Penetrating Thoracic Trauma
Penetrating Thoracic Trauma
Authors:
Kathleen Cowling, DO, MS, FACEP, Clinical Professor, Michigan State University, College of Human Medicine, Attending, Covenant HealthCare, Saginaw, MI.
Mathias J. Christianson, MD, Emergency Medicine Resident, Synergy Medical Education Alliance, Saginaw, MI; Clinical Instructor, Michigan State University College of Human Medicine.
Peer Reviewer:
Jay Menaker, MD, Associate Professor, Department of Surgery, Associate Professor, Department of Emergency Medicine, University of Maryland School of Medicine, Baltimore; Physician Director of Quality Management, R Adams Cowley Shock Trauma Center, Baltimore, MD.
Trauma continues to be a significant cause of morbidity and mortality. Accidental death remained the fifth most common cause of death for all ages in 2009.1 Thoracic injuries reportedly have been involved in up to 75% of all deaths related to trauma and may be directly responsible in up to 25% of these deaths.2-4 Between 4-15% of trauma center admissions are related to penetrating thoracic injuries.5 While the initial resuscitative phase taught in the American College of Surgeon's Advanced Trauma Life Support (ATLS) courses has become a staple in the evaluation of these patients, there are many other aspects to consider when caring for these patients. The initial evaluation and transportation by EMS providers, as well as intermediate hospital work-up prior to transfer to trauma centers, may alter the outcome for these patients. Advances in radiologic evaluation with ultrasound may assist initial management decisions. Observation in asymptomatic patients may be contemplated. Also, emergency department thoracotomy may be considered in the appropriate clinical setting.
The purpose of this issue is to provide guidance in the evaluation and management of the patient presenting with acute penetrating thoracic trauma. This article will discuss the major injury types and methods to detect them. The discussion will address the aforementioned considerations and advancements in the work-up of these cases to help with resource utilization and provide high quality care to patients.
Pre-trauma Center Evaluation and Transfer vs. Direct Trauma Center Transport
Initial presentation to non-trauma centers is common in rural areas. The practice of initial stabilization and transfer to a higher level trauma center is well known and taught in ATLS courses. Studies have shown, however, that when this initial evaluation is prolonged and patients are not promptly transferred to appropriate trauma centers, there is increased risk of mortality.6-10 Sampalis and colleagues found an overall mortality of 8.9% for indirectly transported patients vs. 4.8% for patients directly transported to a trauma center.9 These data may support bypassing the initial hospital and directly transporting to tertiary trauma centers. There are a variety of reasons proposed for which EMS providers stop at non-trauma centers, such as inability to care for such critical patients, the thought that some higher level of care was better than the care that could be provided en route to the tertiary center, or the need for immediate attention for an unstable patient.6 Also, in some areas there are limited resources, and the prolonged transport to the tertiary center may leave gaps in EMS coverage.
For victims of major trauma taken initially to a non-trauma center then subsequently transferred, the most common intervention provided at the initial hospital is endotracheal intubation, followed by chest decompression.11 The additional "procedures" performed commonly included imaging studies and, frequently, CT scanning. The studies were often repeated upon arrival to tertiary centers. One possible reason for this could be more sophisticated scanners or higher quality images at the trauma centers. However, these imaging studies at the initial hospital, combined with the lack of protocol-based care, frequently prolong the transfer time.6
There is some concern that these patients are under-resuscitated due to their transfer and prolonged time to definitive care. The initial resuscitation administered by EMS and the initial facility may give a false sense of security, as patients are often transported and arrive with appropriate appearing vital signs. It has been speculated that this allows some measure of comfort upon arrival at accepting tertiary facilities, and patients may be treated less aggressively.8 Speculation aside, the transferred patients in that study appeared to be treated less aggressively, with extended correction of resuscitation markers such as lactic acid and base deficit. This prolonged correction may increase complication rates and cause transferred patients to incur longer intensive care unit (ICU) and hospital lengths of stay.10
There are studies showing a benefit from permissive hypotension with limited crystalloid use and increased blood product use. This practice is known as damage control resuscitation. This practice utilizes a goal of transfusion of a 1:1 ratio of fresh frozen plasma and units of packed red blood cells, with very limited crystalloid administration. The target systolic blood pressure is 90 mm Hg.12 Two recent studies utilizing this resuscitation concept had an average systolic blood pressure of about 100 mm Hg.12,13 The studies found improved mortality in both 24-hour survival and 30-day survival.12,13 While studies continue, this concept has not yet gained wide acceptance or become the clinical standard of care. Additionally, this approach does not appear to be well studied in transferred trauma patients.
The published observational studies referenced above would support direct transport of critical trauma patients to tertiary trauma centers. When this is not possible, or more immediate attention and stabilization are necessary, efforts should be made to streamline treatments and minimize nontherapeutic imaging studies in an effort to decrease the time to transfer from the intermediate facility to the tertiary center. This would include addressing the airway and intubating the patient, if needed, obtaining a chest X-ray, utilizing bedside ultrasound, if available, and possibly placing a chest tube if needed.
