Abdominal Compartment Syndrome in the Emergency Department
January 1, 2017
Reprints
Author
Roselynn A. Gentles, MD, Emergency Medicine Physician, Cascade Medical, Leavenworth, WA.
Peer Reviewer
Steven M. Winograd, MD, FACEP, St. Barnabas Hospital, Clinical Assistant Professor, Emergency Medicine, New York College of Osteopathic Medicine, Old Westbury, NY.
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Schneider (editor), Dr. Stapczynski (editor), Ms. Fessler (nurse planner), Dr. Gentles (author), Dr. Winograd (peer reviewer), Ms. Mark (executive editor), and Ms. Coplin (executive editor) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Normal intra-abdominal pressure is around 0 mmHg, but normal may be up to 12 mmHg in an adult. Above that, complications may occur. In children, abdominal compartment syndrome may be seen with elevations above 10 mmHg.
- Massive fluid and blood resusucitation in the ED puts patients at risk for abdominal compartment syndrome later in their hospital course.
- Patients at risk for abdominal compartment syndrome include those with severe burns, trauma, pancreatitis, and with recent abdominal surgery.
- Complications seen with abdominal compartment syndrome include respiratory failure, hypotension, renal failure, mesentaric ischemia, and neurologic changes.
Introduction
Intra-abdominal hypertension and abdominal compartment syndrome are common occurrences in both intensive care unit (ICU) and surgical patients. Although not typically observed in the emergency department (ED) as a presenting chief complaint, critically ill patients will go on to develop either intra-abdominal hypertension or abdominal compartment syndrome. Out of this group, a portion may develope abdominal compartment syndrome as a direct result of their initial resuscitation and management. Therefore, it is important for emergency physicians to have a general working knowledge of abdominal compartment syndrome so they can identify risk factors and decrease morbidity and mortality for the duration of the patient’s hospitalization.
Abdominal compartment syndrome is characterized by organ dysfunction resulting from acute and sustained elevation in intra-abdominal pressure.1 Intra-abdominal pressure itself is defined as a steady-state pressure within the abdominal cavity.2 It is dependent on the patient’s respiratory status, whether inhaling or exhaling, in addition to the volume of the solid and hollow organs, whether they are empty or contain fecal matter. Intra-abdominal pressure also is influenced by any tumors or fluid within the abdominal cavity.3 Unfortunately, it is not possible to diagnose a patient with intra-abdominal pressure based on clinical examination.1 Instead, the diagnosis must be suspected in at-risk individuals, and then confirmed with bedside monitoring.
Normal expiratory intraabdominal pressure ranges from sub-atmospheric to 0 mmHg. The Abdominal Compartment Society (formerly the World Society of the Abdominal Compartment Syndrome [WSACS]) accepts the upper limit of intra-abdominal pressure to be 12 mmHg. Levels greater than 12 mmHg are classified as intra-abdominal hypertension. Intra-abdominal hypertension can be hyperacute, acute, subacute, or chronic.2 Hyperacute occurs during a very brief episode (such as during sneezing or laughing). Acute usually progresses over the course of several hours and generally is seen in trauma patients. Subacute occurs over the course of days and is more common in medical patients. Chronic intra-abdominal hypertension occurs over months or years, such as in patients who are pregnant, in cases of morbid obesity,2 or in patients with chronic ascites.4
It is further categorized into four grades.5
Grade I: Intra-abdominal pressure 12-15 mmHg
Grade II: Intra-abdominal pressure 16-20 mmHg
Grade III: Intra-abdominal pressure 21-25 mmHg
Grade IV: Intra-abdominal pressure > 25 mmHg
Special considerations are given to children, as an intra-abdominal pressure of 10-15 mmHg has been associated with organ damage in this population. In general, any intra-abdominal pressure higher than 10 mmHg with associated new organ dysfunction should be considered abdominal compartment syndrome in pediatric patients until proven otherwise.6
Abdominal compartment syndrome occurs when the pressure is elevated enough to cause blood flow circulation to decrease. Although this typically occurs when the intra-abdominal pressure is between 10 mmHg and 15 mmHg, abdominal compartment syndrome technically is not defined until the intra-abdominal pressure increases by more than 20-25 mmHg, there are signs of end organ dysfunction, and abdominal decompression has a beneficial effect.7
History
Although the term abdominal compartment syndrome was not introduced until 1982 by Kron et al, scientists have been making connections between abdominal distension and multi-organ system failure for more than a century. In 1863, Marey noticed that patients who seemed to have an increase in abdominal pressure also went on to develop pulmonary collapse. Later, Henricus used animal models to demonstrate the incidence in respiratory failure in animals with intra-abdominal pressures between 27-46 mmHg. In 1911, Emerson changed the focus from a pulmonary standpoint to that of a cardiovascular one after he began correlating an increased incidence of cardiovascular death in cats, dogs, and rabbits that had an elevated intra-abdominal pressure.8
In 1947, research expanded to human subjects. In 1948, Gross developed the staged abdominal repair (StAR) technique for abdominal wall closure, which is now one of the primary surgical interventions used in abdominal compartment syndrome. It wasn’t for another 32 years until the connection was made by Kron, Harman, and Richards regarding the effects of intra-abdominal hypertension on the renal system as well, which then led to the term “abdominal compartment syndrome.”5
In 2004, WSACS convened a consensus conference consisting of a group of surgeons and intensivists from international hospitals who convened to establish a set of guidelines for identifying, monitoring, and treating abdominal compartment syndrome.
Causes
Abdominal compartment syndrome occurs in a wide variety of patient spectrums. These include trauma, whether direct or indirect to the abdomen, as well as other medical conditions, such as pancreatitis or ascites.
