Fibromyalgia and Other Soft Tissue Pain Syndromes
Nasim Daoud, MD, Assistant Professor of Medicine, Division of Rheumatology, Loma Linda University, Loma Linda, CA
Keith K. Colburn, MD, Professor of Medicine, Division of Rheumatology. Loma Linda University and Loma Linda VA Medical Center, Loma Linda, CA
Peer Reviewer
Glen D. Solomon, MD, Professor and Chair, Department of Internal Medicine, Boonshoft School of Medicine, Wright State University, Dayton, OH
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, Dr. Daoud, Dr. Colburn, and Dr. Solomon report no financial relationships relevant to this field of study.
Executive Summary
- Fibromyalgia is a common musculoskeletal condition that is often seen in primary care practices, with nearly a 8:1 female predominance.
- It is characterized by diffuse, widespread pain with multiple tender points lasting at least 3 months.
- Management of fibromyalgia includes good sleep hygiene, stretching, aerobic exercise, and targeted pharmacologic therapies.
- A careful history and physical examination is usually sufficient to differentiate fibromyalgia from a connective tissue disease, but if clinical uncertainty exists, specific serologic tests should be obtained.
- Many soft tissue manifestations typically present in conjunction with fibromyalgia and complicate the treatment scenarios.
A common cause of chronic musculoskeletal pain lasting for at least 3 months, fibromyalgia syndrome (FMS) is characterized by diffuse, widespread pain and multiple tender points. It is often accompanied by significant fatigue, unrefreshed interrupted sleep, and cognitive dysfunction, along with many other somatic symptoms. The etiology of the pain is not known.
FMS is diagnosed more frequently in women. For example, a study in the United States found a ratio of 6.8:1 female to male predominance, while the ratio varied in other countries. Patients with FMS, like other functional somatic syndromes, appear healthy. There are no obvious abnormalities in laboratory or radiologic studies. FMS may be diagnosed using the American College of Rheumatology (ACR) 2010 or 1990 criteria.1,2 However, eligibility for clinical care does not require a definite diagnosis.
FMS is sometimes divided into primary, secondary, and secondary-concomitant fibromyalgia. The term primary fibromyalgia is most often used when there is no other condition with similar findings that could explain the symptoms attributed to fibromyalgia. Secondary FMS refers to FMS symptoms caused by another condition like hypothyroidism; secondary-concomitant FMS is FMS associated with another disease, such as systemic lupus erythematosus (SLE), but not necessarily caused by that disease. FMS is one of a group of soft tissue pain disorders that affect muscles and soft tissues, such as bursa, tendons, and ligaments.3,4
Diagnosis of FMS
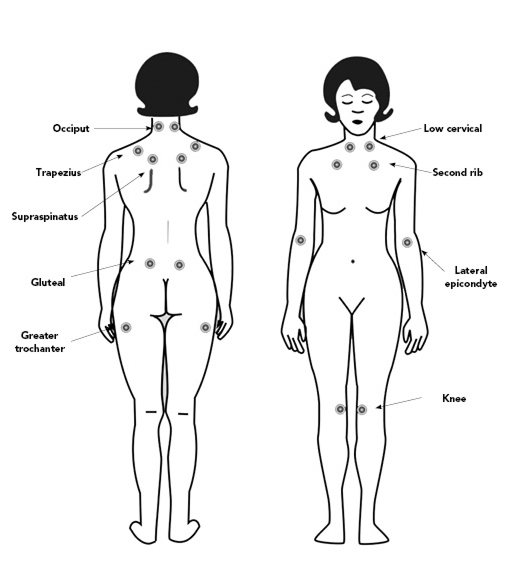
The manifestations of FMS include widespread musculoskeletal pain involving both sides of the body and above and below the waist (see Figure 1). However, the pain initially may be localized in the neck and shoulders. Upon asking FMS patients about the location of the pain, most will answer “I hurt all over,” or “It feels as if I always have the flu.”5,6 Sometimes it is helpful to ask the patient, “Do you have anywhere on your body that doesn’t hurt?”
Figure 1. Tender Point Locations of Fibromyalgia |
|
Adapted from: National Institutes of Arthritis and Musculoskeletal and Skin Diseases |
Other symptoms associated with FMS include fatigue, poor and interrupted sleep, headaches, sicca symptoms, irritable bowel syndrome, interstitial cystitis pain, cognitive problems, depression, and anxiety. Fatigue is especially notable when arising from sleep, but is also marked in the mid-afternoon. The pain and fatigue are aggravated by minor activities and prolonged inactivity. Patients with FMS feel tired and stiff when they get up in the morning and may remark that no matter how many hours they slept, it was not enough. Cognitive disturbances typically include problems with attention, short-term memory, and difficulty performing tasks that require rapid thought changes. In one study, depression and/or anxiety were present in 30-50% of patients at the time of diagnosis.7,8
Differential Diagnosis
Several rheumatic diseases can mimic fibromyalgia, and a clinician would not want to miss these diagnoses because of their potential long-term sequelae, such as progressive joint or other organ damage or a life-threatening disease if untreated. Rather than worrying about every disease that can cause musculoskeletal pain, the clinician should focus on the characteristic features of FMS (see Table 1) that include pain above and below the waist, which is bilateral and axial lasting at least 3 months, and somatic complaints including fatigue, sleep, mood, and cognitive disturbances.
