Adult Vaccination Update for Emergency Physicians
March 15, 2017
Reprints
AUTHOR
Jeffrey Weinstein, MD, FIDSA, CPHQ, Chief Quality Officer, Director, Antimicrobial Stewardship Program, Kettering Medical Center and Sycamore Hospital, Dayton, OH.
PEER REVIEWER
Dean L. Winslow, MD, Professor of Medicine, Division of General Medical Disciplines, Division of Infectious Diseases and Geographic Medicine, Stanford University School of Medicine, Stanford, CA.
Statement of Financial Disclosure
To reveal any potential bias in this publication, and in accordance with Accreditation Council for Continuing Medical Education guidelines, we disclose that Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Dr. Schneider (editor), Dr. Stapczynski (editor), Ms. Fessler (nurse planner), Dr. Weinstein (author), Dr. Winslow (peer reviewer), Ms. Mark (executive editor), and Ms. Coplin (executive editor) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Adult vaccination is one of the most important means of preventing disease in vulnerable populations, but rates of vaccine coverage for adults are still significantly lower than with routine childhood vaccination.
- The effectiveness of many vaccines depends on the ability to vaccinate a large percentage of the population. Vaccination offered from the emergency department can help reach these thresholds by addressing the Advisory Committee on Immunization Practices recommendations.
- Most vaccines work by stimulating the humoral immune system to produce antibody to the pathogen. Future challenge then elicits a protective immune response.
- Influenza and tetanus/diphtheria/pertussis vaccines are recommended for all adults, while others are recommended in subsets of adults depending on age, comorbidities, and certain risk factors.
- Live virus vaccines generally are contraindicated in immunosuppressed patients.
- Pregnant women are relatively immunosuppressed and should not receive live virus vaccines, but all pregnant women should receive the tetanus/diphtheria/acellular pertussis vaccine during each pregnancy.
What I need to know about vaccinations is simple: Give a tetanus booster if it has been more than five years in patients with dirty wounds, and if the patient has a cough, fever, and body aches, ask if they received a flu shot. After all, routine immunization is not part of the emergency department’s (ED) role. This may change. A few EDs and some urgent cares (UCs) are offering influenzal and pneumococcal vaccinations as part of their scope of service. There is also discussion about using the ED or UC visit to screen for and initiate immunizations for other diseases. We present this discussion of adult immunizations so that you will be prepared to respond to questions and concerns as the concept of ED vaccination evolves.
— J. Stephan Stapczynski, MD, Editor
Introduction
It started with a hypothesis. In the late 1700s, Edward Jenner had heard that milkmaids who frequently were exposed to cowpox seemed to be immune to smallpox (variola) infection. Although others had noted this, Jenner proved that immunity could be developed by injecting cowpox into subjects who he later challenged with smallpox.1 The Latin word for cow is vacca — thus the medical term for vaccines. Vaccines are among the single biggest scientific advance in human history, and Jenner’s work is credited with saving millions of lives.
Over recent decades, new advances in vaccination have continued to alter the epidemiology of common infectious diseases in ways previously unimaginable. For instance, childhood meningitis and epiglottitis due to Haemophilus influenzae type b essentially have been eradicated in the United States since universal childhood vaccination against this pathogen began in 1991.2 Chickenpox likewise has been reduced 90% with the widespread uptake of varicella vaccination.3
Since adults and children live together in communities, the beneficial effects of childhood vaccines have extended to elders in significant ways. The death rates among both children and adults declined after the introduction of the varicella vaccine,4 and the adult death rate from invasive pneumococcal disease declined with the introduction of infant pneumococcal conjugate vaccine (PCV13). This is explained, in part, by the concept of herd immunity. When a large enough proportion is immune to a pathogen, even those in the community who have not been vaccinated are afforded some protection.
This article will review the vaccines recommended for all adults as well as those vaccines recommended for special populations such as immunosuppressed patients and pregnant women. In addition, this article will examine strategies that can be employed to increase vaccination rates in adults.
Vaccinations Recommended for Healthy Adults
The vaccines recommended in healthy adults are listed in Figure 1. All shown shaded in yellow are universally recommended. This figure along with the detailed footnotes can be found at: www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. Those vaccines shaded in purple will be described in a later section of this article.
Influenza Vaccine
Influenza A and B cause epidemics annually worldwide with severity determined by the degree of change in the hemagglutinin and, to a lesser degree, the neuraminidase antigens on the viral surface. The predominant strains since 1968 have been H3N2, but a pandemic occurred in 2009 with an antigenic shift to the H1N1 (swine flu) strain.5 During inter-pandemic years, there is antigenic drift such that the prior year’s vaccine is unlikely to remain effective. Thus, influenza vaccination is recommended annually for all adults in the United States who lack a contraindication.
The currently available vaccines include trivalent and quadrivalent intramuscular and intradermal preparations, as well as a high-dose form and a newer recombinant form that has no ovalbumin and therefore is safe for use in patients with severe egg allergies.6 The Advisory Committee on Immunization Practices (ACIP) does not state a preference of which vaccine should be given, although some experts favor high-dose influenza vaccination for those > 65 years of age because it results in better vaccine efficacy.7 A live attenuated influenza vaccine that has been approved for use in those 2-49 years of age is no longer recommended, as preliminary data show only 3% protective efficacy compared to 63% efficacy of the standard inactivated vaccines in children aged 2-17 years.8
In adults, the efficacy of influenza vaccination depends on the status of the host. A meta-analysis of studies performed between 1966 and 2013 determined that parenteral inactivated influenza vaccine had an efficacy of about 60% in healthy adults,9 whereas studies in HIV-infected patients suggest efficacy to prevent laboratory confirmed influenza may be as high as 71-85%.10 The currently marketed influenza vaccines are extremely safe, with the most common side effect being pain at the injection site.
Prior anaphylaxis to an influenza vaccine (which is a rare event) is the only absolute contraindication. As noted above, those with severe egg allergy can receive the recombinant influenza vaccine. The swine flu vaccine given in 1976 led to an increase in cases of Guillain-Barre syndrome (GBS), but a recent study suggests the small increased rates of this syndrome with current vaccines are lower than the rate of GBS seen after naturally occurring influenza infection.11
Tetanus, Diphtheria, Pertussis
For several decades the recommendation for diphtheria/tetanus (Td) vaccination in adults simply was for booster shots every 10 years. Tetanus, although rare in the United States, is most common in older adults whose immunity has waned due to lack of receipt of the Td booster. While diphtheria also remains rare in the United States, there have been epidemics overseas in recent years. Vaccine refusal is one of the factors leading to a resurgence of pertussis in children and adults.12 This resurgence has led to a revision of the recommendations, as the Td booster does not protect against pertussis. As per Figure 2, the ACIP now recommends one tetanus/diphtheria/acellular pertussis (Tdap) booster for all adults in the United States followed by Td boosters every 10 years.
Special consideration regarding Td boosters applies for tetanus-prone wounds. All patients who have received fewer than three doses of tetanus-containing vaccine should receive a booster. Those in this category with major tetanus-prone wounds also should receive tetanus immune globulin administered at a different site. Fully vaccinated patients with minor wounds only require a booster if the most recent tetanus-containing vaccine was given more than 10 years previously, while those with major wounds should receive a booster if their last dose was more than five years previously. Neither of these groups requires tetanus immune globulin.13
As per the general ACIP recommendations, if adult patients with wounds in need of a booster have not had the Tdap, then this preparation should be given. Pain at the injection site is the most common side effect of Tdap vaccine in adults. Low-grade fever is relatively common, while higher grade fever of > 102°F is more common among children. The acellular pertussis vaccine has fewer side effects than the older whole cell pertussis vaccines. The use of Tdap vaccines in pregnant women is discussed in a later section of this article.
