Abdominal Pain in Special Populations
Abdominal Pain in Special Populations
Authors:
Angela M. Mills, MD, Assistant Professor of Emergency Medicine, Department of Emergency Medicine, University of Pennsylvania, Philadelphia.
Esther H. Chen, MD, Associate Professor of Emergency Medicine, Department of Emergency Medicine, University of California–San Francisco, San Francisco General Hospital.
Peer Reviewer:
Catherine A. Marco, MD, FACEP, Professor, Emergency Medicine, Program Director, Emergency Medicine Residency, University of Toledo College of Medicine, OH.
Introduction
A common presenting complaint for patients seeking emergency medical care is acute abdominal pain. Although difficult to diagnose in healthy patients, it is even more challenging in special populations. This article will focus on three distinct populations: patients with altered immunologic function, pregnant women, and post-procedural patients. In immunocompromised patients, the detection of life-threatening abdominal disease may be delayed because of atypical presentations. Atypical symptoms in late pregnancy (as the intra-abdominal contents are displaced) also may delay diagnosis. Furthermore, the anchoring effect of focusing on obstetrical causes of pain in pregnant women also may delay the diagnosis of non-obstetrical, surgical causes of abdominal pain. In post-procedural patients, understanding the complications specific to the individual procedures is necessary for the emergency physician to prioritize the differential diagnosis and order the appropriate diagnostic studies. This article will highlight the abdominal pain characteristics and diagnoses that are unique to these populations and review the clinical presentation, diagnostic evaluation, and management in these patients.
Patients with Altered Immunologic Function
Patients with altered immunologic function range from those with mild immunologic dysfunction (e.g., elderly, diabetic, uremic) to those with moderate to severe immunologic compromise (e.g., acquired immunodeficiency syndrome [AIDS], transplanted patients, active malignancy undergoing chemotherapy). While the differential diagnosis of abdominal pain is similar to that in the general population, these patients may exhibit delayed or atypical presentations of these conditions. Furthermore, transplanted organs lack native innervation, so pain may not even be a reliable sign of pathology in these patients.1 Patients may lack the classic signs of peritonitis and, instead, present with altered mental status and tachycardia.2 Abdominal pain in the moderate to severely immunocompromised group may be caused by opportunistic infections, such as cytomegalovirus (CMV) infection, neutropenic enterocolitis (typhlitis), and intra-abdominal abscesses. Finally, because they are challenging and more likely to have life-threatening disease means that immunosuppressed patients will often receive advanced imaging with abdomen-pelvis computed tomography (CT) as part of their emergency department (ED) evaluation.
HIV/AIDS. Patients with human immunodeficiency virus (HIV) and CD4 counts greater than 200/mm3 are typically considered to have mild to moderate immunologic dysfunction, whereas those with CD4 counts less than 200/mm3, defined as having AIDS, are severely compromised and have a high risk of opportunistic infection and the resultant complications. Highly active antiretroviral therapy (HAART) has greatly diminished the incidence of opportunistic and AIDS-related gastrointestinal infections. In a retrospective study of 108 patients with HIV and undifferentiated abdominal pain (84% on HAART, 44% with AIDS),3 only 7% of patients (10% of those with AIDS) were diagnosed with an opportunistic infection, compared to rates of 41-86% of patients with abdominal pain in the pre-HAART era.4-6 Disseminated mycobacterial disease was the most common diagnosis in this study population; other opportunistic infections included Candidal esophagitis, AIDS cholangiopathy, lymphoma, and intra-abdominal tuberculosis. Well-described causes of abdominal pain in the HIV population, such as cryptosporidiosis, CMV colitis, and Kaposi's sarcoma, were not found in this study group. Moreover, only 9 patients (8%) required operative intervention. Of these, one patient had an abscess caused by Mycobacterium, the only HIV-associated infection. A more recent study of opportunistic gastrointestinal diseases in HIV patients confirmed these findings, demonstrating that 26% of patients on HAART (vs. 80% in the non-HAART group) had an opportunistic infection as the etiology of their pain, most commonly Candidal esophagitis and CMV esophagitis or colitis.7 Lymphoma and Kaposi's sarcoma were less common than expected.
The principal causes of abdominal pain in HIV patients that require operative intervention are appendicitis, cholecystitis, bowel obstructions, and intestinal perforation.8 While the incidence of these conditions is similar in HIV patients on HAART compared to non-HIV patients, the underlying pathology may differ between these groups.7,9 For example, CMV infection in the terminal ileum and colon may cause vasculitis of the bowel wall, which can lead to gastrointestinal hemorrhage, bowel perforation, or toxic megacolon.10 CMV infection also may cause appendicitis, but with more indolent symptoms. Bowel obstruction and intussusception in HIV patients may be due to lymphoma or Kaposi's sarcoma.8,9 While gallstone cholecystitis occurs with similar frequency among HIV and non-HIV patients, acalculous cholecystitis is more frequently found among those with HIV. Patients with HIV also may develop cholangiopathy as an adverse effect of antiretroviral medications or from biliary duct infiltration by opportunistic pathogens leading to biliary obstruction.9
In addition to cholangiopathy, nonspecific abdominal pain is an adverse effect of the same antiretroviral medications that have so successfully decreased gastrointestinal disease in this population. HAART can cause nausea, vomiting, and diarrhea, sometimes severe enough to cause acute dehydration or hemodynamic instability. Certain medications also are associated with specific causes of abdominal pain. Didanosine (Videx) is known to cause acute pancreatitis, and indinavir (Crixivan) may cause nephrolithiasis because it can precipitate in the kidneys.8 These side effects can make the medication regimens so intolerable that some patients discontinue the treatment entirely. Unfortunately, these lapses in treatment also increase the patient's risk of opportunistic infections.
