Priapism: Evaluation and Emergency Management
July 15, 2015
Reprints
AUTHORS
Jonathan Glauser, MD, Faculty, MetroHealth Medical Center, Department of Emergency Medicine, Associate Professor, Case Western Reserve University, Cleveland, OH.
Joseph Lally, MD, Emergency Medicine Resident, MetroHealth Medical Center, Cleveland, OH.
PEER REVIEWER
Steven Winograd, MD, FACEP, Core Faculty, St. Barnabas Hospital, Bronx, NY; Emergency Medicine Residency, Assistant Clinical Professor of Emergency Medicine, NYiTCOM, Westbury, LI, NY, Albert Einstein Medical School Affiliate.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Stapczynski (editor) owns stock in Pfizer, Johnson & Johnson, AxoGen, Walgreens Boots Alliance Inc., and Bristol Myers Squibb. Ms. Mark’s spouse (executive editor) works for a company that creates advertising for Uroplasty. Dr. Schneider (editor), Dr. Glauser (author), Dr. Lally (author), Dr. Winograd (peer reviewer), and Mr. Landenberger (editorial director) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Priapism occurs most commonly in young adult men.
- There is a bimodal incidence of priapism.
- Pharmacologic agents are the most common etiology of priapism in adults who do not have sickle cell disease.
- There are 2 major categories of priapism: ischemic and non-ischemic. Ischemic priapism requires urgent therapy.
- Untreated, ischemic priapism may result in permanent erectile dysfunction. The duration of priapism is the most important predictor of future erectile function.
- Testing of patients with priapism generally entails diagnostic ultrasound and cavernous blood gas analysis.
- Recurrent priapism is usually associated with sickle cell disease and should be treated as ischemic priapism.
- The aim of surgical treatment of low-flow priapism is to provide a shunt between the corpus cavernosum and glans penis or corpus spongiosum.
- Patients with nonischemic priapism can usually be managed conservatively, although patients can be managed with arterial embolization.
Priapism is one of those issues that emergency physicians must know how to recognize and treat. Several things have changed in the care of the patient with priapism recently. First the recognition of three distinct types of priapism: ischemic, nonischemic, and stuttering. Second, it is now recommended that decompression of the corpora take place early in ischemic disease. Adjuvant therapies such as ice, fluids, and analgesics should not delay definitive treatment. Finally, there are new tools to help differentiate ischemic from nonischemic disease, when the diagnosis is in doubt. These include ultrasound and aspiration from the corpus cavernous and running a blood gas analysis on that blood.
It is also important to recognize and warn the patient of possible long-term sequelae of priapism, particularly when the patient presents after many hours. Erectile dysfunction is possible after any episode of priapism, but especially common after 4-12 hours.
This article will review the most recent guidelines for the treatment of priapism.
— Sandra M. Schneider, MD, Editor
Introduction
Priapism by definition is a persistent erection of the penis or clitoris that is not associated with sexual stimulation or desire. The name is derived from the Greek and Roman figure Priapus, a deity of fertility and gardens, son of Zeus and Aphrodite. It was first described in the western literature by Petraens in 1616. The duration of an erection to define priapism, for purposes of this report, will be considered to be four hours or greater.
A relatively infrequent phenomenon, priapism is estimated to occur in as many as 5.3 per 100,000 men per year in the United States.1 Other epidemiologic reports cite an incidence of from 0.5 to 1 case per 100,000 person-years, although these were based upon European data, which may not include many patients with sickle cell disease.2,3 While relatively uncommon, it constitutes a medical emergency. Ischemic priapism is associated with erectile dysfunction and progressive fibrosis of the cavernosal tissues.4,5 The goal of therapy is to achieve detumescence and preserve erectile function. It is perhaps most frequently encountered within the context of sickle cell disease, potentially affecting all ages, but with bimodal peak incidence of between 5 and 10 years in children and 20 to 50 years in adults.6 This entity is, as a rule, rare and unpredictable, and many of the guidelines promulgated are, of necessity, based upon case reports and small case series rather than on controlled trials.5
Priapism can be differentiated into ischemic, nonischemic, and stuttering. Ischemic priapism, also referred to as veno-occlusive or low-flow, is characterized by little or no cavernous blood flow and abnormal cavernous blood gases. Aspirated cavernosal blood is hypoxic, hypercarbic, and acidotic.
Nonischemic priapism is a persistent erection caused by unregulated arterial blood flow. It is typically related to prior trauma, and is not painful. Increased arterial flow into the cavernous tissue leads to the formation of arteriocavernous shunts. There is a smaller chance of future erectile dysfunction with nonischemic priapism as compared with ischemic priapism.
