Diagnosis and Treatment of Skin and Soft Tissue Infections
Authors
Benjamin Lindquist, MD, Stanford/Kaiser Residency Program, Stanford, CA
N. Ewen Wang, MD, Associate Professor Surgery (Emergency Medicine), Associate Director Pediatric Emergency Medicine, Stanford School of Medicine, Stanford, CA
Peer Reviewer
Robert A. Felter, MD, CPE, FAAP, FACPE, Children's Hospital National Medical Center, Washington, DC; Professor of Clinical Pediatrics, Georgetown University College of Medicine
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Dietrich (editor), Dr. Skrainka (CME question reviewer), Dr. Lindquist (author), Dr. Wang (author), Dr. Felter (peer reviewer), Ms. Coplin (executive editor), and Mr. Springston (associate managing editor) report no relationships with companies related to the field of study covered by this CME activity.
Executive Summary
- Staphylococcus aureus and Streptococcus pyogenes infections are more often caused by local invasion, while Haemophilus influenzae and Streptococcus pneumoniae can be spread by hematogenous routes.
- Historically, methicillin-resistant Staphylococcus aureus (MRSA) infections were thought to be limited to patients in direct contact with the healthcare system; however, new data suggest a growing number of community-acquired MRSA (CA-MRSA) infections.
- Whirlpool baths, water slides, or community pools have also been responsible for outbreaks of Pseudomonas aeruginosa infection in healthy children.
- Necrosis, without history of other exposures such as bites, is often pathognomonic of S. pyogenes infections that may require debridement.
- With the surge of CA-MRSA, purulent skin and soft tissue infections continue to increase in prevalence in pediatric patients. One 2008 study showed that 88% of pediatric abscesses cultured S. aureus, with 85% of those being MRSA.
Skin and soft tissue infections encompass a large variety of benign and life-threatening conditions. Some are amenable to treatment on an outpatient basis while others require admission for intravenous antibiotics or emergent surgical debridement. An understanding of the anatomic relationships between skin and surrounding soft tissues is imperative in determining the correct treatments. Similarly, a careful patient history and physical exam is required to tailor treatment to specific patient populations and presumptive pathogens. Other conditions within the differential diagnosis of skin and soft tissue infections include: osteomyelitis, septic arthritis, pyomyositis, deep venous thrombosis, or allergic reaction. The authors provide a comprehensive review of common and life-threatening conditions associated with skin and soft tissue infections in pediatrics.
— Ann M. Dietrich, MD, Editor
Epidemiology
Emergency department (ED) visits for skin and soft tissue infections in children have increased dramatically in the last decade.1,2,3,4,5 Accordingly, from 1997 to 2009, hospital admission for pediatric patients with skin and soft tissue infections increased from 1.9 to 3.4 million annually.2 During that same time, pediatric patients requiring incision and drainage have doubled.2 This growing volume of patients is thought to be largely due to the emergence of community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA).
Similar to other pediatric infectious processes, specific causes of skin and soft tissue infections vary somewhat with age and exposure. Across all ages from neonate to adolescent, Streptococcus and Staphylococcus species remain the most common causes of infection. Organism and disease prevalence vary by season, with certain infections (e.g., impetigo and cellulitis) being more common in the summer months.6 Immunosuppressed children, neonates, and those with chronic diseases (e.g., diabetes) are at increased risk for development of all infections, with skin infections being no exception.7 Similarly, patients with recent surgical interventions or placement of orthopedic devices are also at risk for skin infections, mostly from skin flora.
Pathophysiology and Common Pathogens
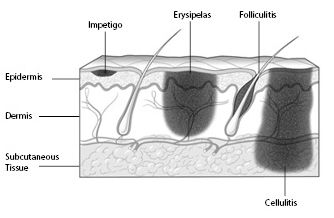
The pathophysiology of infection varies by age and bacteria involved. Most skin and soft tissue infections are the result of local invasion, but, as in neonates, may also be caused by hematogenous spread.6,7,8 For example, S. aureus and Streptococcus pyogenes infections are more often caused by local invasion, while Haemophilus influenzae and Streptococcus pneumoniae can be spread by hematogenous routes.7 In local invasion, bacteria normally present on healthy skin and enter through defects in the epidermal layer to cause infection. Clinical presentation depends on the layer of skin involved (see Figure 1).