Table 1. American College of Rheumatology 2010 Preliminary Diagnostic Criteria for Fibromyalgia |
|
1. Widespread pain. 2. Symptoms have been present at a similar level for at least 3 months. 3. The patient does not have a disorder that would otherwise explain the pain. The ACR 2010 Preliminary Diagnostic Criteria for Fibromyalgia does not include the nine pairs of tender points (see Figure 1) used in the 1990 ACR Classification Criteria, which are at locations that most primary care clinicians and specialists routinely evaluate in patients with soft tissue complaints. These locations include the upper mid-trapezius muscle, the lateral epicondyle (the so-called tennis elbow location), the second costochondral junction, and the greater trochanter (trochanteric bursa of the hip). The amount of pressure should equal 4 kg/cm², which is enough to whiten the nail bed of the examiner's thumb. |
Resources for Patient Education on Fibromyalgia |
|
|
National Center for Complementary and Integrative Health National Institutes of Health |
|
|
American College of Rheumatology |
|
|
Advocates for Fibromyalgia Funding, Treatment, Education, and Research |
|
|
Fibromyalgia Network |
|
|
National Fibromyalgia Association |
|
|
National Fibromyalgia Partnership, Inc. |
|
|
Arthritis Foundation |
|
|
National Institue of Arthritis and Musculoskeletal and Skin Diseases |
|
Conditions that should be considered in the differential diagnosis of FMS include other disorders that may mimic FMS or that may occur as comorbid conditions, as well as a collection of common disorders that often overlap with FMS.10,11 These illnesses, which include myofascial pain, chronic fatigue syndrome, irritable bowel syndrome, pelvic and bladder pain disorders, and temporomandibular pain, are common and can present challenges in developing a differential diagnosis.11 Like FMS, each of these conditions is very common and controversial, since there are few objective abnormalities found on the physical, laboratory, or radiologic examinations. As a result, FMS and these overlapping disorders are sometimes termed “functional somatic syndromes.”
Rheumatic diseases, such as rheumatoid arthritis, Sjögren’s syndrome, and SLE, may present with generalized arthralgias, myalgias, and fatigue, and like FMS most often affect younger women. However, characteristic features of rheumatoid arthritis, such as multiple joint swelling, or of SLE, with a facial rash and multisystem organ inflammation, do not occur in primary FMS. About 30-50% or more of patients with these autoimmune diseases also have FMS (secondary-concomitant FMS), often confusing both patients and doctors.
Ankylosing spondylitis and other inflammatory back conditions may present with axial skeletal pain and stiffness similar to that of FMS and may be misdiagnosed as FMS.
Polymyalgia rheumatica may mimic FMS. However, patients with polymyalgia rheumatica tend to be older at onset and to present more with generalized stiffness than with severe, widespread pain. They also have an elevated erythrocyte sedimentation rate, whereas it is usually normal in patients with FMS.
Myositis and other myopathies cause muscle weakness and fatigue but are not usually associated with diffuse pain, which distinguishes FMS from inflammatory myositis and the metabolic myopathies.
A serum thyroid-stimulating hormone should be ordered in all patients with suspected FMS, since patients with hypothyroidism often complain of generalized aches, fatigue, and interrupted sleep and are classified as having secondary FMS.
Serology Markers in FMS
A careful history and physical examination, rather than screening serologic blood tests, should be sufficient to differentiate FMS from a connective tissue disease. Unless there is strong clinical suspicion of a systemic rheumatic disorder, routine ordering of serologic tests is not recommended since these tests, such as antinuclear antibody (ANA) and rheumatoid factor (RF), may be positive in healthy individuals and in many other diseases. For example, in a prospective evaluation of 118 consecutive patients with FMS for prevalence of Raynaud’s phenomenon, sicca symptoms, ANA, and low complement, the authors found approximately 10-15% of patients with FMS have a positive ANA test; however, 5-10% of healthy women also have a positive ANA and at least a low titer of 0.5 Therefore, the predictive value of a positive ANA is poor in patients who have no signs or symptoms of SLE.
