'Disappearing' Infectious Diseases
'Disappearing' Infectious Diseases
Authors:
Asim Abbasi, MD, Pediatric Emergency Medicine Fellow, University of Rochester Medical Center, Rochester, NY.
Colleen O. Davis, MD, MPH, FAAP, FACEP, Chief, Pediatric Emergency Medicine, University of Rochester Medical Center, Rochester, NY.
Peer Reviewer:
Fredrick M. Abrahamian, DO, FACEP, Associate Professor of Medicine, David Geffen School of Medicine at UCLA, Los Angeles, CA; Director of Education, Department of Emergency Medicine, Olive View–UCLA Medical Center, Sylmar, CA.
This article originally appeared in the May 23, 2011, issue of Emergency Medicine Reports.
Introduction
Vaccination has dramatically reduced the number of cases of chickenpox, measles, polio, mumps, and pertussis treated in primary care settings. Antibiotic treatment of streptococcal pharyngitis has reduced the number of cases of rheumatic fever. As a result, there are generations of physicians who have never encountered patients with these diseases. These diseases are often relegated to the historical section of general medical textbooks. However, recent isolated outbreaks in adults as well as children have brought these diseases back into the forefront1,2 for primary care providers. These outbreaks can be attributed to several factors, including waning immunity; perceived lack of need for booster shots; concern about a causal link between vaccines and neurobehavioral conditions such as autism and attention deficit hyperactivity disorder; and parental desire to decrease their children's exposure to vaccines.
This article will review the recognition and clinical management of several disappearing infectious diseases, including varicella, measles, polio, mumps, pertussis, and rheumatic fever.
Varicella
Overview. Thought to be derived from the Old English word "giccin," which translates to "itching," varicella or chickenpox is caused by the varicella-zoster virus (VZV). VZV belongs to the herpes virus family and, like all other herpes viruses, VZV causes a lifelong infection once introduced into the host. Transmission occurs by inhalation of aerosolized respiratory secretions or contact with fluid from the skin lesions. Varicella zoster disease refers to the primary infection, and herpes zoster disease refers to the reactivation of the latent virus.
Epidemiology. Chickenpox is a highly contagious viral infection. In the pre-vaccine era, complications from the primary disease caused up to 15,000 hospitalizations, with 100-150 deaths annually in the United States.3 With the introduction of the vaccine in 1995, the morbidity and mortality declined significantly.4,5 Outside the United States, increased migration of unvaccinated people has caused an increase in the incidence of varicella infection in adolescents and adults.6 One study from England showed that mortality from VZV has been increasing for the past 30 years and 80% of deaths from chickenpox are in adults.7 This shift in epidemiology is different from our previous experience with chickenpox prior to the vaccine.
Clinical Symptoms. Once infected with VZV, the incubation period, during which the patient is asymptomatic, can last from 10-21 days. Prodromal symptoms consist of fever, general malaise, headache, and abdominal pain and occur for 1-2 days prior to the onset of the skin lesions.
The classic exanthem of chickenpox consists of pruritic macules that become papular and eventually form fluid-filled vesicles. The classic description of "dewdrop on a rose petal" refers to the zone of erythema surrounding the clear blisters. Patients typically have lesions at various stages (macular, papular, or vesicular). This mixture of stages differentiates the varicella exanthem from that of smallpox. The varicella rash usually begins on the face, neck, or trunk and proceeds in a centripetal direction, or outward toward the extremities. In children, the lesions cross the hair line, are found on the scalp, and are also often present in the oropharynx, ears, nares, genitals, and on the eyelids and conjunctiva. Varicella lesions typically do not invade the cornea. The rash disappears within 3 weeks of onset.
Of note, the number of vesicular skin lesions on the body has been shown to be positively correlated with the maximum body temperature and degree of viremia.8 In addition, the varicella rash is often more extensive in older patients and in secondary household contacts who contract the disease.
Differential Diagnosis. Other more common diseases can be mistaken for a varicella rash, including disseminated meningococcus, coxsackievirus or echovirus viral exanthem, drug eruption, papular urticaria/contact dermatitis, insect bites, eczema herpeticum, or dermatitis herpetiformis. Smallpox, although now considered extinct, should be considered in the context of bioterrorism.
Diagnosis. The diagnosis of chickenpox is made clinically. If the diagnosis is uncertain, a Tzanck smear may be performed. Once a fresh vesicle is identified, it should be unroofed and the base scraped with a #15 scalpel blade. The sample can then be placed on a microscope slide for immediate analysis or placed in viral media for transfer to the lab. The presence of multinucleated giant cells indicates VZV but can also be indicative of the herpes simplex virus. Polymerase chain reaction (PCR) testing of the cell scraping is more specific and sensitive for varicella, especially if performed early in the illness.9
Serological testing is also possible, although results may not be available immediately. The utility of serologic testing is limited, but may be helpful in the management of the hospitalized patient. Assays of IgG antibodies to VZV are helpful to determine the immune status of individuals whose history of varicella is unknown or uncertain.
Complications. In the healthy pediatric patient, asymptomatic hepatitis with transient elevation of liver enzymes is a common complication of varicella infection and has no sequelae.10 Itching can lead to secondary bacterial infection of the vesicular rash and is the most common complication; invasive infection with group A streptococcus and toxic shock syndrome have been described in rare cases. Antibiotic therapy has been shown to reduce the risk of life-threatening bacterial superinfection,11 but fatal sepsis or necrotizing fasciitis may still occur. Always check for signs of cellulitis surrounding the varicella lesions.
Cough and/or dyspnea can be a sign of varicella pneumonia, which is the most common cause of death from primary varicella disease; a plain chest radiograph will show interstitial pneumonitis with diffuse bilateral infiltrates and perihilar densities.
Post-varicella acute cerebellar ataxia is the most common neurological complication of varicella, occurring once in every 4000 cases.12 This presents with the gradual onset of gait disturbance, nystagmus, and slurred speech and lasts days to weeks. Encephalitis is another serious complication.13 Nuchal rigidity, altered mental status, and seizures are clinical signs of meningoencephalitis, which is less common than ataxia but may have a chronic course in the immunocompromised patient. Age is a risk factor for central nervous system (CNS) complications of varicella infections. Patients younger than 5 years and older than 20 years are most susceptible to CNS complications.14,15
Hemorrhagic complications of varicella are more commonly seen in adults. Thrombocytopenia during acute primary varicella can be associated with bleeding into skin lesions, petechiae, purpura, epistaxis, hematuria, and gastrointestinal hemorrhage.
