The Great Mimicker: Thyroid Emergencies
September 1, 2017
Reprints
AUTHORS
George Willis, MD, Assistant Professor, Department of Emergency Medicine, University of Maryland, Baltimore.
Sarah Alhizab, MBBS, Department of Emergency Medicine, University of Maryland, Baltimore.
PEER REVIEWER
Jonathan Glauser, MD, Professor, Emergency Medicine, Case Western Reserve University; Senior Faculty, Emergency Medicine Residency, MetroHealth/Cleveland Clinic, OH.
Financial Disclosure
Dr. Farel (CME question reviewer) owns stock in Johnson & Johnson. Ms. Light (nurse planner) reports that she serves as a consultant for Bard Medical. Brian Hocum, PharmD, (pharmacist reviewer) is an employee of United Therapeutics. Dr. Schneider (editor), Dr. Stapczynski (editor), Dr. Willis (author), Dr. Alhizab (author), Dr. Glauser (peer reviewer), Ms. Mark (executive editor), Ms. Coplin (executive editor), and Ms. Hatcher (AHC Media editorial group manager) report no financial relationships with companies related to the field of study covered by this CME activity.
EXECUTIVE SUMMARY
- Thyroid storm is a rare condition that can mimic other more common disorders.
- The two most common clinical features of thyroid storm are fever and altered mental status.
- The most common underlying thyroid disorder in thyroid storm is Graves’ disease.
- Thyroid storm is treated with these classes of medication in sequence: beta-blockers, anti-thyroid agents, inorganic iodine, and corticosteroids.
- Hyperpyrexia in thyroid storm is treated with acetaminophen and, if necessary, passive cooling measures.
- Myxedema coma is a rare condition that is more common in the winter months.
- The most common clinical features of myxedema coma are altered mental status and hypothermia.
- The most common underlying thyroid disorder in myxedema coma is long-standing primary hypothyroidism.
- Myxedema coma is treated with thyroid hormone replacement.
- The hypothermia in myxedema coma is treated with passive warming; active warming measures may exacerbate hypotension.
- The serum thyroid-stimulating hormone is the most readily available test for rapid assessment of thyroid function.
The thyroid gland is one of the human body’s primary regulators of metabolism. Because of the numerous feedback mechanisms in place, the secretion of thyroid hormone is tightly regulated, and metabolism remains relatively balanced throughout life. Thyroid disease is relatively common, but most often is a benign disease with little clinical significance in the emergency setting. However, even a small insult can disrupt this system and throw the regulation out of control, resulting in the secretion of too much or too little thyroid hormone. Both of these situations can result in the thyroid emergencies thyroid storm and myxedema coma.
Although they are relatively uncommon, both of these thyroid emergencies are associated with increased mortality. The problem is that most patients who present with thyroid emergencies will appear to have a much more common disease process. This may result in a delay in the diagnosis and, ultimately, management, contributing to high mortality. It is incumbent upon emergency providers to keep thyroid disease in the differential when evaluating patients presenting with altered mental status and extremes of temperature.
Physiology of Thyroid Hormone
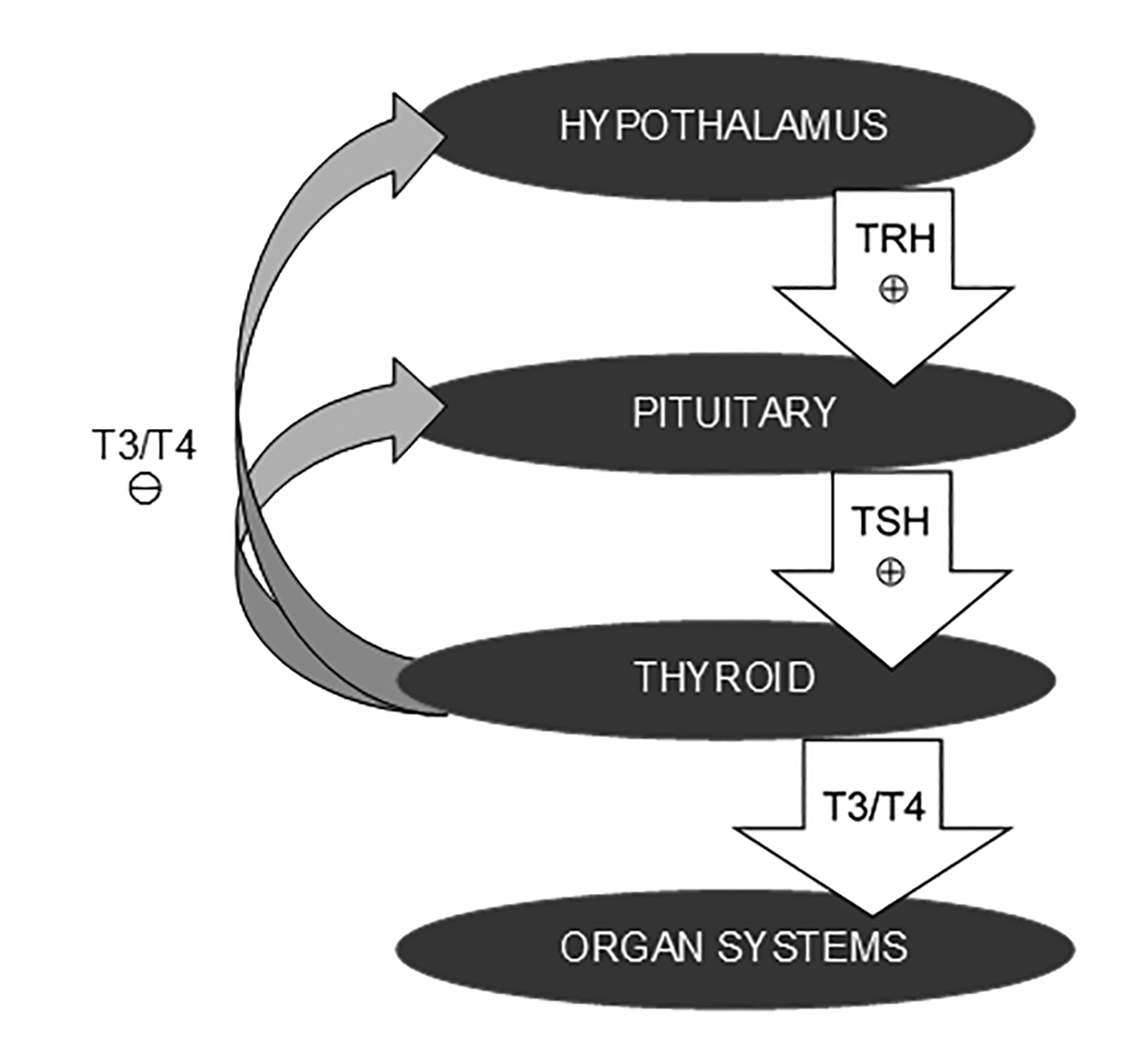
To understand disorders of the thyroid, it is important to comprehend the normal thyroid physiology. Thyroid function is controlled via several feedback mechanisms. The hypothalamus releases thyrotropin-releasing hormone (TRH), which activates the pituitary gland to release thyroid-stimulating hormone (TSH). TSH subsequently stimulates the thyroid gland to secrete thyroid hormones T3 and T4. When there is sufficient circulating thyroid hormone, thyroid hormone negatively feeds back to inhibit the release of TRH from the hypothalamus and the release of TSH from the pituitary gland. This will slow down the release of thyroid hormone from the thyroid gland. This system is called the hypothalamic-pituitary-thyroid axis (HPT axis). (See Figure 1.)