If there is clinical confusion regarding the differential diagnosis of FMS with other systemic connective tissue diseases, more specific serologic tests should be obtained, such as an anti-DNA antibody, anti-Smith antibody test for SLE, or anti-CCP antibody for RA. Thirty-five patients (30%) in the above study had a history of Raynaud’s phenomenon. Primary care physicians should attempt to rule out other rheumatic diseases in patients with Raynaud’s.
Fibromyalgia Management
Fibromyalgia management is directed at using a stepwise approach to reduce the major symptoms, including chronic widespread pain, fatigue, insomnia, and cognitive dysfunction. The first step is educating the patient about the disease, treatment approaches, good sleep hygiene, and the importance of treating comorbidities that may contribute to symptoms, including mood or sleep disorders. In some reports education may have a modest effect on fibromyalgia symptoms, such as fatigue, anxiety, and depression, but will have no effect on pain.13,14
The next step consists of an exercise program, featuring aerobic conditioning, stretching, and gentle strengthening. In a Cochrane collaboration analysis of 34 studies that included exercise, the authors noted that there is moderately good evidence that aerobic-only exercise at recommended intensity levels has positive effects on global well-being, physical function, and possibly pain. The researchers concluded that “supervised aerobic exercise training has beneficial effects on physical capacity and fibromyalgia symptoms.”15
Conservative treatment of FMS would insist on the following: If non-pharmacologic modalities do not result in improvement, drug monotherapy is recommended. Combination drug therapy in most patients unresponsive to monotherapy would be the next step in management. (See Table 2.)
Table 2. Medications Used in Treating Fibromyalgia |
|
|
Sleep Aids |
|
|
Drug |
Dose |
|
Trazadone |
50-150 mg, 2 hours before bedtime |
|
Amitriptyline |
25-200 mg, 2 hours before bedtime |
|
Zolpidem |
2.5-10 mg hs (controversial with some investgators) |
|
Eszopiclone |
1-3 mg hs |
|
Benzodiazepams |
not recommended for physiological addiction reasons |
|
Pain Medications |
|
|
Drug |
Dose |
|
Gabapentin |
up to 3600 mg/day, in divided doses |
|
Pregablin |
up to 450-600 mg/day, in divided doses |
|
Tramadol |
50 -400 mg daily, in divided doses |
|
Pramipexol |
0.25 -4.5mg hs |
|
Ropinirol |
0.5-24 mg hs |
|
Opiates should not be used chronically since they end up lowering the pain threshold and require increasing doses and advancement to stronger drugs, and patients end up hurting worse than ever. |
|
|
Pain/Antidepression Medications |
|
|
Drug |
Dose |
|
Duloxetine |
30-60 mg/day |
|
Milnacipran |
50-100 mg bid |
|
Venlafaxine |
37.5-150 mg/day |
|
Other antidepressants |
|
|
Muscle Relaxants |
|
|
|
|
Antifatigue Drugs |
|
|
|
|
Aerobic Exercise (most important part of treatment) |
|
|
|
Some primary care physicians may be more aggressive in treating FMS patients and initially would include education, aerobic exercise, and medicinal treatment of each of the major symptoms of this syndrome. When polytherapy is considered, therapeutic decisions must be based on data from monotherapy trials and a sound knowledge of the pharmacological profile of each drug. Several well-designed clinical trials have explored specific drug combinations selected on the basis of potential additive or synergistic effects.16
If the patient fails to respond to above treatment, the next step is referral to a physical medicine and rehabilitation evaluation and treatment program. Since this may cause more pain, referral for psychological intervention for pain management, including cognitive behavioral therapy, is very useful.
There is evidence for efficacy of strength training,18 hypnosis,19 and biofeedback19,20,21 in treating FMS. Several well-controlled studies evaluated spa and bath therapies that have been used for centuries to ease pain in the management of FMS.22 Assessment and care in a specialized multidisciplinary program, particularly for patients with disease refractory to other interventions or on chronic opioids, is suggested.
Tender and Trigger Point Injections
FMS pain is widespread and does not seem to be restricted to tender points. It frequently comprises multiple areas of deep tissue pain (trigger points) with much larger adjacent areas of referred pain. Locating those trigger points by palpation of muscles may exhibit a palpable taut band or cord of tense muscle fibers < 4 mm in diameter. Analgesia in areas of extensive nociceptive input has been found to provide long-lasting local as well as general pain relief. Thus, interventions aimed at reducing local FMS pain seem to be effective but need to focus less on tender points and more on trigger points and other body areas of heightened pain and inflammation. Uncontrolled studies suggest that dry needling or soft-tissue injections with lidocaine or saline are equally effective. Local injections in muscular areas of pain are also commonly employed by rheumatologists, who reported that patients “like injections,” but also that the rheumatologists did not know what else to do.23
Soft Tissue Manifestations of FMS
FMS patients have concomitant conditions, such as tendinitis, which lead to shoulder, arm, and leg pain, but are likely symptoms of FMS.24 In one study about the prevalence of soft tissue pain, rotator cuff tendinitis, pes anserinus bursitis, and lateral epicondylitis were significantly more common in patients with primary and secondary FMS than in control subjects. Although soft tissue rheumatism is non-articular pain, patients often attribute their symptoms to closest joints.
