Shoulder Trauma
August 15, 2016
Reprints
AUTHORS
Moira Davenport, MD, Assistant Residency Director, Allegheny General Hospital, Pittsburgh, PA.
Ryan Freedman, MD, MS, Primary Care Sports Medicine Fellow, Duke University; Emergency Medicine Physician, Division of Emergency Medicine, Duke University, Durham, NC.
PEER REVIEWER
Steven M. Winograd, MD, FACEP, St. Barnabas Hospital, Clinical Assistant Professor, Emergency Medicine, New York College of Osteopathic Medicine, Old Westbury, NY.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Stapczynski (editor) owns stock in Pfizer, Johnson & Johnson, Walgreens Boots Alliance Inc., GlaxoSmithKline, Bristol Myers Squibb, and AxoGen. Dr. Schneider (editor), Ms. Fessler (nurse planner), Dr. Davenport (author), Dr. Freedman (author), Dr. Winograd (peer reviewer), Ms. Mark (executive editor), Ms. Coplin (executive editor), and Mr. Landenberger (editorial and continuing education director) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Use provocative tests to identify labral and rotator cuff tears.
- Posterior shoulder dislocations can be missed on anteroposterior radiographs.
- Carefully check for injuries to periarticular structures with any shoulder dislocation.
- Acromioclavicular joint separations usually can be managed conservatively.
The shoulder is the most mobile joint in the human body. Given its exposed location, the shoulder is a prime target for traumatic injury. In addition, mobility comes with a price: the potential for injuries to the supporting structures, muscles, ligaments, and cartilage. Many acute shoulder injuries involve these periarticular structures even if the actual shoulder joint — the glenohumeral articulation — is not affected. This article will discuss common acute injuries to the shoulder and adjacent structures.
Labral Tears
A common shoulder injury that emergency physicians may not appreciate is a tear to the glenoid labrum. The glenoid labrum is a fibrocartilage structure in the shoulder joint that helps to anchor the joint capsule and shoulder ligaments. The labrum can be injured by a single traumatic event or by repetitive motion causing cumulative damage that builds up over time.
Anatomy and Biomechanics
The glenoid labrum adheres to the edge of the glenoid cavity. It increases the shape and coverage area of the glenoid surface. The glenoid labrum increases the contact area between the humeral head and scapula by 2 mm anteroposteriorly and 4.5 mm superoinferiorly.1 In addition to the capsule, the labrum also increases the glenohumeral stability by creating a seal around the humeral head. This creates a negative pressure, up to -32 mmHg, that increases setting of the humeral head into the glenoid fossa.1 The long head of the biceps brachii tendon inserts into the labrum, and is a major source of injury to the labrum.
The labrum is an insertion point for the capsule and glenohumeral ligaments. Depending on the position of the arm, the ligaments can undergo a variety of different levels of stress.
There are anatomic variants of the glenoid, which will not be discussed here, but that can contribute to an inappropriate diagnosis of labral pathology. Interruption of the labrum, such as a tear, can increase the translation of the humeral head on the glenoid.2
Injury
The most common labral tear identified on arthroscopy is the “SLAP lesion” (superior labrum, anterior, and posterior), a term coined by Snyder et al.3 This is an injury located at the biceps anchor and extends anteriorly to posteriorly. Snyder described four types of SLAP lesions. Type I is a frayed chondral rim that is typically the result of repetitive microtrauma rather than a discrete injury. Type II is a tear of the labrum, and the biceps anchor point is detached from the superior rim of the glenoid. Type III is a bucket-handle tear of the superior labrum without detachment of the biceps anchor. Type IV is a bucket-handle labral tear that extends into and splits the biceps. There have been many additions to the classification of SLAP tears, including most prominently those made by Maffet et al, who describe types V–IX that describe extensions of SLAP II tears (SLAP VI–VIII) in addition to a circumferential tear of the labrum off of the glenoid (SLAP IX).4
Several mechanisms are proposed to be responsible for SLAP lesions. These can be divided into acute and repetitive traumatic events. Of importance to emergency providers are the lesions that will cause pain, such as progressive pain that ultimately reaches a level for the patient to seek evaluation or sudden pain after a traumatic event.
Most commonly SLAP tears will cause pain posteriorly or the feeling of instability, and there may be an associated history of popping or clicking. There may be an associated trauma, or patients may have no recollection of an injury. Traumatic events include traction or compression of the shoulder. Some athletes may notice the injury during the act of throwing.3,5-8
Physical examination of the injured or painful shoulder should follow an organized pattern to enhance detection of useful findings. General inspection of the shoulder joint, active and passive range of motion, and strength are part of the standard shoulder examination. Several provocative tests have been described to aid in the diagnosis of labral tears. For the emergency physician, one or a combination of these three tests may be helpful in confirming suspicion for a labral tear.
In 1998, O’Brien described the active compression test, which many physicians use as their primary diagnostic physical exam test for labral pathology. This test requires the patient to be standing, with the shoulder flexed to 90 degrees (the entire arm straightly extended horizontal to the floor), then followed by 10 to 20 degrees of adduction (arm deviated toward the midline). The patient extends his or her thumb and rotates the arm to full pronation (thumb pointing to the floor). The physician then places downward pressure on the patient’s hand, and the patient resists, attempting to hold the arm in position. The provoking of deep pain or a sensation of clicking in the glenohumeral region should be noted. The patient then fully supinates the arm (thumb pointing horizontally outward) and downward pressure then is reapplied. A positive test is one where pain or a clicking sensation is noted when pronated but less so or completely absent when supinated.9,10 The original authors found the active-compression test to be extremely sensitive (100%) and specific (98%) for identifying labral tears.9
The crank test and the anterior slide test are two other commonly performed tests. The crank test is performed by passively elevating the shoulder to 160 degrees in the scapular plane followed by applying an axial load on the humerus while internally and externally rotating the shoulder. A positive test occurs when shoulder pain or a popping sensation is produced.6,11 For the anterior slide test, the patient places his or her hands on the hips with the thumb pointing posteriorly. An anterior-superior force is applied to the patient at the elbow while stabilizing the scapula. Reproduction of the patient’s shoulder pain, a pop, or a click is considered a positive test.6,12
As with most physical examination tests, further studies to replicate initial findings have found reduced sensitivity and specificity. In general, making the diagnosis of a labral tear solely on physical exam is somewhat unreliable. One study evaluating the aforementioned tests found that a combination of suggestive history (popping, clicking, and catching) combined with a positive provocative test, or multiple positive physical tests, improved the specificity for diagnosing a labral tear.6,10
Imaging Studies
Suspicion of shoulder injury in the ED setting should include radiographs of the shoulder joint. These include anteroposterior (AP), axillary, and scapular Y. For labral tears, imaging typically will be normal. However, there may be some bony abnormalities noted that are associated with labral tears, such as Bankart lesions, glenohumeral osteoarthritis, or calcific tendinopathy. Typically, dedicated tests for labral tears will not be performed in the emergency setting. The most accurate imaging study to detect labral tears in a MR arthrogram (MRA) performed with intra-articular injection of gadolinium. The reported sensitivity of an MRA for labral tears is about 88%, with a specificity of about 90%.7,13 A standard MRI performed without contrast has a reported sensitivity for labral tears of about 76%, with a specificity of about 87%. However, the noncontrast MRI has more variability due to the skill of the interpreting radiologist.
Management
Management of isolated labral tears in the emergency setting involves referral to an orthopedic surgeon for discussion of nonoperative vs. operative management. Operative repair can be executed arthroscopically, with repair and/or debridement of the lesion. Although less clinically relevant for the ED physician, there is often concomitant pathology with labral tears, such as rotator cuff tears, resulting in shoulder instability that may need to be addressed surgically.
There is no need for immobilization or stabilization in the ED. If there is suspicion of an acute labral tear, patients should be advised to stop any throwing activities or activities that induce pain. A short course of anti-inflammatory medications to reduce pain and inflammation should be prescribed. Nonoperative patients should be referred to physical therapy for exercises that improve mobility and strength.