Treatment. Previously healthy, immunocompetent children younger than the age of 12 diagnosed with varicella do not require treatment with antiviral agents.16 Treatment is reviewed in Table 1.
Table 1: Treatment of Varicella
Oral Acyclovir
- Healthy, > 12 years old
- Chronic skin conditions
- Chronic pulmonary disease
- Taking long-term salicylate
- Corticosteroids short intermittent or aerosolized
- Secondary household cases
IV Acyclovir
- Immunosuppressed
- Secondary complications
In immunocompromised patients, acyclovir should be initiated within 2-3 days after onset of the rash. Early administration of acyclovir prevents onset of VZV pneumonia17 and decreases complications from the varicella exanthem (secondary bacterial infection).
Children with varicella should not be given aspirin due to the risk of developing Reye's syndrome.
Exposure. Children should be kept home from school or daycare until no new lesions are appearing and all the lesions have completely crusted over. Remind parents to avoid aspirin and all aspirin-containing products.
Unimmunized household contacts who are exposed to varicella should be given the varicella vaccine within 3 days.16 Varicella zoster immunoglobulin is recommended for immunocompromised patients, newborns, and pregnant patients; pregnant women are at significant risk for varicella pneumonitis. One study showed that chickenpox is five times more likely to be fatal in pregnant women than in nonpregnant adults.18
While in the ED, patients with possible varicella infection should be isolated. Primary care providers without evidence of immunity who have been exposed to a varicella patient should not care for high-risk patients for 10 to 21 days after exposure.16
Vaccination. The live-virus vaccine is available as a monovalent vaccine and in combination with the measles, mumps, and rubella (MMR) vaccine. Patients who have only had one dose of the vaccine can have breakthrough varicella with milder symptoms.
Measles
Overview. With the introduction of the measles vaccine in 1963, there was an effective eradication of the endemic disease.19 Reports of measles to the CDC were at an all-time low in 2004.20-22 Cases that occurred involved patients with international travel.23 Since 2004, the highly publicized but incorrect association of MMR vaccine with autism resulted in many parents electing to forego immunization for their children. This has led to a rise in measles and measles-related morbidity and mortality. In 2008, more measles cases were reported than in any other year since 1997; more than 90% of those infected had not been vaccinated or their vaccination status was unknown.20 In addition, a quarter of these outbreaks occurred in patients older than age 20.24
Epidemiology. The word "measles" is derived from the Latin word "misellus," which translates as "miserable." The virus belongs to the family Paramyxoviridae, and is a single-strand RNA virus. It is also known as rubeola or English measles and is not to be confused with rubella, which is German measles. The peak incidence of infection occurs during late winter and early spring and is most common in unvaccinated children younger than 2 years of age. Along with lack of immunization, pregnancy, malnutrition, and vitamin A deficiency have been linked to severe forms of the disease.25-27 The mode of entry into the body is via the respiratory mucosa or conjunctivae. Note that the measles virus can survive in aerosolized droplets for up to an hour. Once infection occurs, the incubation period is 10 days, during which the patient is usually asymptomatic.
Figure 1: Koplik Spots on the Buccal Mucosa
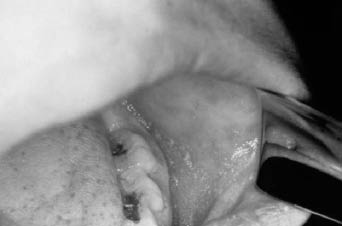
Source: Public Health Image Library, Centers for Disease Control and Prevention.
Clinical Presentation. The onset of symptoms is heralded by a high fever (usually to 104° Fahrenheit), followed by the three Cs: cough, coryza, and conjunctivitis. Koplik spots (see Figure 1), 1-2 mm white spots with a red background, found on the buccal mucosa are pathognomonic for measles. They appear during the prodrome and fade away after 48 hours. Their absence on examination does not exclude a diagnosis. The classic exanthem of measles is an erythematous maculopapular rash that begins on the face and proceeds in a caudal direction, involving the whole body, including the palms and soles. The patient will be most ill during the first 2 days after the onset of the rash. After fusing into large salmon-colored patches, most often on the face and chest, the rash will fade away within a week.
Differential Diagnosis. Because of its similarity to other pediatric exanthems, the differential diagnosis of measles is quite broad and includes infectious mononucleosis, Kawasaki disease, scarlet fever, meningococcemia, parvovirus B19 infection, roseola, rubella, and enteroviral infections. Drug eruptions from sulfonamides or phenobarbital can also mimic the exanthem seen in measles.
Diagnosis. While a clinical diagnosis of measles is often adequate in endemic areas, laboratory tests are usually recommended to confirm the diagnosis where the incidence of measles is low, such as in the United States. Serologic testing is most commonly employed; both IgM and IgG antibodies specific to measles are produced in the initial immune response. An IgM antibody assay obtained on a nasopharyngeal sample has almost 100% specificity if done within several days of onset of the rash. False-negatives may occur if the sample is obtained in the first 2 days of the rash. IgG antibody levels peak within a month and can persist for a long time.28
Complications. Several factors, such as young age (especially younger than 2 years)29 and malnutrition, can increase the severity of complications.
Acute otitis media is the most frequent complication of measles. Pneumonia is the most severe complication and a common cause of death.30 Measles-related giant-cell pneumonia is prevalent among immunocompromised patients with leukemia or HIV/AIDS and can be caused by the measles virus itself, or by superinfection with Staphylococcus aureus or Streptococcus pneumoniae.31 Other respiratory complications of measles include laryngotracheobronchitis (croup), bronchiolitis, and bacterial tracheitis.
