberman P, Decker W, Camargo C, et al. SAFE: A multidisciplinary approach to anaphylaxis education in the emergency department. Ann Allergy Asthma Immunol 2007;98:519-523.
36. Sicherer S, Feldweg A, Somins FE. Precribing epinephrine for anaphylaxis self-treament. Uptodate.com 2010;V18.2 :1-14.
37. Kelso JM. A second dose of epinephrine for anaphylaxis: How often needed and how to carry. J Allergy Clin Immunol 2006;117:464-564.
38. Lieberman P. Use of epinephrine in the treatment of anaphylaxis. Curr Opin Allergy Clin Immunol 2003;3:313-318.
39. Clark S, Kamarthi G, Okafo T, et al. Multi-center study of emergency dpartment visitsfor bee sting allergy [abstract]. J Allergy Clin Immunol 2002;109:S270.
40. Kay AB. Allergy and allergic diseases: Second of two parts. N Engl J Med 2001;344:109-113.
41. Simons FE, Gu X, Simons KJ. Outdated epipen and epiepn jr autoinjectors: Past their prime? J Allergy Immunol 2000;105:1025-1030.
42. Vanden Hoek T, Morrison L, Shuster M, et al. AHA Guidelines for CPR and emergency cardiovascular care. Circulation 2010;122:S832-833.
43. Kanwar M, Irvin C, Frank J, et al. Confusion about epinphrine dosing leading to iatrogenic overdose: A life threatening problem with a potential solution. Ann Emerg Med 2010;55:341-344.
44. Burks A. Factoring PAF in anaphylaxis. N Engl J Med 2008;358:79-81.
Anaphylaxis is a rapid-onset and potentially life-threatening allergic reaction.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