CONCLUSION
Diagnosis of labral tears in the ED isn’t always feasible or possible, but it is important to keep this pathology in mind when dealing with shoulder pain after recent trauma. The ED physician also may see patients who present with chronic shoulder pain who do not have the proper follow-up and thus can be assisted. The most prudent course of action is to manage the acute symptoms and to refer the patient to proper follow-up in the outpatient setting.
Shoulder Dislocation
The shoulder is the most commonly dislocated large joint in the body, and the most common joint dislocation to present to the ED. Shoulder dislocation is classified according to the direction the humeral head has in relation to the glenoid, but up to 98% of cases occur when the humeral head is displaced anteriorly.14 A small percentage of patients present with inferior shoulder dislocation. The presentation of shoulder dislocation occurs in a bimodal age distribution. These two groups include young adult men, and men and women older than 60 years of age.14,15
The cause of anterior shoulder dislocation is typically violent external rotation in abduction, which moves the humeral head anteriorly.15 Younger patients are more apt to have dislocation from sporting injuries, while older patients may have dislocations from falls or seizures. Posterior dislocations classically are associated with seizures, lightning injuries, or electrical injuries.16
Shoulder dislocation requires emergent management in the ED. Patients typically present with severe pain, which should be addressed prior to imaging. Typically, the ED physician can quickly assess for the presence of dislocation upon presentation of the patient to the ED. There is usually a preceding mechanism of injury (i.e., a fall or traction injury).
Anterior Dislocations
Anterior dislocations are a frequent complaint in the emergency setting. Classically, this pathology presents with the patient holding the arm in slight abduction and external rotation. The patient typically is in severe pain. Clinically, an anterior dislocation is easier to recognize than a posterior shoulder dislocation because of physical exam features.
Standard radiographs are usually adequate for diagnosis. (See Figure 1.) The physician can see alignment of the glenoid with the humeral head as well as any fractures associated with the traumatic event. There are four types of anterior dislocations, all characterized according to where the humeral head lies. These include subcoracoid (most common), subglenoid, subclavicular, and intrathoracic (very rare). If anterior shoulder dislocation is suspected after physical exam and history, appropriate X-rays of the shoulder should be ordered. These typically include an AP radiograph and axillary views.
Figure 1. Anterior Shoulder Dislocation
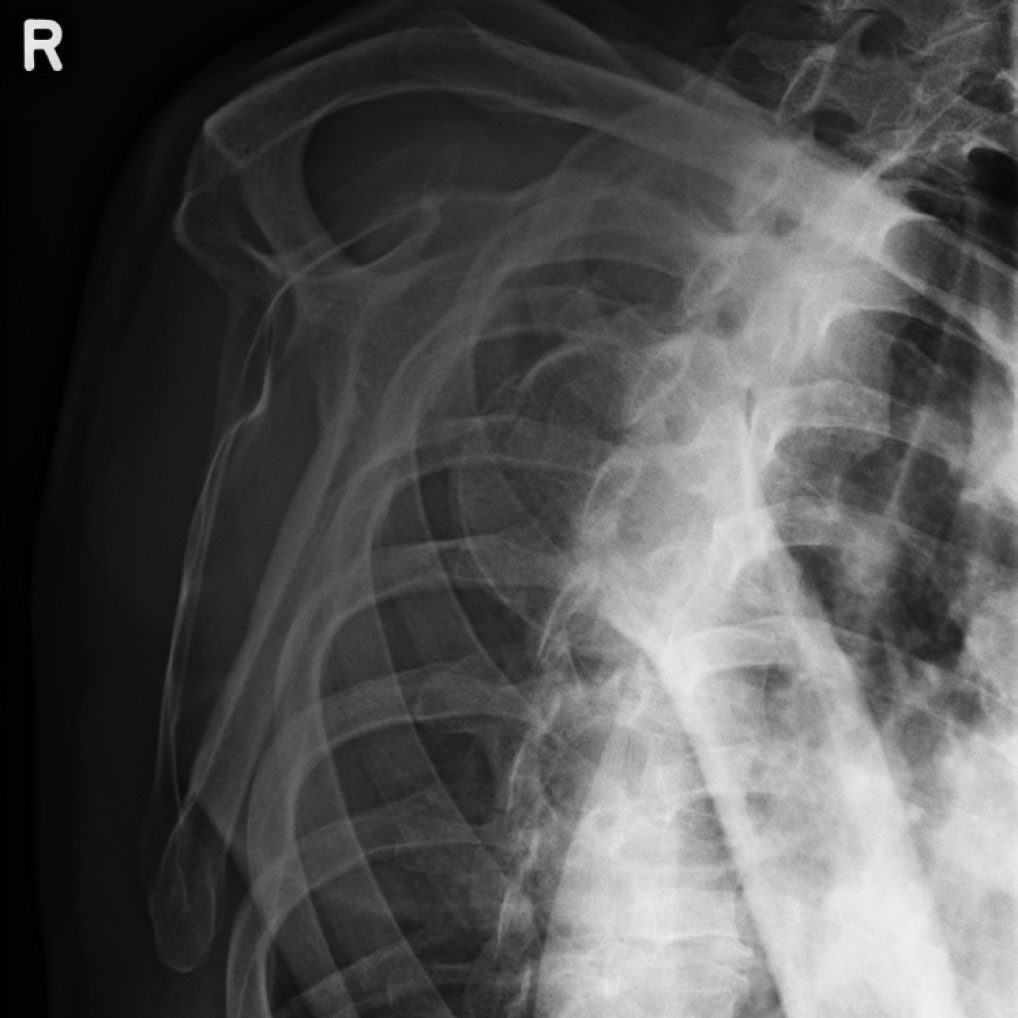
Image courtesy of David Effron, MD
Approximately 5-10% of anterior shoulder dislocations cannot be reduced in the ED, and they will require a visit to the operating room.17 The remainder can be successfully reduced in the ED using a variety of techniques, including traction/countertraction, Stimson technique, external rotation, scapular manipulation, and the modified Cunningham technique. Although some techniques are more effective than others, no technique will yield 100% results. In the emergency setting, physician preference and the clinical situation will dictate which technique to use.
There are two main modalities for analgesia during reduction: conscious sedation and local anesthesia via intra-articular local anesthetics. Conscious sedation is a popular choice among emergency physicians, as this adequately controls the patient’s pain during reduction. It also produces muscle relaxation and helps facilitate the reduction. In the past, conscious sedation was conducted with the use of intravenous opioids combined with benzodiazepines. More recently, conscious sedation has evolved to the use of propofol and ketamine because of their decreased adverse reaction profile. However, there continues to be the risk for adverse reactions, include hypotension, respiratory depression, and agitation, with the use of intravenous procedural sedation. In addition to adverse reactions, evidence shows that there is an increased length of stay for conscious sedation when compared to local analgesia.18 In a systematic review, Finch et al found overwhelming evidence of decreased length of stay with the use of intraarticular lidocaine when compared to IV sedation.18
Posterior Shoulder Dislocations
Posterior shoulder dislocations account for 1-4% of all glenohumeral dislocations.19-21 However, given their rarity and subtle physical exam and radiologic findings, these traumatic injuries often are missed on the initial presentation. A study by Rowe and Zarins showed that the initial treating physician did not recognize 79% of posterior dislocations at time of presentation.22 Furthermore, delayed diagnosis of posterior shoulder dislocations can lead to increased complications such as avascular necrosis and degenerative disease.