Febrile seizures occur in less than 2.5% of children and are usually benign.32 Encephalitis, on the other hand, has a poor prognosis. Three forms of encephalitis have been attributed to measles:33
Progressive infectious encephalitis, associated with immunocompromised patients, is a progressive deterioration that leads to altered mental status and seizures.34
Post-infectious encephalitis is an immune-mediated process that involves demyelination of white matter. There is no direct viral invasion of neural cells in this form of encephalitis.35 Symptoms can include headache, vomiting, altered mental status, and weakness; these usually begin 4 to 14 days after the appearance of the rash.
Subacute sclerosing panencephalitis can occur from one to 35 years after onset of disease, when the measles virus is reactivated.36 Mean time of onset is 7 years. Patients who were infected with measles prior to the age of 2 years have a stronger association with development of subacute sclerosing panencephalitis. Symptoms include a slow onset of behavioral changes, accompanied by myoclonus, ataxia, and visual disturbances. The prognosis is very poor.
Gastrointestinal complications are less frequent than the respiratory or neurological symptoms, and are more prevalent among patients in developing countries.37 These symptoms can include anorexia, diarrhea, weight loss, and precipitation of severe protein calorie malnutrition.
Treatment. Rapid identification and isolation of patients with possible measles is imperative.
Management of primary measles is supportive. Patients with any respiratory symptoms should have a chest X-ray. Judicious use of antipyretics is also recommended. Follow-up with the primary care provider after an initial ED visit and/or hospitalization is important to monitor for post-measles complications.
For malnourished children, especially in developing countries, and for immunocompromised patients, vitamin A supplementation can reduce the morbidity and mortality from measles.38 The American Academy of Pediatrics recommends vitamin A supplementation for any child 6 months to 2 years who is hospitalized for measles and its complications.16,34,39
A recent review suggests that antibiotics will decrease the incidence of otitis media and pneumonia but have no effect on development of diarrhea, conjunctivitis, croup, or mortality.40,41 The use of antivirals, such as ribavirin and interferon, may have some benefit in immunocompromised patients, but the evidence is limited.42
Vaccination. The risk of complications from a primary measles infection is higher among children younger than 1 year of age. Therefore, current guidelines recommend that children 6 months through 11 months of age get vaccinated when exposure is imminent, such as during an outbreak or during international travel.16
Exposure. If a patient with no prior history of immunization presents within 72 hours after an exposure to measles, the vaccine may be administered.
In addition to the vaccine, intramuscular immunoglobulin is recommended for patients at high risk for severe sequelae of the disease if they present within 6 days of exposure.16,43 High-risk patients include pregnant women, children with HIV/AIDS, and infants younger than 1 year of age.
Polio
Overview. The word "polio" originates from the Greek word for "gray," which refers to the gray matter in the spinal cord that is infected during acute poliomyelitis. Although paralysis is a feared complication of this febrile illness, it is a rare complication. Most poliovirus infections are not apparent to the patient.
The poliovirus is an enterovirus, a group that also includes the Coxsackie (of hand-foot-and-mouth disease) and echoviruses. It is spread via the fecal excretion and pharyngeal secretions of the patient, with the former being the major mode of transmission.
Epidemiology. The introduction of the Sabin and Salk vaccine44 in the early 1950s resulted in a marked decrease in the incidence of polio in industrialized countries. Worldwide public health initiatives by the World Health Organization have focused on eradication of the natural form of the disease.
In the United States, the last case of wild poliovirus disease was seen in 197945; since then, all reported cases in the United States have been vaccine-associated. Isolated outbreaks are still common in rural parts of India and Africa, where warfare and political upheaval make distribution of vaccine difficult.
Prior to the industrial revolution, most polio infections occurred in infants. Because infants still had antibodies passively obtained from their mothers, they were spared from developing a primary viremia and the subsequent invasion of the CNS that causes paralysis. This allowed infants to acquire active immunity to polio while having general passive immunity. Advances in public sanitation and personal hygiene protected the public from many infectious diseases, but delayed the patient's infection with the poliovirus.46 Thus, children in developed countries were infected with the poliovirus after their passively gained antibodies had waned. Infection without antibodies led to primary viremia and infection of the CNS.47
Clinical Presentation and Complications. The incubation period can range from 1 to 4 weeks. The most common manifestation is inapparent (subclinical) polio, which occurs in 90–95% of infections and causes no symptoms or sequelae. In 5% of infections, the patient experiences nonspecific symptoms, such as general malaise, fever, anorexia, vomiting, and abdominal pain. The duration of this minor form of the polio illness is usually 3–5 days, and most patients have complete recovery.
A small percentage of patients may recover temporarily, only to have a resurgence of symptoms, including fever, worsening headache, nuchal rigidity, and soreness of back muscles and limbs. This form of polio is known as non-paralytic poliomyelitis and is diagnosed clinically by the presence of nuchal and spinal rigidity.48 Urinary incontinence can occur due to transient paralysis of the bladder. There may be a temporary change in peripheral reflexes, either an increase or decrease, which usually resolves. Recovery is complete, although recent studies have shown that subclinical neuron involvement in the brain and motor neurons of the spinal cord may result in eventual muscle weakness and neuromuscular symptoms many years or even decades later.49
Paralytic poliomyelitis occurs in 0.1% of infected patients. As with non-paralytic poliomyelitis, it occurs in the second part of the biphasic illness after a period of apparent recovery. However, the difference between paralytic and non-paralytic polio is that in the second phase of the illness, the patient will start to have severe myalgias and paresthesias. Paralysis follows over several days. Since the virus invades the anterior horns of the spinal cord, the damage occurs in the lower motor neurons, resulting in flaccid paralysis.50 On exam, the deep tendon reflexes can be hyperactive briefly before disappearing completely. The involvement is asymmetric. Proximal muscles tend to be more involved than distal muscles, with the legs more commonly involved than the arms.
Compared to the involvement of the spinal cord, bulbar poliomyelitis is a form of paralytic poliomyelitis that occurs due to paralysis of the muscles innervated by the cranial nerves. Along with the facial and masticatory muscles, there is paralysis of the soft palate and pharynx.51 The symptoms can include an inability to control secretions, with dysphagia and dyspnea. Involvement of the medulla can lead to dysautonomia and prolonged periods of apnea, resulting in respiratory compromise.