Varicella and Zoster Vaccines
Varicella-zoster virus (VZV) is a herpesvirus that is the cause of chickenpox in non-immune individuals and shingles when it reactivates later in life. Primary chickenpox in children can be a mild disease but also can be associated with severe disease and even death in children, primarily from Streptococcal toxic shock syndrome as a superinfection. Routine varicella vaccination not only can reduce morbidity and mortality in children,14 but by reducing the burden of disease in the community, there is lower likelihood of exposure in pregnant women and immunocompromised adults. These groups of adults are not only at higher risk for severe chickenpox but also have a contraindication to vaccination themselves.
The ACIP does recommend varicella vaccination to all non-immune adults who do not have such a contraindication. The main side effects of varicella vaccination are pain at the injection site and the development of a small number of nearby vesicles. A more diffuse rash can occur, and those affected should avoid close contact with non-immune or immunocompromised individuals.15
The zoster vaccine was developed for use in adults to help prevent shingles, which is a reactivation of previously acquired VZV infection. As adults age or become immunocompromised, the risk of shingles increases. Shingles can present as dermatomal disease or disseminated disease. The latter can cause life-threatening manifestations, including pneumonia, ophthalmic infection, and central nervous system involvement, in immunocompromised patients. In addition, post-herpetic neuralgia with chronic pain is a complication more common in older adults.
The zoster vaccine has an efficacy of approximately 50% for preventing shingles, but nearly 70% for preventing postherpetic neuralgia.16 Although efficacy has been shown for adults 50 years of age and older, the cost effectiveness for those aged 50-60 years is lower17 and, therefore, many payors will only cover the zoster vaccine for those patients older than 60 years.
Contraindications to zoster vaccination include pregnancy and immunocompromised states. However, the vaccine is safe for HIV-infected patients with a CD4 count of > 200 cells/mL3. The most common side effect of the zoster vaccine is pain at the injection site. The only differences between the varicella vaccine and the zoster vaccine are the dose and administration schedule. The varicella vaccine is lower dose and requires a two-dose schedule, whereas the zoster vaccine contains a higher concentration of the attenuated virus but is given as a single shot.
Measles, Mumps, and Rubella
Measles and mumps are viral infections that had been nearly eliminated in the United States by the early 1980s because of successful vaccination strategies in children. Now both diseases occur in epidemics related to under-immunization in certain religious groups18 and the children of adults philosophically opposed to vaccination (“anti-vaxxers”).19 The latter group has remained steadfastly against childhood vaccination for fear of a link to autism despite multiple Institute of Medicine reports and other studies that have failed to find any causal links between either the measles/mumps/rubella (MMR) or vaccine ingredients such as thimerosal and autism spectrum disorder.20 In fact, the original research purporting this link has been proven to be fraudulent.21
Measles, mumps, and rubella all can have serious health consequences in adults as well as children. In addition to the classic presentation with coryza, fever, and rash, measles can cause pneumonia, encephalitis, and subacute sclerosing panencephalitis. Mumps causes the classic parotitis and orchitis but also can be associated with central nervous system disease. While often mild in adults, rubella is feared as it causes a variety of serious birth defects in infants born to mothers infected during pregnancy. The efficacy of the MMR vaccine approaches 95-100%.22
Because infection with measles and mumps was nearly universal before 1957, those people born before then generally are considered to be immune. Per the ACIP, all adults born in or after 1957 should have at least one dose of the MMR unless there is a contraindication such as immunosuppression. A second dose four weeks later is recommended for college students, healthcare workers, and international travelers. Serologic testing should be performed on women of childbearing age, and those not immune to rubella should be vaccinated if not pregnant; vaccination is contraindicated during pregnancy. The most common side effects of MMR vaccination in adults include fever, rash, lymphadenopathy, and joint pain.
Human Papillomavirus Vaccines
Human papillomavirus (HPV) is the major cause of not only genital warts but also urogenital cancers. Because types 16 and 18 are the major etiologic agents of cervical cancer, vaccines initially were developed and approved for use in girls and women. Subsequently HPV vaccines against four and later nine serotypes were developed. Now HPV vaccine is recommended for both sexes for several reasons. In females, vaccination is given primarily to reduce the risk of cervical cancer. In males, vaccination that reduces infections likewise will reduce infection in their sexual partners. Also, male vaccination will reduce the risk for HPV-associated diseases, such as penile, anal, and oral cancers, as well as respiratory papillomatosis.
Vaccination to prevent HPV infection is of course most effective if given before the period of risk — that is to say before the onset of sexual activity. For boys and girls, the vaccine series should be given at ages 11-12 years. Because there are many different serotypes, however, catch-up vaccination is recommended for women up until age 26 years and for men until age 21 years (26 years if HIV-infected). The ACIP currently recommends the use of either the 2-valent, 4-valent, or newer 9-valent vaccine in girls/women, but only the 4-valent or 9-valent vaccine in boys/men (see Figure 1).
Much of the efficacy data on HPV vaccination comes from Australia, which was the first country to establish a national government-funded vaccine campaign. Rates of infection with the four vaccine serotypes (6, 11, 16, 18) fell among women from 28.7% to 6.7% from the pre-vaccine period to the post-vaccine period.23 In the United States, the prevalence of infection in females with these serotypes has been reduced by 64% in the 14- to 19-year-old age group and by 34% in the 20- to 24-year-old age group.24 In males, the efficacy for preventing genital warts among the four vaccine serotypes was 65% in a large international trial.25
The HPV vaccines are very safe, with the main adverse effect being pain at the injection site. There have been reports of syncopal episodes after receipt of the vaccine, and the ACIP now recommends a 15-minute waiting period in a sitting or supine position following vaccination.26
Pneumococcal Vaccines
Streptococcus pneumoniae is a virulent bacterial pathogen that causes pneumonia, bacteremia, and bacterial meningitis in adults and children. Initial pneumococcal vaccines were directed at capsular polysaccharides of the many serotypes of the bacteria. The polysaccharide vaccines are poorly immunogenic in younger children and the immunosuppressed and, therefore, pneumococcal conjugate vaccines have been developed. These vaccines include protein antigens to help induce type-specific antibody production.
At present, the two major vaccines available in the United States are the pneumococcal polysaccharide 23-valent (PPSV23) and the pneumococcal conjugate 13-valent (PCV13) preparations. The serotypes covered include those that are most commonly associated with invasive pneumococcal disease in humans. Dosing and sequencing of these vaccines vary depending on the age of the patient and indication for vaccination.
In general, the PCV13 should be given before the PPSV23 in those never previously vaccinated. Those requiring more than one dose of PPSV23 should not receive the subsequent doses within five years of the previous dose. The ACIP has scheduled review of the pneumococcal vaccine algorithms for adults in 2018. Detailed algorithms are found online at the ACIP Recommended Adult Immunization Schedule: www.cdc.gov/vaccines/hcp/acip-recs/index.html.
The efficacy of the PPSV23 vaccine in older adults has been established in a number of trials and confirmed in a recent meta-analysis.27 The vaccine efficacy to prevent invasive pneumococcal disease was between 45-73% and to prevent pneumococcal pneumonia was between 48-64%. Although these numbers are similar to the efficacy of PCV13, it is thought that the PPSV23 still has a role in adults as it provides protection against pneumococcal strains not included in PCV13. A very large trial of the PCV13 vaccine in 85,000 adults 65 years of age and older found vaccine efficacy to be 45% at reducing vaccine-type pneumococcal pneumonia and 75% at preventing invasive pneumococcal disease.28
Adverse effects of the pneumococcal vaccine include pain, swelling, and/or erythema at the injection site. The only known contraindication to vaccination is a history of anaphylaxis to a previous pneumococcal vaccine.
Vaccinations Recommended for Special Adult Populations
Hepatitis B Vaccine
The hepatitis B vaccine is highly effective at preventing infection, as evidenced by the epidemiology of hepatitis B infection in vaccinated healthcare workers. While hepatitis B is much more transmissible than hepatitis C or HIV, there has been an approximate 30-fold reduction in hepatitis B infections among healthcare workers since universal vaccination began.29 Rates of vaccination in other adults, however, are low, and those who were not vaccinated in childhood remain at risk via sexual exposure or the sharing of needles used for IV drug injection.30
Although a large proportion of patients with acute hepatitis B clear the infection spontaneously, those who develop chronic infection are at risk of progressing to cirrhosis or developing hepatocellular carcinoma. Given the safety of this vaccine, the high burden of disease, and ease of transmission, the hepatitis B vaccine is now a routine childhood vaccine. Adults who should be vaccinated are described in Table 1 as well as in Figures 1 and 2.