To diagnose a posterior shoulder dislocation, it must first be within the physician’s consideration. The mechanism of injury should trigger suspicion of a posterior dislocation in the patient who suffers a powerful blow anteriorly falls on an outstretched hand, has a seizure, or suffers from an electrocution. In presentation of seizure or electrocution, the physician should consider bilateral shoulder injury. The mechanism in seizure and electrocution involves the overpowering of the internal rotators (latissimus dorsi, pectoralis major, subscapularis, and teres major) by the external rotators (infraspinatus and teres minor).19
Physical diagnosis of a posterior dislocation on exam will reveal a patient with his or her arm held in internal rotation and adduction. On palpation, the physician may find a more pronounced coracoid process and a posterior humeral head. These findings may be subtle, so range-of-motion testing is important and typically will reveal a lack of passive or active ability to adduct the shoulder. The patient will not be able to externally rotate the shoulder. In delayed presentation, posterior shoulder dislocation may appear similar to a frozen shoulder. Obtaining a history of any recent trauma will help in the diagnosis of posterior dislocation.
Standard shoulder radiographs are often inferior in diagnosing a posterior dislocation. On true antero-posterior shoulder radiographs, the signs of a posterior dislocation are subtle. The subtle findings include the light bulb, vacant glenoid, rim, and trough line signs. The light bulb sign is a result of the internal rotation of the humeral head causing the projection to look like a light bulb. The vacant glenoid sign shows the glenoid fossa appearing empty. The trough line sign shows a dense vertical line that represents a reverse Hill-Sachs lesion in the superomedial portion of the humeral head. Unfortunately all of these signs are subtle and may be missed by a clinician. The most definitive view on X-ray is the axillary view. This view can be difficult to obtain in patients presenting with shoulder pain in the ED. Clough et al recommend obtaining an axillary or a modified axillary (trauma scenario) view in any patient with a history of seizure, electrocution, or similar trauma who has limited external rotation.23 More advanced imaging, such as CT scans and MRI, should be obtained if there is concern for additional pathology. CT can help clarify any osseous lesions, while MRI is good for identifying rotator cuff pathology.
Figure 2. Posterior Shoulder Dislocation
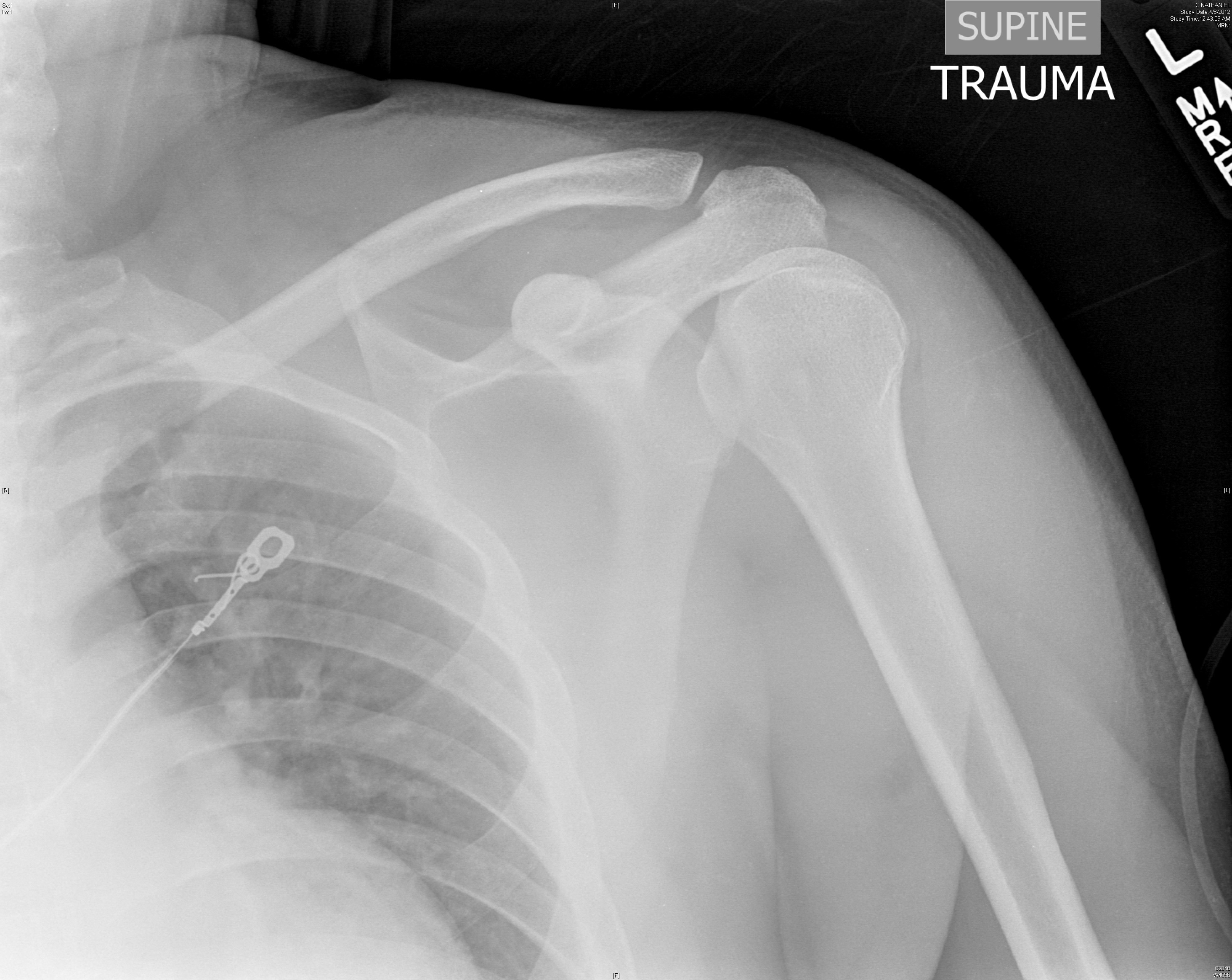
Image courtesy of J. Stephan Stapczynski, MD
Figure 3. Posterior Shoulder Dislocation
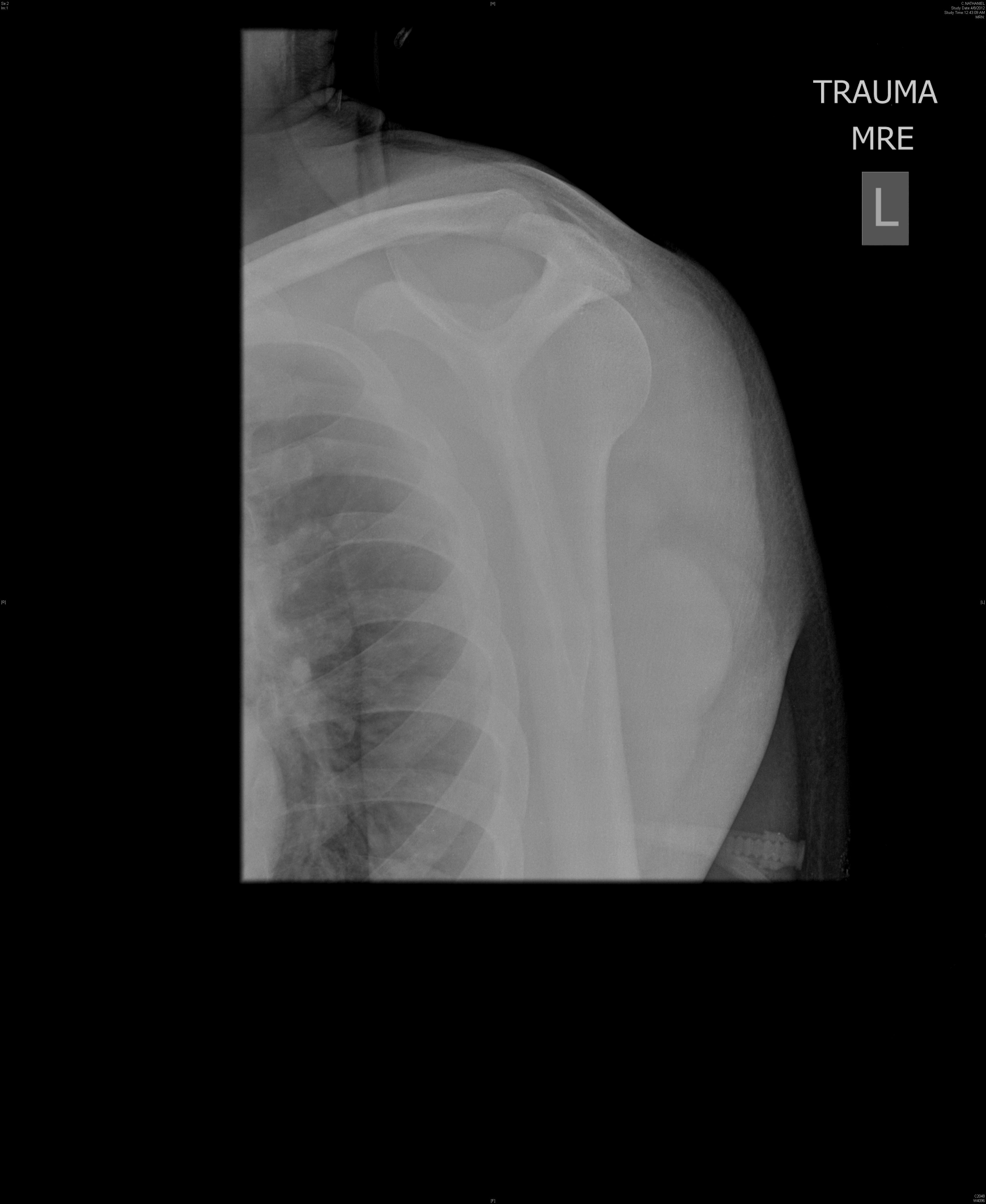
Image courtesy of J. Stephan Stapczynski, MD
Reduction of posterior dislocation should be attempted only if the glenoid defect (reverse Bankart lesion) is less than 20%.20,24 Once concomitant bony injuries have been determined, posterior dislocation can be reduced using conscious sedation or local anesthetic techniques. To reduce a posterior shoulder, gentle traction is placed laterally on the shoulder, with pressure applied to the humeral head pushing it anteriorly. Once in this position, the arm is slowly externally rotated. After successful reduction, the arm should be placed in a position of abduction and external rotation. If the physician is unable to relocate the shoulder or if the patient has a history of recurrent instability, reduction will require an operative approach, likely with repair of any soft tissue injuries (capsule, labrum) that prevent relocation or stability.