Malignancy. Approximately 40% of ED visits by patients with active cancer are for the evaluation of abdominal pain.11 Possible causes of abdominal pain include conditions directly related to the malignancy in addition to complications from the treatment of the malignancy.12 While intra-abdominal malignancies may mechanically cause small or large bowel obstructions depending on their size and location,13 extra-abdominal malignancies (i.e., breast, melanoma) complicated by diffuse peritoneal carcinomatosis can also cause intestinal obstruction.14 Detection of a gastric outlet or large bowel obstruction in a patient who is otherwise healthy or with a prior cancer history should raise the suspicion of an undiagnosed underlying or recurrent cancer.11,13 Moreover, an uncommon condition in healthy adults, such as intussusception, may be a result of a solid tumor acting as a lead point.15 Intestinal perforation may be caused by the transmural erosion of gastrointestinal cancers, metastatic lesions to the bowel, and atypical infections. Pneumoperitoneum from bowel perforation may be challenging to diagnose in these patients because they may not exhibit peritonitis.2 Other causes of abdominal pain include malignant ascites and Budd-Chiari syndrome, a constellation of symptoms due to hepatic venous outflow obstruction secondary to thrombosis.12
Advanced imaging often is necessary for diagnosis in this patient population. Abdominal CT enables the emergency physician to more accurately identify closed-loop or strangulation obstruction, detect an intestinal transition point, and demonstrate vascular thrombosis or bowel pneumatosis.12 By contrast, plain radiography has a sensitivity of 66% in detecting small-bowel obstruction16 and is unable to detect conditions such as strangulated bowel and vascular thrombosis.17
Cancer treatments have well-documented adverse effects. Radiation therapy to the abdomen or pelvis may produce localized intestinal luminal narrowing and progressive occlusive vasculitis. Radiation enteritis comprises a spectrum of diseases, ranging from acute bowel perforation and gastrointestinal hemorrhage to chronic fistulas and strictures.18 Chemotherapeutic agents frequently cause abdominal pain associated with nausea, vomiting, and/or diarrhea and also may cause profound neutropenia. The most common cause of an acute abdomen in neutropenic patients is neutropenic enterocolitis (NEC), also known as typhlitis.19 NEC is a severe necrotizing inflammation of the cecum and adjacent small bowel occurring in profoundly immunocompromised patients, most commonly with chemotherapy-induced neutropenia or bone-marrow transplantation. NEC also has been described in other patient groups with altered immunologic function, including those with AIDS, aplastic anemia, and organ transplantation. In addition, patients exposed to cytotoxic agents, such as vinorelbine (Nabelbine) and the taxanes, have an increased risk of developing NEC.20
Patients with NEC may present with fever, nausea, vomiting, diarrhea, abdominal distension, and right lower-quadrant abdominal pain. They also may present acutely with hypotension and other signs of sepsis. Although these symptoms can be similar to the routine side effects of the chemotherapeutic agents themselves, NEC is more likely to be associated with bowel wall thickening on CT of more than 4 mm; wall thickening of greater than 10 mm portends a very poor prognosis.21,22 Treatment for NEC includes fluid resuscitation, broad-spectrum antibiotics (against enteric gram-negative bacilli, Enterococcus spp., and anaerobes), bowel rest, and parenteral nutrition.20 Surgery is indicated for bowel ischemia and perforation.23 Mortality rates are high (30-50%),24 so prompt recognition may be life-saving.
Solid Organ Transplantation. Like many postsurgical patients, patients who have received a solid organ transplant frequently visit the ED for abdominal pain (31%), associated gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea), and infectious symptoms (17%) such as fever or abnormal wound drainage.1 Regardless of the time lapsed since transplantation, infection (36%) and gastrointestinal or genitourinary pathology (20%) are the most common ED diagnoses. Organ rejection, infection, and drug toxicity must be considered in the post-transplant patient who presents with acute abdominal pain.
The differential diagnosis is dependent upon the time elapsed since transplantation. In the early post-transplant period (< 1 month), post-surgical complications and infections are most common.1 The surgical anastomosis may constrict or leak, causing intestinal obstruction or peritonitis, respectively. Graft injury, such as bile duct ischemia, can develop into a liver abscess. Viral or candidal infections may be derived from the donor organ or a complication of the surgery itself. Clostridium difficile colitis is common during this period, whereas opportunistic infections typically are absent.25 Finally, although graft rejection could occur at any period post-transplant, patients have a slightly higher risk of graft rejection within the first month than in the intermediate or late post-transplant period.1
In the intermediate post-transplant period (1-6 months), viral infections and graft rejection are frequently seen and are the most common reasons for fever.25 During this period, patients suffer from the full effect of immunosuppressants and may develop opportunistic infections, such as CMV colitis and intra-abdominal abscesses caused by fungal (e.g., Candida albicans, Cryptococcus) and bacterial infections (e.g., Nocardia, Legionella). These patients should be considered to be severely immunodeficient and should undergo a thorough search for the source of infection.
Immunosuppressive therapy is routinely tapered down after 6 months in transplant patients with good graft function. The risk of infection declines slightly in this late post-transplant period, although patients continue to be at risk for developing chronic rejection as a result of chronic viral infections.25 In addition, acute diverticulitis is a common gastrointestinal infection seen during this period. This population also may develop post-transplantation lymphoproliferative disorder (PTLD), a lymphoproliferative disorder thought to be associated with Epstein-Barr virus infection and similar to disorders occurring in patients with AIDS. Patients may present with fever, a mononucleosis-like syndrome, gastrointestinal obstruction, bleeding or perforation, and have significant hepatic or pancreatic dysfunction. Disease regression may occur simply by decreasing immunosuppressant therapy, although this disease often requires additional chemotherapy or immunotherapy.25
Pregnancy
Abdominal pain in the pregnant patient poses a unique challenge to the clinician. The differential diagnosis is much broader in pregnant patients than non-pregnant patients. Although both obstetrical and non-obstetrical diseases must be considered, anchoring bias can delay the diagnosis of non-obstetrical surgical conditions by focusing attention only to the potential obstetrical causes of the pain, such as ectopic pregnancy, ovarian torsion, and labor. The clinical presentation and natural progression of many diseases that cause abdominal pain may be altered in pregnancy. CT, now a routine diagnostic tool to evaluate abdominal pain in non-pregnant patients, is often avoided in pregnant women because of the radiation risk to the fetus. This next section of the article will focus on common non-obstetrical etiologies of the acute abdomen in pregnancy and the unique characteristics of disease presentation and progression in this distinct population.
The normal physiologic changes that occur during pregnancy can make diagnosing and managing these patients very challenging. For example, blood plasma volume increases out of proportion to red blood cell mass and creates a state of "physiologic" anemia. Therefore, pregnant women may not exhibit signs of shock (i.e., tachycardia, hypotension) until they have lost a significant amount of blood. In addition, leukocytosis becomes an unreliable sign of infection because it can be normally elevated in pregnancy.26 Gastrointestinal symptoms that typically are associated with some surgical conditions such as nausea, vomiting, anorexia, and constipation can be normal in pregnancy. Particularly in late pregnancy, signs of peritonitis (i.e., abdominal guarding, rigidity, rebound tenderness) can be obscured by a laxity of the abdominal musculature.27 All of these factors may contribute to the diagnostic delay in surgical conditions and increased morbidity in this population.