Table 1. Persons Who Should Be Vaccinated Against Hepatitis B
- Healthcare workers
- Patients with end-stage renal disease or liver disease who are not immune, diabetics depending on risk
- Injection drug users and their sexual partners who are not immune
- Public safety workers at risk for blood and body fluid exposure
- Men who have sex with men
- Patients in a sexually transmitted disease treatment center
- Household contacts of patients with hepatitis B surface antigen positivity
- Adults with multiple sexual partners
The hepatitis B vaccine is given as a series of three injections over six months — alone or in combination with the hepatitis A vaccine. Vaccine response rates are 85-90%, and testing of anti-hepatitis B antibody titers is recommended after vaccination of healthcare workers or others with ongoing risk, such as IV drug users or hemodialysis patients. In general, non-responders require an additional three-dose series. Severe adverse reactions to hepatitis B vaccination are rare.31
Hepatitis A Vaccine
The hepatitis A virus does not cause chronic disease, but acute infection can produce a miserable illness characterized by nausea, malaise, and occasionally severe liver dysfunction. Although less common in the United States than in developing countries, the virus is transmitted via the fecal-oral route so everyone is at risk. Most infections are transmitted by food handlers, so common source outbreaks occur.
The vaccine is recommended as per Figure 2 for patients with chronic liver disease, including hepatitis B or C infections, men who have sex with men, injection drug users, and travelers. For the latter group, the first injection provides protection for a trip, but a second shot six months after the first completes the series and provides prolonged protection.32 If greater than six months has elapsed between doses, the series does not need to be restarted; the second shot is sufficient. Soreness at the injection site is the most common side effect of this vaccine, and severe reactions are extremely uncommon.
Figure 1. Recommended Immunization Schedule for Adults Aged 19 Years or Older by Age Group, United States, 2017
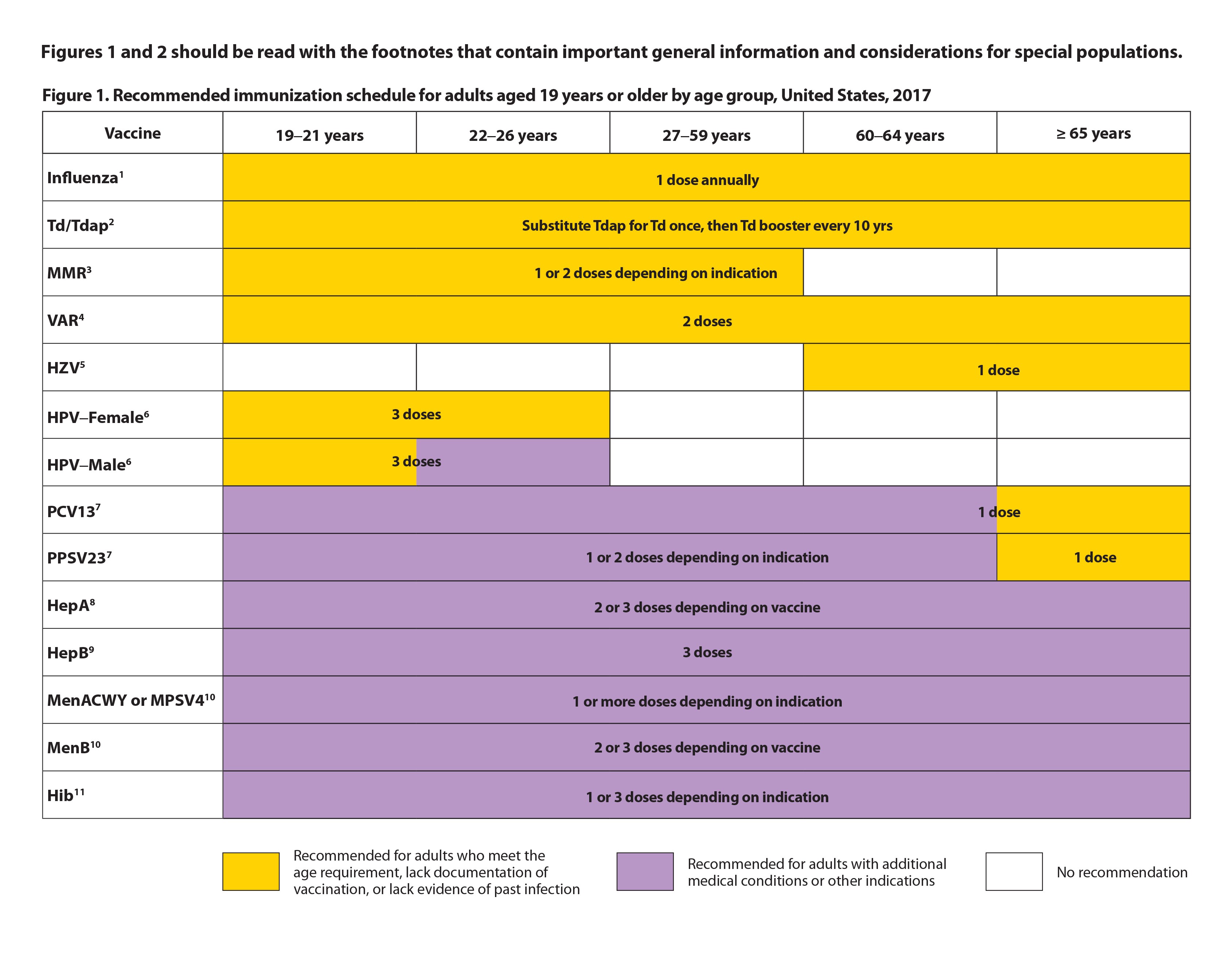
*These figures must be used along with the detailed footnotes found at: www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. SOURCE: Centers for Disease Control and Prevention
Figure 2. Recommended Immunization Schedule for Adults Aged 19 Years or Older by Medical Condition and Other Indications, United States, 2017
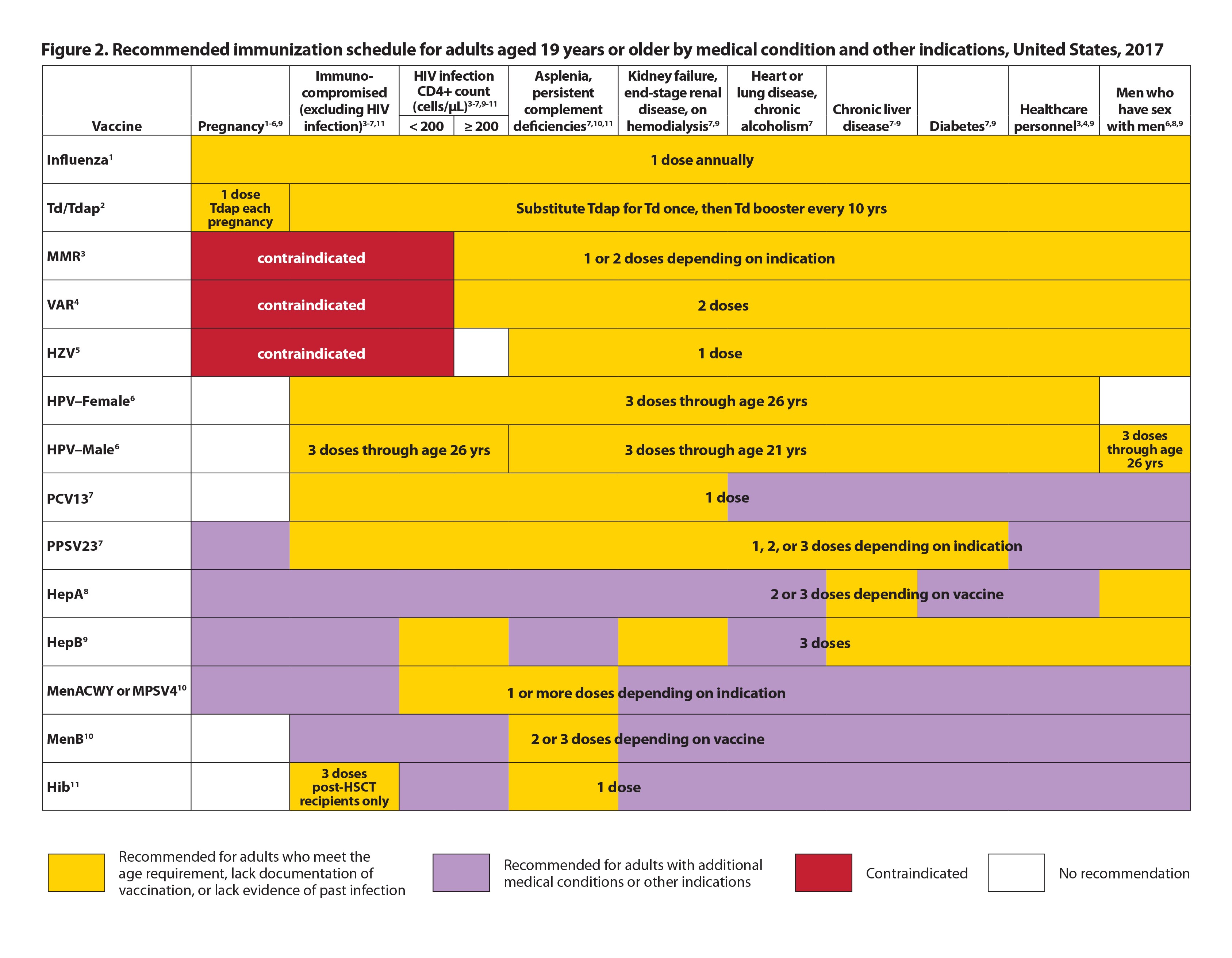
*These figures must be used along with the detailed footnotes found at: www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. SOURCE: Centers for Disease Control and Prevention
Footnotes. Recommended Immunization Schedule for Adults Aged 19 Years or Older, United States, 2017
1. Influenza vaccination
General information
- All persons aged 6 months or older who do not have a contraindication should receive annual influenza vaccination with an age-appropriate formulation of inactivated influenza vaccine (IIV) or recombinant influenza vaccine (RIV).
- In addition to standard-dose IIV, available options for adults in specific age groups include: high-dose or adjuvanted IIV for adults aged 65 years or older, intradermal IIV for adults aged 18 through 64 years, and RIV for adults aged 18 years or older.
- Notes: Live attenuated influenza vaccine (LAIV) should not be used during the 2016–2017 influenza season. A list of currently available influenza vaccines is available at www.cdc.gov/flu/protect/vaccine/vaccines.htm.
Special populations
- Adults with a history of egg allergy who have only hives after exposure to egg should receive age-appropriate IIV or RIV.
- Adults with a history of egg allergy other than hives, e.g., angioedema, respiratory distress, lightheadedness, or recurrent emesis, or who required epinephrine or another emergency medical intervention, may receive age-appropriate IIV or RIV. The selected vaccine should be administered in an inpatient or outpatient medical setting and under the supervision of a healthcare provider who is able to recognize and manage severe allergic conditions.
- Pregnant women and women who might become pregnant in the upcoming influenza season should receive IIV.
2. Tetanus, diphtheria, and acellular pertussis vaccination
General information
- Adults who have not received tetanus and diphtheria toxoids and acellular pertussis vaccine (Tdap) or for whom pertussis vaccination status is unknown should receive 1 dose of Tdap followed by a tetanus and diphtheria toxoids (Td) booster every 10 years. Tdap should be administered regardless of when a tetanus or diphtheria toxoid-containing vaccine was last received.