Figure 1. The Various Feedback Mechanisms in the Hypothalamic-Pituitary-Thyroid Axis

The process of thyroid hormone synthesis starts with transport of iodine into the thyroid follicular cells by the iodine pump (sodium-iodine symporter). Iodine is a critical component of thyroid hormone. Iodine is oxidized by the enzyme thyroid peroxidase (TPO) and then binds to thyroglobulin (Tg) to form monoiodotyrosine (MIT) and diiodotyrosine (DIT). Coupling MIT with DIT will form T3 (triiodothyronine), and coupling two DIT will formT4 (thyroxine). About 10-20% of thyroid hormone released into the circulation is T3, which is the more active form of the hormone, whereas the remainder of hormone is T4, which is the inactive form. The majority of T4 circulates bound to binding proteins and, thus, is inactive. T4 must be peripherally converted into T3 by the enzyme 5-deiodinase to become active.
Thyrotoxicosis and Thyroid Storm
Hyperthyroidism is a state of excessive release of thyroid hormone from the thyroid gland, leading to a heightened metabolic state. Thyroid hormone also can emanate from other sources, such as release from stored thyroid follicles or exogenous hormone intake. Thyrotoxicosis is a hypermetabolic state caused by an excessive increase in circulating thyroid hormone as well as extra-thyroidal sources of thyroid hormone. Patients with hyperthyroidism or thyrotoxicosis can vary in presentation from asymptomatic or subclinical to thyroid storm. Thyroid storm or crisis is defined as the end stage of thyrotoxicosis and is a rare medical emergency that can progress quickly to multi-organ failure and death.
Epidemiology
Prevalence of thyrotoxicosis is about 1.2% in the United States.1 Thyroid storm is rare, with an incidence of 1-3% in patients with thyrotoxicosis.1-3 It is five to 10 times more common in women than in men and more common in patients 20-50 years of age.4,5 Mortality ranges between 10% to 50%, even with treatment.1,5,6 The most common causes of death are heart failure, respiratory failure, disseminated intravascular coagulopathy (DIC), sepsis, and brain injury.4,7
Etiologies
The most common cause of thyrotoxicosis is Graves’ disease, an autoimmune disease in which antibodies attack the thyroid gland, causing it to release excessive thyroid hormone. Graves’ disease accounts for about 60-80% of all causes of thyrotoxicosis.1 Toxic multinodular goiter and thyroiditis are the next most common causes. Other causes of thyrotoxicosis include TSH-secreting tumors in the pituitary or TRH-secreting tumors in the hypothalamus; however, these are relatively rare.8
Pregnancy, which is accompanied by physiologic changes such as increased resting heart rate, diaphoresis, and heat intolerance, is also a known precipitant of thyrotoxicosis and can present with similar symptoms. Even non-viable pregnancies such as ectopic pregnancy and molar pregnancy can incite thyrotoxicosis.9
The exact mechanism leading to development of thyroid storm from thyrotoxicosis still is not well understood. Commonly, a stressor (for instance, sepsis) causes a disruption of the body’s regulatory mechanisms. Other endocrinopathies also can trigger thyroid storm, further clouding the diagnosis.10 Common precipitants of thyroid storm can be found in Table 1.
Table 1. Common Precipitants of Thyroid Storm
- Medications (i.e., amiodarone, lithium, or cessation of antithyroid medications)
- Infections
- Thyroid conditions (i.e., Graves’ disease, thyroiditis, toxic multinodular goiter,
metastatic thyroid cancer, thyroid surgery, treatment with radioactive iodine) - Previous thyroid surgery or manipulation
- Trauma
- Ischemia (i.e., myocardial infarction, stroke)
- Other endocrinopathies (i.e., diabetic ketoacidosis)
- Pregnancy, molar pregnancy
Clinical Manifestations
Patients presenting with thyrotoxicosis commonly manifest many signs and symptoms due to thyroid hormone’s effects on multiple organ systems. It is not uncommon for these patients to have multiple complaints at once, with thyrotoxicosis providing a unifying diagnosis. Common clinical manifestations for each organ system are enumerated in Table 2. Patient presentations often will vary.
Table 2. Clinical Manifestations of Thyrotoxicosis
Organ System |
Symptoms/Signs |
|
Head, eyes, ears, nose, and throat |
Proptosis |
|
Cardiac |
Hypertension with widened pulse pressure Tachycardia Dysrhythmias (atrial fibrillation is the most common) Chest pain Heart failure (usually high output) |
|
Respiratory |
Dyspnea on exertion Orthopnea Pulmonary edema |
|
Gastrointestinal |
Anorexia and weight loss Nausea and vomiting Diarrhea Hepatomegaly Jaundice (poor prognostic indicator) |
|
Neurologic |
Fever and heat intolerance Diaphoresis Anxiety Agitation Delirium Seizure Confusion Stupor Coma |
|
Musculoskeletal |
Muscle weakness |
Clinical manifestations will depend on which organ system is the most affected by the presence of the excess hormone. Other factors that can contribute to thyroid hormone’s effects on different organs include age, comorbidities, and length of the illness.1 Some patients may present with primarily central nervous system (CNS) complaints, such as agitation, whereas others may present with only gastrointestinal (GI) complaints, such as abdominal pain and vomiting. Patients with thyroid storm will present with signs and symptoms of thyrotoxicosis to an exaggerated degree. Because of this array of different presentations, the diagnosis of thyrotoxicosis and thyroid storm often is elusive and requires the emergency provider to be vigilant in considering these diagnoses.