Stuttering or intermittent priapism entails unwanted painful erections with intervening detumescence. These characteristically last less than 3 hours.5
“Malignant priapism” results from infiltration of the penis with cancer which has metastasized, and will not be further discussed here.7
Epidemiology
It has been estimated based upon a review of the Nationwide Emergency Department Sample of 25 and 30 million ED visits in the United States from 2006-2009 identifying 32,462 visits for priapism that the incidence of priapism is approximately 5.34 per 100,000 male subjects per year. The median age in this report was 37 years, with ischemic priapism the most frequent category. Of these, approximately 13.3% resulted in hospitalization/admission for further management.1 It appears to be more common during the summer months, with ischemic priapism being the most common type. A lower incidence of priapism has been reported in other countries, perhaps related to a lower rate of hyperviscosity states such as sickle cell disease, as noted above.2,8 Cohort studies of sickle cell populations indicate lifetime probabilities of developing priapism to be approximately one in three.9,10
Priapism in children is unusual, but can result in erectile dysfunction and penile disfigurement. It is most frequently related to sickle cell disease, but may be associated with leukemia, trauma, idiopathic, or be pharmacologically induced. Neonatal priapism has been reported in 15 per 100,000 live births. Pediatric priapism may be under-reported due to embarrassment.11
The History: Specific Elements
The history will in large part determine the urgency of treatment for priapism. The duration of the erection should be ascertained, as well as the degree of pain. Ischemic priapism is painful. Prior episodes should be determined.
A list of prescription medications should be obtained, including antihypertensives, anticoagulants, and antidepressants. Agents particularly of note include trazodone, antidepressants such as selective serotonin reuptake inhibitors (SSRIs), and other psychotropic drugs. (See Table 1.) Because trazodone has the side effect of causing priapism, it has been used to treat erectile dysfunction.
Table 1. Drugs Implicated as Possible Etiologies of Priapism
Erectile Dysfunction Pharmacotherapies
Antihypertensives
- Hydralazine
- Prazocin
- Guanethidine
- Nifedipine
- Labetalol
- Doxazosin
Antipsychotics
- Chlorpromazine
- Clozapine
- Olanzapine
- Risperidone
- Quetiapine
- Trazodone
- Thioridazine35
Antidepressants
- Trazodone
- Phenelzine citalopram
- Fluoxetine6
Alcohol
Marijuana
Cocaine
Pregabalin36
The patient should be queried about use of recreational drugs such as alcohol, marijuana, and cocaine. A history may reveal the use of vasoactive agents used for intracavernous injection such as papaverine, alprostadil, prostaglandin E, or phentolamine.
There may be a history of hematologic disease, especially sickle cell disease, although leukemia and thalassemias have been implicated, possibly related to blood viscosity. The role of sickle cell trait as a cause for priapism has been less well established.
There may be a history of penile or perineal trauma or straddle injury, predisposing to high-flow priapism.
Causes
There are a number of listed causes for priapism (see Table 2), noting firstly that the most common cause of priapism in childhood, and of stuttering priapism, is sickle cell disease.11
Table 2. Etiologies and Conditions Associated with Priapism19
Ischemic Priapism
- Sickle cell disease, other hemoglobinopathies (thalassemia, G6PD deficiency)
- Drugs
- Neoplastic disease: local or metastatic
- Leukemia, polycythemia
- Multiple myeloma
- Hyperlipidic parenteral nutrition
- Hemodialysis
- Heparin treatment
- Fabry’s disease
- Neurologic conditions: spinal cord injury, general/regional anesthesia, cauda equina compression syndrome34
High-flow (Nonischemic) Priapism
- Trauma
- Intracavernous injection
- Penile revascularization surgery
- Vasoactive drugs
Secondary causes for priapism may be grouped roughly by category and are enumerated here.
Hematologic causes include: sickle cell disease, thalassemia, multiple myeloma, thrombotic thrombocytopenic purpura, leukemia, G6PD deficiency, and polycythemia.
The relationship between priapism and sickle cell trait has been postulated, but for now is unproven.
Neurologic events may cause priapism, including spinal cord injury, spinal shock, and hanging injury. These should be elicited during a neurologic examination and by history.
Past traumatic events may cause high-flow priapism. Specific trauma includes perineal, pelvic, or penile trauma from arteriolar-sinusoidal fistula/pudendal artery laceration or injury to its branches. Priapism tends to be delayed by hours to days following direct or straddle trauma.
Iatrogenic causes include the following: intracavernous injections of papaverine, phentolamine, and prostaglandin E1. The incidence of priapism following intracavernosal injection may be as high as 0.5% per injection.8
Specific drugs listed as causes for priapism are listed by category: antidepressants; PDE5 inhibitors such as sildenafil, vardenafil, or tadalafil; alpha blockers; second-generation antipsychotics (the most common drug cause as a class in adults); methylphenidate; atomoxetine; cocaine; anticoagulants; and some antihypertensives, including hydralazine, prazosin, and guanethidine.13
Recreational substance use has been implicated, specifically alcohol, cocaine, and marijuana. Rare infectious or toxic causes have been reported such as malaria, spider toxins, and scorpion envenomation.