- Adults with an unknown or incomplete history of a 3-dose primary series with tetanus and diphtheria toxoid-containing vaccines should complete the primary series that includes 1 dose of Tdap.
Unvaccinated adults should receive the first 2 doses at least 4 weeks apart and the third dose 6–12 months after the second dose.
- Notes: Information on the use of Td or Tdap as tetanus prophylaxis in wound management is available at www.cdc.gov/mmwr/preview/mmwrhtml/rr5517a1.htm.
Special populations
- Pregnant women should receive 1 dose of Tdap during each pregnancy, preferably during the early part of gestational weeks 27–36, regardless of prior history of receiving Tdap.
3. Measles, mumps, and rubella vaccination
General information
- Adults born in 1957 or later without acceptable evidence of immunity to measles, mumps, or rubella (defined below) should receive 1 dose of measles, mumps, and rubella vaccine (MMR) unless they have a medical contraindication to the vaccine, e.g., pregnancy or severe immunodeficiency.
- Notes: Acceptable evidence of immunity to measles, mumps, or rubella in adults is: born before 1957, documentation of receipt of MMR, or laboratory evidence of immunity or disease. Documentation of healthcare provider-diagnosed disease without laboratory confirmation is not acceptable evidence of immunity.
Special populations
- Pregnant women who do not have evidence of immunity to rubella should receive 1 dose of MMR upon completion or termination of pregnancy and before discharge from the healthcare facility; nonpregnant women of childbearing age without evidence of rubella immunity should receive 1 dose of MMR.
- Adults with primary or acquired immunodeficiency including malignant conditions affecting the bone marrow or lymphatic system, systemic immunosuppressive therapy, or cellular immunodeficiency should not receive MMR.
- Adults with human immunodeficiency virus (HIV) infection and CD4+ T-lymphocyte count ≥200 cells/μl for at least 6 months who do not have evidence of measles, mumps, or rubella immunity should receive 2 doses of MMR at least 28 days apart. Adults with HIV infection and CD4+ T-lymphocyte count <200 cells/μl should not receive MMR.
- Adults who work in healthcare facilities should receive 2 doses of MMR at least 28 days apart; healthcare personnel born before 1957 who are unvaccinated or lack laboratory evidence of measles, mumps, or rubella immunity, or laboratory confirmation of disease should be considered for vaccination with 2 doses of MMR at least 28 days apart for measles or mumps, or 1 dose of MMR for rubella.
- Adults who are students in postsecondary educational institutions or plan to travel internationally should receive 2 doses of MMR at least 28 days apart.
- Adults who received inactivated (killed) measles vaccine or measles vaccine of unknown type during years 1963–1967 should be revaccinated with 1 or 2 doses of MMR.
- Adults who were vaccinated before 1979 with either inactivated mumps vaccine or mumps vaccine of unknown type who are at high risk for mumps infection, e.g., work in a healthcare facility, should be considered for revaccination with 2 doses of MMR at least 28 days apart.
4. Varicella vaccination
General information
- Adults without evidence of immunity to varicella (defined below) should receive 2 doses of single-antigen varicella vaccine (VAR) 4–8 weeks apart, or a second dose if they have received only 1 dose.
- Persons without evidence of immunity for whom VAR should be emphasized are: adults who have close contact with persons at high risk for serious complications, e.g., healthcare personnel and household contacts of immunocompromised persons; adults who live or work in an environment in which transmission of varicella zoster virus is likely, e.g., teachers, childcare workers, and residents and staff in institutional settings; adults who live or work in environments in which varicella transmission has been reported, e.g., college students, residents and staff members of correctional institutions, and military personnel; nonpregnant women of childbearing age; adolescents and adults living in households with children; and international travelers.
- Notes: Evidence of immunity to varicella in adults is: U.S.-born before 1980 (for pregnant women and healthcare personnel, U.S.-born before 1980 is not considered evidence of immunity); documentation of 2 doses of VAR at least 4 weeks apart; history of varicella or herpes zoster diagnosis or verification of varicella or herpes zoster disease by a healthcare provider; or laboratory evidence of immunity or disease.