Fever is present in almost all patients.5 Sometimes the fever can be profoundly elevated. Altered mental status is almost universally present and can vary from agitation and irritability to convulsions, abnormal movements, seizures, and even delirium and coma.1,11,12 The severity of mental status change is associated with worse outcomes.6 The presence of altered sensorium with the fever often will prompt the emergency provider to contemplate alternative diagnoses that are more common, such as CNS infections, sepsis, or toxicologic disorders. While these disease processes are associated with higher morbidity and mortality, emergency providers should consider the diagnosis of thyroid storm as well.
Elderly patients are at particular risk for developing apathetic thyroid storm. In these patients, instead of the typical hyperagitated state often associated with thyroid storm, they will present with apathy and depression and often complain of nonspecific symptoms such as weakness or fatigue.1,13 Emergency providers should consider this diagnosis in elderly patients presenting with these nonspecific symptoms and fever.
Hemodynamic alterations also will be present, including hypertension with a widened pulse pressure and tachycardia, which can include dysrhythmias, most commonly sinus tachycardia and atrial fibrillation. Atrial fibrillation is considered one of the diagnostic criteria in both scoring systems for thyroid storm and is the most common dysrhythmia associated with thyroid storm. The tachycardia usually is out of proportion to the fever. Patients also may complain of palpitations and exercise intolerance. One case report prompts the emergency provider to consider thyroid storm in trauma patients when tachycardia is unexplained.14 They also may present with chest pain, which may be related to catecholamine-induced vasospasm or cardiomyopathy.15
Patients may present with symptoms of heart failure, such as dyspnea on exertion and orthopnea. Heart failure associated with thyrotoxicosis usually is a high-output heart failure where the cardiac output is elevated instead of depressed, as in more common presentations of heart failure. This is due to decreased systemic vascular resistance leading to increased cardiac output causing circulatory overload and subsequent backup of fluid into the lungs, causing pulmonary edema.16 Patients also may develop lower extremity pitting edema and jugular venous distention. If untreated, the left ventricle eventually will succumb to the increased work, and patients will develop depression of the ejection fraction. A small subset of patients with thyrotoxicosis initially will present with low-output heart failure.16 Both types of heart failure frequently improve once the patient becomes euthyroid.16
GI manifestations include nausea, vomiting, and diarrhea, which can be severe enough to cause volume depletion and electrolyte imbalance. Hepatic congestion also may occur, leading to abdominal pain, transaminitis, and hyperbilirubinemia, creating a clinical picture similar to cholangitis, with fever, right upper quadrant pain, and jaundice. The condition can progress to hepatic failure and disturbances in coagulation.17
Patients with thyroid storm can present variably, and the list of possible diagnoses can become quite extensive. Because of the various presentations possible for thyroid storm, it often is difficult to make the diagnosis. Emergency providers should investigate more common and, certainly, all deadly diagnoses. However, emergency providers should have a high suspicion for thyroid storm in patients presenting with altered mental status and high temperature or multiple complaints that have no seemingly organic cause.
Diagnosis
Thyroid function testing is the mainstay in diagnosing thyrotoxicosis and thyroid storm. TSH commonly is used as a screening test to check for thyroid diseases because of its quick turnaround time and its high sensitivity and specificity. In the presence of thyrotoxicosis, the TSH is almost always low because of the excess thyroid hormone negatively feeding back on the pituitary gland. Rarely, the TSH also can be normal or high in the presence of a TSH- or TRH-secreting tumor. Additionally, the TSH can be falsely lowered by medications such as levothyroxine or amiodarone.
Should the TSH be low, a free T4 level should be obtained. An elevated level of free T4 confirms the diagnosis but has a longer turnaround time in the emergency department. Free T4 is the most specific diagnostic test for thyrotoxicosis and thyroid storm. However, there is no definitive laboratory value cutoff that separates thyrotoxicosis from thyroid storm.1,5 Although it is rare, there is a small subset (~5%) of thyroid storm patients who have isolated T3 thyrotoxicosis.1 In these patients, the T3 will be elevated in the setting of a low TSH and a normal free T4. If the clinical suspicion is high for thyroid storm with a low TSH and a normal free T4, checking a total T3 is worthwhile as well.1
In terms of determining which patients warrant thyroid function testing, the provider must use clinical judgment. Scoring systems are available to help guide the provider in making the diagnosis of thyroid storm. In 1993, Burch and Wartofsky proposed a scoring system based on assigning a score for common thyroid storm signs and symptoms according to severity.18 The Burch-Wartofsky Score (BWS) includes dysfunctions in thermoregulatory, cardiovascular, gastrointestinal-hepatic, and central nervous system and presence or absence of a precipitating factor.19,20 This scoring system can be found in Table 3.
Table 3. Burch-Wartofsky Score
|
Total score < 25 is not likely thyroid storm, 25-45 is suggestive of thyroid storm, and > 45 is highly suggestive of thyroid storm. |
|
|
Adapted from: Burch HB, Wartofsky L. Life-threatening thyrotoxicosis. Thyroid storm. Endocrinol Metab Clin North Am 1993;22:263-277. |
|
Signs/Symptoms |
Score |
|
Temperature from 37.2° to > 40° C |
5-30 points |
|
Central nervous system dysfunction from agitation to seizures or coma |
10-30 points |
|
Gastrointestinal dysfunction from nausea/vomiting to jaundice |
10-20 points |
|
Heart rate from 99 to > 140 bpm |
5-25 points |
|
Presence of atrial fibrillation |
10 points |
|
Congestive heart failure from peripheral edema to pulmonary edema |
5-15 points |
|
Presence of an inciting event |
10 points |
Citing the lack of external validation of the BWS and its high false-positive capability, in 2012, the Japanese Thyroid Association (JTA) established a new set of diagnostic criteria that requires the presence of thyrotoxicosis confirmed by thyroid laboratory studies plus a combination of symptoms suggestive of multi-organ failure.2 However, these criteria are based on retrospective data and have not been validated externally. The JTA guidelines are found in Table 4.