Tendinitis is a local pain and dysfunction from inflammation and degeneration. It typically involves the area of the tendon near its point of insertion into the bone or its point of muscular origin.
Enthesitis is inflammation at the site of insertion of ligaments, tendons, joint capsules, and fascia to bone. Common sites for enthesitis are the insertion of the plantar fascia and the Achilles tendon region.
Fasciitis is proliferation and degeneration of fascia. The most common sites include Dupuytren’s palmar contracture and plantar fasciitis.
Bursitis is inflammation of small fluid-filled pads between areas of potential friction in soft tissues. Its purpose is to reduce friction and act as a “shock absorber” and cushion between the bones and adjacent tendons and muscles, protecting the soft tissues from underlying bony prominences. The most common sites of bursitis are the pes anserine bursa (medial aspect of the knee), trochanteric bursa (around the hip area), and the subacromial bursa (in the shoulder area). The following section will discuss the most common soft tissue disorders, including those in FMS regardless of primary or secondary associations, in the shoulders, elbows, hands, hips, knees, and feet.
Disorders of the Shoulder
Rotator cuff disease is responsible for a high proportion of patients with shoulder pain presenting to general practice and causes work absenteeism and claims for sickness benefits. Rotator cuff disease often can be managed in primary health care clinics, although some cases may require help from physical medicine or orthopedics. Careful history and examination followed by pertinent investigation are essential to establish the correct diagnosis. The main aim of treatment is to improve symptoms and restore function of the affected shoulder.
Subacromial bursitis: The subacromial bursa (also known as the sub-deltoid bursa) is located superior to the glenohumeral joint and has as its floor the tendon of the supraspinatus muscle and its roof the acromion. Patients typically complain about pain at rest, exacerbated by use, and refer to the insertion of the deltoid muscle 10 cm down the outer arm. Pain may be severe and can often interrupt sleep.
Rotator Cuff Tendinopathy: Rotator cuff tendinitis almost always represents chronic injury to the supraspinatus (functionally responsible for abduction) and/or infraspinatus (functionally responsible for external rotation) tendons. It develops as a consequence of repetitive activity, generally at or above shoulder height, which leads to microvascular injury and tendon degeneration. Patients describe the shoulder pain as exacerbated by reaching, pushing, pulling, lifting, positioning the arm above the shoulder, or lying on the affected side.
Rotator Cuff Tendon Tears: Tears occur primarily in the supraspinatus tendon, but also happen to the infraspinatus tendon. Those tears are most often in patients older than 40 years of age and who have a history of impingement syndrome.31 This condition occurs as a result of progressive tendon degeneration, traumatic injury, or a combination of these factors. Patients complain of shoulder weakness, pain over the anterolateral shoulder or sometimes the upper back, and a popping or catching sensation when the shoulder is moved. Shoulder function will be preserved if the tear is small in size. If the tear involves both the supraspinatus and infraspinatus tendons, the patient will complain of weakness and loss of function of reaching, pushing, pulling, lifting, and positioning the arm above the shoulder level. A frozen shoulder, or adhesive capsulitis, may develop as a consequence of persistent immobility. The differentiation between a subacromial bursitis, rotator cuff tendinitis, and rotator cuff tear usually can be made on the basis of history, physical exam, and imaging studies.
Diagnosis and management: The diagnosis of the type of shoulder derangement can be made by using a combination of clinical tests. There is no one test that can diagnose rotator cuff damage. After 6 weeks of symptoms, if the clinical history or shoulder examination suggests the possibility of a complete tear of the rotator cuff, ultrasound or magnetic resonance imaging (MRI) will help determine the extent of the lesion. Many clinicians consider musculoskeletal ultrasound to be the gold standard for the initial evaluation of rotator cuff disease. Multiple studies demonstrate ultrasound’s high sensitivity in diagnosing rotator cuff disease, particularly complete tendon tears.33,34 Initial treatment consists of ice, rest, and non-steroidal anti-inflammatory medication for acute pain if necessary. However, strict immobilization is recommended. There is some controversy whether patients should be treated non-operatively, since there is no convincing evidence that surgical treatment is more effective than conservative management, especially in older patients. There is no indication for the surgical treatment of asymptomatic rotator cuff tears.32
Conservative management using qualified physical or occupational therapists is most helpful for developing specific programs to aid in the recovery process. Exercise therapy should be specific and of low intensity but high frequency, combining training, attention to relaxation and posture, and treatment of myofascial trigger points, including stretching. Rehabilitation in a specialized unit can be considered in chronic, treatment-resistant cases. There is little evidence to support the therapeutic use of ultrasound. However, the theory behind ultrasound as a treatment is that it stimulates tendon healing via collagen production.35
Laser therapy in the treatment of rotator cuff tendinopathy is without scientific merit.36 A few small, randomized trials found a short-term benefit from glucocorticoid injections.37,38,39 Subacromial injections with corticosteroids are indicated for persistent or recurrent pain, and symptomatic relief gained from such injections may improve a patient’s effort and compliance with physical therapy.40,41 Corticosteroid injections can be performed blind, landmark-guided or with ultrasound image guidance. A meta-analysis found statistically significant differences in pain and abduction between landmark-guided and ultrasound image guidance steroid injections for adults with shoulder pathology. However, these differences are small and may not represent clinically useful differences.42 Indications for referral for orthopedic evaluation include treatment failures after 3 months of conservative management or suspected diagnosis of a rotator cuff tear, labrum tear, or adhesive capsulitis.