In trauma, both primary and secondary abdominal compartment syndrome can occur. Primary abdominal compartment syndrome involves direct injury to the abdomen, such as a motorcycle accident causing a splenic rupture or a penetrating wound that then requires immediate laparotomy.8,9 Postsurgical complications and ruptured abdominal aortic aneurysms, in addition to acute pancreatitis, also are considered to be examples of primary abdominal compartment syndrome.2,5
Secondary abdominal compartment syndrome develops in any type of trauma or medical patient who requires massive resuscitation, even in those who do not suffer a direct injury to the abdominal compartment. It is caused by elevated pressure and organ dysfunction due to third space edema and resuscitation.8 Examples include head wounds, burns, or septic shock.2,9,10,11 This usually occurs later in the patient’s hospital stay; however, it can present within the first 12 hours after injury, especially in patients who have received massive transfusions. As such, abdominal compartment syndrome should be considered if organ dysfunction develops after initial resuscitation.9
Non-traumatic risk factors for developing abdominal compartment syndrome include acute pancreatitis, ruptured abdominal aortic aneurism, intra-abdominal abscess, and intestinal obstructions.8
A third type of abdominal compartment syndrome is termed recurrent abdominal compartment syndrome, in which the patient has already had a recovery period from the initial insult, but then suffers a relapse due to secondary insults.2,8
Aside from trauma, there are two conditions that present to the ED for resuscitation and go on to develop abdominal compartment syndrome, often as a direct result of emergency medicine management. These are severe acute pancreatitis and burn patients.
About 60-80% of patients presenting to the ED with severe acute pancreatitis (SAP) will have intra-abdominal hypertension.12,13 Approximately 12-30% of these patients then will go on to develop abdominal compartment syndrome.14,15 In acute pancreatitis, most reports show that intra-abdominal hypertension develops within the first 3-5 days after hospital admission.13
There are three main contributors to the risk of intra-abdominal hypertension or abdominal compartment syndrome in these patients. The initial insult is usually iatrogenic and is a direct result of the early aggressive fluid resuscitation with crystalloid boluses. In patients with severe pancreatitis, this compensates for central hypovolemia secondary to third spacing.13 Retroperitoneal edema, ascites, and fluid collections are formed.12 In addition, there usually is some local edema from the pancreatic inflammation as well as an accompanying ileus; however, these often are minimal insults compared the fluid boluses received. Once intra-abdominal hypertension occurs, the cycle continues, as the pancreatic perfusion decreases, which leads to pancreatic necrosis.13
Burns are a special category in secondary intra-abdominal hypertension and abdominal compartment syndrome due to capillary leak (third space fluid) and large volume fluid resuscitation.16 Bowel edema and fluid translocation are worsened by fluid translocation from the intra-abdominal pressure-induced venous hypertension. This leads to intra-abdominal hypertension that usually occurs within the first 48 hours after injury. Abdominal compartment syndrome usually develops later, particularly with septic episodes.17 The reported incidence of abdominal compartment syndrome varies from 30% to 80% of patients.16 This wide range is because patients who have a higher total body surface area burned tend to have a higher incidence of abdominal compartment syndrome than those with less extensive injuries.17 Burn patients are at risk for developing sepsis, which provokes a cascading inflammatory response. Inflammatory mediators, angiotensin II, and vasopressin are released into the circulatory system, causing an increase in mesenteric vascular resistance and ischemic enterocolitis. In addition, if the burns affect the abdomen, they can create a circumferential burn, which leads to decreased abdominal wall compliance as well as extrinsic compression of the abdomen.5
Risk Factors
Although abdominal compartment syndrome usually is due to trauma or abdominal infection, a variety of other components influence the development of abdominal compartment syndrome during the hospital course. Holodinsky et al performed a systematic review of 14 different studies and identified 38 different risk factors for both abdominal compartment syndrome and intra-abdominal hypertension.18 They then grouped these into three themes and eight subthemes. The three themes include: baseline patient characteristics, systemic physiology, and fluid resuscitation.
Patient characteristics that seem to increase the likelihood of developing abdominal compartment syndrome/intra-abdominal hypertension include obesity and age. Morbid obesity can be associated with chronic intra-abdominal pressure elevations.19 Although the exact pathogenesis of elevated intra-abdominal pressure among patients with obesity is not known currently, Lambert et al hypothesized that this association may be due to a direct mass effect of intra-abdominal adipose tissue.17,20 Thus, the “normal” values of intra-abdominal pressure in obese patients should be considered to rest between 7 and 14 mmHg.21 It has been suggested that complications found in obese patients, such as pseudotumor cerebri, hypertension, and gastroesophageal reflux disease (GERD), all could be attributed to an obesity-induced intra-abdominal hypertension syndrome.17
Diagnoses that later present with abdominal compartment syndrome/intra-abdominal hypertension as a complication include sepsis, intra-abdominal infection, abdominal surgery, SAP, cirrhosis, gastrointestinal bleeding, ileus, acidemia, and hypotension.
Large-volume crystalloid resuscitation (> 3L) is the final category. This seems to present more often in patients with trauma as well as in patients with acute pancreatitis. In addition, there was some evidence that poly-transfusion with packed red blood cells also may cause abdominal compartment syndrome, although this may be mitigated by using a massive transfusion protocol that includes a larger ratio of plasma and platelets.22
The Abdominal Compartment Society suggests that any patient with two or more of the risk factors is at a high enough risk to warrant intra-abdominal pressure monitoring.4,23 Risk factors include:
Diminished Abdominal Wall Compliance
- Acute respiratory failure, especially with elevated intrathoracic pressure;
- Abdominal surgery with subjectively tight primary closure;
- Major trauma/burns;
- Prone positioning, head of bed elevated > 30°;
- High body mass index, central obesity;
Increased Intra-luminal Contents
- Gastroparesis;
- Ileus;
- Colonic pseudo-obstruction;
- Volvulus;
Increased Abdominal Contents
- Hemoperitoneum/pneumoperitoneum or intra-peritoneal fluid collections;
- Intra-abdominal abscesses or tumors;
- Liver dysfunction/cirrhosis with ascites;
- Peritoneal dialysis;
Capillary Leak/Fluid Resuscitation
- Acidosis (pH < 7.2);
- Hypotension;
- Hypothermia (core temperature < 33 °C);
- Damage control laparotomy;
- Increased APACHE-II or SOFA score;
- Polytransfusion (> 10 units of blood/24 hours);
- Coagulopathy (platelets < 55,000 mm3), prothrombin time > 15s, partial thromboplastin time > 2 times normal, or INR > 1.5;
- Massive fluid resuscitation (> 5 L/24 hours) or positive fluid balance;
- Pancreatitis;
- Oliguria;
- Sepsis;
- Major trauma/burn;
- Mechanical ventilation.4,23
Complications
Abdominal compartment syndrome has an effect on various areas of the body, not specifically the abdominal area alone. Abdominal compartment syndrome affects the lungs and cardiac output, leads to renal failure, causes mesenteric ischemia, and also has neurologic consequences.