Table 4. Japanese Thyroid Association Score
|
*Must have elevated free T4 and/or free T3 |
|
|
1 – Neurologic dysfunction |
|
|
2 – Temperature > 38° C |
|
|
3 – Heart rate > 130 bpm or atrial fibrillation |
|
|
4 – Class IV heart failure |
|
|
5 – Nausea, vomiting, diarrhea, or a bilirubin > 3.0 |
|
|
Definite |
Criteria 1 plus 1 of any other criterion or 3 of criteria 2-5 |
|
Suspected |
2 of criteria 2-5 or they meet “definite” criteria but without the thyrotoxic labs |
|
Adapted from: Akamizu T, Satoh T, Isozaki O, et al. Diagnostic criteria, clinical features, and incidence of thyroid storm based on nationwide surveys. Thyroid 2012;22:661-679. |
|
BWS and the JTA guidelines are equally acceptable. Nonetheless, there was a report from the United States that proposed that a BWS score of 45 or higher is more sensitive than the JTA in detecting patients who need aggressive therapy.7,21 Therefore, it is recommended to use both criteria to increase the accuracy of the diagnosis of thyroid storm.7
Management
Management of thyroid storm should be initiated immediately once clinical suspicion is high. Waiting for confirmatory laboratory studies delays management and contributes to an increase in mortality. Five goals must be achieved when managing thyroid storm: blocking the systemic symptoms, inhibiting the synthesis and release of thyroid hormone, inhibiting the peripheral conversion of T4 to T3, providing supportive care, and treating the underlying cause or trigger.
Blocking Systemic Symptoms. Thyroid hormone causes an increase in the production and expression of beta receptors onto cells, making them very sensitive to any type of adrenergic response. Consequently, patients with thyrotoxicosis often will have systemic manifestations, including hypertension and tachycardia. These systemic signs and symptoms contribute to the patient’s symptoms as well as complications associated with thyrotoxicosis.
Beta-blockers are the initial pharmacologic agent of choice for systemic blockade. Propranolol, a non-selective beta-blocker, is the preferred agent because of its increased ability to decrease peripheral conversion of T4 to T3. Esmolol, a short-acting IV beta-blocker, or metoprolol also can be used. (See Table 5.)
Table 5. Beta-Blockers and Calcium Channel Blockers in the Treatment of Thyroid Storm
Medication |
Dose |
|
Beta-blockers |
|
|
Propranolol |
60 to 120 mg every 6 hours PO or 0.5 to 1 mg slow IV push, then 1 to 2 mg IV q 15 mins |
|
Esmolol |
0.25 to 0.5 mg/kg IV bolus followed by 0.05 to 0.1 mg/kg/min infusion |
|
Metoprolol |
5 mg IV slow push q 5 to 15 mins |
|
Calcium Channel Blockers |
|
|
Diltiazem |
0.25 to 0.35 mg/kg slow IV push |
Beta-blocker dose is adjusted according to heart rate and blood pressure; therefore, patients should be placed on continuous cardiac monitoring. A heart rate of >150 beats per minute (bpm) is associated with an increase in mortality, according to a nationwide survey conducted in Japan from 2004 to 2008.4,7 Beta-blockers should be discontinued when the heart rate is < 80 bpm or the systolic blood pressure is < 80 mmHg.7
In patients with contraindications to beta-blocker therapy, such as severe asthmatics or chronic obstructive pulmonary disease (COPD) patients, the nondihydroperidine calcium channel blockers are suitable alternatives to beta-blocker therapy without the added benefit of blockage of peripheral T4 to T3.
Inhibiting Thyroid Synthesis and Release. Antithyroid medications work by directly inhibiting thyroid peroxidase, which will decrease the synthesis of new thyroid hormone. There are two classes of medications: thiouracils, such as propylthiouracil, and imidazoles, such as methimazole and carbimazole.
Propylthiouracil (PTU) is recommended by the American Thyroid Association (ATA) because of its additional benefit of inhibiting peripheral conversion of T4 to T3, therefore reducing T3 more than methimazole. PTU is administered orally, via nasogastric tube, or rectally. No IV formulation is available. PTU has a number of possible side effects, including significant ones such as agranulocytosis. (See Table 6.)
Table 6. Propylthiouracil and Methimazole in the Treatment of Thyroid Storm
Medication |
Dose |
|
Propylthiouracil |
Loading dose of 500 to 1,000 mg, then 250 mg every 4 hours (oral) or 400 to 600 mg every 4 hours (rectal) |
|
Methimazole |
60 to 120 mg per day in divided doses every 4 to 6 hours (oral) or 20 to 40 mg every 6 to 8 hours (rectal) or 10 to 30 mg every 6 to 8 hours (IV) |
Methimazole (MMI) is widely used as a first-line agent because of its lower side effect profile. In the United States, an IV formulation of methimazole is not commercially available; however, it can be prepared by a clinical pharmacist mixing methimazole powder with normal saline.
A retrospective analysis on clinical parameters, outcome, and treatment showed no difference in mortality between patients treated with PTU alone (11.1%) vs. MMI alone (10.1%).4 Also, there were no differences in APACHE II (Acute Physiology and Chronic Health Evaluation II) or SOFA (Sequential Organ Failure Assessment) scores.4
Inorganic Iodine. Nonradioactive iodine also decreases thyroid hormone synthesis by inhibiting iodide oxidation and organification that inhibits the release of thyroid hormone from the follicular lumen of the thyroid gland.
ATA guidelines recommend administration of iodine at least one hour after anti-thyroid medications.22 Otherwise, it will provide a substrate for the production of more thyroid hormone and potentially worsen the condition. The recommended dose is large since GI absorption is impaired in patients with thyroid storm. The most commonly used preparation is SSKI® (a saturated solution of potassium iodide 1 g/mL), 5 drops PO administered every six hours. Studies evaluating MMI alone vs. MMI and potassium iodide solution showed that combined treatment normalizes thyroid hormone levels faster than MMI alone.7 Also, patients had lower mortality when receiving potassium iodide.4
Lithium is a suitable alternative to iodide therapy. Lithium inhibits the coupling of iodotyrosine, thus blocking thyroid hormone synthesis. It is administered at a dose of 300 mg orally every six to eight hours. Lithium has a narrow therapeutic range; therefore, it is important to monitor serum levels daily to prevent lithium toxicity.5 (See Table 7.)
Table 7. Potassium Iodide and Lithium in the Treatment of Thyroid Storm
Medication |
Dose |
|
SSKI (potassium iodide solution 1 g/mL) |
5 drops PO every 6 hours |
|
Lithium |
300 mg PO every 6 to 8 hours |
Inhibition of Peripheral Conversion of Thyroid Hormone. In addition to the medications already given to decrease peripheral conversion of T4 to T3, corticosteroids should be administered. Corticosteroids serve two purposes in the treatment of thyroid storm: inhibition of the peripheral conversion of T4 to T3 and treatment of the relative adrenal insufficiency associated with thyrotoxic states. Additionally, corticosteroids help with the management of autoimmune causes of thyroid storm, such as Graves’ disease. There is no evidence that one class of steroids is superior to others.5,7 (See Table 8.)