Associated Injuries and Potential Morbidity of Shoulder Dislocation
The most common injury that occurs concomitantly with an anterior shoulder dislocation is a Bankart lesion (injury to the glenoid rim), which results from the humeral head impacting the anterior-inferior labrum. Bankart lesions can be soft (injury to the labrum) or bony (injury to the glenoid visible as a fracture of the anterior-inferior aspect of the glenoid). The simultaneous lesion on the humeral head is known as the Hill-Sachs lesion. Other injuries of the glenohumeral joint can include anterior labral periosteal sleeve avulsion (ALPSA), humeral avulsion of glenohumeral ligament (HAGL tear), SLAP tear, or a bony Bankart (involvement of glenoid rim). These will not be seen on standard shoulder X-rays, but are important complications that will reduce stability for the shoulder and cause persistent pain. A frequent injury associated with anterior dislocation is rotator cuff tears. In a prospective study, clinicians found that 144 of 240 (60%) patients with anterior shoulder dislocation had rotator cuff tears.25
Figure 4. Chronic Hill-Sachs Deformity
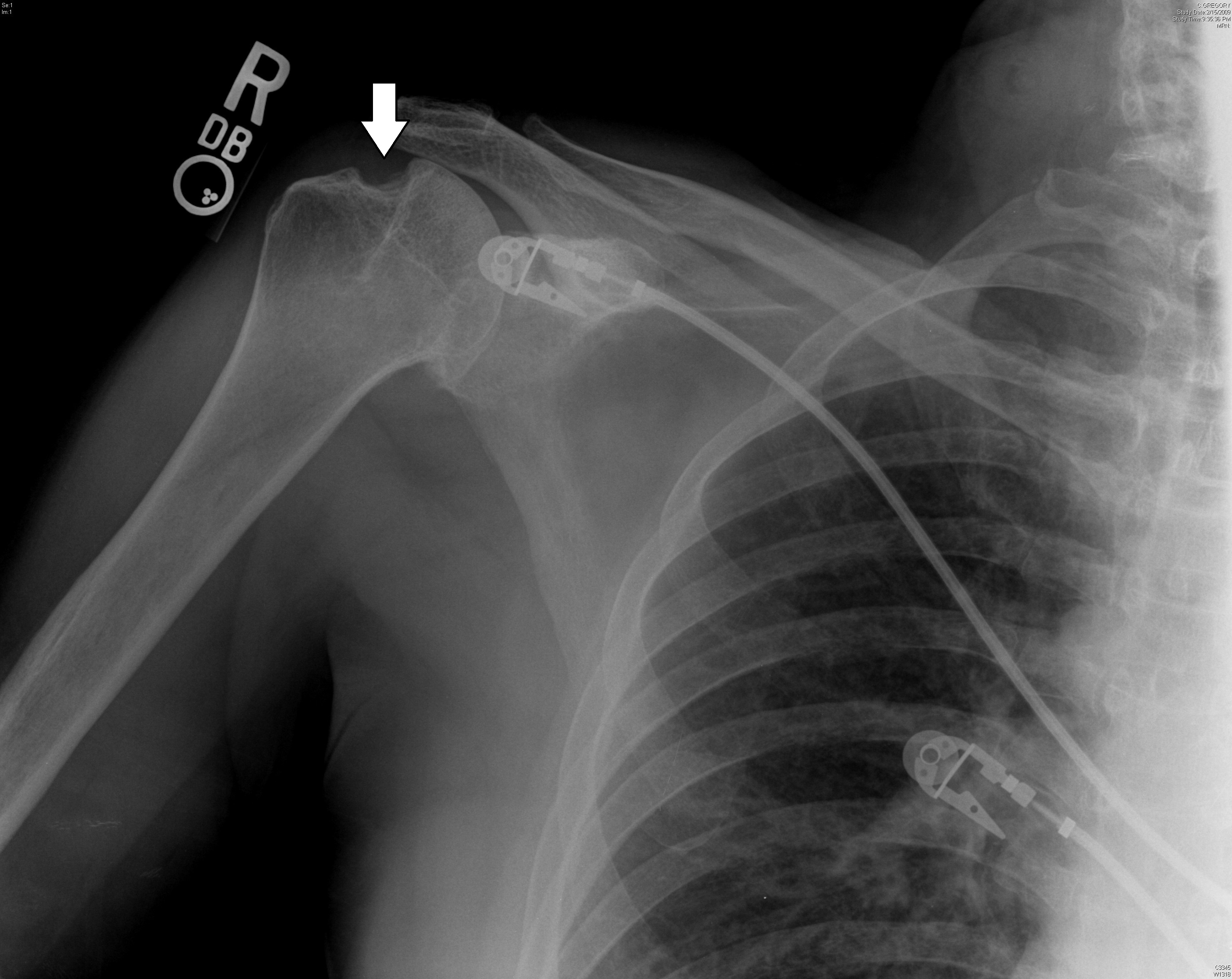
Image courtesy of J. Stephan Stapczynski, MD
Posterior shoulder dislocations can present with other complications. These also include fractures of the proximal humeral shaft, tuberosity, and glenoid and soft tissue injuries. Soft tissue injuries are diagnosed less frequently because standard imaging only includes plain radiographs. More subtle injuries can be found with MR imaging. In many ways, the mechanism of posterior dislocation is the reverse of an anterior shoulder dislocation, so patients with posterior dislocation will have soft tissue injuries that are opposite in location. These soft tissue injuries include reverse Hill-Sachs lesions on the anterior part of the humeral head, reverse osseous Bankart lesion, rotator cuff tears, bicep tendon abnormalities, and injuries to the joint capsule and labrum. In a study of 36 patients who underwent MRI after first-time traumatic posterior shoulder dislocation, multiple patients were found to have additional bony and soft tissue injuries. Approximately 86% of patients had reverse Hill-Sachs lesions, and 20% of patients had full thickness rotator cuff tears.21
In addition to bony and soft tissue injuries, neurovascular structures also can be damaged. The glenohumeral joint sits near the brachial plexus and the axillary vasculature. A review of the literature between 1956-1995 revealed only 22 cases that related anterior shoulder dislocation to axillary artery injury.26 The most common location of the arterial injury in this review was the third part of the axillary artery, defined as the portion of the artery distal to the lower edge of the pectoralis minor muscle. The authors found that the arterial injuries occurred more often in patients older than 50 years of age (86%). All but one of the patients reviewed presented with absent distal pulses.26 Diminished or absent pulses on the dislocated side should be evaluated with vascular injury such as an arteriogram.