Pleural
The increased intra-abdominal pressure causes passive elevation of the diaphragm, which allows transmission of the increased intra-abdominal pressure to the pleural space. This reduces static and dynamic lung compliance, which in turn causes tidal volume, lung compliance, functional residual capacity, and residual volume to decrease. Peak inspiratory pressure increases at levels as low as 15 mmHg, while extrinsic compression of the pulmonary parenchyma occurs between 16 and 30 mmHg.24 Alveolar atelectasis occurs, leading to decreased oxygen transport across the pulmonary membrane. Pulmonary vasculature suffers from the increased pressure, causing a decrease in capillary blood flow as well as arterial hypoxemia. Hypoventilation, hypoxemia, and hypercapnea result.2,6,8,25
Cardiac
Again, with increased intra-abdominal pressure present, the inferior vena cava (IVC) and aorta as well as the splanchic circulation become compressed, leading to a decrease in venous return as well as an increase in afterload (particularly through the pulmonary vasculature). Such reductions have been demonstrated to occur at an intra-abdominal pressure of 10 mmHg.26 With the cephalad displacement of the diaphragm, the intrathoracic pressure increases, resulting in reduced ventricular compliance and cardiac contractility, and most concerning in the ED, a falsely elevated central venous pressure (CVP) and pulmonary artery occlusion pressure (PaOP).2,25 This is because both the PaOP and CVP are measured relative to atmospheric pressure and are actually the sum of both intravascular pressure and intrathoracic pressure. In the presence of intra-abdominal hypertension-induced elevations in intrathoracic pressure, PaOP and CVP tend to be erroneously elevated and no longer reflective of true intravascular volume status.5
Renal
Pre-renal azotemia occurs as perfusion to the kidneys decreases in the setting of increased interstitial and outflow pressure as well as the decrease in cardiac output. Because of the direct pressure on the kidneys from abdominal compartment syndrome, renal derangement occurs. Oliguria has been demonstrated to present at an intra-abdominal pressure of 15 mmHg, while anuria occurs at 30 mmHg.27,28 The renal veins, cortical arterioles, and renal parenchyma are all compressed. As the renal veins are compressed, the blood is shunted away from the renal cortex and functioning glomeruli, causing an increase in renal vascular resistance and reduced glomerular filtration rate. Glomerular and tubular dysfunction occurs. Renin, aldosterone, and antidiuretic hormone levels all increase, which serves to increase the retention of sodium and water, as well as further raising the renal vascular resistance and compounding the original problem. Most disturbing is that fluid resuscitation, loop diuretics, or dopaminergic agents do not seem to affect the pre-renal azotemia, as the glomerular filtration is directly dependent on the amount of pressure present in the abdomen.2,23 Only prompt identification and reversal of the triggering cause of the abdominal compartment syndrome seems to have any effect.2,5,8,25
Mesentery/Gastrointestinal
Increased intra-abdominal pressure leads to direct compression on the splanchic circulation. Hepatic and portal blood flow become compromised at an intra-abdominal pressure of 10 mmHg.29 With the decrease in perfusion, hepatic microcirculatory blood flow is decreased, resulting in a reduction in hepatic mitochondrial function and production of energy substrates. Lactic acid clearance by the liver also is compromised,5 and the liver fails to make clotting factors or protein synthesis.4 Mesenteric and intestinal blood flow reduction occurs at an intra-abdominal pressure of 20 mmHg.29 With the decrease in blood flow to the intestinal mucosa, cellular hypoxia and anaerobic cell metabolism occur, increasing the amount of lactic acid formed as well as promoting translocation of bacteria from the gut to mesenteric lymph noses. This may be one of the primary mechanisms behind the “two hit hypothesis.” The idea is that the body has a generalized inflammatory response to the original insult (infection, trauma, burns, etc). However, any second “hit” (for example, sepsis, surgery, bacterial translocation) causes a massive systemic response, which in turn causes a release of cytokines, the formation of free oxygen radicals, and decreased cellular production of adenosine triphosphate. The cytokines promote vasodilatation and increase in capillary permeability. With the decrease of ATP, the sodium-potassium pump fails, allowing sodium and water to leak back into the cells. The cells swell, the membranes lose their integrity, and intracellular contents are poured into the extracellular environment, leading to more inflammation and tissue edema.30 In addition, with the portal venous system being affected at a lower pressure, the blood that normally would be draining from the abdomen and returning to the cardiac system instead causes visceral edema and engorgement of the small intestines and results in even higher intra-abdominal pressures.5,8
Neurologic
Two mechanisms currently are proposed to describe the concomitant elevated ICP that can occur in primary or secondary abdominal compartment syndrome. First, because of the increased abdominal pressure and resulting collapse of the IVC, the lumbar venous plexus blood flow decreases. This causes a drop in the absorption of the cerebrospinal fluid in the lumbar cisterna region, or an outflow obstruction of the cerebrospinal fluid. Secondly, because of the increase in intrathoracic pressure, the jugular venous pressure increases, which decreases venous outflow and causes increased intracranial blood volume and increased ICP.2,31,32
Abdominal Wall