Bicipital tendinitis: Bicipital tendinitis, or biceps tendinitis, is an inflammatory process of the long head of the biceps tendon. This is a common cause of shoulder pain. The tendon originates intra-articularly at the superior glenoid tubercle, where it is a part of the glenoid labrum. It then extends distally to the musculotendinous junction with the long head biceps muscle. The tendon is exposed on the anterior shoulder as it passes through the humeral bicipital groove and inserts onto the superior aspect of the labrum of the glenohumeral joint. Patients complain of achy anterior shoulder pain, exacerbated by overhead activity, lifting heavy objects, and pushing or pulling with the shoulder elevated. The pain is often referred to the posterior or lateral parts of the shoulder, where if injected will not help the shoulder pain. The treatment of bicipital tendonitis is most efficiently accomplished with a corticosteroid injection around the tender point on the tendon, but not into the tendon itself.
Disorders of the Elbow
The elbow is the articulation between three bones: the humerus, ulna, and radius. Epicondylitis is the most common cause of elbow pain. Lateral epicondylitis is the most common location of pain and tenderness in patients with FMS.24 Olecranon bursitis can also be painful, but happens less frequently. Injury to the pronator teres and flexor carpi radialis muscles, which originate at the medial epicondyle, causes medial epicondylitis (golf elbow). In lateral epicondyle injuries (tennis elbow), the carpi radialis brevis muscle is most commonly involved, but may include the extensor carpi radialis longus extensor digitorum and extensor carpi ulnaris.
Lateral epicondylitis (tennis elbow): Patients with tennis elbow typically report an insidious onset associated with a history of overuse without specific trauma. Patients usually complain of mild to severe pain over the lateral elbow that may radiate down the posterior aspect of the forearm. The pain occurs with simple activities, like picking up or holding very light objects, and worsens with activity and improves with rest. Repetitive motions in which the wrist frequently deviates from a neutral position can cause epicondylitis.
Microscopic evaluation of the tendons does not show signs of inflammation, but rather angiofibroblastic degeneration and collagen disarray. Light microscopy shows both an excess of fibroblasts and blood vessels that are consistent with angiogenesis.55,56
Lateral epicondylitis is diagnosed clinically by the following physical findings on exam: localized tenderness over the lateral epicondyle and proximal wrist extensor muscle mass. Pain is increased with resisted wrist extension with the elbow in full extension. Passive terminal wrist flexion with the elbow in full extension increases pain with resisted supination and hand shaking. Although there are multiple treatment options for management of the lateral epicondylitis, no one single treatment is completely effective, so it is preferable to use a stepwise approach, starting with initial treatment of activity modification, counter force bracing, nonsteroidal anti-inflammatory drugs (NSAIDs) if not contraindicated, and physical therapy. If there is no improvement, glucocorticoid injections or injections with lidocaine can be effective. Multiple studies have shown that lidocaine alone is at least as good as corticosteroids injected near the lateral epicondyle.57 Alternative therapies to reduce pain include extracorporeal shock wave therapy, ultrasound-guided percutaneous radiofrequency thermal lesioning, laser therapy, platelet-rich plasma, botulinum toxin, or applying topical nitrates.58 If these treatments fail, referral to orthopedics for possible surgical relief is recommended.
Disorders of the Hand
The most common soft tissue disorder in the hands is a Dupuytren’s contracture.24 This common, fibroproliferative disorder is characterized by slowly progressive thickening and shortening of the palmar fascia, which appears as a nodule in the palm of the hand. The most affected locations are flexor tendons of the third, fourth, and fifth digits. Ultrasound can be a useful tool to evaluate the thickness and nodules and to guide the intralesional glucocorticoid injection. The intralesional injection of nodules of Dupuytren’s disease with triamcinolone acetonide may modify the progression of the disease;59 however, corticosteroids are associated with a high risk of complications; including tendon ruptures, fat atrophy, and skin discoloration.60 Physical therapy can be used in early stages, but it is not clear whether passive stretching offers any significant clinical benefit or harm. There is no evidence to support that massage, splinting, or exercise will prevent the progression of the contracture.