Initial Evaluation and Management of Common Injury Types
When a trauma patient arrives in the emergency department, the American College of Surgeons Committee on Trauma recommends following the ATLS protocol. This starts with a primary survey, which focuses on immediate life-threatening injuries and the ABCs algorithm.14 The airway is evaluated first. If the patient is unconscious, or there is any doubt that the patient is able to maintain a patent airway, perform immediate endotracheal intubation. The possibility of a spinal cord injury should be considered and spinal precautions maintained. Next, breathing is assessed and the chest is exposed. Observe for discrepancies in movement and examine for tracheal deviation. Auscultation may reveal decreased breath sounds. When there is tracheal deviation or suspicion for tension pneumothorax, perform immediate chest decompression with a large-bore (14-18 gauge) IV needle in the second intercostal space at the midclavicular line. Other signs of tension pneumothorax include hypotension, neck vein distension, and tachycardia.15 Circulation is assessed and control of major source bleeding should be applied. Crystalloid and/or blood is initiated through two large-bore peripheral IV lines. A brief neurological survey should be performed to evaluate any disability, and the patient should be exposed to allow complete visualization and examination.14
After the primary survey and any immediate life-threatening injuries are attended to, the secondary survey and imaging studies are obtained to define injuries and plan treatments.14 Potential injuries in the setting of penetrating thoracic trauma include chest wall injuries, pneumo- and/or hemothorax, pulmonary injuries, tracheobronchial injuries, cardiac injuries, injuries of great vessels, diaphragmatic injuries, and esophageal injuries.2-4,15-19
Rib or sternal fractures may be seen with penetrating trauma at the point of entry and/or exit. They may also contribute to further penetrating injury if there is displacement of the fracture ends causing further damage to internal structures.3,4
Pneumothorax is a common complication of penetrating thoracic trauma. It results from free air in the pleural cavity. There are three types of pneumothorax: simple, communicating or open, and tension pneumothorax. Simple pneumothorax does not result in shift of the mediastinum or change in the hemidiaphragm.4 A pneumothorax is considered small if it is 15% or less of the pleural space, moderate if it is 15-60%, and large if it occupies greater than 60%. Depending on the clinical circumstances, small or occult pneumothoraces may be observed. However, if the patient is intubated or will require surgery with general anesthesia, a chest tube is often placed, as many of these will progress under positive pressure ventilation.4 If none is placed prior to surgery, the anesthesia team should be aware of the presence of pneumothorax. If there is a moderate to large pneumothorax, a chest tube will be required. Recommended chest tubes size should be 36-40 French in traumatic pneumothoraces because of the risk of concomitant hemothorax. This is placed at the fifth intercostal space on the anterior axillary line. This allows drainage of blood and smaller clotted material when placed under 20 cm of water suction.
A communicating or open pneumothorax results from a defect in the chest wall that is at least two-thirds the diameter of the trachea, allowing direct communication with the atmosphere.3,15 This is also referred to as a sucking chest wound. During inspiration, air rushes into the pleural cavity via the wound, causing further collapse of the affected lung. There is minimal expansion of the lung during expiration. An occlusive dressing is applied and an appropriate-sized chest tube placed to prevent development of tension pneumothorax.3,4,15
Tension pneumothorax occurs when there is one-way air leakage from either a chest wall wound or the lung itself. The air continues to accumulate and fills the pleural space, eventually compressing the affected lung and mediastinal structures. This compression may cause tracheal deviation and leads to impaired cardiac function with decreased venous return to the heart and distension of the neck veins with eventual hypotension.3,4,15 Tension pneumothorax is a clinical diagnosis, and treatment should not be delayed for confirmation by chest X-ray. This requires immediate needle decompression followed by chest tube placement.
Bleeding from lung tissue is the most common cause of traumatic hemothorax, followed by laceration of intercostal or internal mammary arteries.4,15 It occurs concomitantly with pneumothorax in as many as 25% of cases.4 Hemothorax may be detectable by chest X-ray with as little as 200-300 mL of blood blunting the costophrenic angles on upright images. In most trauma, however, the X-ray is supine and hemothorax is more difficult to identify. In this case, it is usually seen as a haziness over the affected lung space.3,4 Treatment is by drainage facilitated by placement of a 36-40 French chest tube.
Pulmonary injuries related to penetrating trauma are often due to parenchymal laceration. These are usually self-limited but may cause hemothorax. Treatment is by chest tube placement to drain the blood and monitor for additional hemorrhage.3,4 Fewer than 10% of these cases require operative intervention, usually due to persistent air leak or persistent hemorrhage.2,3 Pulmonary contusion is more common in blunt trauma cases, although it may be present in high-velocity gunshot wounds. Pulmonary contusion results in alveolar hemorrhage and is accompanied by interstitial edema within 1-2 hours.5 Pulmonary contusion results in an infiltrative pattern on chest X-ray. In severe cases, acute respiratory distress syndrome may ensue.3,4
Tracheobronchial injury is an uncommon injury and often fatal before arrival at the emergency department.2-4 With penetrating trauma, it is seen at any level of the tracheobronchial tree, although stab wounds nearly always affect the cervical airway. Suggestive findings of tracheobronchial injury include persistent air leak, cervical subcutaneous emphysema, hemoptysis, or tension pneumothorax.2-4 On chest X-ray, the "fallen lung sign" is considered to be specific for tracheobronchial injury. The affected lung appears to fall away from the hilum instead of toward it, as in the case of pneumothorax.2 Treatment involves fiberoptic bronchoscopic intubation with endotracheal tube placement beyond the injured site.2,15 Also, more than one chest tube may be required to re-expand the affected lung.
Penetrating cardiac injury involves the right ventricle in approximately 43% of cases, which is due its anterior location. The left ventricle is affected in about 34% of cases, and the atria about 20%.4 The mortality rate for these injuries is high, ranging from 60-80%.5 In the United States, penetrating cardiac injuries are more often due to gunshot wounds than stab wounds.16,17 However, as many as 80% of patients with stab wounds develop tamponade.4,5 Entrance wounds located in the area between the clavicles superiorly, infracostal margin inferiorly, and the midclavicular lines laterally (the "cardiac box") should be presumed to have a cardiac injury.3,5 Patient presentation is usually in one of two states: hemorrhagic shock or cardiac tamponade. This is dependent on the injury, with larger injuries that communicate with the pleural cavity allowing continued hemorrhage, and smaller pericardial injuries that seal causing tamponade. This may be distinguished by bedside echocardiogram during the focused assessment with sonography for trauma (FAST) exam. The treatment for communicating rupture is operative intervention and may require emergency department thoracotomy (EDT).2,4
Pericardial tamponade classically presents with Beck's triad: hypotension, muffled heart tones, and distended neck veins.2-4,15 A high index of suspicion is needed, though, because not all three of these signs may be present. For example, muffled heart tones may be difficult to distinguish in the noisy ED. Also, depending on the patient's hemodynamics, there may not be distension of neck veins. As little as 50 mL of blood can cause acute pericardial tamponade.2,4 Pericardial tamponade requires definitive management with pericardial window or thoracotomy, although pericardiocentesis may be temporarily therapeutic if the blood has not clotted. Pericardiocentesis may not stabilize the patient due to inability to remove clotted blood from the pericardium. Patients in extremis may require EDT.