Table 8. Corticosteroids in the Treatment of Thyroid Storm
Medication |
Dose |
|
Hydrocortisone |
100 mg IV q 8 hours |
|
Dexamethasone |
8 mg IV or PO every day |
Supportive Care
Treatment of Hyperpyrexia. Acetaminophen is the initial choice of antipyretic. Emergency providers should avoid salicylates and nonsteroidal anti-inflammatory drugs, as they increase thyroid hormone level by decreasing protein binding to T4, allowing it to circulate freely and potentially be converted to T3. Despite this, there was no significant difference in outcomes between those who received acetaminophen vs. other antipyretic agents.4
When temperatures are excessive or refractory to antipyretics alone, cooling is recommended to help prevent muscle breakdown. However, active cooling, such as ice packs, submersion, or invasive cooling techniques (i.e., cooled IV fluids, cooled bladder irrigation), is contraindicated because of the resultant vasoconstriction and subsequent worsening of hypertensive crisis. Passive cooling, such as undressing the patient, lowering the temperature in the room, and using fans, is recommended to help lower the patient’s temperature.
Convulsions/Seizures. The first step is to ensure airway, breathing, and circulation are intact and to manage hypoglycemia if present. If the patient’s finger stick for glucose is normal, administer benzodiazepines as the initial medication; diazepam or lorazepam are the preferred agents. If the convulsions continue after adequate doses of benzodiazepines, an antiepileptic, such as phenytoin, is recommended. However, phenytoin has been shown to falsely lower the levels of free T4 because the medication affects the laboratory assay. Therefore, bloodwork should be obtained prior to administration of phenytoin. Alternatively, other antiepileptics, such as valproic acid or levetiracetam, can be used. Typically, seizures will abate with anti-thyroid management once euthyroidism is achieved.11
Agitation, Delirium, Psychosis. First-line medications to control agitation are benzodiazepines. The second-generation antipsychotics, such as risperidone and olanzapine, also have been used safely but are available only in oral formulations. For patients who cannot tolerate oral medication, first-generation parenteral antipsychotics, such as haloperidol, can be used cautiously, although there are case reports suggesting that haloperidol can precipitate thyroid storm. Chlorpromazine has the added benefit of decreasing body temperature.23
Heart Failure Treatment. Because heart failure associated with thyroid storm often is high output, typical heart failure treatment may be harmful. Since systemic vascular resistance is already low, preload and afterload reduction commonly used in heart failure has the potential to worsen the patient’s condition. Therefore, avoid preload and afterload reducing medications, such as nitrates and angiotensin-converting enzyme inhibitors, in these patients. Rate control with beta-blockade (or calcium channel blockers) and anti-thyroid medications often will slow down the heart rate and decrease the stroke volume, subsequently reversing the heart failure.16 Although not usually necessary, gentle diuresis with loop diuretics, such as furosemide, is reasonable to help offload the pulmonary edema.
Anticoagulation. Anticoagulation for atrial fibrillation often is based on the CHADS2 score system per the 2014 American Heart Association, American College of Cardiology, and Heart Rhythm Society guidelines. The role of anticoagulation in atrial fibrillation in thyrotoxic patients and patients in thyroid storm has been more controversial.24-26 There have been several case reports that showed patients with hyperthyroidism are at an increased risk of thromboembolism, some reporting up to a 1.4-fold greater risk than in subjects without hyperthyroidism.25 This is true even in young patients who otherwise have no risk factors for thromboembolic disease other than the thyrotoxic atrial fibrillation. This is probably secondary to the hypercoagulable state hyperthyroidism causes through a decrease in activated partial thromboplastin time (PTT), an increase in fibrinogen, and an increase in factor VIII. Consequently, there are no established guidelines to guide the decision to administer anticoagulation. It is recommended that anticoagulation therapy should be initiated in patients who present with thyrotoxic atrial fibrillation if there are no contraindications.24,26
Treatment of Underlying Cause/Clinical Manifestations. Because a stressor is the usual etiology of thyroid storm, the emergency provider must not forget to manage this stressor while managing the thyroid storm. A thorough history and physical examination should investigate any possible inciting events. There should be a low threshold to broaden the diagnostic workup to include laboratory and imaging studies.
If fever is a manifestation of an infection, consider broad-spectrum antibiotics. The most common infections are urinary tract infections and pneumonia. However, until the causative organism is identified, the 2016 JTA guidelines recommend both Gram-positive and Gram-negative coverage.7 Ischemia, which can be myocardial, neurologic, or mesenteric, can result from thyrotoxic atrial fibrillation but also can induce thyroid storm.
Other Therapies. Therapeutic plasma exchange (TPE) can be used to reduce free thyroid hormone level in circulation.4,7,27 It is important to note that TPE is an alternative therapeutic option for cases that are refractory to all other medical treatment and/or cases with rapid deterioration or thyroid storm with neurologic or cardiac manifestation or severe myopathy.27-31
Surgical Management. Patients may require an emergent thyroidectomy for management of their thyroid storm. These include patients who have not responded to intensive medical treatment after 12 to 24 hours or patients who have developed serious side effects from medical treatment (i.e., agranulocytosis).5,32
Special Considerations
Sometimes pregnancy is a precipitating factor for thyroid crisis. Management of thyroid crisis in pregnant women should include fetal monitoring. Avoid inducing labor or delivery, as it may worsen the maternal outcome. The rest of the management is similar to that of the non-pregnant patient. PTU should be used first in pregnant patients in thyroid storm, as methimazole has been implicated as the cause of scalp defects in newborn children.
Disposition and Prognosis
Admit patients with thyroid storm to the intensive care unit. They are at higher risk to develop hemodynamic collapse, even with adequate treatment. Predictors of poor outcome include degree of CNS dysfunction, advanced age, use of plasmapheresis, mechanical ventilation, and elevated APACHE II and SOFA scores.21,33,34
Conclusion
Thyroid storm is a life-threatening emergency that carries 10-30% mortality. The diagnosis often is elusive, and emergency providers need to be vigilant about keeping thyroid storm on their list of potential diagnoses. Management is in a stepwise fashion and should be initiated as soon as the clinical suspicion is high.