Multiple studies showed the effectiveness of injection of Clostridium histolyticum (Xiaflex) at a dose of 0.58 mg per injection for the treatment of Dupuytren’s contracture in a single digit during a 30-day treatment cycle. The injected collagenase extracted from C. histolyticum weakens and dissolves the Dupuytren cord. If the above treatments fail to improve flexibility of the fingers, patients should be referred to the hand surgeon.
Disorders of the Hip
Trochanteric bursitis: Trochanteric bursitis is a common disorder, especially in patients with FMS.24 Trochanteric bursitis is characterized by painful inflammation of the bursa located between the midpoint of the trochanteric process of the femur and the gluteus medius tendon.62 Patients typically complain of lateral hip pain over the outer thigh or of difficulty with walking. The pain may radiate down the lateral aspect of the thigh. The most classic physical finding is point tenderness over the greater trochanter, reproducing the presenting symptom.63 The patient often complains of difficulty sleeping on the affected side while rubbing the thigh when describing the pain. There is no need for laboratory or radiologic workup to establish the diagnosis. A diagnostic injection of a local anesthetic (5 cc of 1-2%) of lidocaine into the trochanteric bursa may be helpful. Treatment of trochanteric bursitis includes rest, avoiding sleeping on the affected side, applying ice topically, injection of corticosteroids and/or local anesthetics, administration of NSAIDs, and application of topical, sustained-release local anesthetic patches. A home exercise program that emphasizes stretching of the iliotibial band and other hip flexors might be helpful.64 Extracorporeal shock wave therapy has helped in patients who failed the above therapy.65 Surgical intervention may be useful in refractory cases.
Soft Tissue Disorders of the Knee
Bursitis of the knee is common in practice. The knee has four major bursae: suprapatellar, prepatellar, infrapatellar, and pes anserine. A common soft tissue tender point of the knee in FMS is over the pes anserine bursa.24
Pes anserine bursitis: The pes anserine bursa is located on proximomedial aspect of the tibia between the superficial medial collateral ligament and hamstring tendons: the sartorius, gracilis, and semitendinosus. This bursa serves as a lubricating sac between these tendons. Pes anserine bursitis may occur with arising from a seated position or with ascending, but usually not while walking on level surfaces. The pain is often localized to a well-defined area of the inner knee.66,67 Patients often point to the area with one finger when describing the pain and the tenderness.
If the tenderness extends from the anserine bursal area to the joint line, it is more likely that the medial collateral ligament is injured or irritated. Pes anserine bursitis is generally a self-limiting condition. Patients are treated successfully with conservative measures, including topical icing every 4-6 hours; stretching and strengthening the adductor, abductor, and quadriceps groups; stretching the hamstrings; and NSAIDs. Intrabursal injection of local anesthetics, corticosteroids, or both constitutes a second line of treatment. Surgical therapy is indicated only in very rare cases.
REFERENCES
- Wolfe F, Clauw D, Fitzcharles MA, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken) 2010;62:600-610.
- American College of Rheumatology Criteria for the Classification of Fibromyalgia. 1990. Available at: https://www.rheumatology.org/Portals/0/Files/1990_Criteria_for_Classification_Fibro.pdf. Accessed Sept. 1, 2015.
- Goldenberg DL. Fibromyalgia syndrome. An emerging but controversial condition. JAMA 1987;257:2782-2787.
- Goldenberg DL. Fibromyalgia syndrome a decade later: What have we learned? Arch Intern Med 1999;159:777-785.
- Goldenberg DL. Diagnosis and differential diagnosis of fibromyalgia. Am J Med 2009;122(12 Suppl):S14-21.
- Björkegren K, Wallander MA, Johansson S, Svärdsudd K. General symptom reporting in female fibromyalgia patients and referents: A population-based case-referent study. BMC Public Health 2009;9:402.
- Glass JM. Cognitive dysfunction in fibromyalgia and chronic fatigue syndrome: New trends and future directions. Curr Rheumatol Rep 2006; 8:425-429.
- Marcus DA, Bernstein C, Rudy TE. Fibromyalgia and headache: An epidemiological study supporting migraine as part of the fibromyalgia syndrome. Clin Rheumatol 2005;2:595-601.
- Colburn KK. Tendonitis, Bursitis, Myofascial Pain and Fibromyalgia. In: Conn’s Current Therapy. Philadelphia: Elsevier Saunders. 2015; 597-600.