General Management. The most important concept for the emergency physician to remember in managing the pregnant patient is how to interpret the symptoms, signs, and laboratory values that can change during pregnancy. Whereas the diagnosis of the acute abdomen can be challenging, the management of pregnant patients with abdominal pain is similar to that for non-pregnant patients, with a few exceptions. For pregnancies greater than 24 weeks gestation, or the time at which the fetus is considered to be viable, fetal heart-rate monitoring is recommended during the patient's evaluation.28 In addition, early obstetrical consultation is recommended for severely injured patients (gestation > 24 weeks), while mildly injured patients can receive their trauma evaluation in the ED and then be sent for fetal heart-rate monitoring by Obstetrics. Medications such as analgesia and antibiotics should not be withheld unnecessarily in pregnant patients, although specific drugs should be checked for potential teratogenicity and adverse perinatal effects. Opioid analgesia (parenteral or oral) is considered to be safe during pregnancy, whereas aspirin and nonsteroidal anti-inflammatory drugs should be avoided. Likewise, antibiotics such as penicillins, cephalosporins, clindamycin, ertapenem, and metronidazole may be safely administered to pregnant patients, unlike fluoroquinolones or tetracyclines.
Imaging. The diagnostic use of CT for pregnant women is a controversial issue. Although ultrasonography and magnetic resonance imaging (MRI) are safe alternatives to CT, ultrasound is more limited in its diagnostic accuracy for certain conditions and MRI is often not as readily available. In pregnant patients with acute abdominal pain, MRI has been found to have a high negative predictive value (98%) in excluding diagnoses requiring surgical or interventional therapy.29 In addition to excluding disease, both MRI and CT often are able to provide an alternative diagnosis. Some physicians believe that CT should be used if the benefits outweigh the risk of imaging, particularly when delaying the diagnosis increases the morbidity or mortality for the patient. A recent retrospective study of abdomen-pelvis CTs performed between 1998 and 2005 showed that CT utilization increased more in pregnant women compared to the general population (about 22%/year/1,000 deliveries vs. 13%/year), although this was not statistically significant.30 The most common indication for CT was suspected appendicitis. In a survey of radiology residency programs in the United States, 96% of respondents indicated that they will perform CT in pregnant patients with abdominal pain when the benefit/risk ratio is high.31 Respondents also preferred CT to MRI in the second and third trimesters for the evaluation of appendicitis, nephrolithiasis, and abscess; MRI was preferred for the detection of appendicitis and abscess in the first trimester. Most radiology departments have policies regarding the use of imaging in the pregnant patient. However, there is no consensus for an imaging algorithm for evaluating pregnant women with abdominal pain.
Appendicitis. Acute appendicitis is the most common non-obstetric surgical disease in the pregnant population, with an incidence of approximately 1 in 750 to 3000 pregnancies, similar to the non-pregnant population.28 While appendicitis may occur at any time during pregnancy, it occurs more frequently in the second trimester.32,33 The differential diagnosis includes ectopic pregnancy, ovarian torsion, and labor. The rate of appendiceal rupture also has been reported to be higher in the pregnant population, in part because of the diagnostic challenges mentioned previously.32,34 Both early delivery and fetal loss are more common in complicated appendicitis, although fetal loss still occurs with negative appendectomy, stressing the importance of accurate diagnosis.35 Maternal mortality from appendicitis has decreased significantly in recent decades.28
Although the classic teaching is that the pain of appendicitis moves from the right lower quadrant to the right upper quadrant as the uterus displaces the intra-abdominal contents, right lower-quadrant abdominal pain is still the most common presenting symptom regardless of gestational age.32,36 As in the nongravid patient, fever and leukocytosis are unreliable indicators of appendicitis.
In the first trimester, ultrasonography is the initial study of choice because the radiation risk to organogenesis is highest in early pregnancy. Ultrasound is more available than MRI. In pregnant patients, ultrasound has been shown to have a sensitivity of 67% to 100% and a specificity of 83% to 96%.37 The sonographic finding for appendicitis is a noncompressible tubular, multilayered structure greater than 6 mm in diameter. (See Figure 1.) A positive study requires no further testing. A negative or indeterminate study may be followed by MRI, CT (if more advanced gestational age), or inpatient observation with serial abdominal exams. Ultrasound can be technically difficult in the third trimester, so its utility in late pregnancy is more limited. A comparison of CT and MRI after prior normal/inconclusive ultrasound showed a sensitivity and specificity of 85.7% and 97.4%, respectively, for CT and 80% and 99%, respectively, for MRI.38 (See Figure 2.) When available, MRI is the preferred imaging study in evaluating pregnant patients with suspected appendicitis. Like CT, it can detect other surgical causes of abdominal pain, with an overall sensitivity of 89% and specificity of 95% for detecting surgical causes of abdominal pain in pregnancy.29 All imaging modalities have limitations and may be indeterminate if the appendix is not visualized.
Figure 1: Appendicitis on Ultrasound
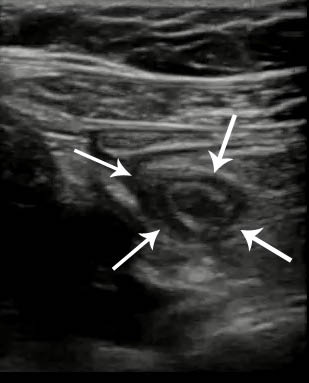
Ultrasound image in the short axis plane of an inflamed appendix (outlined by arrows). It is often described as a "target" sign when viewed in short axis. (Image provided by Nova L. Panebianco, MD, University of Pennsylvania, Philadelphia, PA, published with permission.)