The incidence of great vessel injury in penetrating trauma is about 4% and tends to be more prevalent in gunshot wounds than stab wounds.2,18 The low incidence of emergency department presentation is considered to be due largely to patients exsanguinating prior to arrival at the emergency department. Possible indicators of these injuries include basilar neck wound, transmediastinal gunshot wound, or a pulseless extremity. Penetrating aortic injury is rare and carries a reported mortality of 93% and appears to be more lethal than penetrating cardiac wounds.18 Subclavian vascular injury is the most common great vessel injury, with the subclavian vein being injured more frequently than the artery. Most of these patients are in severe shock and will have massive hemothoraces.18 In these cases, it may be beneficial to autotransfuse the patient if the resources are available. Due to their location behind the clavicle, control of bleeding with pressure is anatomically difficult. If there is an obvious open wound in the supraclavicular area, tamponade to control bleeding may be achieved using 1-2 Foley catheter balloons. Insert one balloon into the wound tract as far as possible and then inflate the balloon. As seen in Figure 1, with the application of traction, this may compress the subclavian vessels. An additional Foley balloon may be placed above the first within the wound tract.18 Patients arriving in extremis will require EDT or urgent thoracotomy in the operating room.
Figure 1: Foley Catheter Balloon Tamponade of Subclavian Vessels
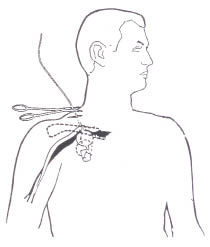
Courtesy of: D. Demetriades, MD, PhD, FACS, Professor of Surgery, Director of Trauma/Emergency Surgery/Surgical Critical Care, LAC+USC Medical Center, University of Southern California, Los Angeles.
Diaphragmatic injury is common after penetrating injury. Any patient with penetrating injury below the nipples and above the costal margin should have high suspicion for diaphragmatic injury.3,4,19 Most often these injuries are occult when associated with penetrating trauma, but there is potential for late herniation, which has potentially severe consequences resulting in pulmonary compromise or strangulation of abdominal contents and includes a mortality rate of up to 36%.15,19 The recommended work-up in stable, asymptomatic patients is laparoscopy because these injuries are frequently missed by common imaging modalities. Diagnostic peritoneal lavage has been used in the past, but there are inconsistent recommended RBC cutoff values, and it is less frequently performed due to the increased use of ultrasound FAST exam and CT imaging.19 If the patient is unstable or requires operative intervention for other injuries, the diaphragm is directly visualized and repaired, if necessary.3,4
Traumatic esophageal perforation is rare; however, most traumatic injuries are due to penetrating trauma.3 A high index of suspicion is necessary because many of these injuries are initially unsuspected and there are severe consequences to delayed diagnosis, especially when diagnosed beyond 24 hours from presentation. Injury patterns most suggestive of esophageal injury include transmediastinal gunshot wounds and near midline posterior stab wounds.3 Signs and symptoms may include chest pain, dysphagia, subcutaneous emphysema, and a systolic mediastinal crunching sound may be auscultated, which is referred to as "Hamman's crunch."2-5 A chest X-ray may reveal mediastinal emphysema, left-sided pleural effusion, or widened mediastinum.4 Diagnostic testing includes water-soluble contrast (e.g., Gastrografin®) esophagogram, followed by barium esophagogram if negative. Water-soluble contrast is less irritating to mediastinal structures if a positive study is found, while barium has increased sensitivity and image quality if the initial study is negative.2,4,5 Alternatively, emergent bedside esophageal endoscopy may be used after a negative water-soluble contrast study. Barium should not precede endoscopy, as it obscures the visual field. Also, CT imaging is an additional adjunct in the stable patient, although not necessary if esophagogram or endoscopy reveal injury. Injured patients require broad-spectrum antibiotics and emergent surgical consultation. Surgical drainage and repair is the standard of care, although some rare cases are managed non-operatively.3,4
Transmediastinal gunshot wounds present a significant diagnostic challenge because of the many vital structures in this region. Often, these patients exsanguinate prior to hospital arrival, and mortality has been reported up to 86% for cardiac injuries, 92% in great vessel injury, and 11% from lung injury.5 Injury patterns suspicious for these scenarios include entrance and exit wounds on opposite hemithoraces, entrance wounds with metallic fragment within the opposite hemithorax, and wounds with metallic fragments seen near the mediastinum indicating a potential tract in this region.20
Patients in extremis upon arrival warrant EDT or urgent thoracotomy, and injury diagnosis takes place in the operative setting. For those with stable hemodynamics, there are various recommended testing modalities. Traditionally, these patients undergo multiple studies, including chest X-ray, angiography, esophagogram and esophageal endoscopy, bronchoscopy, echocardiogram, and subxiphoid pericardial window.3,5,20 More recently, these patients have undergone contrast-enhanced CT scanning to identify potential injuries and then appropriate additional and invasive studies as needed. Additionally, if the wound tract is found not to traverse the mediastinum, additional work-up with invasive studies is unnecessary. The use of CT allows greater gathering of information in one test, which decreases length of time out of the emergency department in these potentially unstable patients.5
Spinal injuries have been reported in nearly 10% of gunshot wound injuries of the head, neck, or trunk.21 Of these, almost two-thirds were considered significant with spinal cord involvement, surgical intervention, or prolonged immobilization.21 Gunshot wounds to the anterior trunk were associated with spinal injury about 5% of the time, and of these, about half were significant. Often these injuries were unsuspected.