|
Figure 1. Layer of Skin Involvement by Infection Type |
|
Source: Amieva-Wang NE, Shandro J, Sohoni A. A Practical Guide to Pediatric Emergency Medicine, Caring for Children in the Emergency Department. Cambridge University Press; 2011. (Copyright Chris Gralapp) |
S. aureus and MRSA
S. aureus colonizes the skin of 30-50% of healthy pediatric patients. Infection is transmitted via direct contact and contaminated surfaces.9 S. aureus contains many surface proteins that allow it to easily bind tissue and instrument surfaces (e.g., sutures, catheters, and prosthetic valves.)7,9 It produces numerous extracellular toxins and enzymes that contribute greatly to its virulence. A few of the important toxins include alpha- and beta-hemolysins, leukocidin, coagulases, bacteriocins, the epidermolytic toxins, staphylokinase, toxic shock syndrome type 1, and the enterotoxins. The epidermolytic toxins can separate adjacent cell layers within the epidermis and cause manifestations of staphylococcal scalded skin syndrome. A variety of enzymes, including hyaluronidase, nuclease, lipase, protease, catalase, lactate dehydrogenase, and lysozyme, are thought to contribute to the spread of local infection.7
S. aureus is split into methicillin-sensitive (MSSA) and MRSA strains, with MRSA thought to be more virulent. Historically, MRSA infections were thought to be limited to patients in direct contact with the healthcare system; however, new data suggest a growing number of CA-MRSA.10 There is a high rate of MRSA colonization of household contacts of pediatric patients with CA-MRSA, suggesting a possible household reservoir of infection.11
Recurrent MRSA infections are also becoming more common and thought to be related to patient colonization and household reservoirs. A study in 2014 by Al-Zubeidi et al suggested that 90% of recurrent infections were from the same strain of bacteria.12 These are promising data, allowing for focused decolonization efforts in eradicating recurrent disease. However, other studies have challenged this concept, suggesting that one-third of patients are colonized by different strains than the one causing infection.13 Ultimately, decolonization is an important concept for the emergency practitioner to understand in addressing treatment options and outpatient follow-up for children with recurrent MRSA skin and soft tissue infections.
Streptococci
Streptococci are gram-positive cocci that are differentiated based on their hemolytic properties (alpha or beta). Beta-hemolytic species are further divided into Lancefield groups based on the composition of their cell walls. Group A streptococcus (otherwise known as S. pyogenes) are a common cause of infection in pediatric patients, especially impetigo and erysipelas. Necrotizing skin infections can also occur with S. pyogenes. Alternatively, S. pneumonia are lancet-shaped, gram-positive, and catalase-negative diplococcic. These are encapsulated organisms posing threat to asplenic or sickle cell patients. Many children also carry S. pneumonia in their nasopharynx.
H. influenza
H. influenzae is a pleomorphic gram-negative coccobacillus that may be capsulated or encapsulated. Young infants and toddlers serve as reservoirs for these organisms, as they colonize the upper respiratory tract. H. influenza type b (Hib) more commonly causes cellulitis than the nontypable strains. This organism must be considered in children with facial cellulitis. In the vaccination era, the frequency of invasive infections has declined, but they are still a potential cause of skin infections, especially in the incompletely immunized or immunocompromised.5,7,9
Pseudomonas aeruginosa
P. aeruginosa is a catalase-positive, gram-negative rod found in the soil, water, and on vegetation. It is an opportunistic pathogen but does colonize a small number of healthy persons on mucosal surfaces, moist body sites, and in the gastrointestinal tract. P. aeruginosa colonizes the respiratory tract of hospitalized patients, patients with endotracheal tubes, patients with chronic respiratory conditions (e.g., cystic fibrosis), or those on broad-spectrum antibiotics. Skin infections most commonly occur on damaged skin, such as in burn victims, soiled wounds, or puncture injuries. The bacterium produces virulent exotoxins with proteolytic or lipolytic activity to allow for localized spread. Whirlpool baths, water slides, or community pools have also been responsible for outbreaks of infection in healthy children.7
History
The practitioner must consider the patient’s age and immunization record when diagnosing and treating skin infections. Types of wounds (e.g., puncture, animal bite) may also give a clue as to which organisms are likely to be causing a particular infection. Understanding geographic locations or travel history will provide insight into particular pathogen prevalence and exposures. A patient’s lifestyle, including intravenous drug use in adolescents, must be elicited. Patients with diabetes, renal failure, HIV, organ transplantation, cirrhosis, and those on chronic immunosuppressive medications are at risk for recurrent infections, severe infections, and infections from opportunistic organisms. Finally, previous treatments used and response can dictate decisions regarding future therapeutic modalities.1,8
Physical Exam
As with any dermatologic condition, a systematic approach to the physical exam is required to make the diagnosis, provide appropriate treatment, and aid physicians on follow-up in assessing infection changes. Abnormal vital signs, such as fever, tachycardia, hypotension, or systemic symptoms, may prompt further investigation and attention to resuscitation. Borders of erythema should be marked to assess for rapid spread while in the ED. With increasing access to electronic medical records, physicians should consider importing pictures of the wounds into the patient chart.