Metabolic causes that have been associated with priapic events include: gout, hemodialysis, total parenteral nutrition, amyloidosis, diabetes, and high lipid content such as intralipid infusion or total parenteral nutrition. Tumors associated with priapism or malignant priapism are rare, with metastatic cancer and testicular cancer reported in association with priapism.11
Pathophysiology
Penile erection entails relaxation of the smooth muscle of the cavernosal arteries and tissue. This leads to increased inflow and decreased venous outflow. Inflow of blood ceases as intracorporal pressure rises to mean arterial pressure.14 In general, the involvement is of the corpora cavernosum only, although occasionally the glans penis and the corpus spongiosum are affected as well. Penile flaccidity is maintained by high resting arterial and cavernosal smooth muscle tone. Tumescence is initiated by relaxation of cavernosal arteries and sinusoidal smooth muscle. The tunica albuginea is stretched, occluding emissary veins during full erection.11,15
Ischemic priapism is synonymous with low-flow or anoxic/veno-occlusive priapism. This is the most common form, and results in a painful, rigid erection. It is associated with failure of detumescence due to impaired relaxation of cavernosal smooth muscle. Since hypoxia and acidosis of the cavernosal tissue ensues, this is essentially a compartment syndrome. Ultrastructural changes in the corporeal smooth muscle may be seen after 12 hours of priapism. The end result of muscle necrosis from priapism is fibrosis.14 This rarely may cause necrosis of the entire penis.16
Parasympathetic and somatic neurons originating from the second to fourth sacral segments stimulate nitric oxide synthase. Nitric oxide acts on cavernosal artery smooth muscle and activates guanylate cyclase, promoting smooth muscle relaxation.15,17 It is therefore postulated that the nitric oxide-based signal transduction pathway is in some way deranged as a result of underlying disease, with decreased nitric oxide bioactivity in the penis. Phosphodiesterase type 5 (PDE5) dysregulation via a cyclic guanosine monophosphate (cGMP)-dependent protein kinase may also be a mechanism of priapism.18 The relationship between nitric oxide as a vascular smooth muscle relaxant and oral phosphodiesterase type 5 (PDE5) inhibitor therapy based on nitric oxide signaling in the penis has translated into the development of PDE5 inhibitor therapy with agents such as sildenafil, vardenafil, and tadalafil.19
Corpus cavernosal ischemia and anoxia result from ischemic priapism, with release of hypoxia-induced growth factors. Changes of impaired corpus cavernosal smooth muscle tone and responses to physiologic and pharmacologic stimuli may develop within 4 hours of the onset of priapism.13,20 This may be a critical time interval during which erectile dysfunction may develop. Later changes may include progressive fibrosis and penile tissue necrosis. The incidence of complete erectile dysfunction following major episodes of ischemic priapism has been reported to range from 30-90% in men.10,14
Nonischemic priapism tends to be less painful and not fully rigid. As noted earlier, this tends to be associated with trauma, such as straddle injury with laceration of the cavernous artery, but also possibly linked to needle injection or to urologic procedures. Congenital arterial malformations have been listed as causing high-flow priapism as well.14 Traumatically induced priapism entails excessive arterial inflow to the penis.13
Treatment of this category is less urgent. Aspiration of the corpus cavernosum has a diagnostic, but not a therapeutic role. Blood aspirated will not be as dark and will not demonstrate the hypoxemia and acidosis of ischemic priapism. This entity is considered by some not to be an emergency, as up to 62% of cases resolve spontaneously.5 Initial management of nonischemic priapism is considered to be observation, but may include ice and compression, or no treatment at all.
Patients with stuttering priapism have a pattern of recurrence over time. Treatment strategies may include early self-injection with sympathomimetic agents. Gonadotropin-releasing hormone antagonists or antiandrogens have been used, as has baclofen, for treatment.
The Physical Examination and Diagnostic Workup
In ischemic priapism, the corpus cavernosum is fully rigid and tender, while the glans penis is soft. In nonischemic (arterial) priapism, the corpora are not as rigid. There may be physical evidence for malignancy in the perineum or abdomen.
A full neurologic examination may be indicated if spinal cord injury is suspected, as noted earlier.
There are a variety of serum tests that may be considered in the diagnosis of priapism, and to elucidate its cause. The most important is cavernosal blood gas analysis (following a 19- or 21-gauge needle aspiration) for oxygen, acidemia, hypercarbia, and pH. (See Table 3.)
Table 3. Considerations for Testing in Priapism
- Serum tests (reticulocyte, CBC with differential)
- Toxicologic screening
- Hemoglobin electropheresis
- Doppler ultrasound
- Penile arteriography, selective pudendal arteriography
- Cavernosonography
- Nuclear imaging
- Blood gas testing on corpus blood
Since priapism is frequently due to hematologic problems, for an initial presentation, consideration may be given to checking a reticulocyte count for established sickle cell crisis, or hemoglobin electrophoresis to diagnose thalassemia major or sickle cell trait/disease.
A CBC may diagnose hematologic disease such as leukemia on demonstrate Howell Jolly bodies on smear, perhaps indicating hyposplenia. A toxicology screen may reveal the presence of psychoactive drugs, if suspected, although this is not usually necessary given an accurate history.
