Osteoporosis
Osteoporosis
Osteoporosis is a highly prevalent metabolic bone disease that affects both men and woman at risk of bone demineralization. It has been recognized as both a qualitative and quantitative bone disorder that imposes an unusually high risk of fracture on affected individuals. Contrary to earlier belief that osteoporosis is a disease of North European, postmenopausal women, it is now recognized that the scope of this disorder extends to include men and women at risk.1 Moreover, awareness of the increased prevalence of this disorder has been heightened as life expectancy is on the rise. It is estimated that in the year 2000 nine million fractures related to osteoporosis occurred worldwide; most of these fractures were in Europe.2 Whether the occurrence of osteoporotic fractures is rising or on the decline remains controversial.
Prior to exhibiting the gross effects of osteoporosis (i.e., skeletal fractures), the impact of this disorder may involve the microstructure of bone. Diagnosis and intervention at this early stage has not been well-studied. Measurement of bone mineral density (BMD) via dual-energy x-ray absorptiometry (DEXA) is the most widely used method to diagnose osteoporosis. DEXA could be applied through widespread population screening, but a more sophisticated approach would be to estimate individual risk of developing osteoporosis and the subsequent likelihood of fracture based on several widely known risk factors.
To better understand osteoporosis, one should be familiar with its underlying pathogenesis and how osteoporosis may lead to compromised bone strength and integrity. Several theories address the complex processes that impact bone mass and quality resulting in osteoporosis.3,4 There is limited conclusive evidence, however, related to this complex disorder, and more research is needed to better understand the underlying processes responsible for the development of fractures. Nevertheless, the following mechanisms offer, at least in part, a reasonable explanation of some of the disease features:
- Increased bone loss by increased resorption and bone turnover
- Decreased compensatory bone formation
- Impact of increased remodeling on the structure of the bone
- Inability to reach peak bone mass, which may depend heavily on genetic factors
- Effect of microfractures
- Change in the microstructure of bone
- Several genetic and environmental determinants and the interplay among these factors5
- The mechanical effect of posture and muscle strength on the bone
One should keep in mind that some of the proposed mechanisms are poorly understood and more research is needed to further delineate the underlying processes responsible for the development of fractures.
Several systemic hormones may, directly or indirectly, contribute to osteoporosis. A lack of estrogen, especially in postmenopausal women, is the classic example of this association. Bone resorption increases as estrogen decreases.6 The relationship between reduced estrogen and osteoporosis is not limited to women as the correlation between estrogen levels and BMD in elderly men is well established.7,8 To a lesser degree but with a similar mechanism, androgen deficiency may be related to increased bone turnover.9,10
Glucocorticoid deficiency may play an important role in osteoporosis by inhibiting bone formation and, to a lesser extent, facilitating increased bone resorption. On the other hand, glucocorticoid excess also is associated with an increased risk for osteoporosis. Additional hormonal effects are to be considered with hypo- or hyperthyroidism, increased parathyroid hormone (PTH), vitamin D deficiency, calcitonin deficiency, and abnormalities in calcium intake and/or absorption.
Low dietary calcium intake may lead to increased fractures, as demonstrated in population studies in the United States. High animal protein intake also has been shown to increase hip fractures in epidemiologic studies.11,12 Several studies have demonstrated that low dietary vitamin K intake is associated with low BMD or increased fractures. Additionally, vitamin K supplementation has been shown to improve the bone turnover profile.13 Low body weight and decreased physical activity also are associated with increased risk of osteoporotic fractures.14 Moreover, cigarette smoking is well known to increase the risk of developing osteoporosis via multiple mechanisms.15,16 Table 1 depicts known risk factors for osteoporosis-related fractures.