The physician must note the location of the infection and the surrounding skin. Well-demarcated edges should be differentiated from non-distinct ones. Streaking erythema or lymph node involvement may signify a more severe infection that could require intravenous antibiotics. The physician must also pay close attention to bone or joint involvement. In cases of facial cellulitis, evidence of orbital cellulitis, deep tissue infection, and mastoiditis must be considered.
Crepitus may be felt in skin and soft tissue infections caused by gas-forming agents and should prompt the physician to consider necrotizing infections. Bullae can be seen in minor infections such as impetigo, but large areas of bullae should raise clinical suspicion for necrotizing infections. The presence of fluctuance signifies an abscess that must be incised and drained. Necrosis, without history of other exposures, such as bites, is often pathognomonic of S. pyogenes infections that may require debridement.8
Diagnostic Adjuncts
Management of most skin infections primarily requires a careful history and physical exam. Further investigation is reserved for cases of ill-appearing children or when there is concern for complications such as necrotizing infections (which will be discussed later), septic arthritis, or osteomyelitis. Routine use of C-reactive protein (CRP), white blood cell count (WBC), and blood cultures is not recommended.14 Debate exists regarding the utilization of CRP as a better indicator of bacterial infection in pediatric patients, with some studies suggesting that CRP has a better predictive value than WBC.15 Imaging (i.e., plain radiographs, ultrasound, computed tomography [CT], or magnetic resonance imaging [MRI]) are reserved for cases in which alternative causes of infection, such as necrotizing infections or osteomyelitis, are of concern.
Disease Types: Impetigo
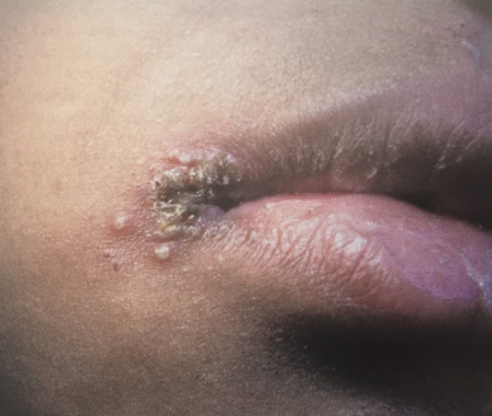
Impetigo is a skin infection caused primarily by S. aureus and S. pyogenes (see Figure 2). It is the most common bacterial skin infection in children between 2 and 5 years of age, but it can occur at any age.5 Infections occur most commonly in the summer months and spread by close physical contact.6 The typical presentation is “honey crusted” lesions on the face and arms. A majority (70%) are non-bullous infections, while some infections contain bullae. Bullous impetigo is caused by S. aureus, and large outbreaks of bullous impetigo have been traced to nurseries.6
|
Figure 2. Honey Crusted Lesions of Impetigo |
|
Source: Amieva-Wang NE, Shandro J, Sohoni A. A Practical Guide to Pediatric Emergency Medicine, Caring for Children in the Emergency Department. Cambridge University Press; 2011. (Courtesy Dr. Hans Kersten). |
The diagnosis of impetigo is clinical. Gram stain and culture of bullae or purulent lesions may be considered in the setting of uncertain diagnosis or treatment failure, but swabs often do not differentiate infection from normal colonization.1 Complications are rare and acute post-streptococcal glomerulonephritis is the most feared complication of this otherwise benign condition. Acute rheumatic fever does not occur after streptococcal skin infections.7
Treatment for impetigo is aimed at eliminating the most common bacterial etiologies of infection (see Table 1). Oral or topical antibiotics can be used.1,5,6,7,9 However, oral therapy is recommended in patients with numerous lesions and for anatomic areas where topical antibiotics are difficult to apply, such as inside the nares.1 The value of systemic antibiotics in preventing acute post-streptococcal glomerulonephritis has not been validated; however, systemic antimicrobials have been recommended during outbreaks to eliminate nephrogenic strains.1
Cellulitis and Erysipelas
Cellulitis and erysipelas are distinct infections that are differentiated by the depth of penetration within the skin. Erysipelas is an infection of the epidermis and superficial dermis that is often well demarcated and thought to be primarily caused by Streptococcus.5 Cellulitis involves the deeper dermis and subcutaneous fat. S. aureus and S. pyogenes account for most cases, with an increasing prevalence of S. aureus, particularly MRSA. Infections are generally caused by local invasion after defect or injury to skin. Cellulitis, however, can be caused by hematogenous spread in neonates and the immunosuppressed. When cellulitis occurs in patients younger than 3 months, the physician must consider group B streptococcal bacteremia.7 H. influenza must be considered in children with facial cellulitis and with chronic disease, namely sickle cell anemia. S. pneumonia skin infections are seen in asplenic patients and those with connective tissue disorders.7
A careful history and physical exam is all that is required to diagnose cellulitis and erysipelas. These lesions most commonly affect lower extremities where injuries to the skin are more common. The child will develop local inflammation at the site of infection as represented by warmth, edema, tenderness, erythema, and sometimes limited mobility.1 It may be difficult to distinguish early cellulitis from an insect bite. In contrast to cellulitis, the inflammatory reaction to insect bites typically is very well circumscribed, smaller, and associated with pruritus. However, defects in the epidermal layer from a bite or sting predispose patients to cellulitis, making it important to give strict return precautions to children with insect bites.