Polioencephalitis is yet another clinical presentation of polio that is distinguished by confusion and altered mental status. Primarily occurring in infants, it is associated with seizures, coma, and spastic paralysis. In contrast to flaccid paralysis, spastic paralysis is due to damage of upper motor neurons. Respiratory insufficiency is common in this form of polio.
Differential Diagnosis. There are many potential causes of acute flaccid limb paralysis, including West Nile virus infection and non-polio enteroviral infection (enterovirus 7158). Guillain-Barré syndrome also can present as flaccid limb paralysis, with or without sensory loss. However, asymmetric flaccid limb paralysis without any sensory loss during or after an acute febrile illness in a nonimmunized child or young adult almost always indicates paralytic poliomyelitis, especially outside the Western hemisphere.
Non-paralytic poliomyelitis can present similar to aseptic meningitis caused by other enteroviruses.52
Post polio syndrome, with fatigue, myalgias, or paralysis, can occur in adults with a history of polio. This may be due to the further loss of anterior horn cells within a population of neurons that were compromised by the previous poliovirus infection. The overall prognosis is good, and this form of polio rarely causes further disability. Persons with this syndrome do not shed poliovirus in their secretions and/or stool and are, therefore, not infectious.
Diagnosis. As with varicella, the diagnosis of poliomyelitis can be made on clinical findings; however, in non-endemic countries confirmatory testing is obtained. The virus is isolated from two stool samples collected 24-48 hours apart. Samples can be obtained from the oropharynx or cerebrospinal fluid (CSF), but are more difficult to grow in culture.
A four-fold rise between the acute and convalescent specimens suggests poliovirus infection. This method cannot distinguish between antibodies induced by vaccine and by the wild virus.
The CSF findings are similar to other viral meningitides: mild elevation of protein and a cell count of 10 to 500/µL with predominant lymphocytes.
Treatment. There are no antiviral medications effective in the treatment of polio. Management is supportive and ranges from simple analgesic treatment for myalgias to aggressive ventilator support for respiratory failure.
Vaccination. The live-virus form given orally (OPV) is associated with vaccine-associated paralytic poliomyelitis (VAPP), especially among immunodeficient children and those receiving their first dose. This is thought to be due to a partial reversion to the neurovirulent phenotype,53 which then goes on to affect the anterior horn of the spinal cord, as in the case of the wild form. Because of this, the inactivated form is the only version currently administered in the United States.
Previously immunized adults traveling to endemic areas should be given one booster dose of inactivated polio vaccine (IPV), and nonimmunized adults should receive at least three doses,54 including two doses given 4 weeks apart and the third one 6 months later.
Mumps
Overview. Mumps presents as an acute viral infection of the salivary glands associated with fever and is caused by the mumps virus, a member of the Paramyxovirus family. Exclusive to human beings, mumps is transmitted by direct contact, respiratory droplets, or fomites, which inoculate the nasopharynx and/or upper respiratory tract.
Epidemiology. The largest epidemic of mumps in the post-vaccine era occurred in 2006 on college campuses in 40 states.55 The incidence was highest among college-age patients between the ages of 18 and 24 years and recipients of two doses of mumps vaccine. Similar to measles and pertussis, mumps is an example of a disease that had a considerable decline in its incidence, but with localized epidemics still occurring due to various factors, such as waning of immunity over time, higher population density, higher herd immunity threshold, and less effective vaccine strains.56,57 As evident from the recent outbreak, the average age of infection is also shifting from childhood to late adolescence/early adulthood.
Clinical Presentation. The incubation period can last from 2 to 3 weeks. A transient viremia can occur prior to the onset of symptoms, during which time the patient is contagious. The prodrome consists of low-grade fever, general malaise, anorexia, myalgia, and headache.
The incubation period is followed by the classic symptoms of painful swelling of the parotid glands. On physical exam, the angle of the mandible is often hidden beneath the swelling, and the opening of Stensen's duct in the oropharynx can be edematous and erythematous. The swelling of the parotid gland can also be associated with otalgia on the ipsilateral side, with upward and outward deviation of the affected ear. In 90% of cases, the contralateral parotid gland will also be affected. Rarely, the submandibular and sublingual glands will also be affected. Resolution of the pain and swelling occurs in a week.
The mumps virus has a strong affinity to the epithelial lining of glands and can also infect the testes. Fifteen to 30% of pre-pubertal males can develop epididymoorchitis up to 6 weeks after the parotitis. Epididymoorchitis is associated with swelling and tenderness of the testicle. Although a feared complication, sterility from mumps orchitis is rare.58 Resolution occurs within a week.
Complications. The most common complication of mumps is infection of the CNS. Fever, headache, vomiting, and nuchal rigidity can be present, indicating meningitis. Alterations in mental status, focal neurological deficits, and seizures occur. Other CNS complications include facial palsy, cerebellar ataxia, transverse myelitis, Guillain-Barré syndrome, and flaccid paralysis. Subclinical pancreatitis is seen. Other less common complications include myocarditis, arthritis, and transient or permanent thyroiditis. Pregnant women have an increased risk of spontaneous fetal abortion. If the neonate is carried to term, there is no increased risk of fetal malformations.
Differential Diagnosis. Other infectious causes of parotitis include S. aureus, cytomegalovirus, Epstein-Barr virus, enteroviruses, and parainfluenza. Noninfectious causes include a parotid duct stone or tumor. Mikulicz syndrome is characterized by abnormal enlargement of glands in the head and neck, including the parotid, lacrimal, and salivary glands.59 This syndrome can occur in association with other underlying disorders such as leukemia, tuberculosis, Hodgkin's lymphoma, and lupus or collagen vascular diseases.
While mumps can cause orchitis, it is not a common cause of testicular pain. Any patient who presents with tenderness and swelling of the testicle should first be evaluated for testicular torsion or sexually transmitted disease.