Brachial plexus, axillary, and radial nerve injuries are also complications of shoulder dislocation. The reported incidence of nerve injuries in patients with shoulder dislocations seen in the ED varies between 21% and 65%.27 They are seen most commonly in luxatio erecta and less commonly in anterior dislocations. They are rare in posterior dislocations, as the neurovascular bundle sits anterior to the glenoid fossa. A full neurological exam should be completed at the time of examination to rule out injury. Most axillary nerve injuries are neuropraxic in nature and should be managed nonoperatively, and recovery of function is expected. Signs of axillary nerve injury may include loss of sensation on the lateral aspect of the shoulder (unreliable) and inability to abduct the shoulder. Radial nerve injury will present with deficits in wrist extension and sensation over the dorsal aspect of the thumb, index, and middle fingers. Symptoms and signs of nerve injury usually resolve with relocation of the shoulder. Those that persist, with the exception of the axillary nerve, should be referred to the orthopedic specialist for consideration of surgical management.27 Emergency clinicians should examine and be aware of the possible concomitant injuries that can occur with glenohumeral dislocation.
Luxatio Erecta
Inferior shoulder dislocation is seen rarely. Making up less than 1% of all shoulder dislocations, luxatio erecta humeri typically present with the shoulder abducted in the “hands up” position.28 It is caused by hyperadduction or direct axial load to the affected extremity.29 In the hyperadduction mechanism, the shoulder is already in an abducted position. A strong hyperadduction force causes the humeral neck to lever across the acromion, thus pushing the humeral head inferiorly. This mechanism typically results in the humeral head rupturing the inferior and middle glenohumeral ligaments and joint capsule. In addition to these injuries, the supraspinatus and infraspinatus also are torn. The direct axial mechanism involves a force on the arm inferiorly, which displaces the humeral head from the glenoid, typically rupturing the inferoglenohumeral ligaments and joint capsule, with frequently associated fracture of the greater tuberosity.30,31
The danger of this dislocation is its complications. In a case series, Mallon et al observed that a fracture of the greater tuberosity or a rotator cuff tear was present in 80% of patients.32 Approximately 60% of the patients had some degree of neurologic compromise, most frequently involving the axillary nerve. Also, 3.3% of the cases had vascular injury, which represents the highest incidence of vascular injury in any type of shoulder dislocation.32
Most of the data gathered for treatment of this pathology are from case reports, as the occurrence is rare. The consensus in the literature advocates assessing neurovascular status and evaluating for occult injury. This typically includes using CT and Doppler studies to identify any fractures or vascular injuries, respectively. Many case reports have reported success using the traction-counter traction method along with analgesia for reduction.28,29,33 In one case report that did not have the luxury of time or safety to undergo procedural sedation due to the emergent nature of the patient’s trauma, an interscalene block was used to help reduce the shoulder quickly so that the patient could fit in the CT scanner.34
Typically, the presentation of luxatio erecta humeri is obvious, but it can be confused with a subglenoid dislocation when looking at radiographs. These two dislocations can be differentiated by clinical exam and observing an abducted humerus on the radiograph.
After reduction and assurance that there is no neurovascular injury, these patients should be placed in a sling for shoulder immobilization. Discharge instructions should include recommendations of close follow-up with an orthopedic surgeon for treatment of the commonly associated injuries, such as rotator cuff tear, fracture, or joint instability.
Newer Technologies
Ultrasound is a potential imaging modality to diagnose shoulder dislocation and confirm reduction. Ultrasound is a safe, non-radiating method to visualize shoulder anatomy. Advantages of ultrasound include real-time imaging, safety, and decreased length of stay. Disadvantages of ultrasound include lack of provider experience, user reliability, decreased ability to view fractures, and a lack of overall studies comparing it with the typical standard of plain radiographs. One study by Abassi et al used a linear probe to compare accuracy of ultrasound diagnosis of anterior shoulder dislocation to the standard of radiography. In this study, ultrasound was 100% sensitive for anterior shoulder dislocations and 100% sensitive for assessing reduction. The sonographers also were able to identify all the fractures seen on plain radiographs.35
As research develops, ultrasound likely will take a more decisive role in the emergency and office setting when diagnosing and treating shoulder dislocation. The main advantage of ultrasound likely will be time reduction for treatment of the patient. With ultrasound confirmation of satisfactory reduction, less time will be spent imaging unreduced shoulders, and eventually ultrasound may replace the need for post-reduction films altogether.
Ultrasound also can assist with intra-articular injections or nerve blocks used to provide analgesia. Its use in the ED and outpatient clinics will be dependent on availability of the technology and training of the physicians using the technology.
Chronic Dislocation
Chronic dislocations (more than three to four weeks) can be associated with capsular adhesions to nearby structures, especially in the elderly population. In a 1941 French study, Calvet et al noted that in a reduction of 91 chronic anterior shoulder dislocations, 68 patients suffered from arterial ruptures.36 In a more recent review of the literature, closed reduction methods showed only satisfactory results in 27 out of 50 patients.37 It is not recommended that chronic shoulder dislocation be treated in the emergent setting. The preferred method of treatment is open reduction, which will require an orthopedic surgeon who can repair any bony, ligamentous, muscular, or cartilaginous defects.
Traumatic Rotator Cuff Tear
The rotator cuff is a combination of muscles that fuse to form a common tendon. It is crucial for movement and the stability of the glenohumeral joint. It is composed of four muscles. These include the abductor: the supraspinatus; the external rotators: infraspinatus and teres minor; and the internal rotator: subscapularis. These four muscles combine to form the common tendon, which is predisposed to both acute and chronic injuries. Chronic injuries are about nine times more common than acute traumatic injuries and tend to be more subtle and from chronic overuse or impingement. Acute tears usually are associated with a specific traumatic injury. There also can be an acute extension of a chronic injury. For the purposes of this article, only acute injuries will be discussed.
Mechanism
Full thickness acute rotator cuff tears require a high-force mechanism of injury. These can include shoulder dislocation or a motor vehicle accident. Lesser trauma typically causes partial thickness tears. The mechanism for these tears may be a fall onto an outstretched arm in an abducted position. Patients sometimes may present after injury while the arm was abducted and flexed above the head. The injury will feel like a sudden tearing sensation in the shoulder with severe pain that radiates into the arm.
Diagnosis
Traumatic rotator cuff tears often can be missed initially in the ED. This likely is due to the subtle physical findings associated with traumatic rotator cuff tear and pain that will present difficulty in examination. On presentation, the patient with an acute rotator cuff tear will have significant shoulder pain. There likely will be a specific traumatic event occurring at the onset of the pain. The patient will have tenderness to palpation over the anterolateral shoulder, and likely will complain of inability to complete overhead activities. This injury will be particularly painful if the patient lies on the affected arm during sleep.