The physical examination generally is self-evident, except that there is a named finding: Piesis sign is present when perineal compression results in penile detumescence, while the erection recurs when perineal pressure is released. This strongly suggests nonischemic priapism in children, perhaps due to prior injury.11,21
Blood gas testing from penile or corpus blood and color duplex ultrasonography are the most reliable methods for distinguishing ischemic from nonischemic priapism. Blood aspirated from the corpus cavernosum is dark and may appear almost black in ischemic priapism. Typically the pO2 in cavernosal blood in men with ischemic priapism will be less than 30 mm Hg, with a pH less than 7.25 and a pCO2 of greater than 60 mm Hg.5
Color duplex Doppler ultrasonography may be used as well to distinguish nonischemic from ischemic (minimal or absent arterial blood flow) priapism. Doppler study and duplex ultrasound of the penis can show systolic and diastolic velocities of the cavernous arteries, with cavernous artery inflow and sinusoidal outflow. Patients with ischemic priapism have little or no blood flow in the cavernosal arteries of the corpora cavernosa, typically demonstrating high resistance, low-flow pattern. In patients with nonischemic priapism, color duplex doppler ultrasonography demonstrates low resistance and normal or high blood flow velocities in the cavernosal arteries.22,23 Ultrasonography can also be used as a screening test, as it may detect cavernous artery fistulas or pseudoaneurysm.5,13 Since antecedent trauma typically entails a straddle injury or direct trauma to the scrotum, the injury is most commonly found in the perineal portion of the corpora cavernosa. The ultrasound is performed in the lithotomy position, starting with the perineum and proceeding along the penile shaft.
Penile arteriography has been performed historically to identify the presence and location of a cavernous artery fistula,24 but now is used primarily for embolization, since ultrasound is effective at diagnosis, and less invasive. A pudendal arteriogram may reveal a blush at the site of injury to the cavernosal artery with nonischemic priapism. Since arteriography is only considered for high-flow priapism resistant to medical therapy, this will seldom be an emergency consideration.
Magnetic resonance imaging of the penis can evaluate smooth muscle viability, and may predict restoration of erectile function.12 This modality can predict nonviable muscle within the corpora, as well as detect rare conditions such as malignant infiltration or cavernosal thrombosis.25 This will generally be ordered by the urologic consultant, as indicated, rather than by the emergency physician.
Management
Ischemic Priapism:
Multiple etiologies of ischemic priapism exist as reviewed previously; however, in the emergency setting, management goals and strategies are the same regardless of underlying cause. Without detumescence and restoration of arterial blood flow, progressive hypoxia of the corpus cavernosum leads to corporal smooth muscle fibrosis and a long-term clinical outcome of erectile dysfunction. Impotence following priapism is correlated with the duration of symptoms. Results from the several studies vary, as all contain relatively small numbers. In one study, only 8% of patients who had symptoms for less than 24 hours became impotent, but 78% with symptoms for more than 72 hours were impotent.2 Another study of 39 patients indicated that all cases with priapism lasting more than 12 hours had problems with subsequent erection quality.26 However the hypoxia and acidosis that is seen as early as 4-6 hours of ischemic priapism has the potential to produce permanent dysfunction.13,27
Table 4. Sympathomimetic Agents of Potential Therapeutic Use in Priapism
- Phenylephrine
- Norepinephrine
- Ephedrine
- Metaraminol
- Epinephrine
Table 5. Other Agents of Potential Use in Medical Management
- Hormonal agents
– Gonadotropin-releasing hormone agonists, androgen receptor
antagonists and antiandrogens (flutamide, bicalutamide, finasteride,
dutasteride)
- Baclofen
- Digoxin
- Terbutaline
- Hydroxyurea (in sickle cell disease)
- PDE5 inhibitors
- Ketamine for sedation in children37
- Methylene blue
- Gabapentin6
- Ketoconazole
The goal of treatment, therefore, is to evacuate anoxic blood, decompress the corpora, and achieve reperfusion clinically manifested as flaccidity and relief of pain. Following successful treatment, it is possible that the penis may continue to feel tumescent secondary to post-ischemic hyperemia and residual edema. If uncertainty of success persists, a repeat penile/corpus cavernosum blood gas or ultrasonography would be appropriate for assessment of flow.25
Conservative management strategies include ejaculation, micturition, ice-pack application, oxygen, and hydration; however, the evidence to support the recommendation of any of these alternative management strategies is limited.28 Oral sympathomimetic medication (terbutaline) has been demonstrated to have success with detumescence in pharmacologic-induced priapism (self-injection for treatment of erectile dysfunction) of short duration (less than 4 hours) compared to placebo.29 Although certain patient populations may have some benefit, the available evidence and American Urological Association (AUA) guidelines do not support their use as primary therapy and should not delay direct intracavernous treatment.5
In patients with a known etiology of priapism, therapy directed at the underlying etiology (i.e., sickle cell vaso-occlusive crisis) also should not take precedence over intracavernous therapy.5 These therapies, such as hydration, packed cell transfusions, or alkalinization, may be done concurrently but not in place of or by delaying intracavernous therapy.
Table 6. Ischemic Versus Nonischemic Priapism: Features19
|
Ischemic |
Non-ischemic |
|
|
Primary Management
Aspiration alone (without sympathomimetic agents) achieved detumescence in 36% of cases. Sympathomimetics alone demonstrated a resolution rate of 58%, whereas a combination of aspiration/irrigation and sympathomimetics yielded 81% resolution.5
Intracavernous treatment should progress in a rapid stepwise fashion. Initial management with aspiration with or without irrigation is recommended as an initial approach. Anesthesia should initially be obtained through a dorsal penile nerve block. (See Figure 1.) The block is performed at the base of the dorsum of the penis using lidocaine. The dorsal nerve of the penis is adjacent to the dorsal artery, just inside Buck’s fascia, so the physician should aspirate to ensure that the artery is not injected. The dorsal nerve block should provide anesthesia to the entire dorsum of the penis.