Table 1: Potential Risk Factors Associated with Osteoporosis and Related Fractures
- Alcoholism
- Certain medications such as glucocorticoids, anticonvulsants, aromatase inhibitors, and heparin
- Hereditary or acquired medical conditions (see Table 2)
- Cigarette smoking
- Dementia
- Diet: low in fruits and vegetables or high in protein, sodium, or caffeine
- Estrogen deficiency states
- Excessive or lack of exercise or decreased mobility
- Family history of osteoporosis or fragility fracture in a first-degree relative
- Frequent, recurrent falls
- Gender: female > male
- Impaired vision
- Increasing age
- Low body weight or excessive weight loss
- Low stores of calcium or vitamin D
- Musculoskeletal abnormalities such as kyphosis
- Personal history of fragility fracture
- Race: white and Asian
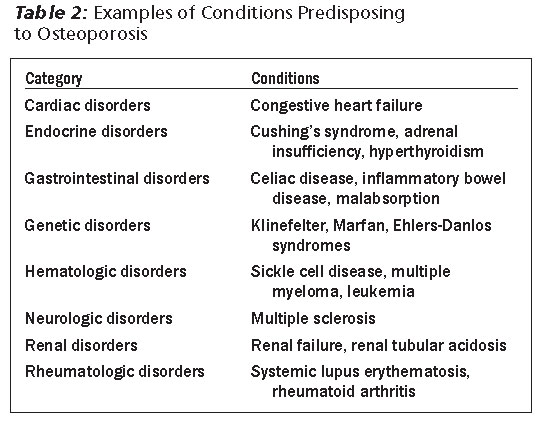
Secondary causes of osteoporosis should be investigated upon diagnosis. Drugs that are known to increase the risk of osteoporosis include glucocorticoids, anticonvulsants, aromatase inhibitors, and heparin. In addition, several systemic conditions may be associated with increased risk of osteoporosis (see Table 2).
Clinical diagnosis of osteoporosis can be made based on the presence of fragility fractures, ones that occur in the setting of normal activities such as fall from standing position, in at-risk individuals when other causes can be excluded. Typical examples of fragility fractures include vertebral and femur neck fractures. Definite diagnosis of osteoporosis, however, is based on the measurement of BMD with DEXA. BMD is expressed in grams of mineral per square centimeter scanned (g/cm2). BMD testing with DEXA yields a calculated value, called a T-score, which is the number of standard deviations above or below the average of peak bone density for one's reference group (e.g., Caucasian women). A Z-score also is calculated based on the patient's expected BMD for age and other characteristics.
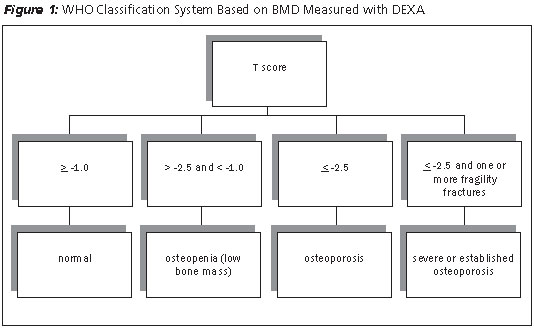
The World Health Organization (WHO) has a widely used classification system that, based on T-score, assigns a diagnosis of normal, osteopenia, osteoporosis, or severe osteoporosis (see Figure 1).17 Of note, although BMD does not necessarily reflect bone quality, the score obtained by DEXA is considered a good predictor of risk of future fracture. Overall and site-specific risks of fracture can be estimated, although DEXA measurement at the hip site may be the best predictor of future hip fracture risk. BMD measurements at the lumbar spine and femoral neck levels are frequently the preferred choice of testing as they seem to be good predictors of fracture risk. Measurement of BMD at the one-third radius site is an acceptable alternative when measurements of the lumbar spine and hip cannot be obtained.18 Other skeletal sites have not been accurate for diagnosing osteoporosis.
An adjusted Z-score based on race or ethnicity has been recommended by the International Society for Clinical Densitometry (ISCD). Z-scores of < -2.0 translate to either a low BMD for age or below the expected range for age. Values > -2.0 are considered within the expected range for age.19
Several organizations have proposed guidelines for osteoporosis screening. The National Osteoporosis Foundation (NOF) has outlined the indications for BMD testing in both men and women in the United States (see Table 3).20 BMD testing should be performed when the results will have an impact on clinical decision-making.