Imaging
For penetrating thoracic trauma, chest X-ray, ultrasound, and CT are commonly utilized. Other imaging techniques include esophageal contrast studies, endoscopy, and bronchoscopy. This section will focus on the common and recent advancements in these techniques.
Chest X-ray is the most commonly performed imaging study for patients with penetrating thoracic trauma.5 It is used in the evaluation of skeletal structures of the chest wall, pneumothorax, hemothorax, and may show evidence of pneumomediastinum as in esophageal and tracheobronchial tree injuries. Most trauma patients present in spinal precautions, and chest X-ray is performed in the supine position. This is problematic in the discovery of smaller to moderate pneumothoraces and, as a result, they are often missed with this modality. The deep sulcus sign was described as a prominent deep lateral costophrenic angle seen on the side with suspected pneumothorax.22 However, studies have shown supine chest X-ray to be insensitive for pneumothorax, with missed diagnosis as high as 54% in one study.23 Also, as previously discussed, hemothorax may be difficult to identify on these supine images, as the blood spreads across the posterior pleural space, creating a hazy appearance.4 Both may be better detected with ultrasound or CT.
The use of ultrasound has become increasingly popular for initial evaluation of trauma patients. It is generally available in most emergency departments and does not require the patient to leave the resuscitation area. It is noninvasive and repeatable for ongoing monitoring.24 The FAST exam traditionally involves ultrasound imaging with four views: subxiphoid pericardium, bilateral subphrenic views of Morrison's pouch and the perisplenic region, and a pelvic view. It has been used to identify pericardial effusion and abdominal and pelvic intraperitoneal fluid. Recently, this exam has been extended to involve imaging of the pleural spaces to screen for pneumothorax that may be missed on plain chest X-ray.
Figure 2: Ultrasound of Pneumothorax
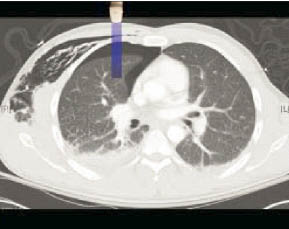
Air gathers in the anterior thoracic space in the supine trauma patient. © 2008, American College of Emergency Physicians. Used with permission.
Pneumothorax location may be dependent on patient position, amount of free air present, or structural problems with the lung itself, such as blebs or atelectasis.5 In erect patients, the air tends to collect in the apical aspect of the pleural space. In supine patients, the free air migrates toward the anterior chest wall.25 Lung ultrasound (as seen in Figure 2) has become a new, more sensitive and specific technique to detect pneumothorax, with sensitivity reported to be 58.9-98.1%, and specificity from 99.2-100%.25-31 Routine sites to examine for pneumothorax are the anteromedial chest at the second intercostal space at the midclavicular line and at the mid-axillary line at approximately the fourth or fifth intercostal space.26,27,32 A variety of transducers, including both linear and convex probes, have been used in these studies, but, most recently, the trend has been to use 3.5-5 MHz convex probes to facilitate extension of the FAST exam into scanning for pneumothoraces, which has been termed EFAST or extended FAST exam. This allows for completing the entire exam without having to change transducers.25,31 (Some examples are shown in Figure 3A–B.)
Figure 3: Ultrasound Pneumothorax
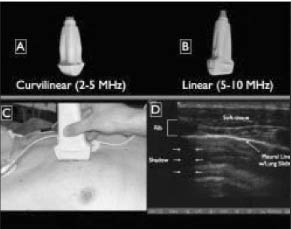
A and B: Examples of transducer probes used for pneumothorax US. C: Orientation of the transducer. D: Bright white pleural line seen below the black ovals which represent the rib shadows of transthoracic US. © 2008, American College of Emergency Physicians. Used with permission.
When scanning, the transducer probe is held perpendicular to the ribs, as in Figure 3C. The probe marker is oriented toward the head. The top of the image is the chest wall. Rib shadows can be identified, and beneath these lies the bright white line representing the pleural interface, as seen in Figure 3D. Beyond this line is the lung parenchymal tissue. In normal lung tissue, this white line is seen moving back and forth with respiration, a phenomenon known as the sliding sign.25-29,31,32 Additionally, normal lung movement causes ultrasonographic B-lines. These so-called "comet-tail" artifacts are produced due to hyperechoic reverberation artifacts and extend perpendicularly from the white line to the distal edge of the ultrasound image.25,26,28,29,32 (See Figure 4.) With a pneumothorax, these findings are not seen. Instead, white echogenic lines are seen at regular intervals parallel to the pleural line. (See Figure 4.) These lines are referred to as A-lines and are the result of ultrasound reverberation within the pneumothorax.26,32 Keep in mind that these are dynamic studies and require ventilation to be seen.32 If a patient is apneic, ventilation must be provided by bag valve mask or endotracheal tube to facilitate the use of ultrasound in identifying pneumothorax.
Figure 4: Comparison of Normal Lung and Pneumothorax
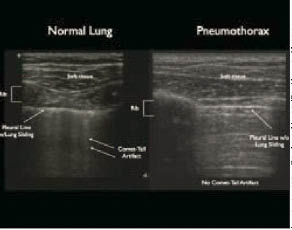
The left image shows normal lung sliding with B-lines seen as perpendicular white lines extending into the far field of US window. The right image shows a pneumothorax with no lung sliding and A-lines which are seen parallel to the pleural line. © 2008, American College of Emergency Physicians. Used with Permission.