Special populations
- Pregnant women should be assessed for evidence of varicella immunity. Pregnant women who do not have evidence of immunity should receive the first dose of VAR upon completion or termination of pregnancy and before discharge from the healthcare facility, and the second dose 4–8 weeks after the first dose.
- Healthcare institutions should assess and ensure that all healthcare personnel have evidence of immunity to varicella.
- Adults with malignant conditions, including those that affect the bone marrow or lymphatic system or who receive systemic immunosuppressive therapy, should not receive VAR.
- Adults with human immunodeficiency virus (HIV) infection and CD4+ T-lymphocyte count ≥200 cells/μl may receive 2 doses of VAR 3 months apart. Adults with HIV infection and CD4+ T-lymphocyte count <200 cells/μl should not receive VAR.
5. Herpes zoster vaccination
General information
- Adults aged 60 years or older should receive 1 dose of herpes zoster vaccine (HZV), regardless of whether they had a prior episode of herpes zoster.
Special populations
- Adults aged 60 years or older with chronic medical conditions may receive HZV unless they have a medical contraindication, e.g., pregnancy or severe immunodeficiency.
- Adults with malignant conditions, including those that affect the bone marrow or lymphatic system or who receive systemic immunosuppressive therapy, should not receive HZV.
- Adults with human immunodeficiency virus (HIV) infection and CD4+ T-lymphocyte count <200 cells/μl should not receive HZV.
Footnotes. Recommended Immunization Schedule for Adults Aged 19 Years or Older, United States, 2017 (continued)
6. Human papillomavirus vaccination
General information
- Adult females through age 26 years and adult males through age 21 years who have not received any human papillomavirus (HPV) vaccine should receive a 3-dose series of HPV vaccine at 0, 1-2, and 6 months. Males aged 22 through 26 years may be vaccinated with a 3-dose series of HPV vaccine at 0, 1–2, and 6 months.
- Adult females through age 26 years and adult males through age 21 years (and males aged 22 through 26 years who may receive HPV vaccination) who initiated the HPV vaccination series before age 15 years and received 2 doses at least 5 months apart are considered adequately vaccinated and do not need an additional dose of HPV vaccine.
- Adult females through age 26 years and adult males through age 21 years (and males aged 22 through 26 years who may receive HPV vaccination) who initiated the HPV vaccination series before age 15 years and received only 1 dose, or 2 doses less than 5 months apart, are not considered adequately vaccinated and should receive 1 additional dose of HPV vaccine.
- Notes: HPV vaccination is routinely recommended for children at age 11 or 12 years. For adults who had initiated but did not complete the HPV vaccination series, consider their age at first HPV vaccination (described above) and other factors (described below) to determine if they have been adequately vaccinated.
Special populations
- Men who have sex with men through age 26 years who have not received any HPV vaccine should receive a 3-dose series of HPV vaccine at 0, 1-2, and 6 months.
- Adult females and males through age 26 years with immunocompromising conditions (described below), including those with human immunodeficiency virus (HIV) infection, should receive a 3-dose series of HPV vaccine at 0, 1–2, and 6 months.
- Pregnant women are not recommended to receive HPV vaccine, although there is no evidence that the vaccine poses harm. If a woman is found to be pregnant after initiating the HPV vaccination series, delay the remaining doses until after the pregnancy. No other intervention is needed. Pregnancy testing is not needed before administering HPV vaccine.
- Notes: Immunocompromising conditions for which a 3-dose series of HPV vaccine is indicated are primary or secondary immunocompromising conditions that might reduce cell-mediated or humoral immunity, e.g., B-lymphocyte antibody deficiencies, complete or partial T-lymphocyte defects, HIV infection, malignant neoplasm, transplantation, autoimmune disease, and immunosuppressive therapy.
7. Pneumococcal vaccination
General information
- Adults who are immunocompetent and aged 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) followed by 23-valent pneumococcal polysaccharide vaccine (PPSV23) at least 1 year after PCV13.
- Notes: Adults are recommended to receive 1 dose of PCV13 and 1, 2, or 3 doses of PPSV23 depending on indication. When both PCV13 and PPSV23 are indicated, PCV13 should be administered first; PCV13 and PPSV23 should not be administered during the same visit. If PPSV23 has previously been administered, PCV13 should be administered at least 1 year after PPSV23. When two or more doses of PPSV23 are indicated, the interval between PPSV23 doses should be at least 5 years. Supplemental information on pneumococcal vaccine timing for adults aged 65 years or older and adults aged 19 years or older at high risk for pneumococcal disease (described below) is available at www.cdc.gov/vaccines/vpd-vac/pneumo/downloads/adult-vax-clinician-aid.pdf. No additional doses of PPSV23 are indicated for adults who received PPSV23 at age 65 years or older. When indicated, PCV13 and PPSV23 should be administered to adults whose pneumococcal vaccination history is incomplete or unknown.
Special populations
- Adults aged 19 through 64 years with chronic heart disease including congestive heart failure and cardiomyopathies (excluding hypertension); chronic lung disease including chronic obstructive lung disease, emphysema, and asthma; chronic liver disease including cirrhosis; alcoholism; or diabetes mellitus; or who smoke cigarettes should receive PPSV23. At age 65 years or older, they should receive PCV13 and another dose of PPSV23 at least 1 year after PCV13 and at least 5 years after the most recent dose of PPSV23.
- Adults aged 19 years or older with immunocompromising conditions or anatomical or functional asplenia (described below) should receive PCV13 and a dose of PPSV23 at least 8 weeks after PCV13, followed by a second dose of PPSV23 at least 5 years after the first dose of PPSV23. If the most recent dose of PPSV23 was administered before age 65 years, at age 65 years or older, administer another dose of PPSV23 at least 8 weeks after PCV13 and at least 5 years after the most recent dose of PPSV23.
- Adults aged 19 years or older with cerebrospinal fluid leak or cochlear implant should receive PCV13 followed by PPSV23 at least 8 weeks after PCV13. If the most recent dose of PPSV23 was administered before age 65 years, at age 65 years or older, administer another dose of PPSV23 at least 8 weeks after PCV13 and at least 5 years after the most recent dose of PPSV23.
- Notes: Immunocompromising conditions that are indications for pneumococcal vaccination are congenital or acquired immunodeficiency including B- or T-lymphocyte deficiency, complement deficiencies, and phagocytic disorders excluding chronic granulomatous disease; human immunodeficiency virus (HIV) infection; chronic renal failure and nephrotic syndrome; leukemia, lymphoma, Hodgkin disease, generalized malignancy, and multiple myeloma; solid organ transplant; and iatrogenic immunosuppression including long-term systemic corticosteroid and radiation therapy. Anatomical or functional asplenia that are indications for pneumococcal vaccination are sickle cell disease and other hemoglobinopathies, congenital or acquired asplenia, splenic dysfunction, and splenectomy. Pneumococcal vaccines should be given at least 2 weeks before immunosuppressive therapy or an elective splenectomy, and as soon as possible to adults who are diagnosed with HIV infection.
8. Hepatitis A vaccination
General information
- Adults who seek protection from hepatitis A virus infection may receive a 2-dose series of single antigen hepatitis A vaccine (HepA) at either 0 and 6–12 months (Havrix) or 0 and 6–18 months (Vaqta). Adults may also receive a combined hepatitis A and hepatitis B vaccine (HepA-HepB) (Twinrix) as a 3-dose series at 0, 1, and 6 months. Acknowledgment of a specific risk factor by those who seek protection is not needed.
Special populations
- Adults with any of the following indications should receive a HepA series: have chronic liver disease, receive clotting factor concentrates, men who have sex with men, use injection or non-injection drugs, or work with hepatitis A virus-infected primates or in a hepatitis A research laboratory setting.
- Adults who travel in countries with high or intermediate levels of endemic hepatitis A infection or anticipate close personal contact with an international adoptee, e.g., reside in the same household or regularly babysit, from a country with high or intermediate level of endemic hepatitis A infection within the first 60 days of arrival in the United States should receive a HepA series.