Treatment should be tailored to local pathogen prevalence (see Table 1). Patient exposure to different bacterium must be elicited and may require special treatment. In mild disease, monotherapy against MSSA and S. pyogenes is often sufficient.1 In cases of severe cellulitis or when associated with suppuration, MRSA coverage should be added. The recommended duration of outpatient antimicrobial treatment is 5 days but must be extended if improvement is not observed.1 Identification of clinical improvement is made easier by marking the erythematous areas and noting progression or recession of the lesion. Hospitalization should be considered in patients with systemic inflammatory response syndrome, as evidenced by tachycardia, fever or hypothermia, tachypnea, or leukocytosis. Children also should be considered for hospitalization if they are ill-appearing, hemodynamically unstable, immunosuppressed, if there is concern for necrotizing or deep infections, or if they are failing outpatient management as evidenced by lack of recession of erythema, increasing pain, or systemic symptoms.
|
Table 1. Antibiotic Treatment Options for Skin and Soft Tissue Infections |
|||
|
Disease |
Antibiotic |
Dosage |
Comments |
|
Cellulitis (MSSA and Strep) |
Nafcillin or oxacillin Cefazolin Clindamycin Dicloxacillin Cephalexin Doxycycline Trimethoprim-Sulfa |
100-150 mg/kg/d in 4 doses IV 50 mg/kg/d in 3 doses IV 10-25 mg/kg/d in 3 doses PO 35-50 mg/kg/d in 4 doses PO 25-50 mg/kg/d in 4 doses PO 100 mg PO BID (> 8 y) 8-12 mg/kg BID PO (TMP) |
Parenteral choice For PCN allergies Bacteriostatic Oral choice PCN allergic Bacteriostatic Bacteriocidal |
|
Purulent and Cellulitis (MRSA) |
Vancomycin Linezolid Clindamycin Doxycycline Trimethoprim-Sulfa |
40 mg/kg/d in 4 doses IV 10 mg/kg every 12 h IV 10-25 mg/kg/d in 3 doses PO 100 mg BID PO (> 8 y) 8-12 mg/kg BID PO (TMP) |
Parenteral choice Bacteriostatic, myelosuppression If resistance < 10-15% Bacteriostatic Bacteriocidal |
|
Erysipelas |
Cephalexin Clindamycin |
25-50 mg/kg/d in 3-4 doses PO 10-25 mg/kg/d in 3 doses PO |
|
|
Penicillin Allergy |
Clindamycin Erythromycin |
10-25 mg/kg/d in 3 doses PO 50 mg/kg/d in 2 doses PO |
|
|
Human Bite |
Penicillin V Cephalexin Amoxicillin-clavulanate |
25-50 mg/kg/d in 3 doses PO 25-50 mg/kg/d in 3-4 doses PO 25 mg/kg/d in 2 doses PO (amox) |
Eikenella corrodens |
|
Cat and Dog Bite |
Amoxicillin-clavulanate |
25 mg/kg/d in 2 doses PO (amox) |
Pasteurella multocida |
|
Puncture and Burn |
Cefepime Piperacillin-tazobactam |
100 mg/kg//d in 2 doses IV 300 mg/kg/d in 3 doses IV (pip) |
Pseudomonas aeruginosa |
|
Saltwater Exposure |
Doxycycline Trimethoprim-Sulfa |
100 mg PO BID (> 8 y) 8-12 mg/kg BID PO (TMP) (< 8 y) |
Vibrio vulnificus |
|
Adapted from: Pickering LK, Long SS. Red Book, 2012 Report of the Committee on Infectious Diseases. Am Acad Pediatrics; 2012; |
|||
|
Impetigo |
Cephalexin Clindamycin Amoxicillin-clavulanate Mupirocin |
25-50 mg/kg/d in 3-4 doses PO 10-25 mg/kg/d in 3 doses PO 25 mg/kg/d in 2 doses PO (amox) Apply to lesions BID |
Only for few lesions |
Purulent Skin and Soft Tissue Infections/Abscesses and the Rise of MRSA
With the surge of CA-MRSA, purulent skin and soft tissue infections continue to increase in prevalence in pediatric patients.16,17,18 One study in 2008 showed that 88% of pediatric abscesses cultured S. aureus, with 85% of those being MRSA.16
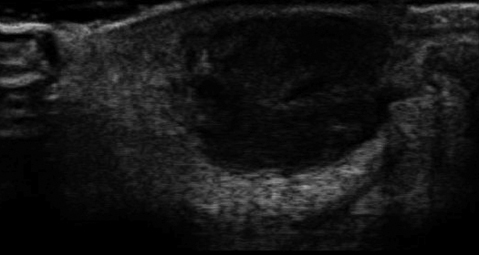