Diagnosis. Patients presenting with fever and parotid swelling should be screened for mumps. The virus can be isolated from nasopharyngeal swabs, urine, blood, or semen soon after the onset of symptoms. Serological confirmation with IgM and IgG testing is recommended due to the difficulty associated with isolation of the virus in culture.60
Measurement of amylase and lipase should be obtained to evaluate for the level of parotid and pancreatic involvement. CSF should be obtained for any concerning neurological symptom. Mumps meningitis is associated with a leukocytosis with predominant lymphocytes and normal to elevated glucose.
Treatment. As with polio, there is no antiviral treatment specific to mumps. Since this disease is benign and self-limited, antipyretic and analgesic treatment is usually sufficient. Steroid administration has not been shown to improve the clinical course of mumps orchitis, although it may help diminish the pain and edema.61
Vaccination. The recent outbreak of mumps in college campuses has raised the question of a third booster dose given in early adolescence.57
Pertussis
Overview. Pertussis originated as a Latin word that translates to "per" (intensive) "tussis" (cough). The "whoop" referred to in the colloquial term "whooping cough" refers to the sudden intake of breath after a coughing fit. The responsible organism is Bordetella pertussis, a gram-negative, aerobic coccobacillus that only infects humans, is spread via aerosolized droplets, and adheres to ciliated respiratory epithelium.
Epidemiology. The introduction of a vaccine in the 1940s led to a dramatic reduction in the incidence of pertussis infections.62 However, immunization has not affected the circulation of B. pertussis. The disease has remained endemic, with breakthrough epidemics occurring every 2–5 years.63 The epidemiology of pertussis infections has changed. In the pre-vaccine era, most reported cases (> 93%) were in children younger than 10 years; in the post-vaccine era, 65% of the reported cases occur in patients older than age 10.64 Modern-day pertussis is a disease affecting all age groups.
In the past 25 years, there has been an increase in the incidence of pertussis.65 The waning of vaccine-induced immunity has allowed continued transmission of the infection among adolescents and adults, with subsequent "passage to susceptible infants."66
Vaccine refusal by parents is cited as a leading cause of the recent pertussis epidemic in California,67 where infants with immature immune systems and the elderly have been at highest risk for mortality.
Clinical Symptoms. The incubation period can range from 3 to 12 days. This stage is followed by the catarrhal stage. The catarrhal stage is indistinguishable from a common upper respiratory infection. Low-grade fever can be present. The patient is most infectious during this stage, which can last up to 2 weeks. The paroxysmal stage is signaled by the paroxysms of coughing. Occasionally, an inspiratory whoop is heard. This is caused by forceful inspiration against a partially closed glottis. Post-tussive emesis is also common in pediatric patients. This paroxysmal phase is the longest phase and can last for 2 to 6 weeks, after which the convalescent phase begins, with symptoms improving slowly over the course of about two weeks.
Due to the lack of mature musculature, infants younger than 3 months often are not able to cough vigorously and will instead choke, gag, gasp, and/or become cyanotic. Periods of apnea may also be noted. Pertussis should be considered in infants who present with apnea, cyanosis, or an acute life-threatening event (ALTE).
Older children and adults with pertussis present with a history of a persistent cough. Studies have shown that pertussis is the cause of 32% of prolonged cough cases in adolescents and adults.68
Conjunctival hemorrhage is seen in pertussis due to the increased intrathoracic pressure from coughing.
Differential Diagnosis. Mycoplasma pneumoniae infections can also present with protracted episodes of coughing, but these are continuous and not sudden and paroxysmal as with pertussis. Patients with a mycoplasma infection have fever and headache. In young infants, a staccato cough and purulent conjunctival discharge suggests a Chlamydia trachomatis infection. Respiratory syncytial virus (RSV) can also present similarly to pertussis, but has more recognizable lower respiratory tract signs, such as rales and wheezing.
Diagnosis. While isolation of B. pertussis in culture remains the gold standard for diagnosis, this can be difficult. Ideal specimen collection requires placing the swab into the posterior pharynx and holding it in place for 15–30 seconds. A dacron or rayon swab should be used. Cotton swabs should not be used because they are toxic to the organism. Special culture media is required to grow the organism, which can take 7-10 days. PCR analysis of these specimens can provide results. Serologic testing comparing acute and convalescent levels of pertussis antibodies is more useful in epidemiological studies and is not employed in a clinic or emergency department setting.
It is important to note that the sensitivities of culture, PCR, and serological testing are highest when a patient presents within 7-10 days of onset of symptoms, and decrease with the duration of the illness. Testing later may lead to a false-negative test.
Leukocytosis, ranging from 15,000 to 100,000 cells/mm3, is a characteristic finding during the catarrhal stage of the disease.69 While the presence of this finding is very suggestive of pertussis (high positive predictive value), lack of this finding does not rule out the diagnosis (low negative predictive value).
Complications. The complications from a primary pertussis infection vary depending on the age of the patient. The most common complications of pertussis are pneumonia, seizures, and encephalopathy. Death can also occur but is most common in infants younger than 4 months. These infants have the highest rate of mortality (1%) from pertussis.70 Prematurity, young maternal age, and underlying cardiac and pulmonary disease are additional risk factors for mortality. Other complications include apnea and bradycardia; this can be due to the increased vagal tone prior to coughing or the obstruction and subsequent hypoxemia during a coughing spell. Pneumonia is the most common complication and a cause of death among infants with pertussis; this can be caused directly by B. pertussis or indirectly by S. aureus, S. pneumoniae, and anaerobic pathogens. Respiratory failure from pneumonia and hemorrhage are the principal causes of mortality. Seizures and encephalopathy are rare complications.
In older patients and adults, complications are primarily from the prolonged, intense coughing spells and include coughing syncope, sleep disturbance, pneumothorax and subcutaneous emphysema, rib fractures, and urinary/fecal incontinence. Carotid dissection has been reported as a severe but rare complication of pertussis in an adult.71
Treatment. Isolation of suspected pertussis is important in the ED. Patients who present with a prolonged cough without a readily identifiable cause should be placed in respiratory isolation until pertussis is ruled out or the patient has been treated for a minimum of 5 days with an appropriate antibiotic. In addition, all health care professionals should observe standard precautions and wear a respiratory mask when examining a patient with a cough when pertussis is suspected or confirmed.