Another method of detecting pneumothorax with ultrasound is using M-mode. Using the same locations with the ultrasound in M-mode, the normal lung will have a "seashore sign," with the chest wall resembling water meeting with the sand of the beach at the pleural interface. The sand represents the lung tissue.32 (See Figure 5A.) With a pneumothorax, there is no beach, and the picture resembles only the water. (See Figure 5B.) An additional tool for identifying normal lung sliding has been the use of power color Doppler imaging.33
Figure 5A: Seashore Sign

This image shows the seashore sign in M-mode with the sand on the beach representing pleural movement below the pleural line. Courtesy of: Robert Blankenship, MD, FACEP, Medical Director, St Vincent Medical Center Northeast, Ultrasound Director, St Vincent Emergency Physicians
Figure 5B: Pneumothorax
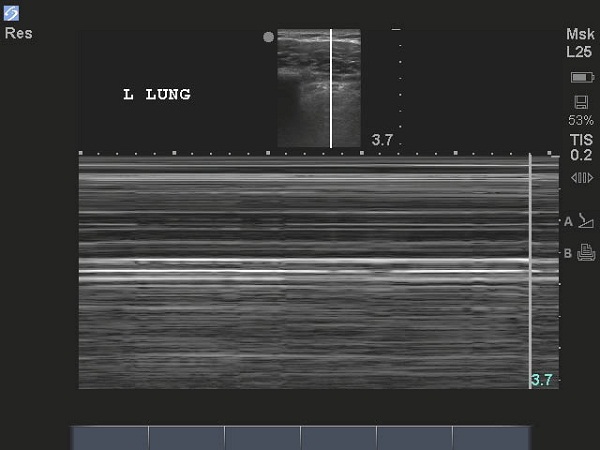
This image shows a pneumothorax in M-mode and lacks the sand/gravel appearance due to the lack of lung sliding. Courtesy of: Robert Blankenship, MD, FACEP, Medical Director, St Vincent Medical Center Northeast, Ultrasound Director, St Vincent Emergency Physicians
There have been attempts at utilizing ultrasound to define the size of pneumothoraces. Lung points are phenomena that describe views with the ultrasound in which half of the image shows normal lung sliding and B-lines, and the other half of the view lacks these signs and shows A-lines. The point where they meet indicates the edge of a pneumothorax.25,31,33 This method does have pitfalls, particularly near the heart or diaphragm where there is potential confusion with the liver or abdominal organs, giving a false lung point.32 A simpler method is to obtain views at the second intercostal space on the midclavicular line, the fourth intercostal space on the anterior axillary line, the sixth intercostal space at the midaxillary line, and the sixth intercostal space at the posterior axillary line. In this case, pneumothorax size was described as small if there was no lung sliding or B-lines at the midclavicular or anterior axillary points, medium if this extended to the midaxillary point, and large if it extended to the posterior axillary point.27 This method appeared accurate for small and large pneumothoraces, but had discrepancies when detecting medium-sized pneumothoraces.
There are pitfalls associated with ultrasound detection of pneumothoraces. This is seen when there is pre-existing lung pathology, such as blebs or pleural adhesions. Blebs or bullous emphysema may show absence of lung sliding even when no pneumothorax exists. Pulmonary adhesions may also impede the normal movement at the pleural line.27,28,32 Traumatic subcutaneous emphysema may confound the image as well. Additionally, heart motion artifact may give the appearance of lung sliding when views are obtained on the left midclavicular line. Remember, the patient must be ventilating to see lung sliding.32
Transthoracic pericardial ultrasound, or transthoracic echocardiography, is the preferred method for diagnosing cardiac injury in stable patients.5,29,34 Prior to echocardiography, the subxiphoid pericardial window was used to evaluate penetrating injuries in close proximity to the heart.29 The subxiphoid pericardial window may still be utilized in equivocal situations. The presence of blood is represented by a black stripe within the pericardium around the heart.34 This may be confounded by areas of clotted blood that will have a mixed echogenic signal within the effusion.29 It is recommended that ultrasound be utilized if pericardiocentesis is performed as a temporizing measure.4,15 The main pitfall with echocardiography in examination for hemopericardium is that there is potential for a false-negative exam if there is a pericardial laceration allowing the effusion to drain into the pleural space.16,29,34,35
CT studies are commonly used in stable patients with transmediastinal injuries. As noted previously, this modality is helpful in distinguishing the tract of penetrating injuries. It may also detect tracheobronchial injuries, vascular injuries, and esophageal injuries. CT is more sensitive than chest X-ray in detection of pneumomediastinum, which can be indicative of tracheal or esophageal injury.5,36 Additional signs of esophageal injury on CT may include defects in the esophageal wall, thickening of the wall, or extravasation of contrast if oral contrast is used.5 CT is also useful and superior to chest X-ray in diagnosing and distinguishing the extent of pulmonary contusions.5 When ultrasound is unavailable, CT may be used to diagnose hemopericardium.
Esophageal contrast studies are indicated for suspected transmediastinal injuries when the tract is confirmed by CT. Endoscopy may also be utilized at the bedside for confirmation of these injuries, although this is operator-dependent and it may be difficult to identify smaller injuries.4
Bronchoscopy is the modality of choice to evaluate tracheobronchial tree injuries.5
Disposition
Disposition of patients with penetrating thoracic trauma may seem obvious, and most people may intuitively think that all of these patients will require admission or at least overnight observation. This may be true for a large portion of these patients; however, some patients may be able to be discharged after an ED evaluation.