Diagnosis is made on clinical exam. Physicians may note erythematous and painful lesions with fluctuance or purulent drainage. In children younger than 2 years of age, abscesses are more commonly found in diaper and buttock areas, making it important to entirely expose these patients.18 In scenarios where diagnosis is in question, imaging modalities such as ultrasound or CT may be employed. Several recent studies have assessed the utility of bedside ultrasound in aiding the physician in the diagnosis of abscesses.19,20,21 Although these studies are small and mostly single center, some have purported a sensitivity of near 97.5% in detecting abscess by ultrasound.19 When the abscess is subclinical, the ultrasound can help change management by identifying fluid collections that would benefit from incision and drainage (see Figure 3).20 However, when the diagnosis is clinically evident on physical exam, the ultrasound is not required to make the diagnosis and initiate treatment.21
|
Figure 3. Hypoechoic Abscess Cavity on Ultrasound |
|
Courtesy of Dr. Romolo Gaspari |
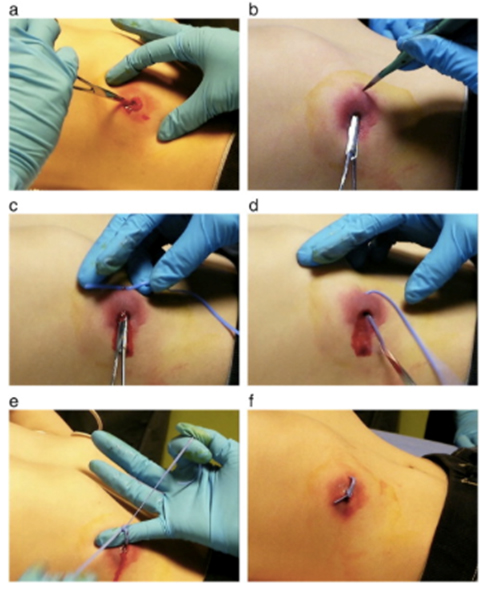
The treatment for abscesses is adequate incision and drainage, where purulent material is entirely removed and induration decreases. Specific techniques for incision and drainage warrant brief mention. Each case is different and some children, depending on age and location of abscess, may require sedation in the ED or management in the operating room. Although incision and drainage with packing has been the standard approach to abscess management for decades, new techniques have been described recently. For example, studies have questioned the utility of packing a simple abscess (< 5 cm) in healthy patients. One such study compared treatments in a prospective, randomized fashion, finding that not packing did not increase morbidity but did decrease the amount of pain and pain medication used by patients.22 Another recent study described the loop technique that utilizes vascular ties to hold the abscess cavity open and allow for continuous drainage (see Figure 4). In 2015, a study compared the two techniques and found that failure was significantly higher in the traditional technique (10.5%) compared to the loop technique (1.4%) as defined by patients requiring admission, intravenous antibiotics, or repeat drainage.23 Although these studies were done on very specific healthy adult populations, they represent a potential paradigm shift for abscess management in pediatric patients where wound management is problematic and re-packing may require sedation.23
|
Figure 4. Loop Drainage Technique |
 |
|
Source: Ladde JG, Baker S, Rodgers CN, Papa L. The loop technique: A novel incision and drainage technique in the treatment of skin abscesses in a pediatric ED. Am J Emerg Med 2015;33:271-276. (Courtesy Dr. Jay Ladde.) |
Several studies have shown that for simple abscesses < 5 cm, fluid cultures are not required.24,25,26,27 However, debate still exists among ED physicians as to the utility of culturing abscesses. The physician must decide whether the culture results will change the management. In typical cases involving healthy patients, cure rates from incision and drainage are high, regardless of which bacteria are causing the infection, and thus it is not necessary to identify the specific cause.24
Similarly, antibiotics should not be routinely used in the treatment of simple abscesses. In one study, 69 immunocompetent children with MRSA abscesses < 5 cm underwent incision and drainage and were empirically started on antibiotics. The authors found no significant difference in response to treatment between patients in which the MRSA isolated from the abscess was susceptible or resistant to the antibiotic.25 Therefore, the clinical improvement was solely related to adequate drainage and not appropriate antibiotic use. Studies like this one have led to an informal consensus that antibiotics are not needed for simple abscesses < 5 cm in healthy patients.24
However, fluid cultures and antibiotics should be used in ill-appearing patients, chronically ill or immunosuppressed patients, patients with large abscesses > 5 cm, and patients with significant surrounding cellulitis.1,24 When antibiotics are used, they should cover CA-MRSA (see Table 1).