Figure 2: Appendicitis on MRI
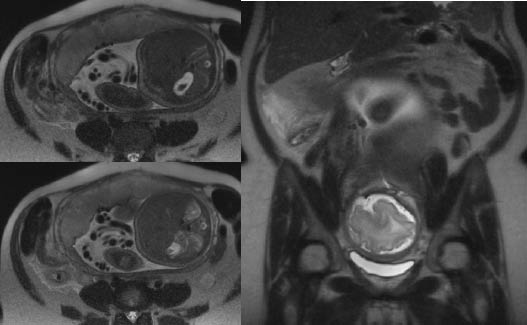
Axial (left) and coronal (right) T2 weighted MRI images through the right lower quadrant in a pregnant patient at 24 weeks gestation demonstrating a distended, thick-walled appendix with periappendiceal fluid (arrows). There is a low signal structure within the appendix, likely an appendicolith or focus of gas. (Image provided by Elizabeth M. Hecht, MD, University of Pennsylvania, Philadelphia, PA, published with permission.)
Treatment for appendicitis is the same in pregnant and non-pregnant patients: intravenous hydration, symptomatic relief with analgesia and antiemetics, antibiotic administration, and operative intervention. Early surgical consultation is recommended.
Acute Biliary Disease. The second most common non-obstetric surgical emergency in pregnancy is acute cholecystitis. Biliary disease is already more common in females, with gallstones present in almost 20% of women by 40 years of age.39 Estrogen increases cholesterol synthesis and progesterone impairs gallbladder emptying and promotes cholestasis.40 However, despite the higher risk of cholestasis, the incidence of acute cholecystitis is no different in pregnant patients compared to non-pregnant patients.
The symptoms of biliary disease in pregnancy are similar to those in the non-pregnant population. They include mid-epigastric or right upper-quadrant abdominal pain, nausea and vomiting, anorexia, and intolerance of fatty foods. Physical examination often reveals right upper-quadrant tenderness and Murphy's sign; jaundice suggests choledocholithiasis and obstruction.
Laboratory values must be interpreted differently in pregnant women. As mentioned previously, the white blood cell count may be normally elevated in pregnancy, as well as alkaline phosphatase. Elevated lipase and amylase suggest associated pancreatitis.
Similar to non-pregnant patients, the differential diagnosis for right upper-quadrant pain is broad and should include pyelonephritis, renal colic, peptic ulcer disease, pancreatitis, appendicitis, viral hepatitis, right lower lobe pneumonia, myocardial infarction, and herpes zoster. Specific conditions to consider in pregnancy are acute fatty liver of pregnancy and hemolytic anemia/elevated liver enzymes/low platelet count (HELLP syndrome).
Ultrasonography is the diagnostic test of choice. Sonographic findings of acute cholecystitis include gallstones (especially if present in the gallbladder neck), pericholecystic fluid, thickened gallbladder wall, common bile duct dilatation, and sonographic Murphy's sign. (See Figure 3.) For more than a decade, emergency physicians have been performing bedside ultrasonography, and the American College of Emergency Physicians (ACEP) formally endorses and supports this modality for right upper-quadrant ultrasonography.41 The sensitivity for gallstone detection by emergency physicians is 90-96%, with a specificity of 88-96%. In addition, pregnant patients with choledocholithiasis and pancreatitis may undergo endoscopic retrograde cholangiography (ERCP) with sphincterotomy, which may be performed successfully with little to no radiation exposure.42
Figure 3: Cholecystitis on Ultrasound
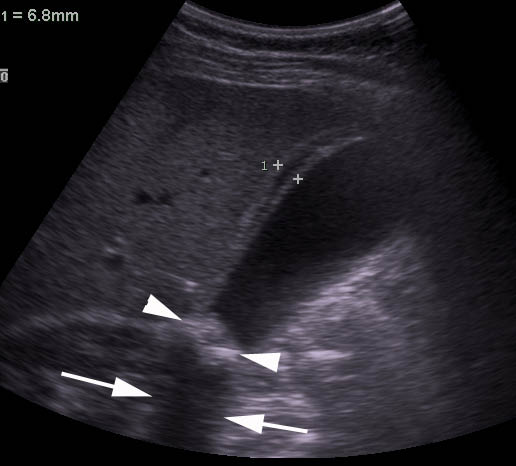
Ultrasonography in longitudinal view demonstrating cholecystitis, specifically gallstones (arrowheads) impacted in the gallbladder neck with posterior shadowing (arrows), and a thickened gallbladder wall with intramural edema (calipers). (Image provided by Anthony J. Dean, MD, University of Pennsylvania, Philadelphia, PA, published with permission.)
Treatment with intravenous hydration, analgesia, antiemetics, and antibiotics is appropriate in the ED. Early surgical consultation is recommended. Historically, the management of pregnant patients with acute biliary disease, particularly symptomatic cholelithiasis, favored conservative treatment; more recently, however, early surgical intervention has been advocated. Delayed cholecystectomy for the pregnant patient with biliary disease has been shown to have increased short- and long-term morbidity, including symptomatic recurrence, longer hospital stays, preterm labor, premature delivery, and fetal loss.43-45 Laparoscopic cholecystectomy has been shown to be safe during pregnancy.44
Bowel Obstruction. Small-bowel obstruction occurs in approximately 1 in 3000 pregnancies, and as in non-pregnant patients, is most commonly caused by adhesions (60-70%).46 Volvulus is significantly more common in pregnancy, accounting for 25% of intestinal obstruction in pregnant patients, compared to 3-5% in non-pregnant patients.47 Volvulus is more likely to occur during times of rapid change in uterine size, such as the early second trimester and the post-partum period. Other less common causes of intestinal obstruction include intussusception, hernia, and malignancy. In pregnant patients who receive operative intervention for obstruction, fetal mortality has been documented at 26% and maternal mortality at 6%.48
The clinical diagnosis of bowel obstruction may be especially challenging in pregnancy. The usual symptoms of crampy abdominal pain, nausea, vomiting, and constipation may be attributed to the normal pregnancy-related symptoms. A large gravid uterus may obscure the physical examination findings of abdominal distension and tenderness. Laboratory tests are often unhelpful in making the diagnosis. Plain abdominal radiography may demonstrate the typical air-fluid levels, progressive bowel dilatation in serial films, or the coffee-bean deformity found with volvulus. (See Figure 4.) However, plain films have a limited sensitivity and specificity for the diagnosis of small-bowel obstruction and may appear normal with closed-loop or strangulated obstructions. If the clinical suspicion is high, more advanced imaging, such as CT or MRI, is better than plain radiography at detecting obstruction. There is a paucity of studies comparing the diagnostic accuracy of CT and MRI. One small prospective study of 28 confirmed bowel obstructions showed a sensitivity and specificity for MRI of 95% and 100%, respectively, and 71% and 71% for helical CT.49
Figure 4: Volvulus on Plain X-ray
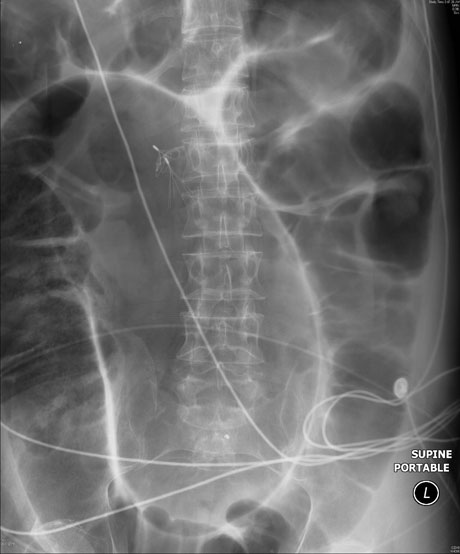
Plain abdominal radiograph demonstrating diffuse distension of the large colon with a "coffee bean" sign suggestive of sigmoid volvulus. (Image provided by Angela M. Mills, MD, University of Pennsylvania, Philadelphia, PA, published with permission.)