The approach to the physical exam should be a stepwise systematic evaluation of the shoulder joint. Inspection should include looking for signs of swelling, effusion, atrophy of muscles, or gross deformities. The physician should palpate for any signs of bony deformity or soft tissue tenderness. Typically, there will be tenderness of the humeral head in the location of the tendon rupture. Passive and active range of motion (ROM) should be the first test conducted on the shoulder. This includes forward flexion, abduction, internal rotation, and external rotation. The most precise way to measure range of motion is with a goniometer. In the emergent setting, subjective assessment of ROM compared to the unaffected shoulder should produce similar results.38 Strength testing should be completed by testing external rotation (infraspinatus), internal rotation (subscapularis), and abduction (supraspinatus). The most common rotator cuff tear involves the supraspinatus; thus, the physical exam will reveal deficits in abduction strength.
A recent literature review found more than 25 special tests described for examination of the rotator cuff.37 It is impossible to perform all of these in a busy emergent setting. Therefore, it is important to make to test each muscle group of the rotator cuff adequately.
For the subscapularis, function can be tested with the lift-off test, belly press test, and the bear hug test. The lift-off test requires the patient to put the hand of the affected shoulder behind the back and to push backward off the back. The belly press test involves the patient forcefully pushing the hand on the belly while the clinician assesses for any signs of weakness, which is manifested by the elbow dropping out of internal rotation. For the bear hug test, the patient places the palm of the hand on the opposite shoulder. The physician then applies an external rotation force. Any weakness is a sign of subscapularis injury.38
The infraspinatus and the supraspinatus are tested commonly with the external rotation lag sign, empty can test (Jobe’s test), and the drop arm test. The external rotation lag sign is tested with the elbow in 90 degrees flexion. The arm is abducted in the scapular plane and the arm is rotated to maximum external rotation by the examiner. If the arm moves back, it is considered a positive test. The empty can test is tested with the arm in 90 degrees in resisted forward flexion and pronation with the thumb pointed down. Pain or weakness indicates a tear of the supraspinatus. For the drop arm test, the physician places the patient’s extended arm in 90 degrees of abduction (or even higher in full abduction) and then instructs the patient to slowly lower the arm to their side under their own power. A normal response is a slow, smooth lowering of the arm under control. An abnormal or positive drop arm test is when the patient has difficulty in sustaining abduction or executing a smooth, controlled lowering, indicating a supraspinatus tear.38
Teres minor is tested with the horn blower’s test. This is a test with the arm held by the clinician at 90 degrees abduction in the scapular plane with the elbow flexed 90 degrees. The patient then attempts resisted external rotation. Any weakness is associated with a teres minor injury.38
Imaging
Once a physical examination confirms suspicion of a rotator cuff injury, the physician may want to evaluate further with imaging studies. Plain radiographs cannot visualize the rotator cuff itself, but there are bony clues to the existence of rotator cuff pathology. In complete tears, the humeral head is displaced superiorly. In more chronic tears, there is sclerosis of the greater tuberosity and acromion, along with spurs. The standard shoulder images (AP, lateral, scapular outlet) should be obtained initially.
There are currently two main modalities to image the rotator cuff itself: ultrasound and MRI. There is no indication for emergent MRI due to rotator cuff pathology; bedside ultrasound may be a good tool to confirm suspicion of an acute rotator cuff tear. In a meta-analysis of 82 articles, Roy et al found that ultrasound, MRI, and MRA had sensitivities of 91%, 90%, 90%, and specificity of 86%, 90%, 90% in diagnosing full or partial thickness tears of the rotator cuff.39
On MRI, there will be an increase signal in T2 in the area of the supraspinatus, with fluid filling in the defect. Criteria for diagnosis of rotator cuff injury in one study were fluid intensity signal on FS T2-weighted images and discontinuity of tendon fibers at or near the insertion of the supraspinatus and infraspinatus tendons.40 MRI is the imaging modality of choice in the outpatient setting.
In the ED, point-of-care ultrasound can be beneficial in aiding a quick and safe diagnosis of acute rotator cuff tear. While the diagnostic capability of ultrasound is user dependent, this is a good way to make an early diagnosis.
Management
Treatment for acute tears requires immobilization in a sling. The patient will need referral to an orthopedic surgeon or sports medicine physician for further imaging (MRI/MRA). Surgical repair is indicated in acute full thickness tears. Shoulder rehabilitation programs are essential both postoperatively and preoperatively.
Overall, rotator cuff injury is a common pathology that will present in both the emergent and outpatient clinical setting. Acute tears of the rotator cuff are more obviously associated with a traumatic injury and are very sensitive to the touch. The patient with an acute rotator cuff tear likely will require analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs) for pain control, sometimes with a short course of opioids for a few days until the severe pain resides. Follow-up is the key for continued evaluation and further treatment of rotator cuff injuries.
Acromioclavicular Joint
The acromioclavicular (AC) joint plays an important role in shoulder pathology. Patients who present after a direct fall onto the shoulder or a fall with an outstretched arm may sustain AC joint injury. The AC joint is strong, but because of its exposed position, it is prone to injury from blunt trauma.The AC joint is involved in 9-12% of all shoulder girdle injuries.41,42
Mechanism
The AC joint is a diarthrodial joint, meaning that it is a type of bony articulation that allows movement and has a synovial membrane. Within the joint is a fibrocartilaginous disk that helps to dissipate the forces from the upper extremity to the axial skeleton.41 The AC joint also is supported by multiple ligaments that include the coracoacromial ligament, coracoclavicular ligament (trapezoid ligament and conoid ligament), and the acromioclavicular ligament. Within the AC structure there are four ligaments: superior, inferior, posterior, and anterior. Additionally, the AC joint owes its support to the deltoid and trapezius muscles, as these cross the joint and play a role in its injury.
There is minimal movement of the AC joint; however, it plays an important role. In 1990, Rockwood and Young were able to obtain a range of 5-8 degrees in a motion they deemed “synchronous scapuloclavicular rotation” in a living patient.41,42 Fukuda et al studied the different ligamentous contributions to joint constraint depending on the stress applied and found that all ligaments contribute significant stability to the joint. Specifically, they noted that most pathologic displacement of the clavicle occurs in the posterior or superior direction, and that in these directions the acriomioclavicular ligament (less displacement and load) and the conoid ligament (larger displacement and induced load) contributed the most constraining forces.43 Multiple biomechanical studies have confirmed that the AC ligaments confer more stability in the antero-posterior plane, while the coracoclavicular (CC) ligaments confer vertical stability.44
AC joint injury is most commonly seen in young males, especially among hockey, football, and rugby players, and among those in motorcycle and bicycle accidents. Most commonly, AC injury relates to a disruption of the ligaments supporting the joint, causing subluxation or dislocations.45,46
Classification
AC joint injuries are classified using Rockwood’s classification, as modified in 1984.41 This classification follows a stepwise involvement/disruption of the AC joint stabilizers. Briefly, type I is classified as a sprain of the joint without complete tear of the ligament. This likely will manifest with AC joint tenderness and a normal X-ray. Type II is a complete tear of the acromioclavicular ligament with a concomitant coracoclavicular ligament sprain, but still intact. On examination, there may be a noticeable separation of the elevated distal end of the clavicle from the acromium, and significant pain, corresponding with X-ray findings of mild elevation of the lateral aspect of the clavicle. Types I and II typically are treated conservatively and can be managed with ice, NSAIDs, and rest until the patient has a painless range of motion. Immobilization devices have been used, but generally are found to be detrimental due to skin breakdown and muscle atrophy and should not be used longer than a period of two weeks. Due to the increased severity of the injury, type II injuries may take up to six to eight weeks to recover, while type I injuries recover in a span of two weeks.41
Type III AC joint dislocation involves complete tear of the AC ligament and the CC ligament, resulting in superior displacement of the distal clavicle relative to the acromion with complete AC dislocation. (See Figure 5.)