Table 3: Indications for Bone Mineral Density Testing by National Osteoporosis Foundation
- Women > 65 years of age
- Men > 70 years of age
- Postmenopausal women with increased risk of fractures
- Men > 50 years of age with increased risk of fractures
- Repeat testing, though still unclear, is recommended every 2 years or longer in individuals with normal BMD.
Serum and urine testing of bone turnover biomarkers assesses the risk of fracture in untreated individuals. Moreover, repeated measurement during the course of FDA-approved therapies may be beneficial in assessing fracture risk reduction. These markers of remodeling include serum C-telopeptide (CTX), urine N-telopeptide (NTX), and bone specific alkaline phosphatase (BSAP).21
Previously confirmed vertebral fractures predict later vertebral and other fractures independent of additional risk factors. Therefore, vertebral assessment imaging of the thoracic and lumbar spine using DEXA should be considered when the presence of such fracture may affect clinical decision-making.19
DEXA scanning remains the most accurate diagnostic method for diagnosis of low BMD. Quantitative ultrasonography is another less expensive and safer technique than DEXA. Quantitative ultrasonography of the calcaneus predicts fractures of the femoral neck, hip, and spine as effectively as DEXA. The threshold for diagnosis, however, is not well defined.22,23
WHO developed an algorithm to estimate the 10-year probability of a hip fracture and the 10-year probability of a major osteoporotic fracture. This algorithm is designed for untreated postmenopausal women and men age 50 or older and is available online at: www.nof.org. The use of BMD from sites other than the hip in this algorithm has not been validated as an estimate of fracture risk and thus is not recommended.
Osteoporosis, though often underdiagnosed in men, has gained increasing interest in the past few years. Early diagnosis of this disorder in men has been particularly challenging as men usually present with osteoporotic fractures. Osteoporosis also is diagnosed based on physical complaints such as back pain or presence of risk factors (see Table 1).24 Due to lack of cohort studies in this setting, and until further comprehensive evidence is available, osteoporosis in men will continue to be overlooked and treatment often will be delayed.
As in women, BMD measurements are also a useful tool in predicting fractures related to osteoporosis in men.25,26 The decision to initiate screening, however, remains the main challenge. Recent guidelines have addressed this matter and more definite statements on screening and prevention have emerged (see "Screening for Osteoporosis").
Hip and spine usually are the preferred sites for testing, as fractures in these two sites could have the greatest impact on quality of life. Additional laboratory testing may be advised in the appropriate setting as suspicion arises and may include testing for myeloproliferative disorders, parathyroid disorders, celiac disease, etc. (see Table 2).
Treatment of osteoporosis in men includes lifestyle measures, calcium and vitamin D supplementations, and hormonal and pharmacologic therapy (see "Prevention and Treatment of Osteoporosis"). Particular attention should be paid to investigate and effectively treat the underlying secondary causes such as long-term glucocorticoid use, hypogonadism, and alcoholism.27,28
The burden of osteoporosis stems from the morbidity and mortality associated with fractures. Hence, the main goal of preventing this disorder is to reduce the occurrence and risk of fractures. This goal can be achieved by the use of medications that promote bone strength and with the adoption of lifestyle interventions aimed at enhancing bone health.
Regular exercise and increased physical activity may play an important role in increasing BMD. This was demonstrated in several studies, especially earlier in life. The impact seems to be more noticeable and sustainable, however, when adherence to a regular program is maintained over prolonged periods of time.29,30 Moreover, the benefit of an exercise program seems to dissipate shortly after the individual stops exercising.31 It has been shown that weight-bearing, aerobic, and resistance exercises all may improve BMD of the spine in postmenopausal women, while walking seems to improve BMD at the hip.
As peak BMD is likely already reached in the elderly, the principal benefit of exercise is to maintain current bone mass and prevent further bone loss. Power training (fast movement with resistance weights) is more effective than strength (slower movement) training.32
Recently investigators examined the impact of skeletal abnormalities and muscle strength on the development of osteoporosis. Exercises, particularly high-impact ones, have been shown to be beneficial during different stages of growth and development.30 Further research is needed to identify the type and frequency of exercise required for osteoporosis prevention. Individualized programs based on an individual's risk factors, age, and BMD may be needed. Until there is persuasive evidence, adherence to a moderate, weight-bearing exercise program most days of the week is reasonable. The intensity of one's program should be adjusted in consultation with a person's primary care physician.