9. Hepatitis B vaccination
General information
- Adults who seek protection from hepatitis B virus infection may receive a 3-dose series of single-antigen hepatitis B vaccine (HepB) (Engerix-B, Recombivax HB) at 0, 1, and 6 months. Adults may also receive a combined hepatitis A and hepatitis B vaccine (HepA-HepB) (Twinrix) at 0, 1, and 6 months. Acknowledgment of a specific risk factor by those who seek protection is not needed.
Special populations
- Adults at risk for hepatitis B virus infection by sexual exposure should receive a HepB series, including sex partners of hepatitis B surface antigen (HBsAg)-positive persons, sexually active persons who are not in a mutually monogamous relationship, persons seeking evaluation or treatment for a sexually transmitted infection, and men who have sex with men (MSM).
- Adults at risk for hepatitis B virus infection by percutaneous or mucosal exposure to blood should receive a HepB series, including adults who are recent or current users of injection drugs, household contacts of HBsAg-positive persons, residents and staff of facilities for developmentally disabled persons, incarcerated, healthcare and public safety workers at risk for exposure to blood or blood-contaminated body fluids, younger than age 60 years with diabetes mellitus, and age 60 years or older with diabetes mellitus at the discretion of the treating clinician.
- Adults with chronic liver disease including, but not limited to, hepatitis C virus infection, cirrhosis, fatty liver disease, alcoholic liver disease, autoimmune hepatitis, and an alanine aminotransferase (ALT) or aspartate aminotransferase (AST) level greater than twice the upper limit of normal should receive a HepB series.
- Adults with end-stage renal disease including those on pre-dialysis care, hemodialysis, peritoneal dialysis, and home dialysis should receive a HepB series. Adults on hemodialysis should receive a 3-dose series of 40 µg Recombivax HB at 0, 1, and 6 months or a 4-dose series of 40 µg Engerix-B at 0, 1, 2, and 6 months.
- Adults with human immunodeficiency virus (HIV) infection should receive a HepB series.
- Pregnant women who are at risk for hepatitis B virus infection during pregnancy, e.g., having more than one sex partner during the previous six months, been evaluated or treated for a sexually transmitted infection, recent or current injection drug use, or had an HBsAg-positive sex partner, should receive a HepB series.
- International travelers to regions with high or intermediate levels of endemic hepatitis B virus infection should receive a HepB series.
- Adults in the following settings are assumed to be at risk for hepatitis B virus infection and should receive a HepB series: sexually transmitted disease treatment facilities, HIV testing and treatment facilities, facilities providing drug-abuse treatment and prevention services, healthcare settings targeting services to persons who inject drugs, correctional facilities, healthcare settings targeting services to MSM, hemodialysis facilities and end-stage renal disease programs, and institutions and nonresidential day care facilities for developmentally disabled persons.
10. Meningococcal vaccination
Special populations
- Adults with anatomical or functional asplenia or persistent complement component deficiencies should receive a 2-dose primary series of serogroups A, C, W, and Y meningococcal conjugate vaccine (MenACWY) at least 2 months apart and revaccinate every 5 years. They should also receive a series of serogroup B meningococcal vaccine (MenB) with either a 2-dose series of MenB-4C (Bexsero) at least 1 month apart or a 3-dose series of MenB-FHbp (Trumenba) at 0, 1–2, and 6 months.
- Adults with human immunodeficiency virus (HIV) infection who have not been previously vaccinated should receive a 2-dose primary series of MenACWY at least 2 months apart and revaccinate every 5 years. Those who previously received 1 dose of MenACWY should receive a second dose at least 2 months after the first dose. Adults with HIV infection are not routinely recommended to receive MenB because meningococcal disease in this population is caused primarily by serogroups C, W, and Y.
- Microbiologists who are routinely exposed to isolates of Neisseria meningitidis should receive 1 dose of MenACWY and revaccinate every 5 years if the risk for infection remains, and either a 2-dose series of MenB-4C at least 1 month apart or a 3-dose series of MenB-FHbp at 0, 1–2, and 6 months.
- Adults at risk because of a meningococcal disease outbreak should receive 1 dose of MenACWY if the outbreak is attributable to serogroup A, C, W, or Y, or either a 2-dose series of MenB-4C at least 1 month apart or a 3-dose series of MenB-FHbp at 0, 1–2, and 6 months if the outbreak is attributable to serogroup B.
- Adults who travel to or live in countries with hyperendemic or epidemic meningococcal disease should receive 1 dose of MenACWY and revaccinate every 5 years if the risk for infection remains. MenB is not routinely indicated because meningococcal disease in these countries is generally not caused by serogroup B.
- Military recruits should receive 1 dose of MenACWY and revaccinate every 5 years if the increased risk for infection remains.
- First-year college students aged 21 years or younger who live in residence halls should receive 1 dose of MenACWY if they have not received MenACWY at age 16 years or older.
- Young adults aged 16 through 23 years (preferred age range is 16 through 18 years) who are healthy and not at increased risk for serogroup B meningococcal disease (described above) may receive either a 2-dose series of MenB-4C at least 1 month apart or a 2-dose series of MenBFHbp at 0 and 6 months for short-term protection against most strains of serogroup B meningococcal disease.
- For adults aged 56 years or older who have not previously received serogroups A, C, W, and Y meningococcal vaccine and need only 1 dose, meningococcal polysaccharide serogroups A, C, W, and Y vaccine (MPSV4) is preferred. For adults who previously received MenACWY or anticipate receiving multiple doses of serogroups A, C, W, and Y meningococcal vaccine, MenACWY is preferred.
- Notes: MenB-4C and MenB-FHbp are not interchangeable, i.e., the same vaccine should be used for all doses to complete the series. There is no recommendation for MenB revaccination at this time. MenB may be administered at the same time as MenACWY but at a different anatomic site, if feasible.
11. Haemophilus influenzae type b vaccination
Special populations
- Adults who have anatomical or functional asplenia or sickle cell disease, or are undergoing elective splenectomy should receive 1 dose of Haemophilus influenzae type b conjugate vaccine (Hib) if they have not previously received Hib. Hib should be administered at least 14 days before splenectomy.
- Adults with a hematopoietic stem cell transplant (HSCT) should receive 3 doses of Hib in at least 4 week intervals 6–12 months after transplant regardless of their Hib history.
- Notes: Hib is not routinely recommended for adults with human immunodeficiency virus infection because their risk for Haemophilus influenzae type b infection is low.
Meningococcal Vaccines
Neisseria meningitidis is a feared pathogen that can cause meningitis or severe sepsis with purpura fulminans. Those with deficiencies in the terminal components of complement have a significantly elevated risk, but healthy adolescents and adults can be affected. Disease can be sporadic or can occur in outbreaks among those living in close quarters, such as a college dormitory.
There are two versions of meningococcal vaccines: a quadrivalent vaccine effective against serogroups A, C, Y, and W135, and a monovalent strain against serogroup B.33 The FDA has approved three commercial versions of the quadrivalent vaccine and two commercial versions of the meningococcal B vaccine.
Current recommendations from the ACIP include the use of both meningococcal vaccines in adolescents and young adults, and most colleges require these for either all incoming students or those residing on campus. The routine childhood vaccine schedule calls for the initial quadrivalent meningococcal vaccine at 11-12 years of age and a booster at age 16 because efficacy seems to wane prior to the age of high school graduation.34
The meningococcal vaccines are all extremely safe with minor reactions reported. There had been early concerns about a causal relationship between the Menactra quadrivalent meningococcal vaccine and GBS, but large-scale studies have revealed no significant increased risk.35
Haemophilus Influenzae Type b Vaccine
The Haemophilus influenzae type b (Hib) vaccine is not routinely indicated for healthy adults. As previously noted, the vaccine has had a tremendous impact on childhood meningitis, but H. influenzae is not a common cause of bacterial meningitis in adults.