Although the abdominal wall itself is not considered a vital organ system, abdominal hypertension does tend to influence its compliance, and thus, further worsen intra-abdominal pressure to abdominal compartment syndrome. Intraperitoneal fluid and visceral edema both distend the abdominal wall and cause decreased compliance. The abdominal wall can become edematous secondary to shock or fluid resuscitations. Blood flow decreases, causing ischemia of the wall, leading to impaired wound healing and possible necrotizing fasciitis.5 Burn patients in particular tend to develop abdominal compartment syndrome, with a major contributor being abdominal wall and decreased compliance causing increased intra-abdominal pressure. These patients are already at risk, as they usually receive massive fluid resuscitation and may develop bowel edema and fulminant sepsis.33
Pregnancy
Intra-abdominal pressures during pregnancy increase, especially during the second and third trimesters, because of the gradual expansion of the fetus and the uterus. Hormonal influences allow the abdominal wall to be stretched slowly, increasing its compliance. In addition, collateral blood flow from the lower extremities and the abdomen develops, nullifying the effects on intra-abdominal pressure of the fetus during pregnancy. Manu et al have suggested that preeclampsia and eclampsia may be manifestations of intra-abdominal hypertension and abdominal compartment syndrome, respectively. A current theory is that an intra-abdominal pressure reduction of renal blood flow would cause renin-angiotensin activation resulting in systemic hypertension. However, bladder pressures are not measured routinely in these patients. In addition, measurements often are considered to be erroneous, as the patient needs to be fully supine to avoid the fetal pressure on the bladder during measurements. This can cause the patient to develop severe hypotension and is not recommended in ill patients.3 Eclampsia itself is alleviated after delivery of the fetus and reduction of the abdominal compartment pressure.34 Because of the lack of specific studies, this hypothesis has yet to be verified.
Measurement
Clinical examination alone has been shown to be highly unreliable in diagnosing abdominal compartment syndrome.1,2,6,23 As such, the Abdominal Compartment Society currently suggests serial measurements of intra-abdominal pressure in patients presenting with risk factors for developing abdominal compartment syndrome.5 During the past 10 years, a variety of methods have been used to measure the intra-abdominal pressure. These include the currently recommended transurethral intravesical technique in addition to transesophageal intragastrical, transvaginal intrauterine, transanal intrarectal, and percutaneous transfemoral to the subdiaphragmatic inferior vena.23,35 intra-abdominal pressure also can be measured via an intraperitoneal catheter installed for ascites drainage or peritoneal dialysis.2
To measure the intra-abdominal pressure via the intra-vesicular route, a pressure transducer and a Foley catheter are connected via a three-way valve. The patient is catheterized, and urine is drained. The catheter then is clamped, and the patient is laid supine. The transducer then is zeroed at the iliac crest in the mid-axillary line. Approximately 15-25 mL (as long as the chosen amount is consistent and does not exceed 25 mL) of saline then is instilled into the bladder, and the pressure is noted after 30-60 seconds at the end of patient exhalation. Most protocols advise doing the measurements every 4-6 hours.4
The Abdominal Compartment Society currently recommends the measurement of intra-abdominal pressure via the bladder in the supine position at end-expiration, ensuring that the abdominal contractions are absent and with the transducer zeroed at the level of the mid-axillary line.23,35 Because of concerns over urinary tract infections and resulting sepsis from the aforementioned recommendation, Tilev examined an intragastric approach to measure abdominal pressure.36 Their study group consisted of 30 patients not at risk for intra-abdominal hypertension and compared pressures intragastrically to those intravesicullarly. They found that there was no statistically significant difference between the measurement results after introduction of 20 mL, 50 mL, or 100 mL of solution into the stomach. However, any fluid volumes greater than 150 mL began to give them skewed results. Complications resulted from air bubbles in the tubing as well as residual material in the stomach clogging the nasogastric tube. Nevertheless, intragastric measurement is an alternative option, particularly when there is transurethral or pelvic injury.
At this time, there are no commercial intra-abdominal pressure monitoring kits available specifically designed for use in children; however, Waele et al suggest using the Abviser Neonate adaptor (ConcaTec Medical) which accommodates the use of feeding tubes as improvised urethral catheters in smaller infants. The reference standard for intermittent intra-abdominal pressure measurement in children is via the bladder using 1 mL/kg as an instillation volume, with minimal instillation volume of 3 mL and a maximum installation volume of 25 mL of sterile saline.
Imaging
Because abdominal compartment syndrome is a diagnosis made based on risk stratification and verification by intravesicular pressure measurement, there are very few reports on imaging leading to the detection of abdominal compartment syndrome.37 However, some CT scans are performed sequentially, tracking the original disease progression. These images can be used to note the development of signs that may suggest or contribute to abdominal compartment syndrome and prompt follow-up if such has not already been detected. These include an elevated diaphragm, increase in ascites, hemoperitoneum, flattened IVC, flattened renal veins, and increased bowel enhancement. The most common findings in a study by Patel et al were an elevated diaphragm, increased bowel enhancement, and flattened renal veins. Flattened IVC and increased bowel enhancement also may be seen in septic shock; however, in patients without hypotension, these may be signs of abdominal compartment syndrome. CT is neither sensitive nor specific, so this modality cannot be used solely to determine if the patient has abdominal compartment syndrome, or the disease severity. In patients with risk factors and for whom the CT scan shows localized fluid collections, hemoperitoneum, or ascites, then abdominal compartment syndrome should be suspected.