The decision to admit these patients should be made based on the severity of illness, abnormality of vital signs, coexistent medical conditions, and the ability to tolerate oral medications. When parenteral antibiotics are needed, vancomycin or clindamycin are often utilized to cover MRSA species.5 However, resistant strains to vancomycin are emerging, and many institutional culture data are waning MRSA susceptibility to clindamycin.1
Necrotizing Soft Tissue Infections
Although rare in the general population and especially rare in healthy children, necrotizing soft tissue infections are the most feared type of skin and soft tissue infection.28 If treatment is delayed, these infections carry a very high mortality rate.
Necrotizing soft tissue infections are classified into two categories based on causative organisms.28 Type 1 infections are polymicrobial with anaerobes, gram-negative rods, and gram-positive cocci. Type 2 necrotizing soft tissue infections are caused by S. pyogenes alone or in combination with S. aureus. Although less common than type 1 infections, these infections occur in healthy and young patients.28 In the past decade, several cases of MRSA necrotizing soft tissue infections have been reported in adults and children.29,30
There are several risk factors associated with the development of necrotizing soft tissue infections. Those include infection from varicella, recent surgery, trauma, chronic illness such as diabetes, and immunosuppression.31 However, as with many deadly diseases, a large portion of patients have no identifiable risk factors.28 A population study between 2001-2005 in Canada, prior to the varicella vaccine, revealed that a majority of cases were complications of varicella infection.31 Other studies have recognized immunosuppression as the biggest risk factor for necrotizing soft tissue infections.32
Diagnosis of necrotizing soft tissue infections can be very challenging even to the seasoned physician. Presenting symptoms are often nonspecific, and patients initially may not appear critically ill. Some patients may even present to the ED prior to the development of skin changes.35 Because of this, patients are often misdiagnosed on initial presentation.28,32 The classic symptom is pain out of proportion to exam.28 According to the Canadian population study, the most common presenting complaints were pain (97%) and rash (73%).28,32 Bullae formation, crepitus, and tissue necrosis occur later, but spread rapidly as the bacterium dissect through fascial planes.28
Laboratory evaluation may be helpful, but is often nonspecific. In 2004, the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) was developed from a retrospective analysis of adult patients showing that patients with a score > 6 had a positive predictive value of 92% for necrotizing soft tissue infections (see Table 2).8,34 Further studies have questioned the external validity of these results in adults, and few studies exist to apply this system to pediatric patients, with some showing no correlation between the score and necrotizing soft tissue infections in children.35 However, a recent case report credited the LRINEC score in diagnosing a necrotizing soft tissue infection in a child with acute lymphoblastic leukemia who did not have skin changes consistent with infection.33 These conflicting results show that further study is required prior to using this scoring system primarily in evaluating pediatric patients with possible necrotizing soft tissue infections.
|
Table 2. Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) Score |
||
|
Points |
||
|
White blood cell count (109/L) < 15 15-25 > 25 |
0 1 2 |
|
|
Hemoglobin level (g/dL) > 13.5 11-13.5 < 11 |
0 1 2 |
|
|
Sodium level (mmol/L) ≥ 135 < 134 |
0 2 |
|
|
Creatinine level (mg/dL) ≤ 1.6 > 1.6 |
0 2 |
|
|
Glucose level (mg/dL) ≤ 180 > 180 |
0 1 |
|
|
Risk Low Intermediate High |
Points ≤ 5 6-7 ≥ 8 |
Probability < 50% 50-75% > 75% |
|
Adapted from: Rajan S. Skin and soft-tissue infections: Classifying and treating a spectrum. Cleve Clin J Med 2012;79:57-66. |
||
|
C-reactive protein (mg/dL) < 150 > 150 |
0 4 |
|
Similar to laboratory testing, imaging is not incredibly useful in the diagnosis of necrotizing soft tissue infections. Plain films may show gas in the soft tissue in approximately 57% of patients with non-S. pyogenes necrotizing soft tissue infections, and therefore the lack of this finding should not delay diagnosis if clinical suspicion is high. MRI and CT are more sensitive modalities in diagnosing necrotizing soft tissue infections, but should not delay surgical intervention in patients with a high clinical suspicion for necrotizing infection.28 Ultrasound can also be used to aid in the diagnosis of necrotizing soft tissue infections and requires much less time to perform. Ultrasound may reveal subcutaneous thickening, air, and fascial fluid.36 Ultimately, the diagnosis of necrotizing soft tissue infections is made in the operating room by identifying a loss of normal resistance in dissecting through fascial planes and the presence of “dishwasher fluid.”28,37