Figure 5. Left Type III AC Separation
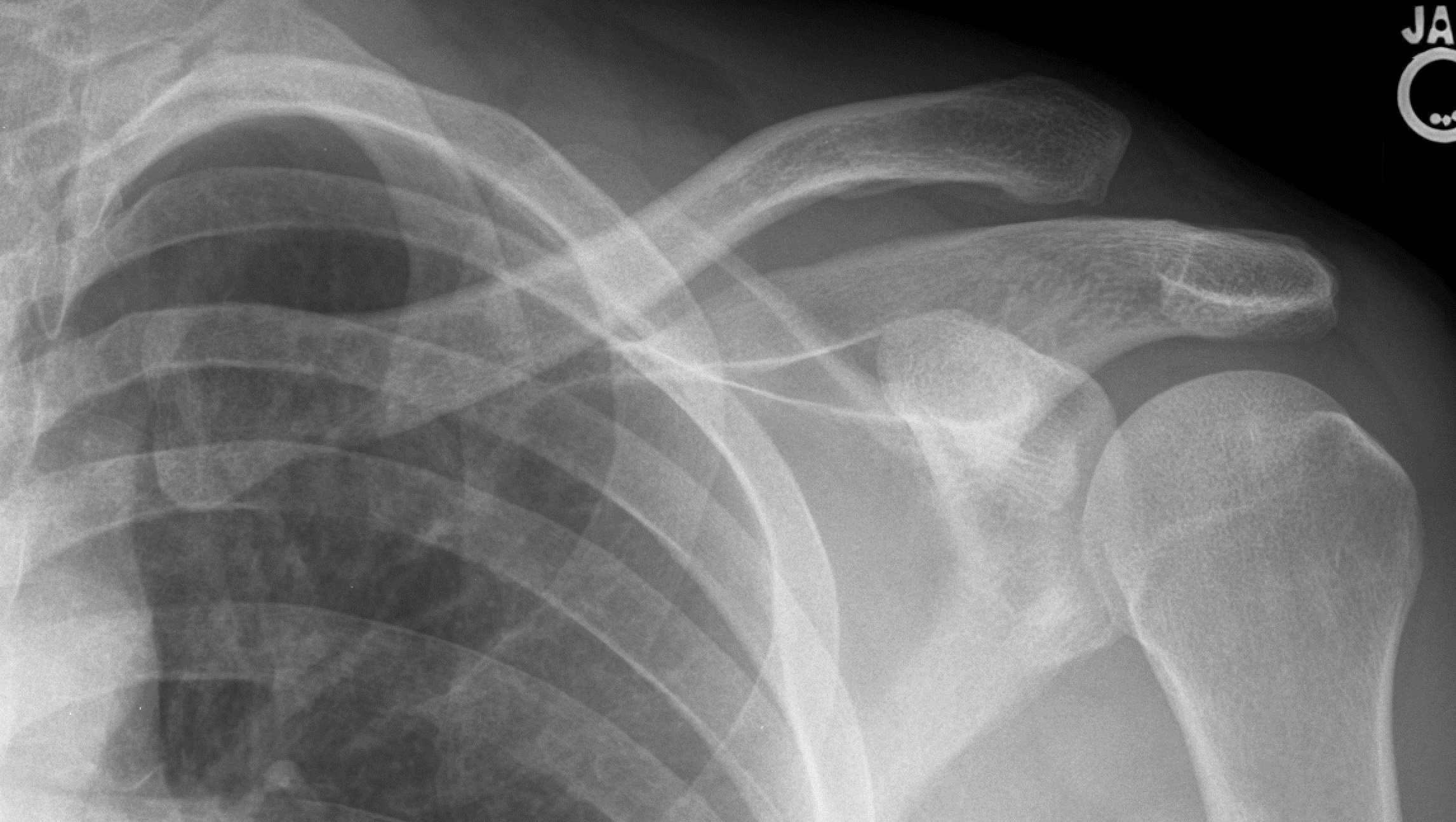
Image courtesy of J. Stephan Stapczynski, MD.
Management of type III AC separation is a current area of discussion. These have been managed both surgically and non-operatively. Proponents of conservative non-operative management argue that patients regain excellent results and painless shoulder function in the setting of decreased complications and decreased return to sport time, while those who favor surgery cite potential for chronic instability and pain with non-operative management. Several meta-analyses and systematic reviews have looked at the clinical outcomes of different management techniques following type III AC dislocation. Their conclusions indicate that non-operative treatment is likely as good as operative treatment, but there is a need for additional robust and quality data to identify potential patients who might benefit from early surgical intervention.47-50 When seeing this injury in the ED, referral to orthopedic surgery is appropriate and necessary to determine further workup.
Types IV-VI are more rare and serious injuries to the AC joint that require surgery. Type IV is a rupture of the AC and CC ligaments with posterior dislocation through the trapezius muscle, which may result in tenting of the skin. Type V is characterized by severe separation in AC distance as seen on radiograph and is a result of injury of the trapezial and deltoid fascia. Type VI is extremely rare and results in inferior dislocation of the distal clavicle in either the subacromial or subcoracoid positions. Type IV is usually the result of a high-energy collision, such as collision with a fixed object, motor vehicle collision, or fall from height, and can be associated with other traumatic injuries. Neurovascular assessment in these injuries is vital and the dislocation may need to be reduced. Types IV-VI likely will need some element of reduction in the ED or by the emergency physician or an orthopedic consult for management. Surgical repair is the treatment.
Diagnosis
Diagnosis of AC joint dislocation hinges upon history, physical exam, and radiographic analysis. Look for history of direct trauma to the area or an axial load mechanism. Standard shoulder radiographs, clinical suspicion, and AC joint tenderness should be enough to make the correct diagnosis. There are special tests that may indicate an AC joint injury. Local AC joint tenderness tends to be the most sensitive, which is intuitive. Other tests that may assist include the O’Brien (previously described for SLAP tears) and Paxinos (squeezing acromion and clavicle), which were found to be 79%/16% sensitive and 50%/90% specific in a study that compared physical exam techniques to radiographs, bone scans, and MRIs.45 The sensitivity and specificity of standard shoulder radiographs in diagnosing AC injury is 41% and 90%, respectively.45,46
AC joint injury may be seen in any acute or urgent care setting, and the ability of the physician to recognize this injury and distinguish it from other shoulder pathology will be of much help to the patient long term. These patients should be referred to a sports medicine physician or an orthopedic surgeon if their injury may require surgery (IV-VI). Appropriate rehabilitation is also important for long-term outcomes of pain and function.
Clavicular Fracture
The clavicle is an integral structure in shoulder mobility and support. It is the only osseous connection between the axial skeleton and the arm. As expected, an injury to this bone can render an entire arm limited in function. Clavicular fractures have an estimated incidence of 24.4 fractures per 100,000 person years in the United States.51 They occur more frequently in males, with men nearly three times as likely to experience a clavicle fracture.51 These fractures are caused most commonly by a fall on an outstretched arm or direct impact.
The majority of these injuries can be treated non-operatively and in a sling. However, it is key for the ED physician to identify those that may need operative repair to relay the urgency to the patient of prompt follow-up.
SUMMARY
The emergency physician will encounter a variety of shoulder injuries in clinical practice. It is useful to consider the mechanism of injury when assessing these patients. After the initial neurovascular evaluation of the involved arm, select from the many examination maneuvers and tests to help further assess the injury. After the imaging is warranted to evaluate for potential fractures or dislocations. Patients with suspected cartilage or ligamentous injury should be referred to an orthopedic or sports medicine specialist for follow up.
REFERENCES
- Clavert P. Glenoid labrum pathology. Orthop Traumatol Surg Res 2015;101(1 Suppl):S19-24.
- Pagnani MJ, Deng XH, Warren RF, et al. Effect of lesions of the superior portion of the glenoid labrum on glenohumeral translation. J Bone Joint Surg Am 1995;77:1003-1010.
- Snyder SJ, Karzel RP, Pizzo WD, et al. SLAP lesions of the shoulder. Arthroscopy 1990;6:274-279.
- Maffet MW, Gartsman GM, Moseley B. Superior labrum-biceps tendon complex lesions of the shoulder. Am J Sports Med 1995;23:93-98.