Figure 1. Schematic Diagram of Penis Anatomy on Cross Section
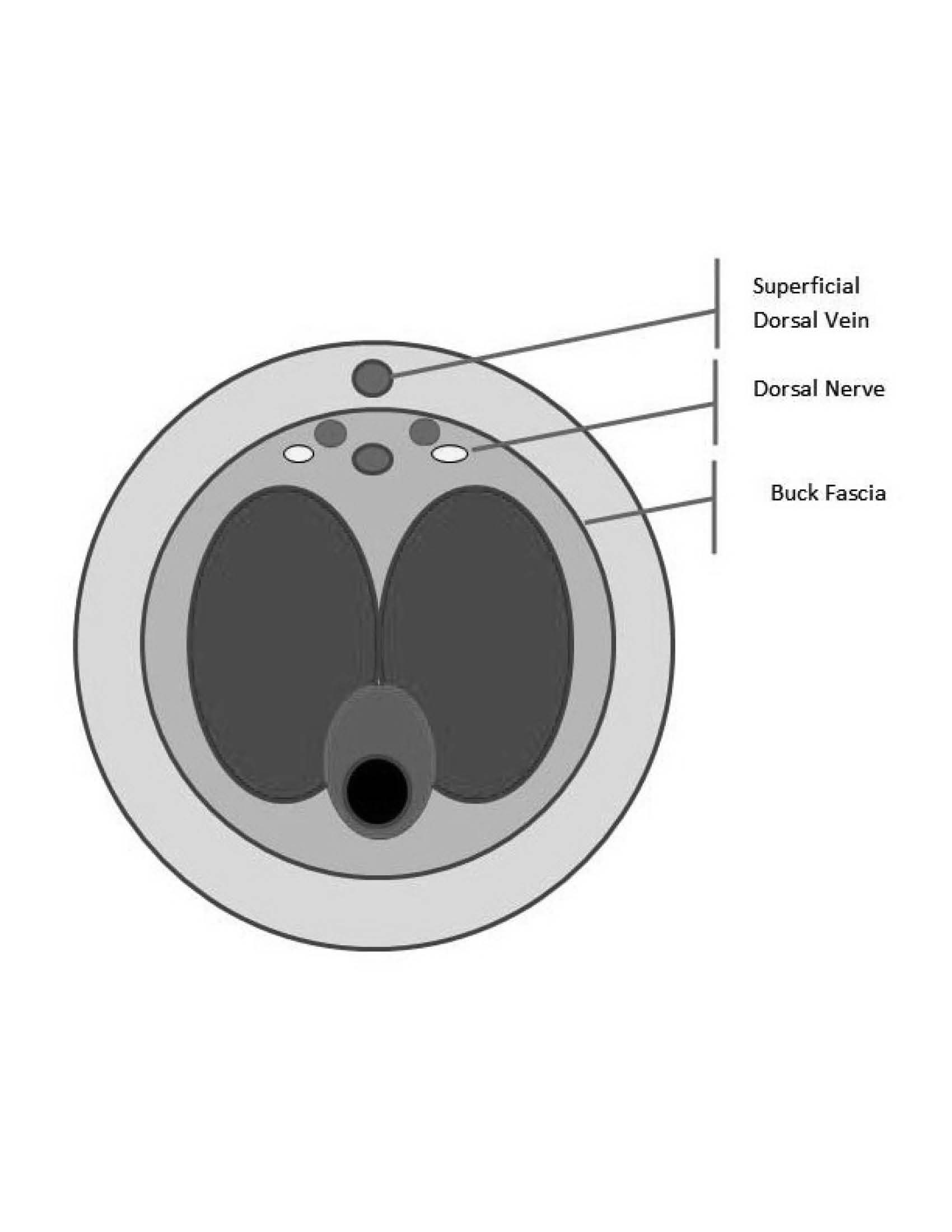
Reprinted with permission from: Mellick L. Male genital emergencies: Part II. Emerg Med Rep 2013;34:103.
Dorsal Nerve Block
A dorsal nerve block can be performed prior to aspiration of blood from the corpora cavernosa. The pudendal nerve (S2-S4) provides sensory innervation to the penis. This nerve passes under the pubis symphysis and divides into the right and left dorsal nerves that travel below the Buck fascia. The right and left dorsal penile nerves are blocked as close to the base of the penis as possible. A 27-gauge needle is used to raise skin wheals at the 2 and 10 o’clock positions with the 1% lidocaine. Next, insert the needle through the center of each skin wheal while directing it toward the center of the shaft. A loss of resistance will occur as the needle penetrates Buck fascia. After aspiration to ensure that the needle is not intravascular, slowly inject approximately 2 mL of local anesthetic on each side. Alternatively, simply inject 2 mL of local anesthetic on each side of the midline while avoiding the superficial dorsal penile vein.
Reprinted with permission from: Mellick L. Male genital emergencies: Part II. Emerg Med Rep 2013;34:103.