Cigarette smoking is known to accelerate bone loss.33 Smoking also may compromise the effectiveness of estrogen treatment in postmenopausal women34,35 and is known to increase the risk of thromboembolic complications in women on HRT. Therefore, smoking cessation is highly recommended in postmenopausal women undergoing treatment with estrogen.
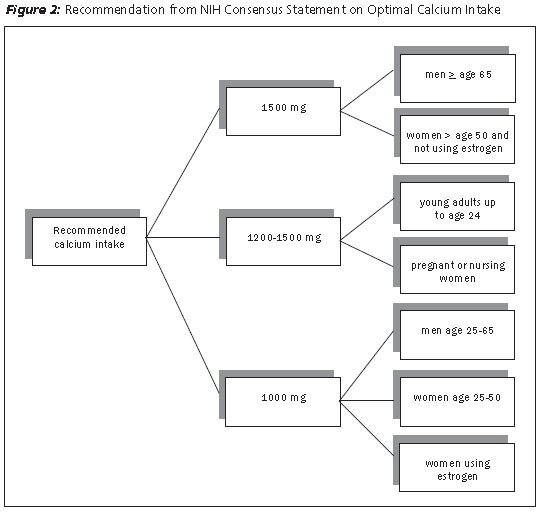
Sufficient daily intake of calcium is essential to maintain healthy bones. Since calcium requirements differ based on factors such as age and menopause status, the amount required to achieve this goal will vary accordingly. The National Institutes of Health (NIH) estimated optimal calcium intake based on gender and hormonal status. See Figure 2 for details.36 Patients should be educated on foods with high calcium content. There does not appear to be clear benefits favoring one calcium formulation, although absorption may differ based on the fasting state, with calcium citrate preparations having a more favorable profile than calcium carbonate formulations.
Vitamin D has gained increasing interest in recent years. It plays an important clinical role in a variety of medical conditions. The more defined role, however, is related to its impact on calcium homeostasis and bone metabolism. With minimal sun exposure, it is generally accepted that a 25-(OH) vitamin D level below 20 ng/ml is inadequate for bone health. Maintaining levels above 20 ng/ml is a reasonable treatment goal for most adults with recommendation of levels above 30 ng/ml in elderly and high-risk individuals. Calcium supplementation may prevent bone loss and changes in biochemical markers of bone turnover in susceptible populations.37,38
It has been shown that mildly low levels of vitamin D, though common, may be associated with bone loss. Supplementation of vitamin D and calcium in vitamin D deficiency has been shown to decrease the risk of hip and non-spine fractures.39,40
In the setting of vitamin D deficiency, treatment should aim to achieve 25-(OH) vitamin D levels of > 30-32 ng/ml to prevent a compensatory rise in the PTH level.41,42 The administration of 50,000 International Units (IU) of vitamin D2 weekly for 8 weeks may achieve this goal. After 8 weeks a level recheck should be done. Once an adequate vitamin D level is reached, maintenance is required with either 800-1000 IU of vitamin D3 daily or 50,000 IU of vitamin D2 once or twice monthly. Concomitant treatment with calcium is recommended.
Several drug classes have been approved for the prevention and treatment of osteoporosis. The choice of a medication or class depends on its efficacy in preventing vertebral and hip fractures, safety, duration of treatment, renal function, and side effects profile.
Bisphosphonates are first-line medications in the management of osteoporosis. They inhibit bone resorption and reduce osteoclast activity.35 A list of FDA-approved medications to prevent and treat osteoporosis is provided in Table 4. The physician should consider indication(s) for treatment and corresponding dosage and duration.