- Burkhart SS, Morgan CD. The peel-back mechanism: Its role in producing and extending posterior type II SLAP lesions and its effect on SLAP repair rehabilitation. Arthroscopy 1998;14:637-640.
- Michener LA, Doukas WC, Murphy KP, et al. Diagnostic accuracy of history and physical examination of superior labrum anterior-posterior lesions. J Athl Train 2011;46:343-348.
- Dodson CC, Altchek DW. SLAP lesions: An update on recognition and treatment. J Orthop Sports Phys Ther 2009;39:71-80.
- Solomon D, Levine W. Slap tears: Pearls and pitfalls in diagnosis and management. Sports Medicine Update 2011:2-3,4,5,6.
- O’Brien SJ, Pagnani MJ, Fealy S, et al. The active compression test: A new and effective test for diagnosing labral tears and acromioclavicular joint abnormality. Am J Sports Med 1998;26:610-613.
- Walsworth MK, Doukas WC, Murphy KP, et al. Reliability and diagnostic accuracy of history and physical examination for diagnosing glenoid labral tears. Am J Sports Med 2008;36:162-168.
- Liu SH, Henry MH, Nuccion SL. A prospective evaluation of a new physical examination in predicting glenoid labral tears. Am J Sports Med 1996;24:721-725.
- Kibler WB. Specificity and sensitivity of the anterior slide test in throwing athletes with superior glenoid labral tears. Arthroscopy 1995;11:296-300.
- Bencardino JT, Beltran J, Rosenberg ZS, et al. Superior labrum anterior-posterior lesions: Diagnosis with MR arthrography of the shoulder. Radiology 2000;214:267-271.
- Chalidis B, Sachinis N, Dimitriou C, et al. Has the management of shoulder dislocation changed over time? Int Orthop 2007;31:385-389.
- Cutts S, Prempeh M, Drew S. Anterior shoulder dislocation. Ann R Coll Surg Engl 2009;91:2-7.
- Kroner K, Lind T, Jensen J. The epidemiology of shoulder dislocations. Arch Orthop Trauma Surg 1989;108:288-290.
- Ufberg JW, Vilke GM, Chan TC, et al. Anterior shoulder dislocations: Beyond traction-countertraction. J Emerg Med 2004;27:301-306.
- Fitch RW, Kuhn JE. Intraarticular lidocaine versus intravenous procedural sedation with narcotics and benzodiazepines for reduction of the dislocated shoulder: A systematic review. Acad Emerg Med 2008;15:703-708.
- Cicak N. Posterior dislocation of the shoulder. J Bone Joint Surg Br 2004;86:324-332.
- Perron AD, Jones RL. Posterior shoulder dislocation: Avoiding a missed diagnosis. Am J Emerg Med 2000;18:189-191.
- Saupe N, White LM, Bleakney R, et al. Acute traumatic posterior shoulder dislocation: MR findings. Radiology 2008;248:185-193.
- Rowe CR, Zarins B. Chronic unreduced dislocations of the shoulder. J Bone Joint Surg Am 1982;64:494-505.
- Clough TM, Bale RS. Bilateral posterior shoulder dislocation: The importance of the axillary radiographic view. Eur J Emerg Med 2001;8:161-163.
- Samilson RL, Prieto V. Posterior dislocation of the shoulder in athletes. Clin Sports Med 1983;2:369-378.
- Atef A, El-Tantawy A, Gad H, et al. Prevalence of associated injuries after anterior shoulder dislocation: A prospective study. Int Orthop 2016;40:519-524.
- Gates JD, Knox JB. Axillary artery injuries secondary to anterior dislocation of the shoulder. J Trauma 1995;39:581-583.
- Perron AD, Ingerski MS, Brady WJ, et al. Acute complications associated with shoulder dislocation at an academic emergency department. J Emerg Med 2003;24:141-145.
- Sogut O, Yigit M, Karayel E, et al. Luxatio erecta humeri: Hands-up dislocation. J Emerg Med 2015;49:e53-e55.
- Begaz T, Mycyk MB. Luxatio erecta: Inferior humeral dislocation. J Emerg Med 2006;31:303-304.
- Davids JR, Talbott RD. Luxatio erecta humeri. A case report. Clin Orthop Relat Res 1990;(252):144-149.
- Matsumoto K, Ohara A, Yamanaka K, et al. Luxatio erecta (inferior dislocation of the shoulder): A report of two cases and a review of the literature. Injury Extra 2005;36:450-453.
- Mallon WJ, Bassett FH, 3rd, Goldner RD. Luxatio erecta: The inferior glenohumeral dislocation. J Orthop Trauma 1990;4:19-24.
- Grate Jr I. Luxatio erecta: A rarely seen, but often missed shoulder dislocation. Am J Emerg Med 2000;18:317-321.
- Brant-Zawadzki G, Herring A. Urgent interscalene brachial plexus block for management of traumatic luxatio erecta in the ED. Am J Emerg Med 2015;33:986.e3-986.e5.
- Abbasi S, Molaie H, Hafezimoghadam P, et al. Diagnostic accuracy of ultrasonographic examination in the management of shoulder dislocation in the emergency department. Ann Emerg Med 2013;62:170-175.
- Calvet J, Leroy M, Lacroix L. Luxations de l’épaule et lésions vasculaires. Journal de Chirurgie 1941;58:337-346.
- Sahajpal DT, Zuckerman JD. Chronic glenohumeral dislocation. J Am Acad Orthop Surg 2008;16:385-398.
- Jain NB, Wilcox RB, 3rd, Katz JN, Higgins LD. Clinical examination of the rotator cuff. PM R 2013;5:45-56.
- Roy JS, Braen C, Leblond J, et al. Diagnostic accuracy of ultrasonography, MRI and MR arthrography in the characterisation of rotator cuff disorders: A meta-analysis. Br J Sports Med 2015;49:1316-1328.
- Edmonds EW, Eisner EA, Kruk PG, et al. Diagnostic shortcomings of magnetic resonance arthrography to evaluate partial rotator cuff tears in adolescents. J Pediatr Orthop 2015;35:407-411.
- Lemos MJ. The evaluation and treatment of the injured acromioclavicular joint in athletes. Am J Sports Med 1998;26:137-144.
- Rockwood CA, Matsen FA. The Shoulder. Philadelphia: Saunders; 1990:413-414.
- Fukuda K, Craig EV, An KN, et al. Biomechanical study of the ligamentous system of the acromioclavicular joint. J Bone Joint Surg Am 1986;68:434-440.
- Saccomanno MF, DE Ieso C, Milano G. Acromioclavicular joint instability: Anatomy, biomechanics and evaluation. Joints 2014;2:87-92.
- Walton J, Mahajan S, Paxinos A, et al. Diagnostic values of tests for acromioclavicular joint pain. J Bone Joint Surg Am 2004;86-A:807-812.
- Bonz J, Tinloy B. Emergency department evaluation and treatment of the shoulder and humerus. Emerg Med Clin North Am 2015;33:297-310.
- Reid D, Polson K, Johnson L. Acromioclavicular joint separations grades I-III: A review of the literature and development of best practice guidelines. Sports Med 2012;42:681-696.
- Smith TO, Chester R, Pearse EO, et al. Operative versus non-operative management following rockwood grade III acromioclavicular separation: A meta-analysis of the current evidence base. J Orthop Traumatol 2011;12:19-27.
- Spencer EE, Jr. Treatment of grade III acromioclavicular joint injuries: A systematic review. Clin Orthop Relat Res 2007;455:38-44.
- Tauber M. Management of acute acromioclavicular joint dislocations: Current concepts. Arch Orthop Trauma Surg 2013;133:985-995.
- Van Tassel D, Owens BD, Pointer L, Moriatis Wolf J. Incidence of clavicle fractures in sports: Analysis of the NEISS Database. Int J Sports Med 2014;35:83-86.
MONOGRAPH: The shoulder is the most mobile joint in the human body. It's also a prime target for traumatic injury.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