Cavaliere et al investigated the use of ultrasonography in the evaluation of abdominal compartment syndrome by setting up a simulation of increased intra-abdominal pressure in healthy volunteers.38 A pelvic stabilizer around the waist was used to create external compression. Intra-abdominal pressure was measured via the gastric route. Notable findings were: 1) the IVC was compressed with significant decrease of both anteroposterior and lateral diameters and deformed (the deformity was worsened by use of noninvasive ventilation); 2) the portal vein was compressed; 3) blood velocities did not change in the IVC, portal vein, right suprahepatic vein, or right external iliac vein.
Treatment
The primary goal of managing abdominal compartment syndrome is to alleviate the pressure in the abdomen. This can be done both medically and surgically, with medical management being the initial intervention, provided the patient is not in danger of immediate deterioration in which case emergent surgery is indicated.
The Abdominal Compartment Society has published guidelines for the management of abdominal compartment syndrome. (See Figure 1.)
Figure 1. Intra-abdominal Hypertension (IAH)/Abdominal Compartment Syndrome (ACS) Management Algorithm
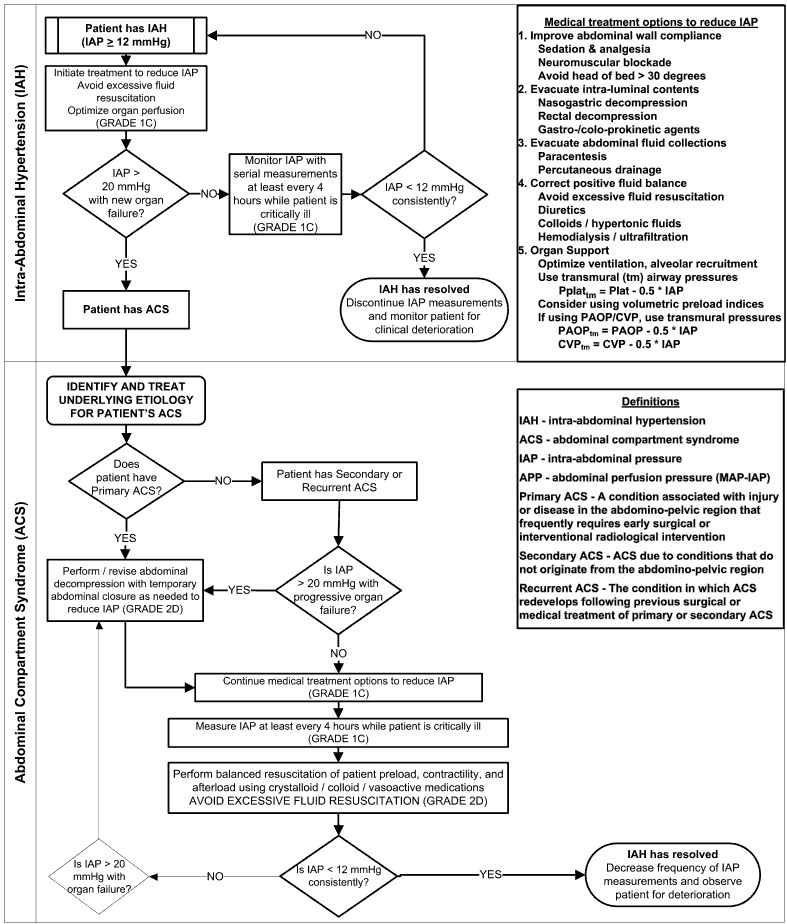
© copyright by WSACS, the Abdominal Compartment Society (www.wsacs.org); adapted from Kirkpatrick et al. Intra-abdominal hypertension and the abdominal compartment syndrome: Updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Medicine 2013, Volume 39, Issue 7, pp 1190–1206.
Medical management currently is divided into five separate treatment parts. These include: evacuate intraluminal contents, evacuate intra-abdominal space-occupying lesions, improve abdominal wall compliance, optimize fluid administration, and optimize systemic/regional perfusion.9,23
Evacuation of intraluminal contents focuses on removing as much pressure from the abdomen through the available orifices naturally present. These include: insertion of a nasogastric tube for gaseous or liquid distension; insertion of a rectal tube or rectal disimpaction; prokinetics such as neostigmine, metoclopramide, and erythromycin; enemas, preferably after perforation has been excluded; and colonic decompression.9,23,39
Evacuation of intra-abdominal space-occupying lesions involves management of the fluid around the organs. Percutaneous catheter decompression is particularly effective in patients with ascites secondary to liver cirrhosis, sepsis, pancreatitis, and in patients suffering from the massive fluid resuscitation and capillary leak that accompanies burn resuscitation.9,40 Sun et al performed a series of bedside percutaneous drainage techniques and noted that to decrease intra-abdominal pressure from 29 to 14 mmHg, an average of 1,800 mL of ascites needed to be removed.41
Improvement of abdominal wall compliance focuses on minimizing the extrinsic compression of the abdomen through sedation, neuromuscular blockade, and body positioning. Currently, there is very limited information on how sedation affects the abdominal compartment pressure; however, given that patients in pain tend to have more muscle contractions/tensing, it is logical to assume that by causing their trunk musculature to relax, the pressure in the abdomen should decrease. Therefore, the Abdominal Compartment Society suggests sedation, although no actual recommendations on the type of sedation exist. Two studies involving propofol vs. dexmedetomidine,42 as well as epidural analgesia vs. opioids,43 currently are available for review. In the former, dexmedetomidine appeared to drop the intra-abdominal pressure much more significantly compared to propofol. In the latter, the epidural analgesia was shown to decrease the intra-abdominal pressure, while the opioid group did not have a change in outcome. In terms of neuromuscular blocking agents, routine use in the ICU is not suggested; however, a single bolus may be given to help reduce intra-abdominal pressure in patients waiting for surgical decompression or as a temporary adjuvant when used with other medical interventions.9 In burn patients with circumferential burns involving the abdominal wall musculature, escharotomies are suggested prior to open decompressive laparotomy.3,40
Although not directly having an effect on the abdomen, intra-abdominal pressure has been noted to slightly increase with elevation of the head of the bed. Current ICU recommendations advise to keep the head of the bed elevated to 30 degrees to prevent aspiration pneumonia. This angle has been shown in observational studies to increase the pressure in the abdomen; therefore, it is suggested that in a patient who appears to be developing abdominal compartment syndrome, lowering the head of the bed to a supine position would be a good initial intervention.9 (See Figure 2.)