The vaccine is recommended in limited situations for adults: asplenic patients (including those with sickle cell disease, as they are functionally asplenic) and adults after hematopoietic stem cell transplantation. In patients undergoing splenectomy, the vaccine should be administered 14 days before surgery or if the surgery is for traumatic rupture, the vaccine should be given 14 days post-operatively.36 A full three-shot series is not required except following stem cell transplantation.
Vaccines Recommended in Immunosuppressed Patients
Vaccines recommended for immunosuppressed patients are listed in Figure 2. However, not all immunosuppressed patients are alike. Asplenic patients are at unique risk for life-threatening infections with encapsulated organisms and thus require meningococcal and Hib vaccines in addition to pneumococcal vaccines.
As a rule of thumb, live vaccines, such as MMR, zoster, and yellow fever, should be avoided in immunosuppressed patients. However, certain live attenuated vaccines are safe and recommended for HIV-infected patients with CD4 counts > 200 since their immune system is strong enough. Live vaccines also may be acceptable in patients after hematopoietic stem cell transplant after two years, but never in solid organ transplant patients.37 The Hib vaccine also is recommended after hematopoietic stem cell transplant.38
All immunosuppressed adults should receive annual influenza vaccination as well as pneumococcal vaccination, regardless of age. This includes cancer patients actively undergoing chemotherapy treatments.39 They also should be given the Tdap vaccine once, and thereafter the Td booster every 10 years.40
Vaccines Recommended for Patients with Chronic Disease
The recommended vaccines for patients with chronic diseases, such as cardiopulmonary, renal, liver, or alcoholism, are also listed in Figure 2. In general, all should receive annual influenza vaccination as well as pneumococcal vaccination according to the recommended schedule. The recommendation for Tdap once followed by the Td booster every 10 years still applies. The MMR vaccine should be given based on the tables to those without proof of immunity. Zoster vaccine is safe to administer at age 60 years unless the patient is immunocompromised.
The most significant difference among these groups is that patients with end-stage renal disease,41 chronic liver disease, and diabetes all should be vaccinated against hepatitis B if not already immune, whereas this is not universally needed for those patients with chronic cardiopulmonary diseases and alcoholism. Non-immune patients with chronic liver disease also should receive the hepatitis A vaccine series.
Vaccines Recommended for Pregnant Women
Pregnancy is a relatively immunocompromising state and, as such, live vaccines, such as MMR and varicella, are contraindicated. Despite this recommendation, fetal infection in women who were inadvertently given a dose of MMR has never been documented.42 Influenza vaccine should be given to all pregnant women lacking a contraindication during the season when the vaccine is available.
In addition, protection against diphtheria, pertussis, and tetanus is essential in pregnant women. Although diphtheria mainly has re-emerged outside of the United States, pertussis (whooping cough) has had a significant resurgence here this decade, affecting both young children and adults.43 Guidelines now recommend that all pregnant women be vaccinated against these pathogens. Those women who previously have not completed the vaccine series should have a full three-shot series, including one dose of the Tdap. All previously vaccinated pregnant women should have a Tdap booster with each pregnancy even when there is a short time interval between pregnancies,44 as young infants need maternal antibody for protection during the first six months of life.
Vaccines Recommended for Healthcare Workers
Well-established guidelines have been published for the vaccinations recommended (and often required) for healthcare personnel.45 Of course, all healthcare workers should be vaccinated against hepatitis B, as this is the most transmissible virus in healthcare settings. Annual influenza vaccination also is critical for healthcare workers. Measles can be transmitted via an airborne route, and outbreaks have occurred in hospital settings in the United States.
Since immunity from childhood vaccines often wanes, the MMR is recommended for non-immune healthcare workers.45 All healthcare personnel also should be updated with the Tdap if they have not had this formulation previously. Finally, immunity against varicella is important for healthcare workers as this is an airborne virus and also because severe disease can ensue in adults.
Vaccines for Travelers
International travelers may be at risk for a variety of infectious diseases, some of which are not present in the United States. Vaccination of travelers is beyond the scope of this review, and information for healthcare providers regarding recommended vaccines for international travel can be found at wwwnc.cdc.gov/travel/destinations/list/.
Strategies to Increase Adult Vaccination Coverage
It is well documented that rates of adult vaccination in the United States are lower than desired. For example, CDC data from 2014 show that only 43.2% of adults older than 19 years of age received the influenza vaccine and only 61.3% of adults older than 65 years of age had appropriate pneumococcal vaccination coverage.46 This compares to goal rates of vaccination under the Healthy People 2020 program of 70% and 90%, respectively.47 This problem is not new, and a variety of methods have been studied to increase vaccine coverage rates in adults both in outpatient and inpatient settings.
In the ED, feasibility and pilot studies have been done to use the opportunity of an emergency visit for influenza and pneumococcal vaccination.48,49,50,51 The large number of ED visits provides the chance to identify vaccination opportunities, educate patients, and even provide vaccinations.52 Because of the potential severity of the disease and the use of a single injection, studies in which vaccinations are provided in the ED have focused on influenza and pneumococcal vaccinations.53 Screening patients for potential eligibility and referring them to a clinic for vaccination is less ideal; as noted in one study, only 10% of patients who were given a prescription referral for pneumococcal vaccination actually followed up.54 In urgent care and ED settings, vaccinations that require a series of injections remain a challenging and heretofore unstudied possibility.
In the inpatient arena, the missed chances for vaccination are also well described. A study of Medicare inpatients from 1998 to 1999 found that the rate of influenza and pneumococcal vaccination during hospitalization for previously unvaccinated individuals was only 2.7% and 0.4%, respectively.55 The low rate of vaccination during hospitalization has been addressed in part by the IMM-2 Core Measure from the Centers for Medicare and Medicaid Services, which translates into financial penalties for hospitals that fall below a threshold influenza vaccination rate before patient discharge. Many hospitals now have standing orders not only for influenza vaccination in season but also for pneumococcal vaccination year-round for appropriate patients.
REFERENCES
- Baxby D. Edward Jenner’s inquiry after 200 years. BMJ 1999;318:390.
- Centers for Disease Control and Prevention. Progress toward elimination of Haemophilus influenzae type b invasive disease among infants and children — United States, 1998-2000. MMWR Morb Mortal Wkly Rep 2002;51:234.
- Guris D, Jumaan AO, Mascola L, et al. Changing varicella epidemiology in active surveillance sites — United States, 1995-2005. J Infect Dis 2008;197(Suppl 2):S71.
- Nguyen HQ, Jumaan AO, Seward JF. Decline in mortality due to varicella after implementation of varicella vaccination in the United States. N Engl J Med 2005;352:450.
- Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team, Dawood FS, Jain S, et al. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009;360:2605-2615.
- Cox MM, Izikson R, Post P, Dunkle L. Safety, efficacy, and immunogenicity of Flublok in the prevention of seasonal influenza in adults. Ther Adv Vaccines 2015;3:97-108.
- Hibberd PL. Seasonal influenza vaccination in adults. Available at: UpToDate.com. Accessed Feb. 4, 2017.
- Centers for Disease Control and Prevention. ACIP votes down use of LAIV for 2016-2017 flu season. CDC Media Statement. June 22, 2016. Available at: www.cdc.gov/media/releases/2016/s0622-laiv-flu.html. Accessed March 2, 2017.
- Demicheli V, Jefferson T, Al-Ansary LA, et al. Vaccines for preventing influenza in healthy adults. Cochrane Database Syst Rev 2014;(3):CD001269.
- Remschmidt C, Wichmann O, Harder T. Influenza vaccination in HIV-infected individuals: Systematic review and assessment of quality of evidence related to vaccine efficacy, effectiveness and safety. Vaccine 2014;32:5585-5592.
- Vellozzi C, Iqbal S, Broder K. Guillain-Barre syndrome, influenza, and influenza vaccination: The epidemiologic evidence. Clin Infect Dis 2014;58:1149.
- Phadke VK, Bednarczyk RA, Salmon DA, Omer SB. Association between vaccine refusal and vaccine-preventable diseases in the United States: A review of measles and pertussis. JAMA 2016;315:1149-1158.