Myxedema Coma
Myxedema coma or myxedema is a rare condition defined as the severe form of hypothyroidism leading to multi-organ dysfunction, including altered sensorium.
Epidemiology
The incidence of myxedema coma is reported at 0.22 per million each year, with a mortality of 30-60%, regardless of treatment.35,36 Delays in the recognition and treatment are associated with worse outcomes. The incidence is certainly underreported, as there are many possible inciting events that mimic myxedema coma and are more common and also fatal (i.e., sepsis). Myxedema coma occurs much more commonly in women than in men, and the vast majority of cases occur in patients 60 years of age or older.37
There also appears to be a seasonal component, as almost 90% of cases occur during the winter months, and there is less of an incidence in areas near the more tropical regions in the world.36,37 About 95% of cases are associated with severe or long-standing primary hypothyroidism that usually is undiagnosed or undertreated. The remaining 5% is caused by deficiency in hormone from the pituitary or the hypothalamus.38 Myxedema coma usually is precipitated by infection, sepsis, trauma, or some medications. These and other common precipitating factors can be found in Table 9.
Table 9. Common Precipitants of Myxedema Coma
- Infection (urinary tract infection, pneumonia)
- Medications (sedative agents, valproic acid toxicity, codeine, benzodiazepines, opiates, marijuana)
- Dehydration in chronic kidney disease/end stage renal disease patient
- Cold exposure, hypothermia (hence, condition is more common in winter)
- Sepsis
- Metabolic acidosis and electrolyte abnormalities
- Ingestion of raw bok choy
- Congestive heart failure, cerebrovascular accident, gastrointestinal bleeding
Clinical Features
Myxedema crisis presents as severe hypothyroidism with signs, symptoms, and laboratory tests suggestive of end-organ dysfunction. The emergency provider should consider myxedema coma in patients presenting with altered mental status and hypothermia with abnormal thyroid function testing.
Patient presentations may vary. Because of the thyroid’s many actions on the organs, various organs may be affected differently by the lack of thyroid hormone. Common signs and symptoms can be found in Table 10.
Table 10. Clinical Manifestations of Myxedema Coma
Organ System |
Symptoms/Signs |
|
Head, eyes, ears, nose, and throat |
Airway edema Macroglossia Facial edema |
|
Cardiac |
Hypotension with narrow pulse pressure Bradycardia QT prolongation or AV blocks Pericardial effusion |
|
Respiratory |
Pleural effusion Hypoventilation Respiratory depression |
|
Gastrointestinal |
Decreased bowel sounds Constipation Weight gain |
|
Neurologic |
Hypothermia Cold intolerance Depression Confusion Stupor Coma |
|
Dermatologic |
Nonpitting edema |
|
Musculoskeletal |
Muscle weakness Delayed relaxation phase of deep tendon reflexes (called the Woltman sign or myxedema reflex) |
|
Hematologic |
Anemia Coagulopathy secondary to acquired von Willebrand syndrome Disseminated intravascular coagulopathy |
Hypothermia is almost always present, found in 75-100% of patients with myxedema coma.36,37 The hypothermia is profound in most circumstances. As stated before, it is most common in the winter months and is thought to be due to a dysfunction in the thermoregulatory system.
Additionally, hemodynamics are affected, as shown by bradycardia and hypotension with a narrow pulse pressure. Hypotension is due to decreased contractility of the heart but also can be due to fluid accumulating in the pericardium to cause cardiac tamponade.39,40 Dysrhythmias also are possible. Bradycardia is common, but QT prolongation can occur as well, leading to torsades de pointes.37,41
Hypoventilation may not manifest as bradypnea or apnea until later in the disease process, but patients may develop signs and symptoms of hypercapnia. Hypoventilation also can be the result of fluid accumulating in the pleural space causing decreased lung volumes.
Alterations in sensorium is another common clinical manifestation. Patients will present with fatigue, confusion, depression, and stupor, eventually leading to a comatose state. The diagnostic problem is that there are multiple possible etiologies for these patients to have altered mental states, such as hypothermia, electrolyte abnormalities (i.e., hyponatremia, hypoglycemia), hypercapnia, hypotension, and the inciting event such as sepsis.
Because of these various presentations, especially with altered mental status, the diagnosis in the emergency department can be difficult. Again, consider myxedema coma in patients with altered mental status and hypothermia.
Diagnostic Tests
Thyroid function testing is necessary to make the diagnosis, although management should commence before confirmatory testing if the clinical suspicion is high. TSH will be elevated if the cause is primary hypothyroidism. The TSH will be normal or low if hypothyroidism is caused by secondary or tertiary hypothyroidism, both of which are rare. Confirmatory testing with T4 and T3 makes the diagnosis, with either or both being low.
Other laboratory studies include a complete blood count, which may demonstrate leukocytosis and anemia. Chemistry commonly will show hyponatremia, hyperkalemia, and hypoglycemia secondary to adrenal insufficiency. They may have an elevated creatinine, transaminitis, and an elevated creatine kinase (CK) from hypothyroid myopathy. The coagulation profile may demonstrate an elevated PTT and bleeding time. The blood gas also may show respiratory acidosis (low pH and elevated PaCO2) secondary to hypoventilation.
Since arrhythmias are common, an ECG should be obtained. The most common finding is sinus bradycardia. QT prolongation also can occur, which can lead to torsades de pointes.41 An echocardiogram may show signs of heart failure or tamponade, as fluid backup is common.39,40 The emergency provider also should look for an inciting event, such as sepsis, and should have a low threshold to obtain diagnostic labs and imaging, searching for a possible cause.
The lack of validated or accepted clinical diagnostic criteria and the rarity of the condition make diagnosing this condition in the emergency department difficult. Two scoring systems exist, neither of which has been validated or is used commonly in practice.42,43 The first uses a combination of cardiac manifestations, CNS dysfunctions, GI disturbance, and laboratory analyses.42 The second uses six variables and has a sensitivity and specificity of 80%.43 The criteria include Glasgow Coma Scale, TSH level, free T4 level, hypothermia, bradycardia, and the presence of a precipitating factor. Until studies validate either of these scoring systems, their use is not recommended.
Management
Resuscitative Measures. Emergency providers should ensure an adequate airway is present. Hypothyroidism may depress respiratory drive and predispose to respiratory acidosis and hypoxic respiratory failure requiring intubation and mechanical ventilation.