Figure 2. Intra-abdominal Hypertension (IAH)/Abdominal Compartment Syndrome (ACS) Management Algorithm

© copyright by WSACS, the Abdominal Compartment Society (www.wsacs.org); adapted from Kirkpatrick et al. Intra-abdominal hypertension and the abdominal compartment syndrome: Updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Medicine 2013, Volume 39, Issue 7, pp 1190–1206.
Fifteen years ago, abdominal compartment syndrome was suggested to be due to under-resuscitation of crystalloids during the initial ED evaluation.25 Current literature suggests that we may have overcompensated in giving too much crystalloid, causing volume overload of critical patients, which then results in pulmonary edema and abdominal compartment syndrome, in addition to other complications. Zhao et al found that patients who were resuscitated with only normal saline and not colloid products had a higher intra-abdominal pressure and developed abdominal compartment syndrome more often than patients who received colloids or a mixture thereof.44 Unfortunately, there is not enough evidence at this time to propose whether a patient should be kept balance neutral or in a negative balance, although several studies have found that patients who had a positive fluid balance suffered a worse outcome, including abdominal compartment syndrome.15 As there is not enough evidence promoting negative or neutral fluid balance, Abdominal Compartment Society currently suggests using a protocol to avoid a positive cumulative balance after initial fluid resuscitation has occurred. Further, as renal dysfunction seems to be a side effect of the continuous pressure in the abdomen, and diuretics seem to have little or no effect in increasing renal perfusion, the Abdominal Compartment Society currently has no recommendations on diuretic use.23 It does suggest using an enhanced ratio of plasma/packed red blood cells for resuscitation of massive hemorrhage instead of copious amounts of crystalloids.
Current burn recommendations advise continuing with the Parkland formula, now called the Consensus Formula. Fluid restriction does not appear to improve the outcome in burn patients.40 Oda et al demonstrated that in patients with severe burn injury, hypertonic lactated saline resuscitation could reduce the risk of secondary abdominal compartment syndrome; however, these findings have not yet been fully researched and thus are not incorporated into the most recent Abdominal Compartment Society guidelines.45 O’Mara et al showed that plasma resuscitated patients maintained an intra-abdominal pressure below 20 mmHg, thus avoiding complications associated with abdominal compartment syndrome, such as renal failure or respiratory compromise.3,46 In general, intra-abdominal hypertension/abdominal compartment syndrome should be suspected in all burn patients with a total body surface area greater than 20%, with intra-abdominal pressure measured every 2-4 hours during the resuscitation period.3
Extracorporeal techniques, such as hemofiltration, are more effective in rapidly moving excess fluid than diuretics. There are no universally accepted levels of urea, creatinine, potassium, or pH at which to start therapy. Initiation of renal replacement therapy should be prompted more by the rate of change of renal parameters and by the patient’s overall condition than by arbitrary levels. Hemofiltration itself involves blood being pumped through an extracorporeal system that incorporates a semi-permeable membrane. Hydrostatic pressure drives plasma and water across the membrane. This method differs from hemodialysis in that hydrostatic pressure, as opposed to the mechanism of diffusion, is used. The excess fluid then is discarded.47
New medical treatment studies include the use of theophylline, tissue plasminogen activator (tPA), and octreotide. Bodnar et al found increased serum adenosine and interleukin 10 to be laboratory markers that are elevated in abdominal compartment syndrome.48 They then hypothesized that theophylline would help reduce the intra-abdominal pressure by counteracting the binding of the adenosine to the adenosine receptors. This, in turn, would improve cardiac contractility and renal function as well as perfusion. They then compared the outcome of two groups of patients with intra-abdominal hypertension: those who received theophylline in addition to the Abdominal Compartment Society recommendations, and those who received the current standard medical treatment. The group that received theophylline had a mortality score of 0% and had a decrease of intra-abdominal pressure by 8.7 mmHg. Tissue plasminogen activator currently is used to help dissolve clots in the body, although it is usually reserved for massive pulmonary emboli, ST elevation myocardial infarction, or ischemic strokes. Horer et al used tPA to evacuate retroperitoneal hematomas and thereby decrease the intra-abdominal pressure.49
Current Abdominal Compartment Society guidelines recommend decompressive laparotomy in cases of overt abdominal compartment syndrome. However, as the mortality rate is close to 50% even after surgery, current suggestions are to medically manage intra-abdominal hypertension as much as possible before proceeding to surgery. In addition, the Abdominal Compartment Society advises against taking patients with a septic abdomen to the operating room unless the risk of abdominal compartment syndrome is high enough that it outweighs the risks of operation.23
A variety of surgical techniques currently are used for surgical decompression. In general, nonoperative techniques should be attempted primarily, but if improvement of the abdominal compartment syndrome is not noted, then surgery within five days of abdominal compartment syndrome onset is suggested.13 Mentula et al reported a 100% mortality rate in patients who had decompressive surgery done more than five days after development of abdominal compartment syndrome. In this case, the organ damage is most likely irreversible; however, there is no definitive guideline for when the procedure should be conducted.50
The most common surgical method is a midline laparostomy in which a vertical incision extending from the xiphoid to pubis is made through all the abdominal wall layers. Alternatively, a transverse bilaterally extended incision a few centimeters below the costal margins also can be used.51 Other techniques include a bilateral subcostal incision as well as a subcutaneous linea alba fasciotomy in which three horizontal incisions are made through the skin and fascia, but does not penetrate the peritoneum. This may serve as a temporary measure to alleviate the pressure and allow time for other medical management techniques to have an effect on the compartment syndrome; however, if the intra-abdominal hypertension continues to rise, a full laparotomy using one of the above techniques is suggested.6 In all of these cases, intra-abdominal pressure still needs to be monitored, as abdominal compartment syndrome still can develop even with an open abdomen if the original cause, such as bleeding, is not controlled.