Bisphosphonates are notorious for causing gastrointestinal (GI) side effects and should be taken on an empty stomach for better oral absorption. When taking bisphosphonates, GI symptoms can be alleviated by switching from a daily to a weekly regimen. Osteonecrosis of the jaw, an infrequent adverse reaction related to the use of bisphosphonates, also is a concern. Osteonecrosis may result from the use of high IV doses to treat complications from a malignant disease.43 Zoledronic acid, which is given IV, has similar effects as other daily oral bisphosphonates. Due to its potency, zoledronic acid is administered in small doses over longer intervals.44,45 Black et al reported that atrial fibrillation, a serious side effect, occurred more frequently in the zoledronic acid group when compared to a control group.44
Controversy has emerged over the years regarding the use of HRT in the prevention and treatment of osteoporosis. For years, the use of HRT was advocated as little was known about the long-term effects of its use. The shift in the paradigm of such practice started to occur following a number of clinical trials including the Women's Health Initiative (WHI) clinical trial. HRT, though decreasing the incidence of fractures, was associated with a higher risk of breast cancer and cardiovascular complications.46-48 It still is unknown at which doses HRT can be deemed effective with minimal potential side effects, and further research is needed in this area. Given the above-mentioned findings, the indiscriminate use of HRT for prevention of osteoporosis should be discouraged and alternative therapies should be considered. Further, FDA has approved raloxifene, a selective estrogen receptor modulator, for the prevention and treatment of osteoporosis in postmenopausal women.
Calcitonin, denosumab, and teriparatide also are approved by FDA for the prevention and treatment of osteoporosis. High-risk individuals take these medications in combination with supplemental calcium and vitamin D. Table 5 provides information on indications, dosing, and drug class. Constant high levels of PTH over prolonged durations, as seen in hyperparathyroidism, have been shown to increase bone resorption. Paradoxically, intermittent exposure seems to have a stimulatory effect on bone formation. Hence, the prolonged use of exogenous PTH (teriparatide) is an effective treatment for osteoporosis.
Understanding the need for screening to investigate the risk of osteoporosis and fractures requires thorough understanding of the potential risk factors that increase the individual's risk of osteoporosis-related fractures (see Table 1). Several health care organizations have adopted screening guidelines to assist providers in making decisions on screening. Given the abundance of risk factors and the burden of fractures on morbidity and mortality, the decision to pursue testing will have to be made based on the risks involved and benefits obtained, as well as the candidacy for pharmacologic treatment, and ultimately should be handled on a case-by-case approach. For instance, a low threshold for screening and intervention should be adopted when dealing with a 65-year-old demented male who is at high risk of falls to prevent the devastating aftermath of potential fractures secondary to undiagnosed osteoporosis.
Table 6: Summary
- Osteoporosis is a metabolic bone disease affecting both men and women.
- Fractures related to osteoporosis may be a major burden for affected individuals.
- Several risk factors are associated with a higher risk of osteoporosis and subsequent fractures.
- Some medications may place the patient at risk for osteoporosis and fractures.
- Certain hereditary and acquired medical conditions are also known to be associated with osteoporosis.
- Diagnosis and management of osteoporosis relies principally on the measurement of bone mineral density (BMD) using dual-energy x-ray absorptiometry (DEXA).
- Biochemical markers and algorithms also assist in decision-making.
- Suboptimal levels of calcium and vitamin D warrant treatment to prevent osteoporosis and fractures.
- The United States Food and Drug Administration (FDA) has approved 10 different medications for the prevention and treatment of osteoporosis.
- Bisphosphonates are considered first-line medications for treating osteoporosis in postmenopausal women.
- Intravenous (IV) zoledronic acid has similar effects to other oral bisphosphonates.
- Hormone replacement therapy (HRT) is no longer considered a first-line treatment for osteoporosis due to the overwhelming evidence of increased risk of breast cancer and cardiovascular disease.
- Screening for osteoporosis is initiated based on risks, benefits, and the candidacy for pharmacologic treatment.
- Generally, screening for osteoporosis targets women > age 65, men > age 70, and individuals who are at high risk of osteoporosis.
Below is a summary of recommendations for osteoporosis screening accounting for different health care organizations' recommendations.49-50 (See Table 6.)
Osteoporosis is a highly prevalent metabolic bone disease that affects both men and woman at risk of bone demineralization.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