- Hibberd PL. Tetanus-diphtheria toxoid vaccination in adults. Available at: UpToDate.com. Accessed Feb. 4, 2017.
- Marin M, Zhang JX, Seward JF. Near elimination of varicella deaths in the US after implementation of the vaccination program. Pediatrics 2011;128:214-220.
- Albrecht MA. Vaccination for the prevention of chickenpox (primary varicella infection). Available at: UpToDate.com. Accessed on Feb. 5, 2017.
- Oxman MN, Levin MJ, Johnson GR, et al. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N Engl J Med 2005;352:2271.
- Le P, Rothberg MB. Cost-effectiveness of herpes zoster vaccine for persons aged 50 years. Ann Intern Med 2015;163:489-497.
- Gastañaduy PA, Budd J, Fisher N, et al. A measles outbreak in an underimmunized Amish community in Ohio. N Engl J Med 2016;375:1343-1354.
- Yang YT, Barraza L, Weidenaar K. Measles outbreak as a catalyst for stricter vaccine exemption legislation. JAMA 2015;314:
1229-1230. - Maglione MA, Das L, Raaen L, et al. Safety of vaccines used for routine immunization of U.S. children: A systematic review. Pediatrics 2014;134:325-337.
- Deer B. Secrets of the MMR scare. The Lancet’s two days to bury bad news. BMJ 2011;342:c7001.
- Hibberd PL. Measles, mumps, and rubella immunization in adults. Available at: UpToDate.com. Accessed on Feb. 8, 2017.
- Tabrizi SN, Brotherton JM, Kaldor JM, et al. Fall in human papillomavirus prevalence following a national vaccination program.
J Infect Dis 2012;206:1645-1651. - Markowitz LE, Liu G, Hariri S, et al. Prevalence of HPV after introduction of the vaccination program in the United States. Pediatrics 2016;137:e20151968.
- Giuliano AR, Palefsky JM, Goldstone S, et al. Efficacy of quadrivalent HPV vaccine against HPV infection and disease in males. N Engl J Med 2011;364:401.
- Cox JT, Palefsky JM. Recommendations for the use of human papillomavirus vaccines. Available at: UpToDate.com. Accessed Feb. 11, 2017.
- Falkenhorst G, Remschmidt C, Harder T, et al. Effectiveness of the 23-valent pneumococcal polysaccharide vaccine (PPV23) against pneumococcal disease in the elderly: Systematic review and meta-analysis. PLoS One 2017;12:e0169368.
- Bonten MJ, Huijts SM, Bolkenbaas M, et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N Engl J Med 2015;372:1114-1125.
- Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention (CDC). Immunization of health-care personnel: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2011;60(RR-7):1-45.
- Harris AM, Iqbal K, Schillie S, et al. Increases in acute hepatitis B virus infections — Kentucky, Tennessee, and West Virginia, 2006-2013. MMWR Morb Mortal Wkly Rep 2016;65:47-50.
- Teo E-K, Lok ASF. Hepatitis B virus vaccination. Available at: UpToDate.com. Accessed Jan. 24, 2017.
- Wu D, Guo CY. Epidemiology and prevention of hepatitis A in travelers. J Travel Med 2013;20:394-399.
- Basta NE, Mahmoud AA, Wolfson J, et al. Immunogenicity of a meningococcal B vaccine during a university outbreak. N Engl J Med 2016;376:220-228.
- ACIP Childhood/Adolescent Immunization Work Group, Akinsanya-Beysolow I, Jenkins R, et al. Advisory Committee on Immunization Practices (ACIP) recommended immunization schedule for persons aged 0 through18 years — United States, 2013. MMWR Suppl 2013;62:2-8.
- Valentgas P, Amato AA, Bohn RL, et. al. Risk of Guillani-Barre syndrome after meningococcal conjugate vaccination. Pharmacoepidemiol Drug Saf 2012;21:1350-1358.
- Howdieshell TR, Heffernan D, Dipiro JT. Surgical infection society guidelines for vaccination after traumatic injury. Surg Infect 2006;7:275.
- L’Huillier AG, Kumar D. Immunizations in solid organ and hematopoeitic stem cell transplant patients: A comprehensive review. Hum Vaccin Immunother 2015;11:2852-2863.
- Ullmann AJ, Schmidt-Hieber M, Bertz H, et al. Infectious diseases in allogeneic haematopoietic stem cell transplantation: Prevention and prophylaxis strategy guidelines 2016. Ann Hematol 2016;95:1435-1455.
- Grohskopf LA, Sokolow LZ, Broder KR, et al. Prevention and control of seasonal influenza with vaccines. MMWR Recomm Rep 2016;65:1-54.
- Centers for Disease Control and Prevention. Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis (Tdap) vaccine in adults aged 65 years and older — Advisory Committee on Immunization Practices (ACIP), 2012. MMWR Morb Mortal Wkly Rep 2012;61:468-470.
- Holley JL. Immunizations in patients with end-stage renal disease. Available at: UpToDate.com. Accessed Jan. 21, 2017.
- McLean HQ, Fiebelkorn AP, Temte JL, Wallace GS. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: Summary recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2013;62(RR-04):1-34.
- Hartzell JD, Blaylock JM. Whooping cough in 2014 and beyond: An update and review. Chest 2014;146:205-214.
- Centers for Disease Control and Prevention (CDC). Updated recommendations for use of tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis vaccine (Tdap) in pregnant women — Advisory Committee on Immunization Practices (ACIP), 2012. MMWR Morb Mortal Wkly Rep 2013;62:131.
- Immunization of health-care personnel: Recommendations of the Advisory Committee on Immunization Practices (ACIP). Advisory Committee on Immunization Practices; Centers for Disease Control and Prevention (CDC) MMWR Recomm Rep 2011;60(RR-7):1-45.
- Williams WW, Lu PJ, O’Halloran A, et al. Surveillance of vaccination coverage among adult populations – United States, 2014. MMWR Surveill Summ 2016;65(No. SS-1):1-36.
- Healthy People 2020: Immunization and Infectious Diseases Objectives. Available at: https://www.healthypeople.gov/2020/topics-objectives/topic/immunization-and-infectious-diseases. Accessed Jan. 29,2017.
- Abraham MK, Perkins J, Vilke GM, Coyne CJ. Influenza in the emergency department: Vaccination, diagnosis, and treatment: Clinical practice paper approved by American Academy of Emergency Medicine Clinical Guidelines Committee. J Emerg Med 2016;50:536-542.
- Cohen V, Jellinek-Cohen SP, Likourezos A, et al. Feasibility of a pharmacy-based influenza immunization program in an academic emergency department. Ann Pharmacother 2013;47:1440-1447.
- Venkat A, Chan-Tompkins NH, Hegde GG, et al. Feasibility of integrating a clinical decision support tool into an existing computerized physician order entry system to increase seasonal influenza vaccination in the emergency department. Vaccine 2010;28:6058-6064.
- Dexheimer JW, Talbot TR 3rd, Ye F, et al. A computerized pneumococcal vaccination reminder system in the adult emergency department. Vaccine 2011;29:7035-7041.
- Thomas MC, Ademolu AO. Considerations for vaccine administration in the emergency department. Am J Health Syst Pharm 2014;71:231-236.
- Rimple D, Weiss SJ, Brett M, Ernst AA. An emergency department-based vaccination program: Overcoming the barriers for adults at high risk for vaccine-preventable diseases. Acad Emerg Med 2006;13:922-930.
- Manthey DE, Stopyra J, Askew K. Referral of emergency department patients for pneumococcal vaccination. Acad Emerg Med 2004;11:271-275.
- Bratzler DW, Houck PM, Jiang H, et al. Failure to vaccinate Medicare inpatients: A missed opportunity. Arch Intern Med 2002;162:2349-2356.
We present this discussion of adult immunizations so that you will be prepared to respond to questions and concerns as the concept of ED vaccination evolves.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