- Hwang E, Barkhuizen A. Update on rheumatologic mimics of fibromyalgia. Curr Pain Headache Rep 2006;10:327.
- Aaron LA, Buchwald D. Chronic diffuse musculoskeletal pain, fibromyalgia and co-morbid unexplained clinical conditions. Best Pract Res Clin Rheumatol 2003;17:563-574.
- Wolfe F, Ross K, Anderson J, et al. Prevalence of fibromyalgia: A survey in five European countries. St Louis: Elsevier; 2009.
- Cedraschi C, Desmeules J, Rapiti E, et al. Fibromyalgia: A randomised, controlled trial of a treatment programme based on self-management. Ann Rheum Dis 2004;63:290-296.
- Alamo MM, Moral RR, Perula de Torres LA, et al. Evaluation of a patient-centered approach in generalized musculoskeletal chronic pain/fibromyalgia patients in primary care. Patient Educ Couns 2002;48:23-31.
- Busch A, Barber K, Overend T, et al. Exercise for treating fibromyalgia syndrome. Cochrane Database Syst Rev 2007;CD003786.
- Calandre EP, Rico-Villademoros F, Rodríguez-López CM. Monotherapy or combination therapy for fibromyalgia treatment? Curr Rheumatol Rep 2012;14:568-575.
- Carville SF, Arendt-Nielsen S, Bliddal H, et al. EULAR evidence-based recommendations for the management of fibromyalgia syndrome. Ann Rheum Dis 2008;67:536-541.
- Haanen HC, Hoenderdos HT, van Romunde LK, et al. Controlled trial of hypnotherapy in the treatment of refractory fibromyalgia. J Rheumatol 1991;18:72-75.
- Drexler AR, Mur EJ, Gunther VC, et al. Efficacy of an EMG-biofeedback therapy in fibromyalgia patients. A comparative study of patients with and without abnormality in (MMPI) psychological scales. Clin Exp Rheumatol 2002;20:677-682.
- van Santen M, Bolwijn P, Verstappen F, et al. A randomized clinical trial comparing fitness and biofeedback training vs basic treatment in patients with fibromyalgia. J Rheumatol 2002;29:575-581.
- Ferraccioli G, Ghirelli L, Scita F, et al. EMG-biofeedback training in fibromyalgia syndrome. J Rheumatol 1987;14:820-825.
- McVeigh JG, McGaughey H, Hall M, et al. The effectiveness of hydrotherapy in the management of fibromyalgia syndrome: A systematic review. Rheumatol Int 2008;29:119-130.
- Hassett A, Gevirtz R. Nonpharmacologic treatment for fibromyalgia: Patient education, cognitive-behavioral therapy, relaxation techniques, and complementary and alternative medicine. Rheum Dis Clin North Am 2009;35:393-407.
- Genc H, Saracoglu M, Duyur B, Erdem HR. The role of tendinitis in fibromyalgia syndrome. Yonsei Med J 2003;44:619-622.
- Kushner I. Overview of soft tissue rheumatic disorders. UpToDate July 2015.
- Bruyn GAW. Musculoskeletal ultrasonography: Clinical applications. UpToDate June 2015.
- Reveille JD. Soft-tissue rheumatism: Diagnosis and treatment. Am J Med 1997;102(1A):23S-29S.
- Staud R. Division of Rheumatology and Clinical Immunology, University of Florida.
- Moraes VY1, Lenza M, Tamaoki MJ, et al. Platelet-rich therapies for musculoskeletal soft tissue injuries. Cochrane Database Syst Rev 2014;4:CD010071. doi: 10.1002/14651858.CD010071.pub3.
- Lenza M, Buchbinder R, Takwoingi Y, et al. Magnetic resonance imaging, magnetic resonance arthrography and ultrasonography for assessing rotator cuff tears in people with shoulder pain for whom surgery is being considered. Cochrane Database Syst Rev 2013;9:CD009020. doi:10.1002/14651858.CD009020.pub.
- Tempelhof S, Rupp S, Seil R. Age-related prevalence of rotator cuff tears in asymptomatic shoulders. J Shoulder Elbow Surg 1999;8:296-299.
- Diercks R, Bron C, Dorrestijn O, et al. Guideline for diagnosis and treatment of subacromial pain syndrome: A multidisciplinary review by the Dutch Orthopaedic Association. Acta Orthop 2014;85:314-322.
- Lew HL, Chen CP, Wang TG, Chew KT. Introduction to musculoskeletal diagnostic ultrasound: Examination of the upper limb. Am J Phys Med Rehabil 2007;86:310-321.
- Teefey SA, Rubin DA, Middleton WD, et al. Detection and quantification of rotator cuff tears. Comparison of ultrasonographic, magnetic resonance imaging, and arthroscopic findings in seventy-one consecutive cases. J Bone Joint Surg Am 2004;86-A:708-716.