Management of intestinal obstruction is similar in pregnant and non-pregnant patients. Fluid resuscitation, electrolyte replacement, bowel rest, and gastric decompression with a nasogastric tube are the mainstays of initial therapy. Operative intervention is indicated for intestinal perforation, gangrenous bowel, or failure of medical management.
Patients Who Undergo Percutaneous Interventions
Percutaneous interventions are minimally invasive procedures performed by both interventional radiologists and non-interventionalists. Abdominal pain and other gastrointestinal symptoms are common reasons for ED visits post-procedure. This portion of the article will focus on the gastrointestinal complications of several common procedures.
Vena Cava Filters. Potential complications of inferior vena cava (IVC) filters include malposition, fracture, infection, access-site thrombosis, IVC thrombosis, and recurrent deep vein thrombosis (DVT). As such, a device-related complication may cause acute abdominal or flank pain in a patient with an IVC filter. Compared to filters placed within the prior six years, older permanent filters are more likely to migrate or fracture (5-30% [migration] and 2% [strut fracture] vs. 0.3-3% and 0%, respectively).50-52 While IVC filter or strut migration may be asymptomatic, patients may also sustain ischemic or hemorrhagic end-organ dysfunction due to vessel obstruction or vessel perforation, respectively. Through-and-through perforation of the IVC is rare. Newer filters have improved anchoring and are less likely to migrate.50,52 When filter migration or significant IVC penetration does occur, patients may present with sharp, tearing flank or groin pain and may have fever or signs of organ damage, such as small-bowel obstruction, duodenal perforation, or gastrointestinal bleeding. CT imaging confirms diagnosis, and urgent consultation with interventional radiology for filter removal is recommended.
Percutaneous Gastrostomy Tubes. Gastrostomy, gastrojejunostomy, and jejunostomy tubes placed percutaneously may be complicated by both major and minor conditions. Major complications occur in approximately 6% of patients and include gastric perforation or hemorrhage, peritonitis due to peri-catheter leakage of gastric contents, deep stomal infection or abscess, and unintentional injury to adjacent organs.53,54 These complications occur within a few days following the intervention, and patients may present with acute abdominal pain associated with fever or signs of severe sepsis. A case series of 370 patients post-gastrostomy-tube placement demonstrated four major complications in patients with significant co-morbidities. Minor complications, occurring in about 8% of patients, include peristomal leakage with skin excoriation, superficial wound infection, and tube malfunction (e.g., dislodgement, balloon rupture, blockage).53,54 Patients with a suspected major complication of a percutaneous gastrostomy tube often require CT for diagnosis. Early consultation with surgery or interventional radiology is indicated for treatment.
Patients also may present to the ED to have a dislodged tube replaced. As feeding tube tracts can narrow or close within hours of removal, tubes should be replaced in a timely fashion unless the tube was recently inserted. A percutaneous endoscopic gastrostomy (PEG) tube requires approximately one to two weeks after placement (up to four weeks in a patient with altered immunologic function) to form a tract.55 Replacement of the tube in an immature tract can lead to misplacement into the peritoneal cavity. Information on the type of tube (e.g., gastrostomy, jejunostomy) and the date of placement can help guide ED management. Partially dislodged tubes may need removal prior to replacement; while most tubes may be removed easily, some may require endoscopic removal. If there is doubt, the provider who placed the tube should be contacted to guide management. Tube replacement may be confirmed by obtaining a plain supine abdominal radiograph after injecting water-soluble contrast through the tube. If a feeding tube is not available, a Foley catheter may be used temporarily to prevent closure of the tract.56
Transvenous Hepatic and Renal Biopsy. Transvenous hepatic and renal biopsies are most commonly performed when there is a contraindication to or failed percutaneous biopsy. The majority of complications from transhepatic biopsies are detected prior to discharge from the procedure and require no intervention.57 Delayed development of a perihepatic hematoma may occur post liver biopsy. Patients present to the ED with abdominal pain radiating to the right shoulder. Right upper-quadrant ultrasonography may be performed to make the diagnosis. (See Figures 5A and 5B.) Acute intervention is indicated for hemodynamic instability or a life-threatening decrease in the hematocrit level. While transient fever may be commonly seen up to 24 hours after the procedure,58 persistent fever associated with gastrointestinal symptoms is concerning for possible intra- or peri-hepatic abscess and needs further evaluation.
Figure 5A: Perihepatic Hematoma on Ultrasound
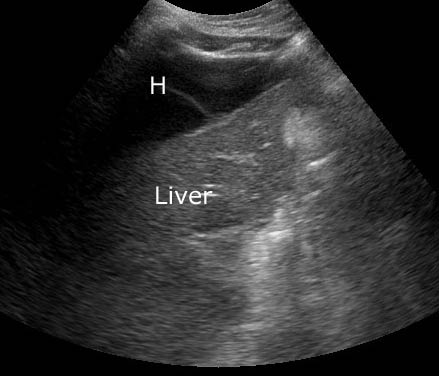
This ultrasound image demonstrates perihepatic hematoma (H) following transvenous hepatic biopsy. (Image provided by Angela M. Mills, MD, University of Pennsylvania, Philadelphia, PA, published with permission.)