A recent Cochrane review72 suggests that administration of antibiotics for the treatment of pertussis is effective in eliminating B. pertussis from patients, thus limiting the spread of infection. However, treatment does not alter the clinical course of the illness. Recommended courses include 3 days of azithromycin, 7-14 days of erythromycin, or 7 days of clarithromycin.
Table 2: Endemic Countries for Mumps, Pertussis, Varicella, Measles, and Polio85
Disease Endemic Areas
Mumps Still endemic in most countries around the world, unknown if endemic in United States
Pertussis Still endemic in most countries around the world, including United States
Varicella Still endemic in most countries around the world, unknown if still endemic to United States
Measles Most countries in Africa, Afghanistan, Bangladesh, India, Indonesia, Pakistan, Thailand, Philippines, Bulgaria, Bosnia, Brazil, Argentina; isolated epidemics in the United States
Polio Afghanistan, Nigeria, India, Pakistan
In neonates, macrolides have been associated with infantile hypertrophic pyloric stenosis. However, the risk of developing life-threatening complications of pertussis infection outweighs the potential risk of pyloric stenosis. Infants younger than 1 month should be treated with azithromycin and monitored for gastrointestinal signs of obstruction with hypertrophic pyloric stenosis. For patients allergic to macrolides, trimethoprim-sulfamethoxazole can be used.
Hospitalization is recommended for patients with pertussis who are younger than 3 months of age and for patients of all ages who present with apnea.
Exposure. Unimmunized adults and adolescents with waning immunity are significant reservoirs of infection with pertussis. Chemoprophylaxis is recommended for neonates and infant household contacts, as it may limit the secondary transmission of the virus.16 Prophylaxis can be achieved with either azithromycin or erythromycin administration. Postexposure prophylaxis with antibiotics for health care workers who had exposure to patients with confirmed pertussis is recommended, even if they had been vaccinated.73
Vaccination. For patients younger than 7 years of age, the current vaccine used in developed countries includes diphtheria, tetanus toxoid, and acellular pertussis (DTaP). This has replaced the original vaccine that consisted of whole-cell pertussis component (DTP) and had adverse side effects.
A booster vaccine containing tetanus toxoid, diphtheria, and acellular pertussis (Tdap) is recommended for adolescents at 11–18 years of age.16 Adults 19–64 years of age should receive a single dose of Tdap.74 Tdap may be given as tetanus prophylaxis.
It is important for EDs to ensure that medical staff members are protected against pertussis to prevent the risk of transmission to susceptible patients. One study showed that emergency department staff had low levels of antibody to pertussis, despite 99% having had documented childhood immunization.75
Rheumatic Fever and Rheumatic Heart Disease
Epidemiology. Acute rheumatic fever results from an autoimmune response to a group A streptococcus (GAS) infection. At the beginning of the century, acute rheumatic fever was the most common cause of acquired heart disease.76 A decline in the incidence of rheumatic fever and heart disease in the Western hemisphere has been linked to improved sanitation, antibiotic treatment of streptococcal infections, and a shift in the prevalent strains of GAS from rheumatogenic to non-rheumatogenic strains.76,77 The current incidence of the disease is 0.5/100,000, although there have been isolated outbreaks in communities.
In developing countries, the incidence of rheumatic fever remains high at 50/100,000 and is still the most common form of acquired heart disease.78 Age at onset influences the order of complications; younger children tend to develop carditis first, whereas older patients tend to develop arthritis first.
Clinical Symptoms and Diagnosis. The Jones criteria79 outline the clinical features of acute rheumatic fever. (See Table 3.)
Table 3: Jones Criteria for Rheumatic Fever
Need 2 major or 1 major + 2 minor and evidence of recent GAS infection
Major
- Carditis
- Polyarthritis
- Erythema marginatum
- Subcutaneous nodules
- Chorea
Minor
- Arthralgia
- Elevated ESR, CRP
- Prolonged PR on ECG
Carditis can present as pericarditis, myocarditis, endocarditis, or congestive heart failure. Mitral regurgitation is the most common type of valvular disorder. The aortic valve can also be affected with aortic regurgitation and/or insufficiency. Myocarditis presents as tachycardia with cardiomegaly or arrhythmias. Pericarditis can manifest as a friction rub, pericardial pain, or cardiomegaly.
The polyarthritis is migratory and involves large joints. The joints can be hot, swollen, and exquisitely tender. Sydenham's chorea consists of sudden, rapid, and purposeless movements of the face and upper extremities.80 This can be the sole presenting symptom for rheumatic fever. Of note, in the United States, acute rheumatic fever is the most common cause of chorea.81 The subcutaneous nodules are small, painless swellings over the body prominences, such as hands, feet, elbows, scalp, and vertebrae. The rash is an erythematous macule with central clearing and serpiginous borders.
For the required diagnosis of a preceding GAS infection, the slide agglutination (also known as Streptozyme or Rapid Strep) can be used. Elevation of antistreptolysin O (> 333 units), anti-DNase B, or antihyaluronidase antibody levels can also be used.
Treatment. Once diagnosis of acute rheumatic fever has been made, 1.2 million units of benzathine penicillin G should be administered intramuscularly or, alternatively, 10 days of oral procaine penicillin.16 Erythromycin can be used in the penicillin-allergic patient. Treatment with antibiotics has not been shown to alter cardiac outcomes from acute rheumatic fever.82
Anti-inflammatory therapy with salicylates has been shown to relieve symptoms of fever, arthritis, and arthralgia. Aspirin can be given in a dose of 80-100 mg/kg/day for 6 weeks, with gradual tapering over a month. Traditionally, prednisone has been used if congestive heart failure develops as a complication of acute rheumatic fever, although there the evidence is lacking.83 There is no specific therapy for chorea, but carbamazepine or valproic acid have been shown to be more effective than haloperidol.84
Conclusion
Infectious diseases such varicella and measles have fallen off the radar of many primary care providers, yet they remain endemic in many developing countries, and outbreaks occur in this country. It is also important to stay informed about health updates and advisories from local and state health departments regarding outbreaks within the geographical scope of the primary care provider.