The asymptomatic patient with no pneumothorax on initial evaluation presents a quandary for many ED and trauma physicians. There are data showing that up to 12% of these patients develop delayed pneumothorax.38 Do all asymptomatic patients need to be kept for extended observation or can they be released? It has been generally accepted that these patients be observed for a period of 6 hours and undergo repeat chest X-ray in 6 hours.39 Recent studies have shown that, in asymptomatic patients with penetrating chest trauma and negative initial chest X-ray, a three-hour repeat chest X-ray is sufficient when compared to six-hour film.40,41 These studies did show interval development of pneumothorax on several patients within the first three hours, but none from the third to sixth hour of observation. There were no reports of delayed pneumothorax beyond discharge.
There is one study that supported immediate release of these asymptomatic patients with no occult pneumothoraces found on initial chest CT.37 In this study, asymptomatic patients with negative chest X-rays had noncontrast chest CT. If this was also negative, they were held for 6 hours and a repeat chest X-ray was obtained. If this was negative, they were released. This study with 118 patients found that if the initial CT was negative, the patient could potentially be discharged immediately, as none of the studied patients developed delayed pneumothorax.
Asymptomatic pneumothorax, also referred to as occult pneumothorax, detected on initial chest X-ray or CT of the chest, generally requires prolonged observation and admission.37,38,40-42 Pneumothoraces of 20-25% have generally fit into this observation group.4,38 It has been shown that up to 8% of these occult findings progress and require chest tube placement.42 It has been recommended that these patients undergo inpatient observation with repeat chest X-ray at 6, 24, and 48 hours, and have chest tube placement if there is progression of the pneumothorax.
It is generally accepted to treat pneumothorax in the setting of the unstable trauma patient with a large-bore (36-40 F) chest tube, as previously noted. This practice is frequently applied to the progressive occult pneumothorax as well. Does the asymptomatic and otherwise stable patient require this significant procedure? Interventional radiologists frequently utilize aspiration techniques and smaller 6-9 French catheters with Heimlich valves when there is pneumothorax after percutaneous needle biopsy.43 This aspiration technique may have some role in more typical trauma patients as well. There has also been some study of spontaneous pneumothorax to suggest that smaller caliber chest tube placement may be equally effective in the treatment of pneumothorax.44-47 These studies utilized 9 and 12 F catheters, as compared to the larger chest tubes typically used. This has not been studied in trauma patients specifically, but could be considered in stable, asymptomatic patients with small pneumothorax.
Patients with severe injuries will require thoracotomy. This is considered urgent when required for unstable patients and is generally within the first few hours after arrival at the hospital.2 There are various indications for thoracotomy. The ATLS instructs that if 1500 mL of blood is drained immediately after chest tube insertion, or there is greater than 200 mL/hr for 2-4 hours, that urgent thoracotomy be considered.15 Variations of this recommendation exist and include initial drainage of greater than 20 mL/kg or ongoing bleeding at rates of greater than 7 mL/kg/hour.4 Additional recommendations include: increasing hemothorax, persistent unstable vital signs with appropriate blood and fluid resuscitation, acute decompensation, cardiac tamponade, persistent air leak, and air embolism.2,4 Caution, however, should be exercised in the setting of coagulopathy, as the thoracotomy may worsen blood losses.
Patients presenting to the emergency department in extremis with penetrating thoracic trauma may require emergency department thoracotomy (EDT). This procedure should only be performed in the setting of appropriate surgical availability with a trauma or thoracic surgeon in close proximity and able to undertake operative intervention immediately after EDT. Indications for EDT are patients arriving after penetrating cardiac and non-cardiac injuries with witnessed or measured physiologic signs of life. These signs of life include: pupillary response, spontaneous respiration, presence of a pulse, measurable blood pressure, extremity movement, and ongoing electrical activity in the heart.49
The overall survival rate with EDT for penetrating thoracic trauma is about 9%, with the highest rates seen for stab wounds (about 17%) compared to gunshot wounds (about 4%). Resuscitation with EDT for multiple cardiac and great vessel injuries is generally futile, with survival rates less than 1%.
The presence of ED signs of life has been shown to be a positive predictor of outcome in studies.50-52 Patients with signs of life on arrival to the ED had survival rates of nearly 12%, as compared to about 3% in those with no signs of life. This was further stratified with about 9% survival if there were signs of life during transport, and 1% with no signs of life in the field. Performance of CPR is a poor predictor of outcome; resuscitation is futile with CPR time of greater than 15 minutes with no signs of life in the setting of penetrating trauma.53,54
Summary
Penetrating thoracic trauma is potentially serious, and where possible, victims should be transported directly to a trauma center. If the patient arrives at a non-trauma-center ED, immediate life threats should be addressed, and patients should be transferred expeditiously to the nearest appropriate trauma center. Some of the common injury patterns observed include chest wall injuries, pneumo- and/or hemothorax, pulmonary laceration, pulmonary contusion, tracheobronchial injuries, cardiac injuries, injuries of great vessels, diaphragmatic injuries, and esophageal injuries. Of these, the pneumo- and hemothorax are particularly common, and chest tube placement is the most common intervention in the management of thoracic trauma.
The imaging of these critical patients is evolving as well. The supine chest X-ray has been shown to be relatively ineffective at identifying pneumothorax. Ultrasound has become more popular in identifying this injury and has been shown to be more reliable. Also, the use of ultrasound in evaluating the pericardial space has become nearly essential in identifying acute cardiac tamponade and has replaced the subxiphoid pericardial window as the method of choice. CT imaging of the chest has been shown to more specifically identify injuries in stable patients and may provide key information, such as the wound tract to plan further work-up and interventions.
With respect to disposition of asymptomatic patients, current literature shows that those with no pneumothorax on initial and three-hour chest X-ray may be discharged. Those who have no pneumothorax on initial chest CT could potentially be discharged from the ED without follow-up chest X-ray. Patients with asymptomatic pneumothorax should be kept for an observation period of 24-48 hours with serial chest X-ray at 6, 24, and 48 hours. Chest tube placement is indicated if there is increasing size or clinical deterioration of these patients. Chest tube size will depend on the current treatment practices locally, but there may be potential use for smaller catheters.