Figure 5B: Perihepatic Hematoma on CT
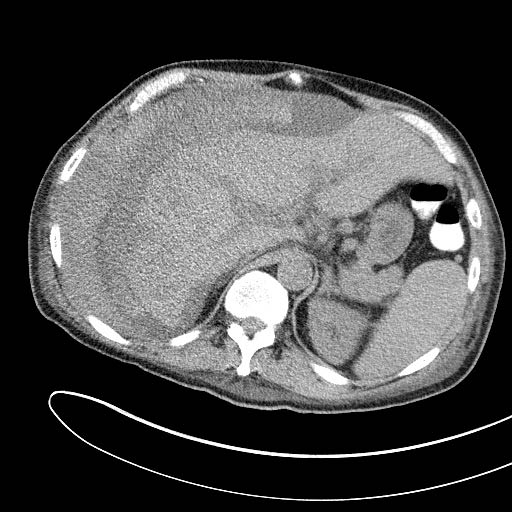
Computed tomography (CT) of the same patient showing perihepatic hematoma post transvenous hepatic biopsy. (Image provided by Angela M. Mills, MD, University of Pennsylvania, Philadelphia, PA, published with permission.)
Complications of transjugular renal biopsies also do not necessitate acute intervention. The majority of patients (66%) develop gross hematuria due to a perinephric hematoma or calyceal hemorrhage, which resolves spontaneously.59,60 Significant hematuria and symptomatic anemia may be caused by an arterio-venous or arterio-calyceal fistula in patients post transvenous biopsy of the kidney.61 These patients may complain of abdominal pain and distension due to clot retention or urethral obstruction. Manual bladder irrigation through a large-bore (20 French or larger) Foley catheter is indicated for these patients. Continuous bladder irrigation may be used, as clots may cause obstruction of the catheter, leading to bladder perforation. Patients with asymptomatic anemia in the absence of hematuria also may present with abdominal or flank pain due to a retroperitoneal hematoma.62 Patients with intractable bleeding or a retroperitoneal bleed warrant urgent consultation with interventional radiology for vessel embolization.
Transjugular Intrahepatic Portosystemic Shunts. In liver failure patients with portal hypertension and its complications, transjugular intrahepatic portosystemic shunts (TIPS) redirect blood from the abnormally high-pressure portal system to the low-pressure caval system.63 Complications from the procedure include inadvertent puncture of the liver capsule (5-30%), gallbladder or bile ducts (5-10%), and right kidney (< 2%).64 These patients may present with acute abdominal or flank pain due to an expanding perihepatic hematoma, acute cholangitis, or perinephric hematoma, respectively. CT may be indicated to detect intra-abdominal sources of bleeding or infection. The ED treatment of TIPS-related complications includes hemodynamic stabilization and treatment of specific findings such as antibiotics for infection and correction of coagulopathies and blood transfusion for significant bleeding.
Uterine Artery Embolization. Patients often seek emergency care post uterine artery embolization (UAE) for abdominal or pelvic pain and vaginal discharge or bleeding.65-67 In addition to these complaints, patients may also develop fever, hot flashes, mood swings, abdominal bloating, dysuria, and fibroid passage. The compilation of these symptoms is known as post-embolization syndrome and is thought to be caused by fibroid infarction.67 Severe post-procedural pain and pelvic infection are the two most common indications for hospitalization.67 In addition to post-embolization syndrome, fibroid passage, and fibroid necrosis, abdominal pain may also be due to the more serious etiology of pelvic infection, including endometritis, myometritis, pelvic abscess, and infected necrotic leiomyoma. MRI with gadolinium is considered the most sensitive imaging study to differentiate a pelvic infection from post-embolization syndrome.68 (See Figure 6.) If MRI is not readily available, CT is the next study of choice.
Figure 6: Endometritis on MRI
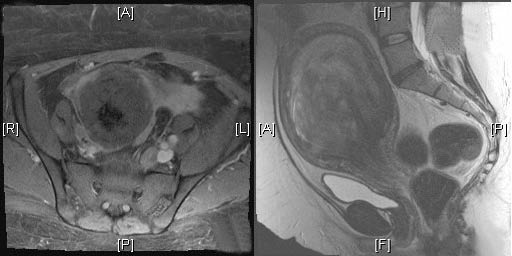
6A. Magnetic resonance imaging (MRI) of the uterus demonstrating gas and inflammation within the endometrium characteristic of endometritis. 6B. MRI of the uterus demonstrating uterine fibroid extrusion. (Image provided by Esther H. Chen, MD, University of California San Francisco, San Francisco, CA, published with permission.)
Percutaneous Biliary Drains. Percutaneous biliary drains may be external (the catheter is upstream from obstruction and bile is drained externally into a drain bag), internal (a metallic or plastic stent crosses obstruction and drains into the bowel with no drain exiting the body), and internal-external (an external catheter crosses the obstruction with side holes both upstream and downstream).69 Patients with these biliary drains may present to the ED with acute abdominal pain caused by intraperitoneal bile leakage or obstruction of the drain, which can lead to ascending cholangitis and intrahepatic abscess, particularly in those with malignant biliary obstruction.70 CT is recommended for the identification of these complications. In the ED, uncapping the external portion of the biliary drain may alleviate an obstruction and the patient's pain.
Conclusions
The ED evaluation and management of abdominal pain in the three distinct populations discussed in this issue can be challenging because atypical and delayed presentations increase their risk of complications compared to the general population. It is helpful to understand the underlying conditions and specific characteristics to prioritize certain diseases in the broad differential diagnosis of abdominal pain. In immunocompromised hosts, the risk of a serious intra-abdominal infection is high, particularly for HIV patients with low CD4 counts not on HAART, transplant patients in the intermediate post-transplantation period, or cancer patients undergoing active chemotherapy with neutropenia. The underlying opportunistic infection can present as surgical processes such as bowel perforation, acute appendicitis, or intestinal obstruction. Pregnant patients undergo physiologic changes that can make interpreting symptoms, physical examination findings, and even laboratory results more difficult. To avoid the radiation risk of CT, physicians can rely on ultrasound and MRI, the latter of which is diagnostically similar to CT for many surgical conditions. CT may be used in the later stages of pregnancy when the benefit of imaging outweighs the radiation risk. For these distinct populations, the reliance on imaging for diagnosis is difficult to avoid because the benefit of making an accurate diagnosis often outweighs the risks associated with the imaging study.