- Rees JD, Wilson AM, Wolman RL. Current concepts in the management of tendon disorders. Rheumatology (Oxford) 2006;45:508-521.
- Jonsson P, Wahlström P, Ohberg L, Alfredson H. Eccentric training in chronic painful impingement syndrome of the shoulder: Results of a pilot study. Knee Surg Sports Traumatol Arthrosc 2006;14:76-81.
- Akgün K, Birtane M, Akarirmak U. Is local subacromial corticosteroid injection beneficial in subacromial impingement syndrome? Clin Rheumatol 2004;23:496-500.
- Karthikeyan S, Kwong HT, Upadhyay PK, et al. A double-blind randomised controlled study comparing subacromial injection of tenoxicam or methylprednisolone in patients with subacromial impingement. J Bone Joint Surg Br 2010;92:77-82.
- Plafki C, Steffen R, Willburger RE, Wittenberg RH. Local anaesthetic injection with and without corticosteroids for subacromial impingement syndrome. Int Orthop 2000;24:40-42.
- Buchbinder R, Green S, Youd JM. Corticosteroid injections for shoulder pain. Cochrane Database Syst Rev 2003;CD004016.
- Hay EM, Thomas E, Paterson SM, et al. A pragmatic randomised controlled trial of local corticosteroid injection and physiotherapy for the treatment of new episodes of unilateral shoulder pain in primary care. Ann Rheum Dis 2003;62:394-399.
- Sage W, Pickup L, Smith TO, et al. The clinical and functional outcomes of ultrasound-guided vs landmark-guided injections for adults with shoulder pathology—a systematic review and meta-analysis. Rheumatology 2013;52:743-751. doi: 10.1093/rheumatology/kes302.
- Patton WC, McCluskey GM 3rd. Biceps tendinitis and subluxation. Clin Sports Med 2001;20:505-529.
- Ahrens PM, Boileau P. The long head of biceps and associated tendinopathy. J Bone Joint Surg Br 2007;89:1001-1009.
- Safran MR, McKaeg DB, Van Camp SP, eds. Biceps tendon injuries. In: Manual of Sports Medicine. Philadelphia: Lippincott Williams & Wilkins; 1998: 347-349.
- Nirschl RP. Elbow tendinosis/tennis elbow. Clin Sports Med 1992;11:851-870.
- Zeisig E, Ohberg L, Alfredson H. Extensor origin vascularity related to pain in patients with tennis elbow. Knee Surg Sports Traumatol Arthrosc 2006;14:659-663.
- Bisset L, Beller E, Jull G, et al. Mobilisation with movement and exercise, corticosteroid injection, or wait and see for tennis elbow: Randomised trial. BMJ 2006;333:939
- Paoloni JA, Appleyard RC, Nelson J, Murrell GA. Topical nitric oxide application in the treatment of chronic extensor tendinosis at the elbow: A randomized, double-blinded, placebo-controlled clinical trial. Am J Sports Med 2003;31:915-920.
- Ketchum LD, Donahue TK. The injection of nodules of Dupuytren’s disease with triamcinolone acetonide. J Hand Surg Am 2000;25:1157-1162.
- Donaldson OW, Pearson D, Reynolds R, Bhatia RK. The association between intraoperative correction of Dupuytren’s disease and residual postoperative contracture. J Hand Surg Eur Vol 2010;35:220-223.
- Namazi H, Abdinejad F. Botulinum Toxin as a novel addition to the antidupuytren armamentarium. Med Hypotheses 2007;68:240-241.
- Brinker MR, Miller MD. The adult hip. In: Fundamentals of Orthopaedics. Philadelphia: WB Saunders; 1999: 269-285.
- Trochanteric bursitis. In: Snider RK, ed. Essentials of Musculoskeletal Care. Rosemont, IL: American Academy of Orthopaedic Surgeons; 1997: 299-303.
- Lustenberger DP, Ng VY, Best TM, Ellis TJ. Efficacy of treatment of trochanteric bursitis: A systematic review. Clin J Sport Med 2011;21:447-453.
- Furia JP, Rompe JD, Maffulli N. Low-energy extracorporeal shock wave therapy as a treatment for greater trochanteric pain syndrome. Am J Sports Med 2009;37:1806-1813.
- Anderson BC. Office Orthopedics for Primary Care: Diagnosis and Treatment. 2nd ed. Philadelphia: WB Saunders; 1999.
- Alvarez-Nemegyei J, Canoso JJ. Evidence-based soft tissue rheumatology IV: Anserine bursitis. J Clin Rheumatol 2004;10:205-206.
MONOGRAPH: Sometimes it's helpful to ask the patient, “Do you have anywhere on your body that doesn’t hurt?”
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.