A 19- or 21-gauge butterfly needle or angiocath attached to a syringe is used (a butterfly is preferred to prevent dislodgment of the needle during syringe change).30,31 Needle insertion placement is based upon personal experience in order to gain access to corpora. (See Figures 2 and 3.) The most referenced approach is at approximately the 2 or 10 o’clock position at the base of the penis, although more distal insertion points along the corpora may be considered. Due to communication between corpora, only one side needs to be aspirated during the initial attempt. Ventral approaches need to be avoided to prevent injury to the corpus spongiosum and urethra, with dorsal approaches avoided to prevent injury to the neuro-vascular bundle. Approaching the corpora via a transglanular approach is also a consideration.25 The needle is advanced into the corpus until blood is aspirated and continued until fresh red oxygenated blood is obtained. Irrigation with normal saline may be considered, although there is no firm evidence for independent benefit of this over aspiration alone.5,27,31 Bilateral access may be obtained, and irrigation and aspiration can be performed simultaneously, although this is not necessary in the majority of cases.25,32
Figures 2 and 3. Anoxic Blood Is Aspirated as Part of Intracavernous Treatment
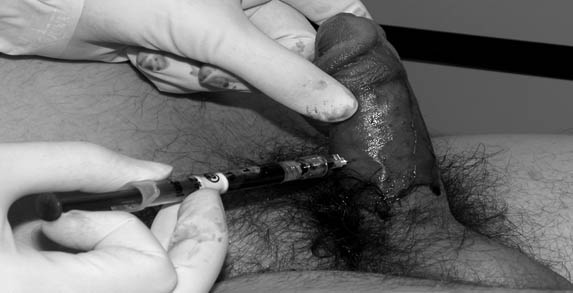
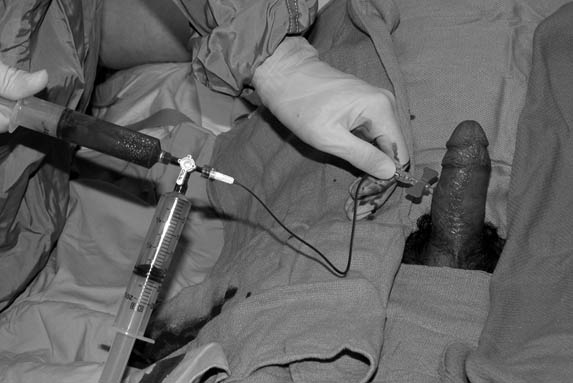
Alpha-adrenergic injection, typically with phenylephrine, may be continued for up to 1 hour.
Images courtesy of Dr. David Effron.
Failure to achieve detumescence after aspiration (with or without irrigation) should prompt intracavernous injection of a sympathomimetic agent. As mentioned previously, combination therapy with sympathomimetics in addition to aspiration significantly increases resolution up to approximately an 81% rate of success.5 AUA guidelines recommend the use of phenylephrine as the agent of choice because of decreased systemic effects. The efficacy of sympathomimetics relies on alpha receptor-mediated vasoconstriction within the corpora cavernosum. As phenylephrine is alpha selective, cardiac inotropic and chronotropic side effects are minimized, although heart rate and blood pressure monitoring are recommended in patients with cardiovascular risk.5,25,27
Phenylephrine should be diluted with normal saline to a concentration of 100-500 mcg/mL and dosed in approximately 1 mL aliquots every 3-5 minutes. Injections may be continued approximately every 3-5 minutes up to one hour prior to consideration of treatment failure.5,27 Higher doses can be considered for younger patients without cardiac risk factors before being considered treatment failure.27 For practical purposes, the clinician should prepare phenylephrine at the time of the aspiration attempt.
Patients should be monitored for known side effects including hypertension, headache, reflex bradycardia, tachycardia, palpitations, and arrhythmias. Patients with malignant or poorly controlled hypertension may be inappropriate candidates for sympathomimetic treatments.5
Secondary Management
The use of surgical shunts for resolution of ischemic priapism is considered only after failure of above nonsurgical treatments.5,13,27,32 There is a direct relationship between primary treatment failure and duration of erection. Prolonged ischemia and acidosis causes a diminished smooth muscle response to sympathomimetics. Erections lasting greater than 48 hours are unlikely to resolve, although intracavernous therapy should still be attempted.5,25,33
Shunting procedures, both percutaneous and surgical, are beyond the scope of usual emergency practice. In general, the goal of the procedure is to create a fistula to establish outflow from the corpora cavernosum in order to bypass the veno-occulsion and to drain de-oxygenated blood. Distal shunts have a higher success rate, are easier to perform, and have fewer complications.27