Current management guidelines suggest closure of the abdomen within two weeks, provided the intra-abdominal hypertension has been controlled.6
Conclusion
Abdominal compartment syndrome is recognized more commonly as an important contributor to morbidity and mortality in critical care patients. These cases arise in trauma, burn, post-surgical, and septic patients. Emergency medicine providers need to be aware that while immediate care often does require use of crystalloids, one must take into account further complications that may evolve with over-hydration. In addition, patients may present via transfer from outlying hospitals having already received multiple boluses of fluids with the initial stages of abdominal compartment syndrome. Clinical examination alone is not sufficient to detect abdominal compartment syndrome, but new-onset organ failure in a patient with risk factors should lead to consideration of a larger, more complicated, and emergent picture.
REFERENCES
- Kirkpatrick A, Brenneman F, McLean R, et al. Is clinical examination an accurate indicator of raised intra-abdominal pressure in critically injured patients? Can J Surg 2000;43:207-211.
- Papavramidis T, Marinis AD, Pliakos I, et al. Abdominal compartment syndrome - Intra-abdominal hypertension: Defining, diagnosing, and managing. J Emerg Trauma Shock 2011;4:279-291.
- Malbrain ML, De laet I, Cheatham M. Consensus conference definitions and recommendations on intra-abdominal hypertension (IAH) and the abdominal compartment syndrome (ACS) — the long road to the final publications, how did we get there? Acta Clin Belg 2007;62 Suppl: 44-59.
- Berry N, Fletcher S. Abdominal compartment syndrome. Continuing Education in Anesthesia, Critical Care & Pain 2012;12(3).
- Cheatham ML. Abdominal compartment syndrome: Pathophysiology and definitions. Scand J Trauma, Resusc Emerg Med 2009;17:10.
- De Waele JJ, Ejike JC, Leppaniemi A, et al. Intra-abdominal hypertension and abdominal compartment syndrome in pancreatitis, paediatrics, and trauma. Anesthesiol Intensive Ther 2015;47:219-227.
- Cheatham ML, White MW, Sagraves SG, et al. Abdominal perfusion pressure: A superior parameter in the assessment of intra-abdominal hypertension. J Trauma 2000;49:621-626.
- Mohapatra B. Abdominal compartment syndrome. Indian J Crit Care Med January-March 2004;8:26-32.
- DeKeulenaer B, Regli A, De Laet I, et al. What’s new in medical management strategies for raised intra-abdominal pressure: Evacuating intra-abdominal contents, improving abdominal wall compliance, pharmacotherapy, and continuous negative extra-abdominal pressure. Anaesthesiol Intensive Ther 2015;47:54-62.
- Ball C, Kirkpatrick AW, McBeth P. The secondary abdominal compartment syndrome: Not just another post-traumatic complication. Can J Surg 2008;51:399-405.
- Kirkpatrick AW, Balogh Z, Ball CG, et al. The secondary abdominal compartment syndrome: Iatrogenic or unavoidable? J Am Coll Surg 2006;202:668-679.
- De Waele JJ, Hoste E, Blot SI, et al. Intra-abdominal hypertension in patients with severe acute pancreatitis. Crit Care 2005;9:R452-457.
- De Waele JJ. Management of abdominal compartment syndrome in acute pancreatitis. Pancreapedia: Exocrine Pancreas Knowledge Base 2015. DOI: 10.3998/panc.2015.29.
- Ke L, Tong ZH, Ni HB, et al. The effect of intra-abdominal hypertension incorporating severe acute pancreatitis in a porcine model. PLoS One 2012;7:e33125.
- Ke L, Ni HB, Sun JK, et al. Risk factors and outcome of intra-abdominal hypertension in patients with severe acute pancreatitis. World J Surg 2012;36:171-178.
- Wise R, Jacobs J, Pilate S, et al. Incidence and prognosis of intra-abdominal hypertension and abdominal compartment syndrome in severely burned patients: Pilot study and review of the literature. Anaesthesiol Intensive Ther 2016;48:95-109.
- Malbrain ML, De Keulenaer BL, Oda J, et al. Intra-abdominal hypertension and abdominal compartment syndrome in burns, obesity, pregnancy, and general medicine. Anaesthesiol Intensive Ther 2015;47:228-240.
- Holodinsky JK, Roberts DJ, Bail CG, et al. Risk factors for intra-abdominal hypertension and abdominal compartment syndrome among adult intensive care unit patients: A systematic review and meta-analysis. Crit Care 2013;17:R249.
- Papavramidis TS, Duros V, Michalopoulos A, et al. Intra-abdominal pressure alterations after large pancreatic pseudocyst transcutaeous drainage. BMC Gastroenterol 2009;9:42.
- Lambert DM, Marceau S, Forse RA. Intra-abdominal pressure in the morbidly obese. Obes Surg 2005;15:1225-1232.
- Malbrain ML, Cheatham ML, Kirkpatrick A, et al. Results from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. Definitions. Intensive Care Med 2006;32:1722-1732.
- Cotton BA, Au BK, Nunez TC, et al. Predefined massive transfusion protocols are associated with a reduction in organ failure and postinjury complications. J Trauma 2009;66:41-49.
- Kirkpatrick AW, Roberts DJ, De Waele J, et al. Intra-abdominal hypertension and the abdominal compartment syndrome: Updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 2013;39:1190-1206.