Other diseases, such as pertussis and acute rheumatic fever, are rare but can be still encountered in the United States. Obtaining an immunization history in a patient who presents with a suspicious infectious disease is crucial. Discharge plans should include follow-up with the primary doctor for assessment of their immunization status, especially in high-risk patients such as infants, pregnant women, the elderly, and the immunocompromised.
References
1. Tugwell BD, et al. Chickenpox outbreak in a highly vaccinated school population. Pediatrics 2004;113:455-459.
2. Measles outbreak in a boarding school Pennsylvania, 2003. MMWR Morb Mortal Wkly Rep 2004;53:306-309.
3. Galil K, et al. Hospitalizations for varicella in the United States, 1988 to 1999. Pediatr Infect Dis J 2002;21:931-935.
4. Nguyen HQ, et al. Decline in mortality due to varicella after implementation of varicella vaccination in the United States. N Engl J Med 2005;352:450-458.
5. Seward JF, et al. Varicella disease after introduction of varicella vaccine in the United States, 1995-2000. JAMA 2002;287:606-611.
6. Fairley CK, Miller E. Varicella-zoster virus epidemiology a changing scene? J Infect Dis 1996;174 Suppl 3:S314-S319.
7. Rawson H, et al. Deaths from chickenpox in England and Wales 1995-7: Analysis of routine mortality data. BMJ 2001;323:1091-1093.
8. Asano Y, et al. Severity of viremia and clinical findings in children with varicella. J Infect Dis 1990;161:1095-1098.
9. Leung J, et al. Evaluation of laboratory methods for diagnosis of varicella. Clin Infect Dis 2010;51:23-32.
10. Ey JL, et al. Varicella hepatitis without neurologic symptoms or findings. Pediatrics 1981;67:285-287.
11. Jackson MA, et al. Complications of varicella requiring hospitalization in previously healthy children. Pediatr Infect Dis J 1992;11:441-445.
12. Salas AA, Nava A. Acute cerebellar ataxia in childhood: Initial approach in the emergency department. Emerg Med J 2010;27:956-957.
13. Arvin AM. Varicella-zoster virus. Clin Microbiol Rev 1996;9:361-381.
14. Preblud SR. Age-specific risks of varicella complications. Pediatrics 1981;68:14-7.
15. Preblud SR. Varicella: Complications and costs. Pediatrics 1986;78:728-735.
16. 2006 Red Book: Report of the Committee on Infectious Diseases. Elk Grove, Illinois; 2006.
17. Balfour HH, Jr. Varicella zoster virus infections in immunocompromised hosts. A review of the natural history and management. Am J Med 1988;85:68-73.
18. Harger JH, et al. Risk factors and outcome of varicella-zoster virus pneumonia in pregnant women. J Infect Dis 2002;185:422-427.
19. Orenstein WA, et al. Measles elimination in the United States. J Infect Dis 2004;189 Suppl 1:S1-3.
20. Update: Measles United States, January-July 2008. MMWR Morb Mortal Wkly Rep 2008;57:893-896.
21. Global measles mortality, 2000-2008. MMWR Morb Mortal Wkly Rep 2009;58:1321-1326.
22. Measles United States, 2004. MMWR Morb Mortal Wkly Rep 2005;54:1229-1231.
23. Muller CP, et al. Reducing global disease burden of measles and rubella: Report of the WHO Steering Committee on research related to measles and rubella vaccines and vaccination, 2005. Vaccine 2007;25:1-9.
24. Sabella C. Measles: Not just a childhood rash. Cleve Clin J Med 2010;77:207-213.
25. Markowitz LE, et al. Vitamin A levels and mortality among hospitalized measles patients, Kinshasa, Zaire. J Trop Pediatr 1989;35:109-112.
26. Feigin RD, Cherry JD. Textbook of Pediatric Infectious Diseases, 3rd ed. Philadelphia: Saunders; 1992.
27. Dossetor J, et al. Persistent measles infection in malnourished children. BMJ 1977;1:1633-635.
28. WHO. Manual for the laboratory diagnosis of measles and rubella virus infection. August 2007.
29. Perry RT, Halsey NA. The clinical significance of measles: A review. J Infect Dis 2004;189 Suppl 1:S4-16.
30. Barkin RM. Measles mortality. Analysis of the primary cause of death. Am J Dis Child 1975;129:307-309.
31. Quiambao BP, et al. Coinfection is common in measles-associated pneumonia. Pediatr Infect Dis J 1998;17:89-93.
32. Makhene MK, Diaz PS. Clinical presentations and complications of suspected measles in hospitalized children. Pediatr Infect Dis J 1993;12:836-840.
33. Barkovich AJ. Pediatric Neuroimaging. New York: Raven Press; 1990.
34. Aicardi J, et al. Acute measles encephalitis in children with immunosuppression. Pediatrics 1977;59:232-239.
35. Johnson RT, et al. Measles encephalomyelitis clinical and immunologic studies. N Engl J Med 1984;310:137-141.
36. Sever JL. Persistent measles infection of the central nervous system: Subacute sclerosing panencephalitis. Rev Infect Dis 1983;5:467-473.
37. Morley DC. Measles in the developing world. Proc R Soc Med 1974;67:1112-5.
38. Hussey GD, Klein M. A randomized, controlled trial of vitamin A in children with severe measles. N Engl J Med 1990;323:160-164.
49. American Academy of Pediatrics Committee on Infectious Diseases: Vitamin A treatment of measles. Pediatrics 1993;91:1014-1015.
40. Kabra SK, et al. Antibiotics for preventing complications in children with measles. Cochrane Database Syst Rev 2008:CD001477.
41. Garly ML, et al. Prophylactic antibiotics to prevent pneumonia and other complications after measles: Community based randomised double blind placebo controlled trial in Guinea-Bissau. BMJ 2006;333:1245.
42. Forni AL, et al. Severe measles pneumonitis in adults: Evaluation of clinical characteristics and therapy with intravenous ribavirin. Clin Infect Dis 1994;19:454-462.
43. Watson JC, et al. Measles, mumps, and rubella vaccine use and strategies for elimination of measles, rubella, and congenital rubella syndrome and control of mumps: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 1998;47:1-57.