Treatment for necrotizing soft tissue infections is surgical. A delay to surgical intervention should not be tolerated in patients in whom clinical concern is high. In a study of 334 pediatric patients in 2012, non-survivors had a longer period from admission to surgical debridement.38 While surgical debridement is the main priority, these patients must also receive broad-spectrum antibiotics, such as vancomycin, piperacillin/tazobactam, and clindamycin, as well as aggressive fluid resuscitation.28
Special Populations
Children who are immunosuppressed deserve special attention. These patients require more cautious observation and treatment since they are prone to opportunistic infections and more severe disease burden. Conservative treatments, such as intravenous antibiotics and admission, should be strongly considered for even the well-appearing patients, as bacteremia and rapid clinical deterioration is common. Similarly, blood and wound cultures should be performed on a majority of these patients.1
Children with foreign bodies, such as external fixation devices or gastric tubes, require special surveillance for infection. Postoperative patients may also need specific treatments such as wound debridement or exploration. These patients are best managed in consultation with the surgical services, as decisions to interrogate a wound or remove an implant is not the sole responsibility of the ED physician.1
Conclusion
ED visits for pediatric skin and soft tissue infections are on the rise. The physician must be able to recognize and diagnose a wide spectrum of diseases, ranging from benign conditions to those that are immediately life threatening. The diagnosis is often clinical, and treatments must be tailored to the most likely organisms present in a given population of patients. Ultimately, physicians must always consider the most dangerous skin and soft tissue infections first, while also looking for complications.
References
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis 2014;59:e10-52.
- Lopez MA, Cruz AT, Kowalkowski MA, Raphael JL. Trends in resource utilization for hospitalized children with skin and soft tissue infections. Pediatrics 2013;131:e718-725.
- Pallin DJ, Espinola JA, Leung DY, et al. Epidemiology of dermatitis and skin infections in United States physicians’ offices, 1993–2005. Clin Infect Dis 2009;49:901-907.
- Pallin DJ, Egan DJ, Pelletier AJ, et al. Increased US emergency department visits for skin and soft tissue infections, and changes in antibiotic choices, during the emergence of community-associated methicillin-resistant Staphylococcus aureus. Ann Emerg Med 2008;51:291-298.
- Amieva-Wang NE, Shandro J, Sohoni A. A Practical Guide to Pediatric Emergency Medicine, Caring for Children in the Emergency Department. Cambridge University Press; 2011.
- Cherry J, Demmler-Harrison GJ, Kaplan SL, et al. Feigin and Cherry’s Textbook of Pediatric Infectious Diseases. Saunders; 2013.
- Fleisher GR, Ludwig S. Textbook of Pediatric Emergency Medicine. Lippincott Williams & Wilkins; 2010.
- Rajan S. Skin and soft-tissue infections: Classifying and treating a spectrum. Cleve Clin J Med 2012;79:57-66.
- Pickering LK, Long SS. Red Book, 2012 Report of the Committee on Infectious Diseases. Amer Academy of Pediatrics; 2012.
- Hsiang MS, Shiau R, Nadle J, et al. Epidemiologic similarities in pediatric community-associated methicillin-resistant and methicillin-sensitive Staphylococcus aureus in the San Francisco Bay Area. J Pediatric Infect Dis Soc 2012;1:200-211.
- Fritz SA, Hogan PG, Hayek G, et al. Staphylococcus aureus colonization in children with community-associated Staphylococcus aureus skin infections and their household contacts. Arch Pediatr Adolesc Med 2012;166:551-557.
- Al-Zubeidi D, Burnham CA, Hogan PG, et al. Molecular epidemiology of recurrent cutaneous methicillin-resistant Staphylococcus aureus infections in children. J Pediatric Infect Dis Soc 2014;3:261-264.
- Rodriguez M, Hogan PG, Burnham CA, Fritz SA. Molecular epidemiology of Staphylococcus aureus in households of children with community-associated S. aureus skin and soft tissue infections. J Pediatr 2014;164:105-111.
- Stevens DL, Bisno AL, Chambers HF, et al; Infectious Diseases Society of America. Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis 2005;41:1373-1406.
- Pulliam PN, Attia MW, Cronan KM. C-reactive protein in febrile children 1 to 36 months of age with clinically undetectable serious bacterial infection. Pediatrics 2001;108:1275-1279.
- Magilner D, Byerly MM, Cline DM. The prevalence of community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA) in skin abscesses presenting to the pediatric emergency department. N C Med J 2008;69:351–354.
- Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Emergency ID net study group. Methicillin-resistant S. aureus infections among patients in the emergency department. N Engl J Med 2006;355:666-674.
- Holsenback H, Smith L, Stevenson MD. Cutaneous abscesses in children: Epidemiology in the era of methicillin-resistant Staphylococcus aureus in a pediatric emergency department. Pediatr Emerg Care 2012;28:684-686.
- Iverson K, Haritos D, Thomas R, Kannikeswaran N. The effect of bedside ultrasound on diagnosis and management of soft tissue infections in a pediatric ED. Am J Emerg Med 2012;30:1347-1351.
- Sivitz AB, Lam SH, Ramirez-Schrempp D, et al. Effect of bedside ultrasound on management of pediatric soft-tissue infection. J Emerg Med 2010;39:637-643.
- Marin JR, Dean AJ, Bilker WB, et al. Emergency ultrasound-assisted examination of skin and soft tissue infections in the pediatric emergency department. Acad Emerg Med 2013;20:545-553.
- O’Malley GF, Dominici P, Giraldo P, et al. Routine packing of simple cutaneous abscesses is painful and probably unnecessary. Acad Emerg Med 2009;16:470-473.
- Ladde JG, Baker S, Rodgers CN, Papa L. The loop technique: A novel incision and drainage technique in the treatment of skin abscesses in a pediatric ED. Am J Emerg Med 2015;33:271-276.
- Abrahamian FM, Shroff SD. Use of routine wound cultures to evaluate cutaneous abscesses for community-associated methicillin-resistant Staphylococcus aureus. Ann Emerg Med 2007;50:66-67.
- Lee MC, Rios AM, Aten MF, et al. Management and outcome of children with skin and soft tissue abscesses caused by community-acquired methicillin-resistant Staphylococcus aureus. Pediatr Infect Dis J 2004;23:123-127.
- Rajendran PM, Young D, Maurer T, et al. Randomized, double-blind, placebo-controlled trial of cephalexin for treatment of uncomplicated skin abscesses in a population at risk for community-acquired methicillin-resistant Staphylococcus aureus infection. Antimicrob Agents Chemother 2007;51:4044-4048.
- Duong M, Markwell S, Peter J, Barenkamp S. Randomized, controlled trial of antibiotics in the management of community-acquired skin abscesses in the pediatric patient. Ann Emerg Med 2010;55:401-407.
- Jamal N, Teach SJ. Necrotizing fasciitis. Pediatr Emerg Care 2011;27:1195-1199.
- Miller LG, Perdreau-Remington F, Rieg G, et al. Necrotizing fasciitis caused by community-associated methicillin-resistant Staphylococcus aureus in Los Angeles. N Engl J Med 2005;352:1445-1453.
- Lalich IJ, Sam-Agudu NA. Community-acquired methicillin-resistant Staphylococcus aureus necrotizing fasciitis in a healthy adolescent male. Minn Med 2010;93:44-46.
- Eneli I, Davies HD. Epidemiology and outcome of necrotizing fasciitis in children: an active surveillance study of the Canadian Paediatric Surveillance Program. J Pediatr 2007;151:79-84, 84 e1.
- Fustes-Morales A, Gutierrez-Castrellon P, Duran-Mckinster C, et al. Necrotizing fasciitis: Report of 39 pediatric cases. Arch Dermatol 2002;138:893-899.
- Song CT, Hamilton R, Song C, et al. Enabling the diagnosis of necrotising fasciitis without associated skin changes in a paediatric patient with acute lymphoblastic leukaemia: The LRINEC score. J Plast Reconstr Aesthet Surg 2015;68:e23-24.
- Wong CH, Khin LW, Heng KS, et al. The LRINEC (Laboratory Risk Indicator for Necrotizing Fasciitis) score: A tool for distinguishing necrotizing fasciitis from other soft tissue infections. Crit Care Med 2004;32:1535-1541.
- Tancevski A, Bono K, Willis L, Klingele K. Necrotizing fasciitis in a pediatric orthopedic population. Orthopedics 2013;36:e741-745.
- Castleberg E, Jenson N, Dinh VA. Diagnosis of necrotizing faciitis with bedside ultrasound: The STAFF exam. West J Emerg Med 2014;15:111-113.
- Sarani B, Strong M, Pascual J, et al. Necrotizing fasciitis: Current concepts and review of the literature. J Am Coll Surg 2009;208:279-288.
- Endorf FW, Garrison MM, Klein MB, et al. Characteristics, therapies, and outcome of children with necrotizing soft tissue infections. Pediatr Infect Dis J 2012;31:221-223.
MONOGRAPH: A comprehensive review of common and life-threatening conditions associated with skin and soft tissue infections in pediatrics.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.