References
1. Unterman S, Zimmerman M, Tyo C, et al. A descriptive analysis of 1251 solid organ transplant visits to the emergency department. West J Emerg Med 2009;10:48-54.
2. Scott-Conner CE, Fabrega AJ. Gastrointestinal problems in the immunocompromised host. A review for surgeons. Surg Endosc 1996;10:959-964.
3. Yoshida D, Caruso JM. Abdominal pain in the HIV infected patient. J Emerg Med 2002;23:111-116.
4. Barone JE, Gingold BS, Arvanitis ML, et al. Abdominal pain in patients with acquired immune deficiency syndrome. Ann Surg 1986;204:619-623.
5. Parente F, Cernuschi M, Antinori S, et al. Severe abdominal pain in patients with AIDS: Frequency, clinical aspects, causes, and outcome. Scand J Gastroenterol 1994;29:511-515.
6. O'Keefe EA, Wood R, Van Zyl A, et al. Human immunodeficiency virus-related abdominal pain in South Africa. Aetiology, diagnosis and survival. Scand J Gastroenterol 1998;33:212-217.
7. Monkemuller KE, Lazenby AJ, Lee DH, et al. Occurrence of gastrointestinal opportunistic disorders in AIDS despite the use of highly active antiretroviral therapy. Dig Dis Sci 2005;50:230-234.
8. Slaven EM, Lopez F, Weintraub SL, et al. The AIDS patient with abdominal pain: A new challenge for the emergency physician. Emerg Med Clin North Am 2003;21:987-1015.
9. Saltzman DJ, Williams RA, Gelfand DV, et al. The surgeon and AIDS: Twenty years later. Arch Surg 2005;140:961-967.
10. Davidson T, Allen-Mersh TG, Miles AJ, et al. Emergency laparotomy in patients with AIDS. Br J Surg 1991;78:924-926.
11. Swenson KK, Rose MA, Ritz L, et al. Recognition and evaluation of oncology-related symptoms in the emergency department. Ann Emerg Med 1995;26:12-17.
12. Ilgen JS, Marr AL. Cancer emergencies: The acute abdomen. Emerg Med Clin North Am 2009;27:381-399.
13. Hirst B, Regnard C. Management of intestinal obstruction in malignant disease. Clin Med 2003;3:311-314.
14. Ripamonti CI, Easson AM, Gerdes H. Management of malignant bowel obstruction. Eur J Cancer 2008;44:1105-1115.
15. Takeuchi K, Tsuzuki Y, Ando T, et al. The diagnosis and treatment of adult intussusception. J Clin Gastroenterol 2003;36:18-21.
16. Maglinte DD, Kelvin FM, Sandrasegaran K, et al. Radiology of small bowel obstruction: Contemporary approach and controversies. Abdom Imaging 2005;30:160-178.
17. Sarr MG, Bulkley GB, Zuidema GD. Preoperative recognition of intestinal strangulation obstruction. Prospective evaluation of diagnostic capability. Am J Surg 1983;145:176-182.
18. Yeoh E. Radiotherapy: Long-term effects on gastrointestinal function. Curr Opin Support Palliat Care 2008;2:40-44.
19. Hill A, Balkin A. Risk factors for gastrointestinal adverse events in HIV treated and untreated patients. AIDS Rev 2009;11:30-38.
20. Rolston KV. Neutropenic enterocolitis associated with docetaxel therapy in a patient with breast cancer. Clin Adv Hematol Oncol 2009;7:527-528.
21. Gorschluter M, Mey U, Strehl J, et al. Neutropenic enterocolitis in adults: Systematic analysis of evidence quality. Eur J Haematol 2005;75:1-13.
22. Spencer SP, Power N, Reznek RH. Multidetector computed tomography of the acute abdomen in the immunocompromised host: A pictorial review. Curr Probl Diagn Radiol 2009;38:145-155.
23. Blijlevens NM. Neutropenic enterocolitis: challenges in diagnosis and treatment. Clin Adv Hematol Oncol 2009;7:528-530.
24. Ullery BW, Pieracci FM, Rodney JR, et al. Neutropenic enterocolitis. Surg Infect (Larchmt) 2009;10:307-314.
25. Fishman JA. Infection in solid-organ transplant recipients. N Engl J Med 2007;357:2601-2614.
26. Branch DW. Physiologic adaptations of pregnancy. Am J Reprod Immunol 1992;28:120-122.
27. Martin C, Varner MW. Physiologic changes in pregnancy: Surgical implications. Clin Obstet Gynecol 1994;37:241-255.
28. Challoner K, Incerpi M. Nontraumatic abdominal surgical emergencies in the pregnant patient. Emerg Med Clin North Am 2003;21:971-985.
29. Oto A, Ernst RD, Ghulmiyyah LM, et al. MR imaging in the triage of pregnant patients with acute abdominal and pelvic pain. Abdom Imaging 2009;34:243-250.
30. Goldberg-Stein S, Liu B, Hahn PF, et al. Body CT during pregnancy: Utilization trends, examination indications, and fetal radiation doses. AJR Am J Roentgenol 2011;196:146-151.
31. Jaffe TA, Miller CM, Merkle EM. Practice patterns in imaging of the pregnant patient with abdominal pain: A survey of academic centers. AJR Am J Roentgenol 2007;189:1128-1134.
32. Mourad J, Elliott JP, Erickson L, et al. Appendicitis in pregnancy: New information that contradicts long-held clinical beliefs. Am J Obstet Gynecol 2000;182:1027-1029.
33. Tracey M, Fletcher HS. Appendicitis in pregnancy. Am Surg 2000;66:555-559; discussion 559-560.
34. Mazze RI, Kallen B. Appendectomy during pregnancy: A Swedish registry study of 778 cases. Obstet Gynecol 1991;77:835-840.
35. McGory ML, Zingmond DS, Tillou A, et al. Negative appendectomy in pregnant women is associated with a substantial risk of fetal loss. J Am Coll Surg 2007;205:534-540.
36. Andersen B, Nielsen TF. Appendicitis in pregnancy: Diagnosis, management and complications. Acta Obstet Gynecol Scand 1999;78:758-762.