There are a variety of named shunts. The Winter shunt involves passing a large-bore biopsy needle percutaneously through the glans penis into the corpus cavernosum. Ebbehoj or T shunts are corporoglandular shunts which use a scalpel blade to create openings through the glans into the corpus cavernosum and openings in the tunica albuginea so that larger fistulae result than using a biopsy needle alone. The Al-Ghorab shunt involves excising a piece of tunica albuginea from the tip of the corpora cavernosa bilaterally, minimizing the likelihood of shunt closure. Proximal shunts are rarely used, but may include anastomosing the saphenous vein, superficial or deep dorsal vein to the corpora cavernosa.25
Nonischemic
The AUA recommends that initial management of nonischemic priapism should be observation alone, as this does not represent a medical or surgical emergency. The penis is not ischemic.5,27 Spontaneous resolution without intervention occurs in greater than 60% of cases, and conservative treatment including rest, ice packs, and local compression are recommended although not proven to increase resolution rates.5,27,32 Patients will require referral to a urologist for discussion of risk and benefits of procedural intervention for persistent symptoms. Selective arterial embolization is recommended as first-line intervention.5,25,31 Complications of this intervention include penile gangrene, gluteal ischemia, and purulent cavernositis, among others.25,31
Stuttering Priapism
Individual episodes or recurrent priapism should be treated following the stepwise approach as with any case of ischemic priapism, since this represents an emergent condition. Long-term treatment is aimed at prevention of recurrent episodes. Repetitive episodes may lead to an accumulation of changes within the corpora leading to permanent erectile dysfunction.25 Follow-up management is deferred to one’s urologic consultants. Available treatments include systemic therapies (hormonal, baclofen, terbutaline, among others), sympathomimetic agents via self-injection, and penile prosthesis.1,12,25,27,32
Figure 4. Management Algorithm5,12,25,27
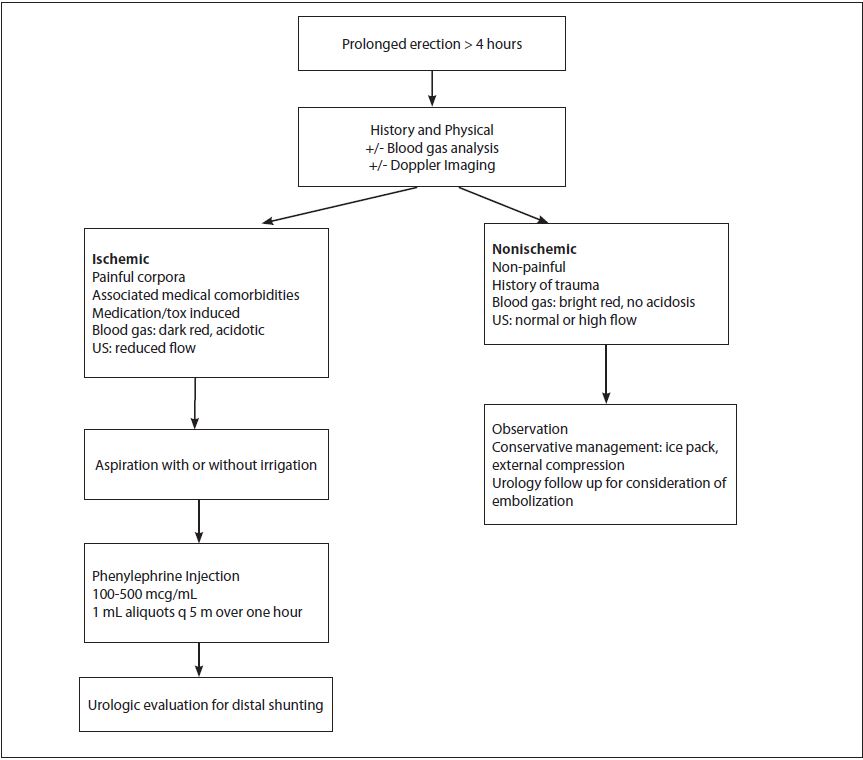
OUTCOMES
Permanent sexual dysfunction is the primary adverse sequela of priapism and directly related to duration of ischemia. Erections secondary to ischemic priapism lasting longer than 24 hours are associated with a rate of erectile dysfunction as high as 90%.14 However, since eventual recovery of erectile function may be seen in up to 44% of patients treated in 24-36 hours, timely treatment is crucial.25
Outcomes in cases of stuttering priapism depend on number and duration of recurrences, as this will reflect the underlying degree of permanent ischemic damage to the corpora. Patients should be counseled early as to the dangers of this disorder and potential for poor outcomes.
Cases of nonischemic priapism have overall good outcomes regarding maintenance of baseline sexual function, as blood flow and oxygenation are maintained, preventing the ischemic pathology. As discussed above, resolution occurs in greater than 60% of cases with observation alone.5,27,32
CONCLUSION
Priapism is a disease entity which emergency physicians will certainly encounter during their careers. With increased usage of pharmacologic and therapeutic agents and interventions, it is likely that it will be more frequently encountered in the future. Ischemic priapism is a true medical emergency, as it carries with it risks of permanent erectile dysfunction and structural damage to the penis. Timely management of veno-occlusive or low-flow priapism therefore is of paramount importance.
REFERENCES
- Roghmann F, Becker A, Sammon JD, et al. Incidence of priapism in emergency departments in the United States. J Urol 2013;190:1275-1280.
- Kulmala RV, Lehtonen TA, Tammela TL. Priapism, its incidence and seasonal distribution in Finland. Scand J Urol Nephrol 1995;29:93.
- Eland IA, van der Lei J, Stricker BH, et al. Incidence of priapism in the general population. Urology 2001;57:970-972.
- El-Bahnasawy MS, Dawood A, Farouk A. Low-flow priapism: Risk factors for erectile dysfunction. BJU Int 2002;89:285.
- Montague DK, Jarow J, Broderick GA, et al. American Urological Association guideline in the management of priapism. J Urol 2003;170:1318-1324.
- Cherian J, Rao AR, Kapasi F, et al. Medical and surgical management of priapism. Postgrad Med J 2006;82:89-94.