Intubation of myxedema coma patients can be difficult. Recall that patients with myxedema coma are prone to hypoventilation. They also may have decreased lung volumes due to compressive forces from pleural effusions or ascites in the abdomen compressing the diaphragm from below. Because of this decrease in ventilation, they have less oxygen reserve, predisposing them to dropping their oxygen saturation faster when using paralytic agents for rapid sequence intubation. Additionally, the mechanical process of intubating may be difficult as a result of upper airway edema and macroglossia.37,44,45 Emergency providers should prepare by having adjunct airway equipment, including a surgical cricothyrotomy kit, when intubating.
Patients in shock will require fluid resuscitation and vasopressors, although vasopressors are less likely to be effective without thyroid hormone replacement therapy first. If hypotension is refractory to vasopressor therapy, before adding a second vasopressor agent or an inotrope, consider the administration of thyroid hormone replacement.
Thyroid Hormone Replacement. Once the diagnosis of myxedema coma is suspected, treatment should be initiated. Waiting for confirmatory testing should not delay care. Thyroid hormone replacement is the mainstay of therapy for myxedema coma. There are two formulations. Thyroxine or T4 is the most commonly used and thyronine or T3 is the alternative.
Thyroxine can be administered intravenously, orally, or via nagogastric tube. Some suggest initiating treatment intravenously for possible gastrointestinal impairment. However, it also has to be converted to T3 in the body, so it has the slowest onset. Thyronine is the more active thyroid hormone and should be avoided in patients who are older or who have previous coronary artery disease, as it will predispose them to developing arrhythmias or myocardial infarction.35
Controversy exists regarding which formulation is the better one to give. Thyroxine usually is given first because of its lower side effect profile. However, in cases of significant hemodynamic compromise, such as shock refractory to vasopressors, thyronine is the better option. Emergency providers should consider adding T3 if there is no improvement with T4 alone. Combination therapy of T4 and T3 has been found to be more successful with dosing being on the lower end for each agent. In a national inpatient database in Japan, there were no differences in mortality between treatment with T4 alone and T4 combined with T3.36 (See Table 11.)
Table 11. Thyroid Hormone Replacement in the Treatment of Myxedema Coma
Medication |
Dose |
|
Thyroxine or T4 |
200 to 500 mcg IV |
|
Thyronine or T3 |
5 to 20 mcg IV then 2.5 to 10 mcg IV q 8 hours |
Adjunctive Therapy. Severe hypothyroidism can impair the synthesis of steroid hormones due to adrenal gland impairment, causing adrenal insufficiency. Therefore, corticosteroids should be administered. Stress dose steroids are administered until adrenal insufficiency can be effectively ruled out. (See Table 12.)
Table 12. Corticosteroids in the Treatment of Myxedema Coma
Medication |
Dose |
|
Hydrocortisone |
100 mg IV q 8 hours |
|
Dexamethasone |
8 mg IV or PO every day |
Treatment of Underlying Precipitating Factor and Supportive Care. When crisis is precipitated by an infection, or infection is suspected, obtain cultures and administer broad-spectrum antibiotics. Infection remains one of the most common inciting events. If a medication is the culprit, it should be discontinued. Treatment of the underlying precipitating factor plays a major role in treatment of the crisis.
Manage hypothermia with passive warming. Active measures, such as warming the patient with a Bair Hugger, warm IV fluids, or other invasive warming techniques, can cause vasodilation and further hypotension leading to shock. Temperature regulation can be achieved by increasing room temperature and placing room temperature blankets on the patient.
If hypoglycemia is present, remember to administer stress dose steroids, as dextrose by itself likely will not maintain the glucose in the setting of adrenal insufficiency. After administration of corticosteroids, dextrose infusions still may be necessary to maintain euglycemia.35
Prognosis and Disposition
All patients with confirmed or suspected myxedema coma should be admitted to the intensive care unit. Predictors of poor outcome include advanced age, persistent bradycardia, patient noncompliance with thyroid medications in pre-existing thyroid disease, presence of cardiovascular disease or complication, persistent hypothermia, and multi-organ impairment with a SOFA score > 6 or APACHE II score > 20.35,46
Conclusion
Myxedema coma has a very high mortality and is underrecognized because of its rarity and its various presentations. Emergency providers should keep myxedema coma on the differential diagnosis for patients presenting hypothermic with altered mental status. Management often involves casting a wide diagnostic net as well as a multi-faceted treatment algorithm.
Summary
Thyroid emergencies are rare but life-threatening conditions. Therefore, it is imperative for emergency providers to recognize them and consider them when faced with an altered patient with extremes of temperature. Mortality and outcomes improve with early diagnosis and treatment.
References
- Devereaux D, Tewelde SZ. Hyper-thyroidism and thyrotoxicosis. Emerg Med Clin North Am 2014;32:277-292.
- Akamizu T, Satoh T, Isozaki O, et al. Diagnostic criteria, clinical features, and incidence of thyroid storm based on nationwide surveys. Thyroid 2012;22:661-679.
- Hampton J. Thyroid gland disorder emergencies: Thyroid storm and myxedema coma. AACN Adv Crit Care 2013;24:325-332.
- Isozaki O, Satoh T, Wakino S, et al. Treatment and management of thyroid storm: Analysis of the nationwide surveys: The taskforce committee of the Japan Thyroid Association and Japan Endocrine Society for the establishment of diagnostic criteria and nationwide surveys for thyroid storm. Clin Endocrinol (Oxf) 2016;84:912-918.
- Chiha M, Samarasinghe S, Kabaker AS. Thyroid storm: An updated review. J Intensive Care Med 2015;30:131-140.
- Swee du S, Chng CL, Lim A. Clinical characteristics and outcome of thyroid storm: A case series and review of neuropsychiatric derangements in thyrotoxicosis. Endocr Pract 2015;21:182-189.
- Satoh T, Isozaki O, Suzuki A, et al. 2016 guidelines for the management of thyroid storm from The Japan Thyroid Association and Japan Endocrine Society (First edition). Endocr J 2016;63:1025-1064.
- Fujio S, Ashari, Habu M, et al. Thyroid storm induced by TSH-secreting pituitary adenoma: A case report. Endocr J 2014;61:1131-1136.
- Moskovitz JB, Bond MC. Molar pregnancy-induced thyroid storm. J Emerg Med 2010;38:e71-76.
- Memon R, Fan W, Snyder R, Krishnamurthy M. Thyroid storm presenting as psychosis: Masked by diabetic ketoacidosis. J Community Hosp Intern Med Perspect 2016;6:31750.
- Lee TG, Ha CK, Lim BH. Thyroid storm presenting as status epilepticus and stroke. Postgrad Med J 1997;73:61.