Patients who are unstable or have ongoing chest tube drainage will require urgent thoracotomy in the operating room for definitive injury diagnosis and repair. Those who present in extremis may benefit from EDT when a trauma or thoracic surgeon is available to take the patient directly to the operating room after initial stabilization. The patients who are most likely to benefit from this procedure have vital signs on arrival at the emergency department, and it still may be of benefit for those with less than 15 minutes of CPR in the setting of penetrating thoracic trauma.
This information should be used to guide clinical practice and provide some direction in clinical decision making in hopes of aiding in providing the best possible patient care to these potentially critical patients.
References
1. Kochanek KD, Xu J, Murphy SL, et al. Deaths: Preliminary data for 2009. National Vital Statistics Reports 2011 March 16 [cited 2011 Nov. 13];59(4):1-51. Available from: http://www.cdc.gov/nchs/data/nvsr/nvsr59/nvsr59_04.pdf.
2. Meredith JW, Hoth JJ. Thoracic trauma: When and how to intervene. Surg Clin North Am 2007;87:95-118.
3. Hoyt DB, Coimbra R, Acosta J. Management of acute trauma. In: Townsend CM Jr., et al, eds. Sabiston Textbook of Surgery, 18th ed. Philadelphia: Saunders Elsevier; 2007:477-520.
4. Eckstein M, Henderson SO. Thoracic trauma. In: Marx JA, et al, eds. Rosen's Emergency Medicine, 7th ed. Philadelphia: Mosby Elsevier; 2009:387-413.
5. Shanmuganathan K, Matsumoto J. Imaging of penetrating chest trauma. Radiol Clin North Am 2006;44:225–238.
6. Garwe T, Cowan LD, Neas BR, et al. Directness of transport of major trauma patients to a level I trauma center: A propensity-adjusted survival analysis of the impact on short-term mortality. J Trauma 2011;70:1118-1127.
7. MacKenzie EJ, Rivara FP, Jurkovich GJ, et al. A national evaluation of the effect of trauma-center care on mortality. N Engl J Med 2006;354:366-378.
8. Nirula R, Maier R, Moore E, et al. Scoop and run to the trauma center or stay and play at the local hospital: Hospital transfer's effect on mortality. J Trauma 2010;69:595-601.
9. Sampalis JS, Denis R, Frechette P, et al. Direct transport to tertiary trauma centers versus transfer from lower level facilities: Impact on mortality and morbidity among patients with major trauma. J Trauma 1997;43:288-296.
10. Young JS, Bassam D, Cephas GA, et al. Interhospital versus direct scene transfer of major trauma patients in a rural trauma system. Am Surg 1998;64:88-92.
11. Helling TS, Davit F, Edwards K. First echelon hospital care before trauma center transfer in a rural trauma system: Does it affect outcome? J Trauma 2010;69:1362-1366.
12. Duchesne JC, barbeau JM, Islam TM, et al. Damage control resuscitation: From emergency department to the operating room. Am Surg 2011;77:201-206.
13. Cotton BA, Reddy N, Hatch QM, et al. Damage control resuscitation is associated with a reduction in resuscitation volumes and improvement in survival in 390 damage control laparotomy patients. Ann Surg 2011;254:598-605.
14. Working Group, Ad Hoc Subcommittee on Advanced Trauma Life Support, American College of Surgeons-Committee on Trauma. Initial assessment and management. In: Advanced Trauma Life Support for Doctors, 8th ed. Chicago: American College of Surgeons; 2008: 1-18.
15. Working Group, Ad Hoc Subcommittee on Advanced Trauma Life Support, American College of Surgeons-Committee on Trauma. Thoracic trauma. In: Advanced Trauma Life Support for Doctors, 8th ed. Chicago: American College of Surgeons; 2008:85-101.
16. Gunay C, Cingoz F, Kuralay E, et al. Surgical challenges for urgent approach in penetrating heart injuries. Heart Surgery Forum 2007;10:473-477. Epub 2007 December.
17. Asensio JA, Berne JD, Demetriades D, et al. One hundred five penetrating cardiac injuries: A 2-year prospective evaluation. J Trauma 1998;44:1073-1082.
18. Demetriades D. Penetrating injuries to the thoracic great vessels. J Card Surg 1997;12:173-180.
19. Leppanieme A, Haapiainen R. Occult diaphragmatic injuries caused by stab wounds. J Trauma 2003;55:646-650.
20. Richardson JD, Flint LM, Snow NJ, et al. Management of transmediastinal gunshot wounds. Surgery 1981;90:671-676.
21. Klein Y, Cohn SM, Soffer D, et al. Spine injuries are common among asymptomatic patients after gunshot wounds. J Trauma 2005;58:833-836.
22. Gordon R. The deep sulcus sign. Radiology 1980;136:25-27.
23. Neff MA, Monk JS, Peters K, et al. Detection of occult pneumothoraces on abdominal computed tomography scans in trauma patients. J Trauma 2000; 49:281-285.
24. Dolich MO, McKenney MG, Varela JE, et al. 2,576 ultrasounds for blunt abdominal trauma. J Trauma 2001;50:108–112.
25. Soldati G, Testa A, Pignataro G, et al. The ultrasonographic deep sulcus sign in traumatic pneumothorax. Ultrasound Med Biol 2006;32:1157-1163.
26. Kirkpatrick AW, Sirois M, Laupland KB, et al. Hand held thoracic sonography for detecting post-traumatic pneumothoraces: The extended focused assessment with sonography for trauma (E-FAST). J Trauma 2004;57:288–295
27. Blaivas M, Lyon M, Duggal S. A prospective comparison of supine chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad Emerg Med 2005;12:844-849.