- Chrouser KJ, Ajiboye OB, Oyetunji TA, Chang DC. Priapism in the United States: The changing role of sickle cell disease. Am J Surg 2011;201:468-474.
- Earle CM, Stuckey BG, Ching HL, et al. The incidence and management of priapism in Western Australia: A 16 year audit. Int J Impot Res 2003;15:272.
- Fowler JE Jr, Koshy M, Strub M, et al. Priapism associated with the sickle cell hemoglobinopathies: Prevalence, natural history and sequelae. J Urol 1991;145:65-68.
- Adeyoju AB, Olujohungbe ABK, Morris J, et al. Priapism in sickle-cell disease: Incidence, risk factors and complications — an international multicenter study. BJU Int 2002;90:898-902.
- Donaldson JF, Rees RW, Steinbrecher HA. Priapism in children: A comprehensive review and clinical guideline. J Pediatr Urol 2014;10:11-25.
- Salonia A, Eardley I, Giuliano F, Hatzichristou D, Moncada I, Vardi Y, Wespes E, Hatzimouratidis K; European Association of Urology. European Association of Urology guidelines on priapism. Eur Urol 2014;65:480-489.
- Burnett AL, Bivalacqua TJ. Priapism: Current principles and practice. Urol Clin N Am 2007;34:631-642.
- Pryor JP, Hehir M. The management of priapism. Br J Urol 1982;54:751-754.
- Lue TF. Male sexual dysfunction. In: McAninch JW, Lue TF, editors. Smith & Tanagho’s General Urology. 18th ed. The McGraw Hill Companies, Inc. 2013:596-616.
- Kwok B, Varol C. Priapism and penile gangrene due to thrombotic thrombocytopenic purpura. Urology 2010;75(1):71-72.
- Dean RC, Lue TF. Physiology of penile erection and pathophysiology of erectile dysfunction. Urol Clin North Am 2005;32:379-395.
- Champion HC, Bivalacqua TJ, Takimoto E, et al. Phosphodiesterase-5A dysregulation in penile erectile tissue is a mechanism of priapism. Proc Natl Acad Sci USA 2005;102:1661-1666.
- Burnett AL. Nitric oxide in the penis — science and therapeutic implications from erectile dysfunction to priapism. J Sex Med 2006;3:578-582.
- Broderick GA, Gordon D, Hypolite J, et al. Anoxia and corporal smooth muscle dysfunction: A model for ischemic priapism. J Urol 1994;151:259-262.
- Mockford K, Weston M, Subramaniam R. Management of high-flow priapism in paediatric patients: A case report and review of the literature. J Paediatr Urol 2007;3:404-412.
- Halls JE, Patel DV, Walkden M, Patel U. Priapism: Pathophysiology and the role of the radiologist. Br J Radiol 2012;85:S79-S85.
- Bertolotto M, Quaia E, Mucelli FP, et al. Color Doppler imaging of posttraumatic priapism before and after selective embolization. Radiographics 2003;23:495-503.
- Bastuba MD, Saenz de Tejada I, Dinlenc CZ, et al. Arterial priapism: Diagnosis, treatment and long-term follow-up. J Urol 1994;151:1231-1237.
- Levey HR, Segal RL, Bivalacqua TJ. Management of priapism: An update for clinicians. Ther Adv Urol 2014;6:230-244.
- Bennett N, Mulhall J. Sickle cell disease status and outcomes of African-American men presenting with priapism. J Sex Med 2008;5:1244-1250.
- Song PH, Moon KH. Priapism: Current updates in clinical management. Korean J Urol 2012;54:816.
- Broderick GA, Kadioglu A, Bivalacqua TJ, Ghanem H, Nehra A, Shamloul R. Priapism: Pathogenesis, epidemiology, and management. J Sex Med 2010;7:476–500.
- Priyadarshi S. Oral terbutaline in the management of pharmacologically induced prolonged erection. Int J Impot Res 2004;16(5):424-426.
- Roberts JR, Hedges JR, Chanmugam AS. Clinical Procedures in Emergency Medicine. Philadelphia, PA: W.B. Saunders, 2004.
- Tay YK, Spernat D, Rzetelski-West K, Appu S, Love C. Acute management of priapism in men. BJU Int 2012;109:15-21.
- Bassett J, Rajfer J. Diagnostic and therapeutic options for the management of ischemic and nonischemic priapism. Rev Urol 2010;12:56-63.
- Berger R, Billups K, Brock G, Broderick GA, et al. Report of the American Foundation for Urologic Disease (AFUD) Thought Leader Panel for evaluation and treatment of priapism. Int J Impot Res 2001;13:S39-S43.
- Ravindran M. Cauda equina compression presenting as spontaneous priapism. J Neurol Neurosurg Psychiatry 1979;42:280.
- Chrouser KJ, Ajiboye OB, Oyetunji TA, Chang DC. Priapism in the United States: The changing role of sickle cell disease. Am J Surg 2011;201:468-474.
- Alsulihem AA, Rabah DM. Priapism associated with pregabalin. Urology Ann 2014;6(4):366-368.
- Jesus LE, Dekermacher S. Priapism in children: Review of pathophysiology and treatment. J Pediatr (Rio J) 2009;85:194-200.
Priapism is one of those issues that emergency physicians must know how to recognize and treat. Several things have changed in the care of the patient with priapism recently.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