- Simon RJ, Friedlander MH, Ivatury RR, et al. Hemorrhage lowers the threshold for intra-abdominal hypertension-induced pulmonary dysfunction. J Trauma 1997;42:398-403.
- De Backer D. Abdominal compartment syndrome. Crit Care 1999;3:R103-R104.
- Richardson JD, Trinkle JK. Hemodynamic and respiratory alterations with increased intra-abdominal pressure. J Surg Res 1976;20:401-404.
- Richards WO, Scovill W, Shin B, Reed W. Acute renal failure associated with increased intra-abdominal pressure. Ann Surg 1983;197:183-187.
- Shenasky JH 2nd. The renal hemodynamic and functional effects of external counterpressure. Surg Gynecol Obstet 1972;134:253-258.
- Diebel LN, Dulchavsky SA, Brown WJ. Splanchnic ischemia and bacterial translocation in the abdominal compartment syndrome. J Trauma 1997;43:852-855.
- Walker J, Criddle LM. Pathophysiology and management of abdominal compartment syndrome. Am J Critl Care 2003;12:367-371.
- Bloomfield GL, Ridings PC, Blocher CR, et al. Effects of increased intra-abdominal pressure upon intracranial and cerebral perfusion pressure before and after volume expansion. J Trauma 1996;40:936-941.
- Sugerman JH, DeMaria EJ, Felton WL 3rd, et al. Increased intra-abdominal pressureand cardiac filling pressure in obesity-associated pseudoumor cerebri. Neurology 1997;49:507-511.
- Kollias S, Stampolidis N, Kourakos P, et al. Abdominal compartment syndrome (ACS) in a severely burned patient. Ann Burns Fire Disasters 2015;28:5-8.
- Richter CE, Saber S, Thung SF. Eclampsia complicated by abdominal compartment syndrome. Am J Perinatol 2009;26:
751-753. - Rooban N, Regli A, Davis WA, De Keulanaer BL. Comparing intra-abdominal pressures in different body positions via a urinary catheter and nasogatric tube: A pilot study. Ann Intensive Care 2012;2 Suppl 1:S11.
- Tilev E. The influence of gastric volume in the measurement of intra-gastric pressure. Trakia Journal of Sciences 2014;12:214-218.
- Patel A, Lall CG, Jennings SG, Sandrasegaran K. Abdominal compartment syndrome. AJR Am J Roentgenol 2007;189:1037-1043.
- Cavaliere F, Cina A, Biasucci D, et al. Sonographic assessment of abdominal vein dimension and hemodynamic changes induced in human volunteers by a model of abdominal hypertension. Crit Care Med 2011;39:344-348.
- Luckianow G, Ellis M, Governale D, Kaplan L. Abdominal compartment syndrome: Risk factors, diagnosis, and current therapy. Critical Care Res Pract 2012;2012:908169.
- Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368-1377.
- Sun ZX, Huang HR, Zhou H. Indwelling catheter and conservative measures in the treatment of abdominal compartment syndrome in fulminant acute pancreatitis. World J Gastroenterol 2006;12:5068-5070.
- Tasdogan M, Memis D, Sut N, Yuksel M. Results of a pilot study on the effects of propofol and dexmedetomidine on inflammatory responses and intraabdominal pressure in severe sepsis. J Clin Anesth 2009;21:394-400.
- Hakobyan RV, Mkhoyan GG. Epidural analgesia decreases intraabdominal pressure in postoperative patients with primary intra-abdominal hypertension. Acta Clin Belg 2008;63:86-92.
- Zhao G, Zhang JG, Wu HS, et al. Effects of different resuscitation fluid on severe acute pancreatitis. World J Gastroenterol 2013;19:2044-2052.
- Oda J, Ueyama M, Yamashita K, et al. Hypertonic lactated saline resuscitation reduces the risk of abdominal compartment syndrome in severely burned patients. J Trauma 2006;60:64-71.
- O’Mara MS, Slater H, Goldforb IW, Caushaj PF. A prospective, randomized evaluation of intra-abdominal pressures with crystalloid and colloid resuscitation in burn patients. J Trauma 2005;58:1011-1018.
- RRT- Baker A, Green R. Renal replacement therapy in critical care. World Federation of Societies of Anaesthesiologists. Anaesthesia Tutorial of the week 194. Aug. 30, 2010. Available at: www.aagbi.org/node/836. Accessed Dec. 18, 2016.
- Bodnar Z, Keresztes T, Kovacs I, et al. Increased serum adenosine and interleukin 10 levels as new laboratory markers of increased intra-abdominal pressure. Langenbecks Arch Surg 2010;395:969-972.
- Hörer T, Skoog P, Pirouzram A, Larzon T. Tissue plasminogen activator-assisted hematoma evacuation to relieve abdominal compartment syndrome after endovascular repair of ruptured abdominal aortic aneurysm. J Endovasc Ther 2012;19:144-148.
- Mentula P, Hienonen P, Kemppainen E, et al. Surgical decompression for abdominal compartment syndrome in severe acute pancreatitis. Arch Surg 2010;145:764-769.
- Leppäniemi A. Surgical management of abdominal compartment syndrome: Indications and techniques. Scand J Trauma Resusc Emerg Med 2009;17:17.
- Atema JJ, Gans SL, Boermeester MA. Systemic review and meta-analysis of the open abdomen and temporary abdominal closure techniques in non-trauma patients. World J Surg 2014;39:912-925.
- Acosta S, Bjarnason T, Petersson U, et al. Multicentre prospective study of facial closure rate after open abdomen with vacuum and mesh-mediated fascial traction. Br J Surg 2011;98:735-743.
Intra-abdominal hypertension and abdominal compartment syndrome are common occurrences in both ICU and surgical patients. It is important for emergency physicians to have a general working knowledge of abdominal compartment syndrome so they can identify risk factors and decrease morbidity and mortality for the duration of the patient’s hospitalization.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