44. Sabin AB. Present position of immunization against poliomyelitis with live virus vaccines. BMJ 1959;1:663-680.
45. Nathanson N. Eradication of poliomyelitis in the United States. Rev Infect Dis 1982;4:940-950.
46. Dauer CC. The changing age distribution of paralytic poliomyelitis. Ann N Y Acad Sci 1955;61:943-955.
47. Nathanson N, Kew OM. From emergence to eradication: The epidemiology of poliomyelitis deconstructed. Am J Epidemiol 2010;172:1213-1229.
48. Kliegman R, Nelson WE. Nelson Textbook of Pediatrics, 18th ed. Philadelphia: Saunders; 2007.
49. Rekand T, et al. Long-term follow-up of patients with nonparalytic poliomyelitis. Arch Phys Med Rehabil 2002;83:533-537.
50. Solomon T, Willison H. Infectious causes of acute flaccid paralysis. Curr Opin Infect Dis 2003;16:375-81.
51. Fox MJ, et al. Bulbar poliomyelitis. N Engl J Med 1952;247:276-279.
52. Mandell GL, et al. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases, 7th ed. Philadelphia, PA: Churchill Livingstone/Elsevier; 2010.
53. Scheld WM, et al. Infections of the Central Nervous System, 2nd ed. Philadelphia: Lippincott-Raven; 1997.
54. Prevots DR, et al. Poliomyelitis prevention in the United States. Updated recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2000;49:1-22; quiz CE1-7.
55. Marin M, et al. Mumps vaccination coverage and vaccine effectiveness in a large outbreak among college students Iowa, 2006. Vaccine 2008;26:3601-3607.
56. Dayan GH, et al. Recent resurgence of mumps in the United States. N Engl J Med 2008;358:1580-1589.
57. Quinlisk MP. Mumps control today. J Infect Dis 2010;202:655-656.
58. Werner CA. Mumps orchitis and testicular atrophy: A factor in male sterility. Ann Intern Med 1950;32:1075-1086.
59. Aleem A, et al. Mikulicz syndrome. J Pak Med Assoc 1997;47:126-129.
60. Gupta RK, et al. Mumps and the UK epidemic 2005. BMJ 2005;330:1132-1135.
61. Masarani M, et al. Mumps orchitis. J R Soc Med 2006;99:573-575.
62. Plotkin SA, Orenstein WA. Vaccines, 4th ed. Philadelphia, PA: Saunders; 2004.
63. Cherry JD. The present and future control of pertussis. Clin Infect Dis 2010;51:663-667.
64. Cherry JD. Epidemiology of pertussis. Pediatr Infect Dis J 2006;25:361-362.
65. Guris D, et al. Changing epidemiology of pertussis in the United States: Increasing reported incidence among adolescents and adults, 1990-1996. Clin Infect Dis 1999;28:1230-1237.
66. Tan T, et al. Epidemiology of pertussis. Pediatr Infect Dis J 2005;24:S10-S18.
67. Roehr B. Whooping cough outbreak hits several U.S. states. BMJ 2010;341:c4627.
68. von Konig CH, et al. Pertussis of adults and infants. Lancet Infect Dis 2002;2:744-750.
69. Sato Y, Arai H. Leukocytosis-promoting factor of Bordetella pertussis. Jpn J Med Sci Biol 1975;28:55-56.
70. Vitek CR, et al. Increase in deaths from pertussis among young infants in the United States in the 1990s. Pediatr Infect Dis J 2003;22:628-634.
71. Skowronski DM, et al. Carotid artery dissection as a possible severe complication of pertussis in an adult: Cinical case report and review. Clin Infect Dis 2003;36:e1-4.
72. Altunaiji S, et al. Antibiotics for whooping cough (pertussis). Cochrane Database Syst Rev 2007:CD004404.
73. Tiwari T, et al. Recommended antimicrobial agents for the treatment and postexposure prophylaxis of pertussis: 2005 CDC Guidelines. MMWR Recomm Rep 2005;54:1-16.
74. Recommended adult immunization schedule United States, 2011. MMWR Morb Mortal Wkly Rep 2011;60:1-4.
75. Wright SW, et al. Pertussis seroprevalence in emergency department staff. Ann Emerg Med 1994;24:413-417.
76. Massell BF, et al. Penicillin and the marked decrease in morbidity and mortality from rheumatic fever in the United States. N Engl J Med 1988;318:280-286.
77. Quinn RW. Comprehensive review of morbidity and mortality trends for rheumatic fever, streptococcal disease, and scarlet fever: The decline of rheumatic fever. Rev Infect Dis 1989;11:928-953.
78. Carapetis JR, et al. Acute rheumatic fever. Lancet 2005;366:155-168.
79. Guidelines for the diagnosis of rheumatic fever. Jones Criteria, 1992 update. Special Writing Group of the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young of the American Heart Association. JAMA 1992;268:2069-2073.
80. Cardoso F. Sydenham's chorea. Curr Treat Options Neurol 2008;10:230-235.
81. Cardoso F, et al. Chorea in fifty consecutive patients with rheumatic fever. Mov Disord 1997;12:701-703.
82. Carter ME, et al. Rheumatic fever treated with penicillin in bactericidal dosage for six weeks. Report of a small controlled trial. BMJ 1962;1:965-967.
83. Albert DA, et al. The treatment of rheumatic carditis: A review and meta-analysis. Medicine (Baltimore) 1995;74:1-12.
84. Genel F, et al. Sydenham's chorea: Clinical findings and comparison of the efficacies of sodium valproate and carbamazepine regimens. Brain Dev 2002;24:73-76.
85. Update on the Global Status of Polio. 2011. Accessed at http://wwwnc.cdc.gov/travel/content/in-the-news/polio-outbreaks.aspx.
Vaccination has dramatically reduced the number of cases of chickenpox, measles, polio, mumps, and pertussis treated in primary care settings. Antibiotic treatment of streptococcal pharyngitis has reduced the number of cases of rheumatic fever. As a result, there are generations of physicians who have never encountered patients with these diseases. These diseases are often relegated to the historical section of general medical textbooks. However, recent isolated outbreaks in adults as well as children have brought these diseases back into the forefront for primary care providers.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