37. Williams R, Shaw J. Ultrasound scanning in the diagnosis of acute appendicitis in pregnancy. Emerg Med J 2007;24:359-360.
38. Basaran A, Basaran M. Diagnosis of acute appendicitis during pregnancy: A systematic review. Obstet Gynecol Surv 2009;64:481-488; quiz 499.
39. Ramin KD, Ramsey PS. Disease of the gallbladder and pancreas in pregnancy. Obstet Gynecol Clin North Am 2001;28:571-580.
40. Gilo NB, Amini D, Landy HJ. Appendicitis and cholecystitis in pregnancy. Clin Obstet Gynecol 2009;52:586-596.
41. Emergency ultrasound guidelines. Ann Emerg Med 2009;53:550-570.
42. Shelton J, Linder JD, Rivera-Alsina ME, et al. Commitment, confirmation, and clearance: New techniques for nonradiation ERCP during pregnancy (with videos). Gastrointest Endosc 2008;67:364-368.
43. Dhupar R, Smaldone GM, Hamad GG. Is there a benefit to delaying cholecystectomy for symptomatic gallbladder disease during pregnancy? Surg Endosc 2010;24:108-112.
44. Jelin EB, Smink DS, Vernon AH, Brooks DC. Management of biliary tract disease during pregnancy: A decision analysis. Surg Endosc 2008;22:54-60.
45. Lu EJ, Curet MJ, El-Sayed YY, et al. Medical versus surgical management of biliary tract disease in pregnancy. Am J Surg 2004;188:755-759.
46. Dietrich CS, 3rd, Hill CC, Hueman M. Surgical diseases presenting in pregnancy. Surg Clin North Am 2008;88:403-419, vii-viii.
47. Augustin G, Majerovic M. Non-obstetrical acute abdomen during pregnancy. Eur J Obstet Gynecol Reprod Biol 2007;131:4-12.
48. Perdue PW, Johnson HW, Jr., Stafford PW. Intestinal obstruction complicating pregnancy. Am J Surg 1992;164:384-388.
49. Beall DP, Fortman BJ, Lawler BC, et al. Imaging bowel obstruction: A comparison between fast magnetic resonance imaging and helical computed tomography. Clin Radiol 2002;57:719-724.
50. Berczi V, Bottomley JR, Thomas SM, et al. Long-term retrievability of IVC filters: Should we abandon permanent devices? Cardiovasc Intervent Radiol 2007;30:820-827.
51. Chung J, Owen RJ. Using inferior vena cava filters to prevent pulmonary embolism. Can Fam Physician 2008;54:49-55.
52. Ray CE, Jr., Kaufman JA. Complications of inferior vena cava filters. Abdom Imaging 1996;21:368-374.
53. Given MF, Hanson JJ, Lee MJ. Interventional radiology techniques for provision of enteral feeding. Cardiovasc Intervent Radiol 2005;28:692-703.
54. Wollman B, D'Agostino HB, Walus-Wigle JR, et al. Radiologic, endoscopic, and surgical gastrostomy: An institutional evaluation and meta-analysis of the literature. Radiology 1995;197:699-704.
55. Minchff TV. Early dislodgement of percutaneous and endoscopic gastrostomy tube. J S C Med Assoc 2007;103:13-15.
56. Kadakia SC, Cassaday M, Shaffer RT. Prospective evaluation of Foley catheter as a replacement gastrostomy tube. Am J Gastroenterol 1992;87:1594-1597.
57. Kalambokis G, Manousou P, Vibhakorn S, et al. Transjugular liver biopsy indications, adequacy, quality of specimens, and complications a systematic review. J Hepatol 2007;47:284-294.
58. Gamble P, Colapinto RF, Stronell RD, et al. Transjugular liver biopsy: A review of 461 biopsies. Radiology 1985;157:589-593.
59. Cluzel P, Martinez F, Bellin MF, et al. Transjugular versus percutaneous renal biopsy for the diagnosis of parenchymal disease: Comparison of sampling effectiveness and complications. Radiology 2000;215:689-693.
60. Misra S, Gyamlani G, Swaminathan S, et al. Safety and diagnostic yield of transjugular renal biopsy. J Vasc Interv Radiol 2008;19:546-551.
61. See TC, Thompson BC, Howie AJ, et al. Transjugular renal biopsy: Our experience and technical considerations. Cardiovasc Intervent Radiol 2008;31:906-918.
62. Fine DM, Arepally A, Hofmann LV, et al. Diagnostic utility and safety of transjugular kidney biopsy in the obese patient. Nephrol Dial Transplant 2004;19:1798-1802.
63. Haskal ZJ, Martin L, Cardella JF, et al. Quality improvement guidelines for transjugular intrahepatic portosystemic shunts. J Vasc Interv Radiol 2003;14:S265-270.
64. Freedman AM, Sanyal AJ, Tisnado J, et al. Complications of transjugular intrahepatic portosystemic shunt: A comprehensive review. Radiographics 1993;13:1185-1210.
65. Ganguli S, Faintuch S, Salazar GM, et al. Postembolization syndrome: Changes in white blood cell counts immediately after uterine artery embolization. J Vasc Interv Radiol 2008;19:443-445.
66. Marshburn PB, Matthews ML, Hurst BS. Uterine artery embolization as a treatment option for uterine myomas. Obstet Gynecol Clin North Am 2006;33:125-144.
67. Pron G, Mocarski E, Bennett J, et al. Tolerance, hospital stay, and recovery after uterine artery embolization for fibroids: The Ontario Uterine Fibroid Embolization Trial. J Vasc Interv Radiol 2003;14:1243-1250.
68. Kitamura Y, Ascher SM, Cooper C, et al. Imaging manifestations of complications associated with uterine artery embolization. Radiographics 2005;25 Suppl 1:S119-132.
69. Covey AM, Brown KT. Palliative percutaneous drainage in malignant biliary obstruction. Part 2: Mechanisms and postprocedure management. J Support Oncol 2006;4:329-335.
70. Wu SM, Marchant LK, Haskal ZJ. Percutaneous interventions in the biliary tree. Semin Roentgenol 1997;32:228-245.
A common presenting complaint for patients seeking emergency medical care is acute abdominal pain. Although difficult to diagnose in healthy patients, it is even more challenging in special populations. This article will focus on three distinct populations: patients with altered immunologic function, pregnant women, and post-procedural patients.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