- Park E, Abraham MK. Altered mental status and endocrine diseases. Emerg Med Clin North Am 2014;32:367-378.
- Parker KI, Loftley A, Charles C, Hermayer K. A case of apathetic thyroid storm with resultant hyperthyroidism-induced hypercalcemia. Am J Med Sci 2013;346:338-340.
- Liang CM, Ho MH, Wu XY, et al. Thyroid storm following trauma: A pitfall in the emergency department. Injury 2015;46:169-171.
- Eliades M, El-Maouche D, Choudhary C, et al. Takotsubo cardiomyopathy associated with thyrotoxicosis: A case report and review of the literature. Thyroid 2014;24:383-389.
- Yamashita Y, Iguchi M, Nakatani R, et al. Thyroid storm with heart failure treated with a short-acting beta-adrenoreceptor blocker, landiolol hydrochloride. Intern Med 2015;54:1633-1637.
- Deng Y, Zheng W, Zhu J. Successful treatment of thyroid crisis accompanied by hypoglycemia, lactic acidosis, and multiple organ failure. Am J Emerg Med 2012;30:2094.e5-6.
- Burch HB, Wartofsky L. Life-threatening thyrotoxicosis. Thyroid storm. Endocrinol Metab Clin North Am 1993;22:263-277.
- Wartofsky L. Clinical criteria for the diagnosis of thyroid storm. Thyroid 2012;22:659-660.
- Klubo-Gwiezdzinska J, Wartofsky L. Thyroid emergencies. Med Clin North Am 2012;96:385-403.
- Angell TE, Lechner MG, Nguyen CT, et al. Clinical features and hospital outcomes in thyroid storm: A retrospective cohort study. J Clin Endocrinol Metab 2015;100:451-459.
- Bahn Chair RS, Burch HB, Cooper DS, et al. Hyperthyroidism and other causes of thyrotoxicosis: Management guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Thyroid 2011;21:593-646.
- Sharp CS, Wilson MP, Nordstrom K. Psychiatric emergencies for clinicians: The emergency department management of thyroid storm. J Emerg Med 2016;51:155-158.
- Traube E, Coplan NL. Embolic risk in atrial fibrillation that arises from hyperthyroidism: Review of the medical literature. Texas Heart Inst J 2011;38:225-228.
- Gonzalez-Bossolo A, Gonzalez-Rivera A, Coste-Sibilia S. Cerebrovascular accident due to thyroid storm: Should we anticoagulate? Case Rep Endocrinol 2016;2016:5218985.
- Petersen AW, Puig-Carrion GD, Lopez-Candales A. Should we revisit anticoagulation guidelines during thyroid storm? Bol Asoc Med P R 2015;107:62-66.
- Muller C, Perrin P, Faller B, et al. Role of plasma exchange in the thyroid storm. Ther Apher Dial 2011;15:522-531.
- McGonigle AM, Tobian AAR, Zink JL, King KE. Perfect storm: Therapeutic plasma exchange for a patient with thyroid storm. J Clin Apher 2017 Jun 13. [Epub ahead of print.]
- Koball S, Hickstein H, Gloger M, et al. Treatment of thyrotoxic crisis with plasmapheresis and single pass albumin dialysis: A case report. Artif Organs 2010;34:E55-58.
- Carhill A, Gutierrez A, Lakhia R, Nalini R. Surviving the storm: Two cases of thyroid storm successfully treated with plasmapheresis. BMJ Case Rep 2012 Oct 19;2012.
- Vyas AA, Vyas P, Fillipon NL, et al. Successful treatment of thyroid storm with plasmapheresis in a patient with methimazole-induced agranulocytosis. Endocr Pract 2010;16:673-676.
- Uchida N, Suda T, Ishiguro K. Thyroidectomy in a patient with thyroid storm: Report of a case. Surg Today 2015;45:110-114.
- Ono Y, Ono S, Yasunaga H, et al. Factors associated with mortality of thyroid storm: Analysis using a national inpatient database in Japan. Medicine (Baltimore) 2016;95:e2848.
- Spaulding SW. Certain clinical features and treatments may be associated with increased mortality in thyroid storm. Clinical Thyroidology 2015;27:306-309.
- Pizzolato E, Peano A, Barutta L, et al. A forgotten life-threatening medical emergency: Myxedema coma. Emerg Care J 2016;12:46-48.
- Ono Y, Ono S, Yasunaga H, et al. Clinical characteristics and outcomes of myxedema coma: Analysis of a national inpatient database in Japan. J Epidemiol 2017;27:117-122.
- Dubbs SB, Spangler R. Hypothyroidism: Causes, killers, and life-saving treatments. Emerg Med Clin North Am 2014;32:303-317.
- Persani L. Clinical review: Central hypothyroidism: Pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab 2012;97:3068-3078.
- Ekka M, Ali I, Aggarwal P, Jamshed N. Cardiac tamponade as initial presenting feature of primary hypothyroidism in the ED. Am J Emerg Med 2014;32:683.e1-3.
- Majid-Moosa A, Schussler JM, Mora A. Myxedema coma with cardiac tamponade and severe cardiomyopathy. Proc (Bayl Univ Med Cent) 2015;28:509-511.
- Schenck JB, Rizvi AA, Lin T. Severe primary hypothyroidism manifesting with torsades de pointes. Am J Med Sci 2006;331:154-156.
- Popoveniuc G, Chandra T, Sud A, et al. A diagnostic scoring system for myxedema coma. Endocr Pract 2014;20:808-817.
- Chiong YV, Bammerlin E, Mariash CN. Development of an objective tool for the diagnosis of myxedema coma. Transl Res 2015;166:233-243.
- Melville JC, Menegotto KD, Woernley TC, et al. Unusual case of a massive macroglossia secondary to myxedema: A case report and literature review. J Oral Maxillofac Surg 2017. [Epub ahead of print.]
- Prado SRSND, Steinman RA, Munir KM, Lamos EM. Supraglottic myxedema: Two cases and a review of the literature. AACE Clinical Case Reports 2017;3:e101-e105.
- Mathew V, Misgar RA, Ghosh S, et al. Myxedema coma: A new look into an old crisis. J Thyroid Res 2011;2011:493462.
Thyroid disease is relatively common, but most often is a benign disease with little clinical significance in the emergency setting. However, even a small insult can disrupt this system and throw the regulation out of control, resulting in the secretion of too much or too little thyroid hormone. Both of these situations can result in the thyroid emergencies thyroid storm and myxedema coma.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