28. Rowan KR, Kirkpatrick AW, Liu D, et al. Traumatic pneumothorax detection with thoracic US: Correlation with chest radiography and CT initial experience. Radiology 2002;225:210–214.
29. Mandavia DP, Joseph A. Bedside echocardiography in chest trauma. Emerg Med Clin North Am 2004;22:601–619.
30. Wilkerson RG, Stone MB. Sensitivity of bedside ultrasound and supine anteroposterior chest radiographs for the identification of pneumothorax after blunt trauma. Acad Emerg Med 2010;17:11-17.
31. Soldati G, Testa A, Sher S, et al. Occult traumatic pneumothorax: Diagnostic accuracy of lung ultrasonography in the emergency department. Chest 2008;133:204-211. Epub 2007 Oct 9.
32. Jones R, Blankenship R. Focus on the EFAST [internet video]. emsono.com [internet]. posted 2010 Dec. 9. [cited 2011 Nov. 13]. Available from: http://www.emsono.com/blogs/cases/archive/2010/12/09/focus-on-the-fast.aspx.
33. Cunningham J, Kirkpatrick AW, Nicolaou S, et al. Enhanced recognition of lung sliding with power color Doppler imaging in the diagnosis of pneumothorax. J Trauma 2002;52:769-771.
34. Rozycki GS, Feliciano DV, Ochsner MG, et al. The role of ultrasound in patients with possible penetrating cardiac wounds: a prospective multicenter study. J Trauma 1999;46:543-552.
35. Ball CG, Williams BH, Wyrzykowski AD, et al. A caveat to the performance of pericardial ultrasound in patients with penetrating cardiac wounds. J Trauma 2009;67:1123–1124.
36. LeBlang SD, Dolich MO. Imaging of penetrating thoracic trauma. J Thorac Imaging 2000;15:128–135.
37. Magnotti L, Weinberg J, Schroeppel T, et al. Initial chest CT obviates the need for repeat chest radiograph after penetrating thoracic trauma. Am Surg 2007;73: 569-573.
38. Ordog GJ, Wasserberger J, Balasubramanium S, et al. Asymptomatic stab wounds of the chest. J Trauma 1994;36:680-684.
39. Weigelt JA, Aurbakken CM, Meier DE, et al. Management of asymptomatic patients following stab wounds to the chest. J Trauma 1982;22:291–294.
40. Seamon M, Medina C, Pieri P, et al. Follow-up after asymptomatic penetrating thoracic injury: 3 hours is enough. J Trauma 2008;65:549-553.
41. Shatz D, Pedraja J, Erbella J, et al. Efficacy of follow-up evaluation in penetrating thoracic injuries: 3- vs. 6-hour radiographs of the chest. J Emerg Med 2001;20:281-284.
42. Muckart DJ. Delayed pneumothorax and haemothorax following observation for stab wounds of the chest. Injury 1985;16:247–248.
43. Wu CC, Maher MM, Shepard JO. Complications of CT-guided percutaneous needle biopsy of the chest: Prevention and management. AJR 2011;196:W678-W682.
44. Benton IJ, Benfield GFA. Comparison of a large and small-calibre tube drain for managing spontaneous pneumothoraces. Respir Med 2009;103:1436-1440. Epub 2009 June 7.
45. Conces DJ Jr, Tarver RD, Gray WC, Pearcy EA. Treatment of pneumothoraces utilizing small caliber chest tubes. Chest 1988;94:55-57.
46. Devanand A, Koh MS, Ong TH, et al. Simple aspiration versus chest-tube insertion in the management of primary spontaneous pneumothorax: A systematic review. Respir Med 2004;98:579-590.
47. Andrivet P, Djedaini K, Teboul JL, et al. Spontaneous pneumothorax: Comparison of thoracic drainage vs. immediate or delayed needle aspiration. Chest 1995;106:335-339.
48. Hassani B, Foote J, Borgundvaag B. Outpatient management of primary spontaneous pneumothorax in the Emergency Department of a community hospital using a small-bore catheter and a Heimlich valve. Acad Emerg Med 2009;16:513-518.
49. Working Group, Ad Hoc Subcommittee on Outcomes, American College of Surgeons-Committee on Trauma. Practice management guidelines for emergency department thoracotomy. J Am Coll Surg 2001;193:303-309
50. Molina EJ, Gaughan JP, Kulp H, et al. Outcomes after emergency department thoracotomy for penetrating cardiac injuries: A new perspective. Interactive CardioVasc Thorac Surg 2008;7:845-848. Epub 2008 July 23.
51. Rhee PM, Acosta J, Bridgeman A, et al. Survival after Emergency Department thoracotomy: Review of published data from the past 25 years. J Am Coll Surg 2000;190:288-298.
52. Seamon MJ, Shiroff AM, Franco M, et al. Emergency department thoracotomy for penetrating injuries of the heart and great vessels: An appraisal of 283 consecutive cases from two urban trauma centers. J Trauma 2009;67:1250-1258.
53. Moore EE, Knudson MM, Burlew CC, et al. Defining the limits of resuscitative emergency department thoracotomy: A contemporary Western Trauma Association perspective. J Trauma 2011;70:334-339.
54. Powell DW, Moore EE, Cothren CC, et al. Is emergency department resuscitative thoracotomy futile care for the critically injured patient requiring prehospital cardiopulmonary resuscitation? J Am Coll Surg 2004;199:211–215.
Trauma continues to be a significant cause of morbidity and mortality. Accidental death remained the fifth most common cause of death for all ages in 2009. Thoracic injuries reportedly have been involved in up to 75% of all deaths related to trauma and may be directly responsible in up to 25% of these deaths.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
